Abstract
Background and Purpose
We assessed the prevalence and potential association of hypertension with multiple sclerosis (MS)-related disability progression.
Methods
This was a retrospective study of 2,813 patients who were followed for 20 years. We modeled the associations of several risk factors with the pattern of disability progression. The primary end point was the rate of disability progression.
Results
In total, 2,396 patients were available for analysis, of which 1,074 (44.8%) scored 4 (EDSS4) on the Expanded Disability Status Scale (EDSS), 717 (29.9%) scored 6 (EDSS6), and 261 (10.9%) scored 8 (EDSS8). The mean times to reach scores of 4, 6, and 8 were 123.5, 163.1, and 218.9 months, respectively. Hypertension was present in 207 (8.6%) patients during follow-up. Hypertension was associated with a higher probability of reaching each EDSS score compared to non-hypertensive patients: 62% vs. 43% for EDSS4 (p<0.01), 51% vs. 28% for EDSS6 (p<0.01), and 17% vs. 10% for EDSS8 (p<0.01). Nevertheless, hypertensive MS patients experienced longer intervals to reach each EDSS score: longer by 51.6, 38.9, and 62.7 months to EDSS4, EDSS6, and EDSS8, respectively (p<0.01) when compared to non-hypertensive MS patients reaching the same EDSS scores.
Conclusions
Disability progression is more prevalent amongst hypertensive MS patients. However, they experience longer time intervals between the stages of disability progression.
Keywords: multiple sclerosis, hypertension, risk factor, epidemiology, disability, atherosclerosis
INTRODUCTION
Multiple sclerosis (MS) is a chronic, frequently progressive, and disabling disease. Both physicians and patients need to be aware of the factors associated with disease occurrence and progression, since MS patients are frequently afflicted with this disease while they are still in the prime of their lives.1,2
Previous retrospective studies have assessed a myriad of risk factors for MS-associated disability progression. The available literature reflects difficulties in establishing concrete risk factors for both disease occurrence and disability progression: for instance, some studies found advanced age at disease onset to be associated with a shorter time to disease progression3,4 while other studies did not find such an association.5,6 Obesity, mainly during early life, was shown to increase the risk of MS development7,8 but in other studies this association was found to be dependent upon certain demographic parameters.9 Smoking was demonstrated to increase the risk of disease occurrence in some studies and to a lesser extent enhance disease progression.10,11,12,13
Some of the aforementioned conditions (e.g., smoking and obesity) are established risk factors for atherosclerosis, cardiovascular, and cerebrovascular disease. Accordingly, it is plausible that they could also be risk factors for the increased occurrence and progression of neurological disability among MS patients.14 However, this assumption could be weakened by most MS patients having a relatively low risk of cardiovascular disease due to them being young females. Few studies have addressed this important issue.
As for the potential association of arterial hypertension (HTN) with MS prevalence and associated disability progression, a recent review by Tettey et al.15 questioned whether vascular comorbidities influence MS clinical disability. Those authors suggested that having type-2 diabetes, HTN, dyslipidemia, or peripheral vascular disease at any point during the disease course could be associated with a greater progression in disability. Conflicting data exist regarding the prevalence of HTN in MS. While some authors argue that the prevalence is similar to that in the general population,16 others claim that the prevalence is lower among MS patients.17
In view of the conflicting data in the available literature, we decided to assess the prevalence and evaluate the potential association of several risk factors related to atherosclerosis in a large cohort of MS patients.
METHODS
Study cohort
The current study was approved by the Chaim Sheba Institutional Review Board committee. The study cohort included MS patients followed in the Multiple Sclerosis Center, Sheba Medical Center, Tel Hashomer, Israel for more than 20 years. The diagnosis for each patient was made accordingly by expert neurologists working in the clinic. Background health data were based on patient interviews performed during their MS clinic visits: family history and smoking status were based on self-reports, while diagnoses of HTN and cardiovascular diseases were also confirmed according to patient records and chronic medications.
