Abstract
Within the interleukin-17 (IL-17) family of cytokines, IL-17A is known to be critical in the host defense against fungal infections; however, the function of the other IL-17 family members in anti-fungal immunity remains largely unknown. Here, we show that IL-17C expression was highly induced in kidney epithelial cells after fungal infection. Mice that lacked IL-17C exhibited increased survival and attenuated kidney tissue damage, although they had similar fungal loads. IL-17C deficiency resulted in decreased pro-inflammatory cytokine expression compared with wild-type control mice. Additionally, IL-17C directly acted on renal epithelial cells in vitro to promote pro-inflammatory cytokine production. Taken together, our data demonstrate that IL-17C is a critical factor that potentiates inflammatory responses and causes host injury during fungal infection.
Keywords: fungal infection, IL-17C, kidney injury, pro-inflammatory cytokine, sepsis
Introduction
Candida albicans is among the most common clinical fungal pathogens, and it can have a tremendous influence on human life and can result in a high morbidity rate.1,2,3 Studies on the host immune responses to C. albicans infection have revealed that both immune deficiency and the over-activation of inflammatory responses contribute to fungus-associated diseases.4 A systemic candidiasis mouse model has been extensively studied, in which mice die of progressive sepsis with multiple organ failure, especially in the kidney, which is the primary infected organ.5 The immunopathological basis of sepsis has been reported to be mediated by the classical pro-inflammatory cytokines interleukin 1 (IL-1) and tumor-necrosis factor-α (TNF-α).6,7
The IL-17 cytokine family includes IL-17A, B, C, D, E, and F; of these, IL-17A is the most extensively studied.8,9,10,11 IL-17A is pathogenic in the development of a variety of inflammatory diseases, such as multiple sclerosis and arthritis.9,12 Moreover, IL-17A secreted from Th17 cells and gamma-delta T cells was found to be important in anti-fungal immune responses during C. albicans infection through stimulating neutrophil recruitment and function.13,14,15 Moreover, IL-17A-deficient mice were shown to be susceptible to fungal infection.16,17 Furthermore, patients with Job's Syndrome, which includes STAT3 mutations that lead to Th17 cell defects, were shown to have an elevated susceptibility to C. albicans infection.18,19 Additionally, Th1 cells and interferon gamma (IFN-γ) production play a critical role in the host defense against fungal infections, and mice with IFN-γ deficiency are highly susceptible to infection.20 These studies suggested that both Th1 and Th17 cells provide protective immune responses against fungal infection. However, the functions of the other IL-17 cytokine family members in systemic candidiasis are still largely unknown.
IL-17C, which is a member of the IL-17 cytokine family, has recently been studied and has emerged as a critical player in mucosal immunity and autoimmunity. IL-17C was first shown to stimulate TNF-α and IL-1β expression in the human THP-1 monocytic cell line.21 Additionally, the overexpression of IL-17C in CD4+ T cells aggravated the pre-existing arthritis,22 which suggests that IL-17C may be a pro-inflammatory cytokine. Three independent groups subsequently demonstrated that IL-17C signals through IL-17RE, which is expressed on epithelial cells and TH17 cells.23,24,25 IL-17C/IL-17RE plays a critical role in promoting protective innate immune responses to intestinal pathogens by inducing antibacterial peptides and pro-inflammatory cytokine expression in colon epithelial cells.25 Further, it potentiates Th17 cell responses in experimental autoimmune encephalomyelitis.24
Because IL-17RE is highly expressed in the kidney,23 which is the primary infected organ in systemic fungal infections, we hypothesized that IL-17C may play a role in fungal infections. In this study, we found that renal epithelial cells expressed IL-17C after fungal infection. Furthermore, Il17c-deficient mice were highly resistant to systemic candidiasis, which was associated with decreased pro-inflammatory cytokine expression, including IL-6 and IL-1β, in the infected kidney tissue. Thus, our results demonstrate that IL-17C-mediated inflammation can be detrimental during fungal infections.
Materials and methods
Mice
Il17c−/− mice and WT controls at six to eight weeks of age on the C57BL/6 background were used for the experiments. All of the mice were maintained under specific pathogen-free conditions. All animal experiments were approved by the Institutional Animal Care and Use Committee at Tsinghua University.
The utilized systemic candidiasis mouse model
Mice were injected via their lateral tail veins with C. albicans (SC5314) cells in sterile saline. The C. albicans cells were grown overnight at 30 °C in yeast, peptone, and dextrose medium for yeast formation. The cells were centrifuged, washed in phosphate-buffered saline (PBS) and counted before intravenous infection. After C. albicans infection, the survival rates were monitored for several days. For fungal-burden determinations, the tissues were weighed, homogenized in PBS and plated on agar plates in a series of diluted solutions. After 48 hr of incubation at 37 °C, the colony-forming unit (CFU) were counted and determined as CFU/g tissue.
RNA isolation and quantitative reverse transcriptase polymerase chain reaction (RT-PCR)
Total RNA was isolated from cells or mouse tissues using TRIzol reagent (Invitrogen, Grand Island, NY, USA) according to the manufacturer's instructions. cDNA was synthesized with oligo(dT), random hexamers, and M-MLV reverse transcriptase (Invitrogen). Then, the cDNA was amplified with real-time PCR using the IQ SYRB Green Supermix (Bio-Rad Laboratories, Hercules, CA, USA) on a CFX96 Real-Time System (Bio-Rad Laboratories). The cytokines, IL-17RE, and kidney injury molecule-1 (KIM-1) expression was normalized to GAPDH, which was utilized as a reference gene, and the C. albicans ITS1-2 rDNA expression in the kidney was normalized to β-actin.
Histology and immunohistochemistry (IHC)
Mice were euthanized post-infection, and the kidneys were removed, fixed with 10% formalin, dehydrated in ethanol and embedded in paraffin. The tissue sections were mounted on glass slides, processed for hematoxylin and eosin (H&E) and periodic acid Schiff staining, and evaluated for kidney pathology and IHC. Two independent pathologists scored the histology in a blinded manner, and the presence of necrosis, inflammation and hyperemia were evaluated according to the following scoring system: 0 = none, 1 = minimal, 2 = moderate, and 3 = severe. The total score was determined by the summation of the scores from the three categories. To analyze the IL-17C or IL-17A expression in the kidney, the sections were pretreated with boiling citrate buffer for 10 min to retrieve the antigens. After cooling, the sections were treated with 3% H2O2 for 15 min and then washed in PBS three times for 5 min each. Then, the sections were stained with rabbit anti-IL-17C (Bioss) or rabbit anti-mouse IL-17A (Santa Cruz Biotechnology, Santa Cruz, CA, USA) antibodies.
