Abstract
Leukocyte telomere lengths (LTLs) are shorter in obese compared with normal weight people. However, it is not known whether sperm telomere length (STL) is related to obesity. The aim of the study was to evaluate the impact of men’s body mass index (BMI) on STL, embryo quality, and clinical outcomes in couples undergoing IVF. In total, 651 couples were recruited, including 345 men with a normal BMI and 306 men with an overweight BMI (normal BMI group: 20–25 kg/m2; overweight BMI group: >28 kg/m2). We found that couples with male’s BMI over 28 kg/m2 exhibited a significantly lower fertilization rate, good-quality embryo rate and clinical pregnancy rate compared to their normal BMI counterparts. The mean STL in the overweight BMI group was also significantly shorter than that of the normal BMI group. The results also showed that individuals with higher BMI had higher ROS (Reactive oxygen species) content and sperm DNA fragmentation rate when compared with normal BMI individuals. Mitochondrial activity was also lower in the overweight BMI group than in the normal BMI group. This is the first report to find that STL is shorter in overweight/obese men, which may account for their poorer treatment outcomes in IVF cycles.
Overweight and obesity are ongoing worldwide epidemics, and their effects on reproductive health have been widely studied in women. Overweight or obese women experience longer times to pregnancy and reduced probabilities of conception relative to their normal weight counterparts1,2. Moreover, they are at an increased risk of early and recurrent miscarriage3. These women generally have reduced oocyte quality, lower developmental potential, and poor clinical outcomes when they undergo assisted reproduction treatment4,5,6. In contrast, few studies have focused on the impact of men’s BMI on assisted reproductive technology (ART) outcomes. In particular, the impact of the male’s BMI on embryonic development during IVF has been rarely studied. Numerous studies have examined the effects of overweight and obesity on semen parameters (e.g., sperm motility, morphology and count) with conflicting results. Several studies have shown a negative association between overweight BMI and standard semen parameters7,8,9, whereas other studies have shown the reverse10,11. Previous studies have also investigated the associations between BMI, testosterone and sex hormone serum levels, and some found that increased BMI was consistently associated with reduced levels of serum sex-hormone-binding globulin, inhibin B, T (testosterone), and FSH (follicle stimulating hormone)12,13. However, other studies did not observe these changes8,10.
Telomeres are highly repeated hexameric (TTAGGG) nucleotide sequences at the ends of chromosomes14. In association with bound proteins, telomeres protect the chromosome ends from being recognized as double-strand breaks requiring repair. One study found that the shortening of long telomeres in sperm can help sustain the critical telomere length required for normal embryonic cleavage post-fertilization15. Another study showed that wild-type oocytes fertilized by sperm with short telomeres from later generations of telomerase-null mice (TR−/−) led to increased embryo fragmentation, low embryo quality, and apoptosis16. Our most recent study also demonstrated that sperm telomere length (STL) is positively associated with embryo quality in IVF17.
Obesity is often accompanied by oxidative stress and inflammation18. Oxidative stress is an important contributing factor to telomere attrition19. Several studies have found significant negative associations between BMI and the telomere length of leukocytes in peripheral blood20,21,22,23. A recent study found that BMI was positively correlated with reactive oxygen species (ROS) production in semen24. However, little is known about the effects of overweight and obesity on STL or the role of these relationships in IVF treatment outcomes. The present study aimed to evaluate the effects of male BMI on fertilization rate, early embryonic development quality, and clinical pregnancy rate in couples undergoing IVF. We found that fertilization rates, high-quality embryo rates and clinical pregnancy rates were lower among couples with men whose BMIs were over 28 kg/m2 than with men whose BMIs were between 20 and 25 kg/m2. In addition, we assessed the mean STL for each patient, and the results showed that this mean value was significantly shorter in the overweight BMI group than in the normal BMI group. We also found that sperm mitochondrial activity was lower but that the sperm DNA fragmentation rate and ROS content in semen were higher in the overweight BMI group than the normal BMI group.
