Abstract
As the recent outbreaks in Edinburgh and Camarthen, UK, have shown, Legionella pneumonia (LP) remains a significant public health problem, which is not only confined to those who have travelled abroad. In both outbreaks and sporadic cases, diagnosis can go unrecognised. We reviewed the demographics, comorbidities, diagnosis, treatment and clinical outcome of LP cases over five years in a district general hospital in northwest England. Over half of LP cases were UK acquired and ‘classic’ clinical features were common. Clinical criteria for diagnosing LP were confirmed, but few sputum samples were sent to reference laboratories, limiting further essential epidemiological mapping of UK cases. Following current UK community-acquired pneumonia guidance would have missed nearly one quarter of LP cases in our series, potentially leading to further morbidity and mortality.
Key Words: descriptive, atypical, Legionella, pneumonia, CURB-65, outcome
Introduction
Specific guidance on the investigation and early diagnosis of Legionella pneumonia (LP) is limited despite this infection comprising nearly 7% of all community-acquired pneumonias (CAP) in some series1 and up to 13% of pneumonias dealt with in outpatient settings.2 Rates of LP are increasing3 and it continues to cause high morbidity and mortality.4 Recent LP outbreaks, such as those in southwest Edinburgh and Camarthen, have been associated with acquisition of infection within the UK and are a stark reminder of the severity of the disease. UK Health Protection Agency (HPA) data have shown that, over the past 31 years (1980–2011), just over half of UK LP cases were associated with recent travel, mainly overseas.5 However, a travel history alone has little predictive value. Moreover, although presentation can include ‘classic’ features of atypical pneumonia, these features are not sufficient to accurately differentiate LP from pneumonia of other aetiology.6
Rapid testing for LP using Legionella urinary antigen (LUA) is now widely available and reporting of positive results to local public health authorities is not only recommended but is also a legal requirement.6 The CURB-65 score7 enables severity assessment and can help optimise antibiotic therapy.
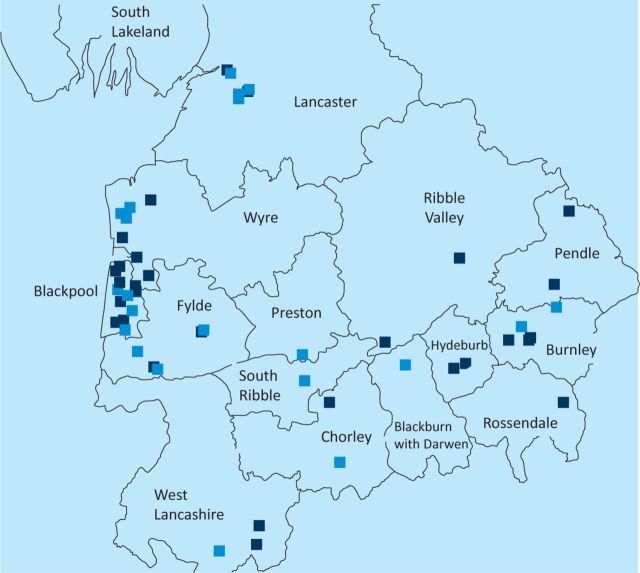
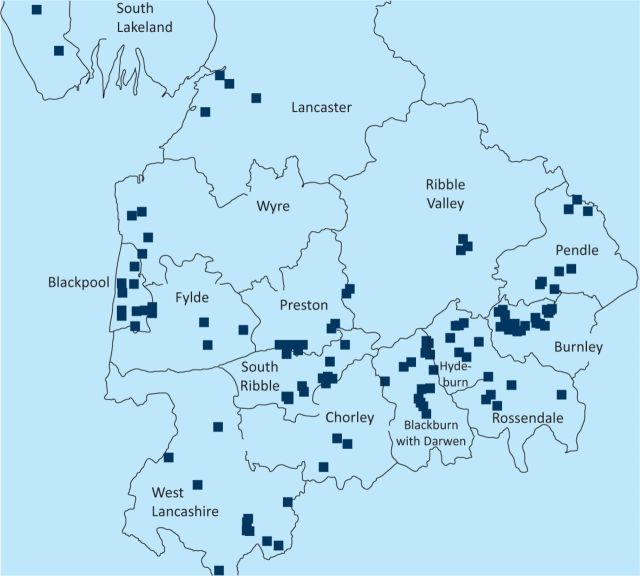
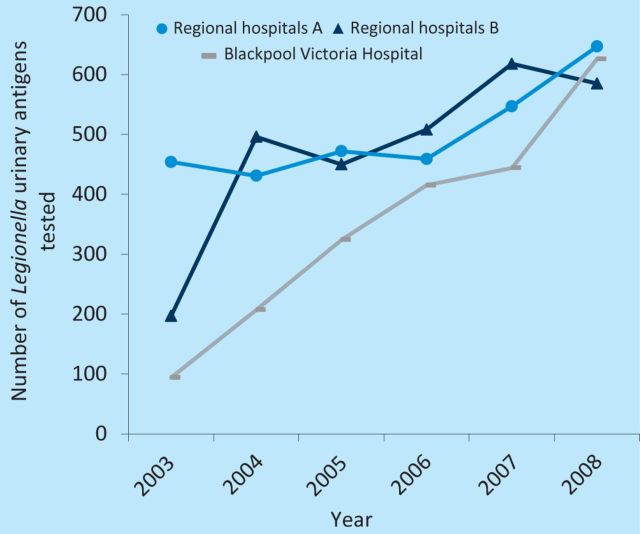
In 2011, in a report from the Cumbria and Lancashire Health Protection Unit of the HPA to the Department of Microbiology of Blackpool Teaching Hospitals, an uneven distribution of LP was noted in the Cumbria and Lancashire region, with clustering of 44% of total cases in the Blackpool area between 2001 and 2009 (Fig 1). This clustering did not appear to be either a UK-acquired community outbreak or related to the location of registered wet cooling systems (also known as industrial water coolants; Fig 2). When compared with other trusts serving similar-sized populations in the north west region, no increased frequency of LUA testing was found in Blackpool Teaching Hospitals that could have accounted for the apparent clustering of cases (Fig 3).
