Abstract
The Drosophila wing imaginal disc is a sac-like structure that is composed of two opposing cell layers: peripodial epithelium (PE, also known as squamous epithelia) and disc proper (DP, also known as pseudostratified columnar epithelia). The molecular mechanism of cell morphogenesis has been well studied in the DP but not in the PE. Although proper Dpp signalling activity is required for proper PE formation, the detailed regulation mechanism is poorly understood. Here, we found that the Dpp target gene sal is only expressed in DP cells, not in PE cells, although pMad is present in the PE. Increasing Dpp signalling activity cannot activate Sal in PE cells. The absence of Sal in the PE is essential for PE formation. The ectopic expression of sal in PE cells is sufficient to increase the PE cell height. Down-regulation of sal in the DP reduced DP cell height. We further demonstrated that the known PE cell height regulator Lines, which can convert PE into a DP cell fate, is mediated by sal mis-activation in PE because sal-RNAi and lines co-expression largely restores PE cell morphology. By revealing the microtubule distribution, we demonstrated that Lines- and Sal-heightened PE cells are morphologically similar to the intermediate cell with cuboidal morphology.
Cell shape changes occur throughout various developmental processes, such as Drosophila gastrulation1,2 and neural tube formation in vertebrates3,4. Reorganization of the microtubule cytoskeleton contributes to cell shape changes5,6,7 and thereby regulates the morphogenesis of tissues or organisms. In the Drosophila wing imaginal disc, all cells have a cuboidal shape in both cell layers in the 1st and early 2nd instar stages. During the late 2nd and 3rd instar stages, one layer of the cells is flattened to squamous epithelium (also known as peripodial epithelium, PE), whereas the opposite layer of cells elongates to columnar epithelium (also known as disc proper, DP). The intermediate cells at the wing disc margin linking the PE and DP exhibit a cuboidal shape8,9. Thus, the wing disc is a sac-like structure composed of two opposing cell layers10,11.
Several signalling pathways have been implicated in epithelium morphogenesis in the wing disc. Decapentaplegic (DPP) signalling activity is required to ensure the correct architecture of DP epithelium12,13. Dpp activity via Rho1 promotes the elongation of DP cells in a cell-autonomous manner9. A T-box transcription factor Optomotor-blind (Omb), one of the Dpp targets, is expressed in a gradient manner along the anterior-posterior axis to ensure correct DP morphology14. Wingless signalling regulates the apical cell circumference of DP cells by a proximo-distal gradient15. Together with Vestigial (Vg), Wingless signalling maintains DP cell elongation during the late larval stage16. DPP signalling is involved in PE cell morphogenesis. Mutant clones lacking Dpp receptor activity cannot form in the eye PE and display reduced size and aberrant shape in the wing PE17. When Daughters against Dpp (Dad), a suppressor of DPP signal transduction, is overexpressed in the wing PE, a portion of the wing PE cells are elongated8.
In addition to Dpp signalling, Hedgehog (Hh) signalling and other putative transcription factors are also important for PE morphogenesis. Clones lacking Hedgehog (Hh) function in the eye PE are smaller than their wild-type twinspots, and the anterior ventral domains of both PE and DP are lacking in these clones11. Similarly, the wing disc is smaller in size and fails to form the PE in hh mutants8. In addition, the putative transcription factor Lines (Lin) promotes elongation of the PE and converts these cells into a DP cell fate. Lin is also necessary for the expression of the Dpp target gene spalt (sal)18.
Although appropriate Dpp signalling activity is necessary for PE morphogenesis8,17, the downstream mediators of Dpp signalling remain unknown. Here, we report that the known Dpp target genes, including sal and omb, are not expressed in PE. The absence of sal but not omb insures correct PE morphogenesis. The ectopic expression of sal in the PE is sufficient to elongate PE. Lin requires sal function to elongate the PE because suppressing sal can rescue the phenotype induced by lin ectopic expression. When the microtubule distribution is visualized, Sal-elongated PE cells are morphologically similar to cubically intermediate cells that normally link the PE and DP layers.