Expanded Disability Status Scale
The Expanded Disability Status Scale (EDSS) was developed by John F. Kurtzke and is used to quantify disability in MS.18 The EDSS quantifies disability in eight functional systems, with its score ranging between 0 (normal neurological examination findings) to 10 (death due to MS). For this study the progression of disability over time was used for patients reaching the following EDSS scores: 4 (EDSS4), which indicates a disability that does not prevent normal activities; 6 (EDSS6), indicating assistance needed for walking; and 8 (EDSS8), indicating being bedridden but with preserved arm function.19
Statistical analysis
The primary analysis tested for associations of selected risk factors for atherosclerosis with MS-associated disability progression. We used a multiway analysis-of-variance model to assess the risk of disability progression to EDSS4, EDSS6, and EDSS8 according to the following variables: smoking status, family history of arterial HTN, family history of ischemic heart disease, family history of diabetes, and the presence of chronic arterial HTN in the patient. The family history of some diseases was considered rather than the personal history due to the youngness of most MS patients when they present with this disease. All these risk factors were assimilated into a multiway analysis model after being corrected according to the age at disease onset. Analysis was performed using SPSS software (version 20.0). A sub analysis was applied to the group of patients who had MS and reported HTN at any stage of follow-up. We assessed whether there was an association between the length of time since HTN was diagnosed and the length of intervals before reaching each of the different stages of disability quantified using the EDSS.
RESULTS
Study cohort
The study cohort initially included 2,813 patients, of which 404 were excluded due to an indefinite MS diagnosis and 13 were excluded due to the disease onset occurring before the age of 18 years. This left 2,396 MS patients eligible for analysis: 1,771 (73.9%) of them had relapsing remitting MS, 118 (4.9%) had primary progressive disease, 376 (15.7%) had secondary progressive disease, 113 (4.7%) were diagnosed as having clinically isolated syndrome, and 2 (<1%) had optic neuritis. The subjects comprised 774 (32.3%) males and 1,622 (67.7%) females; they were aged 46.5±13.5 years (mean±SD), with an age of 31.6±10.9 years at disease onset.
Factors associated with disease progression
EDSS4, EDSS6, and EDSS8 were reached by 1,074 (44.8%), 717 (29.9%), and 261 (10.9%) patients in our cohort after average disease durations of 123.5, 163.1, and 218.9 months, respectively. The frequencies of atherosclerosis risk factors among the complete eligible cohort are presented in Table 1. Arterial HTN was present in 207 (8.6%) patients, a positive family history of HTN was present in 411 (17.1%) patients, a positive family history of diabetes mellitus was present in 472 (19.7%) patients, a positive family history of cardiac disease was present in 328 (13.7%) patients, and a positive personal history of ischemic heart disease was present in 2 (<1%) patients. Information regarding smoking status was available for 2,376 (99.1%) patients of the eligible cohort: 525 (21.9%), 1,296 (54.1%), and 555 (23.1%) were classified as "never smoked," "past smokers," and "current smokers," respectively.
Table 1. Baseline patient characteristics.
| Co-morbidity | EDSS4 (n=1,074) | EDSS6 (n=717) | EDSS8 (n=261) | |||
|---|---|---|---|---|---|---|
| Frequency | Mean duration (months) | Frequency | Mean duration (months) | Mean duration (months) | Mean duration (months) | |
| Arterial HTN | ||||||
| Yes (n=207) | 129 (12%) | 140.1 | 106 (14.7%) | 162.4 | 35 (13.4%) | 237.1 |
| No (n=2,189) | 945 (88%) | 121.2 | 611 (85.3%) | 163.2 | 226 (86.6%) | 216.1 |
| Family history of HTN | ||||||
| Yes (n=411) | 189 (17.6%) | 125.6 | 108 (15.1%) | 148.3 | 32 (12.2%) | 186.3 |
| No (n=1,985) | 885 (82.4%) | 123.1 | 609 (84.9%) | 165.7 | 229 (87.8%) | 235.5 |
| Family history of diabetes | ||||||
| Yes (n=472) | 215 (20.0%) | 123.0 | 139 (19.4%) | 163.4 | 44 (16.9%) | 236.4 |
| No (n=1,924) | 859 (80.0%) | 123.6 | 578 (80.6%) | 163.0 | 217 (83.1%) | 215.4 |
| Family history of cardiac disease | ||||||
| Yes (n=328) | 172 (84%) | 140.3 | 110 (15.3%) | 181.7 | 32 (12.2%) | 272.9 |
| No (n=2,068) | 902 (16%) | 120.3 | 607 (84.7%) | 159.7 | 229 (87.8%) | 211.4 |
| Smoking status | ||||||
| Never (n=525) | 211 (19.8%) | 128.2 | 137 (19.3%) | 170.2 | 50 (19.5%) | 238.0 |
| Past (n=1,296) | 590 (55.2%) | 125.2 | 423 (59.5%) | 163.2 | 162 (63.3%) | 225.0 |
| Current (n=555) | 267 (25.0%) | 116.9 | 151 (21.2%) | 157.9 | 44 (17.2%) | 182.4 |
| Personal history of ischemic heart disease | ||||||
| Yes (n=2) | 1 | 64 | 1 | 75 | 0 | 0 |
| No (n=2,394) | 1,073 | 123.6 | 716 | 163.2 | 261 | 218.9 |
EDSS: Expanded Disability Status Scale, HTN: hypertension.