Fig 1.
Geographical distribution of Legionella cases in northwest England, 2001–2009. Dark blue squares represent travel-associated cases, light blue squares represent community-acquired cases. © Crown copyright and database rights 2012, Ordnance Survey, 100016969.
Fig 2.
Geographical distribution of wet cooling systems in northwest England, 2001–2009. © Crown copyright and database rights 2012, Ordnance Survey, 100016969.
Fig 3.
Number of Legionella urinary antigen (LUA) tests per regional hospital. Regional hospitals A (dark blue line) and B (light blue line) serve populations of a similar size to Blackpool Victoria Hospital (grey line). Data courtesy of the Cumbria and Lancashire Health Protection Unit.
An observational, descriptive, retrospective analysis of all LP cases over the preceding five years was undertaken to examine the apparent clustering of cases in north-west England.
Methods
In total, 26 LP cases with a positive LUA test (BinaxNOW, Inverness Medical, UK) were identified from the pathology database of the Blackpool Teaching Hospitals between July 2006 and July 2011. This time period was chosen because microbiological reporting was standardised to the computerised pathology database from 2006 onwards.
Case-note review was undertaken to gather specific data on demographics, comorbidities, foreign travel history, clinical and microbiological findings, diagnosis, treatment and clinical outcome (Table 1). In addition, information on dates of symptom onset and delay before admission was collated from HPA data.
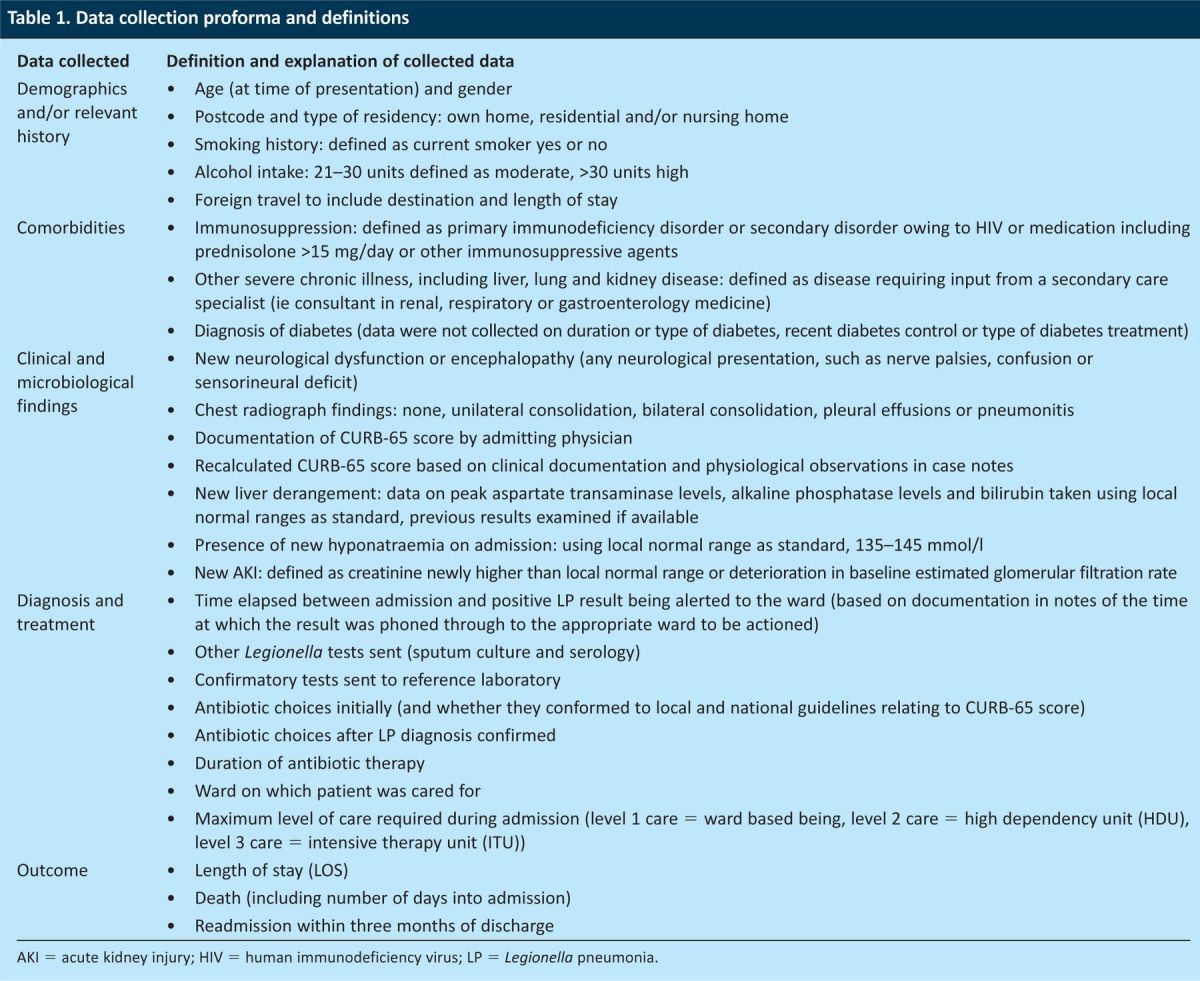
Table 1.
Data collection proforma and definitions
Statistical analysis involved comparisons of means using unpaired t-test for parametric data and Mann-Whitney U test for nonparametric data. Additional simple univariate logistic regression was performed using the STATA program (version 12).
