Abstract
Background
Liraglutide is one of the glucagon-like peptide-1 analogs; there are only a few reports of liraglutide being used for the treatment of insulin allergy. Furthermore, anti-insulin immunoglobulin G antibodies are occasionally detected in patients with diabetes. Hence, we report a case in which switching to liraglutide therapy ameliorated both the symptoms of insulin allergy with hypereosinophilia and the characteristics of insulin antibodies in a patient with type 2 diabetes mellitus.
Case presentation
We present the case of a 70-year-old Japanese man with type 2 diabetes who developed insulin allergy with hypereosinophilia. Anti-insulin antibodies, high glycated hemoglobin levels (approximately 12 %), and high serum insulin levels were detected. Because a change in his insulin treatment was inefficient, treatment with liraglutide to protect residual insulin secretion was started, resulting in improvements in his insulin allergy, serum glycated hemoglobin, insulin, and eosinophil levels. Scatchard plots revealed decreased binding capacity and increased affinity constant for high affinity sites of anti-insulin antibodies.
Conclusions
Liraglutide might be useful for treating insulin allergy and anti-insulin antibodies in patients with type 2 diabetes.
Keywords: Liraglutide, Insulin allergy, Human insulin-specific immunoglobulin E, Hypereosinophilia, Anti-insulin immunoglobulin G antibodies
Background
Insulin allergy is an important adverse effect of insulin treatment in patients with diabetes. Ghazavi and Johnston recently reported that the prevalence of allergic reactions to insulin products is approximately 2 % at present [1]. Although the frequency of insulin allergy is low, the symptoms vary, ranging from local injection site reactions to complete anaphylactic reactions [2, 3]. Given that anaphylactic reactions are occasionally life-threatening, clinicians must act quickly and appropriately when allergy is suspected.
Glucagon-like peptide-1 (GLP-1) analogs have recently been introduced worldwide as a therapeutic option for the treatment of type 2 diabetes. Liraglutide is one such analog that improves blood glucose control by directly affecting both insulin and glucagon secretion in the pancreas [4]. However, there are only a few reports of liraglutide being used for the treatment of insulin allergy, and its effectiveness remains to be completely elucidated in this context [5, 6]. Furthermore, anti-insulin immunoglobulin G (IgG) antibodies [5, 7, 8] and anti-insulin receptor antibodies [5, 9] are occasionally detected in patients with diabetes who have brittle glycemic control or severe insulin resistance. The effectiveness of liraglutide remains unclear in these cases.
We report a case in which switching to liraglutide therapy ameliorated both the symptoms of insulin allergy with hypereosinophilia and the characteristics of insulin antibodies in patients with type 2 diabetes mellitus.
Case presentation
History leading to admission
Our patient is a 70-year-old Japanese man with diabetes. He was admitted to our hospital 4.5 years ago for further investigation and treatment of recent-onset insulin allergy. He had a 30-year history of type 2 diabetes mellitus and an approximately 6-year history of insulin injection administration, with an average glycated hemoglobin (HbA1c) level of 7.0 %. He reported a past history of myocardial infarction, hypertension, and dyslipidemia and had undergone coronary artery bypass grafting at 68 years. During that surgery, intravenous protamine sulfate was administrated, which induced anaphylaxis.
At that time, 4.5 years prior to the current presentation, he was receiving 100 mg/day aspirin, 100 mg/day clopidogrel, 100 mg/day imidapril, 20 mg/day isosorbide mononitrate, 5 mg/day nicorandil, and 10 mg/day pravastatin, and these had been stable for more than 3 years. In addition, he was being treated with 20 U/day insulin lispro and 8 U/day insulin glargine, which had also been unchanged for approximately 3 years. Finally, he had been receiving 0.9 mg/day voglibose to supplement his insulin therapy for approximately 5 years.
Approximately 5 months before his current presentation, he reported that he had started to develop wheals with redness and itching at the injection site, which occurred immediately after injecting insulin. Of note, these symptoms only occurred following the use of insulin lispro and not after insulin glargine. Over the subsequent months, part of the surface of the wheals had hardened and further investigation was performed. Two months before his current admission, blood samples were taken that revealed high serum levels of human insulin-specific immunoglobulin E (IgE; 4.21 U/mL; normal range <0.35 U/mL), hypereosinophilia (total white blood cell count, 6000/μL; total eosinophil granulocyte count, 780; percentage, 13 %), and a high level of total IgE (403 IU/mL; normal range <173 IU/mL). A pathology sample was taken 2 months before his admission, which revealed eosinophilic infiltration in the hardened part of the injection site (Fig. 1). Therefore, just before admission, he was diagnosed with insulin allergy and was switched from insulin lispro plus glargine to insulin aspart plus glargine.
Fig. 1.
