Abstract
Purpose
To compare the efficacy and safety of current treatments in diabetic macular edema (DME).
Methods
PubMed, Embase and CENTRAL were systematically reviewed for randomized controlled trials of current treatments in DME through August 2015. Data on the mean change of best-corrected visual acuity (BCVA) and central macular thickness (CMT) were extracted, and adverse events (AEs) were collected.
Results
A total of 21 trials were included in our network meta-analysis. Intravitreal ranibizumab improved BCVA most significantly (OR: +7.01 95%CI (2.56 to 11.39)) in 6 months and intravitreal aflibercept (+8.19 (5.07 to 11.96)) in 12 months. Intravitreal triamcinolone combined with LASER decreased CMT most significantly (-111.34 (-254.61 to 37.93)) in 6 months and intravitreal aflibercept (-110.83 (-190.25 to -35.27)) in 12 months. Compared with the relatively high rate of ocular AEs in the groups with administration of steroids, systematic AEs occurred more frequently in the groups with vascular endothelial growth factor inhibitors involved.
Conclusions
Our analysis confirms that intravitreal aflibercept is most favorable with both BCVA improvement and CMT decrease than other current therapies in the management of DME within 12 months. Vascular endothelial growth factor inhibitors for DME should be used with caution due to systematic AEs. Combined intravitreal triamcinolone with LASER has a stronger efficacy in decreasing CMT than the other interventions in the early stage after injection. More high-quality randomized controlled trials will be necessary.
Introduction
Diabetic macular edema (DME) is a common manifestation of diabetic retinopathy and a leading cause of serious central visual loss and impairment in diabetic patients [1]. The prevalence rate of DME has been reported to be 29% in patients with a duration of more than 20 years [2]. The chance of spontaneous improvement in the best-corrected visual acuity (BCVA) and decrease in the central macular thickness (CMT) is limited, so the prognosis of DME is generally disappointing.
Since the Early Treatment Diabetic Retinopathy Study (ETDRS) found that laser resulted in a 50% reduction in severe vision loss in 1985, laser has been the gold standard treatment for macular edema [3]. Recently, it was reported that the level of inflammatory factors could be significantly elevated in the vitreous fluid of patients with DME, and vascular endothelial growth factors (VEGFs) had a stronger influence on retinal vascular permeability and severity of DME than the other factors [4–7]. To avoid the ocular side effects related to laser treatment like subretinal fibrosis and laser scars [8], the anti-inflammatory drugs, like steroids and VEGF inhibitors, were used for DME by intravitreal injection.
Steroids are the common, anti-inflammatory, anti-angiogenic, and blood-retinal barriers, which can stabilize medications in the treatment of DME. High rates of intraocular pressure (IOP) increase and cataract progress were found in steroids-treated eyes. In recent years, the introduction of VEGF inhibitors has revolutionized the treatment of DME. Bevacizumab, ranibizumab, pegaptanib and alibercept have been well established via phase II/III clinical trials showing significantly improved vision in many patients with DME [9–13]. Bevacizumab, a humanized full-length monoclonal antibody, inhibits all isoforms of VEGFs. It has been popular because it is more affordable than other anti-VEGF drugs in the treatment of retinal vascular diseases [14]. As a monoclonal antibody, ranibizumab blocks all isoforms of VEGF-A and enhances their affinity. The FDA has approved it for the treatment of DME [15]. Pegaptanib can inhibit VEGF-165 responding for ocular neovascularization and vascular permeability [16], and the FDA has approved it for neovascular age-related macular degeneration [17]. Aflibercept is a soluble protein and blocks all isoforms of VEGFs. Its half-life is prolonged, and its affinity of binding to VEGF-A is more than 100-fold greater than bevacizumab or ranibizumab. Anti-VEGF agents must be given frequently, and this may be associated with a small increased risk of systematic adverse effects like hypertension.
Some researchers performed systematic reviews and standard meta-analyses on therapies of DME [18–20]. However, standard meta-analysis is unable to include all direct and indirect comparisons among steroids, VEGF inhibitors, and LASER which included laser, macular laser, grid laser and focal/grid laser in one analysis, while network meta-analysis can conduct a more accurate ranking and precision for the current therapeutic strategies [21–23]. Therefore, we performed a network meta-analysis to estimate the efficacy and safety of current treatments in DME.
Methods
Search Strategy
We searched and identified the relevant trials from PubMed, Embase and CENTRAL through August 2015 with no language limit. The medical subject heading and keywords used for the search included diabetic macular edema, laser, steroids, vascular endothelial growth factor, bevacizumab, ranibizumab, pegaptanib and aflibercept. We also screened the reference lists of published meta-analyses of DME treatment. BCVA, CMT, and ocular and systematic adverse events (AEs) were the primary endpoints of this meta-analysis.
Eligibility of Studies
All included studies met the following criteria: (a) randomized controlled trials (RCTs); (b) participants with any type of diabetes; (c) follow-up of more than six months; (d) efficacy outcomes including BCVA and CMT; (e) only the articles containing complete data were included, and duplicated publications or reports of one trial were included once.
Investigators extracted data independently and made the final selection based on resolved discrepancies by discussion. A total of 21 trials were included in our network meta-analysis (Table 1).
Table 1. Study characteristics of trials included in the network meta-analysis.