Results
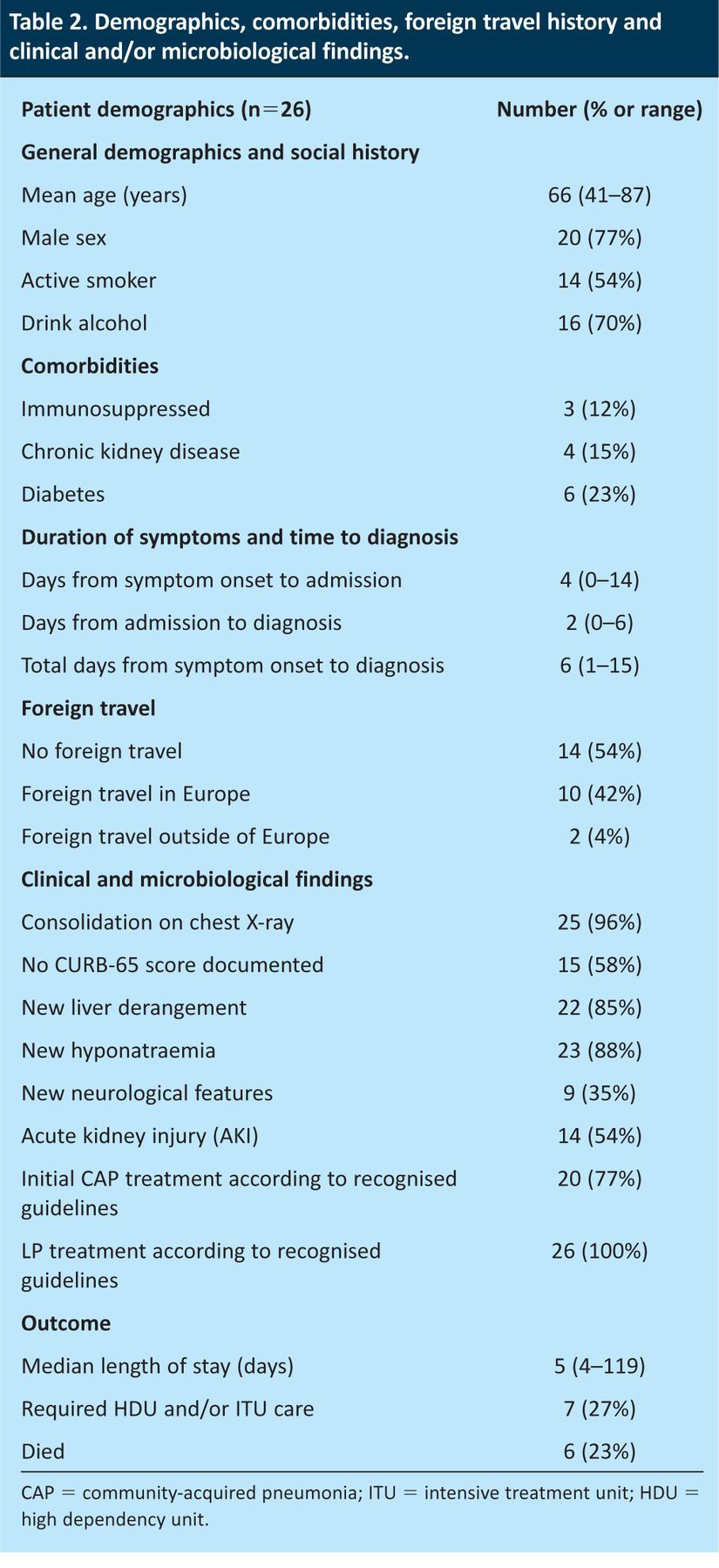
Demographics, history, comorbidity, duration of symptoms before admission and diagnosis, clinical findings, microbiological findings and outcome are summarised in Table 2.
Table 2.
Demographics, comorbidities, foreign travel history and clinical and/or microbiological findings.
Demographics
Of the patients, 20/26 (77%) were men, with a mean age of 66 years (median 66, range 41–87). Women had a higher mean age than men, but this was not significant (women 72 years, 95% confidence interval (CI) 60–87 vs men 63 years, 95% CI 57–68, p=0.1).
Of those that acquired LP in the UK, all residences were defined as ‘own home’ and no clustering was found between these postcodes.
Over half of the patients were current smokers, 14/26 (54%). Of those patients with documented data available, over two-thirds drank alcohol, 16/23 (70%), with over one third being moderate–heavy drinkers (alcohol unit intake >30 units per week).
Comorbidity
Three of the 26 patients (12%) were immunosuppressed: two patients were taking long-term steroids >15 mg daily and another patient with severe rheumatoid arthritis was taking adalimumab (an antitumour necrosis factor agent) and weekly methotrexate. In addition, four of the 26 patients (15%) had existing chronic kidney injury (CKI), with one of these patients being on regular dialysis before admission. Six of the 26 patients (23%) had diabetes.
Foreign travel
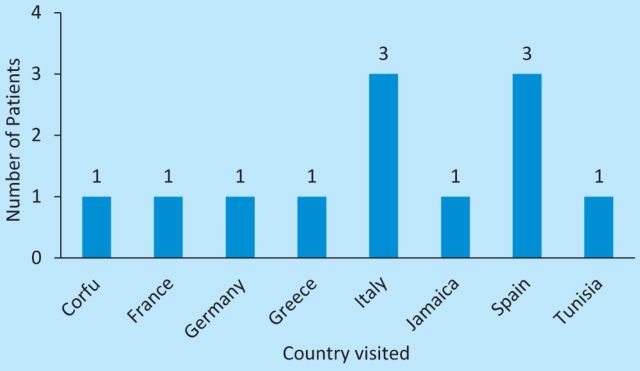
In total, 12/26 (46%) patients reported foreign travel within the previous month: 10/12 within Europe, with Italy and Spain being the most commonly visited destinations. Two patients had travelled beyond Europe (Fig 4).
Fig 4.
Destination of those patients with a relevant travel history (n=12).
Clinical and microbiological findings
In total, 96% (25/26) of patients had consolidation on chest X-ray: this was unilateral in 22/25 patients (88%) and bilateral in 3/25 (12%) patients. No patients had radiographic evidence of pleural effusions or pneumonitis.
Initial CURB-65 score was not documented in 15/26 patients (58%). Of the documented initial CURB-65 scores, the median was 3 (5/11, range 0–4). For those without a documented initial CURB-65 score, their score was subsequently calculated by the investigators using clinical documentation, blood results and physiological observations from case notes (see Discussion).
Of the 26 patients, 22 (85%) had new liver derangement, mainly hepatitic with an isolated increase in aspartate transaminase (AST).