Cytokine measurements
The TNF-α, IL-1β, and IL-6 levels in the serum and extracts of kidney tissue from infected mice or kidney epithelial cell supernatants were measured using enzyme-linked immunosorbent assay kits (eBioscience, San Diego, CA, USA). In some experiments, the kidney epithelial cells were stimulated with IL-17C at final concentration of 100 ng ml−1 for 24 hr.
Mouse primary kidney epithelial cell isolation and stimulation
The kidneys were minced into pieces and incubated for 1 hr at 37 °C in a Dulbecco's Modified Eagle Medium: Nutrient Mixture F-12 (DMEM-F12) digestion buffer that contained 1 mg ml−1 collagenase A (Roche, Indianapolis, IN, USA) on a shaker. The digested tissue was filtered and washed three times. The cell pellet was then resuspended in DMEM-F12, which contained 10% (vol/vol) fetal bovine serum (FBS), penicillin (100 µg ml−1), and streptomycin (100 µg ml−1), and was seeded into 6-well plates. Five days later, the epithelial cells were stimulated for 4 hr in the presence or absence of TNF-α (10 ng ml−1), IL-1α (10 ng ml−1), IL-1β (10 ng ml−1), IL-6 (20 ng ml−1), or IL-17A (50 ng ml−1) or were stimulated for 24 hr in the presence or absence of IL-17C (100 ng ml−1).
CD45+ leukocyte isolation from the kidney
Leukocytes were isolated from the kidney by mincing the tissues into pieces and digested for 1 hr at 37 °C in DMEM-F12 digestion buffer containing 1 mg ml−1 collagenase A (Roche). The digested tissues were passed through a 70-µm filter, washed and centrifuged in a 40/70% Percoll gradient. The leukocytes at the interphase were first incubated with rat IgG anti-mouse CD45 (BD Bioscience, San Diego, CA, USA) and were then purified with an anti-rat IgG Micro Bead Kit (Miltenyi Biotec, Bergisch Gladbach, Germany). The CD45+ cells were purified with an AutoMACS separator (Miltenyi Biotec). Flow cytometric analysis indicated that the isolated cell population contained 90% CD45+ cells.
Flow cytometry
To determine the percentage of neutrophils that had infiltrated the kidney, the kidney single cell suspension, after a Percoll isolation, was incubated at 4 °C for 30 min with the following anti-mouse antibodies: Percp/Cy5.5-conjugated CD45 (30-F11; eBioscience); efluor 450-conjugated Ly6C (HK1.4; eBioscience); PE-conjugated Ly6G (RB6-8C5; eBioscience); APC-conjugated CD11b (M1/70; eBioscience). Flow cytometric analysis was performed with a BD LSRFortessa. The data were analyzed using FlowJo software (Tristar, Phoenix, AZ, USA).
Statistical analysis
The Prism software (GraphPad, CA, USA) was used for all of the statistical analyses. Two-tailed Student's t-tests were used to analyze any differences between the groups. p values less than 0.05 were considered statistically significant.
Results
IL-17C is highly expressed in the kidney after fungal infection
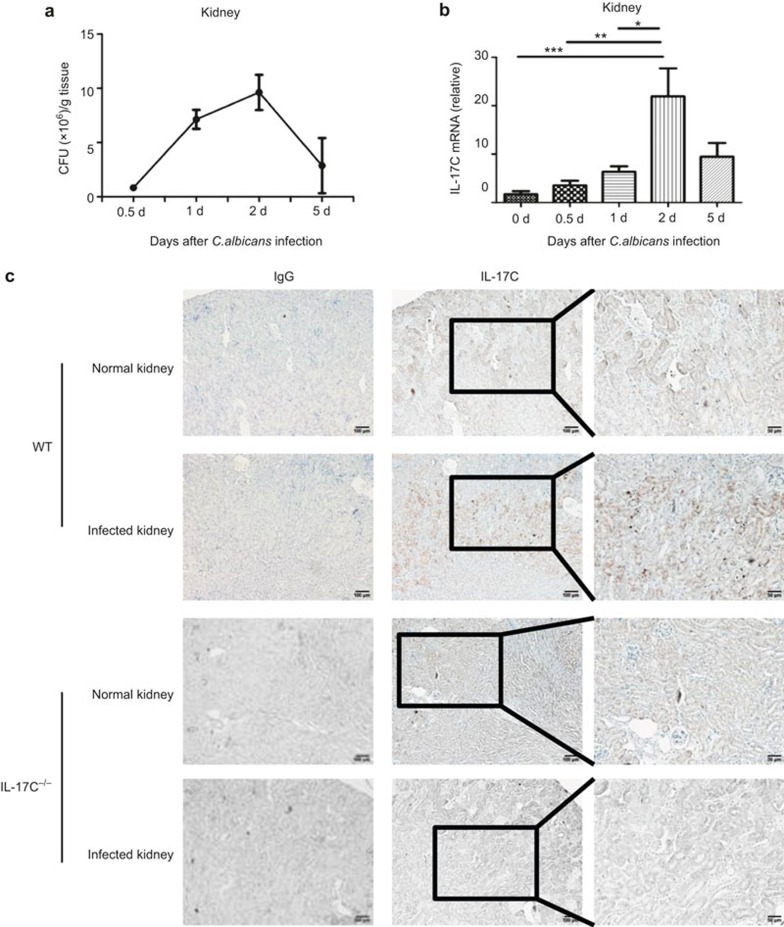
To elucidate the potential role of IL-17C during fungal infections, we first determined the IL-17C expression levels in C. albicans-infected kidney tissue. We challenged the C57BL/6 mice with 5 × 105 CFU of C. albicans and tested the IL-17C mRNA expression at different time points in the kidney, which is the primary target organ.5,26 A CFU assay confirmed the highest fungal burden in the kidney occurred at day 2 (Figure 1a). Moreover, we found that the expression of IL-17C mRNA was highly expressed in the kidneys of the C. albicans-challenged mice, beginning at 12 hr after infection and peaking on day 2, which was consistent with the fungal-burden peak (Figure 1b). However, in the other fungal-infected tissues, such as the lungs, liver, and spleen, there was no correlation between the IL-17C expression kinetics and the fungal burden at different time points after C. albicans challenge (data not shown).
Figure 1.