Results
Basic patient characteristics and IVF cycle outcomes
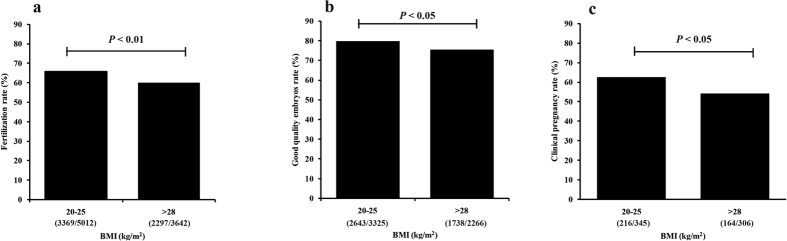
We enrolled 651 patients including 306 patients with an overweight BMI and 345 age-matched patients with a normal BMI undergoing IVF for the first time. The basic patient characteristics are shown in Table 1. The data are presented as the means ± standard deviations. Male’s age, female’s age, female BMI, female basal FSH level, Gn (gonadotropin hormone) duration, Gn dose, oocytes retrieved and embryos transferred did not significantly differ between the two groups. However, the semen parameter analysis showed that although sperm concentration did not exhibit between-group differences, sperm activity (a parameter assessed in semen analysis that consists of the ability of the sperm to swim in a forward direction), semen volume and total sperm count were significantly higher in the normal BMI group than in the overweight BMI group (Table 2). We also compared the IVF cycle outcomes between the two groups and found that the fertilization rate was significantly lower in the overweight BMI group than in the normal BMI group (Fig. 1a). When we compared the high-quality embryo (grade I and grade II) rate between the two groups, we found that it was significantly lower in the overweight BMI group (Fig. 1b). Furthermore, the clinical pregnancy rate was also significantly lower for this group than for the normal BMI group (Fig. 1c). To confirm the relationship between male BMI and pregnancy rate in IVF, we performed a multivariate logistic regression analysis. After adjusting for female BMI, female age, basal FSH level, male age, number of embryos transferred and sperm count, the data showed that males with a BMI >28 kg/m2 were associated with lower clinical pregnancy rates (Table 3).
Table 1. Demographic and IVF cycle characteristics according to male BMI.
| Parameter | Male BMI (kg/m2) | ||
|---|---|---|---|
| 20–25(N = 345) | >28(N = 306) | P | |
| Male Age (years) | 30.4 ± 4.0 | 30.5 ± 3.9 | 0.91 |
| Female Age (years) | 29.7 ± 4.9 | 29.9 ± 3.9 | 0.48 |
| Female BMI (kg/m2) | 22.4 ± 3.1 | 22.3 ± 3.6 | 0.26 |
| Female Basal FSH (IU/L) | 7.4 ± 2.1 | 6.9 ± 2.5 | 0.51 |
| Gn duration (days) | 11.2 ± 1.9 | 11.0 ± 1.9 | 0.13 |
| Gn dosage (IU) | 2,034.8 ± 822.6 | 2,028.5 ± 817.7 | 0.92 |
| Oocytes retrieved (n) | 11.3 ± 5.9 | 11.2 ± 6.5 | 0.63 |
| Embryos transferred (n) | 2.0 ± 0.3 | 1.9 ± 0.5 | 0.32 |
BMI, body mass index; FSH, follicle stimulating hormone; values are shown as the mean ± standard deviation; Student’s t test was used to compare the data between the two groups.
Table 2. Basic semen characteristics of the two BMI groups.
| Parameter | Male BMI (kg/m2) | ||
|---|---|---|---|
| 20–25(N = 345) | >28(N = 306) | P | |
| Sperm concentration (million/ml) | 59.9 ± 54.6 | 59.5 ± 51.8 | 0.83 |
| Semen volume (ml) | 3.08 ± 1.69 | 3.79 ± 1.71 | <0.01 |
| Total sperm count (million) | 234.5 ± 228.3 | 202.7 ± 155.3 | 0.04 |
| Sperm motility (%) | 46.5 ± 18.9 | 39.9 ± 19.1 | <0.001 |
Values are displayed as the mean ± standard deviation; Student’s t test was used to compare the data between the two groups.
Figure 1. Between-group comparison of fertilization, embryonic development and clinical pregnancy rate (the raw data were shown in the figures).
(a) Comparison of fertilization rates between couples with men with normal BMIs (20–25 kg/m2; N = 345) and overweight BMIs (>28 kg/m2; N = 306) (chi-square test). (b) Comparison of high-quality embryo rate on day 3 between couples with men with normal BMIs (N = 345) and those with men with overweight BMIs (N = 306, chi-square test). (c) Comparison of the clinical pregnancy rate between couples with men with normal BMIs (N = 345) and those with men with overweight BMIs (N = 306, chi-square test).
Table 3. Likelihood of clinical pregnancy after IVF, presented as odds ratios (95% confidence intervals).
| Male BMI: >28 kg/m2(N = 306) | |
|---|---|
| Unadjusted | 0.75 (0.64–0.88) |
| P < 0.001 | |
| Adjusted | 0.73 (0.62–0.88) |
| P < 0.01 | |
A multivariate logistic regression was used to investigate whether male BMI affects clinical pregnancy after adjusting for female BMI, female age, number of embryos transferred, basal FSH levels, male age, and sperm count for the overweight BMI group.
Shorter relative STLs in overweight/obese patients
Previous studies, including one from our own lab, have suggested that STL plays an important role in early embryonic development16,17,25. Because obese men usually have shorter LTLs (Leukocyte telomere lengths), we examined the relative STL for each patient. When we compared the STLs between the two groups, we found that the mean relative STL was significantly shorter in the overweight BMI group than the normal BMI group (Fig. 2). Previous studies have also found that STL is positively associated with age.
Figure 2.