Mean serum sodium (Na) at diagnosis was 129.9 mmol/l (range 122–136), with 23/26 patients (88%) showing some degree of biochemical hyponatraemia. In addition, 9/26 (35%) of the patients had new-onset confusion or other neurological features. Mean serum Na in those with neurological features was 128.1 mmol/l (n=9) and 131.7 mmol/l in those without neurological features (n=17); however, this trend was not statistically significant (p=0.09).
Diagnosis and treatment
Patients had a mean of four days between symptom onset and admission to hospital (range 0–14 days). LUA was identified a mean two days into admission (range 0–6 days), with results being phoned by laboratory scientists to the ward directly in all cases. The full time and date of the result given by telephone was documented in notes from 23/26 (88%) patients. The mean total number of days between symptom onset and diagnosis was six (range 1–15).
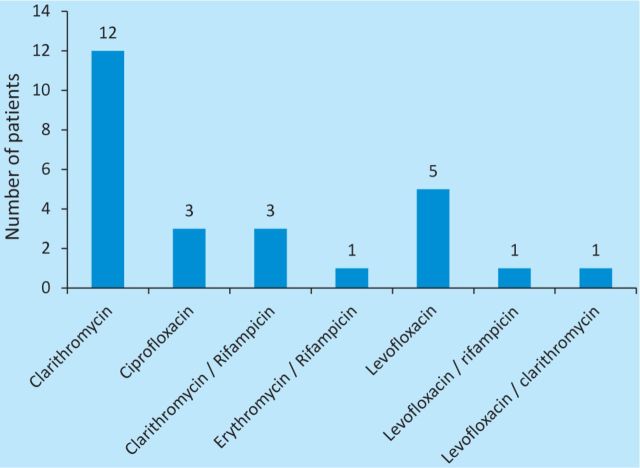
Antibiotic prescriptions in 6/26 patients (23%) did not conform to local or national guidance, with these patients receiving additional antibiotic groups or broader spectrum antibiotic cover than recommended at lower CURB-65 scores (data not shown). Of those who survived until discharge, 20/20 (100%) received appropriate antibiotic cover for LP (defined as 10–14 days of appropriate dose, route and choice of macrolide or quinolone). Antibiotic regimens after LP diagnosis are shown in Fig 5.
Fig 5.
Choice of antibiotic for Legionella cover after diagnosis (n=26). Sputum samples were collected for reference laboratory confirmation in 5/26 patients (19%).
Outcome
The median length of stay (LOS) was five days (range 4–119, mean 18 days). Of the 26 patients, 15 (58%) were treated on a specific respiratory medicine ward. In addition, 7/26 (27%) of the patients required care in a high dependency (HDU) or intensive treatment (ITU) unit. Of the 26 patients, six (23%) died, five of whom died within the first five days of admission. Two of these patients died on ITU, one on HDU, and the remaining two on respiratory wards.
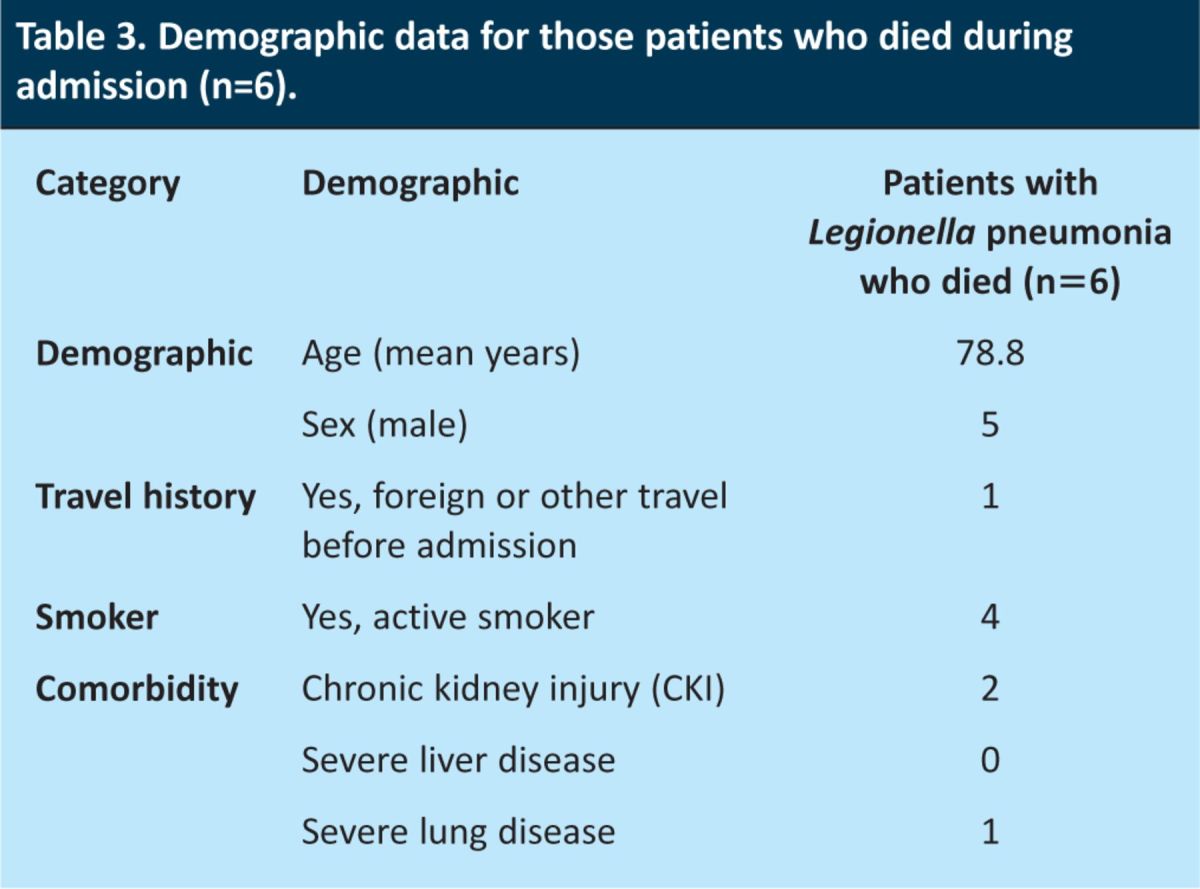
Those who died during admission were significantly older than those who survived to discharge (79 years (95% CI 70–88) vs 60 years (95% CI 55–66), p<0.001). Of those who died, five (83%) had UK community-acquired LP. The demographics of patients who died are shown in Table 3.
