Abstract
Purpose
Dry eye syndrome is commonly thought of as an inflammatory disease, and we have previously presented data showing the effectiveness of topical TNF-α blocker agents for the treatment of this condition. The purpose of this study was to investigate the effectiveness of the TNF-α blocking agent HL036337 compared to cyclosporine A for the treatment of dry eye induced inflammation in order to establish whether HL036337 represents a more effective method for suppressing inflammation. The efficacy of HL036337 and cyclosporine A was determined using an experimental murine dry eye model.
Methods
The TNF-α blocker HL036337 is a modified form of TNF receptor I. Using dry eye induced C57BL/6 mice (n = 45), corneal erosion was measured at day 4 and 7 after topical treatment with cyclosporine A or HL036337. To determine the effective treatment dose, 0.25, 0.5, 1, 2.5, and 5 mg/mL of HL036337 were topically administered twice per day to dry eye induced murine corneas for 1 week.
Results
The optimal concentration of the TNF-α blocker HL036337 for treatment of dry eye induced corneal erosion was determined to be 1 mg/mL. Dry eye induced corneal erosion was improved after 1 week with topically applied cyclosporine A and HL036337 at 1 mg/mL.
Conclusions
HL036337 administered topically at 1 mg/mL effectively improved corneal erosion induced by dry eye. This finding may also suggest that inhibition of TNF-α can improve dry eye syndrome.
Keywords: Corneal erosion, Dry eye, HL036337, Inflammation, Tumor necrosis factor-alpha
Dry eye (DE) syndrome is one of the most frequent diseases encountered by ophthalmologists, and has a prevalence of 30% to 50% in the entire population [1,2,3] and a significant impact on daily life [4,5,6]. The etiology of DE syndrome has not been clearly established despite significant research efforts; however, according to previous studies, DE is likely an immunological disorder [7]. Specifically, there are increased levels of inflammatory cytokines in induced DE, and these cytokines seem to play an important role in the inflammatory response of DE syndrome [7,8,9]. In addition, clinical research on both humans and mice has confirmed the increased expression of cytokines such as IL-1a, IL-1b, IL-6, IL-8, IL-12, and TNF-α on the ocular surface of DEs [10,11,12].
Among the known inflammatory cytokines, TNF-α is understood to play multiple roles in inducing pro-inflammatory reactions and reciprocal stimulation [13]. TNF-α is involved in cellular transport and activation, pathogen resistance, and regulation of immune/inflammatory responses [14]. Accordingly, TNF-α has been utilized to develop a number of different drugs (e.g., TNF-α antagonist infliximab, adalimumab, etanercept, etc.) for the treatment of diseases such as rheumatoid arthritis, Sjögren's syndrome, and ankylosing spondylitis [14,15]. However, TNF-α blockers are not currently utilized as therapeutic agents for the treatment of DE syndrome.
There have been numerous attempts to develop therapeutic agents against DE syndrome, but there remain many areas for improvement. Ji et al. [16] previously developed HL036337 by modifying the TNF-α receptor I, which we investigated as a new treatment for DE syndrome. The purpose of this study was to establish the maximal effective concentration of the TNF-α blocker agent HL036337 via in vivo analysis of DE-induced inflammation on the ocular surface tissue. In addition, we sought to determine whether HL036337 represents a more effective method to suppress inf lammation compared to cyclosporine A (Restasis; Allergan, Parsippany-Troy Hills, NJ, USA).
Materials and Methods
Animals and dry eye induction
The study research protocol was approved by the Institutional Animal Care and Use Committee of Yonsei University College of Medicine. Six-week-old female C57BL/6 mice (Charles River Laboratory, Wilmington, MA, USA) were used in accordance with the standards described in the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research. DE was induced by placing mice in a controlled-environment chamber [17]. Five mice were allocated to each group (control and DE-induced). To achieve maximum ocular surface dryness, mice in the controlled-environment chamber (relative humidity below 20%) were given subcutaneous injections of 0.15 mL scopolamine hydrobromide (10 mg/mL; Sigma-Aldrich Chemical, St. Louis, MO, USA) twice per day for the duration of the experiment. Each experiment was repeated three times, and we calculated average values of expression data from three biological replicates at each time point.