| Study ID | Diabetes Mellitus | Treatment | Follow-up | Total (eyes) | BCVA-6m | BCVA-12m | CMT-6m | CMT-12m | Jadad | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Duration (years) | Type 2 (%) | Intervention | Dosage | Number | ||||||||
| Wells et al. (2015) | 15±3.32 | 88% | IVA | 2.0mg | 9–10 | 12 months | N = 208 | NA | +13.3 ± 11.1 | NA | -169 ± 138 | 4 |
| 17±3.32 | 94% | IVB | 1.25mg | 9–11 | N = 206 | NA | +9.7 ± 10.1 | NA | -101 ± 121 | |||
| 16±3.06 | 90% | IVR | 0.3mg | 9–11 | N = 206 | NA | +11.2 ± 9.4 | NA | -147 ± 134 | |||
| Ishibashi et al. (2015) | 11.21±8.20 | 99.2% | IVR | 0.5mg | 7.8±2.94 | 12 months | N = 133 | NA | +6.6 ± 7.68 | NA | -134.6 ± 131.17 | 7 |
| 11.33±8.05 | 98.5% | IVR+LASER | 0.5mg | 7.0±3.07 | N = 132 | NA | +6.4 ± 10.67 | NA | -171.8 ± 160.85 | |||
| 1.5±0.85 | ||||||||||||
| 11.34±8.85 | 98.5% | LASER | NA | 1.9±1.02 | N = 131 | NA | +1.8 ± 8.27 | NA | -57.2 ± 118.60 | |||
| Berger et al. (2015) | 16.5±9.0 | 88% | IVR | 0.5mg | 9.2±2.8 | 12 months | N = 75 | +7.1 ± 7.83 | +8.9 ± 7.83 | –129.3 ± 118.69 | –143.5 ± 148.25 | 5 |
| 18.5±11.6 | 79.5% | IVR+LASER | 0.5mg | 8.8±2.9 | N = 73 | +5.6 ± 8.58 | +8.2 ± 9.44 | –114.2 ± 113.29 | –152.2 ± 142.47 | |||
| 1.6±1.0 | ||||||||||||
| 16.6±10.7 | 87.5% | LASER | NA | 2.6±2.1 | N = 72 | +0.9 ± 7.68 | +0.3 ± 13.64 | –64.4 ± 117.26 | –107.1 ± 157.34 | |||
| Gillies et al. (2014) | 16.7±10.7 | NA | IVB | 1.25mg | 8.6 | 12 months | N = 42 | NA | +8.9 ± 8.8 | NA | NA | 5 |
| 16.7±10.3 | NA | DDSI | 0.7mg | 2.7 | N = 46 | NA | +5.6 ± 16.3 | NA | NA | |||
| Comyn et al. (2014) | 18.5±4.09 | 81.8% | IVR | 0.5mg | 9.0 | 48 weeks | N = 22 | NA | NA | NA | -131.5 ± 98.0 | 5 |
| 18±2.81 | 100% | LASER | NA | 2.6 | N = 11 | NA | NA | NA | -102.9 ± 88.4 | |||
| Shoeibi et al. (2013) | NA | NA | IVB | 1.25mg | 1 (1–2) | 13.3 ± 3.4 months | N = 41 | NA | +14±16 | NA | -91 ± 224.9 | 7 |
| NA | NA | IVB+IVT | 1.25mg | 1 (1–2) | N = 37 | NA | +9.5±16.5 | NA | -57 ± 225 | |||
| 2mg | 1 | |||||||||||
| Nepomuceno et al. (2013) | 15.9±8.0 | NA | IVR | 0.5mg | 7.67 | 48 weeks | N = 28 | NA | +14.5±2 | NA | -136 ± 23 | 7 |
| 16.2±8.0 | NA | IVB | 1.5mg | 9.84 | N = 32 | NA | +11.5±1 | NA | -126 ± 25 | |||
| Callanan et al. (2013) | NA | NA | DDSI+LASER | 0.7mg | 2 | 12 months | N = 126 | NA | +2.9 ± 11.45 | NA | -102.8 ± 130.86 | 7 |
| ≥1 | ||||||||||||
| NA | NA | LASER | NA | 2.5 | N = 127 | NA | +2.1 ± 12.05 | NA | -125.3 ± 123.38 | |||
| Arevalo et al. (2013) | NA | NA | IVB | 1.25 or 2.5mg | 5.8±3.2 | 24 months | N = 141 | +8.16 ± 17.37 | +11.13 ± 17.65 | NA | NA | 5 |
| NA | NA | LASER | NA | 2.2±1.4 | N = 120 | +4.1 ± 11.3 | +4.41 ± 11.7 | NA | NA | |||
| NA | NA | IVB+LASER | 1.25 or 2.5mg | 6.2±4.9 | N = 157 | +4.9 ± 16.37 | +7.44 ± 16.73 | NA | NA | |||
| Berger et al. (2013) | 17±10.1 | 86% | IVR+LASER | 0.5mg | Monthly | 12 months | N = 78 | NA | +8.0 ± 9.1 | NA | -145.2 ± 143.3 | 5 |
| IVR | 0.5mg | Monthly | N = 81 | NA | +8.7 ± 7.9 | NA | -134.7 ± 142.7 | |||||
| LASER | N = 82 | NA | +0.8 ± 12.3 | NA | -103.6 ± 141.7 | |||||||
| Soheilian et al. (2012) | 10.5±3.2 | NA | IVB | 1.25mg | 3.1±1.6 | 24 months | N = 50 | +10.5±10 | +10.5±13.5 | −36 ± 119 | −40 ± 133 | 7 |
| 10.4±2.6 | NA | IVB+IVT | 1.25mg | 2.6±1.5 | N = 50 | +5±14 | +5±13.5 | −25 ± 108 | −10 ± 145 | |||
| 2mg | ||||||||||||
| 10.5±2.9 | NA | LASER | NA | 1.0±0.1 | N = 50 | -1±16.5 | +1±17 | −11 ± 78 | 6 ± 86 | |||
| Synek et al. (2011) | NA | NA | IVB | 1.25mg | 3 | 24 weeks | N = 30 | +10±10 | NA | –94 ± 170 | NA | 7 |
| NA | NA | IVB+IVT | 1.25mg | 3 | N = 30 | +10±10 | NA | –93 ± 124 | NA | |||
| 2mg | 1 | |||||||||||
| DRCR et al. (2010) | 16±3.32 | 89% | LASER | NA | NA | 3 years; | N = 293 | NA | +3 ± 13 | NA | −102 ± 151 | 7 |
| 18±3.06 | 92% | IVR+LASER | 0.5mg | 2 (0–4) | N = 187 | NA | +9 ± 11 | NA | −131 ± 129 | |||
| 17±3.32 | 89% | IVT+LASER | 4mg | 1 (0–2) | N = 186 | NA | +4 ± 13 | NA | −127 ± 140 | |||
| Mitchell et al. (2011) | 15.23±9.91 | 88.8% | IVR | 0.5mg | 7.0±2.81 | 12 months | N = 116 | NA | +6.1 ± 6.43 | NA | −118.7 ± 115.07 | 6 |
| 14.62±9.84 | 86.4% | IVR+LASER | 0.5mg | 6.8±2.95 | N = 118 | NA | +5.9 ± 7.92 | NA | −128.3 ± 114.34 | |||
| 12.93±9.02 | 87.4% | LASER | NA | ≥3 months | N = 111 | NA | +0.8 ± 8.56 | NA | −61.3 ± 132.29 | |||
| Nguyen et al. (2010) | NA | NA | IVR | 0.5mg | 4 | 2 years | N = 42 | +7.24 ± 4.46 | +6.61 ± 5.58 | NA | NA | 5 |
| NA | NA | LASER | NA | NA | N = 42 | −0.43 ± 4.45 | +2.39 ± 4.0 | NA | NA | |||
| NA | NA | IVR+LASER | 0.5mg | 2 | N = 42 | +3.8 ± 4.04 | +4.81 ± 5.16 | NA | NA | |||
| Michaelides et al. (2010) | NA | 89.5% | LASER | NA | 3 | 12 months | N = 38 | NA | NA | NA | -68 ± 171 | 5 |
| NA | 90.5% | IVB | 1,25mg | 9 | N = 42 | NA | NA | NA | -130 ± 122 | |||
| Massin et al. (2010) | 14.2±4.44 | 97.1% | IVR | 0.3 or 0.5mg | 10.2±2.5 | 12 months | N = 102 | NA | +10.3 ± 9.1 | NA | -194.2 ± 135.1 | 7 |
| 15.1±6.63 | 98% | Placebo | NA | NA | N = 49 | NA | -1.4 ± 14.2 | NA | -48.4 ± 153.4 | |||
| Ahmadieh et al. (2008) | NA | NA | IVB | 1.25mg | 3 | 24 weeks | N = 41 | +9 ± 16.64 | NA | −95.7 ± 172.5 | NA | 6 |
| NA | NA | IVB+IVT | 1.25mg | 3 | N = 37 | +10.5 ± 13.50 | NA | −92.1 ± 125.3 | NA | |||
| 2mg | 1 | |||||||||||
| NA | NA | Placebo | NA | NA | N = 37 | +1.5 ± 16.50 | NA | 34.9 ± 63.9 | NA | |||
| Pappas et al. (2008) | NA | NA | IVT+LASER | 4mg | NA | 6 months | N = 35 | NA | NA | -176.14 ± 46 | NA | 5 |
| NA | NA | IVB | 1.25mg | NA | N = 27 | NA | NA | -87.38 ± 21 | NA | |||
| Lam et al. (2007) | NA | NA | IVT | 4mg | 1 | 6 months | N = 38 | -0.7 ± 10.7 | NA | NA | NA | 5 |
| NA | NA | IVT+LASER | 4mg | 1 | N = 36 | -1.1 ± 10.8 | NA | NA | NA | |||
| 1 | ||||||||||||
| NA | NA | LASER | NA | 1 | N = 37 | -1.6 ± 11.5 | NA | NA | NA | |||
| Audren et al. (2006) | 14.4±8.9 | NA | IVT | 4mg | 1 | 6 months | N = 17 | +6.9 ± 10.7 | NA | NA | NA | 5 |
| Placebo | NA | NA | N = 17 | -2.6 ± 7.8 | NA | NA | NA | |||||
BCVA, mean change in best corrected visual acuity; CMT, mean change in central macular thickness; IVA, intravitreal aflibercept; IVB, intravitreal bevacizumab; IVR, intravitreal ranibizumab; LASER, laser, macular laser, grid laser and focal/grid laser; DDSI, dexamethasone implant; IVT, intravitreal triamcinolone; NA, No available.
