Abstract
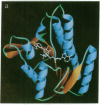
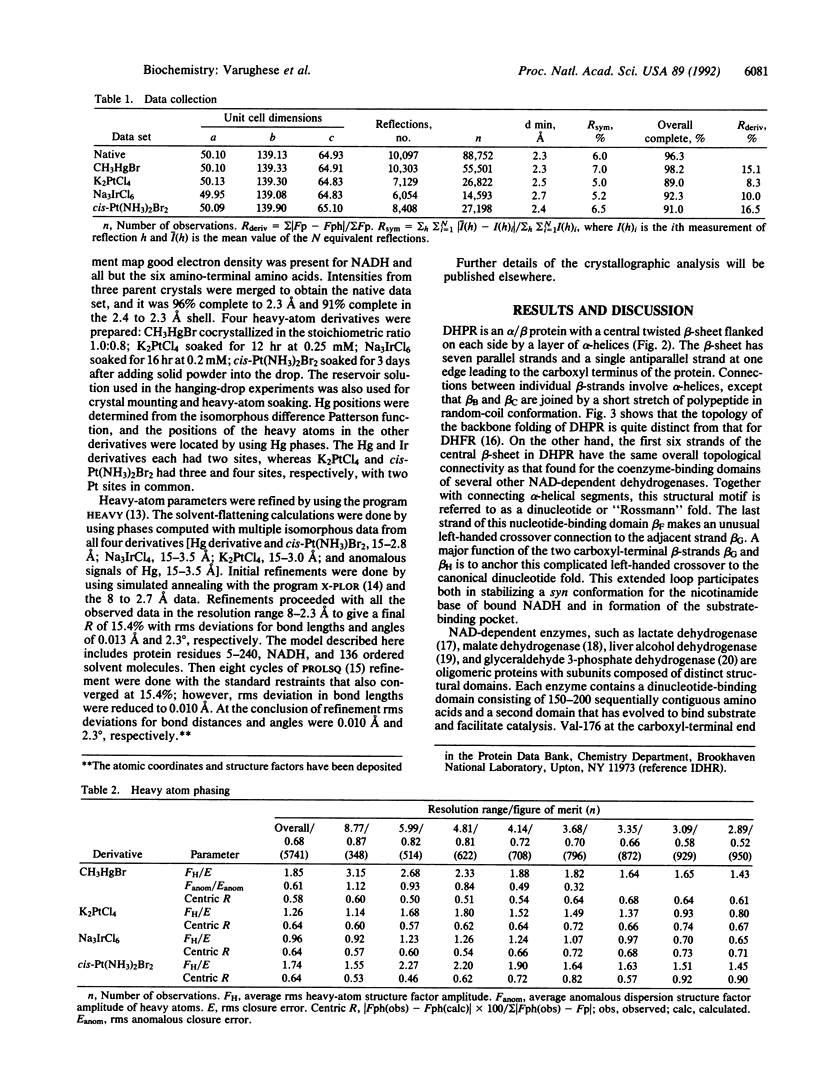
The structure of a binary complex of dihydropteridine reductase [DHPR; NAD(P)H:6,7-dihydropteridine oxidoreductase, EC 1.6.99.7] with its cofactor, NADH, has been solved and refined to a final R factor of 15.4% by using 2.3 A diffraction data. DHPR is an alpha/beta protein with a Rossmann-type dinucleotide fold for NADH binding. Insertion of an extra threonine residue in the human enzyme is associated with severe symptoms of a variant form of phenylketonuria and maps to a tightly linked sequence of secondary-structural elements near the dimer interface. Dimerization is mediated by a four-helix bundle motif (two helices from each protomer) having an unusual right-handed twist. DHPR is structurally and mechanistically distinct from dihydrofolate reductase, appearing to more closely resemble certain nicotinamide dinucleotide-requiring flavin-dependent enzymes, such as glutathione reductase.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Armarego W. L. Hydrogen transfer from 4-R and 4-S (4-3H) NADH in the reduction of d,l-cis-6,7-dimethyl-6,7 (8H) dihydropterin with dihydropteridine reductase from human liver and sheep liver. Biochem Biophys Res Commun. 1979 Jul 12;89(1):246–249. doi: 10.1016/0006-291x(79)90970-7. [DOI] [PubMed] [Google Scholar]
- Armarego W. L., Ohnishi A., Taguchi H. New pteridine substrates for dihydropteridine reductase and horseradish peroxidase. Biochem J. 1986 Mar 1;234(2):335–342. doi: 10.1042/bj2340335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benner S. A. The stereoselectivity of alcohol dehydrogenases: a stereochemical imperative? Experientia. 1982 May 15;38(5):633–636. doi: 10.1007/BF02327092. [DOI] [PubMed] [Google Scholar]
- Brünger A. T., Kuriyan J., Karplus M. Crystallographic R factor refinement by molecular dynamics. Science. 1987 Jan 23;235(4787):458–460. doi: 10.1126/science.235.4787.458. [DOI] [PubMed] [Google Scholar]
- Chou K. C., Maggiora G. M., Némethy G., Scheraga H. A. Energetics of the structure of the four-alpha-helix bundle in proteins. Proc Natl Acad Sci U S A. 1988 Jun;85(12):4295–4299. doi: 10.1073/pnas.85.12.4295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craine J. E., Hall E. S., Kaufman S. The isolation and characterization of dihydropteridine reductase from sheep liver. J Biol Chem. 1972 Oct 10;247(19):6082–6091. [PubMed] [Google Scholar]
- Eklund H., Samma J. P., Wallén L., Brändén C. I., Akeson A., Jones T. A. Structure of a triclinic ternary complex of horse liver alcohol dehydrogenase at 2.9 A resolution. J Mol Biol. 1981 Mar 15;146(4):561–587. doi: 10.1016/0022-2836(81)90047-4. [DOI] [PubMed] [Google Scholar]
- Howells D. W., Forrest S. M., Dahl H. H., Cotton R. G. Insertion of an extra codon for threonine is a cause of dihydropteridine reductase deficiency. Am J Hum Genet. 1990 Aug;47(2):279–285. [PMC free article] [PubMed] [Google Scholar]
- Karplus P. A., Daniels M. J., Herriott J. R. Atomic structure of ferredoxin-NADP+ reductase: prototype for a structurally novel flavoenzyme family. Science. 1991 Jan 4;251(4989):60–66. [PubMed] [Google Scholar]
- Karplus P. A., Schulz G. E. Substrate binding and catalysis by glutathione reductase as derived from refined enzyme: substrate crystal structures at 2 A resolution. J Mol Biol. 1989 Nov 5;210(1):163–180. doi: 10.1016/0022-2836(89)90298-2. [DOI] [PubMed] [Google Scholar]
