Abstract
Background & Aims
Functional status (a patient’s ability to perform activities that meet basic needs, fulfill usual roles, and maintain health and well being) has been linked to outcomes in patients with cirrhosis and can be measured by the Karnofsky performance status (KPS) scale. We investigated the association between KPS score and mortality in patients with cirrhosis.
Methods
We used the United Network for Organ Sharing database to perform a retrospective cohort study of patients listed for liver transplantation in the United States between 2005 and 2015. We used Cox proportional hazards and competing risk regression analyses to examine the association between KPS and mortality and transplantation.
Results
Of 79,092 patients, 44% were in KPS category A (KPS 80%–100%), 43% were in category B (KPS 50%–70%), and 13% were in category C (KPS 10%–40%). Between 2005 and 2015, the proportion of patients in category A decreased from 53% to 35%, whereas the proportions in categories B and C increased from 36% to 49% and from 11% to 16%, respectively. KPS was associated with mortality: compared to patients in KPS category A, the KPS B adjusted hazard ratio [HR] was 1.14 (95% confidence interval [CI], 1.11–1.18) and the KPS C adjusted HR was 1.63 (95% CI, 1.55–1.72). KPS was also associated with liver transplantation; compared to patients in KPS category A, the KPS B adjusted HR was 1.08 (95% CI, 1.06–1.11) and the KPS C adjusted HR was 1.35 (95% CI, 1.30–1.40). In competing risk analysis, only the relationship between KPS and mortality maintained significance and directionality. These relationships were most pronounced in patients without hepatocellular carcinoma.
Conclusions
Among patients with cirrhosis listed for liver transplantation, poor performance status, based on the KPS scale, is associated with increased mortality. In this population, performance status has decreased over time.
Keywords: survival, death, liver disease, UNOS, waitlist
INTRODUCTION
Cirrhosis is a highly morbid condition characterized by complications including ascites, hepatic encephalopathy, malnutrition, and sarcopenia. These complications can impair quality of life, mental health, and physical function. Functional status refers to an individual’s ability to perform activities that meet basic needs, fulfill usual roles, and maintain health and well-being.1 Functional status is diminished in patients with cirrhosis, and for these patients, measures of functional status have prognostic value. Frailty, a closely-related concept indicating impaired physiologic reserve, is associated with mortality, length of stay, and rehabilitation needs in hospitalized patients with cirrhosis.2 Frailty and decreased 6-minute walking distance are associated with death and diminished quality of life in liver transplant candidates.3–5
Functional status can be measured by the Karnofsky performance status (KPS), a simple, 11-point scale expressed as a percentage of physical function ranging from 100% (normal, no complaints, no evidence of disease) to 0% (dead).6 It is widely used in oncology to help guide treatment and clinical trials.7–10 It has good interrater reliability and construct validity.11, 12 It predicts outcomes in multiple populations, including chronic kidney disease,13 heart failure,14 and HIV.15 For patients with liver disease, the closely related Eastern Cooperative Oncology Group (ECOG) performance status is associated with mortality after liver transplant and in patients with hepatocellular carcinoma (HCC).16, 17 Notably, performance status is included in the Barcelona Clinic Liver Cancer staging system.18 Despite its widespread use, the KPS scale has not been examined in patients with cirrhosis.
We aimed to examine the association between KPS and mortality in waitlisted patients with cirrhosis. Our hypothesis was that patients with worse performance status would have greater mortality after adjusting for other measures of disease severity. To address this question, we examined the United Network for Organ Sharing (UNOS) database, which contains information on all patients listed for liver transplantation in the US. Compared to prior studies of frailty, which were all single-center,2–5 the UNOS database offers the advantages of (1) a large population-wide analysis from multiple centers and (2) the use of KPS data collected as part of routine clinical care, allowing for improved generalizability.
METHODS
Study Sample
For this retrospective cohort study, we obtained standard transplant analysis and research files from UNOS, which contains data through September 25, 2015 on all patients listed for solid organ transplant in the US. We limited the study to adults ≥ 18 years of age listed for liver transplant on or after April 1, 2005. Prior to that date, KPS was not routinely recorded.
Of the 110,359 adults, we excluded subjects with previous liver transplant (n = 3,759), multiple simultaneous organ transplant listings (n = 9,831), a non-cirrhosis diagnosis (n = 2,271), and acute liver failure (n = 4,553). Patients who transferred to another center or received a transplant at another center were excluded to avoid double-counting patients with multiple listings (n = 4,226). Similar to previous studies,19 patients with fewer than 5 days of waitlist time were also excluded (n = 8,368), as functional status may not be meaningful in this short time period. Finally, we excluded those missing KPS (n = 4,165) and those with extreme laboratory values suggesting data entry error: body mass index (BMI) < 15 or > 50 kg/m2, serum albumin > 6 g/dL, and serum sodium > 170 meq/L (n = 667). The study was approved by the Indiana University Institutional Review Board.