Data Abstraction and Quality Assessment
We assessed the risk of bias in each study using the Cochrane Collaboration’s tool and evaluated the risk of bias categories based on the following items: (1) sequence generation, (2) allocation concealment, (3) blinding of participants and personnel, (4) incomplete outcomes data, (5) selective reporting, and (6) other bias. We also reassessed the bias using the modified Jadad scale. Randomization, concealment of allocation, double blinding, and withdrawals and dropouts were evaluated with a total of 7 points.
Characteristics of studies, such as author, year of publication, duration and type of diabetes, dosage and number of each intervention, follow-up time, and number of eyes in each intervention were recorded exactly. The details of the efficacy outcome, including the mean change of BCVA and CMT in 6 months and 12 months from the baseline, were captured respectively. When extracting the BCVA data, we converted the logarithm of the minimal angle of resolution (log MAR) into ETDRS letters form.
Statistical Analysis
We performed the multi-treatment meta-analysis within a Bayesian framework by using the Markov Chain Monte Carlo simulation [24]. All data were analyzed by using the Aggregate Data Drug Information System (ADDIS) v1.16.5 (Drugis, Groningen, NL). Forest plots were made in R software (version 3.2.3) with the R2winBUGS package. Statistical heterogeneity was assessed by the I2 using the Higgins–Thompson method [25]: < 25% was no heterogeneity, 25–50% was low heterogeneity, 50–75% was moderate heterogeneity, and > 75% was high heterogeneity.
Node-splitting [26] and pair-wise meta-analyses were used to evaluate the inconsistency of direct comparisons in indirect evidences in the network meta-analysis. The direct and indirect evidences in accordance in the split node were analyzed in a node-splitting assessment. P < 0.05 was considered as significant heterogeneity. The efficacy of the intervention was assessed by the odds ratio (OR) with 95% credibility interval (CI). If 1.0 was not included in 95% CI, the results were considered statistically significant. We calculated the rank’s possibility and ranked the outcome of different inventions according to the estimated effect size.
Results
Literature Search
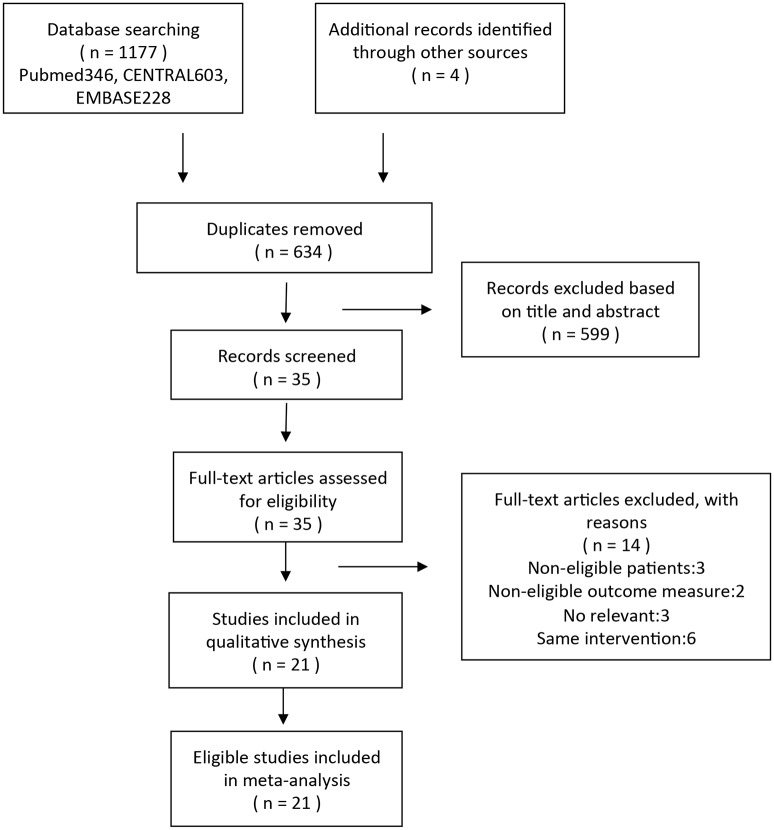
We identified 1181 articles (346 in PubMed, 603 in Embase, 228 in CENTRAL, and 4 additional records through other sources) in the initial search of all databases. These articles were restricted to human subjects and RCTs before August 2015. Except 547 studies that were retrieved for duplicates, there were 634 studies. Based on the titles and abstracts, 599 of them were removed. Of the remained 35 studies, 14 articles were excluded, because the patients in 3 trials and the outcome measures in 2 trials did not meet the inclusion criteria, the interventions in 6 trials were same, and the study types in 3 trials were not relevant with our meta-analysis. A total of 21 trials were identified and were eligible for this net meta-analysis (Fig 1)[27–47].
Fig 1. Flow chart indicating the selection process for this network meta-analysis.
