Abstract
We conducted a prospective study to assess utility of detection of antibodies to phospholipase A2receptor (PLA2R) in the serum of patients with membranous nephropathy. Seventy five patients with biopsy proven membranous nephropathy admitted between January 2011 and September 2014 were studied. Serum anti- PLA2R was tested by indirect immunofluorescence. The test was positive in 45 out of 60 patients with primary membranous nephropathy (PMN) and in none of the 15 patients with secondary membranous nephropathy, with a sensitivity of 75% and specificity of 100% for PMN. Anti PLA2R positivity also showed a significant correlation with quantum of proteinuria and negative correlation with serum albumin. This study has validated detection of serum anti PLA2R in PMN as a non invasive diagnostic tool in Indian patients.
Key words: Anti-phospholipase A2 receptor antibodies, indirect immunofluorescence, membranous nephropathy
Introduction
Membranous nephropathy (MN) is a leading cause of nephrotic syndrome in adults, with a potential for progression to end-stage renal disease in 40–50% of untreated patients in 10–15 years.[1,2] MN can be idiopathic or secondary to a host of causes, viz. infections, drugs, toxins, autoimmune diseases, and malignancy. Diagnostic work-up differs significantly between the two forms. For secondary MN, therapy is focused on underlying cause but primary MN is treated with immunosuppressant drugs.
Primary MN has been a diagnosis of exclusion until 2009, when Beck et al. demonstrated auto antibodies against M-type phospholipase A2 receptor (PLA2R, a membrane glycoprotein located on the podocytes) in 60–80% of such patients.[3,4] This landmark discovery supports the notion that primary MN is an organ limited autoimmune disease. There is paucity of Indian data on the prevalence of anti-PLA2R and its clinical utility in MN. We analyzed the prevalence of this antibody among primary MN and its correlation with disease severity.
Subjects and Methods
Biopsy proven MN patients who presented to our department during the period from October 2011 to September 2014 was included in the study. They were categorized into primary and secondary MN. Investigations were done to find out secondary causes of MN viz. HBsAg, HIV antibody, hepatitis C virus antibody, anti-nuclear antibody, stool for occult blood, mammography in women, prostate specific antigen in men, and upper gastrointestinal endoscopy in selected patients. Those patients without any identifiable secondary cause were categorized as primary MN and those patients who received steroids and immunosuppressants were excluded from the study. Serum anti PLA2R antibody test was done in all patients by indirect immunofluorescence test (IIFT Mosaic; EUROIMMUN AG, Lübeck, Germany). Institutional ethical committee approval was obtained for the study.
Clinical data
Patients were categorized into three groups according to their risk for progression to chronic kidney disease as per KDIGO guidelines. Low risk was defined as urine protein creatinine ratio (uPCR) <4 g/g and estimated glomerular filtration rate (eGFR) >60 ml/min/1.73 m2 (calculated using modified modification of diet in renal disease formula), moderate risk included uPCR 4–8 g/g and eGFR >60 ml/min/1.73 m2, and high-risk as uPCR >8 g/g or eGFR <60 ml/min/1.73 m2.
Demonstration of serum anti-phospholipase A2 receptor antibody
Anti-phospholipase A2 receptor antigen in the glomerulus can be demonstrated by immunohistochemistry and circulating anti-PLA2R antibodies are detected by serological tests. There are three different methods of detection of APLA2R antibody viz. Western blot, indirect immunofluorescence assay (IIF), and ELISA. Advantages of ELISA and Western blot techniques include ability to detect and quantify the circulating antibodies but they are expensive and time consuming. IIF is a rapid, inexpensive qualitative and semi quantitative test. We employed IIFT using EUROIMMUN AG (GERMANY) in the study. IIFT has been shown to be concordant with other methods.[3]
Indirect immunofluorescence employs a mosaic of two different biochips mounted on a slide. One biochip is coated with human embryonic kidney (HEK) 293 cells that express PLA2R-protein and the other HEK 293 without this antigen as substrates. Serum samples were incubated for 30 min on the slide and fluorescein isothiocyanate-conjugated anti human IgG antibody from goat was used to detect bound IgG antibody. The assay is considered positive, when specific cytoplasmic fluorescence of transfected cells occurs in 1 in 10 dilution of serum under fluorescence microscope. In IIF, antibody positivity was graded as mild (intensity 1+), moderate (intensity 2+) and severe (intensity 3+) in 1 in 10 dilution of serum [Figure 1].