Results and Discussion
sal is not expressed in the PE
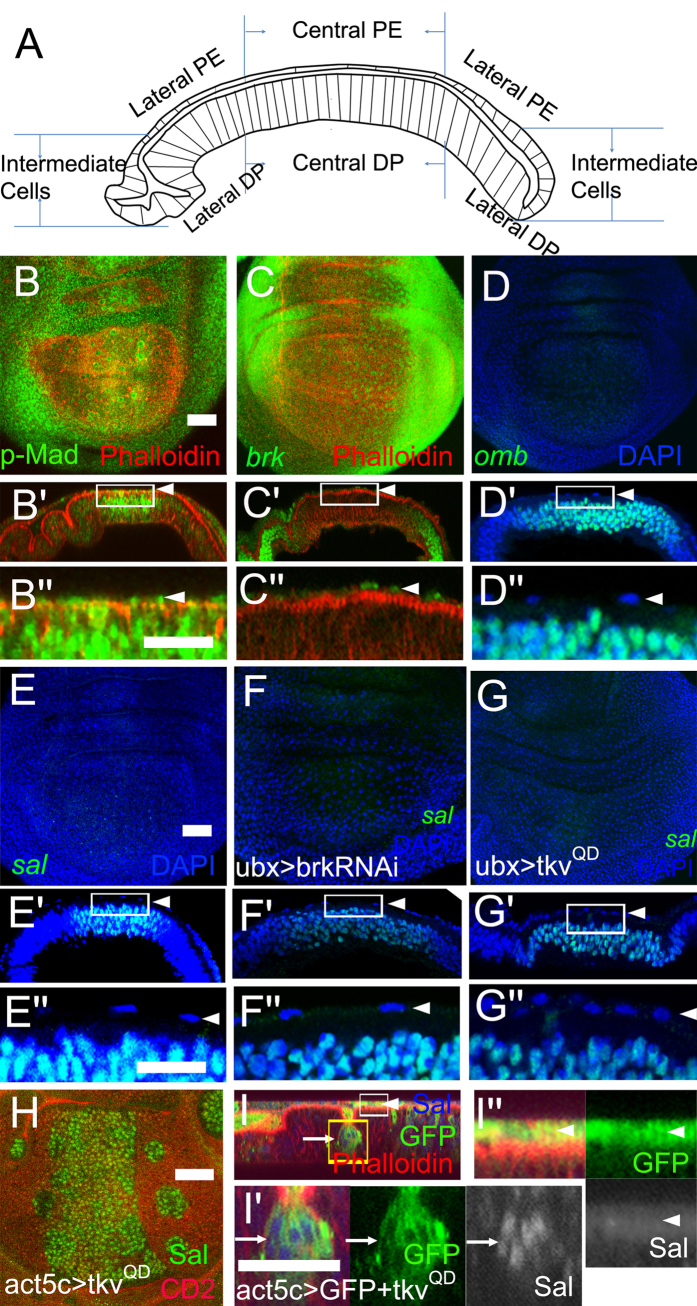
The wing disc is a sac-like structure composed of PE, DP, and intermediate cells linking the PE and DP. An x-z view of the wing disc is schematically presented in Fig. 1A. To investigate the potential role of Dpp signalling in PE morphogenesis, we first revealed the distribution of Dpp signalling activity in late 3rd instar (L3) wing imaginal discs in both the x-y and x-z views. Using an antibody of phospho-Mothers against dpp (pMad)19,20 to reveal Dpp signal transduction activity, we found that Dpp signal transduction was ubiquitously present in both the PE and DP (Fig. 1B–B″)21. The pMad level was relatively reduced in the central PE compared with the central DP (Fig. 1B′,B″, arrowheads). Then, we detected Dpp target gene expression patterns in the PE. The main Dpp target genes are brinker (brk), omb, and sal in the L3 wing discs22,23,24. brk was transcribed in both the PE and DP, with a relatively weaker level in the PE, as indicated by a brk-lacZ reporter (Fig. 1C–C″, arrowheads). However, both omb and sal were transcribed only in the DP, not in the PE (Fig. 1D–E″, arrowheads). These data indicate that the Dpp target genes omb and sal are asymmetrically expressed in the PE and DP. Brk is also a repressor of other Dpp target genes, including sal and omb, and thereby restricts their expression domains to the medial DP region24,25. The presence of Brk in the PE might be a direct cause of the absence of Omb and Sal in the PE. To assess this possibility, brk-RNAi was expressed in the PE. Sal expression was not detectable in the central PE (Fig. 1F′,F″, arrowheads). The efficiency of brk-RNAi was demonstrated by the elevation of Brk targets Omb and Sal in lateral wing discs of C765>brk-RNAi (data not shown). To further confirm that Dpp signalling cannot induce sal expression in the PE, a constitutive active form of the Dpp receptor tkvQDwas expressed in the PE. Sal was not induced in central PE (Fig. 1G–G″). When tkvQDclones were generated, Sal was induced only in clones within the DP (Fig. 1H,I,I′) and not in clones located in the PE (Fig. 1I,I″). Similarly, Omb was not induced in clones located in the PE (Fig. S-1B, arrowhead). Ubiquitous expression of tkvQDfailed to induce Omb in the PE (Fig. S-1C′,C″, arrowheads). These data demonstrate that Dpp signalling cannot activate omb and sal in PE.
Figure 1. sal is not induced by Dpp signalling in the PE.
In this and subsequent figures, late L3 wing discs are used, otherwise developmental stage is indicated. x-y views are oriented with dorsal up and anterior left. Cryosections (x-z views) along the dorsal-ventral boundary are oriented with apical up and anterior left. Scale bars are 50 μM. In x-y views of (B–G) panels are focused on the PE plane. H is focused on the DP plane. Others are x-z views of the wing disc. (A) Schematic drawing of wing disc structure in x-z view. The wing disc is a sac-like structure composed of two opposing cell layers with apical sides facing an internal lumen. (B–B″) pMad staining in the wing disc. Note that pMad is detected in the PE (arrowheads) and the DP. Boxed region in B′ is presented at a higher magnification in B″. (C–C″) brk expression pattern in the wing disc. Note that brk is transcribed in both the PE (arrowheads) and the DP as reveal by the brk-lacZ reporter. Boxed region in C′ is presented at a higher magnification in C″. (D–D″) omb expression pattern in the wing disc. Note that omb is only transcribed in the DP, not the PE (arrowheads), as revealed by the omb-lacZ reporter. Boxed region in D′ is presented at a higher magnification in D″. (E-E″) sal expression pattern in the wing disc. Note that sal is only transcribed in the DP, not in the PE (arrowheads), as revealed by the sal-lacZ reporter. Boxed region in E′ is presented at a higher magnification in E″. (F–F″) Expression of brk-RNAi does not induce Sal in the PE (arrowheads). Boxed region in F′ is presented at a higher magnification in F″. (G) Expression of tkvQD does not induce Sal in the PE (arrowheads). Boxed region in G′ is presented at a higher magnification in G″. (H-I″) Expression of tkvQD in clones induced Sal in the DP (arrow) but not in the PE (arrowhead). Boxed regions in I are presented at higher magnifications in I′ (a DP clone, arrow) and I″ (a PE clone, arrowhead).
