Abstract
Background
Physical exercise effectively attenuates neuropathic pain, and multiple events including the inhibition of activated glial cells in the spinal dorsal horn, activation of the descending pain inhibitory system, and reductions in pro-inflammatory cytokines in injured peripheral nerves may contribute to exercise-induced hypoalgesia. Since fewer GABAergic hypoalgesic interneurons exist in the dorsal horn in neuropathic pain model animals, the recovery of impaired GABAergic inhibition in the dorsal horn may improve pain behavior. We herein determined whether the production of gamma-aminobutyric acid (GABA) and glutamic acid decarboxylase (GAD) in the dorsal horn is restored by treadmill running and contributes to exercise-induced hypoalgesia in neuropathic pain model mice. C57BL/6 J mice underwent partial sciatic nerve ligation (PSL). PSL-Runner mice ran on a treadmill at 7 m/min for 60 min/day, 5 days/week, from two days after PSL.
Results
Mechanical allodynia and heat hyperalgesia developed in PSL-Sedentary mice but were significantly attenuated in PSL-Runner mice. PSL markedly decreased GABA and GAD65/67 levels in neuropils in the ipsilateral dorsal horn, while treadmill running inhibited these reductions. GABA+ neuronal nuclei+ interneuron numbers in the ipsilateral dorsal horn were significantly decreased in PSL-Sedentary mice but not in PSL-Runner mice. Pain behavior thresholds positively correlated with GABA and GAD65/67 levels and GABAergic interneuron numbers in the ipsilateral dorsal horns of PSL-Sedentary and -Runner mice.
Conclusions
Treadmill running prevented PSL-induced reductions in GAD65/67 production, and, thus, GABA levels may be retained in interneurons and neuropils in the superficial dorsal horn. Therefore, improvements in impaired GABAergic inhibition may be involved in exercise-induced hypoalgesia.
Keywords: Gamma-aminobutyric acid, glutamic acid decarboxylase, neuropathic pain, exercise-induced hypoalgesia, treadmill running
Background
Physical exercise has been established as an effective means to attenuate neuropathic pain (NPP) in animal studies.1–6 A previous study reported that treadmill running reduced pain hypersensitivity in chronic constriction injury (CCI) model mice, and the inhibition of microgliosis in the spinal dorsal horn was suggested to contribute to this exercise-induced hypoalgesia (EIH).7 Swimming in partial sciatic nerve ligation (PSL) model mice attenuated pain behaviors and also inhibited the activation of glial cells in the spinal dorsal horn.8 Moreover, treadmill running in spinal nerve ligation (SNL) model rats increased β-endorphin and met-enkephalin contents in the periaqueductal gray matter (PAG) and rostroventral medulla (RVM), suggesting that the descending pain inhibitory system plays an important role in EIH.9 Thus, physical exercise appears to improve NPP by altering cells and systems in the nervous system including the brainstem and spinal dorsal horn; however, the mechanisms underlying EIH have not yet been elucidated in detail.
Gamma-aminobutyric acid (GABA) is the principal inhibitory transmitter in the central nervous system including the spinal dorsal horn. Previous studies showed the loss of GABA-immunoreactive cells and fibers in the spinal dorsal horns of NPP model animals, and impairments in GABAergic inhibition in the spinal dorsal horn are assumed to play an important role in the production of NPP.10–12 GABA is synthesized from glutamate by glutamic acid decarboxylase (GAD). Two distinct isoforms of GAD, GAD65 and GAD67, have been identified, with each isoform being encoded by different genes, namely, Gad2 and Gadl, respectively.13 Although GAD65 and GAD67 are found in GABAergic neurons in the spinal dorsal horn, the predominance of each isoform varies in respective laminae. GAD65 is largely expressed in laminae I–II, while the expression of GAD67 is abundant in deeper laminae.14 In CCI model rats, the density of nerve terminals with GAD65 is reduced in the superficial dorsal horn.15 Furthermore, GAD65 knockout mice show impaired synaptic inhibition and exacerbated pain behaviors.16 A previous study demonstrated that the decrease in GABA in the spinal dorsal horn in CCI model rats was associated with a decline in GAD67.17 Thus, the functional loss of GAD65 and/or GAD67 in the spinal dorsal horn may contribute to the development of NPP via reductions in the GABA inhibitory tone.
Thermal and tactile hypersensitivities are reversed with the administration of GABA to NPP model animals.18 Moreover, pain behaviors in PSL model rats are reduced by spinal cord stimuli that elevate spinal GABA and GAD65 levels.19,20 An injection of the adenoviral vector GAD65 into the trigeminal ganglion of facial pain model rats not only enhances GAD65 and GABA levels but also improves pain behaviors.21 Previous studies have also demonstrated that GAD67 plays critical roles in regulating NPP.17,22,23 Despite growing evidence showing the importance of GABA and GADs in the regulation of NPP, the relevance of both factors to EIH is unknown. Therefore, we herein focused on GABA and GAD65/67 production in the superficial dorsal horn in NPP model mice subjected to treadmill running. The results of the present study showed that exacerbated pain behaviors following PSL surgery were significantly improved in treadmill runners. Furthermore, PSL-induced reductions in GAD65/67 production in the superficial dorsal horn were prevented by treadmill running, leading to the retention of GABA in interneurons and neuropils. Positive correlations were also observed between the thresholds of pain behaviors and GABA and GAD65/67 levels or GABAergic interneuron numbers in the ipsilateral dorsal horns of PSL-Sedentary and -Runner mice. Therefore, we concluded that EIH is achieved, at least in part, by improvements in impaired GABAergic inhibition in the spinal dorsal horn.
Methods
All experiments in this study were approved by the Animal Care Committee of Wakayama Medical University (Ref. No. 642). All experiments conformed to the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 99-158, revised 2002).
Preparation of NPP model mice
Adult C57BL/6 J mice (Japan SLC, Shizuoka, Japan) were used in this study. These mice were housed four to five per cage, under a 12/12-h light-dark cycle and allowed free access to food and water. The evaluation of pain behaviors was initiated on 11-week-old mice, and mice were 13 weeks old in the final treadmill running session. NPP model mice were prepared by PSL according to a previously described method.24 Mice were deeply anesthetized with 2% isoflurane to maintain painless conditions. Under sterile conditions, an incision was made in the skin on the lateral surface of the right thigh, and the muscle layers were parted to expose the sciatic nerve at the mid-thigh level. Approximately one-third to one-half of the sciatic nerve was tightly ligated with 8-0 silk sutures (Alcon Surgical, Fort Worth, TX), and the wound was then closed with 5-0 nylon sutures (Akiyama-Seisakusho, Tokyo, Japan). Sham surgery was performed according to the same procedures described above, except for the execution of PSL.
Experimental groups and treadmill running protocol
Mice were divided into five groups: (1) Naive mice: mice without any artificial surgery, (2) Sham-Sedentary mice: mice without treadmill running after sham surgery, (3) Sham-Runner mice: mice with treadmill running after sham surgery, (4) PSL-Sedentary mice: mice without treadmill running after PSL surgery, and (5) PSL-Runner mice: mice with treadmill running after PSL surgery.