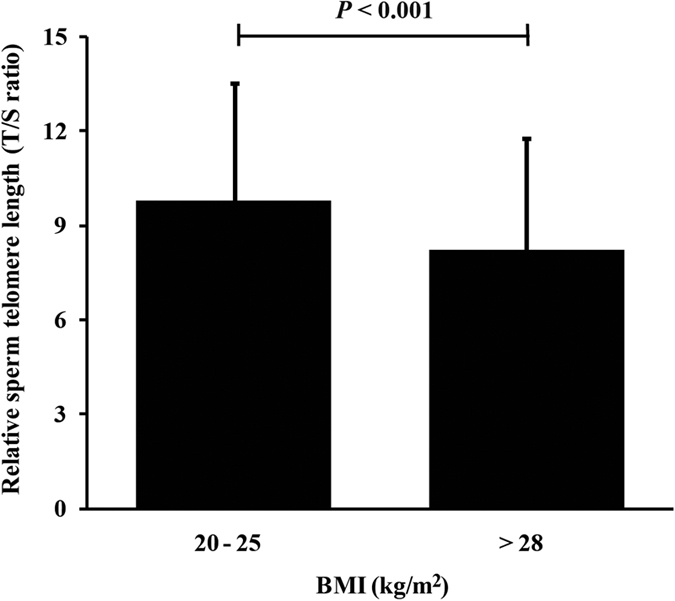
Comparison of relative STL (T/S ratio) in men with normal BMIs (20–25 kg/m2; N = 345) and overweight BMIs (>28 kg/m2; N = 306) (Student’s t test; bars represent standard deviations).
Increased oxidative stress, decreased DNA integrity and mitochondrial activity in the sperm of overweight/obese patients
To assess the levels of oxidative stress in the semen collected from the overweight BMI group, we used the NBT method to detect ROS content. We collected semen from another 104 patients, including 54 patients with a BMI >28 kg/m2 and 50 patients with normal BMIs. The production of formazan in the serum of men with overweight BMIs was significantly higher than that of those with normal BMIs (Supplementary Figure 1a; P < 0.01). This result implies that oxidative stress is prominent in overweight/obese men.
We performed a sperm chromatin dispersion test to determine the DNA fragmentation level for each patient. The results showed that the overweight/obesity group had a higher sperm DNA damage rate than the normal BMI group (Supplementary Figure 1b; P < 0.01). Shorter STLs were also found in the overweight BMI group than in the normal BMI group (Supplementary Figure 1c; P < 0.05). Those findings suggest that overweight/obese men have decreased DNA integrity. For sperm mitochondrial activity, the percentage of sperm with DAB class II and III was lower (Supplementary Table 1; DAB classes II: P < 0.03; DAB classes III: P = 0.04) and the percentage of sperm with DAB class IV was higher in overweight (Supplementary Table 1; P < 0.01) than in normal BMI men, suggesting that overweight/obese patients have decreased sperm mitochondrial activity.
Discussion
Infertility affects approximately 15% of couples worldwide, and male factors account for approximately 50% of all cases26. However, the mechanisms involved are poorly understood. Over the past two decades, ART has become more popular and has helped many infertile couples have their own children. Previous studies have shown that female obesity has a detrimental influence on fertility and treatment outcomes in IVF; however, relatively few studies have focused on the impact of male overweight/obesity during IVF.
Although several recent studies have explored the effects of male BMI on early embryonic quality and clinical pregnancy rate during IVF, they have reported conflicting results. One study including 344 infertile couples stated that men with a BMI over 25 kg/ m2 have a significantly lower chance of having partners achieve clinical pregnancy than men of normal weight; however, day 3 embryo quality was not affected27. Another study including 305 infertile couples found that an elevated paternal BMI was associated with a lower clinical pregnancy rate. However, no significant relationship was found with regard to early embryo development28. Furthermore, one study including 114 couples found no association between male BMI and IVF fertilization rate, embryo quality on day 3, or pregnancy rate29. In the present study, we investigated the effect of overweight/obesity on IVF treatment outcomes and tried to explore the possible mechanisms involved. We studied 651 couples, including 306 couples with a male with an overweight BMI and 345 with a male with a normal BMI. The data showed that sperm activity, semen volume and total sperm count were significantly higher in the normal BMI group than in the overweight BMI group. Although the etiology behind the reduced sperm parameters is not completely clear, several possible factors have been suggested as causes. Some studies found that an elevated BMI can impair spermatogenesis by causing an increase in scrotal temperature30,31, thus leading to impaired semen quality32. Other studies also reported that obesity is associated with increased oxidative stress33,34, an ROS content most likely higher than the usual metabolic rates required to maintain normal biological processes and an increased level of stress in the local testicular environment. Redundant ROS can lead to sperm DNA damage and affect normal sperm function and motility by disturbing mitochondrial genomes35.