Outcomes
The primary outcome was removal from the waitlist due to death, clinical deterioration, or medical unsuitability (combined into a single mortality outcome). We also examined liver transplant as a secondary outcome. Patients removed from the waitlist for improved condition, loss to follow-up, removal in error, transplant refusal, and “other” were censored in survival analyses.
Karnofsky Performance Status
Since April 2005, transplant centers have captured functional status using the KPS scale, which is expressed in 10% increments.6 We further classified KPS into three categories according to the patient’s ability to work or provide self-care as previously described.6, 9 Patients with KPS category A are able to carry on normal activity and work; patients with KPS B are unable to work, but are able to live at home and care for personal needs; and patients with KPS C are unable to provide self-care (Supplementary Table).
Variables
We considered additional variables collected at listing that could influence outcomes. These included age; sex; race/ethnicity (white, black, Hispanic, and other); BMI; diabetes; dialysis; model for end-stage liver disease (MELD) score; serum albumin and sodium; presence of ascites (absent, slight, and moderate) and hepatic encephalopathy (none, grade 1–2, grade 3–4); ABO blood group; transplant region; listing year; and underlying liver disease. The underlying disease was categorized as alcohol, hepatitis C, alcohol/hepatitis C, nonalcoholic steatohepatitis, hepatitis B, cryptogenic cirrhosis, autoimmune hepatitis, primary biliary cirrhosis, primary sclerosing cholangitis, hepatocellular carcinoma, and other. Patients with HCC and another diagnosis (e.g. hepatitis C) were categorized as HCC because of differences in organ allocation.20
Statistical Analysis
Patients were followed from listing to removal from the waitlist for any reason. Those who were still on the waitlist on September 25, 2015, were censored. Categorical variables were reported as proportions and counts. Continuous variables were reported as either means and standard deviations (SD) or medians and ranges. Bivariate comparisons were performed with Pearson’s χ2 test, one-way analysis of variance, or the Kruskal-Wallis test. We used Cox proportional hazards regression to determine the association between KPS and either death or transplantation, with and without adjustment for covariates. The proportional hazards assumption was verified using log-log plots and Schoenfeld residuals. We repeated the models using an alternative outcome, grouping waitlist removals for “other” reasons as due to death or clinical deterioration because many patients removed for “other” reasons actually have deterioration.21 Additionally, we repeated the models including those missing KPS and assigning them to either KPS A or KPS C. We also performed subgroup analyses in patients with and without HCC, given the differences in organ allocation.20 Interaction terms were added to the models to confirm subgroup differences. In addition to considering cause-specific hazards using the Cox models, we used competing risk regression to estimate differences in the cumulative incidence of mortality and transplantation.22 These models account for competing outcomes that prevent the outcome of interest. For instance, death prevents future liver transplantation, and liver transplantation prevents pre-transplant death. In contrast, Cox proportional hazards models assume that outcomes may still occur after censoring. We also estimated the cumulative incidence of death and transplantation while accounting for the competing risks as previously described.23 All p-values were based on 2-sided tests and were considered statistically significant when p < 0.05. Analyses were performed using Stata version 13 (StataCorp LP, College Station, TX).
RESULTS
79,092 patients listed for transplant met the inclusion and exclusion criteria. The mean KPS was 68.6% (SD 20.1). 34,775 patients (44.0%) were category A (KPS 80 – 100%), 33,705 (42.6%) were category B (KPS 50 – 70%), and 10,612 (13.4%) were category C (KPS 10 – 40%). The groups were similar with regards to the prevalence of diabetes, BMI, and serum albumin and sodium (Table 1). Differences in these variables were statistically significant as a result of the large sample size. Group C was younger, had more women, and more Hispanic patients. Group C also had higher illness severity, with more patients on dialysis, greater mean MELD score, and more patients with ascites and hepatic encephalopathy. Group C also had more patients with alcoholic liver disease and fewer with HCC. For the 17,138 patients with HCC, the mean KPS was 80% (SD 15.6); 55.3% were KPS A, 39.7% were KPS B, and 5.0% were KPS C. There was significant regional variation in KPS: the mean KPS ranged from 64% to 75%. The ABO blood groups were similar in the KPS categories. Between 2005 and 2015, the proportion of patients in category A decreased from 53.3 to 35.1% (34% decrease), while the proportion in categories B and C increased from 35.7 to 49.3% (38% increase) and from 11.0 to 15.6% (42% increase), respectively (Figure 1).
Table 1.
