Abstract
Pluripotent stem cells (PSCs) hold great promise in regenerative medicine, disease modeling, functional genomics, toxicological studies and cell-based therapeutics due to their unique characteristics of self-renewal and pluripotency. Novel methods for generation of pluripotent stem cells and their differentiation to the specialized cell types such as neuronal cells, myocardial cells, hepatocytes, and beta cells of the pancreas and many other cells of the body are constantly being refined. Pluripotent stem cell derived differentiated cells, including neuronal cells or cardiac cells are ideal for stem cell transplantation as autologous or allogeneic cells from healthy donors due to their minimum risks of rejection.
DNA damage induced by ionizing radiation (IR), ultraviolet (UV) light, genotoxic stress, and other intrinsic and extrinsic factors trigger a series of biochemical reactions termed as DNA damage response (DDR). In order to maintain genomic stability, and avoid transmission of mutations into progenitors cells, stem cells have robust DNA damage response signaling – a contrast to somatic cells. Stem cell transplantation may over come the late effects related to radiation. This review will particularly focus on differential DNA damage response between stem cells and derived differentiated cells and the possible pathways that determine such differences.
Introduction
Stem cells hold great promise in regenerative medicine, disease modeling, functional genomics, toxicological studies and cell-based therapeutics (1–6). Adult stem cells are rare, quiescent with limited self-renewal and differentiation potential. However, emerging evidence suggests that both quiescent and active stem cell populations coexist in several tissues in separate but nearby compartments (7). The main role of adult stem cells in a particular organ is to replenish cells that are lost during physiological or pathological processes (through disease and injury) (8, 9). Embryonic stem cells (ESC) and induced pluripotent stem cells (iPSCs) exhibit unique characteristics such as robust self-renewal and pluripotency. Self-renewal allows ES cells to grow for extended periods without loss of genomic integrity. Pluripotent stem cells have the ability to differentiate into derivatives of all three germ layers – ectoderm, mesoderm and endoderm – and hence have the ability to generate any tissue specific cell of the body (10–13). This review is particularly focused on demonstrating the differential DNA damage response (DDR) between somatic and pluripotent stem cells. In addition to focusing on our contribution to the stem cell field pertaining to Nitric oxide-cyclic GMP and the DNA damage response field in general, we have discussed major findings in both these areas of research wherever applicable. We however, do apologize to many investigators for omissions made, as we are unable to include all publications in the stem cell and DDR areas.
Embryonic Stem (ES) Cells and induced Pluripotent Stem (iPS) Cells
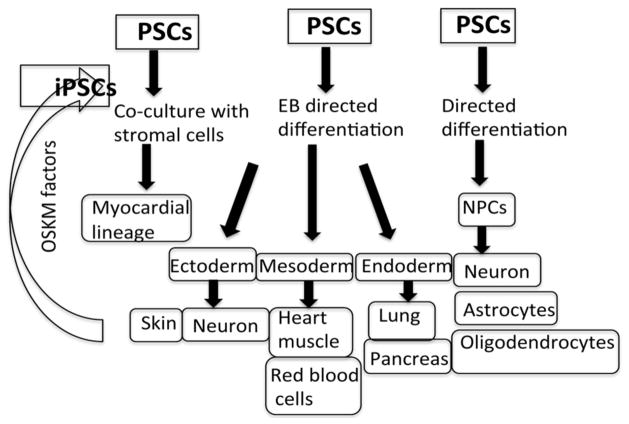
Evans and coworkers (14) and Martin (15) were the first to describe the derivation of mouse ES cells from the blastocyst inner cell mass (ICM) that were able to proliferate indefinitely while maintaining pluripotency. Subsequently, Thompson and co-workers (10) were the first to derive human ES cells from the blastocyst ICM of a human pre-implantation embryo. Both cell lines could proliferate continuously under the appropriate conditions for prolonged periods of time. Furthermore, ES cells have shown to be differentiated into cardiomyocytes, neural progenitors, trophoblastic cells, endothelial cells, hepatocyte-like cells, osteoblasts, hematopoietic lineages, insulin-expressing cells and many other cells of the body (2, 16) using either directed differentiation, EB-directed differentiation or by stromal co-culture methods (10, 17). A simplified version of various protocols (directed, EB directed and stromal co culture method) for the differentiation of derivatives of the three germ layers (ectoderm, mesoderm and endoderm) and induction of pluripotent stem cells from fibroblast (somatic cells) by introduction of OSKM factors (OCT4, SOX2, KLF-4 and c-myc) are presented in Figure 1.
Figure 1.
Pluripotent stem cell self-renewal and pluripotency. Various protocols (directed, EB directed and stromal co culture method) for the differentiation of stem cells into derivatives of the three germ layers (ectoderm, mesoderm and endoderm) and induction of pluripotent stem cells from fibroblast (somatic cells) by introduction of OSKM factors. OSKM= OCT4, SOX2, KLF-4 and c-myc
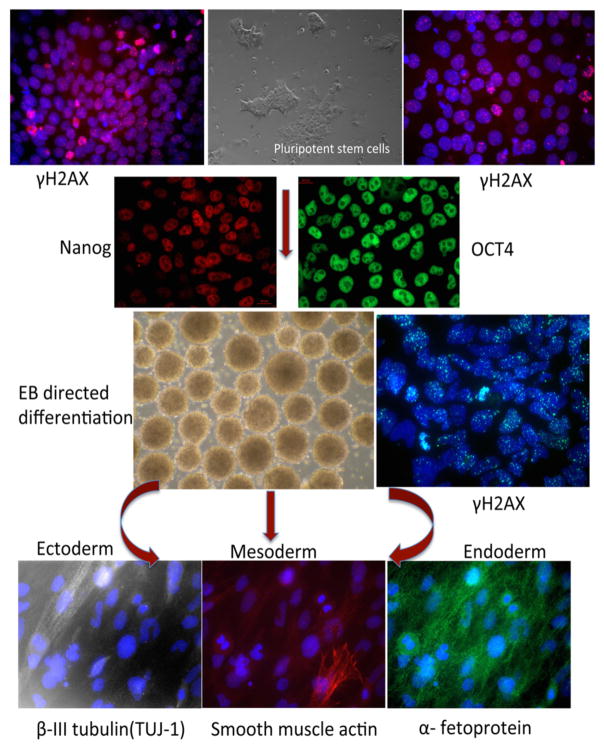
A number of signaling pathways such as Wnt/β catenin (18), PI3K (19, 20), MAPK (19, 21), and Nitric Oxide (22) have shown to be involved in the proliferation and differentiation of stem cells. Our previous work (23–27) has helped to establish the role of NO-cGMP in the proliferation and differentiation of stem cells. Differential expression and functions of various NO signaling components were observed during mouse and human ES cell differentiation (23, 24). Furthermore, the results demonstrated that the exposure of ES cells to NO donors and various soluble guanylyl cyclase (sGC) activators alone or in combination induces differentiation of stem cells into myocardial cells with a robust increase in second messenger cyclic GMP (cGMP) accumulation (25). The aforesaid results suggested that the regulation of sGC expression and activity might be important for directing the stem cell differentiation. This model was further supported by subsequent studies which indicated that the gene encoding the sGCα1 subunit can undergo alternative splicing during ES cell differentiation and that the C-type sGC α1 splice variant is highly expressed in differentiating cells and has an intracellular distribution that varies from the canonical sGCα1 subunit (27). Interestingly, differentiation of ES cells by polyphenol curcumin was partly due to manipulation of the NO-cGMP pathway (26). The results from studies of the NO-cGMP pathway with human ES cells provided the rationale for exploring the role of NO-cGMP signaling in differentiation of iPS cells. Using pharmacological probes such as NO donors, sGC activators and phosphodiesterase inhibitors suggested that iPS cell differentiation might also be regulated by the NO-cGMP pathway (data unpublished). The differentiation of pluripotent stem cells identified by staining for stem cell markers OCT4 and Nanog into multiple lineages, is represented in Figure 2. Pluripotent stem cells are subsequently differentiated using the EB directed method into the cells of the 3 germ layers and detected by staining of β-III tubulin (ectoderm), smooth muscle actin (mesoderm) and α-fetoprotein (endoderm). In response to IR, γ-H2A.X foci formation (surrogate marker for DSBs) were observed in both PSCs and derived differentiated cells.
