Abstract
Purpose
To evaluate radiotherapy (RT) and chemotherapy (CT) treatments of early-stage extranodal natural killer/T-cell lymphoma (ENKTL).
Materials and Methods
Fifty-five patients with stage I or II ENKTL [n=39 (71%) and 16 (29%) patients, respectively] who were treated with RT between 1999 and 2013 were analyzed retrospectively. The median age was 54 years (range, 24–81). Patients were grouped by treatment modality as RT alone [n=19 (35%)], upfront CT plus RT [CT+RT, n=16 (29%)], and concurrent chemoradiotherapy [CCRT, n=20 (36%)]. The median RT dose was 48 Gy. Patient characteristics between each treatment group were well balanced. Patterns of failure and survival were analyzed.
Results
The overall response rate after RT was 94.6%. Ten patients experienced distant failure, and seven experienced local failure comprising five in-field and two out-field failures. The local and distant failure rates in the RT-alone group were the same (16%). In the CT+RT group, the most common failure sites were local (19%). In the CCRT group, the most common failures were distant (25%). At a median follow-up of 56 months (range, 1–178 months), the 5-year overall survival (OS) and progression-free survival rates were 66% and 54%, respectively. The 5-year OS rate for the RT-alone and CT+RT groups were 76% and 69%, respectively, and the 2-year OS rate for the CCRT group was 62% (p=0.388).
Conclusion
In the era of multimodal treatment for ENKTL, RT alone using advanced techniques should be considered for local disease control, whereas maintenance CT regimens should be considered for distant disease control.
Keywords: Lymphoma, radiotherapy, recurrencec
INTRODUCTION
Nasal-type extranodal natural killer (NK)/T-cell lymphoma (ENKTL), formerly referred to as polymorphic reticulosis, midline malignant reticulosis, or T-cell angiocentric lymphoma, was classified as NK/T-cell lymphoma in the 1998 World Health Organization (WHO) classification. According to a nationwide study of malignant lymphomas, ENKTL is the third most common lymphoma in Korea.1 Early-stage ENKTL, characterized by extensive angioinvasion and necrosis, is usually localized to the upper aerodigestive tract and has been associated with Epstein-Barr virus infection.
The optimal management of ENKTL has changed continuously. Before it was classified as a lymphoma, ENKTL was treated using radiotherapy (RT) alone. In a report from our institution,2 patients treated with RT alone experienced an 83.7% overall response rate, but local and systemic failure rates of 50% and 25%, respectively, resulting in a 5-year overall survival (OS) rate of 40%. After being classified as a lymphoma, upfront chemotherapy (CT) was administered as the first-line treatment, although frequent local failures were observed.3 Although combinations of multiple CT agents such as cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP); etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin (EPOCH); and steroid, methotrexate, ifosfamide, L-asparaginase, and etoposide (SMILE), were used with concurrent (CCRT) or sequential chemoradiotherapy (CT+RT), neither the incidence of systemic relapse nor the prognosis were significantly altered by the inclusion of CT.4 Furthermore, the local control rates with CT combinations were comparable to those for RT alone when using modern radiation techniques.4 CCRT followed by maintenance CT was considered as a treatment option. According to the Korean lymphoma consortium,5 CCRT followed by VP-16, ifosfamide, cisplatin, and dexamethasone (VIPD) showed promising outcomes, resulting in a 73% complete response (CR) rate and a 3-year OS rate of 86.2%. None-theless, distant failures did not decrease compared to data for RT alone.
There is no clear consensus on the optimal management for ENKTL, and randomized trials comparing RT alone with CCRT have not been conducted. Presently, the National Comprehensive Cancer Network guidelines recommend RT alone, CCRT, or CT+RT for stage I ENKTL without any known risk factors. This study analyzed clinical outcomes including the failure patterns, survival, and toxicities in patients with stage I and II ENKTL to further evaluate and compare the clinical benefits of RT and CT for the treatment of ENKTL.
MATERIALS AND METHODS
Patients
Between 1999 and 2013, 82 patients with ENKTL underwent RT at Yonsei Cancer Center in Seoul, Korea. After reviewing the pathology and medical records retrospectively, 17 patients with Ann Arbor stage III or IV disease and eight patients with primary tumors in regions other than the head and neck region were excluded. In addition, two patients were excluded as they had undergone salvage therapy after first-line treatment failure. The final analysis population consisted of 55 eligible patients with stage I or II ENKTL.
A pathologic diagnosis of ENKTL was based on either atypical CD56-expressing lymphoid cell proliferation or nuclear Epstein-Barr virus–encoded small mRNA-positive and cytoplasmic cytotoxic molecule–expressing atypical lymphoid cell proliferation. At diagnosis, patients underwent pretreatment evaluations including history taking, physical examination, complete blood count, serum biochemistry analysis including lactate dehydrogenase (LDH) level measurements, bone marrow aspiration and biopsy, computed tomography scanning and/or magnetic resonance imaging (MRI) of the involved lesion, and positron emission tomography (PET). PET was performed in 41 patients (74.5%).
Patient characteristics are summarized in Table 1. The median age was 54 years (range, 24–81 years). Seventeen patients (68%) were younger than 60 years of age. The male/female ratio was 1.6:1. According to the NK/T-cell lymphoma prognostic index (NKPI),6 which includes the presence of "B" symptoms, lesions at stages III or IV, elevated serum LDH concentration, and lymph node involvement, 15 patients (28%) were classified as group III or IV (i.e., those with >2 risk factors).