Baseline Characteristics According to Karnofsky Performance Status
| Characteristic | Karnofsky A n = 34,775 |
Karnofsky B n = 33,705 |
Karnofsky C n = 10,612 |
p-value |
|---|---|---|---|---|
| Age, years, mean (SD) | 55.0 (9.6) | 55.3 (9.0) | 53.7 (10.1) | < 0.001 |
| % Male | 68.7 | 64.8 | 62.1 | < 0.001 |
| Race/ethnicity | < 0.001 | |||
| % White | 71.8 | 72.5 | 68.0 | |
| % Black | 8.4 | 7.2 | 8.3 | |
| % Hispanic | 13.4 | 15.4 | 18.1 | |
| % Other | 6.4 | 4.8 | 5.6 | |
| BMI, kg/m2, mean (SD) | 28.6 (5.4) | 28.8 (5.6) | 28.7 (6.1) | < 0.001 |
| % Diabetes | 24.9 | 27.3 | 25.3 | < 0.001 |
| % Dialysis | 0.3 | 0.6 | 10.1 | < 0.001 |
| MELD score, mean (SD) | 13.3 (5.3) | 15.6 (6.3) | 24.6 (9.7) | < 0.001 |
| Serum albumin, g/dL, mean (SD) | 3.2 (0.7) | 3.0 (0.6) | 3.0 (0.7) | < 0.001 |
| Serum sodium, meq/L, mean (SD) | 136.9 (4.1) | 135.7 (4.7) | 135.1 (6.0) | < 0.001 |
| Ascites | < 0.001 | |||
| % Absent | 38.1 | 23.3 | 15.5 | |
| % Slight | 51.3 | 55.6 | 42.3 | |
| % Moderate | 10.6 | 21.1 | 42.3 | |
| Hepatic encephalopathy | < 0.001 | |||
| % None | 54.6 | 35.1 | 25.3 | |
| % Grade 1–2 | 43.8 | 60.5 | 59.4 | |
| % Grade 3–4 | 1.6 | 4.4 | 15.3 | |
| Liver disease | < 0.001 | |||
| Alcohol | 12.8 | 17.1 | 22.7 | |
| Hepatitis C | 23.6 | 24.8 | 24.7 | |
| Alcohol/Hepatitis C | 4.4 | 6.3 | 6.5 | |
| Nonalcoholic steatohepatitis | 7.0 | 9.5 | 9.1 | |
| Cryptogenic | 5.1 | 5.9 | 6.5 | |
| Hepatocellular carcinoma | 27.2 | 20.2 | 8.0 | |
| Hepatitis B | 1.7 | 1.4 | 2.8 | |
| Primary sclerosing cholangitis | 5.6 | 3.3 | 2.9 | |
| Primary biliary cirrhosis | 3.0 | 2.9 | 2.9 | |
| Autoimmune hepatitis | 2.8 | 2.6 | 3.9 | |
| Other | 6.7 | 6.0 | 10.1 |
Figure 1.
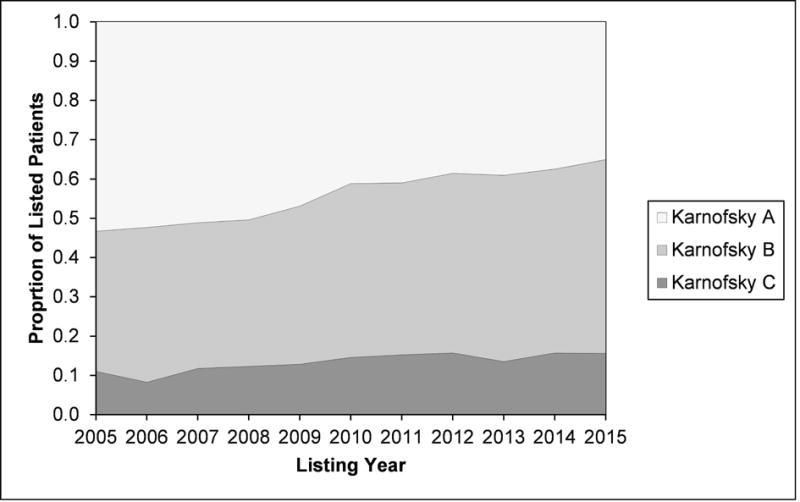
Temporal changes in Karnofsky performance status of patients listed for liver transplantation in the United States.
During a median follow-up of 0.54 years (interquartile range 0.16–1.52 years), 40,344 patients (51.0%) received a transplant, 17,999 (22.8%) were removed from the waitlist for death or deterioration, 5,840 (7.4%) were removed for “other” reasons, and 14,909 (18.9%) were still alive at last follow-up. 5-year cumulative incidence curves show an increase in both mortality and transplant with declining KPS (Figure 2). The 1-year incidence of death is 11.4% for KPS A, 15.5% for KPS B, and 27.4% for KPS C, and the 1-year incidence of transplant is 38.7%, 44.7%, and 53.4%.