Figure 2.
Characterization and DNA damage response of pluripotent stem cells (PSCs) and derived differentiated cells. Immunostaining of PSCs for stem cell markers OCT4 and nanog and their subsequent differentiation using EB directed into the cells of 3 germ layers. The figure shows staining of β-III tubulin (ectoderm), smooth muscle actin (mesoderm) and α-fetoprotein (endoderm). In response to IRγ H2A.X (surrogate marker for DSBs) foci formation was observed in both PSCs and derived differentiated cells.
Transplantation of stem cell-derived differentiated cells into animal disease models of cardiac injury and Parkinson’s disease has indicated beneficial outcomes post-transplantation (28–30). Preclinical studies with human ES-derived oligodendrocytes injected into rats with spinal cord injury also revealed that the condition of the rats improved after the treatment (31). This led to the first clinical trial in patients with spinal cord injuries by Geron using human ES-cell derived oligodendrocytes (http://celltherapies.geron.com/grnopc1_pipeline). Preliminary results of the clinical trial, presented at the American Congress of Rehabilitation Medicine (ACRM), revealed that none of the participants exhibited serious adverse events – albeit no significant changes in the spinal cord and associated neurological conditions (http://celltherapies.geron.com/grnopc1_pipeline).
Induced pluripotent stem cells were initially generated by introducing a set of key transcription factors (OCT4, SOX2, KLF-4 and c-myc) that can reprogram somatic cells, such as fibroblasts to a pluripotent state (12). Subsequently, re-programming of either mouse or human somatic cells was achieved by either the introduction of OSKM factors (12) or a different set of 4 factors (OCT4, SOX2, NANOG and LIN 28) (11, 13, 32–36). Latter, iPS cells without c-myc were generated to avoid a high risk of oncogenesis when using OSKM factors (37). Somatic cells have also been induced to pluripotent cells by the introduction of only two transcription factors (OCT3/4, SOX-2) when combined with valproic acid (VPA), a small molecule histone deacetylase inhibitor (HDAC), which increased the efficiency of primary human fibroblast reprogramming to a pluripotent state (38). Furthermore, since genetic manipulation and nuclear transfer used in reprogramming can limit their use in clinical applications, pluripotent stem cells from mouse somatic cells have also been generated by a combination of seven small molecule compounds. These included several cAMP agonist (Foreskolin, Prostaglandin E2 and Rolipram) and epigenetic modulators (5-azacytidine, sodium butyrate, RG108 and DZnep). These chemically induced pluripotent stem cells; generated from mouse somatic cells, resemble ES cells in terms of their differentiation potential, germ line transmission, gene expression profile and epigenetic profile (39). In addition, a recent study revealed that the chemical reprogramming process requires the early formation of extra-embryonic endoderm (XEN)-like cells and a late transition from XEN-like cells to chemically induced (Ci) PSCs. This is a unique route that fundamentally differs from the pathway of transcription factor-induced reprogramming. These cells are similar to in vivo counterparts at the transcriptional and functional levels (40).
Similar to embryonic stem cells (ES), iPS cells have been differentiated into cells of various lineages including myocardial and neuronal cells (4, 41–45). However, a few studies have shown that lineage specific differentiation is variable between different clones of iPS cells (46). Although iPS cells appear to be similar to ES cells with respect to morphology, global gene expression, protein levels and differentiation potential, several reports suggest that iPS cells do differ from ES cells in gene expression, DNA methylation state and miRNA expression (47–49).
Recent studies have also shown that the direct reprogramming of fibroblasts with a combination of growth and transcription factors has the potential to generate cardiomyocyts, neurons, endothelial cells, hematopoietic stem/progenitor cells and hepatocytes (50). Fibroblasts can be directly differentiated into cardiomyocytes using defined factors (42) and other cell types. Furthermore, a cocktail of 3 transcription factors (Gata4, Mef2c, and Tbx5) was sufficient to reprogram a cardiac fibroblast into induced cardiomyocyte (iCM) like cells (50). Similarly, functional pancreatic beta cells have been generated from pancreatic exocrine cells using a combination of various growth and transcription factors (50). These studies demonstrate that direct reprogramming could aid in studying mechanisms underlying cell fate decisions during the developmental process.
DNA Damage Response
Cells encounter DNA damage through radiation, genotoxic stress, UV exposure, intrinsic and extrinsic insults, which triggers a series of biochemical reactions, termed as DNA damage response (DDR), involved in the sensing and repair of damaged DNA (51–53). Previous studies have shown DDR to be a fundamental and evolutionarily conserved mechanism for preserving the genomic integrity of cells (51, 52). The DDR pathway involves multiple components including: 1) sensors (the MRE11-RAD50-NBS1 (MRN) complex, ataxia-telangiectasia mutated (ATM), ataxia-telangiectasia and Rad3-related (ATR), DNA-dependent protein kinase (DNA-PK); 2) mediators (breast cancer type 1 susceptibility (BRCA1), claspin, p53 binding protein 1 (53BP1), mediator of DNA damage checkpoint 1 (MDC-1); and 3) effectors such as check protein kinase 1 (CHK1) and CHK2) (52, 54, 55). The combined effect of the DDR components results in cell cycle arrest, DNA repair, and/or senescence and induction of programmed cell death or apoptosis (56–58). These responses are coordinated at the transcriptional, post-transcriptional, translational, and post-translational levels. As DDR is important for genomic stability, its dysfunction is associated with tumorigenicity, genetic diseases, and tissue aging (59–61).
