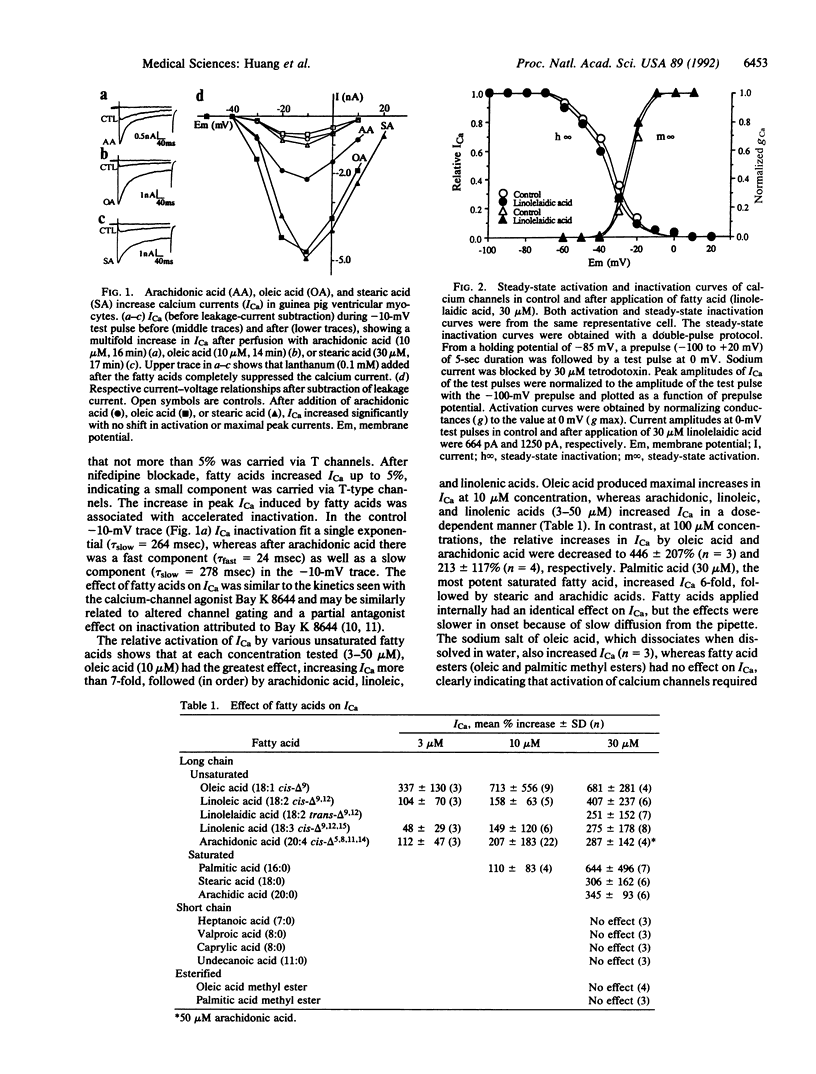
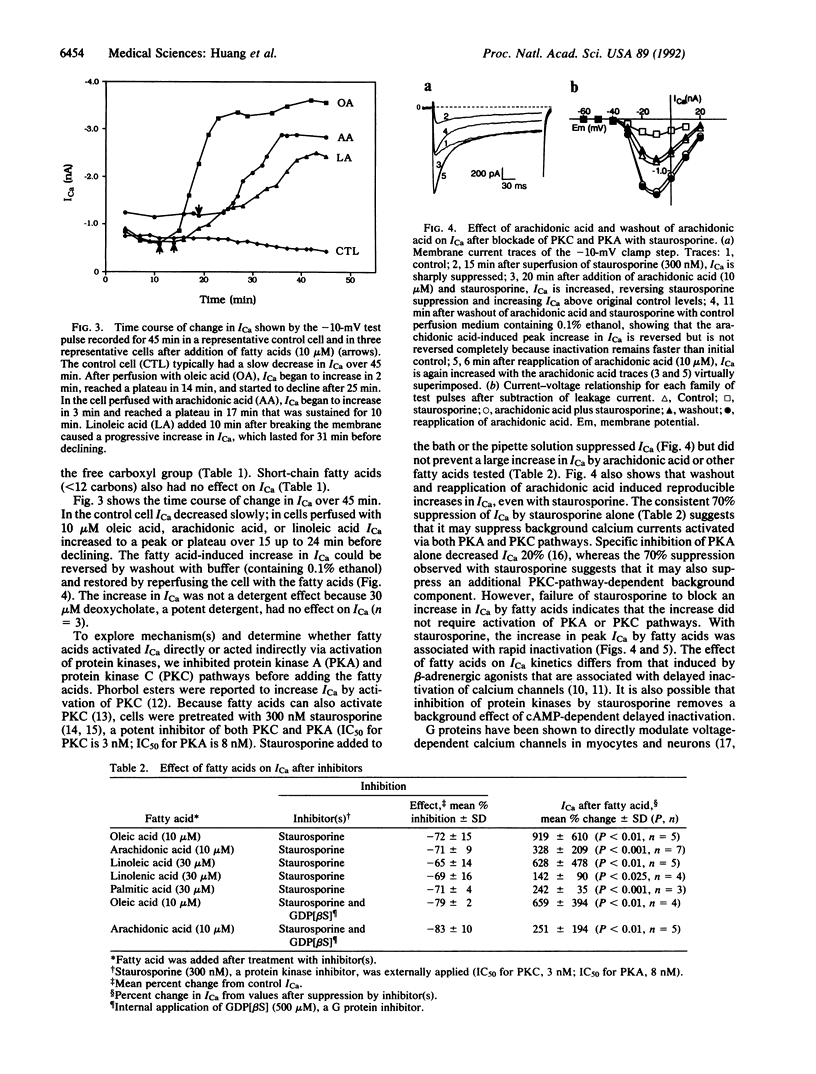
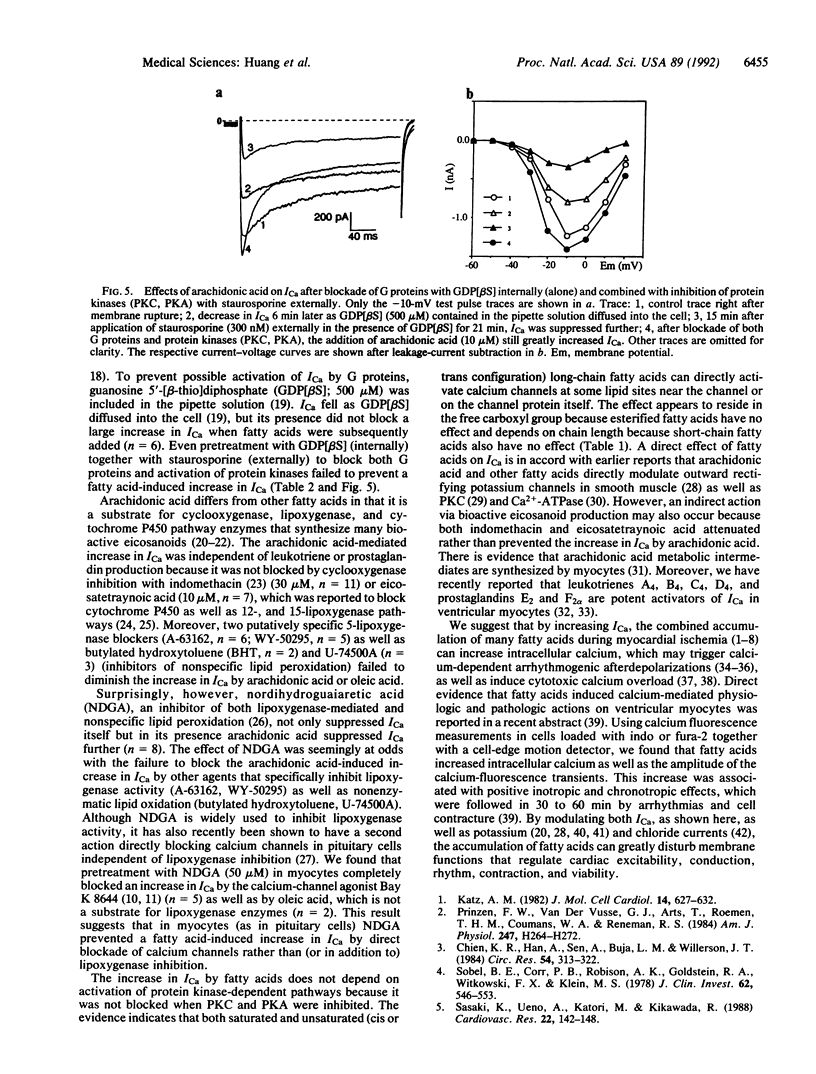
Abstract
Nonesterified fatty acids accumulate at sites of tissue injury and necrosis. In cardiac tissue the concentrations of oleic acid, arachidonic acid, leukotrienes, and other fatty acids increase greatly during ischemia due to receptor or nonreceptor-mediated activation of phospholipases and/or diminished reacylation. In ischemic myocardium, the time course of increase in fatty acids and tissue calcium closely parallels irreversible cardiac damage. We postulated that fatty acids released from membrane phospholipids may be involved in the increase of intracellular calcium. We report here that low concentrations (3-30 microM) of each long-chain unsaturated (oleic, linoleic, linolenic, and arachidonic) and saturated (palmitic, stearic, and arachidic) fatty acid tested induced multifold increases in voltage-dependent calcium currents (ICa) in cardiac myocytes. In contrast, neither short-chain fatty acids (less than 12 carbons) or fatty acid esters (oleic and palmitic methyl esters) had any effect on ICa, indicating that activation of calcium channels depended on chain length and required a free carboxyl group. Inhibition of protein kinases C and A, G proteins, eicosanoid production, or nonenzymatic oxidation did not block the fatty acid-induced increase in ICa. Thus, long-chain fatty acids appear to directly activate ICa, possibly by acting at some lipid sites near the channels or directly on the channel protein itself. We suggest that the combined effects of fatty acids released during ischemia on ICa may contribute to ischemia-induced pathogenic events on the heart that involve calcium, such as arrhythmias, conduction disturbances, and myocardial damage due to cytotoxic calcium overload.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Borgeat P., Hamberg M., Samuelsson B. Transformation of arachidonic acid and homo-gamma-linolenic acid by rabbit polymorphonuclear leukocytes. Monohydroxy acids from novel lipoxygenases. J Biol Chem. 1976 Dec 25;251(24):7816–7820. [PubMed] [Google Scholar]
- Canonico P. L., Schettini G., Valdenegro C. A., MacLeod R. M. Arachidonic acid metabolism and prolactin secretion in vitro: a possible role for the lipoxygenase products. Neuroendocrinology. 1983 Sep;37(3):212–217. doi: 10.1159/000123545. [DOI] [PubMed] [Google Scholar]