The expression patterns of Dpp target genes are well studied in wing DP. Dpp controls target genes (sal and omb) expression indirectly through repression of the transcriptional repressor Brk24,26,27. Dpp target gene expression patterns have not been studied in wing PE to date. The above results revealed that pMad is ubiquitously present in both DP and PE (Fig. 1B–B″). However, brk-lacZ was still present in PE (Fig. 1C–C″). Either suppressing brk or elevating Dpp signalling by expressing tkvQD cannot induce Sal and Omb in the PE (Fig. 1H–I″ and Fig. S-1). Except for Lin (Fig. 2K)18, other factors, such as Bowl, Wg, and EGFR, cannot induce Sal in the PE (Fig. S-2). The factors that suppress sal in the PE require further investigation.
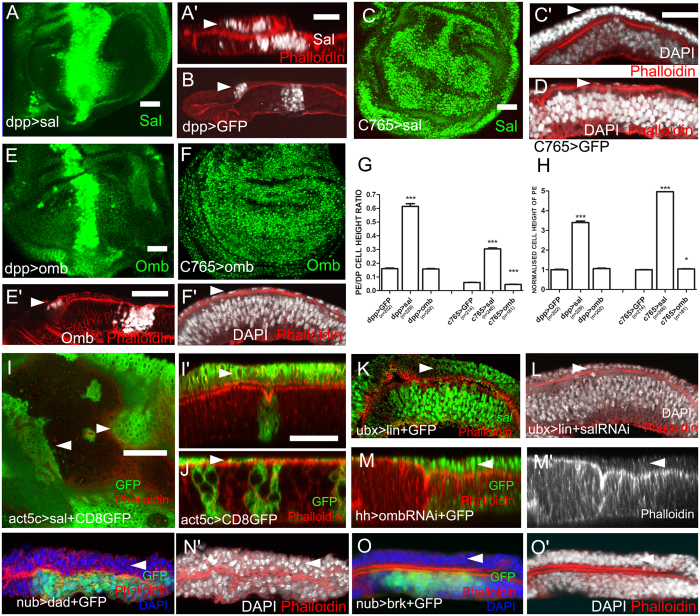
Figure 2. Ectopic sal expression elongates PE.
(A,C,E,F,I) are x-y views, and the remaining images are x-z views. (A) Ectopic expression of sal in the dpp-Gal4 domain induces elongation of lateral PE height (A′, arrowhead) compared with the control (B, arrowhead). (C) Ubiquitous expression of sal elongates PE height (C′, arrowhead) compared with the control (D, arrowhead). Note that the density of DAPI-stained cell nuclei in the PE is considerably increased compared with control. We compared central (flat) cells in C765-Gal4 experiments (C) and lateral (less flat) cells in dpp-Gal4 experiments (A). (E) Expressing omb in the dpp-Gal4 domain does not elongate lateral PE height (E′, arrowhead). (F) Ubiquitous expression of omb does not elongate PE height (F′, arrowhead). (G) The statistics of Image-J program calculated ratio between PE and DP cell heights within one wing disc. (H) The statistical diagram of normalized PE height ratio between gene manipulations and corresponding control. In both (G,H) means ± SEM indicated by *** are significantly different (pairwise comparison of t-tests, p<0.0001). (I) Clones co-overexpressing sal and CD8-GFP cell membrane marker. Clones in the PE exhibit overgrowth (arrowheads) and elongation of PE height (I′, arrowhead). (J) Control clones expressing CD8-GFP. Arrowhead indicates a PE clone. (K) Expression of lin in the PE leads to the activation of sal transcription in the PE (arrowhead) as revealed by the sal-lacZ reporter. (L) Co-expression of salm-RNAi with lin largely rescues PE height. (M) Suppression of omb expression in the posterior DP has a non-autonomous effect on PE height. Arrow indicates PE elongation. (N) Expression of dad in DP induces non-autonomous PE elongation (arrowhead). (O) Expression of brk in DP induces non-autonomous PE elongation (arrowhead).
Absence of sal in the PE is necessary for PE morphogenesis
As omb and sal are expressed only in the DP, not the PE, we therefore asked whether this asymmetric transcription of omb and sal is essential for correct PE formation. To test this possibility, we mis-expressed sal in the PE using the Gal4-UAS system. dpp-Gal4 line is expressed in narrow stripes in the lateral PE (Fig. 2B, arrowhead) and in the middle DP. When sal was ectopically expressed in the dpp-Gal4 domain, the height of lateral PE was notably elongated to a height similar to that of intermediate cells (Fig. 2A′, arrowhead) compared with control (Fig. 2B, arrowhead). Then, we expressed sal driven by C765-Gal4 which is ubiquitously expressed in both DP and PE. A similar elongation phenotype was observed in the PE. The cell height of the central PE was elongated to a height similar to that of cuboidal cells (Fig. 2C′, arrowhead). The extent of elongation as a result of dpp-Gal4 was stronger than that of C765-Gal4. This difference might be due to the differences in Gal4 activity because dpp-Gal4 is stronger than C765-Gal4. The quantification of cell height (Fig. S-3) using the ratio between PE and DP cells within one wing disc revealed a significant increase in sal mis-expression discs (Fig. 2G). Consistently, the PE height ratio between sal mis-expression and control also revealed a significant increase (Fig. 2H). To confirm this result, we generated sal over-expression clones in PE (Fig. 2I, arrowheads). From the x-z cross view, the clonal height was apparently elongated to a height similar to that for cuboidal cells (Fig. 2I′, arrowhead). Therefore, we conclude that sal is sufficient to elongate PE height.