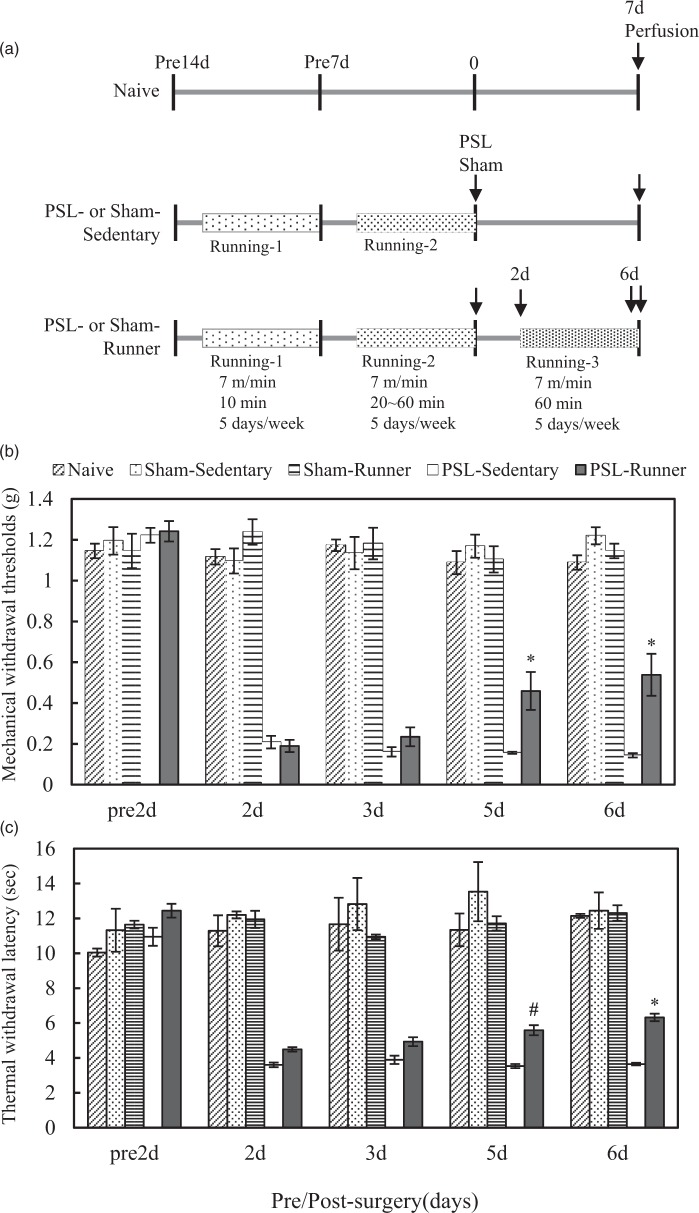
All mice, except for Naive mice, were accustomed to treadmill running for two weeks prior to sham and PSL surgeries. Treadmill running was started at 5 p.m. The treadmill running protocol is presented in Figure 1(a). All mice were acclimated to the treadmill belt for 10 min before starting locomotion. Throughout the treadmill running period, mice were encouraged to continue running and maintain their pace in line with the treadmill belt by gentle hand prodding of the tail or hindquarters. The mice readily responded to this stimulus, and the electrical shock system that forces mice to run was disconnected to avoid bias due to stress. The inclination of the treadmill was set at 0. In the first week, mice were allowed to run at a speed of 7 m/min for 10 min/day for 5 days/week in order to familiarize them with handling and the environment. In the second week, mice ran at a speed of 7 m/min for 20–60 min/day (an increment of 10 min per day), 5 days/week. In the third week, Sham- and PSL-Runner mice ran at a speed of 7 m/min for 60 min/day, 5 days/week, from two days after surgery. Previous studies reported that NPP model mice have the ability to run on a treadmill at a speed of 10 m/min or 31 m/min from three days post-surgery.1,7 In the present study, PSL-Runner mice were able to run on the treadmill at a speed of 7 m/min for 60 min/day from two days post-surgery without any special encouragement to continue running because we designed treadmill running at a slower speed (7 m/min) than those of previous studies, and mice had already experienced treadmill running at the same speed prior to PSL (Figure 1). Mice were sacrificed for the immunohistochemical analysis on the morning after the final treadmill running session.
Figure 1.
Treadmill running protocol and changes in pain behaviors in mice in the five experimental groups. (a) Adult male C57BL/6 J mice were divided into five experimental groups (n = 6). All groups, except for Naive mice, were subjected to Running-1 and Running-2 prior to PSL or Sham surgery. In the Running-1 program, mice ran at a speed of 7 m/min for 10 min/day for 5 days/week, and in the Running-2 program, ran at a speed of 7 m/min for 20 to 60 min/day (an increment of 10 min per day), 5 days/week. PSL- and Sham-Runner mice ran at a speed of 7 m/min for 60 min/day, 5 days/week, from 2 days after surgery (Running-3). Mice were sacrificed for the immunohistochemical analysis on the morning after the final treadmill running session. (b) von Frey and (c) plantar tests were performed in Naive (n = 6), Sham-Sedentary (n = 6), Sham-Runner (n = 6), PSL-Sedentary (n = 6), and PSL-Runner (n = 6) mice two days before and two, three, five, and six days after surgery. (b) Mechanical withdrawal thresholds were significantly higher in PSL-Runner mice than in PSL-Sedentary mice five and six days after PSL (*p < 0.01). (c) Thermal withdrawal latencies were significantly higher in PSL-Runner mice than in PSL-Sedentary mice five and six days after PSL (#p < 0.05, *p < 0.01). The significance of differences among groups was determined by a repeated-measures ANOVA followed by Tukey’s post hoc test.
Evaluation of pain behaviors
Mechanical allodynia and heat hyperalgesia, which are typical symptoms of NPP, were evaluated by von Frey and plantar tests, respectively. In order to dissipate the acute responses of treadmill running on pain behaviors, von Frey and plantar tests were performed at least 14 h after the treadmill running session (the next morning [at 8:30 a.m.]). Prior to the von Frey test, mice were placed in an acrylic glass enclosure with a wire mesh bottom (8.3 × 8.3 × 8.0 cm) and allowed to acclimate for 30 min.
Mechanical allodynia was evaluated using withdrawal thresholds upon a von Frey monofilament stimulation to the plantar surface of the hindpaw (0.023, 0.0275, 0.068, 0.166, 0.407, 0.692, 1.202, 1.479 g). The up-down methods of von Frey monofilament testing were used in the present study.25 The 0.407 -g stimulus was applied first. The von Frey monofilament was applied to the ipsilateral side of the hindpaw for a maximum period of 2 s, and the withdrawal response was observed. When a withdrawal response to a given monofilament was observed, a one-step-thinner filament was applied. The procedure was repeated with sequentially thinner monofilaments until no withdrawal response was observed. At this point, a one-step-thicker monofilament was applied again to confirm the positive response, and, finally, the negative response was confirmed with the one-step-thinner monofilament in order to complete the test. Throughout these procedures, the minimum pressure (g) that evoked a brisk withdrawal response by a stimulation with a thinner von Frey monofilament was taken as the withdrawal threshold.
In accordance with previously described methods,26 heat hyperalgesia was evaluated by the plantar test that measures withdrawal latencies (s) on a radiant thermal stimulus delivered from beneath the glass floor to the plantar surface of the hindpaw (Plantar Test, Model7371, Ugo Basile, Comerio, Italy). The heat stimulus by the projector lamp bulb was focused on the plantar surface of the ipsilateral hindpaw, and the minimum time (s) needed to evoke a brisk withdrawal response of the paw by the heat stimulation was taken as the withdrawal latency. A 20-s cut-off time was used to avoid tissue damage. The von Frey and plantar tests were performed three times for each mouse, and the mean values were designated as the withdrawal thresholds or withdrawal latencies, respectively. In all behavioral evaluations, the investigator was blinded to the titles of the experimental group in order to avoid any bias.