Double-stranded break (DSB) is the most lethal form of DNA damage, which is primarily repaired by non-homologous end joining (NHEJ) (62, 63). NHEJ can occur throughout the cell cycle – predominantly in G1 but also in the S and G2 phases when repair by homologous recombination (HR) is also possible. NHEJ is an error prone repair process that starts by the binding of heterodimer KU70/KU80 to free DNA ends, followed by the recruitment of DNA-dependent protein kinase (DNA-PKcs). The DNA ends are then slightly resected by the Artemis and/or MRE11/RAD50/NBS1 (MRN) nucleases and XRCC4, XLF, and DNA ligase IV recruitment is needed for resealing (53, 64, 65). In contrast, HR repair uses a DNA template in the form of a sister chromatid, homologous chromosome, or repeated sequence for high fidelity repair. The HR pathway process involves BRCA2 and BRCA1, RAD51 and a group of RAD51-related proteins, and the MRE11/RAD50/NBS1 complex (66). The major DNA damage sensors that coordinate the DDR for preservation of the genome are ataxia telangiectasia mutated (ATM) and ataxia telangiectasia and RAD3-related (ATR) (64, 66). Mutation or loss of ATM or ATR leads to cell senescence-accelerated aging, and premature death associated with increased genomic instability (54, 55, 67–71). One of the first events marking the DSB is the phosphorylation of the histone H2A isoform H2AX at S139, also referred to as γ-H2AX, which is mediated by ATM, ATR, or DNA-PKcs (72–75). The formation and resolution of γ-H2AX foci is linked to the presence of DSBs and can act as a surrogate marker for DNA damage and DSB repair. Subsequently, repair proteins such as the MRN complex, ATM, and 53BP1 coordinate cell cycle arrest, DNA repair and apoptosis (64, 76). RAD51 is uniquely associated with repair by HR (66). However, formation of γ-H2AX and 53BP1 IR-induced foci (IRIF) is not specific for either HRR or NHEJ.
Our laboratory has been involved in the mechanistic delineation of DDR in somatic cells, meiotic cells and as well as in cancer models for more than two decades. Here we are listing only some of our pertinent publications (76–84). During that process, we have identified the mammalian ortholog of the Drosophila MOF (males absent on the first) that acetylates Histone H4 Lysine 16 as essential for embryogenesis and oncogenesis. Previous studies have indicated that depletion of human MOF (hMOF) in cell lines causes a defective DDR to IR. Among other defects, these cells show genomic instability, chromosomal aberrations and cell cycle perturbation. Our laboratory experiments also demonstrated that ablation of the mouse mof (mMof) gene led to embryonic lethality and cell death while MOF overexpression resulted in increased proliferation, oncogenic transformation and tumor growth (79). Another laboratory also independently reported that Mof is essential for the progression of embryonic development past the blastocyst stage and is also required for normal chromatin architecture (85). In subsequent studies, histone acetyltransferase MOF was found to be a key regulator of the embryonic stem cell core transcriptional network and for the survival of post-mitotic Purkinje or T-cells (80, 81). Embryonic stem cells with Mof deletion lose characteristic morphology, alkaline phosphate staining, and differentiation potential. Mof deleted cells exhibit aberrant expression of the core transcription factors Nanog, Sox2 and Oct4 (86). More recently, MOF was shown to promote generation of iPS cells by interacting with the H3K4 methyltransferase Wdr5 to promote endogenous oct4 expression during the reprogramming process (87).
DNA Damage Response (DDR) and Stem cells
Maintenance of a robust genomic integrity for stem cells is of vital importance because mutations in stem cells will compromise derived cell lineages and their progenitor cells (88, 89). As mentioned earlier, genomic stability can be achieved by the coordination of multiple processes referred to as DDR which includes cell cycle checkpoints, DNA repair and cell death (62, 64). Recent reviews describing DDR in adult stem cells (90) including hematopoietic stem cells (91) as well as multipotent and pluripotent stem cells (92) have been reported. This review article will mainly focus on DDR signaling in embryonic and induced pluripotent stem cells. Compared to somatic cells, fewer DDR components have been identified in pluripotent stem cells. Studies describing DDR in pluripotent stem cells (PSCs) and either derived differentiated cells or other somatic cells provide a wide range of outcomes (Table 1). However, it is clear that ES cells and PSCs have an enhanced activity of DNA repair proteins and DNA DSB repair (92). Upon exposure to DNA damaging agents (UV, IR, H2O2, psoralen), human ESCs showed efficient DNA repair as compared to fibroblast and HeLa cells (93). Several previous studies have demonstrated that HR is the predominant pathway for DNA repair in both mouse and human ES cells (88, 89, 94, 95), although some studies point to the repair in human ES cells post DNA damage by NHEJ pathway (96) during late G2 phase of the cell cycle (97).
Table 1.
Repair pathways of Pluripotent stem cells versus differentiated cells
In another study pertaining to human ES cells and derived neural cells, the authors showed that the disappearance of γ-H2AX foci occurred at slower rate in hESC as compared to neural and astrocyte cells, which may reflect a more complex DNA repair system (88). Moreover, resolution of Rad51 foci, which is reflective of repair by HR was similar in both human ES cells as well as neural cells (but not in astrocytes). Overall, the study demonstrated that hESC utilize ATR dependent HR repair pathway, whereas astrocytes depend on ATM for NHEJ (88). However, in contrast to the earlier study, using human ES cells as a model, the investigators demonstrated that with in 15 minutes post irradiation, ATM kinase gets phosphorylated and localized to the DNA double strand break sites, leading to phosphorylation of downstream targets Chk2, p53 and Nbs1. Exposure to IR resulted in the temporary arrest of ES cells in the G2 phase in an ATM dependent manner, suggesting that human ES cells activate DNA damage checkpoint (98, 99).
Following IR exposure, iPS cells show activation of checkpoint signaling by exhibiting phosphorylation of ATM, NSB1, Chk2 and p53. This study further showed that ATM is localized to DSBs and that p53 was localized to the nucleus of NANOG positive cells (100). As with a previous study (98) with human ES cells, iPS cells are also temporarily arrested in the G2 phase of the cell cycle and a lack of G1/S arrest is displayed in iPS cells (similar to ES cells). Further, both iPS and ES cells form Rad51 foci and show sister chromatid exchanges, thereby demonstrating repair by HR. Compared to their differentiated derivatives both ES and iPS cells show a higher expression of DNA damage signaling gene expression, checkpoint activation and repair of various types of DNA lesions (100). In addition DNA damage processing in human neonatal stromal cells has been compared with adult stromal cells and induced pluripotent stem cells and the data suggested that there are developmental age-dependent differences in DNA damage repair capacity (101). Liedtke and coworkers also reported that a differentiation-dependent downregulation of repair capacity was observed during osteogenic differentiation in neonatal stromal cells (101). Furthermore, it remains to be established whether DNA damage response plays a role in cellular differentiation.
As described earlier, human stem cells have a highly efficient DNA repair system that becomes less efficient as cells differentiate to more specialized cells. Further studies reveal that stem cells have an anaerobic metabolism that reduces mitochondria number and oxidative stress (102), thereby reducing genomic instability. If DNA damage is not repaired, cells undergo senescence, apoptosis or differentiation to avoid continued growth of stem cells with mutations and genomic alteration (102).