Table 1. Patient Characteristics.
| Variables | Groups | n | % |
|---|---|---|---|
| Sex | Male | 34 | 62 |
| Female | 21 | 38 | |
| Age (yrs) | Median (range) | 54 (24-81) | |
| ≥60 | 17 | 32 | |
| <60 | 38 | 68 | |
| Ann Arbor stage | I | 39 | 71 |
| II | 16 | 29 | |
| Performance status | ECOG 0, 1 | 52 | 94 |
| ECOG 2 | 3 | 6 | |
| Primary site | Sinonasal | 47 | 86 |
| Others | 8 | 14 | |
| Epstein-Barr virus | Yes | 13 | 24 |
| Not checked | 42 | 76 | |
| Serum LDH | Elevated | 26 | 68 |
| Normal | 12 | 32 | |
| B symptoms | Yes | 13 | 24 |
| No | 42 | 76 | |
| IPI | 1 (low) | 21 | 38 |
| 2 (low intermediate) | 28 | 51 | |
| 3 (high-intermediate) | 6 | 11 | |
| 4 (high) | 0 | 0 | |
| NKPI | Group 1 | 19 | 35 |
| Group 2 | 21 | 38 | |
| Group 3 | 13 | 24 | |
| Group 4 | 2 | 4 | |
ECOG, Eastern Cooperative Oncology Group; IPI, international prognostic index; LDH, lactate dehydrogenase; NKPI, NK/T cell lymphoma prognostic index. 6
Patients were subdivided into groups based on the treatment modality as follows: RT alone (n=19, 35%), CT+RT (n=16, 29%), and CCRT (n=20, 36%). Before 1987, our treatment policy for stage I or II ENKTL was to administer involved-field RT alone. After 1987, patients underwent either CT+RT or RT alone. More recently, patients have been treated with CCRT alone or CCRT followed by maintenance CT. Patients' characteristics between the groups were well balanced and are shown in Table 2.
Table 2. Patient Characteristics According to Treatment Modality Groups.
| Variables | Groups | RT alone, n (%) | CT+RT, n (%) | CCRT, n (%) | p value |
|---|---|---|---|---|---|
| Sex | Male | 10 (53) | 12 (75) | 12 (60) | 0.395 |
| Female | 9 (47) | 4 (25) | 8 (40) | ||
| Age (yrs) | ≥60 | 12 (63) | 14 (88) | 12 (60) | 0.169 |
| <60 | 7 (37) | 2 (12) | 8 (40) | ||
| Stage | I | 15 (79) | 11 (69) | 13 (65) | 0.656 |
| II | 4 (21) | 5 (31) | 7 (35) | ||
| Performance | ECOG 0, 1 | 19 (100) | 16 (100) | 17 (85) | 0.102 |
| ECOG 2 | 0 (0) | 0 (0) | 3 (15) | ||
| Primary site | Sinonasal | 15 (79) | 13 (81) | 19 (95) | 0.352 |
| Others | 4 (21) | 3 (19) | 1 (5) | ||
| Serum LDH | Elevated | 5 (71) | 9 (82) | 12 (60) | 0.476 |
| Normal | 2 (29) | 2 (18) | 8 (40) | ||
| B symptoms | Yes | 3 (16) | 3 (19) | 7 (35) | 0.376 |
| No | 16 (84) | 13 (81) | 13 (65) | ||
| IPI | 1 | 10 (53) | 7 (44) | 4 (20) | 0.225 |
| 2 | 8 (42) | 8 (50) | 12 (60) | ||
| 3 | 1 (5) | 1 (6) | 4 (20) | ||
| NKPI | Group 1 | 10 (53) | 5 (31) | 4 (20) | 0.302 |
| Group 2 | 7 (37) | 6 (38) | 8 (40) | ||
| Group 3 | 2 (11) | 4 (25) | 7 (35) | ||
| Group 4 | 0 (0) | 1 (6) | 1 (5) |
RT, radiotherapy; CT, chemotherapy; CCRT, concurrent chemoradiotherapy; ECOG, Eastern Cooperative Oncology Group; IPI, international prognostic index; LDH, lactate dehydrogenase; NKPI, NK/T cell lymphoma prognostic index.6
Treatments
In the CT+RT group, fifteen of 16 patients received maintenance CT after RT. Seven patients were administered CHOP; five were administered cyclophosphamide, vincristine, VP-16, doxorubicin, and prednisone; and four were administered ifosfamide, methotrexate, and VP-16 (IMVP-16). Twenty patients (53%) had cisplatin-based CCRT, 15 of whom were treated with maintenance CT, mainly consisting of VIPD.
Contrast-enhanced computed tomography simulation using a thermoplastic device was mandatory. A mouthpiece was used to distance the tongue from the radiation field in patients who underwent nasal cavity irradiation. All patients underwent involved-site RT (ISRT).7,8 Limited-field ISRT included all gross lesions and adequate margins. Extended-field ISRT included all gross lesions, the bilateral nasal cavities, and the paranasal sinus in cases of stage I disease, and all gross lesions, the bilateral nasal cavities, the paranasal sinus, and the side of involved cervical lymph nodes in cases of stage II disease. In patients with gross lesions outside the nasal cavity, the target volume of extended-field ISRT included the gross lesions, Waldeyer's ring, and the bilateral cervical lymph nodes. Ten patients (18%) underwent two-dimensional RT, 35 (64%) underwent three-dimensional conformal RT (3D-CRT) using 6 MV photons generated by a linear accelerator, and ten (18%) underwent intensity-modulated RT (IMRT) using tomotherapy. Target volume was determined according to the International Commission on Radiation Units and Measurements Report 83.9 We prescribed the radiation dose based on the dose to 95% of the considered volume (D95). Using 3D-CRT, the most common field arrangement was the three-field technique consisting of arrangements weighted in favor of the anterior field and two wedged lateral fields. After a total dose of 45 Gy was irradiated to the gross tumor, a boost dose in the range of 9–18 Gy (up to a total dose of 54–63 Gy) was administered for persistent tumors.
Using IMRT, the simultaneously integrated boost technique was used. Clinical target volume (CTV)1 encompassed the gross lesion, and CTV2 encompassed the margins of CTV1; these were different for the limited and extended-field ISRT. The CTV was modified to reduce the dose to organs at risk. The maximal dose constraints to the spinal cord, optic apparatus, and mandible were <45 Gy, <55 Gy, and <70 Gy, respectively. The larynx, pharynx, and esophagus were also delineated, and the doses were set as low as possible. For the planning target volume, a 0.3 cm margin was applied to the CTV. Two different dose prescriptions were used. For a 20-fraction prescription, the total doses applied to the CTV1 and CTV2 were 48 and 40 Gy, respectively. For a 25-fraction prescription, the total doses applied to the CTV1 and CTV2 were 53 and 45 Gy, respectively.