Study Characteristics
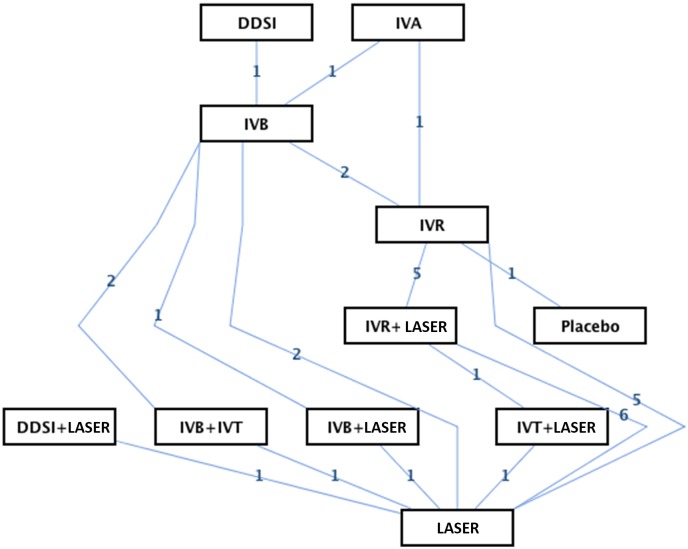
The eligible 21 trials covered 11 different interventions in the treatment of DME and resulted in 16 theoretical comparisons for each of the primary outcomes. We conducted a network of eligible comparisons for the multiple-treatment meta-analysis (Fig 2) and compared the primary endpoints of BCVA and CMT (Tables 2 and 3).
Fig 2. Network of eligible comparisons for the meta-analysis.
Table 2. Network meta-analysis results in BCVA (upper part) and CMT (lower part) at 6 months.
| IVB | 1.30 (-3.09, 5.54) | 4.31 (-2.39, 10.51) | -1.26 (-7.43, 5.43) | 1.30 (-5.08, 8.05) | 3.41 (-4.21, 10.49) | 4.48 (-4.36, 12.59) | 5.69 (1.45, 10.70) | 10.88 (4.08, 17.58) |
| -6.47 (-72.54, 62.87) | IVB+IVT | 2.82 (-4.42, 10.10) | -2.73 (-9.34, 4.83) | -0.16 (-6.73, 7.36) | 1.93 (-5.70, 9.57) | 3.26 (-6.05, 11.94) | 4.31 (-0.81, 10.40) | 9.62 (2.69, 16.67) |
| - | - | IVBLASER | -5.61 (-12.55, 2.31) | -3.02 (-10.08, 4.93) | -0.86 (-9.55, 7.55) | 0.15 (-9.70, 9.59) | 1.39 (-4.33, 8.07) | 6.74 (-2.12, 15.42) |
| 42.28 (-106.95, 193.39) | 49.32 (-105.51, 197.57) | - | IVR | 2.65 (-1.98, 6.92) | 4.54 (-3.29, 11.91) | 5.89 (-3.46, 13.93) | 7.01 (2.56, 11.39) | 12.23 (3.57, 20.22) |
| 28.80 (-120.93, 176.05) | 34.23 (-115.90, 183.08) | - | -14.67 (-123.21, 92.73) | IVR+LASER | 1.91 (-5.97, 9.49) | 3.27 (-5.99, 11.58) | 4.41 (0.04, 8.89) | 9.60 (0.82, 17.75) |
| - | - | - | - | - | IVT | 1.20 (-5.98, 8.22) | 2.49 (-3.61, 9.08) | 7.57 (0.60, 14.05) |
| 87.93 (-15.39, 197.99) | 95.31 (-30.93, 225.58) | - | 46.60 (-141.00, 226.56) | 59.41 (-126.36, 243.64) | - | IVT+LASER | 1.17 (-5.85, 9.26) | 6.43 (-2.80, 15.28) |
| -23.27 (-121.84, 81.43) | -16.45 (-117.29, 86.00) | - | -66.32 (-176.81, 46.22) | -51.41 (-157.00, 63.95) | - | -111.34 (-254.61, 37.93) | LASER | 5.10 (-2.19, 12.07) |
| -131.82 (-231.95, -28.49) | -126.16 (-227.11, -20.62) | - | -174.45 (-349.45, 3.79) | -159.11 (-333.08, 18.42) | - | -220.07 (-368.98, -71.10) | -108.67 (-246.74, 28.57) | Placebo |
IVB, intravitreal bevacizumab; IVR, intravitreal ranibizumab; LASER, laser, macular laser, grid laser and focal/grid laser; IVT, intravitreal triamcinolone.
Table 3. Network meta-analysis results in BCVA (upper part) and CMT (lower part) at 12 months.
| DDSI | 0.41 (-6.98, 8.14) | -7.07 (-13.77, -0.27) | -3.38 (-9.45, 2.45) | 0.53 (-7.14, 8.14) | -1.07 (-8.11, 6.43) | -4.97 (-11.17, 1.35) | -4.09 (-10.39, 2.33) | 0.56 (-6.44, 7.72) | 1.20 (-5.07, 7.84) | 6.74 (-1.38, 14.99) |
| - | DDSI+LASER | -7.38 (-12.97, -2.40) | -3.72 (-8.71, 0.62) | 0.26 (-6.73, 6.32) | -1.36 (-7.24, 4.23) | -5.35 (-9.74, -1.10) | -4.46 (-8.88, -0.33) | 0.17 (-5.01, 5.28) | 0.81 (-3.16, 4.73) | 6.36 (-0.61, 12.96) |
| - | 110.83 (35.27, 190.25) | IVA | 3.71 (0.36, 6.68) | 7.60 (1.73, 13.14) | 5.99 (1.24, 11.17) | 2.07 (-0.97, 5.33) | 2.93 (-0.34, 6.57) | 7.55 (3.23, 12.37) | 8.19 (5.07, 11.96) | 13.84 (7.84, 19.71) |
| - | 56.20 (-9.79, 124.73) | -54.65 (-104.84, -3.26) | IVB | 3.95 (-0.91, 8.61) | 2.32 (-1.58, 6.76) | -1.67 (-3.35, 0.74) | -0.80 (-2.87, 2.08) | 3.85 (0.35, 8.19) | 4.47 (2.56, 7.28) | 10.11 (4.76, 15.79) |
| - | 31.16 (-49.84, 115.46) | -79.38 (-154.61, -4.14) | -25.14 (-82.54, 32.81) | IVB+IVT | -1.60 (-7.72, 4.97) | -5.56 (-10.41, -0.34) | -4.69 (-9.79, 0.75) | -0.10 (-5.67, 6.39) | 0.60 (-4.43, 6.00) | 6.25 (-1.07, 13.46) |
| - | - | - | - | - | IVB+LASER | -3.96 (-8.20, 0.13) | -3.09 (-7.41, 1.09) | 1.55 (-3.65, 6.69) | 2.20 (-1.87, 6.30) | 7.84 (1.01, 14.18) |
| - | 74.43 (12.00, 137.89) | -36.39 (-87.43, 13.00) | 18.24 (-13.25, 47.58) | 43.14 (-17.96, 103.47) | - | IVR | 0.87 (-0.65, 2.45) | 5.48 (2.18, 9.08) | 6.14 (4.74, 7.84) | 11.76 (6.55, 16.73) |
| - | 83.42 (20.92, 146.51) | -27.30 (-84.80, 28.17) | 27.00 (-12.56, 64.73) | 52.15 (-10.76, 114.94) | - | 9.00 (-18.60, 38.21) | IVR+LASER | 4.61 (1.32, 8.02) | 5.28 (3.80, 6.90) | 10.93 (5.43, 16.03) |
| - | 63.27 (-11.16, 141.16) | -46.93 (-119.78, 23.15) | 7.55 (-51.67, 64.19) | 32.69 (-45.99, 108.50) | - | -10.83 (-65.55, 43.23) | -19.72 (-70.09, 30.24) | IVT+LASER | 0.66 (-2.64, 3.95) | 6.25 (-0.15, 12.30) |
| - | 22.23 (-34.26, 80.08) | -88.50 (-143.87, -35.10) | -34.08 (-67.36, -1.11) | -9.18 (-68.11, 49.90) | - | -52.11 (-77.09, -26.21) | -61.14 (-87.68, -34.75) | -41.07 (-91.20, 9.57) | LASER | 5.59 (0.08, 10.71) |
| - | -71.76 (-161.34, 21.75) | -182.93 (-268.84, -96.95) | -128.15 (-202.57, -50.77) | -103.81 (-193.32, -9.21) | - | -145.96 (-213.86, -75.51) | -155.43 (-227.86, -78.77) | -135.36 (-222.74, -46.31) | -94.01 (-165.76, -18.93) | Placebo |
IVA, intravitreal aflibercept; IVB, intravitreal bevacizumab; IVR, intravitreal ranibizumab; LASER, laser, macular laser, grid laser and focal/grid laser; DDSI, dexamethasone implant; IVT, intravitreal triamcinolone.