Table 3.
Demographic data for those patients who died during admission (n=6).
Of those surviving to discharge, 3/20 (15%) were readmitted within three months, all for unrelated medical conditions.
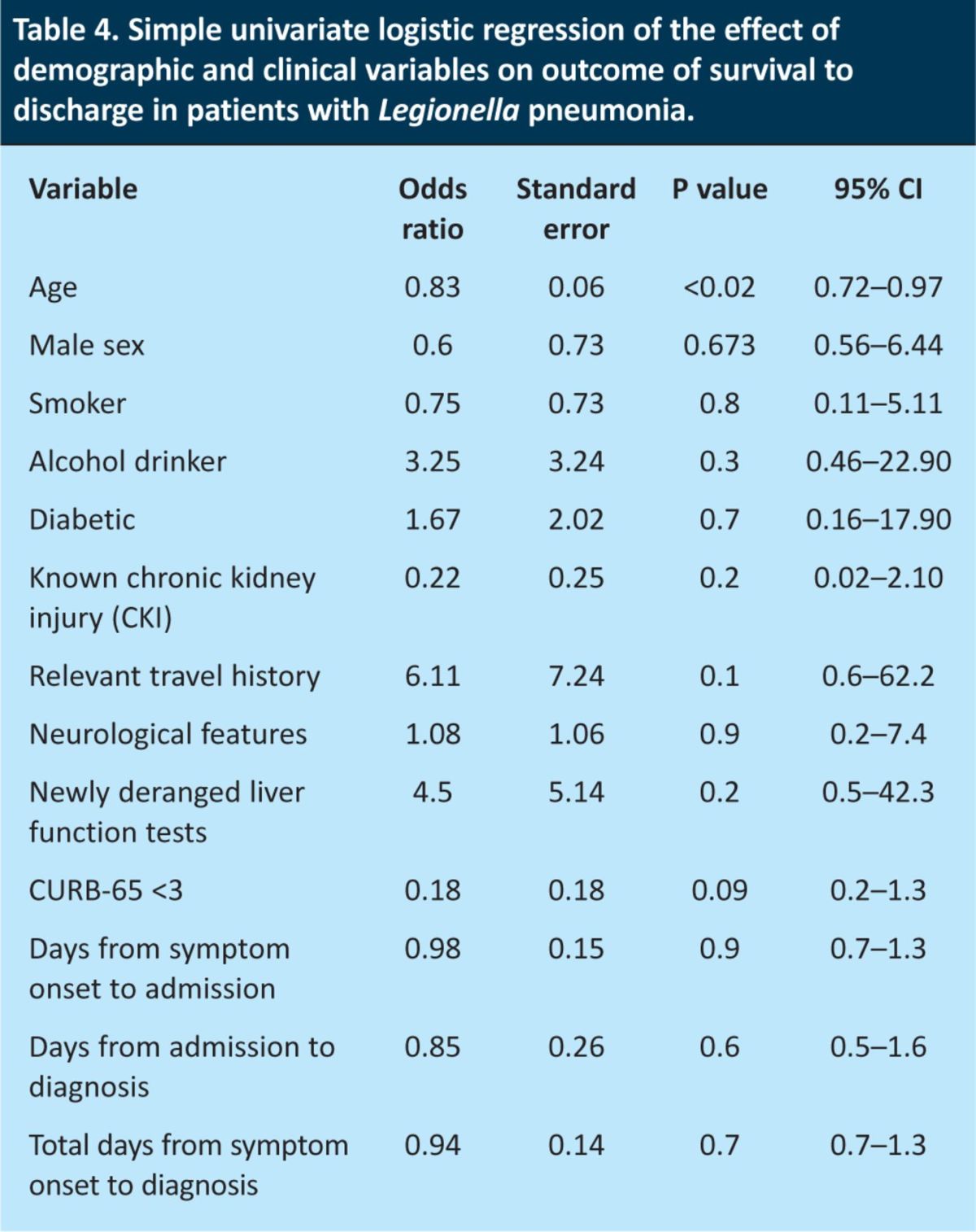
On univariate logistic regression analysis of the effect of demographic and clinical variables on outcome, only lower age was associated with a decreased likelihood of death (OR 0.83 (95% CI 0.72–0.97), p<0.02). There was a tendency towards lower CURB-65 scores (0–2) being associated with decreased likelihood of death, but this did not reach statistical significance (OR 0.18 (95% CI 0.23–1.33), p=0.09). No other demographic or clinical factors were predictive of outcome, including days from symptom onset to admission and diagnosis (Table 4). Subsequently, no multiple logistic regression analysis was performed.
Table 4.
Simple univariate logistic regression of the effect of demographic and clinical variables on outcome of survival to discharge in patients with Legionella pneumonia.
Discussion
After being alerted by the HPA that, between 2001 and 2009, there appeared to be an uneven distribution of the LP cases in the Lancashire and Cumbria region, with 44% being in the Blackpool area, we undertook an observational, descriptive, retrospective analysis of all LP cases in our trust, Blackpool Teaching Hospitals, between July 2006 and July 2011.
LP was first reported after an outbreak at an American Legion conference in 19768 and, since then, the number of LP cases in high-resource settings has continued to increase.9 Legionella pneumophila comprises 90% of Legionellaceae family infections,10 with L. pneumophila serogroup 1 being most common.11 Legionella spp. are heat-stable bacteria that have been associated with colonisation of sources as varied as wet cooling systems, pollution-relieving aerosols,3 asphalt-paving machines,12 spa pools,13 showers, fountains and even windscreen-wiper fluid containers.14
With LP being the cause of up to 7% of CAP and hospital-acquired pneumonias (HAP),1 and with mortality in outbreaks being as high as 20%,4 the disease continues to pose a significant public health risk. A recent outbreak of LP in southwest Edinburgh resulted in high levels of morbidity and three deaths, while a current outbreak in Camarthen, South Wales remains under investigation (www.bbc.co.uk/news/uk-wales-19654519).