The radiation doses ranged from 22 to 63 Gy (median, 48 Gy) at a dose per fraction of 1.8–2.4 Gy within 4–6 weeks. Forty-two patients (76%) received >45 Gy. Assuming an α/β ratio of 10 Gy, 24 patients (43.6%) received 40–45 Gy, 20 patients (36.4%) received 46–50.4 Gy, and nine patients (16.4%) received 54–63 Gy in a 1.8 Gy–equivalent dose. Two patients (3.6%) received <40 Gy. A 68-year-old male, who underwent cisplatin-based CCRT, died from pneumonia exacerbation after 22 Gy. A 50-year-old male, who underwent CT+RT consisting of IMVP-16, refused RT after 23.4 Gy due to grade IV oral mucositis.
Assessment and evaluation
Treatment response was assessed according to the WHO criteria. A CR was defined as the disappearance of all previously measurable lesions and the absence of any new tumor lesions. A partial response (PR) was defined as a decrease of ≥50% in the product of two perpendicular diameters of each measurable lesion. Stable disease was defined as a decrease of <50% or an increase of <25% in tumor size. Progressive disease (PD) was defined as a ≥25% increase in the product of the two diameters of at least one tumor or as the presence of any newly developed lesion.
Treatment failure was categorized as local failure, regional failure, or distant failure. Distant failure was diagnosed based on the clinical and/or radiologic findings and was defined as the appearance of systemic disease at sites other than the head and neck or cervical neck lymph nodes. Local failure was categorized as proposed by Koom, et al.10 into true recurrence (TR) that occurred within RT fields, marginal recurrence that occurred near contiguous areas of the primary site yet just outside the border of the RT field, and elsewhere recurrence (ER) that occurred at other extranodal sites in the head and neck. Regional failure was defined as any recurrence in the cervical neck nodes.
To monitor disease progression and patterns of failure, evaluations were performed 1 month after treatment and every 3–6 months thereafter. RT response was assessed via physical examination (n=4, 7%), computed tomography (n=35, 64%), MRI (n=5, 9%), and PET (n=10, 18%). PET was performed for recent patients. Response evaluation was performed within 3 months after the completion of RT for all patients and at 1 month for patients who received CCRT. Treatment-related toxicities were assessed at every follow-up visit. Toxicities were graded based on the Common Toxicity Criteria Version 4.0 from the National Cancer Institute (NCI-CTC v4.0).
Statistical analyses
All statistical analyses were conducted using SPSS version 20.0 (SPSS Inc., Chicago, IL, USA). OS, progression-free survival (PFS), and local failure-free survival (LFFS) were determined using the Kaplan-Meier method, and the differences were evaluated using the log-rank test. Survival time was measured from the date of diagnosis to the date of death from any cause or the date of last follow-up. PFS was an estimation of the measurement from the date of diagnosis to the date of initial relapse or death. LFFS was an estimation of the measurement from the date of diagnosis to the date of local failure or death. A p-value of <0.05 was considered to be statistically significant.
RESULTS
Response to RT and patterns of treatment failure
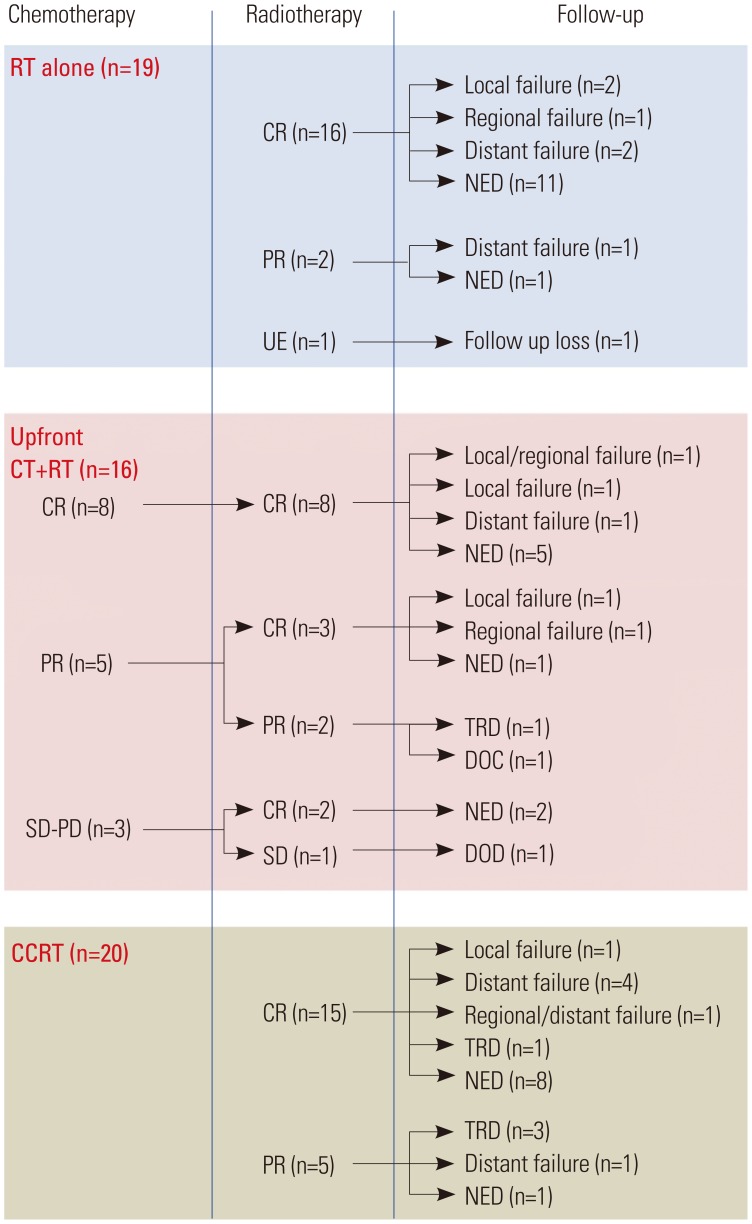
The RT response and treatment outcomes are summarized in Fig. 1. Forty-two patients (76.4%) achieved a CR, and ten (18.2%) achieved a PR. Three patients (5.4%) did not complete the RT and/or died from PD during or immediately after RT.