Figure 2.
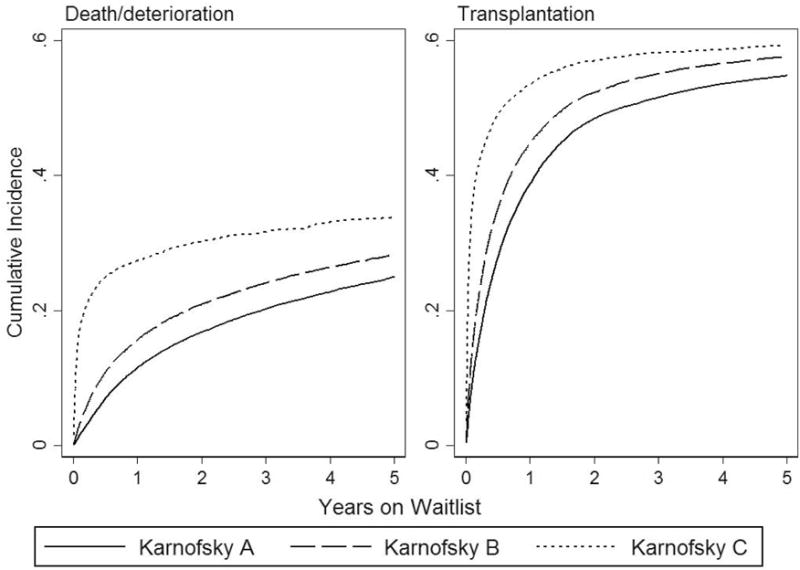
Five-year cumulative incidences of death/deterioration and liver transplantation according to Karnofsky performance status.
In the Cox models, worse KPS was associated with increased risk for death/deterioration and/or transplantation (Table 2). These differences persisted after adjustment for multiple variables. As compared to KPS A, the adjusted hazard ratio (HR) for death/deterioration for KPS B was 1.14 (95% confidence interval [CI], 1.11 – 1.18) and for KPS C was 1.63 (95% CI, 1.55 – 1.72). The association between KPS and transplant appeared less pronounced: KPS B adjusted HR 1.08 (95% CI, 1.06 – 1.11), and KPS C adjusted HR 1.35 (95% CI, 1.30 – 1.40). These relationships were confirmed in analyses where patients removed from the waitlist for “other” reasons were reclassified as death/deterioration and where patients with missing KPS were included and assigned to KPS A and C (data not shown).
Table 2.
Relationship Between Karnofsky Performance Status and Clinical Outcomes
| Unadjusted Hazard Ratioa | Adjusted Hazard Ratiob | Unadjusted Subhazard Ratioa | Adjusted Subhazard Ratiob | |
|---|---|---|---|---|
| Death/deterioration | ||||
| Karnofsky A | 1 (Ref) | 1 (Ref) | 1 (Ref) | 1 (Ref) |
| Karnofsky B | 1.42 (1.37–1.47) | 1.14 (1.11–1.18) | 1.17 (1.14–1.21) | 1.08 (1.04–1.11) |
| Karnofsky C | 3.65 (3.50–3.81) | 1.63 (1.55–1.72) | 1.80 (1.72–1.88) | 1.26 (1.20–1.33) |
|
| ||||
| Transplantation | ||||
| Karnofsky A | 1 (Ref) | 1 (Ref) | 1 (Ref) | 1 (Ref) |
| Karnofsky B | 1.24 (1.22–1.27) | 1.08 (1.06–1.11) | 1.15 (1.12–1.17) | 1.06 (1.03–1.08) |
| Karnofsky C | 2.41 (2.34–2.48) | 1.35 (1.30–1.40) | 1.54 (1.49–1.59) | 1.06 (1.02–1.11) |
|
| ||||
| Death or Transplantation | ||||
| Karnofsky A | 1 (Ref) | 1 (Ref) | N/A | N/A |
| Karnofsky B | 1.29 (1.27–1.32) | 1.10 (1.08–1.12) | ||
| Karnofsky C | 2.74 (2.68–2.81) | 1.44 (1.40–1.48) | ||
Hazard ratios are derived from Cox proportional hazards models; subhazard ratios are derived from competing risks regression models.
Adjusted for age, sex, race, diabetes, dialysis, BMI, blood group, MELD score, serum albumin, sodium, ascites, encephalopathy, underlying liver disease, transplant region, and year of listing.