Treatment regimen and tissue preparations
Ten days after beginning the DE induction protocol, we confirmed the presence of grade 3 or higher corneal erosions, after which mice were divided into a normal group (n = 5), DE-induced group (n = 5), HL036337 0.25 mg/mL treatment group (n = 5), HL036337 0.5 mg/mL treatment group (n = 5), HL036337 1 mg/mL treatment group (n = 5), HL036337 2.5 mg/mL treatment group (n = 5), HL036337 5 mg/mL treatment group (n = 5), Restasis treatment group (n = 5), and vehicle (sodium acetate, pH 5.5) treatment group (n = 5). DE-induced mice were treated by topical administration into the eyeballs at a volume of 10 µL twice per day.
At the end of the study period, mice were euthanized in a CO2 chamber and corneas, lymph nodes (LNs), and lacrimal glands were collected. All tissues were stored at -70℃ for quantitative reverse transcription (RT)-polymerase chain reaction (PCR).
Corneal erosion scoring
Corneal erosion grading was performed before treatment and again on treatment days 4 and 7. Briefly, at the end of treatment, 0.5% fluorescein (Alcon, Hünenberg, Switzerland) was applied to the lower conjunctival sac of the mice and after 3 minutes corneal fluorescein staining was examined with biomicroscopy. Punctate staining was evaluated using the Oxford Scheme grading system, which assigns a grade of 0 to 4. Corneal grading higher than grade 3 by the Oxford Scheme grading system was regarded as proper induction of DE.
Real-time polymerase chain reaction
Corneas, LNs, and lacrimal glands were collected from each of the five mice per group and compared. RNA was isolated with the RNeasy Micro Kit (Qiagen, Hilden, Germany) and reverse transcribed using the Superscript III Kit (Invitrogen, Carlsbad, CA, USA). Real-time PCR was performed using TaqMan Universal Master Mix and the following preformulated primers: IFN-γ (Mm01168134_m1; Applied Biosystems, Foster City, CA, USA), IL-6 (Mm00446190_m1), IL-1b (Mm00434228_m1), TNF-α (Mm004443260_g1), GAPDH (Mm99999915_g1).
Statistics
Statistical analysis was performed using SPSS ver. 21.0 (IBM Co., Armonk, NY, USA), and all data were expressed as the mean ± standard deviation. Differences between groups were examined by Student's t-test, Kruskal-Wallis test, or ANOVA where appropriate. Values of p less than 0.05 were considered statistically significant.
Results
Induction of the dry eye
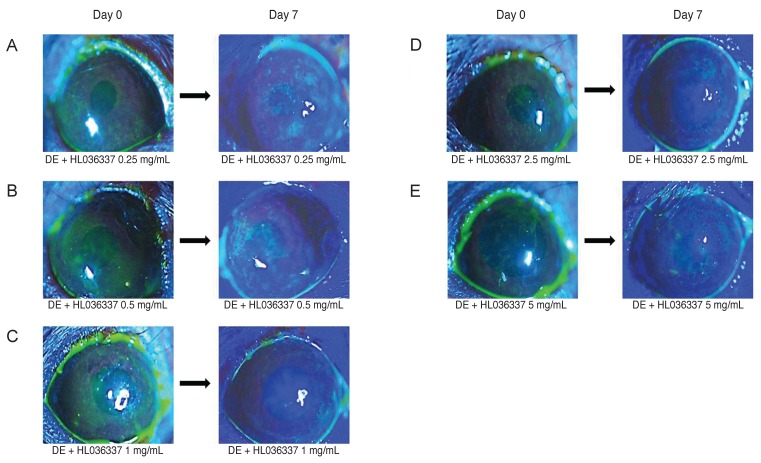
DE was induced in the test group by subcutaneous injection of scopolamine hydrobromide 0.15 mL twice per day and harboring them in controlled-environment chamber at a humidity less than 20%. After inducing DE, 1-mL 0.5% fluorescein was applied into mouse conjunctival sac, and corneal fluorescein staining was examined using slit lamp microscopy. We observed erosion above grade 3 in most of the test group mice, confirming the effective induction of DE using the study protocol (Fig. 1A-1E).