IL-17C is expressed by renal epithelial cells following systemic fungal infection. The fungal burden (a) and IL-17C expression (b) in the kidneys of fungal infected mice (n = 4 for each time point) on 0.5-, 1-, 2-, and 5-day post-infection with 5 × 105 CFU of C. albicans. (c) IL-17C expression in normal and infected kidney of wild-type (WT) mice and Il17c−/− mice was analyzed with IHC at day 2 after intravenous infection with 5 × 105 CFU of C. albicans. Original magnifications: 10× (left and middle) and 20× (right), scale bars: 100 µm for 10× and 50 µm for 20×. The data shown are representative of two independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001 (one-way analysis of variance).
To determine the type of cells in the kidney that express IL-17C during fungal infection, we measured the IL-17C protein expression in the kidney at day 2 with IHC. IL-17C was highly expressed by epithelial cells in the kidney after fungal infection (Figure 1c). Previous studies have indicated an important role for IL-17A in fungal infections,13,16,17,27 and Th17 cells are widely accepted as the major source of IL-17A in this model.28,29 Consistently, we also found high IL-17A expression levels in the kidney (Supplementary Figure 1a), and it was highly expressed on infiltrating inflammatory cells rather than on the epithelial cells (Supplementary Figure 1b). Furthermore, we detected higher IL-25 expression levels in the kidney following fungal infection (Supplementary Figure 1c). In summary, the IL-17C expression in the renal epithelial cells during systemic candidiasis suggested a potential role for IL-17C during fungal infections.
Il17c-deficient mice had increased survival following C. albicans infection
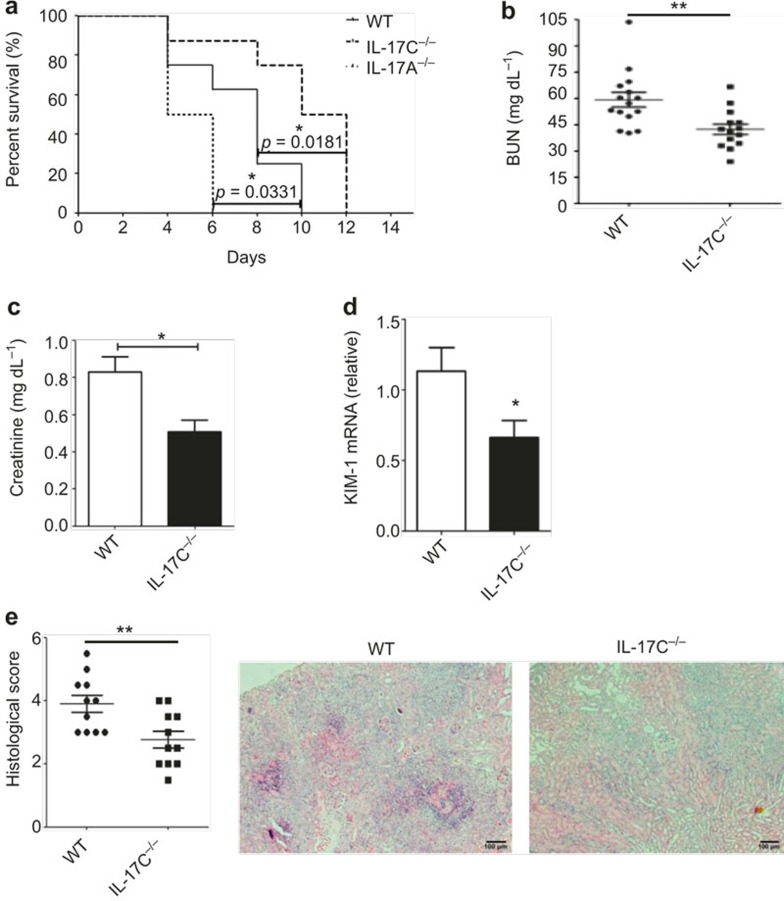
To investigate the role of IL-17C in C. albicans infection, we challenged C57BL/6 WT, Il17c-deficient and Il17a-deficient mice with 3.5 × 105 CFU of C. albicans and monitored the survival rates of these three groups. The IL-17C deficiency significantly increased the survival of the C. albicans infected mice compared with the WT controls (Figure 2a). In contrast, IL-17A deficiency dramatically accelerated the death of the fungal-infected mice (Figure 2a), which is in accordance with the reported protective role of IL-17A in systemic candidiasis.14,16 Moreover, when the mice were intravenously injected with a higher (5 × 105) or lower (2 × 105) C. albicans CFU level, the Il17c−/− mice consistently showed an increased resistance to fungal infection (Supplementary Figure 2).
Figure 2.
Il17c-deficient mice are highly resistant to C. albicans infection. (a) Survival curves of WT (n = 8), Il17c−/− (n = 8) and Il17a−/− mice (n = 6) that were intravenously infected with 3.5 × 105 CFU of C. albicans (SC5314). (b–c) Kidney failure in WT and Il17c-deficient mice was analyzed with BUN (b) and creatinine (c) measurements in mouse serum at day 3 after intravenous infection with 3.5 × 105 CFU of C. albicans. (d) Relative KIM-1 mRNA expression in WT and Il17c−/− mice at day 3 after intravenous infection with 3.5 × 105 CFU of C. albicans was analyzed with real-time RT-PCR. (e) Histological scores of WT and Il17c−/− mouse kidneys at day 3 after intravenous infection with 3.5 × 105 CFU of C. albicans were measured. Representative microscopic pictures of H&E-stained kidneys from mice infected with C. albicans for three days are shown on the right. The scale bar represents 100 µm. The data shown were obtained from three independent experiments. *p < 0.05, **p < 0.01 (t-test).
To further assess the kidney failure, we analyzed the blood urea nitrogen (BUN) and serum creatinine concentrations as well as the KIM-1 mRNA expression, which is a marker of tubular epithelial damage.30 Compared with the WT mice, the Il17c−/− mice exhibited attenuated kidney injury with a significant reduction in the serum BUN and creatinine concentrations as well as lower KIM-1 expression levels in the kidneys of the infected mice on day 3 (Figure 2b–d). Histological analysis with H&E staining also showed that kidney damage was decreased in the Il17c−/− mice (Figure 2e). Hence, the results demonstrated that the Il17c-deficient mice had less severe kidney failure with a higher survival rate than did the WT mice after C. albicans infection, which indicates a distinct role from that of IL-17A.