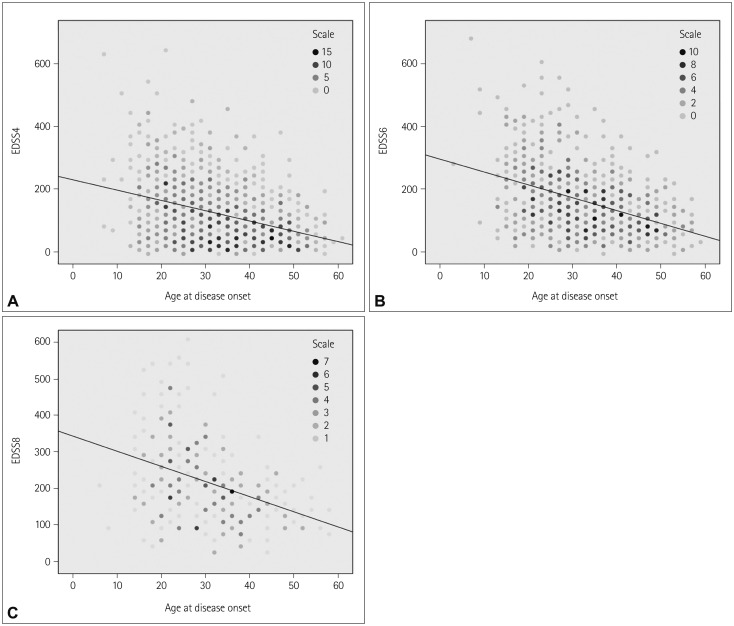
Regression analysis showed that being older at disease onset was associated with more rapid disability progression, with each additional year of age at disease onset shortening the times to reach EDSS4, EDSS6, and EDSS8 by 3.36, 4.23, and 4.08 months, respectively (p<0.01) (Fig. 1).
Fig. 1. Regression analysis of disease progression to EDSS4 (A), EDSS6 (B), and EDSS8 (C) according to age at disease onset. EDSS: Expanded Disability Status Scale.
Among patients with a personal history of arterial HTN that was reported or diagnosed at any time point during follow-up, the risk of progression to each EDSS score was higher relative to non-hypertensive patients (Table 2): 62.3% vs. 43.2% to reach EDSS4 (p<0.01), 51.2% vs. 27.9% to reach EDSS6 (p<0.01), and 16.9% vs. 10.3% to reach EDSS8 (p<0.01).
Table 2. Progression of patients to each EDSS score according to their personal history of arterial HTN.
| Arterial HTN | EDSS4 | EDSS6 | EDSS8 |
|---|---|---|---|
| Yes (n=207) | 62.3% | 51.2% | 16.9% |
| No (n=2,189) | 43.2% | 27.9% | 10.3% |
| Probability* | p<0.01 | p<0.01 | p<0.01 |
*Using the chi-square test.
EDSS: Expanded Disability Status Scale, HTN: hypertension.