Fig. 1. Corneal erosion was measured followed by fluorescein cornea surface staining. Each group included six to eight corneas randomly selected from both eyes dry eye (DE) induction was performed for 10 days, after which corneal erosion in DE induced mice was measured and photographed. After 7 days of treatment with topical HL036337, corneal fluorescein staining was examined with a slit lamp biomicroscope and photos were taken. (A) Treatment with HL036337 0.25 mg/mL, (B) HL036337 0.5 mg/mL, (C) HL036337 1 mg/mL, (D) HL036337 2.5 mg/mL, and (E) HL036337 5 mg/mL. Representative photographs from each group are shown.
Treatment of HL036337 on dry eye-induced mice
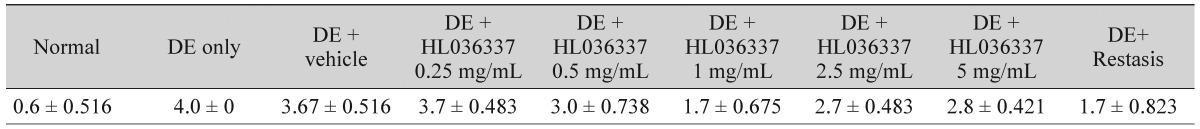
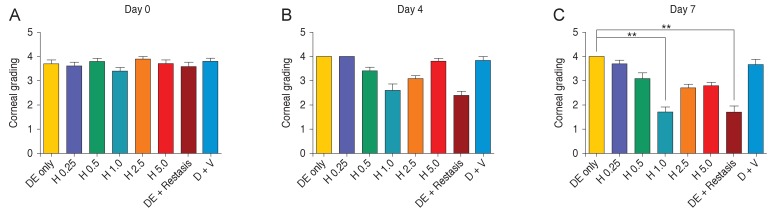
The TNF-α blocker agent HL036337 was administered topically on the corneas of DE-induced mice for 1 week and corneal changes were observed. Test groups were divided into HL036337 0.25, 0.5, 1, 2.5, and 5 mg/mL treatment groups. After 7 days of treatment, the corneal erosion grade was 3.7 ± 0.483 in the HL036337 0.25 mg/mL treatment group, 3.0 ± 0.738 in the HL036337 0.5 mg/mL treatment group, 1.7 ± 0.675 in the HL036337 1 mg/mL treatment group, 2.7 ± 0.483 in the HL036337 2.5 mg/mL treatment group, and 2.8 ± 0.421 grade in the HL036337 5 mg/mL treatment group. Decreased corneal erosion was most apparent in the HL036337 1 mg/mL treatment group (Table 1 and Fig. 2A-2C).
Table 1. Corneal erosion grading after 7 days of topical eye drops treatment.
Values are presented as mean ± standard deviation.
After 10 days of dry eye (DE) induction, topical eye drops were given to the mice and corneal fluorescein staining was examined with a slit lamp biomicroscope on treatment day 7. Groups were divided as follows: normal, DE induced, DE + vehicle, DE + HL036337 0.25 mg/mL, DE + HL036337 0.5 mg/mL, DE + HL036337 1 mg/mL, DE + HL036337 2.5 mg/mL, DE + HL036337 5 mg/mL, and DE + Restasis. Punctate staining was evaluated in a masked fashion using the Oxford Scheme grading system, where a grade of 0 to 4 was given for each cornea. Each experiment was repeated three times (p < 0.05).
Fig. 2. After induction of dry eye (DE) for 10 days, topical eye drops were given to the mice groups and corneal fluorescein staining was examined with a slit lamp biomicroscope at day 0 , 4, and 7. The mice were then divided into groups as follows: DE induced, DE + HL036337 0.25 mg/mL (H 0.25), DE + HL036337 0.5 mg/mL (H 0.5), DE + HL036337 1 mg/mL (H 1.0), DE + HL036337 2.5 mg/mL (H 2.5), DE + HL036337 5 mg/mL (H 5.0), DE + Restasis, and DE + vehicle (D + V). Punctate staining was evaluated in a masked fashion using the Oxford Scheme grading system, where a grade of 0 to 4 was given for each cornea. Each experiment was repeated three times. Student's t-test, **p < 0.05. (A) Corneal erosion grading before topical eye drops treatment. (B) Corneal erosion grading after 4 days of topical eye drops treatment. (C) Corneal erosion grading after 7 days of topical eye drops treatment.