- Kaufman S., Holtzman N. A., Milstien S., Butler L. J., Krumholz A. Phenylketonuria due to a deficiency of dihydropteridine reductase. N Engl J Med. 1975 Oct 16;293(16):785–790. doi: 10.1056/NEJM197510162931601. [DOI] [PubMed] [Google Scholar]
- Levy H. R., Ejchart A., Levy G. C. Conformations of nicotinamide coenzymes bound to dehydrogenases determined by transferred nuclear Overhauser effects. Biochemistry. 1983 Jun 7;22(12):2792–2796. doi: 10.1021/bi00281a004. [DOI] [PubMed] [Google Scholar]
- Lockyer J., Cook R. G., Milstien S., Kaufman S., Woo S. L., Ledley F. D. Structure and expression of human dihydropteridine reductase. Proc Natl Acad Sci U S A. 1987 May;84(10):3329–3333. doi: 10.1073/pnas.84.10.3329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthews D. A., Webber S., Whiteley J. M. Preliminary x-ray diffraction characterization of crystalline rat liver dihydropteridine reductase. J Biol Chem. 1986 Mar 15;261(8):3891–3893. [PubMed] [Google Scholar]
- Murthy M. R., Garavito R. M., Johnson J. E., Rossmann M. G. Structure of lobster apo-D-glyceraldehyde-3-phosphate dehydrogenase at 3.0 A resolution. J Mol Biol. 1980 Apr 25;138(4):859–872. doi: 10.1016/0022-2836(80)90069-8. [DOI] [PubMed] [Google Scholar]
- Presnell S. R., Cohen F. E. Topological distribution of four-alpha-helix bundles. Proc Natl Acad Sci U S A. 1989 Sep;86(17):6592–6596. doi: 10.1073/pnas.86.17.6592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raag R., Appelt K., Xuong N. H., Banaszak L. Structure of the lamprey yolk lipid-protein complex lipovitellin-phosvitin at 2.8 A resolution. J Mol Biol. 1988 Apr 5;200(3):553–569. doi: 10.1016/0022-2836(88)90542-6. [DOI] [PubMed] [Google Scholar]
- Schiering N., Kabsch W., Moore M. J., Distefano M. D., Walsh C. T., Pai E. F. Structure of the detoxification catalyst mercuric ion reductase from Bacillus sp. strain RC607. Nature. 1991 Jul 11;352(6331):168–172. doi: 10.1038/352168a0. [DOI] [PubMed] [Google Scholar]
- Wang B. C. Resolution of phase ambiguity in macromolecular crystallography. Methods Enzymol. 1985;115:90–112. doi: 10.1016/0076-6879(85)15009-3. [DOI] [PubMed] [Google Scholar]
- Webb L. E., Hill E. J., Banaszak L. J. Conformation of nicotinamide adenine dinucleotide bound to cytoplasmic malate dehydrogenase. Biochemistry. 1973 Dec 4;12(25):5101–5109. doi: 10.1021/bi00749a013. [DOI] [PubMed] [Google Scholar]
- Webber S., Hural J., Whiteley J. M. Preliminary studies on the primary structure of rat liver dihydropteridine reductase. Biochem Biophys Res Commun. 1987 Mar 13;143(2):582–586. doi: 10.1016/0006-291x(87)91393-3. [DOI] [PubMed] [Google Scholar]
- White J. L., Hackert M. L., Buehner M., Adams M. J., Ford G. C., Lentz P. J., Jr, Smiley I. E., Steindel S. J., Rossmann M. G. A comparison of the structures of apo dogfish M4 lactate dehydrogenase and its ternary complexes. J Mol Biol. 1976 Apr 25;102(4):759–779. doi: 10.1016/0022-2836(76)90290-4. [DOI] [PubMed] [Google Scholar]
- You K. S. Stereospecificity for nicotinamide nucleotides in enzymatic and chemical hydride transfer reactions. CRC Crit Rev Biochem. 1985;17(4):313–451. doi: 10.3109/10409238509113625. [DOI] [PubMed] [Google Scholar]