In the present study, we also found that couples with a male BMI >28 kg/m2 had significantly lower fertilization rates, high-quality embryo rates and clinical pregnancy rates compared with their normal weight counterparts. A recent meta-analysis study in which thirty papers were included demonstrated that paternal obesity negatively affects male fertility and assisted reproduction outcomes, as shown by significantly reduced fertility in the general population and reduced rates of live birth from ART, as well as increased rates of nonviable pregnancy36. Combined with previous studies27,28, our results suggest that male adiposity impairs the treatment outcomes of IVF cycles, although the clear causal mechanism remains unclear. Some researchers have tried to explore the mechanisms of paternal obesity on early embryonic development using a mouse model of high fat diet. One study found some physiological changes to the preimplantation embryo, including significant delays in cell cycle progression, decreased blastocyst development and cell number, and increased glycolytic rate37. Functional studies examining blastocyst attachment, growth and implantation showed that blastocysts derived from the sperm of obese males displayed significantly reduced outgrowth on fibronectin and delayed fetal development following embryo transfer38. All the above results demonstrated that paternal obesity can affect embryonic development. However, it is not very clear which of the changes in sperm due to obesity ultimately affects early embryonic development.
More and more studies are focusing on investigating the role of telomere length and the telomerase system in reproduction. In telomerase-null mice, the fertilization of wild-type oocytes with sperm with short telomeres from late generations resulted in increased embryo fragmentation, low embryo quality, and apoptosis16. One study in humans showed that telomere lengths in cumulus cells could predict highly competent and high-quality oocytes39. Another study demonstrated that telomerase was a better predictor of pregnancy outcomes following IVF than telomere length40, and our recent study also found that STL was positively associated with early embryonic development in IVF cycles17. In addition, several studies have reported the effects of overweight/obesity on LTL20,21,22,23. We measured the STL of each patient; thus, this study is the first to evaluate how overweight/obesity affects relative telomere length in human sperm. Interestingly, we found that the mean STL was significantly shorter in males with a higher BMI compared with controls.
In the present study, we also investigated the ROS content in the semen of 54 overweight/obese men and 50 normal weight men. The results demonstrated that the former group had higher ROS levels in their semen than the latter group, which is consistent with previous findings showing that BMI is positively correlated with ROS content in sperm24. The present study also found that the sperm mitochondrial activity was lower and the DNA fragmentation rate higher among the overweight/obesity group than the normal weight group, which was consistent with a previous study41. Although ROS plays an important role in modulating essential sperm function, imbalances in ROS levels could lead to dysfunction of the mitochondria42 and DNA damage43. One study showed that when a human telomere sequence was inserted into a plasmid, it caused up to 7-fold more oxidation-induced DNA cleavage in the telomere sequence than a control sequence44. Another study found that telomere shortening is significantly increased under mild oxidative stress when compared with that observed under normal conditions in fibroblasts45. Over-expression of the extracellular superoxide dismutase gene in human fibroblasts decreased the peroxide content, further decreasing the rate of telomere shortening. All the results demonstrated that telomere length was sensitive to oxidative stress. Additionally, some studies found that high-energy intake can enhance oxidative stress within the testicular environment46,47. Intracellular ROS can also affect telomere function indirectly by their interactions with telomerase, which can lead to a loss of activity of telomerase reverse transcriptase48,49. Moreover, white adipose tissue can secrete various hormones, including leptin50, that increase sperm oxidative metabolism51, thus reducing sperm motility by decreasing axonemal protein phosphorylation, as well as by lipid peroxidation52. All the results of the present study combined with those of previous studies suggest that shorter TL, lower mitochondrial activity and higher DNA fragments in the sperm of high BMI patients may be due to increased ROS content.
In summary, we found that STL is closely associated with BMI and that higher BMIs are associated with shorter STLs, decreased mitochondrial activity and increased DNA fragmentation, which might be a result of increased ROS content. All of these changes might lead to lower fertilization rates, embryo quality and clinical pregnancy rates for patients undergoing IVF. Taken together, these findings help us partly understand the effects of paternal overweight/obesity on IVF treatment outcomes. We suggest that obesity increases inflammation and oxidative stress, which might cause shortening of sperm telomere length, ultimately leading to poor early embryo quality and reduced pregnancy rates. However, additional studies are required to determine the detailed causal mechanisms involved.
Experimental Methods
Patient selection
Our sample consisted of 306 overweight males (>28 kg/m2) and 345 age-matched males of normal weight (20–25 kg/m2). All patients were undergoing treatment at our reproductive medical center for first fresh IVF cycles. All patients had normal chromosome karyotypes. None of the men had Y chromosome micro-deletions or any evident causes of spermatogenic impairment. The FSH levels of all of the women were below 10 IU/L. Women were excluded when they had damaged ovarian function that was present prior to the study, such as surgery or use of harmful medications. After assessing the STL in these patients, we collected semen from another 104 patients, including 54 patients with a BMI >28 kg/m2 and 50 age-matched patients with normal BMIs to assess the impact of BMI on oxidative stress, mitochondrial activity and DNA fragmentation rate in the semen. This study was approved by the Ethics Committee of the First Affiliated Hospital of Zhengzhou University, and informed consent about the usage of data and semen was obtained from all participants. Finally, all the methods used in this study were carried out in accordance with the approved guidelines.