Treatment of HL036337, vehicle and Restasis on dry eye-induced mice
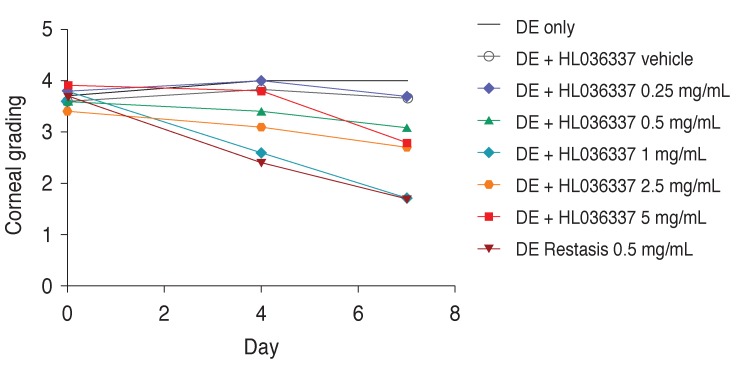
Three groups of DE-induced mice were treated topically with HL036337 1 mg/mL, vehicle, or Restasis and the effect of the treatments on corneal erosion were compared. The vehicle treatment group exhibited no significant difference compared to a mock-treated control group and exhibited no improvement in corneal erosion. On the contrary, treatment with either HL036337 1 mg/mL or Restasis induced improvement in corneal erosion on the fourth and seventh day compared to the first day. Specifically, corneal erosion grades were below grade 2 in both treatment groups on day 7 (Table 1 and Fig. 3).
Fig. 3. Determination of clinical data. Corneal erosion treatment was evaluated for a week with topical application and compared to that of Restasis. Punctate staining was evaluated in a masked fashion using the Oxford Scheme grading system, where a grade of 0 to 4 was given for each cornea. Each experiment was repeated three times. DE = dry eye. p < 0.05.
Changes in cytokine levels in cornea and lymph node after HL036337 treatment
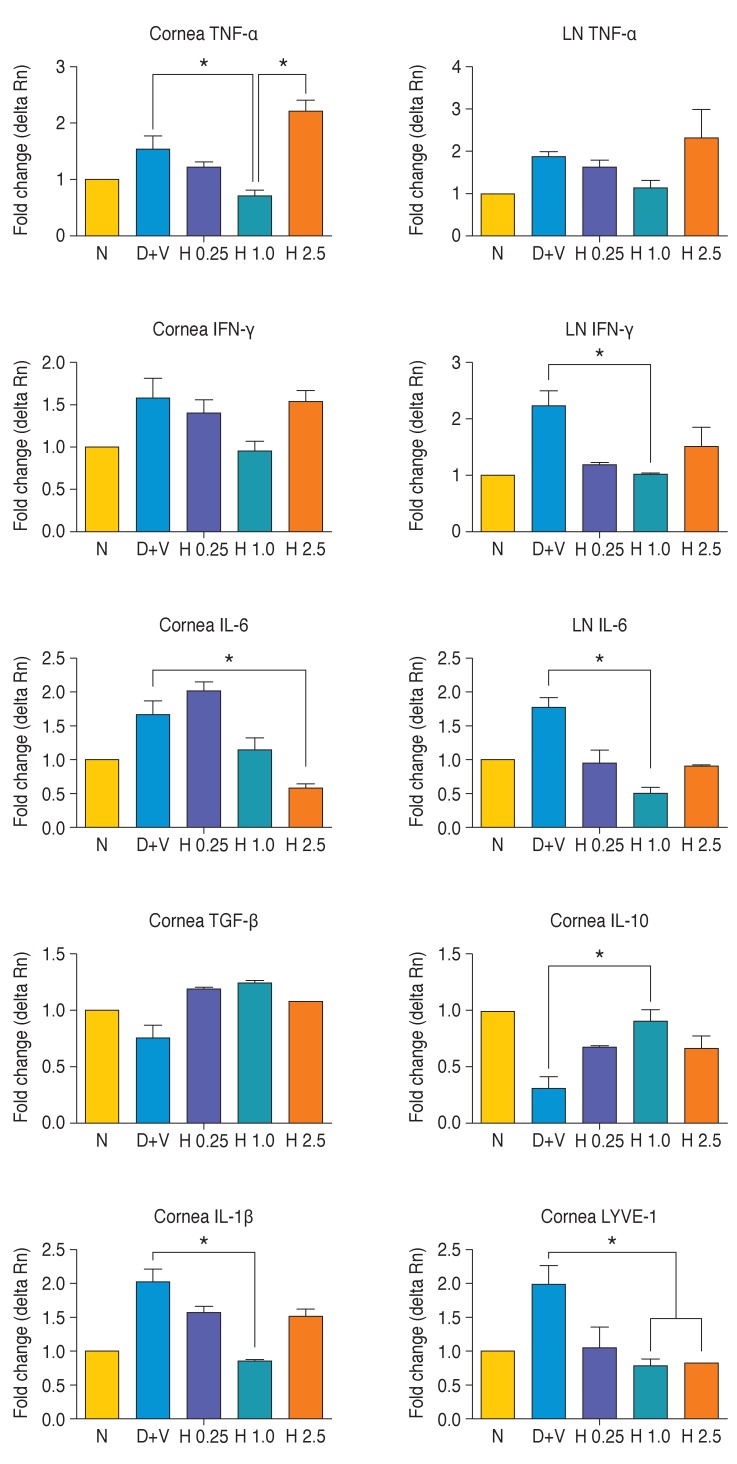
Following DE induction for 10 days, topical eye drops were given to the mice groups and inflammatory cytokines in corneas and LNs were determined by quantitative RT-PCR on day 7. Among the cytokines evaluated, expression of TNF-α, IL-6, IFN-γ, and IL-1b were decreased on the ocular surface and LNs of