Table 3 presents the associations of different risk factors for atherosclerosis with the rate of progression to the different EDSS scores after correction for the age at disease onset. The presence of comorbid arterial HTN at any time during follow-up prolonged the times to reach EDSS4, EDSS6, and EDSS8 by 51.6, 38.9, and 62.7 months, respectively (all p<0.01). A family history of heart disease prolonged the times to reach EDSS4, EDSS6, and EDSS8 by 29.7, 24.1, and 48.1 months, respectively (p<0.01, p<0.02, and p<0.05). Current smoking was associated with shortened times to reach EDSS4 and EDSS8 (7.6 and 65.1 months earlier, respectively, vs. never smoked; p<0.05 and p<0.01). Smoking status was not found to be associated with a shorter time to reach EDSS6. A family history of diabetes and a family history of arterial HTN were not associated with significant changes in the time to reach each of the EDSS scores assessed in this study.
Table 3. Atherosclerosis risk factors associated with progression of MS after correction for age at disease onset*.
| Risk factor | ΔTime (months) to reach EDSS4 | ΔTime (months) to reach EDSS6 | ΔTime (months) to reach EDSS8 |
|---|---|---|---|
| Arterial HTN | +51.6 (p<0.01) | +38.9 (p<0.01) | +62.7 (p<0.01) |
| Family history of diabetes mellitus | NS | NS | NS |
| Family history of heart disease | +29.7 (p<0.01) | +24.1 (p<0.05) | +48.1 (p>0.05) |
| Family history of HTN | NS | −26.3 (p<0.02) | NS |
| Smoking | −11.4 (p<0.05) | NS | −65.1 (p<0.01) |
*Using a regression model of risk factors.
EDSS: Expanded Disability Status Scale, HTN: hypertension, MS: multiple sclerosis, NS: not significant.
DISCUSSION
This study evaluated the association between several risk factors for atherosclerosis and the progression of MS-associated disability as measured by the EDSS score. In our cohort, being older at disease onset was associated with increased risk of disability progression in MS. This association, which has also been reported previously,20,21,22 could be explained by neurological disability arising from both MS-related neurological damage and also atherosclerosis- and arteriolosclerosis-associated damage.
Since being older was unequivocally shown to be associated with a worse prognosis, given the potential association with other risk factors for MS disability progression, we applied statistical correction for age at disease onset (a potential confounder) to avoid statistical bias. The risk factor for atherosclerosis that was found to be most significantly related to the disease-associated neurological disability progression was comorbid arterial HTN. The prevalence of arterial HTN amongst MS patients in our cohort was relatively low (8.6%), as anticipated for this population of relatively young patients (7.7–9.2%).23 Nevertheless, in the small population that does suffer from this comorbidity, we found it to be associated with an increased risk of reaching higher levels of disability. This finding, which applied to each EDSS score tested, is reasonable if atherosclerosis and arteriolosclerosis contribute to neurodegeneration. However, the association of HTN with the rate of disability progression was an unexpected finding that should be further questioned and investigated. It reflects a slower propagation of neurological deficits in hypertensive patients relative to normotensive MS patients that reached the same EDSS scores. There are several hypothetical explanations for this finding. First, antihypertensive medications may have a neuroprotective role, partially preventing some of the measured neurological damage. Second, hypertensive MS patients are (at least potentially) cared for and treated more often by their primary physicians, potentially referring them earlier to medical attention relative to non-hypertensive MS patients.
The literature lacks sound data on the established risk factors for MS occurrence and related disability progression. A recent meta-analysis assessing environmental factors associated with MS found that there was strong consistent evidence of associations with MS occurrence only for infectious mononucleosis and smoking.24 Marrie et al.25 concluded that despite the relatively high prevalence of vascular comorbidities in the MS population, there are major gaps in understanding the epidemiology of such comorbidities in these patients. In other reviews, Wens et al.26 reached a similar conclusion, while Tettey et al.15 suggested that cardiovascular comorbidities affect the course of MS. One aim of the current study was to better characterize this possible association. Some of our results are similar to those of previous studies, while others could be considered as preliminary findings that necessitate further investigations.