Characteristics of the 21 included studies are presented in Table 1. The 21 eligible RCTs contained a total of 4307 eyes, including 428 eyes with 6-month follow-up and 3879 eyes with 12-month follow-up. The data of BCVA and CMT in 6 and 12 months were recorded respectively for the network meta-analysis.
Risk of Bias
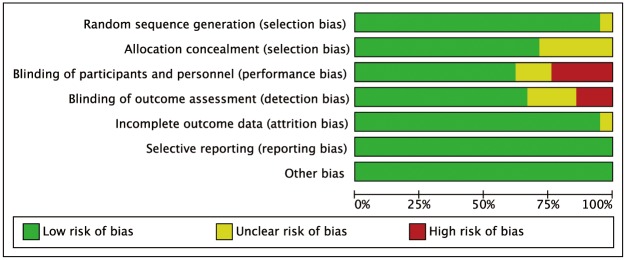
The biases of the 21 included studies were assessed by using the Cochrane Collaboration’s tool as shown in Figs 3 and 4. One of the trials did not describe the method used for generating the allocation sequence [36]; six not for allocation concealment [27, 29, 36, 39, 40, 44]; three not for blinding of participants and personnel [29, 40, 44]; four not for blinding of outcome assessment [29, 40, 44, 47]; one not for incomplete outcome data [45]. Therefore, the risk of bias was considered unclear. In all, five trials performed a high risk of bias in blinding of participants and personnel [30, 35, 41, 46, 47], and three studies featured a high risk of outcome assessment [27, 31, 35].
Fig 3. Risk of bias graph.
Fig 4. Risk of bias summary.
The modified Jadad scale of 21 trials is listed in Table 1, showing similar results to the bias using the Cochrane Collaboration’s tool.
Visual Acuity
Since BCVA is the primary efficacy and progress indicator, and CMT is an important anatomical indicator in treating DME, data of BCVA and CMT were analyzed. For the baseline BCVA and CMT were not strictly matched, the mean change of BCVA and CMT rather than BCVA and CMT themselves was analyzed as the primary outcome (Tables 2 and 3).
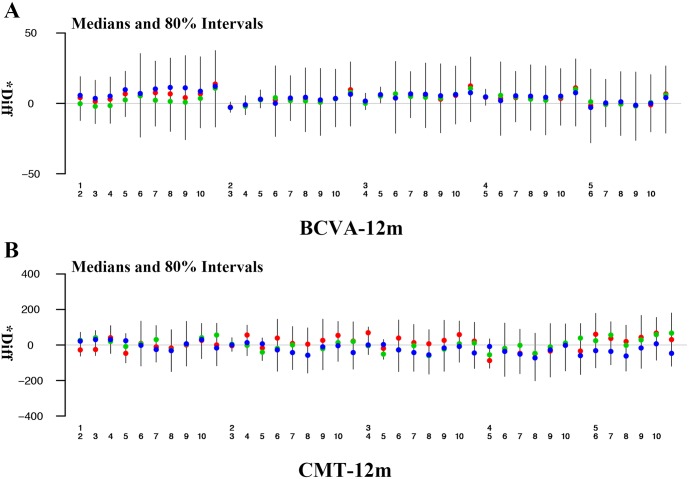
The results of mean BCVA changes in 6 months from baseline showed that intravitreal ranibizumab (IVR) 7.01 (2.56 to 11.39) was best, followed by intravitreal bevacizumab (IVB) 5.69 (1.45 to 10.70) and IVR plus LASER 4.41 (0.04 to 8.89) when compared with LASER alone (Table 2). Both IVR and IVB were significantly superior to LASER alone. All comparisons showed no significant heterogeneity (all p>0.05, S1 Table). In 12 months, intravitreal aflibercept (IVA) 8.19 (5.07 to 11.96) was best, followed by IVR 6.14 (4.74 to 7.84) and IVR plus LASER 5.28 (3.80 to 6.90) when compared with LASER alone (Table 3, Fig 5). All of them were significantly superior to LASER alone. However, there was significant heterogeneity in the comparison between IVB and LASER (p = 0.04, S1 Table). IVR and IVA were superior to other therapies on the basis of mean BCVA changes from baseline in 6 and 12 months. Ranking based on simulations is available in the Supplemental Materials and Methods (See S2 Table).
Fig 5. Forest plots for BCVA and CMT in 12 months.
Abbreviations: 1, IVA; 2, IVB; 3, IVR; 4, IVR+LASER; 5, LASER; 6, DDSI; 7, IVB+IVT; 8, DDSI+LASER; 9, IVB+LASER; 10, IVT+LASER; 11, Placebo.
Central Macular Thickness
Analyses of mean CMT changes in 6 months from baseline showed that intravitreal triamcinolone (IVT) plus LASER -111.34 (-254.61 to 37.93) was best, followed by IVR -66.32 (-176.81 to 46.22) and IVR plus LASER -51.41 (-157.00 to 63.95), when compared with LASER alone (Table 2). None of them was statistically superior to LASER alone. In 12 months, IVA -110.83 (-190.25 to -35.27) was best, followed by IVR plus LASER -83.42 (-146.51 to -20.92) and IVR -74.43 (-137.89 to -12.00), when compared with DDSI+ LASER (Table 3, Fig 5). And all of IVA, IVR and IVR plus LASER were significantly superior to DDSI+LASER. All comparisons showed no significant heterogeneity in 12 months (all p>0.05, S1 Table). IVT plus LASER was better than IVR and IVR plus LASER on the basis of mean CMT changes from baseline in 6 months. Meanwhile, IVA was better than IVR and IVR plus LASER on the basis of mean CMT changes from baseline in 12 months. Ranking based on simulations is available in the Supplemental Materials and Methods (See S2 Table).