Improved survival in Il17c-deficient mice was not attributed to reduced fungal burden or neutrophil infiltration
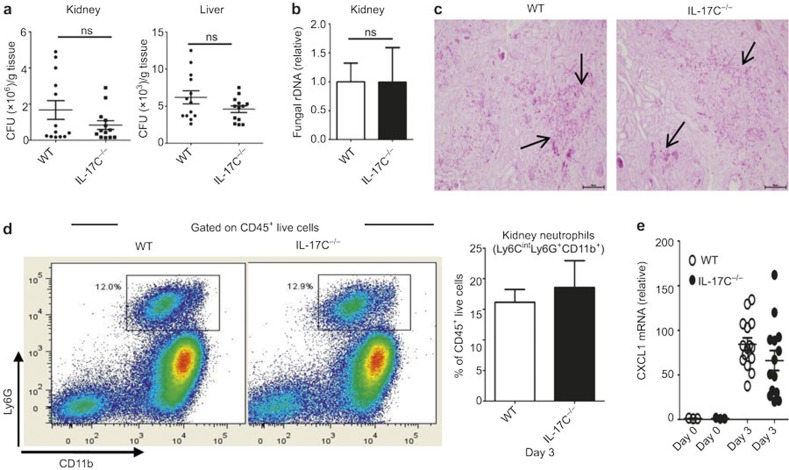
Because the kidney injury in the fungus-challenged mice was thought to correlate with the fungal burden level,5 we further determined the fungal burden with a CFU assay and analyzed the C. albicans ITS1-2 rDNA levels.31 However, to our surprise, despite the decreased kidney failure in the Il17c-deficient mice after C. albicans challenge, the fungal burden in the kidney and liver of these mice was not significantly different from that in the WT mice on days 2 and 3 (Figure 3a and b, Supplementary Figure 3). Moreover, no difference was observed in the C. albicans distribution in the kidney, as determined with periodic-acid Schiff staining (Figure 3c). Together, our data indicate that IL-17C deficiency resulted in improved survival, attenuated kidney tissue damage and decreased renal failure after systemic C. albicans infection; however, these phenomena not caused by reduced fungal burden.
Figure 3.
IL-17C deficiency improves survival without affecting tissue fungal burden and neutrophil infiltration. (a) CFU assays of paired mouse kidneys (left) and liver (right) tissues from WT and Il17c-deficient mice were determined at day 3 after intravenous infection with 3.5 × 105 CFU of C. albicans. (b) The fungal loads in the WT and Il17c-deficient mouse kidneys at day 3 after intravenous infection with 3.5 × 105 CFU of C. albicans. The ITS1-2 rDNA levels were analyzed with real-time PCR and normalized to β-actin DNA. (c) Kidney histopathology was analyzed with periodic acid-Schiff (PAS) staining at day 3 after intravenous infection with 3.5 × 105 CFU of C. albicans. Original magnifications: 20×, scale bars, 100 µm. (d) Representative FACS plots (left) and the statistical analysis (right) of Ly6CintLy6G+CD11b+ neutrophils in WT and Il17c-deficient mouse kidneys at day 3 after 3.5 × 105 CFU C. albicans infection. (e) Relative mRNA expression of CXCL1 in WT and Il17c-deficient kidneys that were intravenously infected with 3.5 × 105 CFU C. albicans at day 3 after infection. The data were obtained from at least three independent experiments.
Studies regarding invasive candidiasis have demonstrated that neutrophil infiltration could exacerbate kidney injury.32 Because IL-17C has been reported to induce neutrophil infiltration,23,33 we determined the infiltrated neutrophil percentage in the kidneys on day 3. As shown in Figure 3d, IL-17C deficiency did not affect neutrophil infiltration in the kidney. Because CXCL1 is the major chemokine that induces neutrophil infiltration,34 we also determined the CXCL1 mRNA expression in the kidney after fungal infection. Consistent with the neutrophil data, no differences in CXCL1 mRNA expression were observed (Figure 3e). Taken together, these data indicate that reduced kidney failure in the Il17c-deficient mice was not attributed to decreased neutrophil infiltration.
IL-17C deficiency results in reduced pro-inflammatory cytokine expression
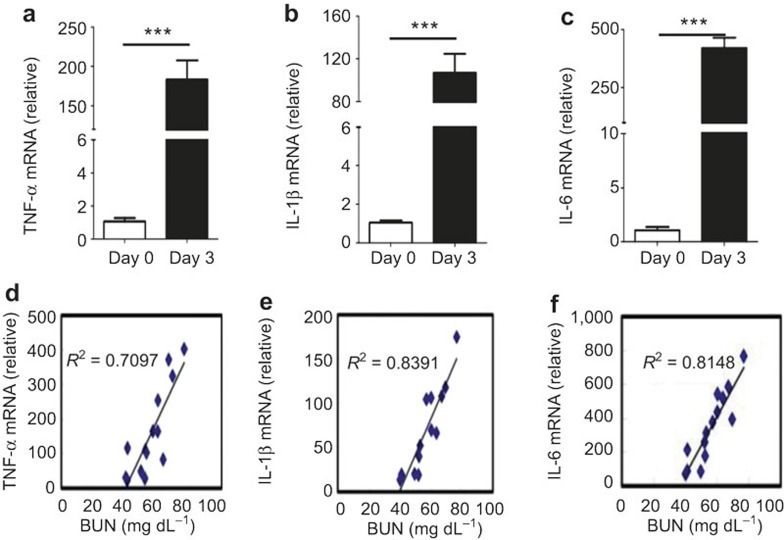
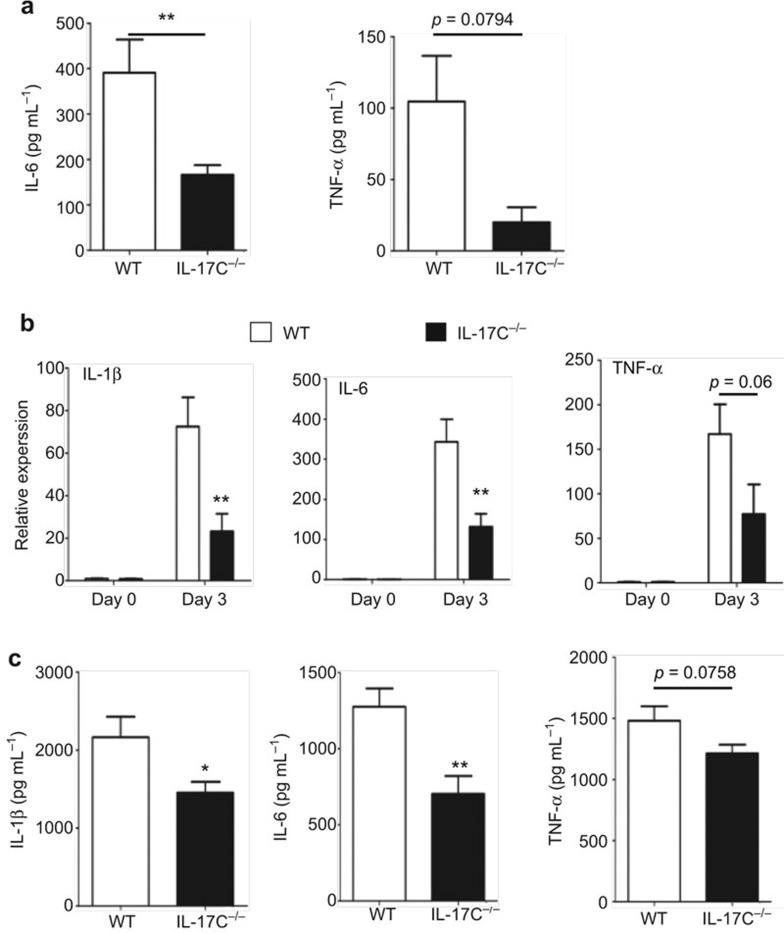
Although inflammatory cytokines, such as TNF-α, IL-1β, and IL-6 can help the immune system eliminate pathogens,25 their excessive production can result in tissue damage.7,22,35,36 Sepsis mediated by the pro-inflammatory cytokines IL-1 and TNF-α was reported as the major cause of death in mice with systemic candidiasis.5,6,37,38 These studies led us to the hypothesis that the systemic fungus infection-induced mortality in mice could be the result of excessive proinflammatory cytokine expression. To assess this hypothesis, we measured the expression of these pro-inflammatory cytokines in the kidney three days after C. albicans infection. We found that TNF-α, IL-1β, and IL-6 were highly induced in WT mice after fungal infection, with 200-fold increased TNF-α expression and 500-fold increased IL-6 expression compared with normal mice (Figure 4a–c). A correlation analysis between the kidney injury (with the BUN concentration as the readout) and the pro-inflammatory cytokine expression in the kidney revealed that the kidney injury was positively correlated with the pro-inflammatory cytokine expression (Figure 4d–f).