Immunohistochemistry and quantitative analysis of immunopositive cells
The primary antibodies used in this study were as follows: GABA (rabbit polyclonal antibody, dilution 1:500, Sigma, Product#A2052, Batch#103M4793), GAD65 + 67 (GAD65/67) (rabbit polyclonal antibody, dilution 1:200, Abcam, ab49832), and NeuN (mouse monoclonal antibody, dilution 1:1000, Millipore, Cat.#MAB377, Lot#NG1876252).
On the day of sacrifice, mice were perfused transcardially with 50 ml of saline solution followed by 80 ml of ice-cooled 4% paraformaldehyde in 0.1 M phosphate-buffered saline (PBS). The lumbar spinal cord (L4-5) was removed and placed in the same fixative for 2 h. The spinal cord was transferred to 30% sucrose (w/v) in 0.1 M PBS for 24 h for cryoprotection and then embedded in OCT compound using dry-ice-cooled hexane. Serial 10 -µm-thick spinal cord sections were prepared in a cryostat and mounted serially on 3-amino-propylethoxysilane-coated slides. The sections were processed for an immunofluorescence analysis according to previous methods.27 Sections were washed in 0.1 M PBS and incubated in 0.1 M PBS with 10% normal goat serum and 1% Triton X-100 at room temperature in order to block nonspecific staining. Sections for double-immunofluorescence staining were incubated simultaneously with two kinds of primary antibodies diluted with 0.1 M PBS containing 5% normal goat serum and 0.3% Triton X-100 at 4℃ for 48 h. After being incubated with the primary antibodies, sections were washed in 0.1 M PBS containing 0.1% Triton X-100 and then incubated with the secondary antibodies diluted with 0.1 M PBS containing 5% normal goat serum and 0.1% Triton X-100 at room temperature for 3 h. The secondary antibodies used for the detection of specific signals for NeuN or GABA and GAD65/67 were Alexa Fluor 568-labeled goat anti-mouse IgG (1:500 Jackson ImmunoResearch) and Alexa Fluor 488-labeled goat anti-rabbit IgG (1:500, Jackson ImmunoResearch), respectively. Sections were washed in 0.1 M PBS and mounted in Vectashield mounting medium with DAPI (Vector Labs, Burlingame, CA) in order to visualize nuclei. Negative control sections, which were processed without being incubated with the primary antibodies, showed no significant positive immunoreactivities. Fluorescent signals were detected using a confocal microscope (LSM700, Carl Zeiss, Oberkochen, Germany) equipped with an argon-helium laser and fluorescence microscope (BZ9000, KEYENCE, Osaka, Japan).
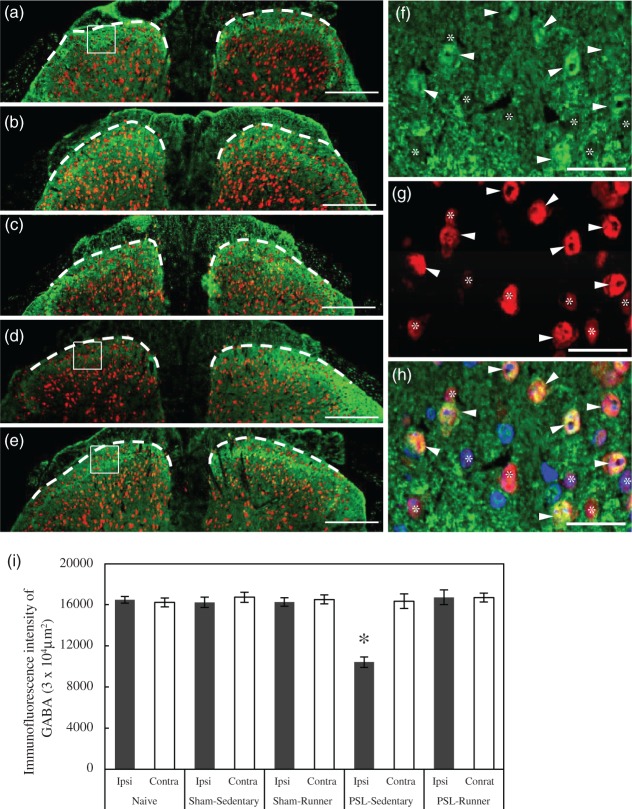
The quantification of double-immunopositive cells for GABA and NeuN in the superficial dorsal horn was performed according to previously described methods.28 Immunofluorescence images of the superficial dorsal horns were acquired using a fluorescence microscope at a magnification of 60×. A square of 104 µm2 in size was placed on the lateral, central, and medial parts of the superficial dorsal horns in microscope images using ImageJ software (version 1.48, National Institutes of Health, USA), and the number of immunopositive cells within it was counted. In this study, GABA+ neurons were defined as NeuN+ neurons with similar or more intense GABA immunoreactivities than those of the neuropils surrounding neurons (Figure 2(f) to (h)). The total number of GABA+ neurons within the squares of these three parts was calculated as the value of a spinal cord section, and the mean value of three randomly selected spinal cord sections from six sections per spinal cord was regarded as the number of GABA+ neurons per mouse.
Figure 2.
Changes in GABA immunoreactivities in the superficial dorsal horn following treadmill running. Lumbar spinal cord (L4-5) sections in (a) Naive, (b) Sham-Sedentary, (c) Sham-Runner, (d) PSL-Sedentary, and (e) PSL-Runner mice were immunostained with GABA (green) and NeuN (red) antibodies. The right and left sides of the dorsal horns indicate the contralateral and ipsilateral sides, respectively. The dotted lines drawn in each photomicrograph indicate the boundary between the dorsal horn and white matter. Bars: (a–e) = 200 µm. (f–h) Lumbar spinal cord sections in Naive mice were immunostained with GABA and NeuN antibodies, and nuclei were stained with DAPI. (f) and (g) indicate the immunoreactivities of GABA (green) and NeuN (red) in the superficial dorsal horn, respectively, and (h) three images including DAPI were merged. A square located in (a) indicates the typical position acquired in enlarged photomicrographs of the superficial dorsal horn. Arrowheads and asterisks indicate GABA+ NeuN+ and GABA− NeuN+ neurons, respectively. Bars: (f-h) = 20µm. (i) As shown in (a), a square of 104 µm2 in size was placed on the lateral, central, and medial parts of the superficial dorsal horns in microscope images, and the immunofluorescence intensity of GABA within it was quantified. GABA immunoreactivity was significantly weaker in PSL-Sedentary mice than in the other groups (n = 6, *p < 0.01). The significance of differences among groups was determined by a one-way ANOVA and Tukey’s post hoc test.
The immunofluorescence intensities of GABA and GAD65/67 in the spinal dorsal horn were quantified as follows. Immunofluorescence images of the superficial dorsal horn were acquired using a fluorescence microscope at a magnification of 60 x, and the acquisition conditions of the immunofluorescence images, such as the number of recording pixels, electrical shutter speed, gain, white balance, and black balance were fixed throughout the image analysis. A square of 104 µm2 in size was placed on the lateral, central, and medial parts of the superficial dorsal horns in microscope images and the immunofluorescence intensity within it was measured using ImageJ software (version 1.48, National Institutes of Health, USA). The immunofluorescence intensity of a neuron that showed negative immunoreactivity with GABA or GAD65/67 immunostaining was used to normalize the background level among the sections, and this value was subtracted from the values for the immunofluorescence intensities of GABA or GAD65/67 within the same square. Total immunofluorescence intensities within the squares of these three parts were calculated as the value of a spinal cord section, and the mean value of three randomly selected spinal cord sections from six sections per spinal cord was regarded as the immunofluorescence intensity per mouse. In order to avoid any bias in the results of the quantitative analysis, the investigators were blinded to all of the experimental groups throughout the quantitative analyses.