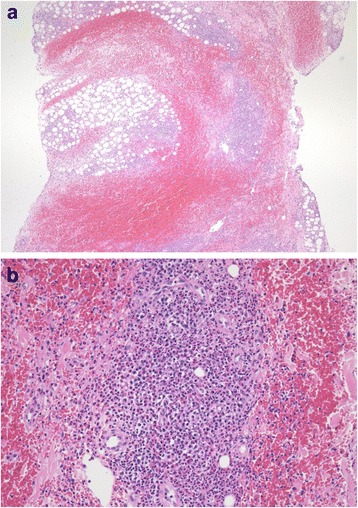
Pathological findings of insulin injection site. Specimen obtained from the injection site of a 70-year-old man with diabetes. Hematoxylin–eosin staining showing predominant infiltration of eosinophils and the simultaneous infiltration of lymphocytic cells in the subcutaneous tissue. a Low-power micrograph 40×. b High-power micrograph 200×
Admission for further investigation
He was admitted to our hospital for treatment of his poor glycemic control and to evaluate his residual ability of insulin secretion. On admission, his physical examination was largely unremarkable (height, 157.1 cm; body weight, 53.0 kg; body mass index, 21.5 kg/m2; body temperature, 36.5 °C; blood pressure, 104/62 mmHg; and pulse, 75 beats/minute, regular), although there was induration with slight redness of his abdomen. His laboratory data at that time are shown in Table 1. Of note, his fasting plasma glucose level was 346 mg/dL, and his HbA1c level was 12.3 %. In addition, we detected high serum insulin, anti-insulin IgG antibody, and anti-insulin receptor antibody levels. Although there were no further symptoms of local allergy (wheals, redness, and itching) after injecting with insulin aspart, his postprandial hyperglycemia continued at approximately 350 mg/dL. Therefore, he was switched to insulin glulisine (24 U/day) plus glargine (22 U/day) after admission. On day 17 of admission, his 24-hour blood glucose profile was 88, 241, 241, 295, 297, and 292 mg/dL at 7:30 a.m., 10:00 a.m., 11:30 a.m., 2:00 p.m., 5:30 p.m., and 8:00 p.m., respectively, with an energy intake of 1600 kcal. On days 18 and 19, his urinary excretion of C-peptide was 42.8 to 51.5 μg/day (normal range 40 to 100 μg/day). On day 19, a glucagon stimulation test was performed, and his serum C-peptide response ranged from 3.2 to 3.6 ng/mL (Table 2). On day 22, although his high glucose levels persisted, he was discharged because we had confirmed his ability to secrete insulin. Although his hypereosinophilia was not resolved before his discharge, he had no further allergic symptoms (wheals, redness, and itchiness) at the injection site for glulisine, and the area of induration had almost disappeared.
Table 1.
Laboratory data on admission
| Parameter | Result (normal range) | Unit | Parameter | Result (normal range) | Unit |
|---|---|---|---|---|---|
| WBC | 5400 (2800–8800) | /μL | γGTP | 32 (10–47) | IU/L |
| Neutrophil | 60 (32–79) | % | Ch-E | 378 (214–466) | IU/L |
| Eosinophil | 15 (0–6) | % | T-Bil | 0.9 (0.2–1.2) | mg/dL |
| Lymphocyte | 16 (18–59) | % | Na | 138 (138–146) | mEq/L |
| Monocyte | 7(0–8) | % | K | 4.9 (3.6–4.9) | mEq/L |
| Basophil | 2(0–2) | % | Cl | 99 (99–109) | mEq/L |
| RBC | 406×104 (366–478) | /μL | BUN | 19 (8–22) | mg/dL |
| Hb | 13.0 (11.6–14) | g/dL | Cre | 1.0 (0.4–0.7) | mg/dL |
| Hct | 38.0 (34.1–41.7) | % | UA | 4.7 (2.3–7) | mg/dL |
| Plt | 15.2×104 (14.7–34.1) | /μL | CK | 209 (62–287) | IU/L |
| AMY | 46 (42–132) | IU/L | |||
| TP | 7.2 (6.7–8.3) | g/dL | CRP | 0.62 (0–0.3) | mg/dL |
| Alb | 3.6 (3.9–4.9) | g/dL | TC | 149 (150–219) | mg/dL |
| AST | 33 (13–33) | IU/L | TG | 70 (30–150) | mg/dL |
| ALT | 28 (6–27) | IU/L | HDL-C | 50 (49–74) | mg/dL |
| FPG | 346 (70–109) | mg/dL | ANA | Negative (<40×) | |
| F-INS | 9774 (5–10) | μIU/mL | CH50 | <12 (25-48) | CH50/mL |
| F-CPR | 4.9 (0.6–2.1) | ng/mL | IgG | 1697 (870–1700) | mg/dL |
| HbA1c | 12.3 (4.7–6.2) | % | Anti-SS-A antibody | Negative | |
| Glycoalbumin | 48.3 (12.4–16.3) | % | Anti-SS-B antibody | Negative | |