The proportion of patients with LP requiring care in HDU or ITU facilities is significant. Nearly one-fifth of CAP cases in ITU were related to LP in some series, higher than was found in those with CAP but not admitted to ITU.15,16 However, with early recognition and appropriate treatment, it is estimated that mortality can be reduced to below 5%.17–19
Patients with LP are more likely to be male20 and smokers,21 a trend that was followed in our cohort (77% male, 54% smokers). Although Legionella is known to affect any age group,22 the mean average age in our series was 65 years, with affected women being older than men.
Little evidence exists as to the effect of comorbidities on either acquisition or severity of LP. British23 and Spanish24 studies have demonstrated chronic illness and worsening kidney function to be associated with poorer outcome, as is chronic lung disease. There is little evidence for the severity of LP in immunosuppressed patients, although one study following up patients with systemic lupus erythematosus taking infliximab reported a death secondary to LP.25 However, only a small number in our series were im-munosuppressed or had chronic kidney disease.
Over two-thirds of our cohort drank alcohol, 38% of whom were moderate to heavy drinkers. Alcoholism has previously been recognised as a predictor of more severe disease.24
In our study, 23% of patients had diabetes, which is higher than the 4.88% background local rate.26 The apparent LP–diabetes association is biologically plausible, because diabetes impairs immune function27 and has been linked with increasing susceptibility to other infections, such as tuberculosis.28 However, it is more likely within our series that this association is confounded by age.
In patients with LP in the UK, infection is often associated with a travel history abroad (commonly to southern Europe).29 Nearly half (46%) of our cohort had undergone foreign travel within the previous month (Fig 4), mostly within Europe, but with two patients travelling further afield. Conversely, an interesting finding from our series was that 54% of patients had UK-acquired LP, none of whom had any relevant ‘within-UK’ travel history. This number is similar to that previously demonstrated in UK public health data. However, in contrast to these previous data, which had suggested that fewer than 10% of cases were related to travel inside the UK, none of the patients with UK-acquired LP in our series reported such travel.30 This is likely to be biased by lack of documentation in clinical notes.
Recent evidence suggests that wider availability of LUA test has led to increased diagnosis of nosocomial LP that might otherwise have gone unnoticed;31 however, all patients in the current series had confirmed CAP. No postcode clustering was found in UK-acquired cases to account for a geographically linked pattern of LP acquisition or transmission (data not shown). In addition, after liaison with local environmental health department officers, who reviewed previous Legionella sources and their updated register of wet cooling systems, neither did the Cumbria and Lancashire Health Protection Unit find any associated geographical clustering of wet cooling systems in the region. However, It was acknowledged that it was unlikely that all wet cooling systems would be registered with the appropriate authorities for analysis. Other potential explanations, such as local weather patterns, were not examined within the confines of this study, and the HPA remains vigilant to new LP cases and continues to monitor the local situation.
A variety of well-recognised clinical and biochemical features are suggestive of LP, including fever, gastrointestinal disturbance, confusion, meningoencephalitis, new neurological features (such as nerve palsies, including Guillain–Barré syndrome), deranged liver function tests, hyponatraemia and kidney failure.8,32–34 Many of our cohort had a combination of these ‘classic’ features, indicating that they continue to be relevant in clinical assessment: 88% had biochemical hyponatraemia; 54% had AKI, as shown in other series;33 85% had hepatitis liver derangement (only 7% showed a cholestatic picture); and 35% had new-onset confusion or other neurological features. Although there was a trend in our series towards lower mean average serum Na in those patients presenting with neurological features than those without, this did not reach statistical significance.
Of note for the clinician, two patients with LP and neurological features presented with marked cerebellar syndrome, an uncommon but recognised presentation.35
LUA testing is rapid, highly specific and recommended as a first-line diagnostic test for early Legionella diagnosis.36 However, LUAs only identify L. pneumophila Serogroup 1 and have moderate overall sensitivity (74% in a recent meta-analysis),37 which improves with greater disease severity.18 The mean average of two days until a positive LUA result in our study is likely to reflect ward-based delay in requesting the test, rather than a lab-based delay in reporting the result; however, it was not possible to verify this from analysis of patients’ case notes. Although on regression analysis no association between outcome and time from symptom onset to admission or diagnosis was found, this is likely to be related to the size of our patient cohort being too small to identify any possible effect.
Most patients (25/26, 96%) had consolidation on chest X-ray: in 22/25 patients (88%), this was unilateral, a propensity that has been noted in other studies.38
Despite wide recognition in the UK of the CURB-65 score and its incorporation into both local and national CAP guidelines, admission CURB-65 score was not documented in over half of the patients. Furthermore, the antibiotic prescription was inappropriate for the severity of disease (as per documented CURB-65 score) in 23% of patients. Of particular note, a significant proportion of patients had a recalculated CURB-65 score of 0–2 (62%). Table 5 shows the demographics of this subgroup. Of these patients, who had either risk factors for, or travel history suggestive of, LP, 77% received appropriate Legionella cover. However, 23% of these patients with a CURB-65 score <2 had neither relevant travel history nor any documented evidence of high clinical Legionella suspicion, but still went on to have a LUA test sent and began treatment with antibiotics that included cover against Legionella (a macrolide in all cases). It is unclear whether the choice of macrolide in these cases was the result of suspicion of ‘atypical’ pneumonia, but it is clear that strict adherence to national CURB-65 guidance in these patients with low CURB-65 scores would have meant not testing or treating for atypical pneumonias, including Legionella. Current UK guidance recommends Legionella testing in ‘severe’ pneumonia (CURB-65 >3), when there is high clinical suspicion, or during outbreaks.6 There was no known acute outbreak of Legionella locally at this time and so guidance would have missed nearly one-quarter (6/26) of Legionella cases in our series. It may be postulated that, despite their low CURB-65 scores, these patients had other clinical features that made the ‘clinical suspicion’ for potential Legionella diagnosis high (eg failure to respond to first-line beta-lactam or aminoglycoside antibiotics in the community, profound hyponatraemia, deranged liver function tests, neurological signs or multisystem involvement). However, it was not possible to elicit this subjective ‘clinical suspicion’ from the patient case notes with any certainty.