- Capdevila J., Gil L., Orellana M., Marnett L. J., Mason J. I., Yadagiri P., Falck J. R. Inhibitors of cytochrome P-450-dependent arachidonic acid metabolism. Arch Biochem Biophys. 1988 Mar;261(2):257–263. doi: 10.1016/0003-9861(88)90340-2. [DOI] [PubMed] [Google Scholar]
- Chien K. R., Han A., Sen A., Buja L. M., Willerson J. T. Accumulation of unesterified arachidonic acid in ischemic canine myocardium. Relationship to a phosphatidylcholine deacylation-reacylation cycle and the depletion of membrane phospholipids. Circ Res. 1984 Mar;54(3):313–322. doi: 10.1161/01.res.54.3.313. [DOI] [PubMed] [Google Scholar]
- Fedida D., Noble D., Shimoni Y., Spindler A. J. Inward current related to contraction in guinea-pig ventricular myocytes. J Physiol. 1987 Apr;385:565–589. doi: 10.1113/jphysiol.1987.sp016508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fish R. D., Sperti G., Colucci W. S., Clapham D. E. Phorbol ester increases the dihydropyridine-sensitive calcium conductance in a vascular smooth muscle cell line. Circ Res. 1988 May;62(5):1049–1054. doi: 10.1161/01.res.62.5.1049. [DOI] [PubMed] [Google Scholar]
- Franson R. C., Pang D. C., Weglicki W. B. Modulation of lipolytic activity in isolated canine cardiac sarcolemma by isoproterenol and propranolol. Biochem Biophys Res Commun. 1979 Oct 12;90(3):956–962. doi: 10.1016/0006-291x(79)91920-x. [DOI] [PubMed] [Google Scholar]
- Gunn M. D., Sen A., Chang A., Willerson J. T., Buja L. M., Chien K. R. Mechanisms of accumulation of arachidonic acid in cultured myocardial cells during ATP depletion. Am J Physiol. 1985 Dec;249(6 Pt 2):H1188–H1194. doi: 10.1152/ajpheart.1985.249.6.H1188. [DOI] [PubMed] [Google Scholar]
- Hescheler J., Rosenthal W., Trautwein W., Schultz G. The GTP-binding protein, Go, regulates neuronal calcium channels. 1987 Jan 29-Feb 4Nature. 325(6103):445–447. doi: 10.1038/325445a0. [DOI] [PubMed] [Google Scholar]
- Hwang T. C., Guggino S. E., Guggino W. B. Direct modulation of secretory chloride channels by arachidonic and other cis unsaturated fatty acids. Proc Natl Acad Sci U S A. 1990 Aug;87(15):5706–5709. doi: 10.1073/pnas.87.15.5706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kameyama M., Hescheler J., Hofmann F., Trautwein W. Modulation of Ca current during the phosphorylation cycle in the guinea pig heart. Pflugers Arch. 1986 Aug;407(2):123–128. doi: 10.1007/BF00580662. [DOI] [PubMed] [Google Scholar]