Statistical analysis
Quantitative data are presented as the mean ± standard error of the mean. A repeated-measures analysis of variance (ANOVA) followed by Tukey’s post hoc test was used to compare withdrawal thresholds or withdrawal latencies among the different experimental groups. Multiple comparisons of the number of immunopositive cells and immunofluorescence intensities among different experimental groups were performed using a one-way ANOVA followed by Tukey’s post hoc test. Differences were considered significant at #p < 0.05 and *p < 0.01.
Results
Effects of treadmill running on pain behaviors
We performed von Frey and plantar tests to examine the effects of treadmill running on pain behaviors in NPP model mice (Figure 1(b) and (c)). No marked differences in the pre-surgical data of withdrawal thresholds or withdrawal latencies were found among mice assigned to the five experimental groups. Naive, Sham-Sedentary, and Sham-Runner mice also did not display any significant alterations in withdrawal thresholds or withdrawal latencies throughout the experimental period, indicating that our treadmill running protocol had no effects on sensory sensitivity in mice without PSL.
Withdrawal thresholds (0.208 ± 0.031 g) and withdrawal latencies (3.60 ± 0.133 s) were markedly lower than pre-surgical values (withdrawal threshold = 1.222 ± 0.036 g; withdrawal latency = 10.94 ± 0.519 s) in PSL-Sedentary mice two days after PSL, and these decreased thresholds and latencies were maintained throughout the experimental period. Withdrawal thresholds in PSL-Runner mice were significantly higher at five days (0.459 ± 0.093 g) and six days (0.538 ± 0.103 g) than those in PSL-Sedentary mice (five days = 0.156 ± 0.005 g; six days = 0.144 ± 0.011 g) (p < 0.01) (Figure 1(b)). Withdrawal latencies in PSL-Runner mice five and six days after PSL (five days = 5.589 ± 0.289 s; six days = 6.322 ± 0.213 s) were also significantly higher than those in PSL-Sedentary mice (five days = 3.533 ± 0.117 s, p < 0.05; six days = 3.644 ± 0.073 s, p < 0.01) (Figure 1(c)). These results show that the treadmill running protocol used in this study effectively alleviated the pain behaviors of PSL model mice.
Effects of treadmill running on GABA production
In order to clarify the effects of treadmill running on GABA production in the superficial dorsal horn, transverse sections of the lumbar spinal cord were analyzed by immunolabeling. In Naive, Sham-Sedentary, and Sham-Runner mice, robust GABA+ immunoreactivities were observed in the superficial laminae of the dorsal horn (2(a) to (c)). GABA immunolabeling was markedly weaker in the ipsilateral side than in the contralateral side in PSL-Sedentary mice (Figure 2(d)); however, this reduction was clearly restored in PSL-Runner mice (Figure 2(e)).
Enlarged images of the superficial dorsal horn of Naive mice showed that a number of rounded GABA+ cells were colocalized with neuronal nuclei (NeuN)+ neurons and that neuropils consisting of dendrites, axons, and nerve terminals surrounding NeuN+ neurons were also densely immunostained (Figure 2(f) to (h)), which was consistent with previously reported immunolabeling patterns.17,29 We quantified GABA immunoreactivities in the superficial laminae including neurons and neuropils using an image analysis (Figure 2(i)). No significant difference was observed in GABA immunoreactivities in Naive, Sham-Sedentary, and Sham-Runner mice. A significant decrease was noted in GABA immunoreactivities in the ipsilateral side of PSL-Sedentary mice (Figure 2(i), p < 0.01), and this was prevented by treadmill running.
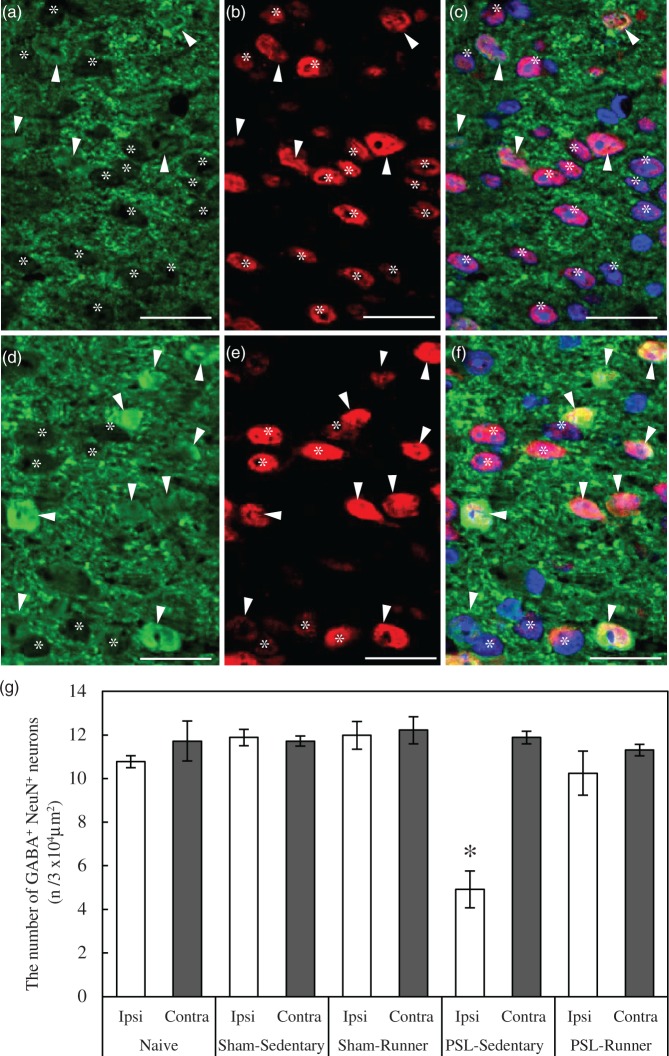
Inhibitory interneurons in the dorsal horn use GABA and/or glycine as neurotransmitters, and a previous study showed that nearly all of the inhibitory interneurons in the superficial dorsal horn are GABA-immunoreactive cells.30 Double immunostaining with GABA and NeuN antibodies revealed that GABA + NeuN + and GABA− NeuN+ neurons were detected in the superficial dorsal horn of Naive mice (Figure 2(f) to (h)). Therefore, we considered GABA+ NeuN+ neurons to be GABAergic inhibitory interneurons and investigated the effects of treadmill running on these interneurons (Figure 3(a) to (f)). Although no significant differences were observed in the number of GABA+ NeuN+ neurons in Naive, Sham-Sedentary, and Sham-Runner mice (Figure 3(g)), the number of GABAergic interneurons in PSL-Sedentary mice was significantly lower in the ipsilateral side than in the contralateral side (Figure 3(a) to (c), (g), p < 0.01). This reduction was clearly counteracted in PSL-Runner mice (Figure 3(d) to (f), (g)). These results show that treadmill running restores decreases in GABA levels in interneurons and neuropils in the superficial dorsal horn of PSL-Runner mice.
Figure 3.
Changes in GABA+ NeuN+ neurons in the superficial dorsal horn following treadmill running. (a,b) Immunoreactivities of GABA (green) and NeuN (red) in the ipsilateral superficial dorsal horn of PSL-Sedentary mice, and (c) three images including DAPI were merged. A typical position acquired in enlarged photomicrographs (a–c) in the ipsilateral dorsal horn was marked by the square located in Figure 3(d). (d,e) Immunoreactivities of GABA (green) and NeuN (red) in the ipsilateral superficial dorsal horn of PSL-Runner mice, and (f) three images including DAPI were merged. A typical position acquired in enlarged photomicrographs (d–f) in the ipsilateral dorsal horn was marked by the square located in Figure 3(e). Arrowheads and asterisks indicate GABA+ NeuN+ and GABA− NeuN+ neurons, respectively. Bars: (a–f) = 20 µm. (g) As shown in Figure 3(a), (d), and (e), squares of 104 µm2 in size were placed on the lateral, central, and medial parts, respectively, of the superficial dorsal horn in microscope images, and the number of GABA+ NeuN+ neurons within it was counted. The number of GABA+ NeuN+ neurons in the ipsilateral dorsal horn was significantly lower in PSL-Sedentary mice than in the other groups (n = 6, *p < 0.01). The significance of differences among groups was determined by a one-way ANOVA and Tukey’s post hoc test.