Adverse Events
Of the 21 trials with 4307 eyes involved, the most common ocular AEs were IOP increase and cataract progress. In 13 trials, IOP increase was reported, and the incidence of IOP increase was 47.7% in the IVT plus LASER group [43, 46], 7.6% in the IVR plus LASER group [29, 39, 43], 7.5% in the IVB group [27, 30, 41], 7.2% in the IVA group [27], and 4.8% in the IVR group [27, 29, 39]. Furthermore, cataract progression was reported in 8 trials, and the rate was 26.9% in the IVT plus LASER group [43], 8% in the IVR group [29], 6.5% in the IVB group [30, 33, 37], 6.3% in the IVR plus LASER group [28, 43], and 0.5% in the IVA group [27]. The groups with administration of steroids had a higher incidence of IOP increase and cataract progress than the groups with use of VEGF inhibitors in the treatment of DME.
Hypertension, angina, and myocardial infarction were reported as systematic AEs potentially related to VEGF inhibition. The incidence of hypertension was reported in 8 trials, being 12.5% in the IVA group [27], 7.6% in the IVR group [27–29, 33, 39, 42], 6.2% in the IVB group [27, 30, 41], and 4.9% in the IVR plus LASER group [27, 28]. The rate of angina was 0.8% in the IVR group [28]. Myocardial infarction was reported in 3 trials, with a rate of 1.9% in the IVA group, 0.5% in the IVB group [27], and 1.2% in the IVR group [27, 39, 42].
Consistency of Network-model and Sensitivity Analysis
Based on direct versus indirect evidences, we compared the effect estimate using the node-splitting and pair-wise meta-analysis (S1 Table). No inconsistencies were observed. We performed the sensitivity analysis of comparison of random and fixed effects model that was more accurate. The outcome was not changed (S3 Table). Those data suggested that our model was very robust.
Discussion
In this network meta-analysis concerning 21 trials and 4307 eyes, we reviewed published information and analyzed the efficacy and safety of therapeutic approaches in the management of DME. It was indicated that the most efficacious treatment was IVA based on the mean BCVA and CMT changes in 12 months, followed by IVR. This was similar to a recent publication showing that greater BCVA improvement was seen with aflibercept than with bevacizumab or ranibizumab [27].
Meanwhile, we found that IVT combined with LASER was best in decreasing CMT in 6 months with no statistical significance. One of the reasons is that IVT plus LASER might have a stronger anti-inflammatory and anti-angiogenic effect in early stage after injection. This only affects the CMT decrease, but has no impact on BCVA improvement [19]. The other reason may be the bias of 3 trials concerning IVT plus LASER [43, 44, 46]. It was a median risk for no description of allocation concealment, blinding of participants and personnel, and blinding of outcome assessment in the report of Pappas et al, but was a high risk of blinding of participants and personnel according to the results of Lam et al. Moreover, there was no trial reporting CMT data related to the IVA treatment in 6 months. Therefore, clinicians should notice that IVT combined with LASER might have benefits in the early stage after injection for decreasing CMT.
There was no statistically significant difference between IVR and IVR plus LASER in either BCVA improvement or CMT decrease during the follow-up period. Another finding was that LASER was superior to placebo only in decreasing CMT in 12 months, suggesting that the role of LASER in the treatment of DME should be reappraised. Meanwhile, 13 trials involved laser treatment in our study, including macular laser (4 trials), grid laser (3 trials), and focal/grid laser (6 trials). Although focal and grid laser photocoagulations belong to macular laser treatment [48], there may be heterogeneity in these two macular laser treatments. Our meta-analysis was based on an assumption that the LASER therapies were same, and clinicians should pay attention to this.
Among ocular AEs, high IOP occurred more frequently in the IVT plus LASER group (47.7%) than the anti-VEGF groups (4.8%-7.2%), while cataract progression was also mostly found in the IVT plus LASER (47.7%) group than the anti-VEGF groups (4.8%-7.2%). Those data were consistent with the notion that there were more ocular AEs in the steroids treatment groups [49]. The incidence of hypertension and myocardial infarction in the anti-VEGF groups was higher than the steroids groups. Diabetic patients are known to be at a higher risk of cardiovascular comorbidities, which are susceptible to systemic complications in addition to DME. The data were in keeping with previously reported AEs occurring with anti-VEGF treatment [11].
Our network meta-analysis focused on the drugs commonly used in patients with DME, and the results of this work may be important for clinical treatment. However, there are also limitations that need to be taken into account. The challenge in the network meta-analysis is that characteristics of the included 21 trials could not be matched, such as the duration of diabetics, the dosage and number of each intervention and the type of laser treatment. Moreover, the eligible 21 trials did not present the outcomes of patients in different types of diabetes separately, so it remains unknown whether any specific type of diabetes could alter the outcomes. The definition and details of AEs were not always reported in each involved study, so that it was not possible to assess the exact incidence of AEs. Therefore, the data available can only indicate the relative safety of every intervention for DME. To more accurately assess the efficacy of these treatments, additional high-quality RCTs will be necessary.
Conclusions
Our analysis confirms that intravitreal aflibercept is most favorable for BCVA improvement and CMT decrease compared with other current therapies in the management of DME within 12 months. VEGF inhibitors for DME should be used with caution due to systematic AEs. Combined IVT with LASER has a stronger efficacy in decreasing CMT than the other interventions in the early stage after injection. More high-quality randomized controlled trials will be necessary.
Supporting Information
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Acknowledgments
The authors thank Ping Lin for her linguistic and editorial assistance.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This study was partly supported by a grant from the National Natural Science Foundation of China (81570820).