In competing risk regression, where death and transplantation are considered competing outcomes rather than being censored, the association between KPS and death/deterioration was confirmed (Table 2). This association was also present in the adjusted model: KPS B adjusted subhazard ratio (SHR) 1.08 (95% CI, 1.04 – 1.11), and KPS C adjusted SHR 1.26 (95% CI, 1.20 – 1.33). KPS was also associated with transplantation in the univariate model, but in the adjusted model, the association was markedly attenuated and without directionality (SHR 1.06 for both KPS B and C).
The addition of interaction terms to the Cox models for mortality and transplantation confirmed significant differences in those with and without HCC. Results of the multivariable Cox models in those with and without HCC are shown in Figure 3. For those without HCC, worsening KPS was associated with increased mortality and transplantation. These relationships also held for patients with HCC, although the associations were attenuated, and, for KPS B, the associations with transplantation and combined death or transplantation lost statistical significance. Associations with mortality were similar when patients with “other” removals were reclassified as having died and when patients with missing KPS were included and assigned to KPS A and C (data not shown).
Figure 3.
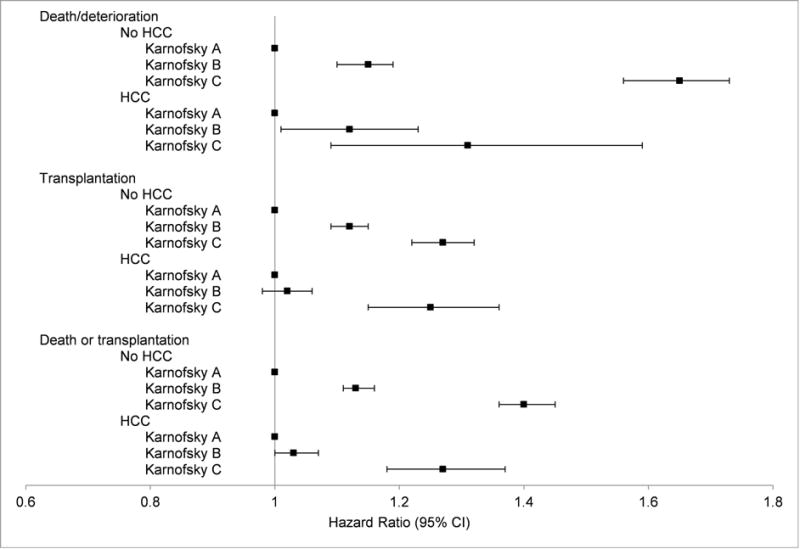
Relationship between Karnofsky performance status and clinical outcomes in patients with and without hepatocellular carcinoma. Estimates and confidence intervals are based on the multivariable Cox proportional hazards models.
In competing risk models, for those without HCC, there remained an increase in the risk of death with worsening KPS: KPS B adjusted SHR 1.07 (95% CI, 1.03 – 1.10) and KPS C adjusted SHR 1.28 (95% CI, 1.21 – 1.35) (Figure 4). This relationship was also seen in the univariate model for the HCC subgroup (KPS B unadjusted SHR 1.30; 95% CI, 1.18 – 1.42; KPS C SHR 1.74; 95% CI, 1.45 – 2.08), but not after adjustment (Figure 4). In those without HCC, worsening KPS was also associated with greater likelihood of transplantation in univariate analysis (KPS B unadjusted SHR 1.33; 95% CI, 1.29 – 1.36; KPS C SHR 2.00; 95% CI, 1.92 – 2.07). However, after adjustment, KPS did not have a consistent association with transplant.
Figure 4.
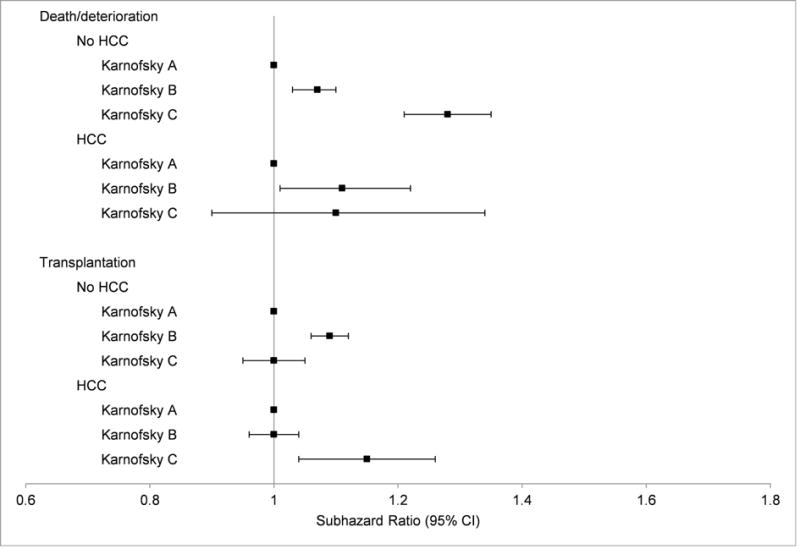
Relationship between Karnofsky performance status and clinical outcomes in patients with and without hepatocellular carcinoma. Estimates and confidence intervals are based on the multivariable competing risk models.