Effects of treadmill running on GAD65/67 production
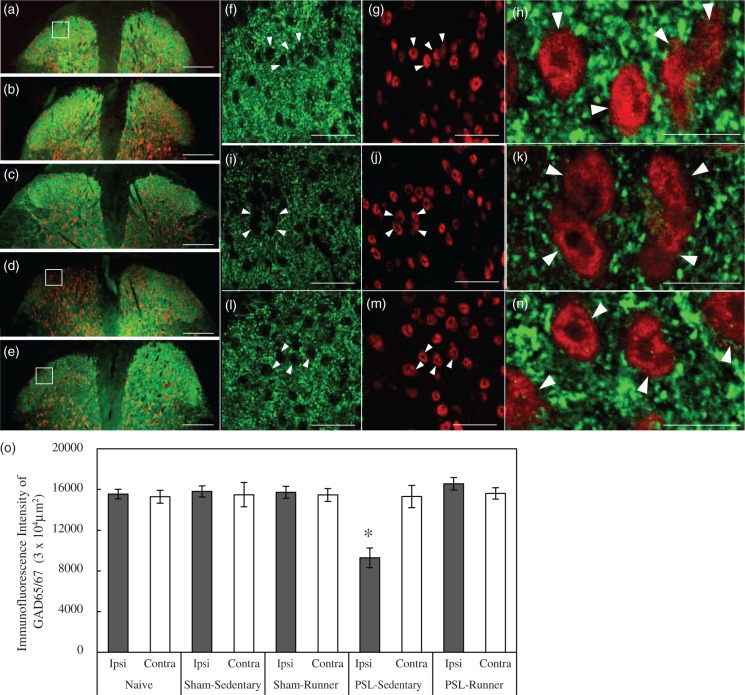
Most axon terminals in the spinal cord contain GAD65 and GAD67 isoforms, and both isoforms play critical roles in the production and attenuation of chronic pain.14 Therefore, we used an antibody (GAD65/67) that labels the GAD65 and GAD67 isoforms. When the lumbar spinal cord in Naive mice was immunostained, specific GAD65/67 immunoreactivities were widely observed in the dorsal horn (Figure 4(a)), and, in enlarged images, intense GAD65/67 immunoreactivities were detected as punctate structures in neuropils in the superficial dorsal horn (Figure 4(f) to (h)). Neuronal cell bodies did not show sufficient immunolabeling, which is consistent with previous findings (Figure 4(f) to (h)), and may be explained by rapid axonal transport into terminals and/or the usage of detergents such as Triton X-100 in immunohistochemical procedures.14,31
Figure 4.
Changes in GAD65/67 immunoreactivities in the superficial dorsal horn following treadmill running. Lumbar spinal cord (L4-5) sections in (a) Naive, (b) Sham-Sedentary, (c) Sham-Runner, (d) PSL-Sedentary, and (e) PSL-Runner mice were immunostained with GAD65/67 (green) and NeuN (red) antibodies. The right and left sides of the dorsal horns correspond to the contralateral and ipsilateral sides, respectively. The square located in (a) was enlarged in (f): GAD65/67 (green) and (g): NeuN (red). The square located in (d) was enlarged in (i): GAD65/67 (green) and (j): NeuN (red), and the square located in (e) was also enlarged in (l): GAD65/67 (green) and (m): NeuN (red). NeuN+ neurons (arrowheads) in (g), (j), and (m) were enlarged in (h), (k), and (n), respectively. Bars: (a–e) = 200 µm, (f, g, i, j, l, and m) = 30 µm, (h, k, and n) = 10 µm. Intense GAD65/67 immunoreactivities were detected in neuropils. (o) The immunofluorescence intensity of GAD65/67 in the superficial dorsal horn was quantified as described in the Methods section. GAD65/67 immunoreactivity levels were significantly lower in PSL-Sedentary mice than in the other groups (n = 6, *p < 0.01). The significance of differences among groups was determined by a one-way ANOVA and Tukey’s post hoc test.
No significant differences were observed in GAD65/67 immunolabeling in the dorsal horns of Naive, Sham-Sedentary, and Sham-Runner mice (Figure 4(a) to (c)). GAD65/67 immunoreactivity was markedly lower in the ipsilateral side than in the contralateral side of PSL-Sedentary mice (Figure 4(d), (i) to (k)); however, this reduction was prevented by treadmill running (Figure 4(e), (l) to (n)). An image analysis revealed a significant reduction in GAD65/67 levels in the ipsilateral side of PSL-Sedentary mice (Figure 4(o), p < 0.01) but not in that of PSL-Runner mice (Figure 4(o)). Thus, the present results show that treadmill running restores PSL-induced reductions in GAD65/67 production, and, thus, GABA levels may be retained in the interneurons and neuropils of the superficial dorsal horn.
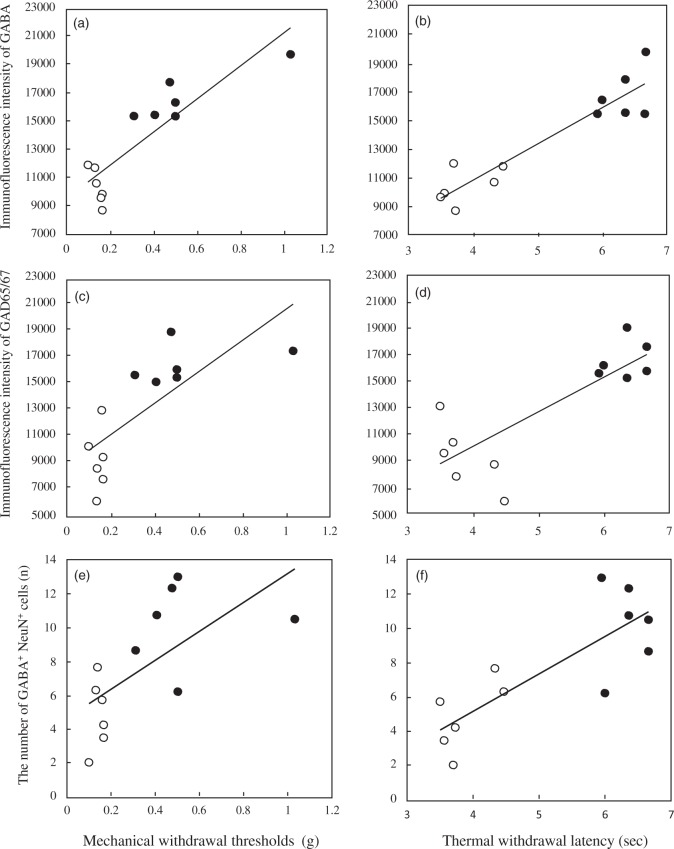
We then investigated whether increases in GABA and GAD65/67 levels in the superficial dorsal horn following treadmill running accelerated the attenuation of pain behaviors. A positive correlation was observed between GABA levels and the thresholds of von Frey (Figure 5(a), p < 0.001) or plantar tests (Figure 5(b), p < 0.001), as well as between GAD65/67 levels and the thresholds of von Frey (Figure 5(c), p < 0.01) or plantar tests (Figure 5(d), p < 0.01) in PSL-Sedentary and -Runner mice. Positive correlations were also observed between the number of GABA+ NeuN+ neurons and the thresholds of von Frey (Figure 5(e), p < 0.05) or plantar tests (Figure 5(f), p < 0.01) in PSL-Sedentary and -Runner mice. These results suggest that increases in GABA and GAD65/67 levels and GABAergic interneurons in the superficial ipsilateral dorsal horn may contribute to EIH.