Unlike the effect of sal mis-expression, the elongation of PE height in case of omb ectopic expression was not apparent (Fig. 2E′,F′, arrowheads), however, the differences in the PE/DP ratio and normalized PE height for C765>omb wing discs are statistically significant (Fig. 2G,H). Strong overexpression of omb induces severe extrusion and basal delamination, and cell motility can thicken the wing disc28. Although we used a relatively weaker UAS-omb line, the side effect from cell movement may remain, thus leading to the statistic difference in the PE measurement. A previous report demonstrates that if Dpp signalling is suppressed in the PE by Ubx-Gal4 driven dad, a portion of the PE cells are elongated to a cuboidal shape8. Therefore, suppressing Dpp signalling in the PE and expressing sal in the PE exhibit similar effects. When carefully assessing the Ubx-Gal4 expression domain, a portion of the expressing cells were, surprisingly, located in the DP (see Fig. S-4). Thus, a non-autonomous effect from a loss of Dpp signalling in the DP in the Ubx>dad wing disc is reasonable. Because when dad-expressing clones were generated within PE, the height of PE did not increase (Fig. S-5). When Omb-RNAi was driven by hh-Gal4 which is expressed in PE and the posterior compartment of DP, the posterior DP height was reduced. Interestingly, the height of opposite PE was increased (Fig. 2M,M′, arrowheads). Thus, it is possible that there is a connection between DP and PE during cell morphogenesis. To directly confirm the non-autonomous effect on PE elongation, the DP height was shortened by expression of either dad or brk within the DP specific nub-Gal4 domain. Consistently, the PE height was apparently increased (Fig. 2N–O′, arrowheads).
sal mediates the role of lin in the elongation of PE height
Previous studies have revealed that mis-expressing lin induces ectopic sal expression in the PE18. Thus, sal may mediate lin’s role in PE elongation. First, we repeated the experiment of lin mis-expression in the PE and consistently observed the elongation phenotype in the PE (Fig. 2K, arrowhead). Then, we revealed the transcription state of sal using a sal-lacZ reporter. sal was apparently transcribed in the PE (Fig. 2K, arrowhead). The sal gene complex is composed of two functionally redundant genes: spalt major (salm) and spalt-related (salr)29. We subsequently performed a rescue experiment by co-expressing lin and salm-RNAi. The morphology of the wing imaginal discs was rescued to an approximate normal state, and PE height was no longer elongated (Fig. 2L, arrowhead). We also measured the cell heights of the corresponding genotypes. sal down-regulation largely rescued the abnormal cell height induced by lin mis-expression (Fig. S-6). These results indicate that sal mediates the role of lin in promoting PE elongation. However, Lin elongated PE to a greater extent than Sal did, according to the statistic measurements. Other mediators may be involved downstream of Lin. Since that Ubx-Gal4 line is also expressed in part of the DP cells (Fig. S-4), potential non-autonomous effects between DP and PE can not be ruled out.
sal affects DP height
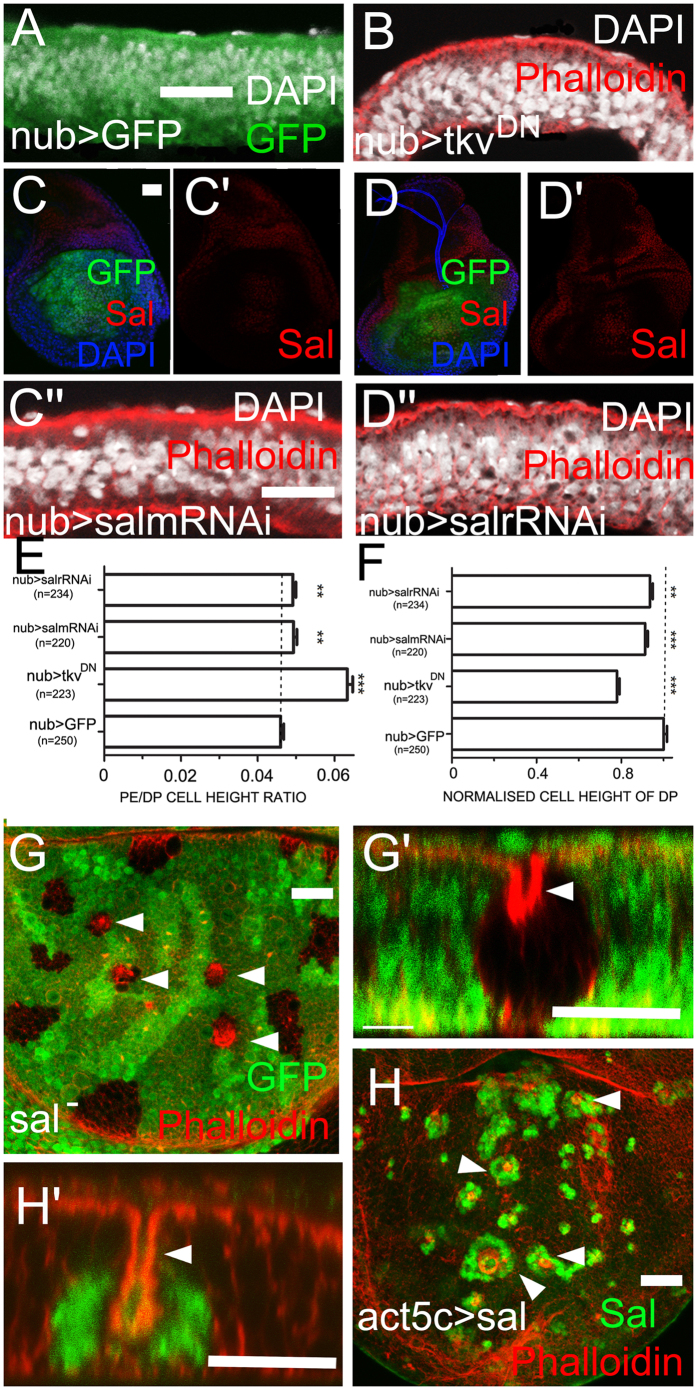
Given that the mis-expression of sal in the PE elongates cell height, we assessed whether down-regulating Dpp-Sal signalling in DP is sufficient to shorten the DP. nub-Gal4 is only expressed in the wing pouch region in the DP7,18,30. When Dpp signalling was mildly inhibited by expressing a dominant negative form of the Dpp receptor, tkvDN, in the nub-Gal4 domain, DP cell height was reduced (Fig. 3B,E,F). Unlike the strong inhibition of Dpp signalling by expressing dad (Fig. 2N), the non-autonomous effect on PE height was not apparent in nub>tkvDN. The Dpp signalling activities in discs of nub>tkvDNand nub>dad were revealed by anti-Sal staining (Fig. S-7). The DP height was slightly reduced when sal was down-regulated either by salm-RNAi or salr-RNAi; however, the extent of this reduction was weaker than that of tkvDN (Fig. 3C–F). Then, we generated sal mutant clones marked by the loss of GFP in