Of those who survived until discharge, 20/20 (100%) received appropriate Legionella therapy as documented in Fig 5, and all of these patients had microbiology clinician involvement.
Full typing and resistance profiling of both the urine and sputum samples of LP patients at reference laboratories is recommended. However, sputum testing occurred in only 19% of our cases. LP remains a significant public health problem and acquisition of quality epidemiological data will require the vigilance of physicians to send further confirmatory tests that might not be of direct benefit to the patient but of benefit to the wider community. This issue could be brought to the clinician's attention on reports of a positive diagnosis or through phone calls from the microbiology department to the patient's attending physician.
Overall morbidity and mortality were high, with nearly one-third of patients requiring HDU or ITU care. Median LOS in those treated on HDU or ITU was 31 days, as opposed to five days if managed on a ward. Nearly one-quarter of the patients died, mostly early into admission, with five of the deaths being within the first five days. Although data from outbreak studies have shown mortality rates of up to 20%,4 the mortality rate seen in our cohort of unrelated cases is higher. The patients who died were older than the cohort average (79 vs 65 years, p=0.017), half had severe chronic illness and two-thirds were ongoing smokers. The difference in mortality rates between our cohort and those of outbreaks may relate to heightened clinical suspicion during outbreaks.
This study has several limitations. First, it describes a patient group of limited numbers, enabling only minimal statistical analysis. Second, its retrospective design means that it is reliant on data collection from documentation in clinical case notes. Finally, no comparison was made with demographic, clinical and outcome data of patients with non-Legionella pneumonias within the same study setting.
Conclusions
In our study, over half of Legionella infections were UK-acquired, indicating that a travel history alone is an insufficient clinical indicator for Legionella disease. Classic atypical features were common, as was the well-recognised association with hyponatraemia, a greater degree of which was seen in those with neurological derangement. Only a few sputum samples were sent to a reference laboratory, limiting further epidemiological mapping of UK Legionella cases. There was high morbidity and mortality, with nearly one-third of patients requiring HDU or ITU care and over one-quarter dying, mainly within the first week of admission. Although generally poorly documented, in patients for whom a CURB-65 score was recorded, over half had a score of 0–2, which would indicate neither requesting of a Legionella test nor empirical LP treatment, according to current UK guidelines; therefore, LP cases might be being missed, untreated or both.
Our study highlights the need for clinicians to consider a diagnosis of LP, be aware that travel history or low CURB-65 score do not preclude the diagnosis, treat suspected LP early and appropriately and be vigilant in collection of samples for essential epidemiological profiling.
References
- 1.Fang GD, Fine M, Orloff J, et al. New and emerging aetiologies for community-acquired pneumonia with implications for therapy. A prospective multi-center study of 359 cases. Medicine 1990; 69: 307–16. [DOI] [PubMed] [Google Scholar]
- 2.Blanquer J, Blanquer R, Borras R, et al. Etiology of community acquired pneumonia in Valencia, Spain: a multicenter prospective study. Thorax 1991; 46: 508–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Joseph CA, Ricketts KD. Legionnaires disease in Europe, 2007–2008. Euro Surveill 2010; 15: 19493. [PubMed] [Google Scholar]
- 4.Nygård K, Werner-Johansen Ø, Rønsen S, et al. An outbreak of legionnaires disease caused by long-distance spread from an industrial air scrubber in Sarpsborg, Norway. Clin Infect Dis 2008; 46: 61–9. [DOI] [PubMed] [Google Scholar]
- 5.Health Protection Agency, 2012. www.hpa.org.uk/web/HPAweb&HPAwebStandard/HPAweb_C/1195733748327 [Accessed 30 January 2013].
- 6.Lim WS, Baudouin SV, George RC, et al. BTS guidelines for the management of community acquired pneumonia in adults: update 2009. Thorax 2009; 64:iii1–55. 10.1136/thx.2009.121434 [DOI] [PubMed] [Google Scholar]
- 7.British Thoracic Society Standards of Care Committee. BTS guidelines for the management of community acquired pneumonia in adults. Thorax 2001; 56:iv:IV1–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fraser DW, Tsai TR, Orenstein W, et al. Legionnaires' disease: description of an epidemic of pneumonia. N Engl J Med 1977; 297: 1189–97. [DOI] [PubMed] [Google Scholar]
- 9.Neil K, Berkelman R. Increasing incidence of legionellosis in the United States, 1990–2005: changing epidemiologic trends. Clin Infect Dis. 2008; 47: 591–9. [DOI] [PubMed] [Google Scholar]
- 10.Fields BS, Benson RF, Besser RE. Legionella and Legionnaires' disease: 25 years of investigation. Clin Microbiol Rev 2002; 15: 506–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Plouffe JF, File TM, Jr, Breiman RF, et al. Reevaluation of the definition of Legionnaires' disease: use of the urinary antigen assay. Community Based Pneumonia Incidence Study Group. Clin Infect Dis 1995; 20: 1286–91. [DOI] [PubMed] [Google Scholar]