Previous studies have shown that hESC are very sensitive to IR with the maximum number of cells undergoing cell death within 7 hrs of IR exposure. In contrast, ES cell derived NSC and neurons were more resistant to IR (103). NSCs, which survived IR, went through senescence and astrocyte characters suggesting distinct genotoxic stress response between ES cells and their differentiated progeny (103). It has been shown that a relatively low dose of IR (1 Gy) can induce apoptosis in hESC while 2 Gy of IR caused a 60% decrease in the hESC survival 5 days post IR exposure and elevated hESC metabolic activity. In addition, 2 and 5 Gy of IR exposure elevated the reactive oxygen species (ROS) and reactive nitrogen species (RNS) for one-week post irradiation. However, other study showed that although irradiated hESCs undergo significant apoptosis after irradiation, they continue to remain pluripotent and maintain the ability to form all three germ layers (such as the formation of teratomas in vivo). Such studies are thought to have implications for tracking cellular regenerative therapies using radiotracer reporter probes (104). Many DNA damaging metabolites, including ROS and aldehydes, cause interstrand crosslinks (ICLs). A recent report established that iPSCs require FA pathway for their growth in vitro and CHK1 was shown to be a critical regulator iPSCs hypersensitivity to DNA damage. Hyperactive CHK1 signaling restricts iPSC self-renewal in the absence of error free DNA repair (104), hence maintaining the genomic stability.
In contrast to somatic cells, pluripotent stem cells exhibit an abbreviated cell cycle profile with very short G1 phase but a longer S phase suggesting that DDR may differ between differentiated cells and stem cells. In normal circumstances, G1 checkpoint activation prevents DNA damaged cells from entering into S-phase. However, the human ESC studies have been contradictory. In one study, the response to IR-induced DNA damage arrested human ESCs in the G2 phase of the cell cycle (105). While a second study indicated, that irradiated human ESCs undergo cell cycle arrest prior to DNA synthesis, thereby demonstrating their ability to activate the G1/S checkpoint (106). An additional study has shown that human ESCs have a leaky G1/S checkpoint (107). Studies have demonstrated that PSCs control genomic integrity by apoptosis rather than DNA repair including both ESCs and iPSCs. These were more susceptible to apoptosis than differentiated cells in spite of their robust repair rates. (108). Another study has reported that upon DNA damage human ESCs undergo rapid p53 mediated apoptosis compared to differentiated cells, an effect attributed to mitochondrial priming (109).
Several stem cell pathways either involved in self-renewal or differentiation have been proposed to cross talk with DNA damage response specific at the choice of selecting DNA DSB. Although it is not clear that any stem cell factor/s are involved in regulating the DNA DSB pathway, recent information indicates that LSD1 and MOF are playing role in stem cell differentiation as well as DNA DSB pathway choice.
Although free radicals in general induce DNA damage, which can result in terminal differentiation most probably by suppressing transcription, however, no clear mechanistic details supporting such argument is available. However, recent extensive review in DNA double-strand break response in stem cells (110) has discussed about the mechanisms by which ESCs respond to DSBs induced by IR and reactive oxygen species (ROS) compared to somatic cells. Further more, Nagaria and coworkers have discussed whether DSB response is altered during the process of reprogramming in iPSCs and the role of DDR in reprogramming of such cells (110). Our unpublished data indicates that damage induced at transcription by CRISPR is preferentially repaired. It is yet to be established, whether nitric oxide signaling influences such DSB repair in transcribing genes, which is currently under intensive investigation.
On the other hand, adult stem cells (ASC) exhibit a lower DNA repair capacity depending on their type and developmental stage. Since adult stem cells are in the quiescent state of the cell cycle, they exhibit a lower level of sensitivity to DNA damage compared to the rapidly growing ES cells (90). In response to DNA damage, adult stem cells progress to the G1 phase of the cell cycle. Therefore, damage is repaired by error prone NHEJ. As a consequence of this, DNA damage accumulation in ASCs can be transmitted into progenitor cells (90, 91). Furthermore, aging contributes to the impairment of stem cell function and age-dependent accumulation of DNA damage in stem cells and in their supporting niche cells seems to be partly responsible for stem cell dysfunction (51). The factors that contribute to the accumulation of DNA damage during aging in adult stem cells include DNA replication errors, telomere shortening, repair system failure and aneuploidy (111, 112) as well as genetic aberrations associated with the early stage development of cancer (51, 98). Cancer stem cells are a small rare population of cells within the bulk of a tumor. These cancer-initiating cells exhibit resistance to DNA damaging agents, chemotherapeutic drugs and possibly other therapeutic modalities. Cancer stem cells and typical stem cells share differentiation potential and response to DNA damage, which is important for cancer therapy with genotoxic agents. Maintenance of the adult stem cell pool is important for normal development, thereby reducing the process of neurodegeneration and premature aging. Defects in the DNA repair processes are manifested in clinical phenotypes of many human genetic diseases (102).
Summary
Perusal of literature reveals that pluripotent stem cells exhibit robust DNA damage response signaling including checkpoint activation, DNA repair, senescence or apoptosis to maintain genomic stability. Pluripotent stem cells have changed regenerative medicine as a great number of disease specific PSCs, especially in the area neurodegenerative and cardiac diseases, have been generated. Reprogramming has further enhanced our understanding pertaining to transcription factors, epigenetic regulators, and micro RNAs that are involved in the cell fate determination in such a way that somatic cells can be directly reprogrammed to generate neurons, cardiomyocytes, endothelial cells and hematopoietic stem/progenitor cells and hepatocytes for cell based therapies. We and others, have shown that PSCs exhibit better DNA damage repair kinetics than derived differentiated cells or somatic cells and their repair by the HR repair pathway is the method of choice for PSCs (some of the data from our group is unpublished). However, some studies do indicate a role for NHEJ during late G2 phase in repair by PSCs. Although a majority of human PSCs undergo apoptosis in response to DNA damage, a subpopulation can proliferate with mutations and damaged DNA that can lead to daughter cells with altered genome. Therefore, PSCs and derived differentiated cells need to be tested prior to their use in clinical medicine. Understanding the molecular mechanism of the DDR in PSCs will improve the production, quality and utility of stem cells in the rapidly expanding area of regenerative medicine.
Acknowledgments
This work was supported by the funds from National Institutes of Health, National Cancer Institute Grants R01CA129537, R01CA154320 and GM109768.