| GAD antibody | 0.3 (0–5.0) | U/mL | Anti-ds-DNA antibody | Negative | IU/mL |
| Human IgE | 5.57 (0–0.34) | U/mL | RF | <3 (<15) | IU/mL |
| IgG-anti-insulin antibodies | 14.5 (<0.3) | % | |||
| Anti-insulin receptor antibodies | Positive | Urine test | |||
| ACTH | 11.6 (7.2–63.3) | pg/mL | U-Glucose | (4+) | |
| Cortisol | 17.9 (6.2–19.4) | μg/dL | U-Protein | Negative | |
| TSH | 1.747 (0.5–5.0) | μIU/mL | U-Blood | Negative | |
| FT4 | 2.11 (0.9–1.7) | ng/dL | U-Ketone | Negative | |
ACTH adrenocorticotropic hormone, Alb albumin, ALT alanine aminotransferase, AMY amylase, ANA anti-nuclear antibody, Anti-ds-DNA anti-double-stranded DNA, anti-SS-A anti-Sjögren’s syndrome A, anti-SS-B anti-Sjögren’s syndrome B, AST aspartate aminotransferase, BUN blood urea nitrogen, Ch-E cholinesterase, CK creatine, Cl chloride, Cre creatinine, CRP C-reactive protein, F-CPR fasting C-peptide, F-INS fasting insulin, FPG fasting plasma glucose, FT4 free thyroxine, GAD glutamic acid decarboxylase, γ-GTP γ-glutamyl transpeptidase, Hb hemoglobin, HbA1c glycated hemoglobin, Hct hematocrit, HDL-C high density lipoprotein cholesterol, IgE immunoglobulin E, IgG immunoglobulin G, K potassium, Na sodium, Plt platelets, RBC red blood cells, RF rheumatoid factor, T-Bil total bilirubin, TC total cholesterol, TG triglyceride, TP total protein, TSH thyroid-stimulating hormone, UA uric acid, WBC white blood cells
Table 2.
An evaluation of the patient’s insulin secretion ability
| (a) Glucagon stimulation test | ||
| Time (minutes) | 0 | 6 |
| PG (pg/mL) | 112 | 122 |
| CP (ng/mL) | 3.2 | 3.6 |
| (b) Urinary C-peptide excretion | ||
| Day 18 (after admission) | Day 19 | Normal range |
| 42.8 μg/day | 51.5 μg/day | (40–100) |
CP C-peptide, PG plasma glucose
Progress since discharge and the introduction of liraglutide
Figure 2 summarizes his clinical course before and after the introduction of liraglutide. After discharge, although glulisine and glargine were continued, his glycemic control worsened, with fasting and postprandial hyperglycemia levels of approximately 200 and 300 mg/dL, respectively, and high serum insulin levels persisted (Fig. 2). Therefore, 2 months after his admission, we added 0.3 mg liraglutide, discontinued the insulin glulisine, and added 2 mg/day glimepiride and 500 mg/day metformin to decrease his total insulin requirements. However, he also started to develop intermittent induration at the site of insulin glargine injection, and although there was no evidence of wheals or redness, his hypereosinophilia persisted. Based on these findings, we reduced the dose of glargine (32→22→26→10 U/day) and increased the dose of liraglutide (0.3→0.6→0.9 mg/day; Fig. 2). This was followed by improvements in his HbA1c, serum insulin, and eosinophil levels; therefore, we decided to completely discontinue the insulin glargine after 3 months of the liraglutide treatment. On his new regimen of 0.9 mg/day liraglutide in combination with oral antidiabetic agents, his HbA1c level was maintained at approximately 7.0 %. The induration had almost completely disappeared 1 month after the insulin injections were discontinued. In addition, his body weight decreased by 1.7 kg after 2 months of liraglutide treatment. Table 3 indicates his diabetes-related medical history.
Fig. 2.
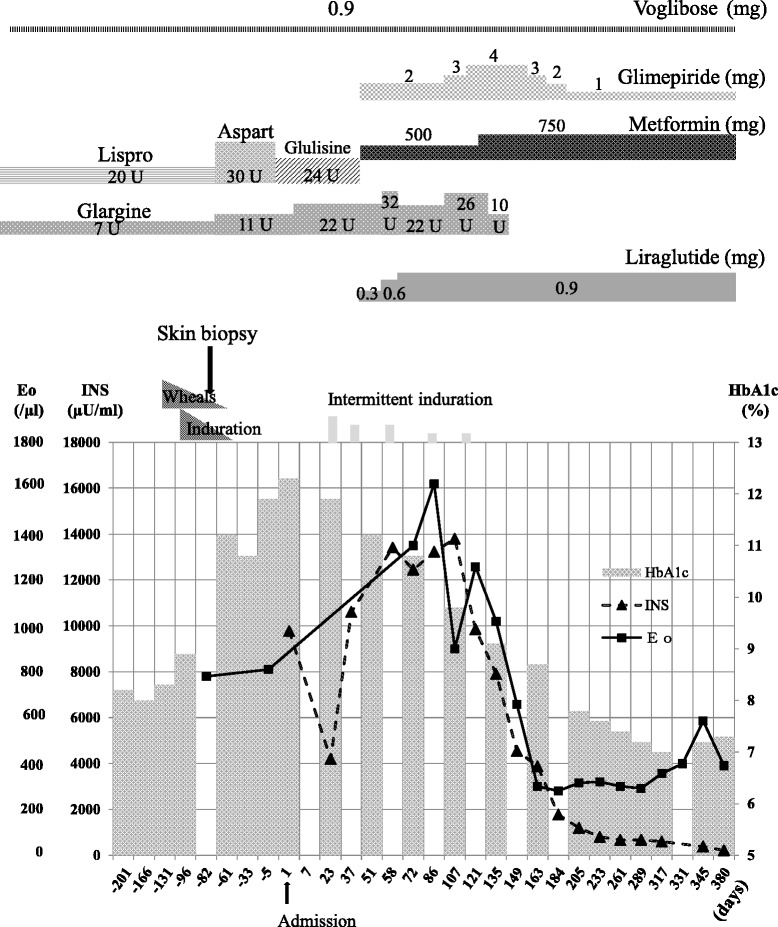