Figure 4.
Kidney failure following systemic fungal infection was positively correlated with pro-inflammatory cytokines expression. (a–c) TNF-α (a), IL-1β (b), and IL-6 (c) mRNA expression in the kidneys from normal and fungal-infected WT mice at day 3 after intravenous infection with 3.5 × 105 CFU of C. albicans. (d–f) The correlation analysis of serum inflammatory cytokine expression, including TNF-α (d), IL-1β (e), and IL-6 levels (f) as well as the BUN concentration at day 3 following infection with 3.5 × 105 CFU of C. albicans. The data shown were obtained from three independent experiments. ***p < 0.001 (t-test).
Because IL-17C was identified as inducing TNF-α, IL-1β, and IL-6 expression in colon tissue,25 we considered whether the increased survival and attenuated kidney failure in Il17c-deficient mice after infection were attributed to a reduction in the pro-inflammatory cytokine levels. We challenged the WT mice and Il17c-deficient mice with 3.5 × 105 CFU of C. albicans and compared the proinflammatory cytokine expression in the kidney tissue and serum on day 3. IL-6 production was significantly reduced in the Il17c-deficient mouse serum compared with the WT mice (Figure 5a). Furthermore, we determined the TNF-α, IL-1β, and IL-6 mRNA and protein levels in the WT and Il17c-deficient mouse kidneys. We found that the IL-6 and IL-1β expression levels were significantly lower in the Il17c-deficient mouse kidneys than in the WT mice (Figure 5b and c); however, other cytokines, including IL-10, IFN-γ, and IL-17A, were not significantly different between the groups (Supplementary Figure 4). Notably, TNF-α expression was slightly decreased in the serum and kidneys of the Il17c-deficient mice (Figure 5). Taken together, these results indicate that IL-17C is a critical factor that exacerbates inflammation that may cause host damage.
Figure 5.
IL-17C enhances the inflammatory cytokine expression in the serum and kidneys following infection. WT and Il17c-deficient mice were intravenously infected with 3.5 × 105 CFU of C. albicans. Three days later, pro-inflammatory cytokine expression levels in the serum and kidney tissues were measured. (a) WT or Il17c−/− mouse serum TNF-α and IL-6 ELISA results after fungal infection are shown. (b) Real-time RT-PCR analysis of IL-1β, IL-6, and TNF-α expression levels in WT and Il17c-deficient mouse kidneys. (c) Cytokine levels, including IL-1β, IL-6, and TNF-α, in the extracts of homogenized kidneys from infected WT and Il17c-deficient mice at day 3 are displayed. All of the data shown were obtained from three independent experiments. *p < 0.05, **p < 0.01 (t-test).
IL-17C regulates inflammatory cytokine expression in renal epithelial cells
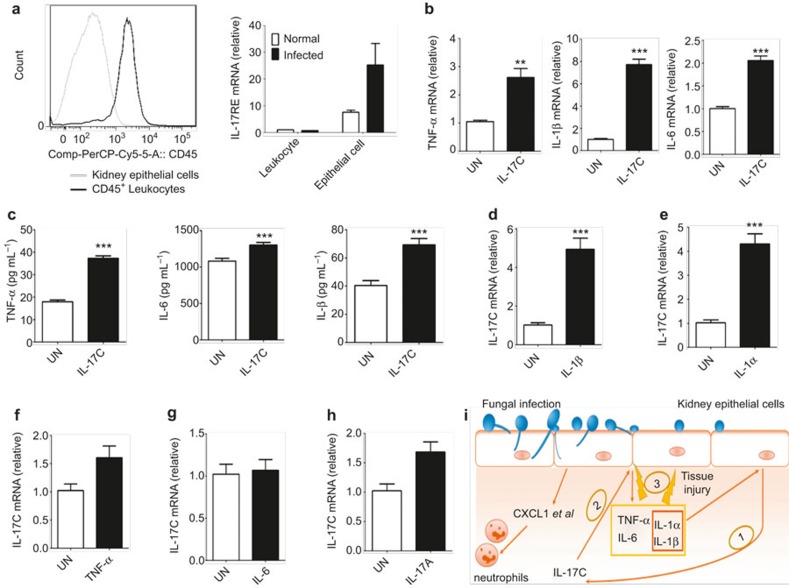
To further determine how IL-17C functions in fungal infections, we analyzed the expression of IL-17RE, the IL-17C receptor, in leukocytes and epithelial cells isolated from normal and infected WT mice with real-time RT-PCR (Figure 6a). We found that the kidney epithelial cells expressed IL-17RE and that this expression was further upregulated following infection (Figure 6a). Furthermore, LPS could also upregulate IL-17RE expression in epithelial cells, which indicated that the IL-17RE upregulation in epithelial cells was not Candida specific (Supplementary Figure 5). These data suggest that IL-17C can directly act on kidney epithelial cells. To test this hypothesis, we isolated kidney epithelial cells and examined their response to IL-17C stimulation. Consistently, IL-17C induced pro-inflammatory cytokine mRNA expression, such as TNF-α, IL-1β, and IL-6 in the kidney epithelial cells (Figure 6b). Additionally, the expression of these pro-inflammatory cytokines in the kidney epithelial cell supernatants was significantly increased 24 hr after IL-17C stimulation (Figure 6c).