References
- 1.Inoue H, Nagata N, Kurokawa H, Yamanaka S. iPS cells: a game changer for future medicine. EMBO J. 2014;33(5):409–17. doi: 10.1002/embj.201387098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lerou PH, Daley GQ. Therapeutic potential of embryonic stem cells. Blood Rev. 2005;19(6):321–31. doi: 10.1016/j.blre.2005.01.005. [DOI] [PubMed] [Google Scholar]
- 3.Sanchez Alvarado A, Yamanaka S. Rethinking differentiation: stem cells, regeneration, and plasticity. Cell. 2014;157(1):110–9. doi: 10.1016/j.cell.2014.02.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhang J, Wilson GF, Soerens AG, Koonce CH, Yu J, Palecek SP, et al. Functional cardiomyocytes derived from human induced pluripotent stem cells. Circ Res. 2009;104(4):e30–41. doi: 10.1161/CIRCRESAHA.108.192237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yamanaka S. Induced pluripotent stem cells: past, present, and future. Cell Stem Cell. 2012;10(6):678–84. doi: 10.1016/j.stem.2012.05.005. [DOI] [PubMed] [Google Scholar]
- 6.Wong WT, Sayed N, Cooke JP. Induced pluripotent stem cells: how they will change the practice of cardiovascular medicine. Methodist Debakey Cardiovasc J. 2013;9(4):206–9. doi: 10.14797/mdcj-9-4-206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li L, Clevers H. Coexistence of quiescent and active adult stem cells in mammals. Science. 2010;327(5965):542–5. doi: 10.1126/science.1180794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Weissman IL, Anderson DJ, Gage F. Stem and progenitor cells: origins, phenotypes, lineage commitments, and transdifferentiations. Annu Rev Cell Dev Biol. 2001;17:387–403. doi: 10.1146/annurev.cellbio.17.1.387. [DOI] [PubMed] [Google Scholar]
- 9.Seita J, Weissman IL. Hematopoietic stem cell: self-renewal versus differentiation. Wiley Interdiscip Rev Syst Biol Med. 2010;2(6):640–53. doi: 10.1002/wsbm.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282(5391):1145–7. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 11.Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131(5):861–72. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 12.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663–76. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 13.Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318(5858):1917–20. doi: 10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- 14.Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292(5819):154–6. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- 15.Martin GR. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci U S A. 1981;78(12):7634–8. doi: 10.1073/pnas.78.12.7634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Reubinoff BE, Pera MF, Fong CY, Trounson A, Bongso A. Embryonic stem cell lines from human blastocysts: somatic differentiation in vitro. Nat Biotechnol. 2000;18(4):399–404. doi: 10.1038/74447. [DOI] [PubMed] [Google Scholar]
- 17.Dambrot C, Passier R, Atsma D, Mummery CL. Cardiomyocyte differentiation of pluripotent stem cells and their use as cardiac disease models. Biochem J. 2011;434(1):25–35. doi: 10.1042/BJ20101707. [DOI] [PubMed] [Google Scholar]
- 18.Singh AM, Li FQ, Hamazaki T, Kasahara H, Takemaru K, Terada N. Chibby, an antagonist of the Wnt/beta-catenin pathway, facilitates cardiomyocyte differentiation of murine embryonic stem cells. Circulation. 2007;115(5):617–26. doi: 10.1161/CIRCULATIONAHA.106.642298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sauer H, Rahimi G, Hescheler J, Wartenberg M. Role of reactive oxygen species and phosphatidylinositol 3-kinase in cardiomyocyte differentiation of embryonic stem cells. FEBS Lett. 2000;476(3):218–23. doi: 10.1016/s0014-5793(00)01747-6. [DOI] [PubMed] [Google Scholar]
- 20.Klinz F, Bloch W, Addicks K, Hescheler J. Inhibition of phosphatidylinositol-3-kinase blocks development of functional embryonic cardiomyocytes. Exp Cell Res. 1999;247(1):79–83. doi: 10.1006/excr.1998.4309. [DOI] [PubMed] [Google Scholar]
- 21.Li J, Wang G, Wang C, Zhao Y, Zhang H, Tan Z, et al. MEK/ERK signaling contributes to the maintenance of human embryonic stem cell self-renewal. Differentiation. 2007;75(4):299–307. doi: 10.1111/j.1432-0436.2006.00143.x. [DOI] [PubMed] [Google Scholar]
- 22.Krumenacker JS, Murad F. NO-cGMP signaling in development and stem cells. Mol Genet Metab. 2006;87(4):311–4. doi: 10.1016/j.ymgme.2005.10.009. [DOI] [PubMed] [Google Scholar]
- 23.Krumenacker JS, Katsuki S, Kots A, Murad F. Differential expression of genes involved in cGMP-dependent nitric oxide signaling in murine embryonic stem (ES) cells and ES cell-derived cardiomyocytes. Nitric Oxide. 2006;14(1):1–11. doi: 10.1016/j.niox.2005.06.010. [DOI] [PubMed] [Google Scholar]
- 24.Mujoo K, Krumenacker JS, Wada Y, Murad F. Differential expression of nitric oxide signaling components in undifferentiated and differentiated human embryonic stem cells. Stem Cells Dev. 2006;15(6):779–87. doi: 10.1089/scd.2006.15.779. [DOI] [PubMed] [Google Scholar]
- 25.Mujoo K, Sharin VG, Bryan NS, Krumenacker JS, Sloan C, Parveen S, et al. Role of nitric oxide signaling components in differentiation of embryonic stem cells into myocardial cells. Proc Natl Acad Sci U S A. 2008;105(48):18924–9. doi: 10.1073/pnas.0810230105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mujoo K, Nikonoff LE, Sharin VG, Bryan NS, Kots AY, Murad F. Curcumin induces differentiation of embryonic stem cells through possible modulation of nitric oxide-cyclic GMP pathway. Protein Cell. 2012;3(7):535–44. doi: 10.1007/s13238-012-2053-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sharin VG, Mujoo K, Kots AY, Martin E, Murad F, Sharina IG. Nitric oxide receptor soluble guanylyl cyclase undergoes splicing regulation in differentiating human embryonic cells. Stem Cells Dev. 2011;20(7):1287–93. doi: 10.1089/scd.2010.0411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Min JY, Yang Y, Converso KL, Liu L, Huang Q, Morgan JP, et al. Transplantation of embryonic stem cells improves cardiac function in postinfarcted rats. J Appl Physiol (1985) 2002;92(1):288–96. doi: 10.1152/jappl.2002.92.1.288. [DOI] [PubMed] [Google Scholar]
- 29.Kim JH, Auerbach JM, Rodriguez-Gomez JA, Velasco I, Gavin D, Lumelsky N, et al. Dopamine neurons derived from embryonic stem cells function in an animal model of Parkinson’s disease. Nature. 2002;418(6893):50–6. doi: 10.1038/nature00900. [DOI] [PubMed] [Google Scholar]
- 30.Min JY, Yang Y, Sullivan MF, Ke Q, Converso KL, Chen Y, et al. Long-term improvement of cardiac function in rats after infarction by transplantation of embryonic stem cells. J Thorac Cardiovasc Surg. 2003;125(2):361–9. doi: 10.1067/mtc.2003.101. [DOI] [PubMed] [Google Scholar]