Fig. 1. Summary of treatment outcomes and treatment failures. RT, radiotherapy; CR, complete response; PR, partial response; UE, unevaluable; CT, chemotherapy; SD, stable disease; PD, progressive disease; NED, no evidence of disease; TRD, treatment-related death; DOC, death due to other cause; DOD, death due to disease; CCRT, concurrent chemoradiotherapy.
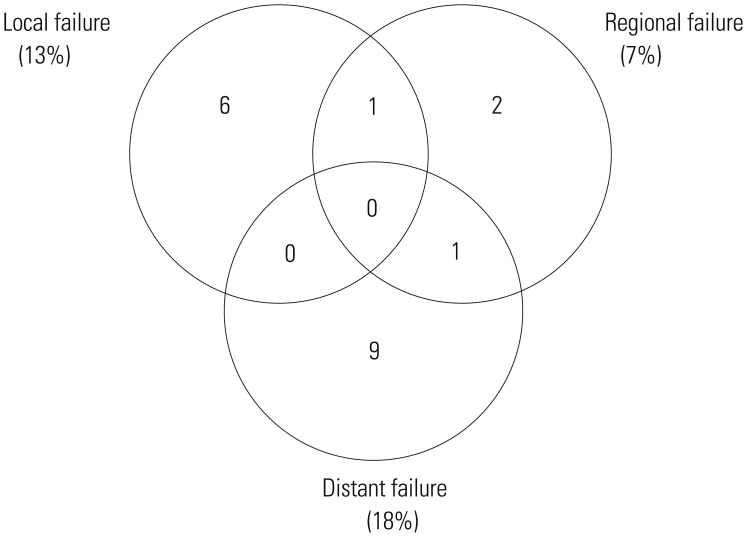
Nineteen patients experienced treatment failure (Fig. 2). Two patients had multiple failure sites and 17 had a single failure site. The most common failure was distant failure occurring in ten patients (eight had skin metastasis, one had lung metastasis, and one had para-aortic lymph node metastasis). Four patients (7%) experienced regional failure: three had stage I disease and one had stage II disease. Except for two patients who had primary tumors of the tonsil area, 37 patients with stage I disease received extended-field ISRT, and 3 of the 37 patients (8%) experienced untreated regional node failure. Of the 16 patients with stage II disease, 14 who received bilateral neck node irradiation did not experience regional failure. However, one of two patients (50%) who received involved neck node irradiation experienced untreated regional node failure.
Fig. 2. Patterns of failure.
The characteristics of seven patients (11%) who experienced local failure, comprising TR in five patients and ER in two patients, are summarized in Table 3. Five patients had stage I disease, and two patients had stage II disease. Three patients received upfront CT followed by RT, three patients received RT alone, and one patient received CCRT. Six patients received less than 50 Gy, mostly 45 Gy in 25 fx. Six of seven patients were successively salvaged, and they survived for a long period of time. Three patients were salvaged with re-irradiation (Patients 4, 6, and 7), two patients with CT (Patients 2 and 5), and one patient (Patient 1) with CT and RT. Initial RT dose/re-irradiation dose were 50.4/40, 45/34.8, and 45/44 Gy, respectively. We prescribed relatively lower doses due to the possibility of radiation toxicity; however, the results of re-irradiation were successful, and radiation toxicity did not occur. One patient (Patient 2) who was initially treated with cisplatin-based CCRT was salvaged with IMVP-16, and the other patient (Patient 5) who was initially treated with IMVP-16 and RT was salvaged with EPOCH. One patient (Patient 1) who was initially treated with IMVP-16 and RT was salvaged with EPOCH and re-irradiation of 41.4 Gy.
Table 3. Details of Patients Who Experienced Local Failure.
| # | Age | Sex | Stage | Primary site | Treatment group | RT dose | Failure site | Time to failure | Salvage treatment | Current status |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 52 | M | IA | Rt. nasal cavity | CT+RT | 45 Gy/25 fx | Rt. mandible (ER) | 6 months | CT, reRT (41.4 Gy) | NED 112 months |
| 2 | 52 | F | IA | Lt. nasal cavity | CCRT | 44 Gy/20 fx | Rt. nasal cavity (TR) | 81 months | CT | NED 87 months |
| 3 | 44 | F | IA | Rt. nasal cavity | CT+RT | 45 Gy/25 fx | Rt. nasal cavity (TR) | 33 months | CT | DOD 88 months with regional, distant failures |
| 4 | 36 | F | IA | Rt. nasal cavity | RT alone | 50.4 Gy/28 fx | Rt. nasal cavity (TR) | 65 months | reRT (40 Gy) | NED 133 months |
| 5 | 46 | M | IIA | Nasopharynx | CT+RT | 45 Gy/25 fx | Lt. nasal cavity (ER) | 9 months | CT | NED 98 months |
| 6 | 54 | M | IIA | Oropharynx | RT alone | 45 Gy/25 fx | Oropharynx (TR) | 7 months | reRT (34.8 Gy) | NED 178 months |
| 7 | 74 | F | IA | Rt. nasal cavity | RT alone | 45 Gy/25 fx | Lt. nasal cavity (ER) | 149 months | RT (44 Gy) | NED 159 months |
CT, chemotherapy; RT, radiotherapy; CCRT, concurrent chemoradiotherapy; ER, elsewhere recurrence; fx, fractions; reRT, re-radiotherapy; NED, no evidence of disease; TR, true recurrence; DOD, dead of disease.