- Katz A. M. Membrane-derived lipids and the pathogenesis of ischemic myocardial damage. J Mol Cell Cardiol. 1982 Nov;14(11):627–632. doi: 10.1016/0022-2828(82)90160-2. [DOI] [PubMed] [Google Scholar]
- Kim D., Clapham D. E. Potassium channels in cardiac cells activated by arachidonic acid and phospholipids. Science. 1989 Jun 9;244(4909):1174–1176. doi: 10.1126/science.2727703. [DOI] [PubMed] [Google Scholar]
- Kim R. S., LaBella F. S. The effect of linoleic and arachidonic acid derivatives on calcium transport in vesicles from cardiac sarcoplasmic reticulum. J Mol Cell Cardiol. 1988 Feb;20(2):119–130. doi: 10.1016/s0022-2828(88)80025-7. [DOI] [PubMed] [Google Scholar]
- Korn S. J., Horn R. Nordihydroguaiaretic acid inhibits voltage-activated Ca2+ currents independently of lipoxygenase inhibition. Mol Pharmacol. 1990 Oct;38(4):524–530. [PubMed] [Google Scholar]
- Kurachi Y., Ito H., Sugimoto T., Shimizu T., Miki I., Ui M. Arachidonic acid metabolites as intracellular modulators of the G protein-gated cardiac K+ channel. Nature. 1989 Feb 9;337(6207):555–557. doi: 10.1038/337555a0. [DOI] [PubMed] [Google Scholar]
- Lewis R. A., Austen K. F., Soberman R. J. Leukotrienes and other products of the 5-lipoxygenase pathway. Biochemistry and relation to pathobiology in human diseases. N Engl J Med. 1990 Sep 6;323(10):645–655. doi: 10.1056/NEJM199009063231006. [DOI] [PubMed] [Google Scholar]
- Mechmann S., Pott L. Identification of Na-Ca exchange current in single cardiac myocytes. Nature. 1986 Feb 13;319(6054):597–599. doi: 10.1038/319597a0. [DOI] [PubMed] [Google Scholar]
- Mitra R., Morad M. A uniform enzymatic method for dissociation of myocytes from hearts and stomachs of vertebrates. Am J Physiol. 1985 Nov;249(5 Pt 2):H1056–H1060. doi: 10.1152/ajpheart.1985.249.5.H1056. [DOI] [PubMed] [Google Scholar]
- Murakami K., Chan S. Y., Routtenberg A. Protein kinase C activation by cis-fatty acid in the absence of Ca2+ and phospholipids. J Biol Chem. 1986 Nov 25;261(33):15424–15429. [PubMed] [Google Scholar]
- Mészàros J., Pappano A. J. Electrophysiological effects of L-palmitoylcarnitine in single ventricular myocytes. Am J Physiol. 1990 Apr;258(4 Pt 2):H931–H938. doi: 10.1152/ajpheart.1990.258.4.H931. [DOI] [PubMed] [Google Scholar]
