Abstract
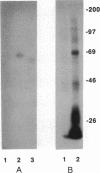
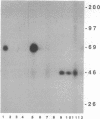
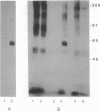
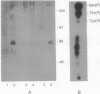
When T cells are activated by the T-cell antigen receptor, a number of cellular proteins are phosphorylated on tyrosine. We investigated whether any of these proteins were present on the surface of activated T cells. Using the human leukemic T-cell line Jurkat and normal peripheral blood lymphocytes, we identified a 67-kDa cell surface glycoprotein in anti-phosphotyrosine immunoprecipitates, after treatment of the cells with CD3 antibody. When cell lysates were depleted of CD5 by sequential immunoprecipitation, the 67-kDa phosphotyrosyl polypeptide was no longer precipitated by the phosphotyrosine antibody. Western blot analysis of anti-phosphotyrosine precipitates confirmed that this glycoprotein was CD5. It was possible that CD5 was present in the anti-phosphotyrosine immunoprecipitates due to its physical association with phosphotyrosyl proteins rather than being directly tyrosine-phosphorylated itself. However, Western blot analysis of anti-CD5 immunoprecipitates with phosphotyrosine antibody and phosphoamino acid analysis demonstrated that CD5 was indeed phosphorylated on tyrosine after stimulation of the cells with CD3 antibody and was concomitantly phosphorylated on serine and threonine. Tyrosine phosphorylation of CD5 was maximal 2 min after CD3 stimulation and returned to baseline levels by 60 min. CD5 is expressed on the cell surface of all mature T cells and a small proportion of B lymphocytes and has recently been identified as the ligand for CD72, a receptor present on the surface of all B cells. The present data suggest that tyrosine phosphorylation may be involved in B-cell-T-cell communication.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chalupny N. J., Ledbetter J. A., Kavathas P. Association of CD8 with p56lck is required for early T cell signalling events. EMBO J. 1991 May;10(5):1201–1207. doi: 10.1002/j.1460-2075.1991.tb08061.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan A. C., Irving B. A., Fraser J. D., Weiss A. The zeta chain is associated with a tyrosine kinase and upon T-cell antigen receptor stimulation associates with ZAP-70, a 70-kDa tyrosine phosphoprotein. Proc Natl Acad Sci U S A. 1991 Oct 15;88(20):9166–9170. doi: 10.1073/pnas.88.20.9166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatila T. A., Geha R. S. Phosphorylation of T cell membrane proteins by activators of protein kinase C. J Immunol. 1988 Jun 15;140(12):4308–4314. [PubMed] [Google Scholar]
- Cooke M. P., Abraham K. M., Forbush K. A., Perlmutter R. M. Regulation of T cell receptor signaling by a src family protein-tyrosine kinase (p59fyn). Cell. 1991 Apr 19;65(2):281–291. doi: 10.1016/0092-8674(91)90162-r. [DOI] [PubMed] [Google Scholar]
- Crabtree G. R. Contingent genetic regulatory events in T lymphocyte activation. Science. 1989 Jan 20;243(4889):355–361. doi: 10.1126/science.2783497. [DOI] [PubMed] [Google Scholar]
- DeFranco A. L. Between B cells and T cells. Nature. 1991 Jun 20;351(6328):603–604. doi: 10.1038/351603a0. [DOI] [PubMed] [Google Scholar]
- Glaichenhaus N., Shastri N., Littman D. R., Turner J. M. Requirement for association of p56lck with CD4 in antigen-specific signal transduction in T cells. Cell. 1991 Feb 8;64(3):511–520. doi: 10.1016/0092-8674(91)90235-q. [DOI] [PubMed] [Google Scholar]
- Griffith L. C., Schulman H., Tagawa M. In vivo and in vitro phosphorylation of murine lymphocyte differentiation antigen CD5. Biochem Biophys Res Commun. 1989 Mar 15;159(2):536–541. doi: 10.1016/0006-291x(89)90026-0. [DOI] [PubMed] [Google Scholar]
- Hayakawa K., Hardy R. R. Normal, autoimmune, and malignant CD5+ B cells: the Ly-1 B lineage? Annu Rev Immunol. 1988;6:197–218. doi: 10.1146/annurev.iy.06.040188.001213. [DOI] [PubMed] [Google Scholar]
- Horvath A. R., Elmore M. A., Kellie S. Differential tyrosine-specific phosphorylation of integrin in Rous sarcoma virus transformed cells with differing transformed phenotypes. Oncogene. 1990 Sep;5(9):1349–1357. [PubMed] [Google Scholar]