Figure 6.
IL-17C amplifies inflammatory cytokine expression in renal epithelial cells. (a) Representative CD45 cell surface expression is displayed, as determined by flow cytometric analysis from isolated kidney epithelial cells and CD45+ leukocytes (left). IL-17RE mRNA expression real-time RT-PCR analysis from leukocytes and kidney epithelial cells that were isolated from normal and infected WT mouse kidneys at day 3 after infection with 3.5 × 105 CFU of C. albicans (right). (b) Real-time RT-PCR analysis of TNF-α, IL-1β, and IL-6 mRNA expression from kidney epithelial cells treated for 24 hr with IL-17C (100 ng ml−1). (c) ELISA results for TNF-α, IL-1β, and IL-6 in supernatants of kidney epithelial cells that were stimulated with IL-17C (100 ng ml−1) for 24 hr. (d–h) Real-time RT-PCR analysis of IL-17C expression in mouse kidney epithelial cells that were treated for 4 hr with IL-1β (10 ng ml−1), IL-1α (10 ng ml−1), TNF-α (10 ng ml−1), IL-6 (20 ng ml−1), and IL-17A (50 ng ml−1). (i) The fungal infection model induced kidney inflammation that was regulated by IL-17C. The data shown are representative of two independent experiments. **p < 0.01, ***p < 0.001 (t-test).
To further elucidate the regulation of IL-17C expression by kidney epithelial cells during fungal infection, we stimulated isolated kidney epithelial cells with various pro-inflammatory cytokines, including TNF-α, IL-1α, IL-1β, IL-6, and IL-17A. We found that IL-1α and IL-1β but not TNF-α, IL-6, or IL-17A induced IL-17C mRNA expression (Figure 6d–h). Taken together, our data indicate that IL-17C serves as an inflammation amplifier during fungal infection.
Discussion
Systemic candidiasis is a disastrous disease that has a high worldwide mortality rate.26,39 Recently, it was reported that mice with systemic candidiasis die of sepsis, the hallmark of which is tissue damage and failure of multiple organs, especially the kidneys.5,6 Although pro-inflammatory cytokines, including TNF-α and IL-1β, have been identified as a major cause of sepsis,6,7 the regulatory mechanisms for sepsis during systemic candidiasis is not well understood. In this study, we identified for the first time that IL-17C is a detrimental amplifier of inflammation during systemic candidiasis.
One of the pivotal findings in our study is the pathological role of IL-17C in systemic fungal infection. Based on discoveries by our group and other investigators, IL-17C has diverse functions from promoting anti-bacterial immune responses to intestinal pathogens at the epithelial surface to aggravating inflammatory diseases, including psoriasis and EAE.23,24,25 Because our previous study showed that IL-17C can potentiate Th17 cell responses in an EAE model24 and other literature has implicated important roles for Th17 cells in the host defense against C. albicans,13,14 we measured the Th17 cell responses after systemic C. albicans infection and found that Th17 cells were not induced in WT mice in this acute systemic infection model (data not shown). This result indicated that although Il17a−/− mice are indeed more susceptible to systemic fungal infections,14,16 the cellular source of IL-17A could be cell types other than Th17 cells, such as NKT cells, IL-17-secreting innate lymphoid cells, and γδ T cells, through in vitro experiments14 or different fungal infection models, such as the oropharyngeal candidiasis model,13 have suggested a crucial protective role for Th17 cells. Taken together, these findings indicate that IL-17C may not play any role in Th17 cell development in the acute systemic fungal infection model, thus contrasting the EAE model. In our current study, by using Il17c-deficient mice, we found that IL-17C plays a pathological role in systemic fungal infections through promoting pro-inflammatory cytokine production, such as TNF-α, IL-1β, and IL-6, which in turn exacerbates kidney injury and leads to a higher mortality rate. The Th17 cytokine IL-17A, but not IL-22, has been shown to play a critical role in the protective immune responses against C. albicans by regulating the production of G-CSF to promote the maturation of neutrophils, which play a crucial role in the host defense against infectious disease.17,40 Notably, as a member of the IL-17 cytokine family, IL-17C in our study was demonstrated to play a significantly distinct role from IL-17A, which suggested that Il17a-deficient mice are more susceptible to C. albicans infection. Additionally, the cellular sources of IL-17A and IL-17C are completely different. In our IHC data (Supplementary Figure 1b), consistent with previous studies, infiltrating immune cells, including Th17 cells, IL-17A-secreting ILCs,27,28,29 and γδT cells, are the major sources of IL-17A.41,42 Nevertheless, IL-17C was almost exclusively expressed by kidney epithelial cells (Figure 1c). Therefore, our study has identified divergent functions among the different IL-17 cytokine family members during fungal infections. Furthermore, we found increased expression of IL-25, a cytokine that was shown to play critical roles in host immune response against intestinal helminthes,43,44 in fungus-infected kidneys compared with normal kidneys (Supplementary Figure 1c). Although IL-25 could provide partial protection against oral candidiasis by promoting the ILC2 immune response,45 it remains to be determined whether it plays any roles in systemic candidiasis.