References
- 1.Williams R, Airey M, Baxter H, Forrester J, Kennedy-Martin T, Girach A. Epidemiology of diabetic retinopathy and macular oedema: a systematic review. Eye. 2004. October;18(10):963–83. [DOI] [PubMed] [Google Scholar]
- 2.Klein R, Klein BE, Moss SE, Davis MD, DeMets DL. The Wisconsin epidemiologic study of diabetic retinopathy. IV. Diabetic macular edema. Ophthalmology. 1984. December;91(12):1464–74. [DOI] [PubMed] [Google Scholar]
- 3.Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Archives of ophthalmology. 1985. December;103(12):1796–806. [PubMed] [Google Scholar]
- 4.Funatsu H, Noma H, Mimura T, Eguchi S, Hori S. Association of vitreous inflammatory factors with diabetic macular edema. Ophthalmology. 2009. January;116(1):73–9. 10.1016/j.ophtha.2008.09.037 [DOI] [PubMed] [Google Scholar]
- 5.Ozaki H, Hayashi H, Vinores SA, Moromizato Y, Campochiaro PA, Oshima K. Intravitreal sustained release of VEGF causes retinal neovascularization in rabbits and breakdown of the blood-retinal barrier in rabbits and primates. Experimental eye research. 1997. April;64(4):505–17. [DOI] [PubMed] [Google Scholar]
- 6.Miyamoto K, Khosrof S, Bursell SE, Moromizato Y, Aiello LP, Ogura Y, et al. Vascular endothelial growth factor (VEGF)-induced retinal vascular permeability is mediated by intercellular adhesion molecule-1 (ICAM-1). The American journal of pathology. 2000. May;156(5):1733–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Behl T, Kotwani A. Exploring the various aspects of the pathological role of vascular endothelial growth factor (VEGF) in diabetic retinopathy. Pharmacological research. 2015. September;99:137–48. 10.1016/j.phrs.2015.05.013 [DOI] [PubMed] [Google Scholar]
- 8.Schatz H, Madeira D, McDonald HR, Johnson RN. Progressive enlargement of laser scars following grid laser photocoagulation for diffuse diabetic macular edema. Archives of ophthalmology. 1991. November;109(11):1549–51. [DOI] [PubMed] [Google Scholar]
- 9.Agarwal A, Afridi R, Hassan M, Sadiq MA, Sepah YJ, Do DV, et al. Novel Therapies in Development for Diabetic Macular Edema. Current diabetes reports. 2015. October;15(10):75 10.1007/s11892-015-0652-z [DOI] [PubMed] [Google Scholar]
- 10.Sultan MB, Zhou D, Loftus J, Dombi T, Ice KS, Macugen Study G. A phase 2/3, multicenter, randomized, double-masked, 2-year trial of pegaptanib sodium for the treatment of diabetic macular edema. Ophthalmology. 2011. June;118(6):1107–18. 10.1016/j.ophtha.2011.02.045 [DOI] [PubMed] [Google Scholar]
- 11.Brown DM, Nguyen QD, Marcus DM, Boyer DS, Patel S, Feiner L, et al. Long-term outcomes of ranibizumab therapy for diabetic macular edema: the 36-month results from two phase III trials: RISE and RIDE. Ophthalmology. 2013. October;120(10):2013–22. 10.1016/j.ophtha.2013.02.034 [DOI] [PubMed] [Google Scholar]
- 12.Diabetic Retinopathy Clinical Research N, Scott IU, Edwards AR, Beck RW, Bressler NM, Chan CK, et al. A phase II randomized clinical trial of intravitreal bevacizumab for diabetic macular edema. Ophthalmology. 2007. October;114(10):1860–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Korobelnik JF, Do DV, Schmidt-Erfurth U, Boyer DS, Holz FG, Heier JS, et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2014. November;121(11):2247–54. 10.1016/j.ophtha.2014.05.006 [DOI] [PubMed] [Google Scholar]
- 14.Regnier SA, Larsen M, Bezlyak V, Allen F. Comparative efficacy and safety of approved treatments for macular oedema secondary to branch retinal vein occlusion: a network meta-analysis. BMJ open. 2015. June 5;5(6):e007527 10.1136/bmjopen-2014-007527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Das A, McGuire PG, Rangasamy S. Diabetic Macular Edema: Pathophysiology and Novel Therapeutic Targets. Ophthalmology. 2015. July;122(7):1375–94. 10.1016/j.ophtha.2015.03.024 [DOI] [PubMed] [Google Scholar]
- 16.Ishida S, Usui T, Yamashiro K, Kaji Y, Ahmed E, Carrasquillo KG, et al. VEGF164 is proinflammatory in the diabetic retina. Invest Ophthalmol Vis Sci. 2003. May;44(5):2155–62. [DOI] [PubMed] [Google Scholar]
- 17.Campa C, Harding SP. Anti-VEGF compounds in the treatment of neovascular age related macular degeneration. Current drug targets. 2011. February;12(2):173–81. [DOI] [PubMed] [Google Scholar]
- 18.Kleijnen J, Korobelnik JF, Lang SH, Birnie R, Leadley RM, Misso K, et al. Systematic review and mixed treatment comparison of intravitreal aflibercept with other therapies for diabetic macular edema (DME). BMC Ophthalmol. 2015. May 15;15:52 10.1186/s12886-015-0035-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang XL, Chen J, Zhang RJ, Wang WJ, Zhou Q, Qin XY. Intravitreal triamcinolone versus intravitreal bevacizumab for diabetic macular edema: a meta-analysis. Int J Ophthalmol. 2013. August 18;6(4):546–52. 10.3980/j.issn.2222-3959.2013.04.26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ford JA, Lois N, Royle P, Clar C, Shyangdan D, Waugh N. Current treatments in diabetic macular oedema: systematic review and meta-analysis. BMJ open. 2013. March 1;3(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bhatnagar N, Lakshmi PV, Jeyashree K. Multiple treatment and indirect treatment comparisons: An overview of network meta-analysis. Perspectives in clinical research. 2014. October;5(4):154–8. 10.4103/2229-3485.140550 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lumley T. Network meta-analysis for indirect treatment comparisons. Statistics in medicine. 2002. August 30;21(16):2313–24. [DOI] [PubMed] [Google Scholar]
- 23.Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. Journal of clinical epidemiology. 2011. February;64(2):163–71. 10.1016/j.jclinepi.2010.03.016 [DOI] [PubMed] [Google Scholar]
- 24.Salanti G, Higgins JP, Ades AE, Ioannidis JP. Evaluation of networks of randomized trials. Statistical methods in medical research. 2008. June;17(3):279–301. [DOI] [PubMed] [Google Scholar]
- 25.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. Bmj. 2003. September 6;327(7414):557–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dias S, Welton NJ, Caldwell DM, Ades AE. Checking consistency in mixed treatment comparison meta-analysis. Statistics in medicine. 2010. March 30;29(7–8):932–44. 10.1002/sim.3767 [DOI] [PubMed] [Google Scholar]
- 27.Wells JA, Glassman AR, Ayala AR, Jampol LM, Aiello LP, Antoszyk AN, et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. The New England journal of medicine. 2015. March 26;372(13):1193–203. 10.1056/NEJMoa1414264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ishibashi T, Li X, Koh A, Lai TY, Lee FL, Lee WK, et al. The REVEAL Study: Ranibizumab Monotherapy or Combined with Laser versus Laser Monotherapy in Asian Patients with Diabetic Macular Edema. Ophthalmology. 2015. July;122(7):1402–15. 10.1016/j.ophtha.2015.02.006 [DOI] [PubMed] [Google Scholar]