Patterns of failure according to treatment modality are shown in Table 4. For the RT-alone group, local and distant failure occurred at the same frequency. For the CT+RT group, local failure was most common. For the CCRT group, distant failure was most common.
Table 4. Patterns of Failure According to Treatment Modality Groups.
| Patterns of failure | Treatment groups | p value | ||
|---|---|---|---|---|
| RT alone (n=19) | CT+RT (n=16) | CCRT (n=20) | ||
| Local failure | 3 (16%) | 3 (19%) | 1 (5%) | 0.361 |
| Regional node failure | 1 (5%) | 2 (13%) | 1 (5%) | 0.67 |
| Distant failure | 3 (16%) | 1 (6%) | 5 (25%) | 0.345 |
RT, radiotherapy; CT, chemotherapy; CCRT, concurrent chemoradiotherapy.
Survival analysis and prognostic factors
The median follow-up time was 26 months (range, 1–178 months) for all patients and 56 months (range, 1–178 months) for patients who were alive at the time of the analysis.
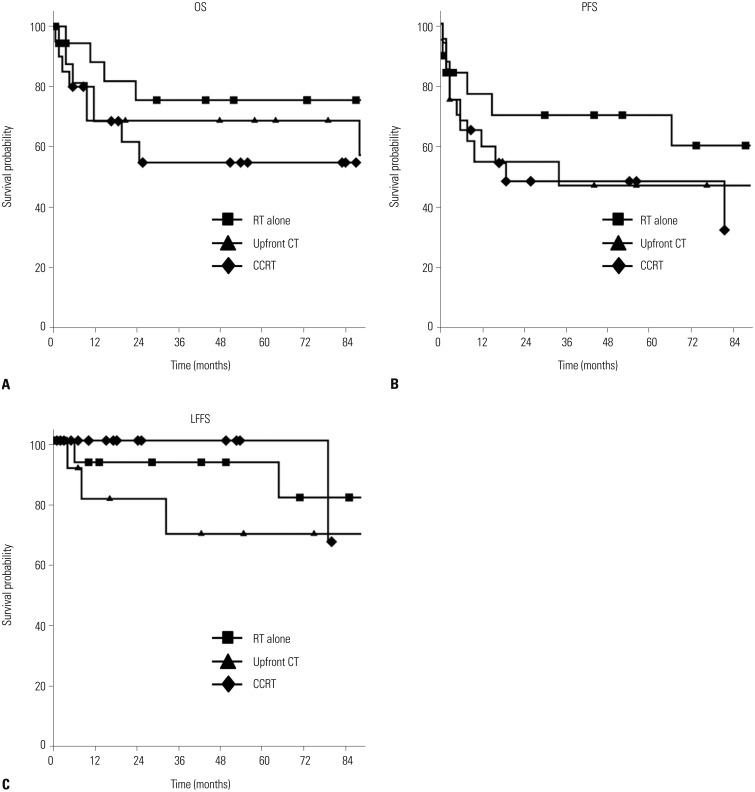
The median follow-up times for surviving patients of the RT-alone, CT+RT, and CCRT groups were 73 months (range, 1–178 months), 64.5 months (range, 3–133 months), and 38.5 months (range, 6–86 months), respectively. The 5-year OS and PFS rates for all 55 patients were 66% and 54%, respectively. The 5-year OS rate for the RT-alone, CT+RT, and CCRT groups were 76%, 69%, and 55%, respectively (p=0.388). The 5-year PFS rates for the RT-alone, CT+RT, and CCRT groups were 70%, 47%, and 48%, respectively (p=0.48). The 5-year LFFS rates for the RT-alone, CT+RT, and CCRT groups were 93%, 69%, and 100%, respectively (p=0.338) (Fig. 3).
Fig. 3. Overall survival (OS) (A), progression-free survival (PFS) (B), and local failure-free survival (LFFS) (C). RT, radiotherapy; CT, chemotherapy; CCRT, concurrent chemoradiotherapy.
The clinical and treatment factors assessed for potential prognostic impact are shown in Table 5. Among these, achieving a CR after RT was the only statistically significant prognostic factor affecting 5-year OS. Patients classified as IPI group 3 (high-intermediate) or NKPI group 4 showed a trend toward worse 5-year OS rates than those of other groups, although the differences were not statistically significant (p=0.052 and 0.434, respectively).
Table 5. Prognostic Factors for OS.
| Factor | 5-yr OS rate | p value (univariate) |
|---|---|---|
| Age (yrs) | 0.5 | |
| <54 | 72% | |
| ≥54 | 61% | |
| Sex | 0.9 | |
| Male | 66% | |
| Female | 66% | |
| Primary site (sinonasal vs. others) | 0.2 | |
| Sinonasal | 63% | |
| Other | 83% | |
| Ann Arbor stage (I vs. II) | 0.5 | |
| I | 68% | |
| II | 60% | |
| Systemic B symptoms | 0.7 | |
| Yes | 66% | |
| No | 67% | |
| Epstein-Barr virus | 0.37 | |
| Yes | 79% | |
| No | 59% | |
| RT dose | 0.3 | |
| <50 Gy | 63% | |
| ≥50 Gy | 70% | |
| RT response | 0.02 | |
| CR | 74% | |
| Non CR | 42% | |
| Treatment sequence | 0.4 | |
| RT alone | 76% | |
| Upfront CT+RT | 69% | |
| CCRT | 55% |
OS, overall survival; RT, radiotherapy; CR, complete response; CT, chemotherapy; CCRT, concurrent chemoradiotherapy.
Toxicity
Radiation-induced toxicities were minimal. Grade 1 or 2 mucositis was observed in 17 patients [31%; RT alone (2 patients), CT+RT (3), and CCRT (12)]. Grade 1 or 2 nausea or vomiting was observed in 13 patients [24%; RT alone (1 patient), CT+RT (3), and CCRT (9)]. However, a 50-year-old male patient in the CT+RT group experienced grade 4 oral mucositis during RT and withdrew from RT treatment.