- 31.Keirstead HS, Nistor G, Bernal G, Totoiu M, Cloutier F, Sharp K, et al. Human embryonic stem cell-derived oligodendrocyte progenitor cell transplants remyelinate and restore locomotion after spinal cord injury. J Neurosci. 2005;25(19):4694–705. doi: 10.1523/JNEUROSCI.0311-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Okita K, Ichisaka T, Yamanaka S. Generation of germline-competent induced pluripotent stem cells. Nature. 2007;448(7151):313–7. doi: 10.1038/nature05934. [DOI] [PubMed] [Google Scholar]
- 33.Maherali N, Sridharan R, Xie W, Utikal J, Eminli S, Arnold K, et al. Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell. 2007;1(1):55–70. doi: 10.1016/j.stem.2007.05.014. [DOI] [PubMed] [Google Scholar]
- 34.Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, Hochedlinger K, et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature. 2007;448(7151):318–24. doi: 10.1038/nature05944. [DOI] [PubMed] [Google Scholar]
- 35.Lowry WE, Richter L, Yachechko R, Pyle AD, Tchieu J, Sridharan R, et al. Generation of human induced pluripotent stem cells from dermal fibroblasts. Proc Natl Acad Sci U S A. 2008;105(8):2883–8. doi: 10.1073/pnas.0711983105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Park IH, Zhao R, West JA, Yabuuchi A, Huo H, Ince TA, et al. Reprogramming of human somatic cells to pluripotency with defined factors. Nature. 2008;451(7175):141–6. doi: 10.1038/nature06534. [DOI] [PubMed] [Google Scholar]
- 37.Nakagawa M, Koyanagi M, Tanabe K, Takahashi K, Ichisaka T, Aoi T, et al. Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. Nat Biotechnol. 2008;26(1):101–6. doi: 10.1038/nbt1374. [DOI] [PubMed] [Google Scholar]
- 38.Huangfu D, Osafune K, Maehr R, Guo W, Eijkelenboom A, Chen S, et al. Induction of pluripotent stem cells from primary human fibroblasts with only Oct4 and Sox2. Nat Biotechnol. 2008;26(11):1269–75. doi: 10.1038/nbt.1502. [DOI] [PubMed] [Google Scholar]
- 39.Hou P, Li Y, Zhang X, Liu C, Guan J, Li H, et al. Pluripotent stem cells induced from mouse somatic cells by small-molecule compounds. Science. 2013;341(6146):651–4. doi: 10.1126/science.1239278. [DOI] [PubMed] [Google Scholar]
- 40.Zhao Y, Zhao T, Guan J, Zhang X, Fu Y, Ye J, et al. A XEN-like State Bridges Somatic Cells to Pluripotency during Chemical Reprogramming. Cell. 2015;163(7):1678–91. doi: 10.1016/j.cell.2015.11.017. [DOI] [PubMed] [Google Scholar]
- 41.Nakagawa M, Takizawa N, Narita M, Ichisaka T, Yamanaka S. Promotion of direct reprogramming by transformation-deficient Myc. Proc Natl Acad Sci U S A. 2010;107(32):14152–7. doi: 10.1073/pnas.1009374107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ieda M, Fu JD, Delgado-Olguin P, Vedantham V, Hayashi Y, Bruneau BG, et al. Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell. 2010;142(3):375–86. doi: 10.1016/j.cell.2010.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Schenke-Layland K, Rhodes KE, Angelis E, Butylkova Y, Heydarkhan-Hagvall S, Gekas C, et al. Reprogrammed mouse fibroblasts differentiate into cells of the cardiovascular and hematopoietic lineages. Stem Cells. 2008;26(6):1537–46. doi: 10.1634/stemcells.2008-0033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Narazaki G, Uosaki H, Teranishi M, Okita K, Kim B, Matsuoka S, et al. Directed and systematic differentiation of cardiovascular cells from mouse induced pluripotent stem cells. Circulation. 2008;118(5):498–506. doi: 10.1161/CIRCULATIONAHA.108.769562. [DOI] [PubMed] [Google Scholar]
- 45.Zwi L, Caspi O, Arbel G, Huber I, Gepstein A, Park IH, et al. Cardiomyocyte differentiation of human induced pluripotent stem cells. Circulation. 2009;120(15):1513–23. doi: 10.1161/CIRCULATIONAHA.109.868885. [DOI] [PubMed] [Google Scholar]
- 46.Karagiannis P, Yamanaka S. The fate of cell reprogramming. Nat Methods. 2014;11(10):1006–8. doi: 10.1038/nmeth.3109. [DOI] [PubMed] [Google Scholar]
- 47.Chin MH, Mason MJ, Xie W, Volinia S, Singer M, Peterson C, et al. Induced pluripotent stem cells and embryonic stem cells are distinguished by gene expression signatures. Cell Stem Cell. 2009;5(1):111–23. doi: 10.1016/j.stem.2009.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Doi A, Park IH, Wen B, Murakami P, Aryee MJ, Irizarry R, et al. Differential methylation of tissue- and cancer-specific CpG island shores distinguishes human induced pluripotent stem cells, embryonic stem cells and fibroblasts. Nat Genet. 2009;41(12):1350–3. doi: 10.1038/ng.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Deng J, Shoemaker R, Xie B, Gore A, LeProust EM, Antosiewicz-Bourget J, et al. Targeted bisulfite sequencing reveals changes in DNA methylation associated with nuclear reprogramming. Nat Biotechnol. 2009;27(4):353–60. doi: 10.1038/nbt.1530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sadahiro T, Yamanaka S, Ieda M. Direct cardiac reprogramming: progress and challenges in basic biology and clinical applications. Circ Res. 2015;116(8):1378–91. doi: 10.1161/CIRCRESAHA.116.305374. [DOI] [PubMed] [Google Scholar]
- 51.Behrens A, van Deursen JM, Rudolph KL, Schumacher B. Impact of genomic damage and ageing on stem cell function. Nat Cell Biol. 2014;16(3):201–7. doi: 10.1038/ncb2928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jackson SP, Bartek J. The DNA-damage response in human biology and disease. Nature. 2009;461(7267):1071–8. doi: 10.1038/nature08467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Pandita TK, Richardson C. Chromatin remodeling finds its place in the DNA double-strand break response. Nucleic Acids Res. 2009;37(5):1363–77. doi: 10.1093/nar/gkn1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Pandita TK. ATM function and telomere stability. Oncogene. 2002;21(4):611–8. doi: 10.1038/sj.onc.1205060. [DOI] [PubMed] [Google Scholar]
- 55.Pandita TK. A multifaceted role for ATM in genome maintenance. Expert Rev Mol Med. 2003;5(16):1–21. doi: 10.1017/S1462399403006318. [DOI] [PubMed] [Google Scholar]
- 56.Hoeijmakers JH. DNA damage, aging, and cancer. N Engl J Med. 2009;361(15):1475–85. doi: 10.1056/NEJMra0804615. [DOI] [PubMed] [Google Scholar]
- 57.Ciccia A, Elledge SJ. The DNA damage response: making it safe to play with knives. Mol Cell. 2010;40(2):179–204. doi: 10.1016/j.molcel.2010.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Harper JW, Elledge SJ. The DNA damage response: ten years after. Mol Cell. 2007;28(5):739–45. doi: 10.1016/j.molcel.2007.11.015. [DOI] [PubMed] [Google Scholar]
- 59.Bartkova J, Horejsi Z, Koed K, Kramer A, Tort F, Zieger K, et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature. 2005;434(7035):864–70. doi: 10.1038/nature03482. [DOI] [PubMed] [Google Scholar]