Our study was subject to several limitations. Its retrospective design meant that the causality of the associations could not be determined. Further studies are therefore needed to characterize the casual relationships. Also, prospective studies should test a potential association of certain antihypertensive treatments with a slower rate of disability progression of MS patients. Other limitations relate to the quality of follow-up and the accuracy of data, which are shared by all retrospective studies on which a large volume of research relies on. We did not include other risk factors for atherosclerosis such as diabetes and dyslipidemia in our study due to statistical constraints, but plan to examine them in future studies. In addition, we did not have information about the antihypertensive medications taken by our patients during this long surveillance period, and so we were unable to determine any possible effect of such medications on the progression of MS.
Footnotes
Conflicts of Interest: The authors have no financial conflicts of interest.
References
- 1.Confavreux C, Vukusic S. The clinical course of multiple sclerosis. Handb Clin Neurol. 2014;122:343–369. doi: 10.1016/B978-0-444-52001-2.00014-5. [DOI] [PubMed] [Google Scholar]
- 2.Goodin DS. The epidemiology of multiple sclerosis: insights to disease pathogenesis. Handb Clin Neurol. 2014;122:231–266. doi: 10.1016/B978-0-444-52001-2.00010-8. [DOI] [PubMed] [Google Scholar]
- 3.Koch M, Uyttenboogaart M, van Harten A, De Keyser J. Factors associated with the risk of secondary progression in multiple sclerosis. Mult Scler. 2008;14:799–803. doi: 10.1177/1352458508089361. [DOI] [PubMed] [Google Scholar]
- 4.Newbould RD, Nicholas R, Thomas CL, Quest R, Lee JS, Honeyfield L, et al. Age independently affects myelin integrity as detected by magnetization transfer magnetic resonance imaging in multiple sclerosis. Neuroimage Clin. 2014;4:641–648. doi: 10.1016/j.nicl.2014.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tremlett H, Zhao Y, Rieckmann P, Hutchinson M. New perspectives in the natural history of multiple sclerosis. Neurology. 2010;74:2004–2015. doi: 10.1212/WNL.0b013e3181e3973f. [DOI] [PubMed] [Google Scholar]
- 6.Stellmann JP, Neuhaus A, Lederer C, Daumer M, Heesen C. Validating predictors of disease progression in a large cohort of primary-progressive multiple sclerosis based on a systematic literature review. PLoS One. 2014;9:e92761. doi: 10.1371/journal.pone.0092761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Versini M, Jeandel PY, Rosenthal E, Shoenfeld Y. Obesity in autoimmune diseases: not a passive bystander. Autoimmun Rev. 2014;13:981–1000. doi: 10.1016/j.autrev.2014.07.001. [DOI] [PubMed] [Google Scholar]
- 8.Kavak KS, Teter BE, Hagemeier J, Zakalik K, Weinstock-Guttman B New York State Multiple Sclerosis Consortium. Higher weight in adolescence and young adulthood is associated with an earlier age at multiple sclerosis onset. Mult Scler. 2015;21:858–865. doi: 10.1177/1352458514555787. [DOI] [PubMed] [Google Scholar]
- 9.Wesnes K, Riise T, Casetta I, Drulovic J, Granieri E, Holmøy T, et al. Body size and the risk of multiple sclerosis in Norway and Italy: the EnvIMS study. Mult Scler. 2015;21:388–395. doi: 10.1177/1352458514546785. [DOI] [PubMed] [Google Scholar]
- 10.O'Gorman C, Broadley SA. Smoking and multiple sclerosis: evidence for latitudinal and temporal variation. J Neurol. 2014;261:1677–1683. doi: 10.1007/s00415-014-7397-5. [DOI] [PubMed] [Google Scholar]
- 11.Jawahar R, Oh U, Eaton C, Wright N, Tindle H, Lapane KL. Association between smoking and health outcomes in postmenopausal women living with multiple sclerosis. Mult Scler Int. 2014;2014:686045. doi: 10.1155/2014/686045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Belbasis L, Bellou V, Evangelou E, Ioannidis JP, Tzoulaki I. Environmental risk factors and multiple sclerosis: an umbrella review of systematic reviews and meta-analyses. Lancet Neurol. 2015;14:263–273. doi: 10.1016/S1474-4422(14)70267-4. [DOI] [PubMed] [Google Scholar]