Clinical course. A diagrammatic representation of the treatment and clinical course of a 70-year-old man with insulin allergy. He presented 5 months before his admission with wheals, redness, and itching immediately after insulin lispro injection that progressed to an area of induration. A skin biopsy and blood tests confirmed an allergy, and after trialing different insulin regimens, we eventually introduced 0.3 mg liraglutide, discontinued the short-acting insulin, and added glimepiride and metformin. However, induration intermittently occurred after insulin glargine injections, so we eventually cross-tapered him off glargine and onto a liraglutide dose of 0.9 mg. Thereafter, his glycated hemoglobin, insulin, and eosinophil levels gradually improved. Eo eosinophil count, HbA1c glycated hemoglobin, INS insulin dose
Table 3.
Timeline
| Time | Diabetes-related medical history |
|---|---|
| 1980s | Onset of type 2 diabetes mellitus |
| 2009 | Coronary artery bypass grafting was conducted, anaphylaxis induced by intravenous protamine sulfate administration |
| May 2011 | Onset of allergy |
| July 2011 | A skin biopsy and blood tests confirmed an allergy |
| 24 October 2011 | Admission to our hospital |
| 14 December 2011 | Liraglutide was additionally started |
| 21 March 2012 | Insulin discontinued |
| April 2012 | The symptoms of insulin allergy almost disappeared |
Supplemental investigation by Scatchard analysis
We also elucidated the changes in the characteristics of anti-insulin IgG antibodies before and after treatment with liraglutide. Scatchard analysis of the anti-insulin IgG antibodies in our case indicated that there was a high binding capacity and a low affinity constant for the high affinity sites before liraglutide therapy (Fig. 3). After 1 year of treatment with liraglutide, the Scatchard plot indicated that the binding capacity decreased and the affinity constant for high affinity sites increased (Fig. 3). Of interest, the anti-insulin receptor antibodies had completely disappeared within 1 year after the injection of liraglutide.
Fig. 3.
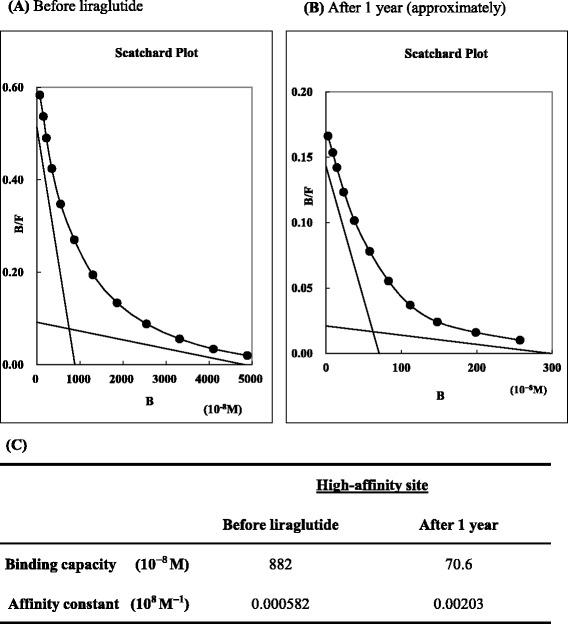
Scatchard plot analysis showing changes in the characteristics of anti-insulin immunoglobulin G antibodies before and after treatment with liraglutide. a The Scatchard plot analysis before the treatment of liraglutide. The binding capacity (10−8 M) of 882 (R1) and affinity constant (108 M−1) of 0.000582 (K1) for high affinity sites and the binding capacity (10−8 M) of 4830 (R2) and affinity constant (108 M−1) of 0.0000190 (K2) for low affinity sites are shown. b The Scatchard plot analysis approximately 1 year after starting liraglutide. The binding capacity (10−8 M) of 70.6 (R1) and affinity constant (108 M−1) of 0.00203 (K1) for high affinity sites and the binding capacity (10−8 M) of 298 (R2) and affinity constant (108 M−1) of 0.0000704 (K2) for low affinity sites are shown. c It is notable, particularly for high affinity sites, that the binding capacity (10−8 M) has changed from 882 to 70.6 (R1), and that the affinity constant (108 M−1) has changed from 0.000582 to 0.00203 (K1). Therefore, liraglutide appeared to induce a decrease in the binding capacity and an increase in the affinity constant for high affinity sites of anti-insulin immunoglobulin G antibodies
Discussion
There are two important aspects to this case. First, we provide a rare description of insulin allergy associated with hypereosinophilia, anti-insulin IgG antibodies, and anti-insulin receptor antibodies in a patient with type 2 diabetes. Second, the marked efficacy of liraglutide suggests that it is useful in all patients with type 2 diabetes.