Hematologic toxicities occurred during CCRT or maintenance CT. Of 36 patients who received CT, grade 1 or 2 leukopenia was observed in eight patients [22%; CT+RT (1 patient), CCRT (7)], and grade 3 or 4 leukopenia or thrombocytopenia was observed in four patients (11%) in the CCRT group. There were five treatment-related deaths [CT+RT (1 patient), CCRT (4)]. During maintenance CT, one patient in the CT+RT group died from gram-positive sepsis, whereas four patients in the CCRT group died from neutropenic fever and pneumonia aggravation.
DISCUSSION
Several studies have shown that RT alone produces a good CR rate in patients with early-stage ENKTL; however, frequent distant and local failures within 2 years of RT completion have been a major obstacle to successful treatment.2,10 Subsequent studies showed that ENKTL responded poorly to anthracycline-based chemotherapies, and patients exhibited frequent local failure and disease progression,11,12 which was thought to be due to the expression of a multidrug-resistant P-glycoprotein in ENKTL.13 With the application of dose-intensified upfront CT, the CR rate improved, although the 3-year OS and distant failure rates did not improve.14 Therefore, trials of CCRT that used CT as a radiosensitizer were designed.5,15 Although doses of >50 Gy were suggested in an RT alone setting,10 lower doses were recommended during CCRT to reduce potential toxicities. Despite these advancing treatment paradigms, there have been no conclusive studies comparing RT alone with CCRT.
This study evaluated the role of RT and CT for the treatment of ENKTL. The study cohort reflected the changing treatment paradigms over time. The RT-alone group was mostly treated between 1999 and 2004, the RT+CT group between 2002 and 2006, and the CCRT group between 2007 and 2013. Although the present study contained potential biases associated with the retrospective analysis of patients treated without a consistent protocol, the results contained herein showed a remarkable improvement in treatment outcomes compared with his-torical data for RT alone. We previously reported that 50% of patients treated with RT alone experienced local failure and that the 5-year OS rate was 40%.2,10 However, more recently, Li, et al.16 showed that only 5% of patients treated with RT alone experienced local failure and that the 5-year OS rate was 80%, findings of which are congruent with those in the present study. Here, the local failure rate of RT alone was 16% and the 5-year OS rate was 76%.
Improvements in the outcome of RT alone are likely due to the development of imaging modalities that have enabled a more complete determination of disease extent for treatment planning. IMRT and image-guided radiotherapy (IGRT) have been used recently in almost all cases involving the head and neck area, and most patients with ENKTL underwent limited or extended-field ISRT.16,17,18,19 ISRT is newly defined concept proposed by the International Lymphoma Radiation Oncology Group. ISRT encompasses the involved sites of disease based on detectable nodal and extranodal extension involvement using imaging modalities, which results in relatively smaller volumes than the larger, nodal station–derived volumes of previous extended-field and involved-field RT. In ENKTL, the volume of ISRT is similar to that of involved-field RT. In the present study, all patients underwent extended-field ISRT, and the majority of patients underwent IGRT with 3D-CRT (64%) or IMRT (16%). In addition, to determine tumor extent accurately, diagnostic MRI was fused with simulation-computed tomography.
Using these modern RT techniques and advanced imaging modalities resulted in, across the whole study cohort, seven patients (11%) experiencing local failure during follow-up and only one of these patients experiencing local failure after CCRT. However, the median follow-up period after CCRT was shorter than that of RT alone or CT+RT; therefore, a longer-term follow-up is needed to validate whether CCRT reduces local failure more than RT alone or CT+RT.
A detailed review of patients who experienced local failure after RT provides clues concerning the determination of the optimal RT dose and field. In the patients who experienced local failure, both TRs and ERs were noted. Of the four patients who experienced TRs, three received a dose of <50 Gy (Patients 2, 3, and 6). As previously reported,10 RT should be used with a tumor dose of ≥50 Gy to intensify local treatment. Despite treating two of these patients (Patients 2 and 3) with CT, the RT dose of <50 Gy appeared to be insufficient, although the addition of CT with the lower-dose RT might explain the later recurrences in Patients 2 and 3 (81 and 31 months, respectively) compared with that in Patient 6 (7 months). In the patient who received a dose of >50 Gy (Patient 4), the recurrence occurred relatively late (65 months), suggesting that higher-dose irradiation would be preferable. One patient (Patient 5) with primary disease at the nasopharynx experienced failure at the left nasal cavity, this patient was treated with CT+RT before the introduction of IMRT; thus, the radiation field encompassed only the pharynx and bilateral cervical lymph nodes yet not the nasal cavity, suggesting insufficient field coverage. One patient who had primary disease at the right nasal cavity (Patient 1) experienced failure at the right mandible, and one patient who had primary disease at the right nasal cavity (Patient 7) experienced failure at the left nasal cavity. The radiation fields in both of these patients were tightly defined in order to protect the organs at risk (both eyes and lenses). These cases of ER implied that extended-field ISRT with generous margins is needed to control early-stage ENKTL. However, our experience does not justify the need for prophylactic irradiation of uninvolved sites, and we did not accumulate enough evidence concerning the optimal radiation volume.
Salvage treatment after local failure was successful. Either a CT regimen different from that initially used or re-irradiation could be an option for salvage treatment for recurrent disease. We prescribed a relatively lower dose (<50 Gy) due to the possibility of radiation toxicity, and no radiation toxicity occurred. CT was also effective for locally recurrent disease; however, it was associated with considerable toxicities and required a longer treatment time. Using modern RT techniques such as IMRT and IGRT with advanced imaging modalities, salvage treatment with re-irradiation could be considered in cases of the local recurrent disease without distant failure.