- Ordway R. W., Walsh J. V., Jr, Singer J. J. Arachidonic acid and other fatty acids directly activate potassium channels in smooth muscle cells. Science. 1989 Jun 9;244(4909):1176–1179. doi: 10.1126/science.2471269. [DOI] [PubMed] [Google Scholar]
- Piomelli D., Volterra A., Dale N., Siegelbaum S. A., Kandel E. R., Schwartz J. H., Belardetti F. Lipoxygenase metabolites of arachidonic acid as second messengers for presynaptic inhibition of Aplysia sensory cells. Nature. 1987 Jul 2;328(6125):38–43. doi: 10.1038/328038a0. [DOI] [PubMed] [Google Scholar]
- Prinzen F. W., Van der Vusse G. J., Arts T., Roemen T. H., Coumans W. A., Reneman R. S. Accumulation of nonesterified fatty acids in ischemic canine myocardium. Am J Physiol. 1984 Aug;247(2 Pt 2):H264–H272. doi: 10.1152/ajpheart.1984.247.2.H264. [DOI] [PubMed] [Google Scholar]
- Revtyak G. E., Buja L. M., Chien K. R., Campbell W. B. Reduced arachidonate metabolism in ATP-depleted myocardial cells occurs early in cell injury. Am J Physiol. 1990 Aug;259(2 Pt 2):H582–H591. doi: 10.1152/ajpheart.1990.259.2.H582. [DOI] [PubMed] [Google Scholar]
- Roth D. M., Lefer D. J., Hock C. E., Lefer A. M. Effects of peptide leukotrienes on cardiac dynamics in rat, cat, and guinea pig hearts. Am J Physiol. 1985 Sep;249(3 Pt 2):H477–H484. doi: 10.1152/ajpheart.1985.249.3.H477. [DOI] [PubMed] [Google Scholar]
- Rüegg U. T., Burgess G. M. Staurosporine, K-252 and UCN-01: potent but nonspecific inhibitors of protein kinases. Trends Pharmacol Sci. 1989 Jun;10(6):218–220. doi: 10.1016/0165-6147(89)90263-0. [DOI] [PubMed] [Google Scholar]
- Sanguinetti M. C., Kass R. S. Voltage-dependent block of calcium channel current in the calf cardiac Purkinje fiber by dihydropyridine calcium channel antagonists. Circ Res. 1984 Sep;55(3):336–348. doi: 10.1161/01.res.55.3.336. [DOI] [PubMed] [Google Scholar]
- Sasaki K., Ueno A., Katori M., Kikawada R. Detection of leukotriene B4 in cardiac tissue and its role in infarct extension through leucocyte migration. Cardiovasc Res. 1988 Feb;22(2):142–148. doi: 10.1093/cvr/22.2.142. [DOI] [PubMed] [Google Scholar]
- Seifert R., Schächtele C., Rosenthal W., Schultz G. Activation of protein kinase C by cis- and trans-fatty acids and its potentiation by diacylglycerol. Biochem Biophys Res Commun. 1988 Jul 15;154(1):20–26. doi: 10.1016/0006-291x(88)90643-2. [DOI] [PubMed] [Google Scholar]