the DP. The intensity of F-actin labelled by Phalloidin was much stronger in the clone regions (Fig. 3G, arrowheads). The x-z cross-section showed that the apical side of sal mutant clones in the DP was retracted toward the basal side (Fig. 3G′, arrowhead). These data suggested that Dpp-Sal signalling is required to maintain DP elongation. Interestingly, a similar retraction phenotype was also observed in sal-overexpressing clones (Fig. 3H,H′). Therefore, both sal loss- and gain-of-function clones induce an apical retraction phenotype in the DP31. This phenotype is observed in both omb loss- and gain-of-function clones in the DP14. Omb exhibits a graded distribution in the DP along the A/P axis and specifies unknown apically distributed adhesion molecules. A continuous Omb level is essential for maintaining the epithelial integrity of the wing disc14. Therefore, sharp discontinuity in either Omb or Sal levels in the DP induces apical retraction of cells. To confirm this conclusion, we generated a sharp discontinuity of Sal in the DP using dpp-Gal4 driven UAS-sal. Sal continuity was disrupted at the A/P boundary and where deep apical folds were formed (Fig. S-8). The expression domain of sal in the DP is narrower than that of omb and vg. Beyond the sal domain, omb and vg can ensure the correct cell morphogenesis in the DP. Clones lacking Vg function are also extruded from the DP layer32.
Figure 3. sal affects DP height.
(C,C′, D,D′, G,H) are x-y views. Other images are x-z views. (A) GFP expressing control. (B) Expression of UAS-tkvDNreduces DP height. (C–C″) Expression of salm-RNAi (nub>salm-RNAi) reduces Sal levels in the DP (C and C′) and slightly reduces DP height (C″). (D) Expression of salr-RNAi (nub>salr-RNAi) reduced Sal levels in the DP (D and D′) and slightly reduces DP height (D″). The changes in PE height are not apparent but are statistically significant based on the PE/DP height ratio (E,F). (G) sal mutant clones (absence of GFP, arrowheads) in the medial DP undergo apical retraction (G′, arrowhead). (H) sal-overexpressing clones (absence of GFP, arrowheads) in the medial DP undergo apical retraction (H′, arrowheads).
Mis-expressing sal converts the PE into a cuboidal shape
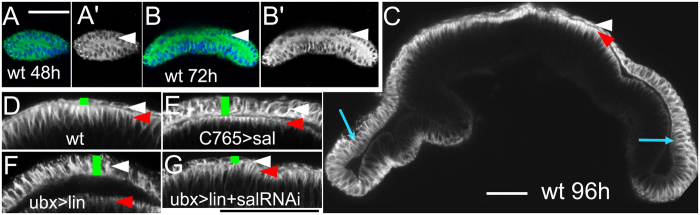
Microtubule cytoskeleton is polarized during cell morphogenesis in the wing imaginal disc33. To reveal microtubule-based cytoskeleton changes induced by either lin or sal mis-expression and the microtubule dynamics during normal development, we monitored the microtubule level via antibody staining. In the 2nd instar, all cells were cuboidal shape, and microtubules were uniformly distributed (Fig. 4A, arrowhead). During the early 3rd instar, the cell shape begins to differentiate. PE cells were largely shortened (Fig. 4B,C, white arrowheads), whereas DP cells were remarkably elongated8,9 (Fig. C, red arrowhead). Correlating with DP elongation, the microtubule network was asymmetrically enriched to the apical side of the DP (Fig. 4C,D, red arrowheads). When lin was mis-expressed in the PE by Ubx-Gal4, sal was activated in the PE (Fig. 2K). Both direct and indirect sal expression (Fig. 4E,F) induced PE height elongation (comparison between green bars indicated PE height in Fig. 4D–F) with an even microtubule distribution (Fig. 4E,F white arrowheads). The microtubule levels of PE were increased compared with the wild type PE (comparison between white arrowheads indicated positions in Fig. 4D–F). The microtubule distribution in Sal-elongated PE was similar to that in very lateral PE and intermediate cells (Fig. 4C, blue arrows) in the L3 stage or undifferentiated cells in earlier larval stages (Fig. 4A, arrowhead). In the rescue experiment, lin and salm-RNAi were co-expressed. PE height was restored (Fig. 4G, green bar), and the microtubule levels in the PE (Fig. 4G, white arrowhead) were similar to that in wild type PE (Fig. 4D, white arrowhead). Therefore, based on the cell height and microtubule distribution, Sal mis-expression converts the PE into a cuboidal cell shape.
Figure 4. Microtubule dynamics during cell height change.