Figure 5.
Relationships between pain behavior thresholds and GABA and GAD65/67 levels or GABAergic interneuron numbers in superficial dorsal horns of PSL-Sedentary and -Runner mice. (a) A positive correlation was observed between GABA levels and the thresholds of von Frey (a, r = 0.874, n = 12, p < 0.001) or plantar tests (b, r = 0.929, n = 12, p < 0.001). A positive correlation was observed between GAD65/67 levels and the thresholds of von Frey (c, r = 0.753, n = 12, p < 0.01) or plantar tests (d, r = 0.817, n = 12, p < 0.01). A positive correlation was observed between the number of GABA + NeuN + neurons and thresholds of von Frey (e, r = 0.645, n = 12, p < 0.05) or plantar tests (f, r = 0.813, n = 12, p < 0.01). Open and closed circles indicate the values of PSL-Sedentary and -Runner mice, respectively.
Discussion
PSL, CCI, and SNL are frequently used as peripheral nerve injury models to generate NPP, and physical exercise attenuates mechanical allodynia and heat hyperalgesia in these model animals. Exercise paradigms such as treadmill running and swimming are now primarily used in EIH research, and their effectiveness has been demonstrated in many related studies.1,2,6,8,9,32 The effectiveness of EIH appears to depend on the timing of the initiation of treadmill running after surgery. For example, when CCI or sciatic nerve crush model animals started to run on a treadmill three days after surgery, significant improvements in pain behaviors were observed within four days post-exercise (within seven days of surgery).1,3 When treadmill running was initiated seven days after surgery in SNL-model rats in another study, the attenuation of pain hypersensitivity was initially observed 14 days post-exercise.9 In the present study, PSL mice started to run on a treadmill two days after surgery at a running speed of 7 m/min for 60 min, which corresponds to approximately 64% of maximal oxygen uptake (VO2max) based on a previous study by Schefer and Talan.33 Mechanical allodynia and heat hyperalgesia were significantly attenuated three days post-exercise. Thus, the present results, in accordance with previous studies, further confirmed the ability of treadmill running to produce EIH and suggest that treadmill running started at an early stage after surgery accelerates the development of EIH.
Although previous studies found marked reductions in GABA levels and the loss of GABA neurons in the spinal dorsal horn of NPP model animals,10–12 contradictory findings have also been reported.29,34 Therefore, the fate of GABAergic neurons in the dorsal horn following peripheral nerve injury remains unclear due to the difficulties associated with immunolabeling GABAergic neurons and inconsistent methods used to analyze GABA levels. In contrast to GABA, the number of enhanced green fluorescent protein (EGFP)+ GAD67 neurons was reported to be significantly decreased in the superficial dorsal horn of SNL model mice seven days post-surgery.35 Lorenzo et al.15 also showed that the density of axon terminals with GAD65 in the dorsal horn of CCI model rats was markedly reduced in laminae I–II from five days post-surgery, with the largest decrease occurring after approximately three to four weeks. A previous study demonstrated marked reductions in GAD65 but not GAD67 in the ipsilateral dorsal horn of NPP model rats within six days of surgery.36 Therefore, further studies to investigate changes in GADs and GABA associated with peripheral nerve injury in the dorsal horn will be important for elucidating the implications of GABAergic inhibition on the production of NPP. The present study showed that PSL surgery not only significantly decreased GAD65/67 levels but also induced marked reductions in GABA levels in the superficial dorsal horn. Therefore, our results suggest that reductions in GABA and GAD65/67 levels in the dorsal horn are sufficient for abolishing GABAergic inhibition, leading to NPP in PSL-Sedentary mice.
A previous study reported a marked reduction in GAD65 immunoreactivity in the ipsilateral dorsal horn in CCI and spared nerve injury model rats, and this reduction was attributed to apoptotic cell death, which was the most prominent seven days post-surgery.36 In contrast, other studies found no significant loss in GABAergic neurons in the superficial dorsal horn 14 and 28 days post-surgery in spared nerve injury and CCI model rats,29,34 and all the apoptotic cells observed after nerve injury were microglia.37 Thus, the loss of GABAergic neurons via apoptotic cell death in NPP model animals remains controversial. In the present study, significant decreases were observed in the number of GABAergic interneurons in the superficial dorsal horn of PSL-Sedentary mice. Cell death does not account for the loss of GABAergic interneurons, because immunostaining with a cleaved caspase-3 antibody, an indicator of apoptotic cells did not reveal any positive immunoreactivity in the dorsal horn of PSL-Sedentary mice seven days post-surgery (data not shown). Another reasonable explanation for the reduction observed in GABAergic interneurons involves the epigenetic modification of GABA synthesis in GABAergic interneurons in response to the altered neuronal environment after nerve injury. Especially, a feature in class IIa subfamily member of histone deacetylases (HDACs) including HDAC4 is synaptic activity-dependent nucleo-cytoplasmic shuttling,38 and in neurons and muscle cells, HDAC4 is imported into the nucleus from the cytoplasm in response to reductions in neural inputs.39–41 In the nucleus, HDAC4 acts as a transcriptional repressor, while the nuclear export of HDAC4 relieves transcriptional repression.42 A previous study demonstrated that reductions in histone H3 acetylation in the Gad2 promoter in the brainstem of chronic pain model animals resulted in its transcriptional repression, and some HDACs including HDAC4 may be associated with the regulatory regions of Gad2.16 Moreover, the neuronal activity of GABAergic neurons in the dorsal horn is markedly impaired in CCI model animals.22 Based on these findings, our results indicate that reduced neuronal inputs into GABAergic interneurons in PSL-Sedentary mice may result in the nuclear import of HADC4, and, thus, the down-regulation of GAD65 by hypoacetylated Gad2 via its transcriptional repression. Since GAD65 principally accelerates the production of chronic pain over GAD67,15,16,36 the impaired production of GAD65 via epigenetic modifications to Gad2 in GABAergic interneurons may account for the exacerbated pain behaviors observed in PSL-Sedentary mice.
Multiple mechanisms have been proposed for EIH. Almeida et al.8 showed that the reduced phosphorylation of phospholipase Cγ-1 and inhibition of activated glial cells in the spinal dorsal horn in PSL model mice may play a role in the development of EIH. Swimming in PSL model mice also attenuates pain behaviors by reducing pro-inflammatory cytokines in injured sciatic nerves.32 Furthermore, inhibitory activity mediated by brain-derived neurotrophic factor, opioids, and serotonin may underlie EIH.4,8,9 The present study showed that treadmill running at approximately 64% VO2max not only induced an increase in GABA levels in interneurons accompanied by an increase of GAD65/67 but also reversed enhanced pain behaviors. We also found positive correlations between pain behaviors and GABA and GAD65/67 levels or GABAergic interneuron numbers in the superficial ipsilateral dorsal horn. Thus, our results suggest that the increased production of GABA via the up-regulation of GADs in interneurons in response to treadmill running produces EIH. Dai et al.43 showed that when c-Fos immunostaining was used to localize treadmill running-activated neurons in the cat spinal dorsal horn, labeled cells were widely detected in the dorsal horn, especially laminae III-IV,43 indicating that dorsal horn neurons are activated during treadmill running due to afferent feedback from the exercising limb muscles via group Ia–IV muscle afferents that project into distinct laminae.44–47 Based on these findings, our results suggest that an increase in the neural input into GABAergic interneurons in the dorsal horn by treadmill running allows for the nuclear export of HDAC4 and thereby contributing to the maintenance of GAD65 production. Therefore, the neuronal activity-dependent epigenetic modification causing the hyperacetylation of Gad2 in GABAergic interneurons may play a role in EIH.