IVF procedure
The semen analysis was carried out according to the WHO protocol (2010) on the day that the oocytes were retrieved (Day 0). Insemination was conducted by placing cumulus–oocyte complexes (COCs) together with sperm selected via density gradient centrifugation. The cumulus cells were removed after 4 h. Fertilization was confirmed after 16–18 h of insemination. Each Day 3 embryo was evaluated and given a grade from 1 to 5. High-quality embryos included grade I and grade II embryos, which were defined as having six to eight symmetrical equal size blastomeres with no fragmentation (grade I) or less than 10% fragmentation (grade II).
Telomere length measurement
The average STL was determined in genomic DNA using a real-time PCR for each patient. The PCR reactions were conducted using the 7500 Real-Time PCR System (Applied Biosystems, USA). The telomeric primers, control gene primers (36B4, single-copy gene) and PCR settings were the same as described before17. Each sample was conducted in triplicate, and a standard curve was created by serial dilutions of known amounts of reference DNA for each reaction. The telomere to single (T/S) copy ratio was used to calculate the relative telomere length. Laboratory personnel who were blind to the case-control status and the outcome assessment performed all of the measurements.
ROS production assay
A standardized protocol was used to perform a photometric nitro blue tetrazolium (NBT) assay to measure seminal ROS production via formazan production53. After washing the semen in phosphate-buffered saline (PBS) at 300 g for 5 min, the sperm was re-suspended in 200 μl PBS. Then, 100 μl 0.1% NBT (Sigma-Aldrich, USA) was added and incubated at 37 °C for 45 min. The sperm were then washed twice in PBS at 500 g for 5 min. Exactly 60 μl DMSO (Sigma-Aldrich) and 2 M KOH were used to solubilize the intracellular formazan product. The resulting color reaction was measured with a microplate reader at 630 nm (BIO-RAD, Finland). ROS production was expressed as micro-grams of formazan per 107 spermatozoa. A standard curve was obtained from known concentrations of formazan substrate solubilized in DMSO.
Mitochondrial activity assay
Sperm mitochondrial activity was detected using the method described by Hrudka54. Briefly, the semen was diluted 1:3 in PBS with 1 mg/mL 3, 3′-diaminobenzidine (DAB) after semen liquefaction and then transferred to an incubator for 1 h at 37 °C. A 10 μl smear was added to a slide. Then, the air-dried slide was fixed using 10% formaldehyde for 10 min. Exactly 500 sperm were counted for each sample using an Olympus BX51 microscope. The sperm were classified based on the stained length of the midpiece. Classes I to III corresponded to 100%, more than 50%, and less than 50% of the midpiece being stained, respectively. Class IV had no staining in the midpiece.
DNA fragmentation determination
A sperm chromatin dispersion (SCD) kit was used to detect the DNA fragmentation rate in the sperm according to the manufacturer’s recommendations (BRED, Life Science Technology Inc., Shenzhen, China). Briefly, 60 μl of the diluted raw semen sample was added to an Eppendorf tube and mixed completely with fused agarose heated at 80 °C for 20 min. Exactly 30 μl of the mixture was added to a pre-coated slide, and the slide was cooled at 4 °C for 5 min. The slide was incubated in solution A for 7 min and solution B for 25 min and was then washed in distilled water for 5 min. After dehydration treatment via a series of ethanol concentrations (70–90–100%), the slide was incubated in a mixture of Wright’s solution and PBS for 16 min. The slide was then washed and air-dried. Exactly 500 sperm were counted for each sample using an Olympus BX51 microscope. Sperm nuclei with large- or medium-sized halos were considered non-fragmented, whereas sperm with small nuclei or without halos were considered to contain fragmented DNA.
Statistical analyses
The demographic data are presented as the means ± SDs for the two groups. The Kolmogorov–Smirnov normality test was used to examine deviations from normal distributions. Student’s t test was used to analyze normally distributed data, and Mann-Whitney U test was used to analyze non-parametric data. A chi-square test was used to analyze qualitative data. A general linear regression model was used to assess the relationship between STL and patient BMI. A multivariate logistic regression was used to investigate whether male BMI affects clinical pregnancy after adjusting for female BMI, female age, the number of embryos transferred, basal FSH levels, male age, and sperm count for the overweight BMI group. All statistical analyses were performed using SPSS software Version 16.0 (SPSS Inc., Chicago, IL, USA). A value of P < 0.05 was considered significant.
Additional Information
How to cite this article: Yang, Q. et al. Effect of paternal overweight or obesity on IVF treatment outcomes and the possible mechanisms involved. Sci. Rep. 6, 29787; doi: 10.1038/srep29787 (2016).