HL036337 treated mice in a dose dependent manner compared to the DE-induced group (p < 0.05) (Fig. 4). However, an increase in TNF-α, IFN-γ, and IL-1b expression was noted in the tissues of mice treated with 2.5 mg/mL HL036337. In addition, LYVE-1, a marker suggestive of lymphangiogenesis, was significantly decreased in corneas treated with HL036337 compared to the DE-induced group (p < 0.05) (Fig. 4). Conversely, expression of the immunoregulatory cytokines TGF-β and IL-10 were increased in corneas treated with HL036337 (p < 0.05) (Fig. 4).
Fig. 4. After 10 days of dry eye (DE) induction, topical eye drops were given to the mice groups and inflammatory cytokines in corneas and lymph nodes (LNs) were determined by quantitative reverse transcription polymerase chain reaction at day 7. Groups were divided as follows: normal (N), DE + vehicle (D + V), DE + HL036337 0.25 mg/mL (H 0.25), DE + HL036337 1 mg/mL (H 1.0), and DE + HL036337 2.5 mg/mL (H 2.5). Six corneas and six to eight cervical LNs from five mice were included in each group. Each experiment was repeated three times. *p < 0.05.
Discussion
Previously, Ji et al. [16] reported the development of HL036337 by modifying the TNF-α receptor I. Specifically, HL036337 is a synthesized peptide that resembles the TNF-α binding region of the TNF-α receptor I that was selected from various candidate molecules generated by site-directed mutagenesis and use of substituted amino acid sequences of human TNFRSF1A, P19438 [16].