- 13.Gustavsen MW, Page CM, Moen SM, Bjølgerud A, Berg-Hansen P, Nygaard GO, et al. Environmental exposures and the risk of multiple sclerosis investigated in a Norwegian case-control study. BMC Neurol. 2014;14:196. doi: 10.1186/s12883-014-0196-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Marrie RA, Rudick R, Horwitz R, Cutter G, Tyry T, Campagnolo D, et al. Vascular comorbidity is associated with more rapid disability progression in multiple sclerosis. Neurology. 2010;74:1041–1047. doi: 10.1212/WNL.0b013e3181d6b125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tettey P, Simpson S, Jr, Taylor BV, van der Mei IA. Vascular comorbidities in the onset and progression of multiple sclerosis. J Neurol Sci. 2014;347:23–33. doi: 10.1016/j.jns.2014.10.020. [DOI] [PubMed] [Google Scholar]
- 16.Marrie RA, Yu BN, Leung S, Elliott L, Caetano P, Warren S, et al. Rising prevalence of vascular comorbidities in multiple sclerosis: validation of administrative definitions for diabetes, hypertension, and hyperlipidemia. Mult Scler. 2012;18:1310–1319. doi: 10.1177/1352458512437814. [DOI] [PubMed] [Google Scholar]
- 17.Simpson RJ, McLean G, Guthrie B, Mair F, Mercer SW. Physical and mental health comorbidity is common in people with multiple sclerosis: nationally representative cross-sectional population database analysis. BMC Neurol. 2014;14:128. doi: 10.1186/1471-2377-14-128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS) Neurology. 1983;33:1444–1452. doi: 10.1212/wnl.33.11.1444. [DOI] [PubMed] [Google Scholar]
- 19.Kurtzke JF. Historical and clinical perspectives of the expanded disability status scale. Neuroepidemiology. 2008;31:1–9. doi: 10.1159/000136645. [DOI] [PubMed] [Google Scholar]
- 20.Kurtzke JF. Clinical definition for multiple sclerosis treatment trials. Ann Neurol. 1994;36(Suppl):S73–S79. doi: 10.1002/ana.410360717. [DOI] [PubMed] [Google Scholar]
- 21.Pankuweit S, Wädlich A, Meyer E, Portig I, Hufnagel G, Maisch B. Cytokine activation in pericardial fluids in different forms of pericarditis. Herz. 2000;25:748–754. doi: 10.1007/pl00001993. [DOI] [PubMed] [Google Scholar]
- 22.Kis B, Rumberg B, Berlit P. Clinical characteristics of patients with late-onset multiple sclerosis. J Neurol. 2008;255:697–702. doi: 10.1007/s00415-008-0778-x. [DOI] [PubMed] [Google Scholar]
- 23.Joffres M, Falaschetti E, Gillespie C, Robitaille C, Loustalot F, Poulter N, et al. Hypertension prevalence, awareness, treatment and control in national surveys from England, the USA and Canada, and correlation with stroke and ischaemic heart disease mortality: a cross-sectional study. BMJ Open. 2013;3:e003423. doi: 10.1136/bmjopen-2013-003423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Belbasis L, Bellou V, Evangelou E, Ioannidis JP, Tzoulaki I. Environmental risk factors and multiple sclerosis: an umbrella review of systematic reviews and meta-analyses. Lancet Neurol. 2015;14:263–273. doi: 10.1016/S1474-4422(14)70267-4. [DOI] [PubMed] [Google Scholar]
- 25.Marrie RA, Reider N, Cohen J, Stuve O, Trojano M, Cutter G, et al. A systematic review of the incidence and prevalence of cardiac, cerebrovascular, and peripheral vascular disease in multiple sclerosis. Mult Scler. 2015;21:318–331. doi: 10.1177/1352458514564485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wens I, Dalgas U, Stenager E, Eijnde BO. Risk factors related to cardiovascular diseases and the metabolic syndrome in multiple sclerosis - a systematic review. Mult Scler. 2013;19:1556–1564. doi: 10.1177/1352458513504252. [DOI] [PubMed] [Google Scholar]