DISCUSSION
Patients with cirrhosis suffer from physical debility that can interfere with activities and impair quality of life. In this study of patients listed for transplant, worse functional status as measured by KPS was associated with increased mortality. KPS was also associated with greater propensity to receive a liver transplant, though this relationship did not hold after considering death as a competing outcome.
These findings add to the literature linking functional status to outcomes for patients with cirrhosis. Carey et al. showed that 6-minute walking distance predicts waitlist mortality.4 Lai and colleagues also demonstrated increased waitlist mortality with increasing frailty, measured by performance-based testing and patient self-report.3 In hospitalized patients with cirrhosis, frailty assessments were associated with 90 day mortality.2 Beyond confirming the relationship between functional status and mortality, this study provides novel additions. Compared to previous single-center studies, this study reflects outcomes across US transplant centers. The large sample size also allows adjustment for more confounders than could be achieved previously. Finally, the UNOS database can be used to examine trends in functional status amongst all patients on the national waitlist. The KPS scale, which has not been previously examined in this context, is widely used clinically and is ubiquitous in oncology.9 It is simple and can be performed by any provider, with good reliability and validity.11, 12 It is also already recorded widely for patients with cirrhosis at the time of transplant listing.
An important addition is the comparison of mortality and transplant. These related outcomes have a complex relationship, and can be modeled using both Cox regression, which treats outcomes independently, and competing risks regression, which considers outcomes together. Both methods are valuable and provide complementary insight.24 When considered independently, both mortality and transplant are more likely with declining KPS. However, when considered together, those with worse KPS do not appear more likely to receive a transplant, since the SHR approaches one and loses directionality. Reasons for this relationship are likely complex, and may include a balance between (1) the desire to transplant those with poor performance status who are most in need and (2) a reluctance to transplant sicker patients due to worse potential post-transplant outcomes.16 Although the relationship between KPS and death is also attenuated when considering competing outcomes, the directionality is maintained. Therefore, KPS may have a stronger influence on mortality as compared to transplant.
Differences in these relationships in patients with and without HCC are also important. In Cox models, the relationships between KPS and both outcomes were maintained, although attenuated for the HCC group. In competing risks models, the only relationship that maintained significance and directionality was for mortality in those without HCC. This suggests that KPS may be more important in patients without HCC. This finding may relate to the selection of patients with HCC for transplant. These patients have lower native MELD scores, higher transplant rates, and lower mortality as compared to non-HCC patients.20 We found that the HCC group had higher KPS, consistent with guidelines favoring transplant for HCC patients with better performance status.18
Two important findings related to KPS in the waitlist population deserve mention. First, the proportion with KPS B and C is increasing, with a concomitant decrease in KPS A. These changes mirror trends in the broader population, which is aging,25 accumulating comorbidities,26,27 and requiring more help with activities of daily living.28 Taken with the increased waitlist mortality for those with worse KPS, this trend is cause for concern. Similar projected changes in the organ donor population could also lead to declining numbers of liver transplants, further impacting waitlist mortality.29 Finally, patients with worse KPS may be less likely to be considered for transplant by risk-averse programs given the negative impact on post-transplant outcomes.16 Together, these trends could result in fewer transplants and increased waitlist mortality in the future. In this context, interventions to improve functional status may have an important role for patients with cirrhosis. Another important finding is the KPS regional variation. This variation may reflect known regional differences in disease severity, accompanied by differences in mortality and transplant rates.20 Alternatively, both regional variation and temporal trends in KPS may simply reflect differences in KPS assessment. In particular, the temporal trend might reflect improved assessment by transplant physicians as they accumulate experience with KPS.
Although this study provides novel information, it does have several limitations. First, the study only includes patients listed for transplant in the US. Additional studies of patients in other populations are needed to generalize the findings. KPS is recorded by individual transplant centers at listing, and many providers nationwide contribute to these KPS assessment. KPS has excellent interrater reliability in cancer populations;11, 12 however, in other populations its test characteristics are less well-established, and it has not been previously tested in cirrhosis. The closely related ECOG performance status has been studied in cirrhosis, and is associated with post-transplant mortality and HCC mortality.16, 17 However, in these studies reliability was not specifically measured. In our study, suboptimal reliability could account for regional variation and temporal trends in KPS. However, the potential for interrater variability is limited to a degree by the KPS categorization. Only ratings that differ across categories would impact the findings (e.g. KPS 40% vs. 50%), while variations within categories would have no impact (e.g. 50% vs. 60%). Nevertheless, further study of KPS validity and reliability are needed to support its use in the cirrhosis population. Finally, although KPS appears to influence mortality more than transplant, the absolute cumulative incidence of transplant is greater than death for all KPS categories. Therefore, prognosis cannot be assessed on the basis of KPS alone. Despite these limitations, this study benefits from a large sample size, which provides a comprehensive picture of patients listed for transplantation in the US. The large sample size also allows for analyses adjusting for multiple important confounders.