In our case, we believed that our patient’s insulin allergy was predominantly a type 1 immediate reaction based on the fact that insulin lispro induced wheals, itching, and redness immediately after injection. Insulin allergy is typically classified into the following: type 1, which is local and immediate; type 3, which is related to autoimmune disease; and type 4, which is a delayed-type reaction. Symptoms typically manifest within 15 minutes after insulin injection in type 1 reactions, approximately 6 hours after type 3 reactions, and 8 to 24 hours after type 4 reactions [2]. In our case, we confirmed that insulin lispro induced immediate symptoms but could not detect whether insulin aspart, glulisine, or glargine induced those reactions.
We did not perform either a skin prick test or an intradermal reaction test because of his past history of anaphylaxis. Out of concern for his safety, we substituted this with a non-invasive drug-induced lymphocyte stimulation test, which is normally considered best suited for delayed (type 4) reactions. Although this test was only positive for insulin glulisine, false-negatives have been detected in some cases, and we considered that insulin glargine was also associated with delayed-type allergy. This was because insulin glargine induced occasional induration and because its discontinuation resulted in improvement of both his induration and hypereosinophilia.
Based on the combination of these results, we concluded that our patient probably had a delayed-type allergic reaction that was complicated with a predominant immediate-type allergic reaction. Unfortunately, we did not obtain the list of additives in either the lispro or glargine, which could have been important given his history of protamine anaphylaxis. Further investigation of the cause of the allergic reactions could not be performed (for example, to other insulin preparations, including human neutral protamine Hagedorn, or the additives in the insulin types).
Previous reports of type 1 and type 4 allergic reactions to insulin indicate that hypereosinophilia does not necessarily occur. Therefore, the increase of eosinophilia in both local pathological findings and systemic peripheral blood is characteristic of this case. In general, although numerous eosinophils tend to be detected in local inflammation, such as allergy and parasitic infection, the complete role of eosinophils has not been elucidated [10]. For example, although hypereosinophilia has been observed in invasive parasitic infection, it has recently been reported to be partially defensive in vivo during such an infection [10]. In clinical cases of hypereosinophilia, clinicians must quickly and accurately diagnose the cause [11]. Potential causes in the differential diagnosis of this case included adrenal crisis, parasitic infection, and blood disease, but there was no evidence in support of these diagnoses. In addition, because hypereosinophilia simultaneously occurred with the observed clinical course of insulin allergy, we considered insulin allergy the more likely cause. This was consistent with the report by Nagai et al., who reported a case of immediate-type allergy against human insulin associated with marked eosinophilia in a patient with type 2 diabetes [12]. In that case, there was also the possibility of hypereosinophilia-induced renal dysfunction. Although there was no evidence of organ damage in this case, clinicians should remain vigilant for hypereosinophilic syndrome [11, 13] and be prepared to initiate appropriate therapy. Currently, treatment options for insulin allergy include switching to another insulin device [2], steroid therapy [2], continuous subcutaneous insulin infusion therapy [14], hyposensitization therapy [15], pancreas transplantation [16], and anti-IgE therapy [17]. However, reports on the treatment of insulin allergy with hypereosinophilia are limited, and new options are needed. Therefore, we consider that liraglutide might be a breakthrough agent in the treatment of insulin allergy with hypereosinophilia in patients with type 2 diabetes. Indeed, a Medline search indicated that at the time of writing, we are the first to report the beneficial effects of liraglutide in this setting.
Treatment with liraglutide requires the residual ability to secrete insulin, but we do not know the appropriate range of insulin secretion that is necessary. Previously, Kozawa et al. reported that the cut-off value for predicting the efficacy of liraglutide was 1.1 for the C-peptide index, which was defined as (fasting C-peptide/glucose)×100, and 1.5 ng/mL for fasting C-peptide [18]. This index was also shown to be a predictive marker for the beta-cell area of the human pancreas [18, 19]. Using a glucagon stimulation test, Usui et al. reported that a ΔC-peptide value of 1.34 ng/mL was a useful cut-off point for switching from insulin to liraglutide without developing hyperglycemia [20]. In this case, although the fasting C-peptide level was high, the ΔC-peptide in the glucagon stimulation test had a low value. Therefore, liraglutide alone could not inhibit postprandial hyperglycemia, and the antidiabetic drugs glimepiride, metformin, and voglibose were combined to achieve complete control. Moreover, the additional benefits of liraglutide include the prevention of body weight gain due to its appetite-suppressing and gastrointestinal peristaltic movement-suppressing effects. Liraglutide and other GLP-1 analogs have been reported to decrease body weight [21, 22]. In our case, our patient’s body weight decreased by only 1.7 kg, but we could not administer liraglutide at a dose of more than 0.9 mg/day. Worldwide, liraglutide is administered at a maximum dose of 1.8 mg/day; however, Japan has approved that liraglutide is administered at a maximum dose of only 0.9 mg/day.