Conclusions
We herein demonstrated that mechanical allodynia and heat hyperalgesia were significantly attenuated in PSL-Runner mice. Furthermore, treadmill running prevented PSL-induced reductions in GAD65/67 production, leading to the retention of GABA in interneurons and neuropils in the superficial dorsal horn. Therefore, we conclude that improvements in impaired GABAergic inhibition represent a new pathway for EIH. In addition, electrophysiological analysis to demonstrate the exercise-induced functional alterations in GABAergic inhibitory system that leads to EIH would be necessary to further support the conclusion. On the other hand, GABAergic interneurons in the spinal dorsal horn may be the dominant candidate for the development of EIH. In addition, both isoforms at the protein and mRNA levels are present in the RVM, and these GABAergic RVM neurons massively project into the spinal dorsal horn.16,48–52 These GABAergic RVM neurons were also shown to participate in pain inhibition.16 Therefore, GABAergic neurons in the RVM may be another potential candidate involved in the development of EIH and warrant further study. Furthermore, although the potential mechanisms other than GABAergic inhibitory system in producing EIH have been proposed, we do not know whether such mechanisms also work under the treadmill running protocol used in this study. In addition, we have not examined yet whether the impaired GABA and GAD65/67 productions are also improved by other treadmill running protocols. The answers would expand our knowledge and understanding of the mechanisms underlying EIH.
Author Contributions
KK, ES and FT planned this study. KK and ST performed the behavioral experiments. KK performed immunofluorescence staining and the image analysis. KK and ES wrote the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by research grants from KAKENHI (Grants-in-Aid for Scientific Research [C] 15K01427 and 15K08677 of the Japan Society for the Promotion of Science).
References
- 1.Bobinski F, Martins DF, Bratti T, et al. Neuroprotective and neuroregenerative effects of low-intensity aerobic exercise on sciatic nerve crush injury in mice. Neuroscience 2011; 194: 337–348. [DOI] [PubMed] [Google Scholar]
- 2.Chen YW, Li YT, Chen YC, et al. Exercise training attenuates neuropathic pain and cytokine expression after chronic constriction injury of rat sciatic nerve. Anesth Analg 2012; 114: 1330–1337. [DOI] [PubMed] [Google Scholar]
- 3.Cobianchi S, Casals-Diaz L, Jaramillo J, et al. Differential effects of activity dependent treatments on axonal regeneration and neuropathic pain after peripheral nerve injury. Exp Neurol 2013; 240: 157–167. [DOI] [PubMed] [Google Scholar]
- 4.Korb A, Bonetti LV, da Silva SA, et al. Effect of treadmill exercise on serotonin immunoreactivity in medullary raphe nuclei and spinal cord following sciatic nerve transection in rats. Neurochem Res 2010; 35: 380–389. [DOI] [PubMed] [Google Scholar]
- 5.Shankarappa SA, Piedras-Renteria ES, Stubbs EB., Jr Forced-exercise delays neuropathic pain in experimental diabetes: effects on voltage-activated calcium channels. J Neurochem 2011; 118: 224–236. [DOI] [PubMed] [Google Scholar]
- 6.Shen J, Fox LE, Cheng J. Swim therapy reduces mechanical allodynia and thermal hyperalgesia induced by chronic constriction nerve injury in rats. Pain Med 2013; 14: 516–525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cobianchi S, Marinelli S, Florenzano F, et al. Short- but not long-lasting treadmill running reduces allodynia and improves functional recovery after peripheral nerve injury. Neuroscience 2010; 168: 273–287. [DOI] [PubMed] [Google Scholar]
- 8.Almeida C, DeMaman A, Kusuda R, et al. Exercise therapy normalizes BDNF upregulation and glial hyperactivity in a mouse model of neuropathic pain. Pain 2015; 156: 504–513. [DOI] [PubMed] [Google Scholar]
- 9.Stagg NJ, Mata HP, Ibrahim MM, et al. Regular exercise reverses sensory hypersensitivity in a rat neuropathic pain model: role of endogenous opioids. Anesthesiology 2011; 114: 940–948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Castro-Lopes JM, Tavares I, Coimbra A. GABA decreases in the spinal cord dorsal horn after peripheral neurectomy. Brain Res 1993; 620: 287–291. [DOI] [PubMed] [Google Scholar]
- 11.Eaton MJ, Plunkett JA, Karmally S, et al. Changes in GAD- and GABA-immunoreactivity in the spinal dorsal horn after peripheral nerve injury and promotion of recovery by lumbar transplant of immortalized serotonergic precursors. J Chem Neuroanat 1998; 16: 57–72. [DOI] [PubMed] [Google Scholar]
- 12.Ibuki T, Hama AT, Wang XT, et al. Loss of GABA-immunoreactivity in the spinal dorsal horn of rats with peripheral nerve injury and promotion of recovery by adrenal medullary grafts. Neuroscience 1997; 76: 845–858. [DOI] [PubMed] [Google Scholar]
- 13.Erlander MG, Tillakaratne NJ, Feldblum S, et al. Two genes encode distinct glutamate decarboxylases. Neuron 1991; 7: 91–100. [DOI] [PubMed] [Google Scholar]
- 14.Mackie M, Hughes DI, Maxwell DJ, et al. Distribution and colocalisation of glutamate decarboxylase isoforms in the rat spinal cord. Neuroscience 2003; 119: 461–472. [DOI] [PubMed] [Google Scholar]
- 15.Lorenzo LE, Magnussen C, Bailey AL, et al. Spatial and temporal pattern of changes in the number of GAD65-immunoreactive inhibitory terminals in the rat superficial dorsal horn following peripheral nerve injury. Mol Pain 2014; 57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang Z, Cai YQ, Zou F, et al. Epigenetic suppression of GAD65 expression mediates persistent pain. Nat Med 2011; 17: 1448–1455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vaysse L, Sol JC, Lazorthes Y, et al. GABAergic pathway in a rat model of chronic neuropathic pain: modulation after intrathecal transplantation of a human neuronal cell line. Neurosci Res 2011; 69: 111–120. [DOI] [PubMed] [Google Scholar]
- 18.Eaton MJ, Martinez MA, Karmally S. A single intrathecal injection of GABA permanently reverses neuropathic pain after nerve injury. Brain Res 1999; 835: 334–339. [DOI] [PubMed] [Google Scholar]
- 19.Cui JG, O’Connor WT, Ungerstedt U, et al. Spinal cord stimulation attenuates augmented dorsal horn release of excitatory amino acids in mononeuropathy via a GABAergic mechanism. Pain 1997; 73: 87–95. [DOI] [PubMed] [Google Scholar]
- 20.Ultenius C, Song Z, Lin P, et al. Spinal GABAergic mechanisms in the effects of spinal cord stimulation in a rodent model of neuropathic pain: is GABA synthesis involved? Neuromodulation 2013; 16: 114–120. [DOI] [PubMed] [Google Scholar]
- 21.Vit JP, Ohara PT, Sundberg C, et al. Adenovector GAD65 gene delivery into the rat trigeminal ganglion produces orofacial analgesia. Mol Pain 2009; 5: 42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Leitner J, Westerholz S, Heinke B, et al. Impaired excitatory drive to spinal GABAergic neurons of neuropathic mice. PLoS One 2013; 8: e73370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Meisner JG, Marsh AD, Marsh DR. Loss of GABAergic interneurons in laminae I-III of the spinal cord dorsal horn contributes to reduced GABAergic tone and neuropathic pain after spinal cord injury. J Neurotrauma 2010; 27: 729–737. [DOI] [PubMed] [Google Scholar]