All the images are x-z views. Stains are anti-α-tubulin and DAPI. White arrowheads indicate the PE layers. Red arrowheads indicate the apical side of DP cells. Blue arrows indicate the intermediate cells. Green bars in (D–G) indicate the cell height of PE. (A–C) Time course of microtubule dynamics in wild type control wing discs. PE height starts to flatten at the early L3 stage (B, arrowhead). Microtubules are polarized to the apical side of the DP (C,D, red arrowheads) compared with the even distribution in PE (C,D, white arrowheads) and intermediate cells (C, blue arrows). (E) Microtubules are enhanced in the PE (white arrow) when sal is expressed. (F) lin expression induces similar microtubule changes upon sal expression. Note that the cell height and microtubule levels of the PE (E,F, white arrows) are similar to that of intermediate cells (C, blue arrows). (G) Co-expression of sal-RNAi and lin rescues PE height and microtubule levels similar to that noted in wild type.
During tissue morphogenesis, cell-shape changes always accompany microtubule cytoskeleton rearrangement33,34,35,36,37. Dpp signalling activity has been proposed to play a basic function in microtubule organization. Dpp signalling is graded in the DP along the A/P axis, with higher levels in the medial DP region, which is enriched in apical microtubules12,13. Thus, a correlation is noted between Dpp signalling activity and microtubule levels in the DP. Clones with loss-of-function of Dpp receptors in the DP appear extruded (cell height is severely shortened) and exhibit reduced apical microtubule levels12,13. Consistently, clones with both loss- and gain-of-function sal (Fig. 3 and Fig. S-9) and omb in the DP consistently exhibit severe apical retraction with shortened cell height and loss of apical microtubule enrichment14. Therefore, our data support the hypothesis that Dpp-Omb/Sal signalling activity plays a more general function in microtubule-based cell morphogenesis. Other transcription factors that induce reductions in DP cell height also correlate with the loss of apical microtubule enrichment. The Tbx6 subfamily gene cluster Dorsocross (Doc) initiates wing hinge/blade fold formation. In the Doc expression domain in the DP, cells are shortened from the apical side with severe loss of apical microtubules38.
Methods
Drosophila stocks
The mutant allele was deficiency Df(2 L)32FP-5, which removes both sal and salr39. The following transgenes were used: dpp-Gal440, nub-Gal430, C765-Gal441, Ubx-Gal442, UAS-GFP (Bloomington Stock Center), UAS-CD8-GFP43, UAS-salm22, UAS-omb23 (weaker line omb595), UAS-lines44, UAS-tkvDN45, UAS-tkvQD41, UAS-dad46, UAS-brk27, UAS-wg (Bloomington Stock Center), UAS-EGFRCA47, UAS-salm-RNAi (VDRC 3029), UAS-salr-RNAi (VDRC 28386), UAS-brk-RNAi (VDRC 2919), UAS-bowl-RNAi (VDRC 18428), and flip-in AYGal448. Enhancer trap lines used were sal-lacZ49, omb-lacZ23 and brkX47-lacZ26.
Transgene expression and clone generation
Larvae were raised at 25 °C. For the efficient expression of RNAi and UAS transgenes driven by the weak C765-Gal4, larvae were raised at 29 °C.
Marked clones of mutant cells were generated by Flp-mediated mitotic recombination50 and by subjecting 1st instar larvae to a 35.5 °C heat-shock for 30 min. Transgenes were expressed using the Gal4–UAS system51.
Larval genotypes for clone generation:
y w hs-flp; ubi-GFP FRT40/Df(2 L)32FP-5 FRT40
y w hs-flp; act5c>CD2>GAL4/UAS-GFP; UAS-tkvQD/+
y w hs-flp; act5c>CD2>GAL4/UAS-CD8-GFP; UAS-salm/+
y w hs-flp; act5c>CD2>GAL4/UAS-CD8-GFP; UAS-dad/+
Immunohistochemistry
Dissected wing imaginal discs were fixed and stained with antibodies according to standard procedures. The following primary antibodies were used: rabbit anti-pMad, 1:200 (Cell Signalling); mouse anti-α-Tubulin, 1:2000 (Sigma); rabbit anti-β-galactosidase, 1:2000 (Promega); mouse anti-Ubx, 1:200 (DSHB); rabbit anti-Omb, 1:1000 and rabbit anti-Sal, 1:500 (a gift from Coralia Pérez Fernández); mouse anti-EGFR, 1:200 (abcam); and mouse anti-Wg 1:200 (DSHB). Secondary antibodies (diluted 1:200) included goat anti-mouse DyLight 488 and goat anti-mouse DyLight 549 (Agrisera) and goat anti-rabbit DyLight Cy5 (Jackson ImmunoResearch). Cell nuclei were stained with DAPI (1:500, Sigma). F-actin was visualized with Rhodamine-phalloidin, 1:2000 (Sigma). Images were collected using a Leica TCS SP2 AOBS confocal microscope. Cell height was calculated based on high resolution confocal images using the Image-J program.
Wing disc cryosectioning
After secondary antibody staining, discs were re-fixed for 30 minutes in 4% paraformaldehyde, washed, and stored in 30% sucrose solution at 4 °C overnight. Discs were oriented in Tissue-Tek (Sakura Finetek), frozen and cut into 20-μm sections on a cryostat (YD-1900, YIDI, China).
Additional Information
How to cite this article: Tang, W. et al. Asymmetric distribution of Spalt in Drosophila wing squamous and columnar epithelia ensures correct cell morphogenesis. Sci. Rep. 6, 30236; doi: 10.1038/srep30236 (2016).
Supplementary Material
Acknowledgments
We thank Tsinghua University, the Bloomington Stock Center and VDRC Stock Center for fly stocks and Adi Salzberg and Coralia Pérez Fernández for the Sal antibody. This research was supported by the National Natural Science Foundation of China (NSFC31372255), the 973 program (2013CB127603) and the Chinese Universities Scientific Fund (2016QC079).