In our case, it is possible that both the insulin allergy and the presence of anti-insulin IgG antibodies themselves aggravated our patient’s glycemic control. Therefore, we tested his human leukocyte antigen-antigen D related (HLA-DR) type based on the report by Uchigata et al. that insulin autoimmune syndromes are strongly associated with HLA-DR4 [23]. We showed that his HLA-DR types were DR-9 and DR-15 and not DR-4; thus, we did not consider HLA-DR to be a potential causative factor for producing anti-insulin IgG antibodies. Instead, we concluded that the anti-insulin IgG antibodies resulted from the exogenous insulin injection; thus, we used liraglutide without insulin to decrease the amount of anti-insulin IgG antibodies and to avoid further production.
Moreover, Scatchard analysis of the anti-insulin IgG antibodies in our patient indicated that there was a high binding capacity and a low affinity constant for high affinity sites. At high affinity sites, Eguchi reported two important patterns of Scatchard analysis of anti-insulin IgG antibodies: (1) high (11.5 to 53.2) binding capacity (10−8 M) and low (0.04 to 0.21) affinity constant (108 M−1) in insulin autoimmune syndrome; and (2) low (0.12 to 1.1) binding capacity (10−8 M) and high (1.45 to 7.11) affinity constant (108 M−1) in patients with diabetes requiring insulin therapy [24]. Therefore, the binding capacity and low affinity constant for high affinity sites are clinically important. Our patient had extremely high binding capacity and a low affinity constant compared with previous reports. In particular, his extremely high binding capacity was consistent with his high serum insulin levels.
After insulin therapy was discontinued and liraglutide was started, the characteristics of his anti-insulin IgG antibodies changed. Specifically, there was a notable decrease in the binding capacity and an increase in the affinity constant for high affinity sites (Fig. 3c). Hara et al. have reported that antibody-mediated insulin resistance was treated by ceasing insulin administration [25], whereas Tamura et al. reported that liraglutide ameliorated the anti-insulin IgG antibodies in a woman with type 2 diabetes receiving hemodialysis [26]. In our case, it remains unclear how liraglutide might have affected our patient’s anti-insulin IgG antibodies; however, a recent report has suggested that GLP-1 receptor agonists interact with the immune system. In fact, Hadjiyanni et al. reported that GLP-1 receptor signaling selectively regulated murine lymphocyte proliferation and maintained peripheral regulatory T cells in vivo [27]. In humans, the effects of GLP-1 receptor agonists have been investigated in patients with psoriasis [28], but the detailed mechanism of action is unclear. Further reports are needed to improve our understanding of these agents.
Anti-insulin IgG antibodies have often been reported to induce hypoglycemia [8, 21, 23, 24]. Unfortunately, we could not measure serum free insulin levels directly and do not know whether the anti-insulin receptor antibodies really affected glucose metabolism. Although Kim et al. reported that anti-insulin receptor antibodies often coexist with anti-insulin IgG antibodies in Korean patients with insulin autoimmune syndrome [29], the clinical significance of this finding has been not determined. Acquired anti-insulin receptor antibodies induce severe insulin resistance, called “type B insulin resistance,” which was first reported in 1976 [30]. However, we found no other symptoms or complications of type B insulin resistance, such as acanthosis nigricans or autoimmune disease (Table 1). In addition, the total insulin dose was smaller than that reported in previous studies on type B insulin resistance [9, 30]. Therefore, we considered the possibility that the presence of anti-insulin receptor antibodies is a false positive in this case. It is also possible that the method of measurement led to a false positive result for anti-insulin receptor antibodies because we used the insulin binding inhibition method [31]. Perhaps the high insulin levels and high anti-insulin IgG antibodies levels had some unknown influence on the measured values.
Conclusions
In conclusion, our case suggests that liraglutide is a useful treatment for insulin allergy associated with hypereosinophilia and anti-insulin IgG antibodies in patients with type 2 diabetes. However, similar case reports are limited, and further reports are clearly needed.
Abbreviations
GLP-1, glucagon-like peptide-1; HbA1c, glycated hemoglobin; HLA-DR, human leukocyte antigen-antigen D related; IgE, immunoglobulin E; IgG, immunoglobulin G
Acknowledgements
We gratefully acknowledge our patient who allowed us to publish his case. This work was supported in part by a Grant-in-Aid Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology (HS). The contents are solely the responsibility of the authors and do not represent the official views of the funding institutions.
Authors’ contributions
HH and HS were responsible for the original manuscript design and draft, and the acquisition of data. HH, HS, and TW were involved in drafting the manuscript. HS was involved in revision of the manuscript for important intellectual content. HH, EO, NK, TK, NM, and KH were the physicians involved in patient management. All authors read and approved the final manuscript.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Contributor Information
Hiroyuki Hirai, Email: hiroyuki@fmu.ac.jp.