Supplementary Material
Acknowledgments
We would like to thank all the patients who participated in this study. This work was funded by grants from the National Natural Science Foundation of China (Grants 31271605 and 31471404 to Yingpu Sun), National Science Foundation for Young Scientists of China (Grant 31401274 to Qingling Yang), and the Youth Innovation Fund of the First Affiliated Hospital of Zhengzhou University (to Qingling Yang).
Footnotes
Author Contributions Y.S. and Q.Y. designed the experiment. F.Z., N.Z., R.B. and G.Y. collected the semen. F.Z. and Q.Y. performed the experiments and prepared all figures and tables. Y.S., Q.Y. and L.L. drafted the manuscript.
References
- Hassan M. A. & Killick S. R. Is previous aberrant reproductive outcome predictive of subsequently reduced fecundity? Human reproduction 20, 657–664, doi: 10.1093/humrep/deh670 (2005). [DOI] [PubMed] [Google Scholar]
- van der Steeg J. W. et al. Obesity affects spontaneous pregnancy chances in subfertile, ovulatory women. Human reproduction 23, 324–328, doi: 10.1093/humrep/dem371 (2008). [DOI] [PubMed] [Google Scholar]
- Lashen H., Fear K. & Sturdee D. W. Obesity is associated with increased risk of first trimester and recurrent miscarriage: matched case-control study. Human reproduction 19, 1644–1646, doi: 10.1093/humrep/deh277 (2004). [DOI] [PubMed] [Google Scholar]
- Ku S. Y. et al. Clinical efficacy of body mass index as predictor of in vitro fertilization and embryo transfer outcomes. Journal of Korean medical science 21, 300–303 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenoble C., Guibert J., Lefebvre G. & Dommergues M. [Effect of women’s weight on the success rate of in vitro fertilization]. Gynecologie, obstetrique & fertilite 36, 940–944, doi: 10.1016/j.gyobfe.2008.06.023 (2008). [DOI] [PubMed] [Google Scholar]
- Loveland J. B., McClamrock H. D., Malinow A. M. & Sharara F. I. Increased body mass index has a deleterious effect on in vitro fertilization outcome. Journal of assisted reproduction and genetics 18, 382–386 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kort H. I. et al. Impact of body mass index values on sperm quantity and quality. Journal of andrology 27, 450–452, doi: 10.2164/jandrol.05124 (2006). [DOI] [PubMed] [Google Scholar]
- Macdonald A. A., Stewart A. W. & Farquhar C. M. Body mass index in relation to semen quality and reproductive hormones in New Zealand men: a cross-sectional study in fertility clinics. Human reproduction 28, 3178–3187, doi: 10.1093/humrep/det379 (2013). [DOI] [PubMed] [Google Scholar]
- Belloc S. et al. High body mass index has a deleterious effect on semen parameters except morphology: results from a large cohort study. Fertility and sterility 102, 1268–1273, doi: 10.1016/j.fertnstert.2014.07.1212 (2014). [DOI] [PubMed] [Google Scholar]
- Aggerholm A. S., Thulstrup A. M., Toft G., Ramlau-Hansen C. H. & Bonde J. P. Is overweight a risk factor for reduced semen quality and altered serum sex hormone profile? Fertility and sterility 90, 619–626, doi: 10.1016/j.fertnstert.2007.07.1292 (2008). [DOI] [PubMed] [Google Scholar]
- Thomsen L., Humaidan P., Bungum L. & Bungum M. The impact of male overweight on semen quality and outcome of assisted reproduction. Asian journal of andrology 16, 749–754, doi: 10.4103/1008-682X.125398 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winters S. J. et al. Inhibin-B levels in healthy young adult men and prepubertal boys: is obesity the cause for the contemporary decline in sperm count because of fewer Sertoli cells? Journal of andrology 27, 560–564, doi: 10.2164/jandrol.05193 (2006). [DOI] [PubMed] [Google Scholar]
- Pauli E. M. et al. Diminished paternity and gonadal function with increasing obesity in men. Fertility and sterility 90, 346–351, doi: 10.1016/j.fertnstert.2007.06.046 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moyzis R. K. et al. A highly conserved repetitive DNA sequence, (TTAGGG)n, present at the telomeres of human chromosomes. Proceedings of the National Academy of Sciences of the United States of America 85, 6622–6626 (1988). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Achi M. V., Ravindranath N. & Dym M. Telomere length in male germ cells is inversely correlated with telomerase activity. Biology of reproduction 63, 591–598 (2000). [DOI] [PubMed] [Google Scholar]
- Liu L., Blasco M., Trimarchi J. & Keefe D. An essential role for functional telomeres in mouse germ cells during fertilization and early development. Developmental biology 249, 74–84 (2002). [DOI] [PubMed] [Google Scholar]