- 60.Liang Y, Lin SY, Brunicardi FC, Goss J, Li K. DNA damage response pathways in tumor suppression and cancer treatment. World J Surg. 2009;33(4):661–6. doi: 10.1007/s00268-008-9840-1. [DOI] [PubMed] [Google Scholar]
- 61.Rass U, Ahel I, West SC. Defective DNA repair and neurodegenerative disease. Cell. 2007;130(6):991–1004. doi: 10.1016/j.cell.2007.08.043. [DOI] [PubMed] [Google Scholar]
- 62.Scott SP, Pandita TK. The cellular control of DNA double-strand breaks. J Cell Biochem. 2006;99(6):1463–75. doi: 10.1002/jcb.21067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kumar R, Horikoshi N, Singh M, Gupta A, Misra HS, Albuquerque K, et al. Chromatin modifications and the DNA damage response to ionizing radiation. Front Oncol. 2012;2:214. doi: 10.3389/fonc.2012.00214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Valerie K, Povirk LF. Regulation and mechanisms of mammalian double-strand break repair. Oncogene. 2003;22(37):5792–812. doi: 10.1038/sj.onc.1206679. [DOI] [PubMed] [Google Scholar]
- 65.Richardson C, Horikoshi N, Pandita TK. The role of the DNA double-strand break response network in meiosis. DNA Repair (Amst) 2004;3(8–9):1149–64. doi: 10.1016/j.dnarep.2004.05.007. [DOI] [PubMed] [Google Scholar]
- 66.Thompson LH, Schild D. Recombinational DNA repair and human disease. Mutat Res. 2002;509(1–2):49–78. doi: 10.1016/s0027-5107(02)00224-5. [DOI] [PubMed] [Google Scholar]
- 67.Wong KK, Maser RS, Bachoo RM, Menon J, Carrasco DR, Gu Y, et al. Telomere dysfunction and Atm deficiency compromises organ homeostasis and accelerates ageing. Nature. 2003;421(6923):643–8. doi: 10.1038/nature01385. [DOI] [PubMed] [Google Scholar]
- 68.Murga M, Bunting S, Montana MF, Soria R, Mulero F, Canamero M, et al. A mouse model of ATR-Seckel shows embryonic replicative stress and accelerated aging. Nat Genet. 2009;41(8):891–8. doi: 10.1038/ng.420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Pandita TK, Dhar S. Influence of ATM function on interactions between telomeres and nuclear matrix. Radiat Res. 2000;154(2):133–9. doi: 10.1667/0033-7587(2000)154[0133:ioafoi]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 70.Pandita TK, Hall EJ, Hei TK, Piatyszek MA, Wright WE, Piao CQ, et al. Chromosome end-to-end associations and telomerase activity during cancer progression in human cells after treatment with alpha-particles simulating radon progeny. Oncogene. 1996;13(7):1423–30. [PubMed] [Google Scholar]
- 71.Pandita TK, Pathak S, Geard CR. Chromosome end associations, telomeres and telomerase activity in ataxia telangiectasia cells. Cytogenet Cell Genet. 1995;71(1):86–93. doi: 10.1159/000134069. [DOI] [PubMed] [Google Scholar]
- 72.Shiloh Y, Ziv Y. The ATM protein kinase: regulating the cellular response to genotoxic stress, and more. Nature reviews Molecular cell biology. 2013;14(4):197–210. [PubMed] [Google Scholar]
- 73.Kastan MB. DNA damage responses: mechanisms and roles in human disease: 2007 G.H.A. Clowes Memorial Award Lecture. Mol Cancer Res. 2008;6(4):517–24. doi: 10.1158/1541-7786.MCR-08-0020. [DOI] [PubMed] [Google Scholar]
- 74.Kastan MB, Lim DS. The many substrates and functions of ATM. Nature reviews Molecular cell biology. 2000;1(3):179–86. doi: 10.1038/35043058. [DOI] [PubMed] [Google Scholar]
- 75.Pandita TK, Lieberman HB, Lim DS, Dhar S, Zheng W, Taya Y, et al. Ionizing radiation activates the ATM kinase throughout the cell cycle. Oncogene. 2000;19(11):1386–91. doi: 10.1038/sj.onc.1203444. [DOI] [PubMed] [Google Scholar]
- 76.Gupta A, Hunt CR, Hegde ML, Chakraborty S, Udayakumar D, Horikoshi N, et al. MOF Phosphorylation by ATM Regulates 53BP1-Mediated Double-Strand Break Repair Pathway Choice. Cell reports. 2014;8(1):177–89. doi: 10.1016/j.celrep.2014.05.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Horikoshi N, Kumar P, Sharma GG, Chen M, Hunt CR, Westover K, et al. Genome-wide distribution of histone H4 Lysine 16 acetylation sites and their relationship to gene expression. Genome Integr. 2013;4(1):3. doi: 10.1186/2041-9414-4-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Gupta A, Sharma GG, Young CS, Agarwal M, Smith ER, Paull TT, et al. Involvement of human MOF in ATM function. Mol Cell Biol. 2005;25(12):5292–305. doi: 10.1128/MCB.25.12.5292-5305.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Gupta A, Guerin-Peyrou TG, Sharma GG, Park C, Agarwal M, Ganju RK, et al. The mammalian ortholog of Drosophila MOF that acetylates histone H4 lysine 16 is essential for embryogenesis and oncogenesis. Mol Cell Biol. 2008;28(1):397–409. doi: 10.1128/MCB.01045-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Gupta A, Hunt CR, Pandita RK, Pae J, Komal K, Singh M, et al. T-cell-specific deletion of Mof blocks their differentiation and results in genomic instability in mice. Mutagenesis. 2013;28(3):263–70. doi: 10.1093/mutage/ges080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Kumar R, Hunt CR, Gupta A, Nannepaga S, Pandita RK, Shay JW, et al. Purkinje cell-specific males absent on the first (mMof) gene deletion results in an ataxia-telangiectasia-like neurological phenotype and backward walking in mice. Proc Natl Acad Sci U S A. 2011;108(9):3636–41. doi: 10.1073/pnas.1016524108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Sharma GG, So S, Gupta A, Kumar R, Cayrou C, Avvakumov N, et al. MOF and histone H4 acetylation at lysine 16 are critical for DNA damage response and double-strand break repair. Mol Cell Biol. 2010;30(14):3582–95. doi: 10.1128/MCB.01476-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Pandita TK, Westphal CH, Anger M, Sawant SG, Geard CR, Pandita RK, et al. Atm inactivation results in aberrant telomere clustering during meiotic prophase. Mol Cell Biol. 1999;19(7):5096–105. doi: 10.1128/mcb.19.7.5096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Scherthan H, Jerratsch M, Dhar S, Wang YA, Goff SP, Pandita TK. Meiotic telomere distribution and Sertoli cell nuclear architecture are altered in Atm- and Atm-p53-deficient mice. Mol Cell Biol. 2000;20(20):7773–83. doi: 10.1128/mcb.20.20.7773-7783.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Thomas T, Dixon MP, Kueh AJ, Voss AK. Mof (MYST1 or KAT8) is essential for progression of embryonic development past the blastocyst stage and required for normal chromatin architecture. Mol Cell Biol. 2008;28(16):5093–105. doi: 10.1128/MCB.02202-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Li X, Li L, Pandey R, Byun JS, Gardner K, Qin Z, et al. The histone acetyltransferase MOF is a key regulator of the embryonic stem cell core transcriptional network. Cell Stem Cell. 2012;11(2):163–78. doi: 10.1016/j.stem.2012.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Mu X, Yan S, Fu C, Wei A. The Histone Acetyltransferase MOF Promotes Induces Generation of Pluripotent Stem Cells. Cell Reprogram. 2015;17(4):259–67. doi: 10.1089/cell.2014.0102. [DOI] [PubMed] [Google Scholar]