Footnotes
Author Contributions J.S. developed the concept and designed the experiments. W.T. performed the experiments. D.W. and J.S. analysed the data and wrote the manuscript.
References
- Leptin M. Drosophila gastrulation: from pattern formation to morphogenesis. Annu. Rev. Cell Dev. Biol. 11, 189–212 (1995). [DOI] [PubMed] [Google Scholar]
- Sawyer J. M. et al. Apical constriction: a cell shape change that can drive morphogenesis. Dev. Biol. 341, 5–19 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Detrait E. R. et al. Human neural tube defects: developmental biology, epidemiology, and genetics. Neurotoxicol. Teratol. 27, 515–524 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowery L. A. & Sive H. Strategies of vertebrate neurulation and a re-evaluation of teleost neural tube formation. Mech. Dev. 121, 1189–1197 (2004). [DOI] [PubMed] [Google Scholar]
- Basu R. & Chang F. Shaping the actin cytoskeleton using microtubule tips. Curr. Opin. Cell Biol. 19, 88–94 (2007). [DOI] [PubMed] [Google Scholar]
- Lecuit T. et al. Two distinct mechanisms for long-range patterning by Decapentaplegic in the Drosophila wing. Nature 381, 387–393 (1996). [DOI] [PubMed] [Google Scholar]
- del Álamo Rodríguez D., Terriente J., Galindo M. I., Couso J. P. & Díaz-Benjumea F. J. Different mechanisms initiate and maintain wingless expression in the Drosophila wing hinge. Development 129, 3995–4004 (2002). [DOI] [PubMed] [Google Scholar]
- McClure K. D. & Schubiger G. Developmental analysis and squamous morphogenesis of the peripodial epithelium in Drosophila imaginal discs. Development 132, 5033–5042 (2005). [DOI] [PubMed] [Google Scholar]
- Widmann T. J. & Dahmann C. Dpp signalling promotes the cuboidal-to-columnar shape transition of Drosophila wing disc epithelia by regulating Rho1. J. Cell Sci. 122, 1362–1373 (2009). [DOI] [PubMed] [Google Scholar]
- Cohen S. M. In The development of Drosophila melanogaster Vol. 2, 747-841 (1993).
- Cho K. O., Chern J., Izaddoost S. & Choi K.-W. Novel signalling from the peripodial membrane is essential for eye disc patterning in Drosophila. Cell 103, 331–342 (2000). [DOI] [PubMed] [Google Scholar]
- Shen J. & Dahmann C. Extrusion of cells with inappropriate Dpp signalling from Drosophila wing disc epithelia. Science 307, 1789–1790 (2005). [DOI] [PubMed] [Google Scholar]
- Gibson M. C. & Perrimon N. Extrusion and death of DPP/BMP-compromised epithelial cells in the developing Drosophila wing. Science 307, 1785–1789 (2005). [DOI] [PubMed] [Google Scholar]
- Shen J., Dahmann C. & Pflugfelder G. O. Spatial discontinuity of optomotor-blind expression in the Drosophila wing imaginal disc disrupts epithelial architecture and promotes cell sorting. BMC Dev. Biol. 10, 23 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaiswal M., Agrawal N. & Sinha P. Fat and Wingless signalling oppositely regulate epithelial cell-cell adhesion and distal wing development in Drosophila. Development 133, 925–935 (2006). [DOI] [PubMed] [Google Scholar]
- Widmann T. J. & Dahmann C. Wingless signalling and the control of cell shape in Drosophila wing imaginal discs. Dev. Biol. 334, 161–173 (2009). [DOI] [PubMed] [Google Scholar]
- Gibson M. C., Lehman D. A. & Schubiger G. Lumenal transmission of decapentaplegic in Drosophila imaginal discs. Dev. Cell 3, 451–460 (2002). [DOI] [PubMed] [Google Scholar]
- Nusinow D., Greenberg L. & Hatini V. Reciprocal roles for bowl and lines in specifying the peripodial epithelium and the disc proper of the Drosophila wing primordium. Development 135, 3031–3041 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiersdorff V., Lecuit T., Cohen S. M. & Mlodzik M. Mad acts downstream of Dpp receptors, revealing a differential requirement for dpp signalling in initiation and propagation of morphogenesis in the Drosophila eye. Development 122, 2153–2162 (1996). [DOI] [PubMed] [Google Scholar]
- Guo Z., Driver I. & Ohlstein B. Injury-induced BMP signalling negatively regulates Drosophila midgut homeostasis. J. Cell Biol. 201, 945–961 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanimoto H., Itoh S., ten Dijke P. & Tabata T. Hedgehog creates a gradient of DPP activity in Drosophila wing imaginal discs. Mol. Cell 5, 59–71 (2000). [DOI] [PubMed] [Google Scholar]
- de Celis J. F., Barrio R. & Kafatos F. C. A gene complex acting downstream of dpp in Drosophila wing morphogenesis. Nature 381, 421–424 (1996). [DOI] [PubMed] [Google Scholar]
- Grimm S. & Pflugfelder G. O. Control of the gene optomotor-blind in Drosophila wing development by decapentaplegic and wingless. Science 271, 1601–1604 (1996). [DOI] [PubMed] [Google Scholar]
- Minami M., Kinoshita N., Kamoshida Y., Tanimoto H. & Tabata T. brinker is a target of Dpp in Drosophila that negatively regulates Dpp-dependent genes. Nature 398, 242–246 (1999). [DOI] [PubMed] [Google Scholar]
- Müller B., Hartmann B., Pyrowolakis G., Affolter M. & Basler K. Conversion of an extracellular Dpp/BMP morphogen gradient into an inverse transcriptional gradient. Cell 113, 221–233 (2003). [DOI] [PubMed] [Google Scholar]
- Campbell G. & Tomlinson A. Transducing the Dpp morphogen gradient in the wing of Drosophila: regulation of Dpp targets by brinker. Cell 96, 553–562 (1999). [DOI] [PubMed] [Google Scholar]