Figure 1.
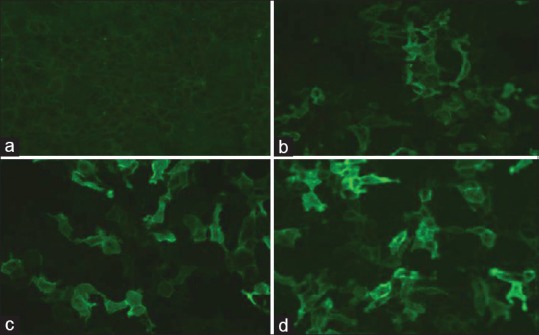
Various grades of staining of anti-phospholipase A2 receptor (anti-PLA2R) antibodies by Indirect Immunofluorescence. Detection of serum anti-PLA2R antibodies by immunofluorescence in 1:10 dilution of serum by florescence microscope graded as (a) negative, (b) mild (1 + intensity), (c) moderate (2+ intensity), (d) severe (3+ intensity)
Statistical analysis
Data were presented as the mean ± standard deviation. Statistical significance was considered when P < 0.05. Student's t-test and analysis of variance were used for parametric analysis. Categorical variables were described as frequencies or percentages, and the data were analyzed with Chi-square test. Correlation between two parameters was assessed using Pearson coefficient of correlation. All the statistical analyses were done using GraphPad software (© Graphpad software, Inc.).
Results
Seventy-five patients were included in the study. Mean duration of follow-up was 18 months (range 1–34 months). Secondary causes were noted in 15 patients (20%), and the remaining 60 (80%) patients were presumed to have primary MN. The causes of secondary MN in the study were systemic lupus nephritis (n = 11), rheumatoid arthritis (n = 1), malignancy (n = 1), hepatitis B virus (n = 1) and probable native drugs (n = 1). Among Primary MN, 38 were males (M:F ratio: 2:1). The mean age of the study group was 43.5 years (range16–67 years). Hypertension and diabetes at presentation were noted in 10 and 3 patients respectively. Mean uPCR was 5.5 (range 2.3–10.2 g protein/g creatinine). Hypoalbuminemia (serum albumin <3 g/dl) was seen in 20 (33.9%) and renal dysfunction (eGFR <60 ml/min) was found in 15 (25.4%) patients. Deep venous thrombosis was seen in 4 (6.7%) patients at presentation.
Prevalence of anti-phospholipase A2 receptor antibody and sensitivity
Serum PLA2R antibody was positive in 45 of 60 patients with primary MN but in none of the patients with secondary MN. Prevalence of serum anti-PLA2R for primary MN in our study was 75%.
Correlation between clinical features and anti-phospholipase A2 receptor antibody reactivity
Table 1 shows the demographic and clinical characteristics of primary MN patients at the time of presentation. Patients with antibody reactivity had severe proteinuria compared to those without antibody reactivity (uPCR 5.98 g/g vs. 4.3 g/g, P = 0.0006). The serum albumin level was significantly lower in patients with antibody reactivity (3.04 g/dl vs. 3.5 g/dl P = 0.0001). A significant proportion of patients with antibody positivity had nephrotic range of proteinuria than those who tested negative (79.2% vs. 20.8% P = 0.0001). There was a significant correlation between antibody positivity with quantum of proteinuria (r = 0.465, P = 0.02 Figure 2]. The proportion of high-risk patients increases with increasing intensity of staining [Figure 3].
Table 1.
Clinical and laboratory profile of patients with iMN according to anti-PLA2R Ab reactivity
Figure 2.