- 12.Coscollá M, Fenollar J, Escribano I, González-Candelas F. Legionellosis outbreak associated with asphalt paving machine, Spain, 2009. Emerg Infect Dis 2010; 16: 1381–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Groothuis DG, Havelaar AH, Veenendaal HR. A note on legionellas in whirlpools. J Appl Bacteriol 1985; 58: 479–81. 10.1111/j.1365-2672.1985.tb01489.x [DOI] [PubMed] [Google Scholar]
- 14.Wallensten A, Oliver I, Ricketts K, et al. Windscreen wiper fluid without added screenwash in motor vehicles: a newly identified risk factor for Legionnaires' disease. Eur J Epidemiol 2010; 25: 661–5. [DOI] [PubMed] [Google Scholar]
- 15.Mundy LM, Auwaerter PG, Oldach D, et al. Community-acquired pneumonia: impact of immune status. Am J Respir Crit Care Med 1995; 152: 1309–15. 10.1097/00019048-199603000-00010 [DOI] [PubMed] [Google Scholar]
- 16.Lieberman D, Schlaeffer F, Boldur I, et al. Multiple pathogens in adult patients admitted with community-acquired pneumonia: a one year prospective study of 346 consecutive patients. Thorax 1996; 51: 179–84. 10.1136/thx.51.2.179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fernández JA, López P, Orozco D, Merino J. Clinical study of an outbreak of Legionnaire's disease in Alcoy, Southeastern Spain. Eur J Clin Microbiol Infect Dis 2002; 21: 729–35. [DOI] [PubMed] [Google Scholar]
- 18.Garrido Blázquez RM, Parra Espinosa FJ, Francés Alemany L, et al. Antimicrobial chemotherapy for Legionnaires disease: levofloxacin versus macrolides. Clin Infect Dis 2005; 40: 800–6. [DOI] [PubMed] [Google Scholar]
- 19.Mykietiuk A, Carratalà J, Fernández-Sabé N, et al. Clinical outcomes for hospitalized patients with Legionella pneumonia in the antigenuria era: the influence of levofloxacin therapy. Clin Infect Dis 2005; 40: 794–9. 10.1086/428059 [DOI] [PubMed] [Google Scholar]
- 20.Falcó V, de Sevilla Fernández T, Alegre J, et al. Legionella pneumophila. A cause of severe community-acquired pneumonia. Chest 1991; 100: 1007–11. [DOI] [PubMed] [Google Scholar]
- 21.Pedro-Botet ML, Sabria M. Legionellosis. Semin Respir Crit Care Med 2005; 26: 625–34. 10.1055/s-2005-925526 [DOI] [PubMed] [Google Scholar]
- 22.Marston BJ, Plouffe JF, File TM, Jr, et al. Incidence of community-acquired pneumonia requiring hospitalization: results of a population-based active surveillance study in Ohio. The Community-Based Pneumonia Incidence Study Group. Arch Intern Med 1997; 157: 1709–18. 10.1001/archinte.157.15.1709 [DOI] [PubMed] [Google Scholar]
- 23.Woodhead MA, Macfarlane JT. Legionnaires' disease: a review of 79 community acquired cases in Nottingham. Thorax 1986; 41: 635–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.el-Ebiary M, Sarmiento X, Torres A, et al. Prognostic factors of severe Legionella pneumonia requiring admission to ICU. Am J Respir Crit Care Med 1997; 156: 1467–72. [DOI] [PubMed] [Google Scholar]
- 25.Aringer M, Houssiau F, Gordon C, et al. Adverse events and efficacy of TNF-alpha blockade with infliximab in patients with systemic lupus erythematosus: long-term follow-up of 13 patients. Rheumatology 2009; 48: 1451–4. [DOI] [PubMed] [Google Scholar]
- 26.National Health Services Information Centre, 2010. www.ic.nhs.uk/searchcatalogue?productid=7331&q=%22National+diabetes+audit%22&sort=Relevance&size=10&page=1#top [Accessed 30 January 2013].
- 27.Geerlings S, Hoepelman A. Immune dysfunction in patients with diabetes mellitus (DM). FEMS Immuno Med Microbiol 1999; 26: 259–65. [DOI] [PubMed] [Google Scholar]
- 28.Jeon CY, Murray MB. Diabetes mellitus increases the risk of active tuberculosis: a systematic review of 13 observational studies. PLoS Med 2008; 5: e152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Naik FC, Ricketts KD, Harrison TG, Joseph CA. Legionnaires' disease in England and Wales 2007. Health Protection Rep 2009; 3: 14. [Google Scholar]
- 30.Joseph CA, Harrison TG, Ilijic-Car D, Bartlett CL. Legionnaires' disease in residents of England and Wales: 1998. Commun Dis Public Health 1999; 2: 280–4. [PubMed] [Google Scholar]
- 31.Lepine LA, Jernigan DB, Butler JC, et al. A recurrent outbreak of nosocomial legionnaire's disease detected by urinary antigen testing: evidence for long-term colonization of a hospital plumbing system. Infect Control Hosp Epidemiol 1998; 19: 905–10. [PubMed] [Google Scholar]
- 32.Helms CM, Viner JP, Sturm RH, et al. Comparative features of pneumococcal, mycoplasmal, and Legionnaires' disease pneumonias. Ann Intern Med 1979; 90: 543–7. [DOI] [PubMed] [Google Scholar]
- 33.Fernández-Sabé N, Rosón B, Carratalà J, et al. Clinical diagnosis of Legionella pneumonia revisited: evaluation of the Community-Based Pneumonia Incidence Study Group scoring system. Clin Infect Dis 2003; 37: 483–9. [DOI] [PubMed] [Google Scholar]
- 34.Mulazimoglu L, Yu VL. Can Legionnaires disease be diagnosed by clinical criteria? A critical review. Chest 2001; 120: 1049–53. 10.1378/chest.120.4.1049 [DOI] [PubMed] [Google Scholar]
- 35.Shelburne SA, Kielhofner MA, Tiwari PS. Cerebellar involvement in legionellosis. South Med J 2004; 97: 61–4. 10.1097/01.SMJ.0000104843.78816.49 [DOI] [PubMed] [Google Scholar]
- 36.Sim WS, Baudouin SV, George RC, et al. BTS guidelines for the management of community acquired pneumonia in adults: update 2009. Thorax 2009; 64:iii1–iii55. 10.1136/thx.2009.1214. [DOI] [PubMed] [Google Scholar]
- 37.Shimada T, Noguchi Y, Jackson JL, et al. Systematic review and meta-analysis: urinary antigen tests for Legionellosis. Chest 2009; 136: 1576–85. [DOI] [PubMed] [Google Scholar]
- 38.Tan MJ, Tan JS, Hamor RH, et al. The radiologic manifestations of Legionnaire's disease. The Ohio Community-Based Pneumonia Incidence Study Group. Chest 2000; 117: 398–403. [DOI] [PubMed] [Google Scholar]