- Jaźwińska A., Kirov N., Wieschaus E., Roth S. & Rushlow C. The Drosophila gene brinker reveals a novel mechanism of Dpp target gene regulation. Cell 96, 563–573 (1999). [DOI] [PubMed] [Google Scholar]
- Shen J. et al. The orthologous Tbx transcription factors Omb and TBX2 induce epithelial cell migration and extrusion in vivo without involvement of matrix metalloproteinases. Oncotarget 5, 11998 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reuter D. et al. Regulation, function and potential origin of the Drosophila gene spalt adjacent, which encodes a secreted protein expressed in the early embryo. Chromosoma 104, 445–454 (1996). [DOI] [PubMed] [Google Scholar]
- Calleja M., Moreno E., Pelaz S. & Morata G. Visualization of gene expression in living adult Drosophila. Science 274, 252–255 (1996). [DOI] [PubMed] [Google Scholar]
- Milán M., Pérez L. & Cohen S. M. Short-range cell interactions and cell survival in the Drosophila wing. Dev. Cell 2, 797–805 (2002). [DOI] [PubMed] [Google Scholar]
- Baena-Lopez L. & García-Bellido A. Control of growth and positional information by the graded vestigial expression pattern in the wing of Drosophilamelanogaster. Proc. Natl. Acad. Sci. USA 103, 13734–13739 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eaton S., Wepf R. & Simons K. Roles for Rac1 and Cdc42 in planar polarization and hair outgrowth in the wing of Drosophila. J. Cell Biol. 135, 1277–1289 (1996). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brodu V., Baffet A. D., Le Droguen P.-M., Casanova J. & Guichet A. A developmentally regulated two-step process generates a noncentrosomal microtubule network in Drosophila tracheal cells. Dev. Cell 18, 790–801 (2010). [DOI] [PubMed] [Google Scholar]
- Jankovics F. & Brunner D. Transiently reorganized microtubules are essential for zippering during dorsal closure in Drosophila melanogaster. Dev. Cell 11, 375–385 (2006). [DOI] [PubMed] [Google Scholar]
- Pope K. L. & Harris T. J. C. Control of cell flattening and junctional remodeling during squamous epithelial morphogenesis in Drosophila. Development 135, 2227–2238 (2008). [DOI] [PubMed] [Google Scholar]
- Booth A. J. R., Blanchard G. B., Adams R. J. & Röper K. A dynamic microtubule cytoskeleton directs medial actomyosin function during tube formation. Dev. Cell 29, 562–576 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sui L., Pflugfelder G. O. & Shen J. The Dorsocross T-box transcription factors promote tissue morphogenesis in the Drosophila wing imaginal disc. Development 139, 2773–2782 (2012). [DOI] [PubMed] [Google Scholar]
- de Celis J. F., Barrio R. & Kafatos F. C. Regulation of the spalt/spalt-related gene complex and its function during sensory organ development in the Drosophila thorax. Development 126, 2653–2662 (1999). [DOI] [PubMed] [Google Scholar]
- Shen W. & Mardon G. Ectopic eye development in Drosophila induced by directed dachshund expression. Development 124, 45–52 (1997). [DOI] [PubMed] [Google Scholar]
- Nellen D., Burke R., Struhl G. & Basler K. Direct and long-range action of a DPP morphogen gradient. Cell 85, 357–368 (1996). [DOI] [PubMed] [Google Scholar]
- Pallavi S. K. & Shashidhara L. S. Egfr/Ras pathway mediates interactions between peripodial and disc proper cells in Drosophila wing discs. Development 130, 4931–4941 (2003). [DOI] [PubMed] [Google Scholar]
- Lee T. & Luo L. Mosaic analysis with a repressible cell marker for studies of gene function in neuronal morphogenesis. Neuron 22, 451–461 (1999). [DOI] [PubMed] [Google Scholar]
- Hatini V., Green R. B., Lengyel J. A., Bray S. J. & DiNardo S. The Drumstick/Lines/Bowl regulatory pathway links antagonistic Hedgehog and Wingless signalling inputs to epidermal cell differentiation. Genes Dev. 19, 709–718 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haerry T. E., Khalsa O., O'Connor M. B. & Wharton K. A. Synergistic signalling by two BMP ligands through the SAX and TKV receptors controls wing growth and patterning in Drosophila. Development 125, 3977–3987 (1998). [DOI] [PubMed] [Google Scholar]
- Tsuneizumi K. et al. Daughters against dpp modulates dpp organizing activity in Drosophila wing development. Nature 389, 627–631 (1997). [DOI] [PubMed] [Google Scholar]
- Queenan A. M., Ghabrial A. & Schupbach T. Ectopic activation of torpedo/Egfr, a Drosophila receptor tyrosine kinase, dorsalizes both the eggshell and the embryo. Development 124, 3871–3880 (1997). [DOI] [PubMed] [Google Scholar]
- Pignoni F. & Zipursky S. L. Induction of Drosophila eye development by decapentaplegic. Development 124, 271–278 (1997). [DOI] [PubMed] [Google Scholar]
- Kühnlein R. P. et al. spalt encodes an evolutionarily conserved zinc finger protein of novel structure which provides homeotic gene function in the headand tail region of the Drosophila embryo. EMBO J. 13, 168–179 (1994). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu T. & Rubin G. M. Analysis of genetic mosaics in developing and adult Drosophila tissues. Development 117, 1223–1237 (1993). [DOI] [PubMed] [Google Scholar]
- Brand A. H. & Perrimon N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.