In the present study, distant failure remained a dominant pattern of failure, despite the incorporation of CT. Wang, et al.20 reported on CT+RT consisting of gemcitabine, oxaliplatin, and L-asparaginase followed by involved-field RT, and the overall response rate was 96.3%, with 11% of patients experiencing distant failure. In addition, the Korean Lymphoma Consortium published the results of CCRT for early-stage ENKTL,5 consisting of weekly cisplatin-based CCRT followed by maintenance VIPD CT. The results were outstanding, showing an 83.3% overall response rate, an 85% 3-year OS rate, an 86% 3-year PFS rate, and a 10% distant failure rate. However, the grade IV toxicities rate was 41% during maintenance CT, and the short follow-up period provoked much controversy. In another study, the Korean Lymphoma Consortium published data concerning CCRT followed by L-asparaginase-containing maintenance CT,15 which demonstrated a 90% overall response rate, a 73% 5-year PFS rate, and a 60% 5-year OS rate with manageable toxicities. The Japan Clinical Oncology Group published a study of CCRT consisting of 50 Gy of RT with concurrent dexamethasone, etoposide, ifosfamide, and carboplatin CT,21 for which the overall response rate was 81% and the 2-year OS rate was 78%; however, the distant failure rate was 33% and not reduced by the addition of CT. In the present study, the overall distant failure rate was 18%, which was superior to the majority of the above studies for RT alone, CT+RT, and CCRT, yet remains high. A PET scan was performed in the majority of our patients, which might explain the lower distant failure rate observed.
High-risk factors for distant failure have not been determined. However, elevated expression of cyclooxygenase-2 was identified recently as a predictive factor for higher distant failure rates in an immunohistochemistry-based evaluation.22 ENKTL is generally very aggressive, and if left untreated it is uniformly fatal. Several investigators have classified ENKTL into two subgroups: nasal and extra-nasal/nasal-type, the former being characterized by locoregional aggressiveness, and the latter being mostly extra-nasal and associated with early multifocal distant dissemination.23 The high prevalence of distant failure in the present cohort might have been due to subtype heterogeneity, as eight of our 55 patients had primary disease in the extra-nasal area. However, differences in the distant failure rate between the two subtypes were not observed (data not shown). Seeking a prognostic factor to predict early distant failure is needed for patients who might benefit from the administration of CT.
The present study had several limitations. First, this was an institutional-based retrospective study of patients treated without a consistent protocol. There were differences in treatment protocols, RT modalities and doses, and CT regimens. However, this heterogeneity made it possible to speculate on the role of RT and CT for the treatment of early-stage ENKTL. Second, due to the rarity of the disease, the number of patients included was small, which made it difficult to uncover statistically significant prognostic factors. The only significant prognostic factor appeared to be achieving a CR after RT. However, patients classi-fied as IPI group 3 and NKPI group 4 showed worse OS than other groups, although the difference was not significant. There were more patients classified as IPI group 3 and NKPI group 4 in the CCRT group, and this imbalance in patients' characteristics should be noted.
In conclusion, the outcome of early-stage ENKTL has improved with advances in RT techniques. The local control rate in the RT-alone group was similar to that in patients who also underwent CT. However, systemic CT did not decrease the distant failure rate and was associated with considerable toxicities. In the era of multimodal treatment for ENKTL, RT alone using advanced techniques should be considered for local disease control, whereas maintenance treatment containing effective CT regimens should be considered for distant disease control.
Footnotes
This research was presented at the 41st annual meeting of the Korean Cancer Association with International Cancer Conference, June 19, 2015, Seoul, Korea.
The authors have no financial conflicts of interest.
References
- 1.Kim JM, Ko YH, Lee SS, Huh J, Kang CS, Kim CW, et al. WHO classification of malignant lymphomas in Korea: report of the third nationwide study. Korean J Pathol. 2011;45:254–260. [Google Scholar]
- 2.Kim GE, Cho JH, Yang WI, Chung EJ, Suh CO, Park KR, et al. Angiocentric lymphoma of the head and neck: patterns of systemic failure after radiation treatment. J Clin Oncol. 2000;18:54–63. doi: 10.1200/JCO.2000.18.1.54. [DOI] [PubMed] [Google Scholar]
- 3.Kim WS, Song SY, Ahn YC, Ko YH, Baek CH, Kim DY, et al. CHOP followed by involved field radiation: is it optimal for localized nasal natural killer/T-cell lymphoma? Ann Oncol. 2001;12:349–352. doi: 10.1023/a:1011144911781. [DOI] [PubMed] [Google Scholar]
- 4.You JY, Chi KH, Yang MH, Chen CC, Ho CH, Chau WK, et al. Radiation therapy versus chemotherapy as initial treatment for localized nasal natural killer (NK)/T-cell lymphoma: a single institute survey in Taiwan. Ann Oncol. 2004;15:618–625. doi: 10.1093/annonc/mdh143. [DOI] [PubMed] [Google Scholar]