Emi Ogata, Email: ogtem@fmu.ac.jp.
Nobuyuki Kikuchi, Email: nobuyuk@fmu.ac.jp.
Teruyuki Kohno, Email: tkono@fmu.ac.jp.
Noritaka Machii, Email: noritaka@fmu.ac.jp.
Koji Hasegawa, Email: koji-h@fmu.ac.jp.
Tsuyoshi Watanabe, Email: twat0423@fmu.ac.jp.
Hiroaki Satoh, Email: hiroakis-tky@umin.ac.jp.
References
- 1.Ghazavi MK, Johnston GA. Insulin allergy. Clin Dermatol. 2011;29(3):300–5. doi: 10.1016/j.clindermatol.2010.11.009. [DOI] [PubMed] [Google Scholar]
- 2.Heinzerling L, Raile K, Rochlitz H, Zuberbier T, Worm M. Insulin allergy: clinical manifestations and management strategies. Allergy. 2008;63(2):148–55. doi: 10.1111/j.1398-9995.2007.01567.x. [DOI] [PubMed] [Google Scholar]
- 3.Blanco C, Castillo R, Quiralte J, Delgado J, García I, de Pablos P, et al. Anaphylaxis to subcutaneous neutral protamine Hagedorn insulin with simultaneous sensitization to protamine and insulin. Allergy. 1996;51(6):421–4. [PubMed] [Google Scholar]
- 4.Seino Y, Yabe D. Glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1: Incretin actions beyond the pancreas. J Diabetes Investig. 2013;4(2):108–30. doi: 10.1111/jdi.12065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kawanami D, Ito T, Watanabe Y, Kinoshita J, Sakamoto M, Isaka T, et al. Successful control of a case of severe insulin allergy with liraglutide. J Diabetes Investig. 2013;4(1):94–6. doi: 10.1111/j.2040-1124.2012.00239.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fujishiro M, IzumidaY, Takemiya S, Kuwano Y, Takamoto I, Suzuki R, Yamauchi T, Ueki K, Kadowaki T. A case of insulin allergy successfully managed using multihexamer-forming insulin degludec combined with liraglutide. Diabet Med. 2015 Oct 20. doi:10.1111/dme.12998. [DOI] [PubMed]
- 7.Berson SA, Yalow RS, Bauman A, Rothschild MA, Newerly K. Insulin-I131 metabolism in human subjects: demonstration of insulin binding globulin in the circulation of insulin treated subjects. J Clin Invest. 1956;35(2):170–90. doi: 10.1172/JCI103262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Iizuka K, Tomita R, Horikawa Y, Takeda J. A case of glycemic instability and insulin allergy due to anti-insulin antibodies in a patient with type 2 diabetes. Diabetol Int. 2012;3(4):233–8. doi: 10.1007/s13340-012-0077-8. [DOI] [Google Scholar]
- 9.Yamada H, Asano T, Kusaka I, Kakei M, Ishikawa S. Type B insulin resistance syndrome with fasting hypoglycemia and postprandial hyperglycemia. Diabetol Int. 2015;6(2):144–48. doi: 10.1007/s13340-014-0190-y. [DOI] [Google Scholar]
- 10.Blanchard C, Marc E. Rothenberg biology of the eosinophil. Adv Immunol. 2009;101:81–121. doi: 10.1016/S0065-2776(08)01003-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Roufosse F, Weller P. Practical approach to the patient with hypereosinophilia. J Allergy Clin Immunol. 2010;126(1):39–44. doi: 10.1016/j.jaci.2010.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nagai Y, Mori T, Abe T, Nomura G. Immediate-type allergy against human insulin associated with marked eosinophilia in type 2 diabetic patient. Endocr J Jun. 2001;48(3):311–6. doi: 10.1507/endocrj.48.311. [DOI] [PubMed] [Google Scholar]
- 13.Uriuhara A, Morita S, Kuroda S, Fumoto M, Araki S, Aoyama N, et al. Severe insulin-resistant diabetes mellitus associated with hypereosinophilic syndrome. Intern Med. 1994;33(10):632–6. doi: 10.2169/internalmedicine.33.632. [DOI] [PubMed] [Google Scholar]
- 14.Nagai T, Nagai Y, Tomizawa T, Mori M. Immediate-type human insulin allergy successfully treated by continuous subcutaneous insulin infusion. Intern Med. 1997;36(8):575–8. doi: 10.2169/internalmedicine.36.575. [DOI] [PubMed] [Google Scholar]
- 15.Matheu V, Perez E, Hernández M, Díaz E, Darias R, González A, et al. Insulin allergy and resistance successfully treated by desensitisation with aspart insulin. Clin Mol Allergy. 2005;23(3):16. doi: 10.1186/1476-7961-3-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Malaise J, Leonet J, Goffin E, Lefebvre C, Tennstedt D, Vandeleene B, et al. Pancreas transplantation for treatment of generalized allergy to human insulin in type 1 diabetes. Transplant Proc. 2005;37(6):2839. doi: 10.1016/j.transproceed.2005.05.020. [DOI] [PubMed] [Google Scholar]