Mice with systemic candidiasis died of sepsis with kidney failure,5 a disease that is closely related to excessive pro-inflammatory cytokine expression. The Il17c−/− mice infected with C. albicans exhibited decreased mortality that was associated not with reduced fungal loads but with the reduced pro-inflammatory cytokine production. We thus further assessed whether IL-17C assumed a pathological role by promoting pro-inflammatory cytokine expression. We found that the kidney tissue TNF-α, IL-1β, and IL-6 and serum IL-6 expression levels were dramatically decreased in Il17c−/− mice (Figure 5) with significantly attenuated kidney failure (Figure 2b–d). Notably, the expression levels of other cytokines, including IL-10, IFN-γ, and IL-17A, in the kidneys of WT and Il17c-deficient mice were not significantly different from each other (Supplementary Figure 4). These data firmly identified a specific cytokine expression profile that is regulated by IL-17C. Therefore, defining the regulatory mechanism of sepsis-associated hyper-inflammation that is targeted by IL-17C may provide us with additional understanding regarding the immune pathology of fungal infections.
Furthermore, we considered the target cells of IL-17C during fungal infections. Because both leukocytes and epithelial cells can produce pro-inflammatory cytokines following infection, we measured IL-17RE expression in isolated leukocytes and epithelial cells from normal or infected kidneys and found that IL-17RE, the receptor for IL-17C, was primarily expressed by kidney epithelial cells (Figure 6a). These data indicated that kidney epithelial cells are the primary targets of IL-17C regulation, although leukocytes can also express pro-inflammatory cytokines after Candida infection. Additionally, in kidney epithelial cells that were cultured in vitro, IL-17C treatment could indeed directly enhance pro-inflammatory cytokine production, including TNF-α, IL-1β, and IL-6 (Figure 6b and c). We also detected that IL-17C was produced by kidney epithelial cells in response to the pro-inflammatory cytokines IL-1α and IL-1β, which were triggered by fungal infection (Figure 6d and e). Therefore, our results have established that IL-17C induces pro-inflammatory cytokine expression in kidney epithelial cells. These data are consistent with previous findings that IL-17C targets epithelial cells in immune responses.23,25,46
In summary, our study has demonstrated that IL-17C acts in an autocrine manner to promote pro-inflammatory cytokine expression in a positive regulatory loop to amplify inflammatory responses to a fungal infection that is detrimental to the host (Figure 6i). Dysregulated expression or signaling by IL-17C may contribute to human disorders that are associated with systemic fungal infections, whereas targeting IL-17C may serve as a novel treatment for sepsis that is associated with systemic fungal infection.
Footnotes
Supplementary information of this article can be found on Cellular & Molecular Immunology website: http://www.nature.com/cmi.
Supplementary Information
References
- Brown GD, Denning DW, Gow NA, Levitz SM, Netea MG, White TC. Hidden killers: human fungal infections. Sci Transl Med 2012; 4: 165rv13. [DOI] [PubMed] [Google Scholar]
- Brown GD, Denning DW, Levitz SM. Tackling human fungal infections. Science 2012; 336: 647. [DOI] [PubMed] [Google Scholar]
- Perfect JR. The impact of the host on fungal infections. Am J Med 2012; 125: S39–S51. [DOI] [PubMed] [Google Scholar]
- Romani L. Immunity to fungal infections. Nat Rev Immunol 2011; 11: 275–288. [DOI] [PubMed] [Google Scholar]
- Spellberg B, Ibrahim AS, Edwards JE, Filler SG. Mice with disseminated candidiasis die of progressive sepsis. J Infect Dis 2005; 192: 336–343. [DOI] [PubMed] [Google Scholar]
- Cohen J. The immunopathogenesis of sepsis. Nature 2002; 420: 885–891. [DOI] [PubMed] [Google Scholar]
- Netea M. Proinflammatory cytokines and sepsis syndrome: not enough, or too much of a good thing? Trends Immunol 2003; 24: 254–258. [DOI] [PubMed] [Google Scholar]
- Dong C. Regulation and pro-inflammatory function of interleukin-17 family cytokines. Immunol Rev 2008; 226: 80–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin W, Dong C. IL-17 cytokines in immunity and inflammation. Emerg Microbes Infect 2013; 2: e60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwakura Y, Ishigame H, Saijo S, Nakae S. Functional specialization of interleukin-17 family members. Immunity 2011; 34: 149–162. [DOI] [PubMed] [Google Scholar]
- Kolls JK, Lindén A. Interleukin-17 family members and inflammation. Immunity 2004; 21: 467–476. [DOI] [PubMed] [Google Scholar]
- Park H, Li Z, Yang XO, Chang SH, Nurieva R, Wang Y-H et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat Immunol 2005; 6: 1133–1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conti HR, Shen F, Nayyar N, Stocum E, Sun JN, Lindemann MJ et al. Th17 cells and IL-17 receptor signaling are essential for mucosal host defense against oral candidiasis. J Exp Med 2009; 206: 299–311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saijo S, Ikeda S, Yamabe K, Kakuta S, Ishigame H, Akitsu A et al. Dectin-2 recognition of alpha-mannans and induction of Th17 cell differentiation is essential for host defense against Candida albicans. Immunity 2010; 32: 681–691. [DOI] [PubMed] [Google Scholar]
- Dejima T, Shibata K, Yamada H, Hara H, Iwakura Y, Naito S et al. Protective role of naturally occurring interleukin-17A-producing γδ T cells in the lung at the early stage of systemic candidiasis in mice. Infect Immun 2011; 79: 4503–4510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang W, Na L, Fidel PL, Schwarzenberger P. Requirement of interleukin-17A for systemic anti-Candida albicans host defense in mice. J Infect Dis 2004; 190: 624–631. [DOI] [PubMed] [Google Scholar]