- 29.Berger A, Sheidow T, Cruess AF, Arbour JD, Courseau AS, De Takacsy F. Efficacy/safety of ranibizumab monotherapy or with laser versus laser monotherpay in DME. Canadian Journal of Ophthalmology. 2015. June;50(3):209–16. 10.1016/j.jcjo.2014.12.014 [DOI] [PubMed] [Google Scholar]
- 30.Gillies MC, Lim LL, Campain A, Quin GJ, Salem W, Li J, et al. A randomized clinical trial of intravitreal bevacizumab versus intravitreal dexamethasone for diabetic macular edema: the BEVORDEX study. Ophthalmology. 2014. December;121(12):2473–81. 10.1016/j.ophtha.2014.07.002 [DOI] [PubMed] [Google Scholar]
- 31.Comyn O, Sivaprasad S, Peto T, Neveu MM, Holder GE, Xing W, et al. A randomized trial to assess functional and structural effects of ranibizumab versus laser in diabetic macular edema (the LUCIDATE study). American journal of ophthalmology. 2014. May;157(5):960–70. 10.1016/j.ajo.2014.02.019 [DOI] [PubMed] [Google Scholar]
- 32.Shoeibi N, Ahmadieh H, Entezari M, Yaseri M. Intravitreal Bevacizumab with or without Triamcinolone for Refractory Diabetic Macular Edema: Long-term Results of a Clinical Trial. Journal of ophthalmic & vision research. 2013. April;8(2):99–106. [PMC free article] [PubMed] [Google Scholar]
- 33.Nepomuceno AB, Takaki E, Paes de Almeida FP, Peroni R, Cardillo JA, Siqueira RC, et al. A prospective randomized trial of intravitreal bevacizumab versus ranibizumab for the management of diabetic macular edema. American journal of ophthalmology. 2013. September;156(3):502–10.e2. 10.1016/j.ajo.2013.04.026 [DOI] [PubMed] [Google Scholar]
- 34.Callanan DG, Gupta S, Boyer DS, Ciulla TA, Singer MA, Kuppermann BD, et al. Dexamethasone intravitreal implant in combination with laser photocoagulation for the treatment of diffuse diabetic macular edema. Ophthalmology. 2013. September;120(9):1843–51. 10.1016/j.ophtha.2013.02.018 [DOI] [PubMed] [Google Scholar]
- 35.Berger A, Sheidow T, Li R, Rehel B, Takacsy F, Courseau AS. A canadian 12-month, phiiib study of ranibizumab combination or monotherapy in visual impairment due to diabetic macular edema: Preliminary analysis ("respond"). Canadian Journal of Diabetes [Internet]. 2013; 37:[S48 p.]. Available from: http://onlinelibrary.wiley.com/o/cochrane/clcentral/articles/646/CN-01024646/frame.html, http://www.canadianjournalofdiabetes.com/article/S1499-2671(13)01088-5/pdf. [Google Scholar]
- 36.Arevalo JF, Lasave AF, Wu L, Diaz-Llopis M, Gallego-Pinazo R, Alezzandrini AA, et al. Intravitreal bevacizumab plus grid laser photocoagulation or intravitreal bevacizumab or grid laser photocoagulation for diffuse diabetic macular edema: results of the Pan-american Collaborative Retina Study Group at 24 months. Retina (Philadelphia, Pa). 2013. February;33(2):403–13. [DOI] [PubMed] [Google Scholar]
- 37.Soheilian M, Garfami KH, Ramezani A, Yaseri M, Peyman GA. Two-year results of a randomized trial of intravitreal bevacizumab alone or combined with triamcinolone versus laser in diabetic macular edema. Retina (Philadelphia, Pa). 2012. February;32(2):314–21. [DOI] [PubMed] [Google Scholar]
- 38.Synek S, Veselý P. Intravitreal Bevacizumab with or without Triamcinolone for Refractory Diabetic Macular Oedema. Collegium antropologicum. 2011. July;35(3):841–5. [PubMed] [Google Scholar]
- 39.Mitchell P, Bandello F, Schmidt-Erfurth U, Lang GE, Massin P, Schlingemann RO, et al. The RESTORE study: ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology. 2011. April;118(4):615–25. 10.1016/j.ophtha.2011.01.031 [DOI] [PubMed] [Google Scholar]
- 40.Nguyen QD, Shah SM, Khwaja AA, Channa R, Hatef E, Do DV, et al. Two-year outcomes of the ranibizumab for edema of the mAcula in diabetes (READ-2) study. Ophthalmology. 2010. November;117(11):2146–51. 10.1016/j.ophtha.2010.08.016 [DOI] [PubMed] [Google Scholar]
- 41.Michaelides M, Kaines A, Hamilton RD, Fraser-Bell S, Rajendram R, Quhill F, et al. A prospective randomized trial of intravitreal bevacizumab or laser therapy in the management of diabetic macular edema (BOLT study) 12-month data: report 2. Ophthalmology. 2010. June;117(6):1078–1086.e2. 10.1016/j.ophtha.2010.03.045 [DOI] [PubMed] [Google Scholar]
- 42.Massin P, Bandello F, Garweg JG, Hansen LL, Harding SP, Larsen M, et al. Safety and efficacy of ranibizumab in diabetic macular edema (RESOLVE Study): a 12-month, randomized, controlled, double-masked, multicenter phase II study. Diabetes care. 2010. November;33(11):2399–405. 10.2337/dc10-0493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Diabetic Retinopathy Clinical Research N, Elman MJ, Aiello LP, Beck RW, Bressler NM, Bressler SB, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010. June;117(6):1064–1077.e35. 10.1016/j.ophtha.2010.02.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pappas GD, Adam CI, Papageorgioy E, Kefalogiannis N, Fanouriakis H. Triamcinolone and Grid Laser versus Bevacizumab Alone for the Treatment of Diabetic Macular Edema. Iovs [Internet]. 2008:[ARVO E- abstract 3483 pp.]. Available from: http://onlinelibrary.wiley.com/o/cochrane/clcentral/articles/124/CN-00746124/frame.html. [Google Scholar]
- 45.Ahmadieh H, Ramezani A, Shoeibi N, Bijanzadeh B, Tabatabaei A, Azarmina M, et al. Intravitreal bevacizumab with or without triamcinolone for refractory diabetic macular edema; a placebo-controlled, randomized clinical trial. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2008. April;246(4):483–9. [DOI] [PubMed] [Google Scholar]
- 46.Lam DS, Chan CK, Mohamed S, Lai TY, Lee VY, Liu DT, et al. Intravitreal triamcinolone plus sequential grid laser versus triamcinolone or laser alone for treating diabetic macular edema: six-month outcomes. Ophthalmology. 2007. December;114(12):2162–7. [DOI] [PubMed] [Google Scholar]
- 47.Audren F, Erginay A, Haouchine B, Benosman R, Conrath J, Bergmann JF, et al. Intravitreal triamcinolone acetonide for diffuse diabetic macular oedema: 6-month results of a prospective controlled trial. Acta ophthalmologica Scandinavica. 2006. October;84(5):624–30. [DOI] [PubMed] [Google Scholar]
- 48.Dowler JG. Laser management of diabetic retinopathy. J R Soc Med. 2003. June;96(6):277–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kriechbaum K, Prager S, Mylonas G, Scholda C, Rainer G, Funk M, et al. Intravitreal bevacizumab (Avastin) versus triamcinolone (Volon A) for treatment of diabetic macular edema: one-year results. Eye. 2014. January;28(1):9–15; quiz 16. 10.1038/eye.2013.242 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.