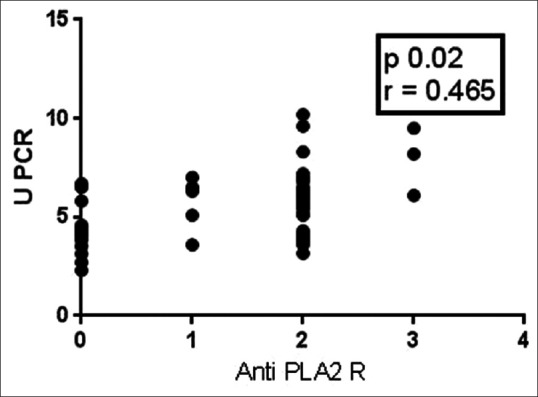
Correlation between proteinuria and anti-phospholipase A2 receptor intensity (Pearson correlation)
Figure 3.
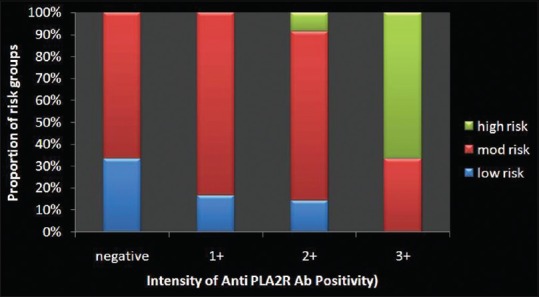
Proportion of high-risk patients increases with more intense staining of anti-phospholipase A2 receptor
Discussion
Membranous nephropathy is characterized by the formation of immune deposits over the glomerular basement membrane, which causes membrane like thickening. The immune deposits consist of IgG, predominantly IgG4 against unidentified antigens, and a search that had been fruitless for five decades. Beck et al. made a path-breaking discovery that M type PLA2R expressed on the podocyte is a major target antigen in primary MN.[3] PLA2R, a glycoprotein of the mannose receptor family is a type I transmembrane receptor for secretory phospholipase. The predominant circulating immunoglobulin against PLA2 is IgG4 which co-localizes within the glomerular immune deposits. In short, Beck et al. hypothesized that in human primary MN, subepithelial deposits form in situ by binding of circulating anti-PLA2R antibodies with PLA2R antigen expressed on the surface of the podocyte, which causes immune-mediated injury resulting in proteinuria. In our study, 75% of patients with primary MN had circulating auto antibodies against PLA2R in their serum using IIF. Prevalence of PLA2R antibodies in patients with primary MN varies between 52% to 82% in various studies[4,5,6,7,8,9] conducted in different ethnic populations. There is paucity of Indian data. The only available study by Ramachandran et al. was done in 36 patients with primary MN which showed a prevalence of serum anti-PLA2R (ELISA) and PLA2R antigen in glomerular tissue of 58.3% and 83.3% respectively.[10]
Our study found significant differences in baseline characteristics between anti PLA2R positive and negative cohorts at time of presentation similar to studies done elsewhere.[7,8,9] Nephrotic proteinuria was present in all those patients who were positive for anti PLA2R antibodies. Proteinuria and hypoalbuminemia were severe in those patients having antibody positivity when compared to those patients showing negative for the same, but there was no significant difference in age, sex and renal function between the groups. This finding was consistent with other studies.[7,8,9] Our study also validates the observation that anti PLA2R reactivity correlates with disease severity. Many studies have demonstrated the correlation of anti PLA2R levels and proteinuria using western blot and ELISA techniques. We found weak (r = 0.46) but significant correlation (P = 0.02) with IIFT method between anti PLA2R levels and quantum of proteinuria. These findings were in concordance with studies conducted in different ethnic population using ELISA and Western blot techniques.[11,12] Anti PLA2R antibodies were not detected in about 25% of our patients with primary MN. During the follow-up period, none developed any clinically identifiable secondary cause for MN. In our study, antibody test was done at the time of presentation itself when the disease was very much active. One reason attributed to less prevalence of antibodies was delay in serum sampling after renal biopsy. The mean time interval between kidney biopsy and performance of the test was only 2 weeks in contrast to other studies where the antibody test was done after significant delay[7,13] (mean time interval: 6–8 weeks). Hence, there could be other unidentified glomerular antigens against which antibodies are formed in primary MN. Recently in yet another breakthrough, circulating auto antibodies against THSD7A (thrombospondin type 1 domain containing 7A) was identified in primary MN patients who were negative for anti PLA2R antibodies.[14]
In a study by Qin et al.,[15] serum anti PLA2R antibodies was found to be positive in 6 of 46 patients with apparent secondary causes viz SLE, hepatitis B infection and malignancy. But in our study none of the 15 patients with secondary MN showed positivity for antibodies. Also, none of the 45 patients who tested positivity developed any features suggestive of secondary cause for MN during the follow-up period.