- Kagami S, Rizzo HL, Kurtz SE, Miller LS, Blauvelt A. IL-23 and IL-17A, but not IL-12 and IL-22, are required for optimal skin host defense against Candida albicans. J Immunol 2010; 185: 5453–5462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma CS, Chew GYJ, Simpson N, Priyadarshi A, Wong M, Grimbacher B et al. Deficiency of Th17 cells in hyper IgE syndrome due to mutations in STAT3. J Exp Med 2008; 205: 1551–1557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milner JD, Brenchley JM, Laurence A, Freeman AF, Hill BJ, Elias KM et al. Impaired T(H)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature 2008; 452: 773–776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- d'Ostiani CF, Del Sero G, Bacci A, Montagnoli C, Spreca A, Mencacci A et al. Dendritic cells discriminate between yeasts and hyphae of the fungus Candida albicans: implications for initiation of T helper cell immunity in vitro and in vivo. J Exp Med 2000; 191: 1661–1674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Chen J, Huang A, Stinson J, Heldens S, Foster J et al. Cloning and characterization of IL-17B and IL-17C, two new members of the IL-17 cytokine family. Proc Natl Acad Sci U S A 2000; 97: 773–778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi Y, Fujio K, Shoda H, Okamoto A, Tsuno NH, Takahashi K et al. IL-17B and IL-17C are associated with TNF-α production and contribute to the exacerbation of inflammatory arthritis. J Immunol 2007; 179: 7128–7136. [DOI] [PubMed] [Google Scholar]
- Ramirez-Carrozzi V, Sambandam A, Luis E, Lin Z, Jeet S, Lesch J et al. IL-17C regulates the innate immune function of epithelial cells in an autocrine manner. Nat Immunol 2011; 12: 1159–1166. [DOI] [PubMed] [Google Scholar]
- Chang SH, Reynolds JM, Pappu BP, Chen G, Martinez GJ, Dong C. Interleukin-17C promotes Th17 cell responses and autoimmune disease via interleukin-17 receptor E. Immunity 2011; 35: 611–621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song X, Zhu S, Shi P, Liu Y, Shi Y, Levin SD et al. IL-17RE is the functional receptor for IL-17C and mediates mucosal immunity to infection with intestinal pathogens. Nat Immunol 2011; 12: 1151–1158. [DOI] [PubMed] [Google Scholar]
- Szabo EK, MacCallum DM. The contribution of mouse models to our understanding of systemic candidiasis. FEMS Microbiol Lett 2011; 320: 1–8. [DOI] [PubMed] [Google Scholar]
- Gladiator A, Wangler N, Trautwein-Weidner K, LeibundGut-Landmann S. Cutting edge: IL-17-secreting innate lymphoid cells are essential for host defense against fungal infection. J Immunol 2013; 190: 521–525. [DOI] [PubMed] [Google Scholar]
- Wüthrich M, Deepe GS, Klein B. Adaptive immunity to fungi. Annu Rev Immunol 2012; 30: 115–148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Acosta-Rodriguez EV, Rivino L, Geginat J, Jarrossay D, Gattorno M, Lanzavecchia A et al. Surface phenotype and antigenic specificity of human interleukin 17-producing T helper memory cells. Nat Immunol 2007; 8: 639–646. [DOI] [PubMed] [Google Scholar]
- Ichimura T. Kidney injury molecule-1 (KIM-1), a putative epithelial cell adhesion molecule containing a novel immunoglobulin domain, is up-regulated in renal cells after injury. J Biol Chem 1998; 273: 4135–4142. [DOI] [PubMed] [Google Scholar]
- Iliev ID, Funari VA, Taylor KD, Nguyen Q, Reyes CN, Strom SP et al. Interactions between commensal fungi and the C-type lectin receptor Dectin-1 influence colitis. Science 2012; 336: 1314–1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lionakis MS, Fischer BG, Lim JK, Swamydas M, Wan W, Richard Lee C-C et al. Chemokine receptor Ccr1 drives neutrophil-mediated kidney immunopathology and mortality in invasive candidiasis. PLoS Pathog 2012; 8: e1002865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurst SD, Muchamuel T, Gorman DM, Gilbert JM, Clifford T, Kwan S et al. New IL-17 family members promote Th1 or Th2 responses in the lung: in vivo function of the novel cytokine IL-25. J Immunol 2002; 169: 443–453. [DOI] [PubMed] [Google Scholar]
- De Filippo K, Dudeck A, Hasenberg M, Nye E, van Rooijen N, Hartmann K et al. Mast cell and macrophage chemokines CXCL1/CXCL2 control the early stage of neutrophil recruitment during tissue inflammation. Blood 2013; 121: 4930–4937. [DOI] [PubMed] [Google Scholar]
- McInnes IB, Schett G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat Rev Immunol 2007; 7: 429–442. [DOI] [PubMed] [Google Scholar]
- Biburger M, Tiegs G. α-Galactosylceramide-induced liver injury in mice is mediated by TNF-α but independent of Kupffer cells. J Immunol 2005; 175: 1540–1550. [DOI] [PubMed] [Google Scholar]
- Dinarello CA. Proinflammatory and anti-inflammatory cytokines as mediators in the pathogenesis of septic shock. CHEST J 1997; 112: 321S–329S. [DOI] [PubMed] [Google Scholar]
- Imai Y, Kuba K, Neely GG, Yaghubian-Malhami R, Perkmann T, van Loo G et al. Identification of oxidative stress and Toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell 2008; 133: 235–249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shoham S, Levitz SM. The immune response to fungal infections. Br J Haematol 2005; 129: 569–582. [DOI] [PubMed] [Google Scholar]
- Ley K, Smith E, Stark MA. IL-17A-producing neutrophil-regulatory Tn lymphocytes. Immunol Res 2006; 34: 229–242. [DOI] [PubMed] [Google Scholar]
- Roark CL, French JD, Taylor MA, Bendele AM, Born WK, O'Brien RL. Exacerbation of collagen-induced arthritis by oligoclonal, IL-17-producing γδ T cells. J Immunol 2007; 179: 5576–5583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamada S, Umemura M, Shiono T, Tanaka K, Yahagi A, Begum MD et al. IL-17A produced by γδ T cells plays a critical role in innate immunity against Listeria monocytogenes infection in the liver. J Immunol 2008; 181: 3456–3463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fallon PG, Ballantyne SJ, Mangan NE, Barlow JL, Dasvarma A, Hewett DR et al. Identification of an interleukin (IL)-25-dependent cell population that provides IL-4, IL-5, and IL-13 at the onset of helminth expulsion. J Exp Med 2006; 203: 1105–1116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao A, Urban JF, Sun R, Stiltz J, Morimoto M, Notari L et al. Critical role of IL-25 in nematode infection-induced alterations in intestinal function. J Immunol 2010; 185: 6921–6929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Y, Guo L, Qiu J, Chen X, Hu-Li J, Siebenlist U et al. IL-25-responsive, lineage-negative KLRG1 hi cells are multipotential ‘inflammatory' type 2 innate lymphoid cells. Nat Immunol 2014; 16: 161–170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reynolds JM, Martinez GJ, Nallaparaju KC, Chang SH, Wang Y-H, Dong C. Cutting edge: regulation of intestinal inflammation and barrier function by IL-17C. J Immunol 2012; 189: 4226–4230. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.