In conclusion, KPS is associated with mortality in patients with cirrhosis listed for liver transplantation. We also found that KPS amongst patients listed for transplant in the US has been worsening over the last decade. These novel findings underscore the importance of physical functioning in patients with cirrhosis and its potential growing impact on outcomes in this population. Future studies are needed to assess interventions to improve functional status and improve outcomes.
Supplementary Material
Acknowledgments
Grant Support: This publication was made possible, in part, with support from the Indiana Clinical and Translational Sciences Institute funded, in part by Grant Number KL2 TR001106 from the National Institutes of Health, National Center for Advancing Translational Sciences, Clinical and Translational Sciences Award.
This work was supported in part by Health Resources and Services Administration contract 234-2005-370011C. The content is the responsibility of the authors alone and does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
Abbreviations
- BMI
body mass index
- CI
confidence interval
- ECOG
Eastern Cooperative Oncology Group
- HCC
hepatocellular carcinoma
- HR
hazard ratio
- KPS
Karnofsky performance status
- MELD
model for end-stage liver disease
- SD
standard deviation
- SHR
subhazard ratio
- UNOS
United Network for Organ Sharing
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosures: The authors have no potential conflicts to disclose.
Author Contributions: Eric S. Orman: study concept and design, acquisition of data, analysis and interpretation of data, drafting of the manuscript, critical revision of the manuscript for important intellectual content, statistical analysis; Marwan Ghabril: study concept and design, analysis and interpretation of data, critical revision of the manuscript for important intellectual content; Naga Chalasani: study concept and design, analysis and interpretation of data, critical revision of the manuscript for important intellectual content.
References
- 1.Leidy NK. Functional status and the forward progress of merry-go-rounds: toward a coherent analytical framework. Nurs Res. 1994;43:196–202. [PubMed] [Google Scholar]
- 2.Tapper EB, Finkelstein D, Mittleman MA, et al. Standard assessments of frailty are validated predictors of mortality in hospitalized patients with cirrhosis. Hepatology. 2015;62:584–90. doi: 10.1002/hep.27830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lai JC, Feng S, Terrault NA, et al. Frailty predicts waitlist mortality in liver transplant candidates. Am J Transplant. 2014;14:1870–9. doi: 10.1111/ajt.12762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carey EJ, Steidley DE, Aqel BA, et al. Six-minute walk distance predicts mortality in liver transplant candidates. Liver Transpl. 2010;16:1373–8. doi: 10.1002/lt.22167. [DOI] [PubMed] [Google Scholar]
- 5.Derck JE, Thelen AE, Cron DC, et al. Quality of life in liver transplant candidates: frailty is a better indicator than severity of liver disease. Transplantation. 2015;99:340–4. doi: 10.1097/TP.0000000000000593. [DOI] [PubMed] [Google Scholar]
- 6.Karnofsky DA, Abelmann WH, Craver LF, et al. The use of the nitrogen mustards in the palliative treatment of carcinoma. With particular reference to bronchogenic carcinoma. Cancer. 1948;1:634–656. [Google Scholar]
- 7.Carson KA, Grossman SA, Fisher JD, et al. Prognostic factors for survival in adult patients with recurrent glioma enrolled onto the new approaches to brain tumor therapy CNS consortium phase I and II clinical trials. J Clin Oncol. 2007;25:2601–6. doi: 10.1200/JCO.2006.08.1661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Marechal R, Demols A, Gay F, et al. Prognostic factors and prognostic index for chemonaive and gemcitabine-refractory patients with advanced pancreatic cancer. Oncology. 2007;73:41–51. doi: 10.1159/000120627. [DOI] [PubMed] [Google Scholar]
- 9.Peus D, Newcomb N, Hofer S. Appraisal of the Karnofsky Performance Status and proposal of a simple algorithmic system for its evaluation. BMC Med Inform Decis Mak. 2013;13:72. doi: 10.1186/1472-6947-13-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Maltoni M, Pirovano M, Scarpi E, et al. Prediction of survival of patients terminally ill with cancer. Results of an Italian prospective multicentric study Cancer. 1995;75:2613–22. doi: 10.1002/1097-0142(19950515)75:10<2613::aid-cncr2820751032>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- 11.Mor V, Laliberte L, Morris JN, et al. The Karnofsky Performance Status Scale. An examination of its reliability and validity in a research setting. Cancer. 1984;53:2002–7. doi: 10.1002/1097-0142(19840501)53:9<2002::aid-cncr2820530933>3.0.co;2-w. [DOI] [PubMed] [Google Scholar]