- Hsi E. D., Siegel J. N., Minami Y., Luong E. T., Klausner R. D., Samelson L. E. T cell activation induces rapid tyrosine phosphorylation of a limited number of cellular substrates. J Biol Chem. 1989 Jun 25;264(18):10836–10842. [PubMed] [Google Scholar]
- Huang H. J., Jones N. H., Strominger J. L., Herzenberg L. A. Molecular cloning of Ly-1, a membrane glycoprotein of mouse T lymphocytes and a subset of B cells: molecular homology to its human counterpart Leu-1/T1 (CD5). Proc Natl Acad Sci U S A. 1987 Jan;84(1):204–208. doi: 10.1073/pnas.84.1.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imboden J. B., June C. H., McCutcheon M. A., Ledbetter J. A. Stimulation of CD5 enhances signal transduction by the T cell antigen receptor. J Clin Invest. 1990 Jan;85(1):130–134. doi: 10.1172/JCI114402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imboden J. B., Stobo J. D. Transmembrane signalling by the T cell antigen receptor. Perturbation of the T3-antigen receptor complex generates inositol phosphates and releases calcium ions from intracellular stores. J Exp Med. 1985 Mar 1;161(3):446–456. doi: 10.1084/jem.161.3.446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones N. H., Clabby M. L., Dialynas D. P., Huang H. J., Herzenberg L. A., Strominger J. L. Isolation of complementary DNA clones encoding the human lymphocyte glycoprotein T1/Leu-1. 1986 Sep 25-Oct 1Nature. 323(6086):346–349. doi: 10.1038/323346a0. [DOI] [PubMed] [Google Scholar]
- June C. H., Fletcher M. C., Ledbetter J. A., Samelson L. E. Increases in tyrosine phosphorylation are detectable before phospholipase C activation after T cell receptor stimulation. J Immunol. 1990 Mar 1;144(5):1591–1599. [PubMed] [Google Scholar]
- June C. H., Fletcher M. C., Ledbetter J. A., Schieven G. L., Siegel J. N., Phillips A. F., Samelson L. E. Inhibition of tyrosine phosphorylation prevents T-cell receptor-mediated signal transduction. Proc Natl Acad Sci U S A. 1990 Oct;87(19):7722–7726. doi: 10.1073/pnas.87.19.7722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- June C. H., Rabinovitch P. S., Ledbetter J. A. CD5 antibodies increase intracellular ionized calcium concentration in T cells. J Immunol. 1987 May 1;138(9):2782–2792. [PubMed] [Google Scholar]
- Kemp B. E., Pearson R. B. Protein kinase recognition sequence motifs. Trends Biochem Sci. 1990 Sep;15(9):342–346. doi: 10.1016/0968-0004(90)90073-k. [DOI] [PubMed] [Google Scholar]
- Kiener P. A., Mittler R. S. CD45-protein tyrosine phosphatase cross-linking inhibits T cell receptor CD3-mediated activation in human T cells. J Immunol. 1989 Jul 1;143(1):23–28. [PubMed] [Google Scholar]
- Koretzky G. A., Picus J., Schultz T., Weiss A. Tyrosine phosphatase CD45 is required for T-cell antigen receptor and CD2-mediated activation of a protein tyrosine kinase and interleukin 2 production. Proc Natl Acad Sci U S A. 1991 Mar 15;88(6):2037–2041. doi: 10.1073/pnas.88.6.2037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Ledbetter J. A., June C. H., Martin P. J., Spooner C. E., Hansen J. A., Meier K. E. Valency of CD3 binding and internalization of the CD3 cell-surface complex control T cell responses to second signals: distinction between effects on protein kinase C, cytoplasmic free calcium, and proliferation. J Immunol. 1986 Jun 1;136(11):3945–3952. [PubMed] [Google Scholar]
- Ledbetter J. A., Schieven G. L., Uckun F. M., Imboden J. B. CD45 cross-linking regulates phospholipase C activation and tyrosine phosphorylation of specific substrates in CD3/Ti-stimulated T cells. J Immunol. 1991 Mar 1;146(5):1577–1583. [PubMed] [Google Scholar]
- Ledbetter J. A., Tonks N. K., Fischer E. H., Clark E. A. CD45 regulates signal transduction and lymphocyte activation by specific association with receptor molecules on T or B cells. Proc Natl Acad Sci U S A. 1988 Nov;85(22):8628–8632. doi: 10.1073/pnas.85.22.8628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ley S. C., Davies A. A., Druker B., Crumpton M. J. The T cell receptor/CD3 complex and CD2 stimulate the tyrosine phosphorylation of indistinguishable patterns of polypeptides in the human T leukemic cell line Jurkat. Eur J Immunol. 1991 Sep;21(9):2203–2209. doi: 10.1002/eji.1830210931. [DOI] [PubMed] [Google Scholar]