- 17.Cavelti-Weder C, Muggli B, Keller C, Babians-Brunner A, Biason-Lauber A, Donath MY, et al. Successful use of omalizumab in an inadequately controlled type 2 diabetic patient with severe insulin allergy. Diabetes Care. 2012;35(6):e41. doi: 10.2337/dc12-0115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kozawa J, Inoue K, Iwamoto R, Kurashiki Y, Okauchi Y, Kashine S, et al. Liraglutide is effective in type 2 diabetic patients with sustained endogenous insulin-secreting capacity. J Diabetes Investig. 2012;3(3):294–7. doi: 10.1111/j.2040-1124.2011.00168.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Meier JJ, Menge BA, Breuer TG, Müller CA, Tannapfel A, Uhl W, et al. Functional assessment of pancreatic β-cell area in humans. Diabetes. 2009;58(7):1595–603. doi: 10.2337/db08-1611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Usui R, Yabe D, Kuwata H, Fujiwara S, Watanabe K, Hyo T, et al. Retrospective analysis of safety and efficacy of insulin-to-liraglutide switch in Japanese type 2 diabetes: A caution against inappropriate use in patients with reduced β-cell function. J Diabetes Investig. 2013;4(6):585–94. doi: 10.1111/jdi.12111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jendle J, Nauck MA, Matthews DR, Frid A, Hermansen K, Düring M, et al. LEAD-2 and LEAD-3 Study Groups: Weight loss with liraglutide, a once-daily human glucagon-like peptide-1 analogue for type 2 diabetes treatment as monotherapy or added to metformin, is primarily as a result of a reduction in fat tissue. Diabetes Obes Metab. 2009;11(12):1163–72. doi: 10.1111/j.1463-1326.2009.01158.x. [DOI] [PubMed] [Google Scholar]
- 22.Chen JH, Tang WH, Lee CH. Effective weight loss after treatment with a glucagon-like peptide-1 receptor agonist in a morbidly obese and diabetic patient before bariatric surgery: a case report. J Med Case Rep. 2014;8:304. doi: 10.1186/1752-1947-8-304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Uchigata Y, Kuwata S, Tokunaga K, Eguchi Y, Takayama-Hasumi S, Miyamoto M, et al. Strong association of insulin autoimmune syndrome with HLA-DR4. Lancet. 1992;339(8790):393–4. doi: 10.1016/0140-6736(92)90080-M. [DOI] [PubMed] [Google Scholar]
- 24.Eguchi Y. Scatchard analysis of insulin autoantibodies in the insulin autoimmune syndrome (Japanese) J Tokyo Wom Med Univ. 1989;59:1296–305. [Google Scholar]
- 25.Hara K, Tobe K, Uchigata Y, Nakazono M, Yasuda K, Terauchi Y, et al. Antibody-mediated insulin resistance treated by cessation of insulin administration. Intern Med. 2000;39(2):143–5. doi: 10.2169/internalmedicine.39.143. [DOI] [PubMed] [Google Scholar]
- 26.Tamura Y, Kimbara Y, Funatsuki S, Mabuchi S, Kodera R, Yoshimoto A, et al. A case of insulin antibody-induced glucose instability in an elderly woman with type 2 diabetes on hemodialysis, successfully ameliorated with liraglutide. Diabetol Int. 2013;4(1):71–5. doi: 10.1007/s13340-012-0100-0. [DOI] [Google Scholar]
- 27.Hadjiyanni KA, Siminovitch JS, Drucker DJ. Glucagon-like peptide-1 receptor signaling selectively regulates murine lymphocyte proliferation and maintenance of peripheral regulatory T cells. Diabetologia. 2010;53(4):730–40. doi: 10.1007/s00125-009-1643-x. [DOI] [PubMed] [Google Scholar]
- 28.Al-Badri MR, Azar ST. Effect of glucagon-like peptide-1 receptor agonists in patients with psoriasis. Ther Adv Endocrinol Metab. 2014;5(2):34–8. doi: 10.1177/2042018814543483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kim SW, Won HK, Seok H, Lee BW, Jung CH, Lee WJ, et al. High prevalence of both anti-insulin and anti-insulin receptor antibodies in Korean patients with insulin autoimmune syndrome. Diabetes Res Clin Pract Nov. 2012;98(2):e12–5. doi: 10.1016/j.diabres.2012.09.019. [DOI] [PubMed] [Google Scholar]
- 30.Kahn CR, Flier JS, Bar RS, Archer JA, Gorden P, Martin MM, et al. The syndromes of insulin resistance and acanthosis nigricans – insulin-receptor disorders in man. N Engl J Med. 1976;294:739–45. doi: 10.1056/NEJM197604012941401. [DOI] [PubMed] [Google Scholar]
- 31.Filier JS, Kahn CR, Roth J, Bar RS. Antibodies that impair insulin receptor binding in an unusual diabetic syndrome with severe insulin resistance. Science. 1975;190:63–5. doi: 10.1126/science.170678. [DOI] [PubMed] [Google Scholar]