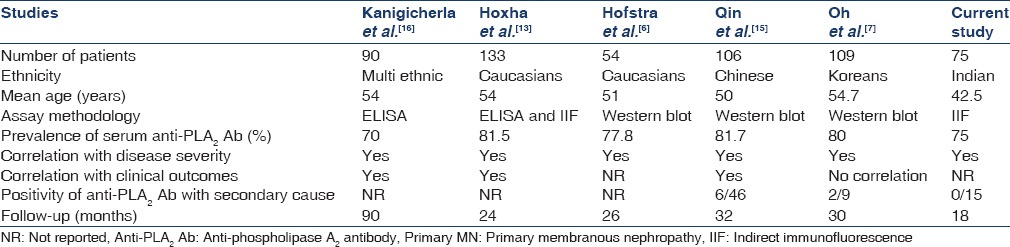
Several studies have already demonstrated close association of serum anti-PLA2R antibodies with proteinuria, and hence it can be used as a marker of disease activity [Table 2]. A study by Hoxha et al.[13] confirmed the presence of higher PLA2R antibody level as an independent risk factor for nonremission of proteinuria and demonstrated that decrease in PLA2R antibody level is associated with decrease in proteinuria. The disappearance of antibodies has been demonstrated in patients who undergo spontaneous remission. Hence, this test also can serve as a marker of response to therapy as well.
Table 2.
Comparison of various studies in patients with PrimaryMN with current study
There are several limitations of our study. Though we categorized the positive patients according to the degree of intensity, we could not measure the antibody titer in serial dilution for logistic reasons. We did not do tissue staining of PLA2R antigen and its correlation with serum antibody levels. Also, we could not serially repeat the test to assess its correlation with clinical outcome. In conclusion, serum anti PLA2R is a reliable specific marker for the diagnosis of primary MN, and it correlates with disease activity also. Anti-PLA2R positivity negates the need for evaluation of secondary causes of MN. This study has validated utility of the test in Indian population.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
References
- 1.Jha V, Ganguli A, Saha TK, Kohli HS, Sud K, Gupta KL, et al. A randomized, controlled trial of steroids and cyclophosphamide in adults with nephrotic syndrome caused by idiopathic membranous nephropathy. J Am Soc Nephrol. 2007;18:1899–904. doi: 10.1681/ASN.2007020166. [DOI] [PubMed] [Google Scholar]
- 2.Torres A, Domínguez-Gil B, Carreño A, Hernández E, Morales E, Segura J, et al. Conservative versus immunosuppressive treatment of patients with idiopathic membranous nephropathy. Kidney Int. 2002;61:219–27. doi: 10.1046/j.1523-1755.2002.00124.x. [DOI] [PubMed] [Google Scholar]
- 3.Beck LH, Jr, Bonegio RG, Lambeau G, Beck DM, Powell DW, Cummins TD, et al. M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. N Engl J Med. 2009;361:11–21. doi: 10.1056/NEJMoa0810457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ronco P, Debiec H. Antigen identification in membranous nephropathy moves toward targeted monitoring and new therapy. J Am Soc Nephrol. 2010;21:564–9. doi: 10.1681/ASN.2009121220. [DOI] [PubMed] [Google Scholar]
- 5.Hoxha E, Harendza S, Zahner G, Panzer U, Steinmetz O, Fechner K, et al. An immunofluorescence test for phospholipase-A2-receptor antibodies and its clinical usefulness in patients with membranous glomerulonephritis. Nephrol Dial Transplant. 2011;26:2526–32. doi: 10.1093/ndt/gfr247. [DOI] [PubMed] [Google Scholar]