- 5.Kim SJ, Kim K, Kim BS, Kim CY, Suh C, Huh J, et al. Phase II trial of concurrent radiation and weekly cisplatin followed by VIPD chemotherapy in newly diagnosed, stage IE to IIE, nasal, extranodal NK/T-Cell Lymphoma: Consortium for Improving Survival of Lymphoma study. J Clin Oncol. 2009;27:6027–6032. doi: 10.1200/JCO.2009.23.8592. [DOI] [PubMed] [Google Scholar]
- 6.Lee J, Suh C, Park YH, Ko YH, Bang SM, Lee JH, et al. Extranodal natural killer T-cell lymphoma, nasal-type: a prognostic model from a retrospective multicenter study. J Clin Oncol. 2006;24:612–618. doi: 10.1200/JCO.2005.04.1384. [DOI] [PubMed] [Google Scholar]
- 7.Illidge T, Specht L, Yahalom J, Aleman B, Berthelsen AK, Constine L, et al. Modern radiation therapy for nodal non-Hodgkin lymphoma-target definition and dose guidelines from the International Lymphoma Radiation Oncology Group. Int J Radiat Oncol Biol Phys. 2014;89:49–58. doi: 10.1016/j.ijrobp.2014.01.006. [DOI] [PubMed] [Google Scholar]
- 8.Specht L, Yahalom J, Illidge T, Berthelsen AK, Constine LS, Eich HT, et al. Modern radiation therapy for Hodgkin lymphoma: field and dose guidelines from the international lymphoma radiation oncology group (ILROG) Int J Radiat Oncol Biol Phys. 2014;89:854–862. doi: 10.1016/j.ijrobp.2013.05.005. [DOI] [PubMed] [Google Scholar]
- 9.Prescribing, Recording, and Reporting Photon-Beam Intensity-Modulated Radiation Therapy (IMRT): Contents. J ICRU. 2010;10:NP. [Google Scholar]
- 10.Koom WS, Chung EJ, Yang WI, Shim SJ, Suh CO, Roh JK, et al. Angiocentric T-cell and NK/T-cell lymphomas: radiotherapeutic viewpoints. Int J Radiat Oncol Biol Phys. 2004;59:1127–1137. doi: 10.1016/j.ijrobp.2003.12.006. [DOI] [PubMed] [Google Scholar]
- 11.Kim BS, Kim TY, Kim CW, Kim JY, Heo DS, Bang YJ, et al. Therapeutic outcome of extranodal NK/T-cell lymphoma initially treated with chemotherapy--result of chemotherapy in NK/T-cell lymphoma. Acta Oncol. 2003;42:779–783. doi: 10.1080/02841860310010682. [DOI] [PubMed] [Google Scholar]
- 12.Kim SJ, Kim BS, Choi CW, Seo HY, Seol HR, Sung HJ, et al. Treatment outcome of front-line systemic chemotherapy for localized extranodal NK/T cell lymphoma in nasal and upper aerodigestive tract. Leuk Lymphoma. 2006;47:1265–1273. doi: 10.1080/10428190600565651. [DOI] [PubMed] [Google Scholar]
- 13.Drénou B, Lamy T, Amiot L, Fardel O, Caulet-Maugendre S, Sasportes M, et al. CD3- CD56+ non-Hodgkin's lymphomas with an aggressive behavior related to multidrug resistance. Blood. 1997;89:2966–2974. [PubMed] [Google Scholar]
- 14.Lee SH, Ahn YC, Kim WS, Ko YH, Kim K, Park K. The effect of preirradiation dose intense CHOP on anthracyline resistance in localized nasal NK/T-cell lymphoma. Haematologica. 2006;91:427–428. [PubMed] [Google Scholar]
- 15.Kim SJ, Yang DH, Kim JS, Kwak JY, Eom HS, Hong DS, et al. Concurrent chemoradiotherapy followed by L-asparaginase-containing chemotherapy, VIDL, for localized nasal extranodal NK/T cell lymphoma: CISL08-01 phase II study. Ann Hematol. 2014;93:1895–1901. doi: 10.1007/s00277-014-2137-6. [DOI] [PubMed] [Google Scholar]
- 16.Li YX, Wang H, Jin J, Wang WH, Liu QF, Song YW, et al. Radiotherapy alone with curative intent in patients with stage I extranodal nasal-type NK/T-cell lymphoma. Int J Radiat Oncol Biol Phys. 2012;82:1809–1815. doi: 10.1016/j.ijrobp.2010.10.040. [DOI] [PubMed] [Google Scholar]
- 17.Isobe K, Uno T, Tamaru J, Kawakami H, Ueno N, Wakita H, et al. Extranodal natural killer/T-cell lymphoma, nasal type: the significance of radiotherapeutic parameters. Cancer. 2006;106:609–615. doi: 10.1002/cncr.21656. [DOI] [PubMed] [Google Scholar]
- 18.Li YX, Liu QF, Wang WH, Jin J, Song YW, Wang SL, et al. Failure patterns and clinical implications in early stage nasal natural killer/T-cell lymphoma treated with primary radiotherapy. Cancer. 2011;117:5203–5211. doi: 10.1002/cncr.26167. [DOI] [PubMed] [Google Scholar]
- 19.Wang H, Li YX, Wang WH, Jin J, Dai JR, Wang SL, et al. Mild toxicity and favorable prognosis of high-dose and extended involved-field intensity-modulated radiotherapy for patients with early+stage nasal NK/T-cell lymphoma. Int J Radiat Oncol Biol Phys. 2012;82:1115–1121. doi: 10.1016/j.ijrobp.2011.02.039. [DOI] [PubMed] [Google Scholar]
- 20.Wang L, Wang ZH, Chen XQ, Li YJ, Wang KF, Xia YF, et al. First-line combination of gemcitabine, oxaliplatin, and L-asparaginase (GELOX) followed by involved-field radiation therapy for patients with stage IE/IIE extranodal natural killer/T-cell lymphoma. Cancer. 2013;119:348–355. doi: 10.1002/cncr.27752. [DOI] [PubMed] [Google Scholar]
- 21.Yamaguchi M, Tobinai K, Oguchi M, Ishizuka N, Kobayashi Y, Isobe Y, et al. Phase I/II study of concurrent chemoradiotherapy for localized nasal natural killer/T-cell lymphoma: Japan Clinical Oncology Group Study JCOG0211. J Clin Oncol. 2009;27:5594–5600. doi: 10.1200/JCO.2009.23.8295. [DOI] [PubMed] [Google Scholar]
- 22.Shim SJ, Yang WI, Shin E, Koom WS, Kim YB, Cho JH, et al. Clinical significance of cyclooxygenase-2 expression in extranodal natural killer (NK)/T-cell lymphoma, nasal type. Int J Radiat Oncol Biol Phys. 2007;67:31–38. doi: 10.1016/j.ijrobp.2006.07.1387. [DOI] [PubMed] [Google Scholar]
- 23.Kwong YL. Natural killer-cell malignancies: diagnosis and treatment. Leukemia. 2005;19:2186–2194. doi: 10.1038/sj.leu.2403955. [DOI] [PubMed] [Google Scholar]