- 12.Schag CC, Heinrich RL, Ganz PA. Karnofsky performance status revisited: reliability, validity, and guidelines. J Clin Oncol. 1984;2:187–93. doi: 10.1200/JCO.1984.2.3.187. [DOI] [PubMed] [Google Scholar]
- 13.McClellan WM, Anson C, Birkeli K, et al. Functional status and quality of life: predictors of early mortality among patients entering treatment for end stage renal disease. J Clin Epidemiol. 1991;44:83–9. doi: 10.1016/0895-4356(91)90204-m. [DOI] [PubMed] [Google Scholar]
- 14.Johnson MJ, Bland JM, Davidson PM, et al. The relationship between two performance scales: New York Heart Association Classification and Karnofsky Performance Status Scale. J Pain Symptom Manage. 2014;47:652–8. doi: 10.1016/j.jpainsymman.2013.05.006. [DOI] [PubMed] [Google Scholar]
- 15.O’Dell MW, Lubeck DP, O’Driscoll P, et al. Validity of the Karnofsky Performance Status in an HIV-infected sample. J Acquir Immune Defic Syndr Hum Retrovirol. 1995;10:350–7. [PubMed] [Google Scholar]
- 16.Jacob M, Copley LP, Lewsey JD, et al. Functional status of patients before liver transplantation as a predictor of posttransplant mortality. Transplantation. 2005;80:52–7. doi: 10.1097/01.tp.0000163292.03640.5c. [DOI] [PubMed] [Google Scholar]
- 17.Hsu CY, Lee YH, Hsia CY, et al. Performance status in patients with hepatocellular carcinoma: determinants, prognostic impact, and ability to improve the Barcelona Clinic Liver Cancer system. Hepatology. 2013;57:112–9. doi: 10.1002/hep.25950. [DOI] [PubMed] [Google Scholar]
- 18.Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020–2. doi: 10.1002/hep.24199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Berry K, Taylor J, Liou IW, et al. Portal vein thrombosis is not associated with increased mortality among patients with cirrhosis. Clin Gastroenterol Hepatol. 2015;13:585–93. doi: 10.1016/j.cgh.2014.10.010. [DOI] [PubMed] [Google Scholar]
- 20.Northup PG, Intagliata NM, Shah NL, et al. Excess mortality on the liver transplant waiting list: unintended policy consequences and Model for End-Stage Liver Disease (MELD) inflation. Hepatology. 2015;61:285–91. doi: 10.1002/hep.27283. [DOI] [PubMed] [Google Scholar]
- 21.Goldberg D, French B, Trotter J, et al. Underreporting of liver transplant waitlist removals due to death or clinical deterioration: results at four major centers. Transplantation. 2013;96:211–6. doi: 10.1097/TP.0b013e3182970619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fine JP, Gray RJ. A Proportional Hazards Model for the Subdistribution of a Competing Risk. Journal of the American Statistical Association. 1999;94:496–509. [Google Scholar]
- 23.Choudhury JB. Non-parametric confidence interval estimation for competing risks analysis: application to contraceptive data. Stat Med. 2002;21:1129–44. doi: 10.1002/sim.1070. [DOI] [PubMed] [Google Scholar]
- 24.Dignam JJ, Zhang Q, Kocherginsky M. The use and interpretation of competing risks regression models. Clin Cancer Res. 2012;18:2301–8. doi: 10.1158/1078-0432.CCR-11-2097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ortman JM, Velkoff VA, Hogan H. An Aging Nation: The Older Population in the United States. Washington, DC: U.S Census Bureau; 2014. pp. 25–1140. [Google Scholar]
- 26.Menke A, Casagrande S, Geiss L, et al. Prevalence of and Trends in Diabetes Among Adults in the United States, 1988–2012. Jama. 2015;314:1021–9. doi: 10.1001/jama.2015.10029. [DOI] [PubMed] [Google Scholar]
- 27.Flegal KM, Carroll MD, Kit BK, et al. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999–2010. Jama. 2012;307:491–7. doi: 10.1001/jama.2012.39. [DOI] [PubMed] [Google Scholar]
- 28.Martin LG, Freedman VA, Schoeni RF, et al. Trends in disability and related chronic conditions among people ages fifty to sixty-four. Health Aff (Millwood) 2010;29:725–31. doi: 10.1377/hlthaff.2008.0746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Orman ES, Mayorga ME, Wheeler SB, et al. Declining liver graft quality threatens the future of liver transplantation in the United States. Liver Transpl. 2015;21:1040–50. doi: 10.1002/lt.24160. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