- 6.Hofstra JM, Beck LH, Jr, Beck DM, Wetzels JF, Salant DJ. Anti-phospholipase A2 receptor antibodies correlate with clinical status in idiopathic membranous nephropathy. Clin J Am Soc Nephrol. 2011;6:1286–91. doi: 10.2215/CJN.07210810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Oh YJ, Yang SH, Kim DK, Kang SW, Kim YS. Autoantibodies against phospholipase A2 receptor in Korean patients with membranous nephropathy. PLoS One. 2013;8:e62151. doi: 10.1371/journal.pone.0062151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Akiyama S, Akiyama M, Imai E, Ozaki T, Matsuo S, Maruyama S. Prevalence of anti-phospholipase A2 receptor antibodies in Japanese patients with membranous nephropathy. Clin Exp Nephrol. 2014 doi: 10.1007/s10157-014-1054-2. DOI 10.1007/s10157-014-1054-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Segarra-Medrano A, Jatem-Escalante E, Quiles-Pérez MT, Salcedo MT, Arbós-Via MA, Ostos H, et al. Prevalence, diagnostic value and clinical characteristics associated with the presence of circulating levels and renal deposits of antibodies against the M-type phospholipase A2 receptor in idiopathic membranous nephropathy. Nefrologia. 2014;34:353–9. doi: 10.3265/Nefrologia.pre2013.Dec.12291. [DOI] [PubMed] [Google Scholar]
- 10.Ramachandran R, Sharma V, Verma A, Nada R, Jha V, Gupta KL. PLA2R in Glomerular deposit and anti PLA2R antibodies in Indian patients with active IMN. Nephrology. 2014;19(Suppl 2):23–76. [APSN abstract] [Google Scholar]
- 11.Hofstra JM, Wetzels JF. Anti-PLA2R antibodies in membranous nephropathy: Ready for routine clinical practice? Neth J Med. 2012;70:109–13. [PubMed] [Google Scholar]
- 12.Hofstra JM, Debiec H, Short CD, Pellé T, Kleta R, Mathieson PW, et al. Antiphospholipase A2 receptor antibody titer and subclass in idiopathic membranous nephropathy. J Am Soc Nephrol. 2012;23:1735–43. doi: 10.1681/ASN.2012030242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hoxha E, Thiele I, Zahner G, Panzer U, Harendza S, Stahl RA. Phospholipase A2 receptor autoantibodies and clinical outcome in patients with primary membranous nephropathy. J Am Soc Nephrol. 2014;25:1357–66. doi: 10.1681/ASN.2013040430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tomas NM, Beck LH, Jr, Meyer-Schwesinger C, Seitz-Polski B, Ma H, Zahner G, et al. Thrombospondin type-1 domain-containing 7A in idiopathic membranous nephropathy. N Engl J Med. 2014;371:2277–87. doi: 10.1056/NEJMoa1409354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Qin W, Beck LH, Jr, Zeng C, Chen Z, Li S, Zuo K, et al. Anti-phospholipase A2 receptor antibody in membranous nephropathy. J Am Soc Nephrol. 2011;22:1137–43. doi: 10.1681/ASN.2010090967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kanigicherla D, Gummadova J, McKenzie EA, Roberts SA, Harris S, Nikam M, et al. Anti-PLA2R antibodies measured by ELISA predict long-term outcome in a prevalent population of patients with idiopathic membranous nephropathy. Kidney Int. 2013;83:940–8. doi: 10.1038/ki.2012.486. [DOI] [PubMed] [Google Scholar]