- Shuba Y. M., Hesslinger B., Trautwein W., McDonald T. F., Pelzer D. Whole-cell calcium current in guinea-pig ventricular myocytes dialysed with guanine nucleotides. J Physiol. 1990 May;424:205–228. doi: 10.1113/jphysiol.1990.sp018063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sobel B. E., Corr P. B., Robison A. K., Goldstein R. A., Witkowski F. X., Klein M. S. Accumulation of lysophosphoglycerides with arrhythmogenic properties in ischemic myocardium. J Clin Invest. 1978 Sep;62(3):546–553. doi: 10.1172/JCI109159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamaoki T., Nomoto H., Takahashi I., Kato Y., Morimoto M., Tomita F. Staurosporine, a potent inhibitor of phospholipid/Ca++dependent protein kinase. Biochem Biophys Res Commun. 1986 Mar 13;135(2):397–402. doi: 10.1016/0006-291x(86)90008-2. [DOI] [PubMed] [Google Scholar]
- Toyo-oka T., Hara K., Nakamura N., Kitahara M., Masaki T. Ca overload and the action of calcium sensitive proteases, phospholipases and prostaglandin E2 in myocardial cell degradation. Basic Res Cardiol. 1985 May-Jun;80(3):303–315. doi: 10.1007/BF01907906. [DOI] [PubMed] [Google Scholar]
- Tsien R. W., Bean B. P., Hess P., Lansman J. B., Nilius B., Nowycky M. C. Mechanisms of calcium channel modulation by beta-adrenergic agents and dihydropyridine calcium agonists. J Mol Cell Cardiol. 1986 Jul;18(7):691–710. doi: 10.1016/s0022-2828(86)80941-5. [DOI] [PubMed] [Google Scholar]
- Weglicki W. B., Waite B. M., Stam A. C., Jr Association of phospholipase A with a myocardial membrane preparation containing the (Na + +K + )-Mg 2+ -ATPase. J Mol Cell Cardiol. 1972 Jun;4(3):195–201. doi: 10.1016/0022-2828(72)90057-0. [DOI] [PubMed] [Google Scholar]
- Wier W. G., Cannell M. B., Berlin J. R., Marban E., Lederer W. J. Cellular and subcellular heterogeneity of [Ca2+]i in single heart cells revealed by fura-2. Science. 1987 Jan 16;235(4786):325–328. doi: 10.1126/science.3798114. [DOI] [PubMed] [Google Scholar]
- Yatani A., Codina J., Imoto Y., Reeves J. P., Birnbaumer L., Brown A. M. A G protein directly regulates mammalian cardiac calcium channels. Science. 1987 Nov 27;238(4831):1288–1292. doi: 10.1126/science.2446390. [DOI] [PubMed] [Google Scholar]