Although there are several TNF-α blockers that can bind TNF-α, none have been able to induce significant improvement in DE [18,19,20]. Furthermore, in some cases, etanercept not only bound to and neutralized TNF-α, but also stabilized TNF-α and extended its half-life [18,19,20]. This might be because temporary binding at a relatively low affinity is unable to sufficiently neutralize TNF-α activity. Importantly, the structural homology of HL036337 allows it to bind free TNF-α in ocular surface tissues, thereby neutralizing its activity [16]. Indeed, HL036337 binds tightly to TNF-α, and exhibits improved tissue penetration and higher tissue concentration in cornea, suggesting it may have improved effectiveness on the ocular surface [16].
In the present study, corneal erosion was reduced following administration of HL036337 to mice with induced DE. This study was also significant in that it established the optimal concentration of HL036337 for the treatment of DE. Specifically, we determined using a mouse in vivo study that the TNF-α blocker HL036337 at a concentration of 1 mg/mL is the most effective agent to reduce corneal erosion compared to other agents at different concentrations. In addition, when compared using corneal erosion staging, HL036337 1 mg/mL exhibited similar counter-erosive effects as Restasis, which is currently available on the market [21].
HL036337 displayed a linear dosage-effect relation at concentrations up to 1 mg/mL, but not at concentrations above 1 mg/mL. As HL036337 is a binding peptide for TNF-α, it likely follows the dose-effect pattern of similar competitive inhibitors, which typically exhibits an effect reaching a saturation point. However, in our study, the effect of HL036337 was actually decreased at concentrations higher than 1 mg/mL rather than reaching the saturation point and forming plateau (Figs. 2 and 4). There are several potential explanations for this observation. First, it is known that TNF-α inhibitors may result in paradoxical induction of autoimmunity, a situation in which the immune system produces antibodies against the inhibitor agent, leading to a loss of efficacy. Consistent with this possibility, several studies have reported that TNF inhibitors may result in production of neutralizing antibodies or auto-antibodies, which in some cases led to allergic reactions and loss of drug-efficacy [22,23]. Thus, in the present study, higher concentrations of HL036337 might have produced neutralizing or autoantibodies. Second, as HL036337 induced a decrease in the levels of free TNF-α, higher doses of HL036337 might have triggered homeostatic compensation of TNF-α production. Lastly, high dose HL036337 might have stimulated production of inflammatory cytokines. Further studies are needed to evaluate these possibilities.
Restasis, which consists of 0.05% cyclosporine A, is an immunosuppressive agent and recognized as an effective treatment in individuals with chronic DE patients [21]. When compared with measured corneal erosion stage level, 1 mg/mL HL036337 showed similar counter-erosive effects as Restasis. This result suggests that HL036337 could be used to regulate immune suppression reaction on the ocular surface.
The mechanism action of cyclosporine A differs from HL036337. Specifically, cyclosporine A forms a heterodimeric complex inside T cell and acts on several targets that play central roles in signaling pathways, including the calcineurin/NFAT pathway and JNK/p38 activation pathways [24]. As a result, cyclosporine A is able to inhibit T cells with high specificity. On the other hand, HL036337 binds directly to TNF-α outside of the cell. Based on the results of the present study, this mechanism may suggest that blockade of TNF-α is a new therapeutic target for the treatment of DE syndrome. Given the different mechanisms of action of cyclosporine A and HL036337, future studies should investigate the combination of these two agents for the treatment of DE syndrome.
In the present study, analysis of inflammatory cytokines in DE-induced mice using RT-PCR showed decreased levels of TNF-α, IL-6, IFN-γ, IL-1b on the ocular surface and LN in the HL036337 treatment group. This result suggested that HL036337 can reduce inflammatory cytokines. On the other hand, expression of the immunoregulatory cytokines TGF-β and IL-10 in corneas was increased by treatment with HL036337. In addition, we observed reduced expression of LYVE-1 in the corneas of mice treated with HL036337, suggesting that the agent may inhibit transportation of inflammatory mediators from LN. These findings will require further investigation into the mechanisms of immune suppression by HL036337.
In conclusion, the present study describes the reduction of corneal erosion by HL036337 treatment on DE-induced mice and reveals the best concentration of HL036337. We provide evidence of improved ocular surface erosion and inhibition of some inflammatory mediators by blocking TNF-α with HL036337. This result implies that proper inhibition of TNF-α can lead to improvement of DE disease. Therefore, HL036337 should be investigated further as a strategy to treat DE syndrome. Lastly, human trials will be needed to verify our in vivo results.