- 88.Adams BR, Golding SE, Rao RR, Valerie K. Dynamic dependence on ATR and ATM for double-strand break repair in human embryonic stem cells and neural descendants. PloS one. 2010;5(4):e10001. doi: 10.1371/journal.pone.0010001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Serrano L, Liang L, Chang Y, Deng L, Maulion C, Nguyen S, et al. Homologous recombination conserves DNA sequence integrity throughout the cell cycle in embryonic stem cells. Stem Cells Dev. 2011;20(2):363–74. doi: 10.1089/scd.2010.0159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Mandal PK, Blanpain C, Rossi DJ. DNA damage response in adult stem cells: pathways and consequences. Nat Rev Mol Cell Biol. 2011;12(3):198–202. doi: 10.1038/nrm3060. [DOI] [PubMed] [Google Scholar]
- 91.Weiss CN, Ito K. DNA damage: a sensible mediator of the differentiation decision in hematopoietic stem cells and in leukemia. Int J Mol Sci. 2015;16(3):6183–201. doi: 10.3390/ijms16036183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wyles SP, Brandt EB, Nelson TJ. Stem cells: the pursuit of genomic stability. Int J Mol Sci. 2014;15(11):20948–67. doi: 10.3390/ijms151120948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Maynard S, Swistowska AM, Lee JW, Liu Y, Liu ST, Da Cruz AB, et al. Human embryonic stem cells have enhanced repair of multiple forms of DNA damage. Stem Cells. 2008;26(9):2266–74. doi: 10.1634/stemcells.2007-1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Lan ML, Acharya MM, Tran KK, Bahari-Kashani J, Patel NH, Strnadel J, et al. Characterizing the radioresponse of pluripotent and multipotent human stem cells. PloS one. 2012;7(12):e50048. doi: 10.1371/journal.pone.0050048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Tichy ED, Pillai R, Deng L, Liang L, Tischfield J, Schwemberger SJ, et al. Mouse embryonic stem cells, but not somatic cells, predominantly use homologous recombination to repair double-strand DNA breaks. Stem Cells Dev. 2010;19(11):1699–711. doi: 10.1089/scd.2010.0058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Adams BR, Hawkins AJ, Povirk LF, Valerie K. ATM-independent, high-fidelity nonhomologous end joining predominates in human embryonic stem cells. Aging (Albany NY) 2010;2(9):582–96. doi: 10.18632/aging.100197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Bogomazova AN, Lagarkova MA, Tskhovrebova LV, Shutova MV, Kiselev SL. Error-prone nonhomologous end joining repair operates in human pluripotent stem cells during late G2. Aging (Albany NY) 2011;3(6):584–96. doi: 10.18632/aging.100336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Momcilovic O, Knobloch L, Fornsaglio J, Varum S, Easley C, Schatten G. DNA damage responses in human induced pluripotent stem cells and embryonic stem cells. PloS one. 2010;5(10):e13410. doi: 10.1371/journal.pone.0013410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Momcilovic O, Choi S, Varum S, Bakkenist C, Schatten G, Navara C. Ionizing radiation induces ataxia telangiectasia mutated-dependent checkpoint signaling and G(2) but not G(1) cell cycle arrest in pluripotent human embryonic stem cells. Stem Cells. 2009;27(8):1822–35. doi: 10.1002/stem.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Rocha CR, Lerner LK, Okamoto OK, Marchetto MC, Menck CF. The role of DNA repair in the pluripotency and differentiation of human stem cells. Mutat Res. 2013;752(1):25–35. doi: 10.1016/j.mrrev.2012.09.001. [DOI] [PubMed] [Google Scholar]
- 101.Liedtke S, Biebernick S, Radke TF, Stapelkamp D, Coenen C, Zaehres H, et al. DNA damage response in neonatal and adult stromal cells compared with induced pluripotent stem cells. Stem Cells Transl Med. 2015;4(6):576–89. doi: 10.5966/sctm.2014-0209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Zou Y, Zhang N, Ellerby LM, Davalos AR, Zeng X, Campisi J, et al. Responses of human embryonic stem cells and their differentiated progeny to ionizing radiation. Biochemical and biophysical research communications. 2012;426(1):100–5. doi: 10.1016/j.bbrc.2012.08.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wilson KD, Sun N, Huang M, Zhang WY, Lee AS, Li Z, et al. Effects of ionizing radiation on self-renewal and pluripotency of human embryonic stem cells. Cancer Res. 2010;70(13):5539–48. doi: 10.1158/0008-5472.CAN-09-4238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Chlon TM, Ruiz-Torres S, Maag L, Mayhew CN, Wikenheiser-Brokamp KA, Davies SM, et al. Overcoming Pluripotent Stem Cell Dependence on the Repair of Endogenous DNA Damage. Stem Cell Reports. 2016;6(1):44–54. doi: 10.1016/j.stemcr.2015.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Filion TM, Qiao M, Ghule PN, Mandeville M, van Wijnen AJ, Stein JL, et al. Survival responses of human embryonic stem cells to DNA damage. J Cell Physiol. 2009;220(3):586–92. doi: 10.1002/jcp.21735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Barta T, Vinarsky V, Holubcova Z, Dolezalova D, Verner J, Pospisilova S, et al. Human embryonic stem cells are capable of executing G1/S checkpoint activation. Stem Cells. 2010;28(7):1143–52. doi: 10.1002/stem.451. [DOI] [PubMed] [Google Scholar]
- 107.Nouspikel T. Genetic instability in human embryonic stem cells: prospects and caveats. Future Oncol. 2013;9(6):867–77. doi: 10.2217/fon.13.22. [DOI] [PubMed] [Google Scholar]
- 108.Luo LZ, Gopalakrishna-Pillai S, Nay SL, Park SW, Bates SE, Zeng X, et al. DNA repair in human pluripotent stem cells is distinct from that in non-pluripotent human cells. PloS one. 2012;7(3):e30541. doi: 10.1371/journal.pone.0030541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Liu JC, Guan X, Ryan JA, Rivera AG, Mock C, Agrawal V, et al. High mitochondrial priming sensitizes hESCs to DNA-damage-induced apoptosis. Cell Stem Cell. 2013;13(4):483–91. doi: 10.1016/j.stem.2013.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Nagaria P, Robert C, Rassool FV. DNA double-strand break response in stem cells: mechanisms to maintain genomic integrity. Biochim Biophys Acta. 2013;1830(2):2345–53. doi: 10.1016/j.bbagen.2012.09.001. [DOI] [PubMed] [Google Scholar]
- 111.Baker DJ, Dawlaty MM, Wijshake T, Jeganathan KB, Malureanu L, van Ree JH, et al. Increased expression of BubR1 protects against aneuploidy and cancer and extends healthy lifespan. Nat Cell Biol. 2013;15(1):96–102. doi: 10.1038/ncb2643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Baker DJ, Weaver RL, van Deursen JM. p21 both attenuates and drives senescence and aging in BubR1 progeroid mice. Cell reports. 2013;3(4):1164–74. doi: 10.1016/j.celrep.2013.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]