- 24.Seltzer Z, Dubner R, Shir Y. A novel behavioral model of neuropathic pain disorders produced in rats by partial sciatic nerve injury. Pain 1990; 43: 205–218. [DOI] [PubMed] [Google Scholar]
- 25.Horiguchi N, Ago Y, Hasebe S, et al. Isolation rearing reduces mechanical allodynia in a mouse model of chronic inflammatory pain. Pharmacol Biochem Behav 2013; 113: 46–52. [DOI] [PubMed] [Google Scholar]
- 26.Imbe H, Okamoto K, Donishi T, et al. Involvement of descending facilitation from the rostral ventromedial medulla in the enhancement of formalin-evoked nocifensive behavior following repeated forced swim stress. Brain Res 2010; 1329: 103–112. [DOI] [PubMed] [Google Scholar]
- 27.Kami K, Senba E. In vivo activation of STAT3 signaling in satellite cells and myofibers in regenerating rat skeletal muscles. J Histochem Cytochem 2002; 50: 1579–1589. [DOI] [PubMed] [Google Scholar]
- 28.Clark AK, Gentry C, Bradbury EJ, et al. Role of spinal microglia in rat models of peripheral nerve injury and inflammation. Eur J Pain 2007; 11: 223–230. [DOI] [PubMed] [Google Scholar]
- 29.Polgar E, Hughes DI, Riddell JS, et al. Selective loss of spinal GABAergic or glycinergic neurons is not necessary for development of thermal hyperalgesia in the chronic constriction injury model of neuropathic pain. Pain 2003; 104: 229–239. [DOI] [PubMed] [Google Scholar]
- 30.Polgar E, Durrieux C, Hughes DI, et al. A quantitative study of inhibitory interneurons in laminae I-III of the mouse spinal dorsal horn. PLoS One 2013; 8: e78309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhao C, Eisinger B, Gammie SC. Characterization of GABAergic neurons in the mouse lateral septum: a double fluorescence in situ hybridization and immunohistochemical study using tyramide signal amplification. PLoS One 2013; 8: e73750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kuphal KE, Fibuch EE, Taylor BK. Extended swimming exercise reduces inflammatory and peripheral neuropathic pain in rodents. J Pain 2007; 8: 989–997. [DOI] [PubMed] [Google Scholar]
- 33.Schefer V, Talan MI. Oxygen consumption in adult aged C57BL/6J mice during acute treadmill exercise of different intensity. Exp Gerontol 1996; 31: 387–392. [DOI] [PubMed] [Google Scholar]
- 34.Polgar E, Todd AJ. Tactile allodynia can occur in the spared nerve injury model in the rat without selective loss of GABA or GABA(A) receptors from synapses in laminae I-II of the ipsilateral spinal dorsal horn. Neuroscience 2008; 156: 193–202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yowtak J, Wang J, Kim HY, et al. Effect of antioxidant treatment on spinal GABA neurons in a neuropathic pain model in the mouse. Pain 2013; 154: 2469–2476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Moore KA, Kohno T, Karchewski LA, et al. Partial peripheral nerve injury promotes a selective loss of GABAergic inhibition in the superficial dorsal horn of the spinal cord. J Neurosci 2002; 22: 6724–6731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Polgar E, Hughes DI, Arham AZ, et al. Loss of neurons from laminas I-III of the spinal dorsal horn is not required for development of tactile allodynia in the spared nerve injury model of neuropathic pain. J Neurosci 2005; 25: 6658–6666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fitzsimons HL. The Class IIa histone deacetylase HDAC4 and neuronal function: nuclear nuisance and cytoplasmic stalwart? Neurobiol Learn Mem 2015; 123: 149–158. [DOI] [PubMed] [Google Scholar]
- 40.Chawla S, Vanhoutte P, Arnold FJ, et al. Neuronal activity-dependent nucleocytoplasmic shuttling of HDAC4 and HDAC5. J Neurochem 2003; 85: 151–159. [DOI] [PubMed] [Google Scholar]
- 41.Cohen TJ, Waddell DS, Barrientos T, et al. The histone deacetylase HDAC4 connects neural activity to muscle transcriptional reprogramming. J Biol Chem 2007; 282: 33752–33759. [DOI] [PubMed] [Google Scholar]
- 42.Cohen TJ, Barrientos T, Hartman ZC, et al. The deacetylase HDAC4 controls myocyte enhancing factor-2-dependent structural gene expression in response to neural activity. FASEB J 2009; 23: 99–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Paroni G, Cernotta N, Dello Russo C, et al. PP2A regulates HDAC4 nuclear import. Mol Biol Cell 2008; 19: 655–667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dai X, Noga BR, Douglas JR, et al. Localization of spinal neurons activated during locomotion using the c-fos immunohistochemical method. J Neurophysiol 2005; 93: 3442–3452. [DOI] [PubMed] [Google Scholar]
- 45.Jankowski MP, Rau KK, Ekmann KM, et al. Comprehensive phenotyping of group III and IV muscle afferents in mouse. J Neurophysiol 2013; 109: 2374–2381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ling LJ, Honda T, Shimada Y, et al. Central projection of unmyelinated (C) primary afferent fibers from gastrocnemius muscle in the guinea pig. J Comp Neurol 2003; 461: 140–150. [DOI] [PubMed] [Google Scholar]
- 47.Maxwell DJ, Riddell JS. Axoaxonic synapses on terminals of group II muscle spindle afferent axons in the spinal cord of the cat. Eur J Neurosci 1999; 11: 2151–2159. [DOI] [PubMed] [Google Scholar]
- 48.Watson AH, Bazzaz AA. GABA and glycine-like immunoreactivity at axoaxonic synapses on 1a muscle afferent terminals in the spinal cord of the rat. J Comp Neurol 2001; 433: 335–348. [DOI] [PubMed] [Google Scholar]
- 49.Morgan MM, Whittier KL, Hegarty DM, et al. Periaqueductal gray neurons project to spinally projecting GABAergic neurons in the rostral ventromedial medulla. Pain 2008; 140: 376–386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pedersen NP, Vaughan CW, Christie MJ. Opioid receptor modulation of GABAergic and serotonergic spinally projecting neurons of the rostral ventromedial medulla in mice. J Neurophysiol 2011; 106: 731–740. [DOI] [PubMed] [Google Scholar]
- 51.Winkler CW, Hermes SM, Chavkin CI, et al. Kappa opioid receptor (KOR) and GAD67 immunoreactivity are found in OFF and NEUTRAL cells in the rostral ventromedial medulla. J Neurophysiol 2006; 96: 3465–3473. [DOI] [PubMed] [Google Scholar]
- 52.Aicher SA, Hermes SM, Whittier KL, et al. Descending projections from the rostral ventromedial medulla (RVM) to trigeminal and spinal dorsal horns are morphologically and neurochemically distinct. J Chem Neuroanat 2012; 43: 103–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hossaini M, Goos JA, Kohli SK, et al. Distribution of glycine/GABA neurons in the ventromedial medulla with descending spinal projections and evidence for an ascending glycine/GABA projection. PLoS One 2012; 7: e35293. [DOI] [PMC free article] [PubMed] [Google Scholar]