- Yang Q. et al. Sperm telomere length is positively associated with the quality of early embryonic development. Human reproduction 30, 1876–1881, doi: 10.1093/humrep/dev144 (2015). [DOI] [PubMed] [Google Scholar]
- Furukawa S. et al. Increased oxidative stress in obesity and its impact on metabolic syndrome. The Journal of clinical investigation 114, 1752–1761, doi: 10.1172/JCI21625 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Zglinicki T. Oxidative stress shortens telomeres. Trends in biochemical sciences 27, 339–344 (2002). [DOI] [PubMed] [Google Scholar]
- Lee M., Martin H., Firpo M. A. & Demerath E. W. Inverse association between adiposity and telomere length: The Fels Longitudinal Study. American journal of human biology : the official journal of the Human Biology Council 23, 100–106, doi: 10.1002/ajhb.21109 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Attas O. S. et al. Adiposity and insulin resistance correlate with telomere length in middle-aged Arabs: the influence of circulating adiponectin. European journal of endocrinology / European Federation of Endocrine Societies 163, 601–607, doi: 10.1530/EJE-10-0241 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt S. C. et al. Leukocyte telomeres are longer in African Americans than in whites: the National Heart, Lung, and Blood Institute Family Heart Study and the Bogalusa Heart Study. Aging cell 7, 451–458, doi: 10.1111/j.1474-9726.2008.00397.x (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valdes A. M. et al. Obesity, cigarette smoking, and telomere length in women. Lancet 366, 662–664, doi: 10.1016/S0140-6736(05)66630-5 (2005). [DOI] [PubMed] [Google Scholar]
- Tunc O., Bakos H. W. & Tremellen K. Impact of body mass index on seminal oxidative stress. Andrologia 43, 121–128, doi: 10.1111/j.1439-0272.2009.01032.x (2011). [DOI] [PubMed] [Google Scholar]
- de Frutos C. et al. Spermatozoa telomeres determine telomere length in early embryos and offspring. Reproduction 151, 1–7, doi: 10.1530/REP-15-0375 (2016). [DOI] [PubMed] [Google Scholar]
- Agarwal A., Mulgund A., Hamada A. & Chyatte M. R. A unique view on male infertility around the globe. Reproductive biology and endocrinology : RB&E 13, 37, doi: 10.1186/s12958-015-0032-1 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merhi Z. O. et al. Male adiposity impairs clinical pregnancy rate by in vitro fertilization without affecting day 3 embryo quality. Obesity 21, 1608–1612, doi: 10.1002/oby.20164 (2013). [DOI] [PubMed] [Google Scholar]
- Bakos H. W., Henshaw R. C., Mitchell M. & Lane M. Paternal body mass index is associated with decreased blastocyst development and reduced live birth rates following assisted reproductive technology. Fertility and sterility 95, 1700–1704, doi: 10.1016/j.fertnstert.2010.11.044 (2011). [DOI] [PubMed] [Google Scholar]
- Colaci D. S. et al. Men’s body mass index in relation to embryo quality and clinical outcomes in couples undergoing in vitro fertilization. Fertility and sterility 98, 1193–1199 e1191, doi: 10.1016/j.fertnstert.2012.07.1102 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasturi S. S., Tannir J. & Brannigan R. E. The metabolic syndrome and male infertility. Journal of andrology 29, 251–259, doi: 10.2164/jandrol.107.003731 (2008). [DOI] [PubMed] [Google Scholar]
- Hammoud A. O. et al. Male obesity and alteration in sperm parameters. Fertility and sterility 90, 2222–2225, doi: 10.1016/j.fertnstert.2007.10.011 (2008). [DOI] [PubMed] [Google Scholar]
- Jung A. & Schuppe H. C. Influence of genital heat stress on semen quality in humans. Andrologia 39, 203–215, doi: 10.1111/j.1439-0272.2007.00794.x (2007). [DOI] [PubMed] [Google Scholar]
- Davi G. & Falco A. Oxidant stress, inflammation and atherogenesis. Lupus 14, 760–764 (2005). [DOI] [PubMed] [Google Scholar]
- Dandona P., Aljada A., Chaudhuri A., Mohanty P. & Garg R. Metabolic syndrome: a comprehensive perspective based on interactions between obesity, diabetes, and inflammation. Circulation 111, 1448–1454, doi: 10.1161/01.CIR.0000158483.13093.9D (2005). [DOI] [PubMed] [Google Scholar]
- Agarwal A., Nandipati K. C., Sharma R. K., Zippe C. D. & Raina R. Role of oxidative stress in the pathophysiological mechanism of erectile dysfunction. Journal of andrology 27, 335–347, doi: 10.2164/jandrol.05136 (2006). [DOI] [PubMed] [Google Scholar]
- Campbell J. M., Lane M., Owens J. A. & Bakos H. W. Paternal obesity negatively affects male fertility and assisted reproduction outcomes: a systematic review and meta-analysis. Reproductive biomedicine online 31, 593–604, doi: 10.1016/j.rbmo.2015.07.012 (2015). [DOI] [PubMed] [Google Scholar]