- Lozano F., Alberola-Ila J., Places L., Gallart T., Vives J. Phosphorylation-mediated changes in the electrophoretic mobility of CD5 molecules. Eur J Biochem. 1990 Oct 24;193(2):469–477. doi: 10.1111/j.1432-1033.1990.tb19361.x. [DOI] [PubMed] [Google Scholar]
- McAteer M. J., Lagarde A. C., Georgiou H. M., Bellgrau D. A requirement for the CD5 antigen in T cell activation. Eur J Immunol. 1988 Jul;18(7):1111–1117. doi: 10.1002/eji.1830180721. [DOI] [PubMed] [Google Scholar]
- Park D. J., Rho H. W., Rhee S. G. CD3 stimulation causes phosphorylation of phospholipase C-gamma 1 on serine and tyrosine residues in a human T-cell line. Proc Natl Acad Sci U S A. 1991 Jun 15;88(12):5453–5456. doi: 10.1073/pnas.88.12.5453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel M. D., Samelson L. E., Klausner R. D. Multiple kinases and signal transduction. Phosphorylation of the T cell antigen receptor complex. J Biol Chem. 1987 Apr 25;262(12):5831–5838. [PubMed] [Google Scholar]
- Pingel J. T., Thomas M. L. Evidence that the leukocyte-common antigen is required for antigen-induced T lymphocyte proliferation. Cell. 1989 Sep 22;58(6):1055–1065. doi: 10.1016/0092-8674(89)90504-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudd C. E. CD4, CD8 and the TCR-CD3 complex: a novel class of protein-tyrosine kinase receptor. Immunol Today. 1990 Nov;11(11):400–406. doi: 10.1016/0167-5699(90)90159-7. [DOI] [PubMed] [Google Scholar]
- Samelson L. E., Phillips A. F., Luong E. T., Klausner R. D. Association of the fyn protein-tyrosine kinase with the T-cell antigen receptor. Proc Natl Acad Sci U S A. 1990 Jun;87(11):4358–4362. doi: 10.1073/pnas.87.11.4358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snow E. C., Mond J. J., Subbarao B. Enhancement by monoclonal anti-Lyb-2 antibody of antigen-specific B lymphocyte expansion stimulated by TNP-Ficoll and T lymphocyte-derived factors. J Immunol. 1986 Sep 15;137(6):1793–1796. [PubMed] [Google Scholar]
- Spertini F., Stohl W., Ramesh N., Moody C., Geha R. S. Induction of human T cell proliferation by a monoclonal antibody to CD5. J Immunol. 1991 Jan 1;146(1):47–52. [PubMed] [Google Scholar]
- Subbarao B., Mosier D. E. Induction of B lymphocyte proliferation by monoclonal anti-Lyb 2 antibody. J Immunol. 1983 May;130(5):2033–2037. [PubMed] [Google Scholar]
- Thomas Y., Glickman E., DeMartino J., Wang J., Goldstein G., Chess L. Biologic functions of the OKT1 T cell surface antigen. I. The T1 molecule is involved in helper function. J Immunol. 1984 Aug;133(2):724–728. [PubMed] [Google Scholar]
- Trevillyan J. M., Lu Y. L., Atluru D., Phillips C. A., Bjorndahl J. M. Differential inhibition of T cell receptor signal transduction and early activation events by a selective inhibitor of protein-tyrosine kinase. J Immunol. 1990 Nov 15;145(10):3223–3230. [PubMed] [Google Scholar]
- Van de Velde H., von Hoegen I., Luo W., Parnes J. R., Thielemans K. The B-cell surface protein CD72/Lyb-2 is the ligand for CD5. Nature. 1991 Jun 20;351(6328):662–665. doi: 10.1038/351662a0. [DOI] [PubMed] [Google Scholar]
- Wallace D. L., Beverley P. C. Phenotypic changes associated with activation of CD45RA+ and CD45RO+ T cells. Immunology. 1990 Mar;69(3):460–467. [PMC free article] [PubMed] [Google Scholar]
- Wang C. Y., Good R. A., Ammirati P., Dymbort G., Evans R. L. Identification of a p69,71 complex expressed on human T cells sharing determinants with B-type chronic lymphatic leukemic cells. J Exp Med. 1980 Jun 1;151(6):1539–1544. doi: 10.1084/jem.151.6.1539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss A., Imboden J., Hardy K., Manger B., Terhorst C., Stobo J. The role of the T3/antigen receptor complex in T-cell activation. Annu Rev Immunol. 1986;4:593–619. doi: 10.1146/annurev.iy.04.040186.003113. [DOI] [PubMed] [Google Scholar]
- Weiss A., Koretzky G., Schatzman R. C., Kadlecek T. Functional activation of the T-cell antigen receptor induces tyrosine phosphorylation of phospholipase C-gamma 1. Proc Natl Acad Sci U S A. 1991 Jul 1;88(13):5484–5488. doi: 10.1073/pnas.88.13.5484. [DOI] [PMC free article] [PubMed] [Google Scholar]