- Binder N. K., Mitchell M. & Gardner D. K. Parental diet-induced obesity leads to retarded early mouse embryo development and altered carbohydrate utilisation by the blastocyst. Reproduction, fertility, and development 24, 804–812, doi: 10.1071/RD11256 (2012). [DOI] [PubMed] [Google Scholar]
- Binder N. K., Hannan N. J. & Gardner D. K. Paternal diet-induced obesity retards early mouse embryo development, mitochondrial activity and pregnancy health. PloS one 7, e52304, doi: 10.1371/journal.pone.0052304 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng E. H. et al. Evaluation of telomere length in cumulus cells as a potential biomarker of oocyte and embryo quality. Human reproduction 28, 929–936, doi: 10.1093/humrep/det004 (2013). [DOI] [PubMed] [Google Scholar]
- Wang W. et al. Telomerase activity is more significant for predicting the outcome of IVF treatment than telomere length in granulosa cells. Reproduction 147, 649–657, doi: 10.1530/REP-13-0223 (2014). [DOI] [PubMed] [Google Scholar]
- Fariello R. M. et al. Association between obesity and alteration of sperm DNA integrity and mitochondrial activity. BJU international 110, 863–867, doi: 10.1111/j.1464-410X.2011.10813.x (2012). [DOI] [PubMed] [Google Scholar]
- Wang X. et al. Alterations in mitochondria membrane potential and oxidative stress in infertile men: a prospective observational study. Fertility and sterility 80 Suppl 2, 844–850 (2003). [DOI] [PubMed] [Google Scholar]
- Kodama H., Yamaguchi R., Fukuda J., Kasai H. & Tanaka T. Increased oxidative deoxyribonucleic acid damage in the spermatozoa of infertile male patients. Fertility and sterility 68, 519–524 (1997). [DOI] [PubMed] [Google Scholar]
- Henle E. S. et al. Sequence-specific DNA cleavage by Fe2+-mediated fenton reactions has possible biological implications. The Journal of biological chemistry 274, 962–971 (1999). [DOI] [PubMed] [Google Scholar]
- Serra V., von Zglinicki T., Lorenz M. & Saretzki G. Extracellular superoxide dismutase is a major antioxidant in human fibroblasts and slows telomere shortening. The Journal of biological chemistry 278, 6824–6830, doi: 10.1074/jbc.M207939200 (2003). [DOI] [PubMed] [Google Scholar]
- Bakos H. W., Mitchell M., Setchell B. P. & Lane M. The effect of paternal diet-induced obesity on sperm function and fertilization in a mouse model. International journal of andrology 34, 402–410, doi: 10.1111/j.1365-2605.2010.01092.x (2011). [DOI] [PubMed] [Google Scholar]
- Rato L. et al. Pre-diabetes alters testicular PGC1-alpha/SIRT3 axis modulating mitochondrial bioenergetics and oxidative stress. Biochimica et biophysica acta 1837, 335–344, doi: 10.1016/j.bbabio.2013.12.008 (2014). [DOI] [PubMed] [Google Scholar]
- Haendeler J. et al. Antioxidants inhibit nuclear export of telomerase reverse transcriptase and delay replicative senescence of endothelial cells. Circulation research 94, 768–775, doi: 10.1161/01.RES.0000121104.05977.F3 (2004). [DOI] [PubMed] [Google Scholar]
- Zhu H., Fu W. & Mattson M. P. The catalytic subunit of telomerase protects neurons against amyloid beta-peptide-induced apoptosis. Journal of neurochemistry 75, 117–124 (2000). [DOI] [PubMed] [Google Scholar]
- Wang P., Mariman E., Renes J. & Keijer J. The secretory function of adipocytes in the physiology of white adipose tissue. Journal of cellular physiology 216, 3–13, doi: 10.1002/jcp.21386 (2008). [DOI] [PubMed] [Google Scholar]
- Lampiao F. & du Plessis S. S. Insulin and leptin enhance human sperm motility, acrosome reaction and nitric oxide production. Asian journal of andrology 10, 799–807, doi: 10.1111/j.1745-7262.2008.00421.x (2008). [DOI] [PubMed] [Google Scholar]
- Agarwal A., Makker K. & Sharma R. Clinical relevance of oxidative stress in male factor infertility: an update. American journal of reproductive immunology 59, 2–11, doi: 10.1111/j.1600-0897.2007.00559.x (2008). [DOI] [PubMed] [Google Scholar]
- Tunc O., Thompson J. & Tremellen K. Development of the NBT assay as a marker of sperm oxidative stress. International journal of andrology 33, 13–21, doi: 10.1111/j.1365-2605.2008.00941.x (2010). [DOI] [PubMed] [Google Scholar]
- Hrudka F. Cytochemical and ultracytochemical demonstration of cytochrome c oxidase in spermatozoa and dynamics of its changes accompanying ageing or induced by stress. International journal of andrology 10, 809–828 (1987). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.