Acknowledgements
This work was supported by the Industrial Strategic Technology Development Program (project no. 10040233, molecular engineering and drug development of anti-TNF antibody fragment for local inflammatory disease) funded by the Ministry of Knowledge Economy (Korea).
Footnotes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
References
- 1.Uchino M, Schaumberg DA, Dogru M, et al. Prevalence of dry eye disease among Japanese visual display terminal users. Ophthalmology. 2008;115:1982–1988. doi: 10.1016/j.ophtha.2008.06.022. [DOI] [PubMed] [Google Scholar]
- 2.Guo B, Lu P, Chen X, et al. Prevalence of dry eye disease in Mongolians at high altitude in China: the Henan eye study. Ophthalmic Epidemiol. 2010;17:234–241. doi: 10.3109/09286586.2010.498659. [DOI] [PubMed] [Google Scholar]
- 3.Kim WJ, Kim HS, Kim MS. Current trends in the recognition and treatment of dry eye: a survey of ophthalmologists. J Korean Ophthalmol Soc. 2007;48:1614–1622. [Google Scholar]
- 4.Schiffman RM, Walt JG, Jacobsen G, et al. Utility assessment among patients with dry eye disease. Ophthalmology. 2003;110:1412–1419. doi: 10.1016/S0161-6420(03)00462-7. [DOI] [PubMed] [Google Scholar]
- 5.Buchholz P, Steeds CS, Stern LS, et al. Utility assessment to measure the impact of dry eye disease. Ocul Surf. 2006;4:155–161. doi: 10.1016/s1542-0124(12)70043-5. [DOI] [PubMed] [Google Scholar]
- 6.Song JS, Hyon JY, Lee D, et al. Current practice pattern for dry eye patients in South Korea: a multicenter study. Korean J Ophthalmol. 2014;28:115–121. doi: 10.3341/kjo.2014.28.2.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brignole F, Pisella PJ, Goldschild M, et al. Flow cytometric analysis of inflammatory markers in conjunctival epithelial cells of patients with dry eyes. Invest Ophthalmol Vis Sci. 2000;41:1356–1363. [PubMed] [Google Scholar]
- 8.Stern ME, Beuerman RW, Fox RI, et al. The pathology of dry eye: the interaction between the ocular surface and lacrimal glands. Cornea. 1998;17:584–589. doi: 10.1097/00003226-199811000-00002. [DOI] [PubMed] [Google Scholar]
- 9.Massingale ML, Li X, Vallabhajosyula M, et al. Analysis of inflammatory cytokines in the tears of dry eye patients. Cornea. 2009;28:1023–1027. doi: 10.1097/ICO.0b013e3181a16578. [DOI] [PubMed] [Google Scholar]
- 10.Boehm N, Riechardt AI, Wiegand M, et al. Proinflammatory cytokine profiling of tears from dry eye patients by means of antibody microarrays. Invest Ophthalmol Vis Sci. 2011;52:7725–7730. doi: 10.1167/iovs.11-7266. [DOI] [PubMed] [Google Scholar]
- 11.Lam H, Bleiden L, de Paiva CS, et al. Tear cytokine profiles in dysfunctional tear syndrome. Am J Ophthalmol. 2009;147:198–205.e1. doi: 10.1016/j.ajo.2008.08.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen Y, Zhang X, Yang L, et al. Decreased PPAR-γ expression in the conjunctiva and increased expression of TNF-α and IL-1β in the conjunctiva and tear fluid of d ry eye mice. Mol Med Rep. 2014;9:2015–2023. doi: 10.3892/mmr.2014.2041. [DOI] [PubMed] [Google Scholar]
- 13.Aggarwal BB. Signalling pathways of the TNF superfamily: a double-edged sword. Nat Rev Immunol. 2003;3:745–756. doi: 10.1038/nri1184. [DOI] [PubMed] [Google Scholar]
- 14.Cope AP, Londei M, Chu NR, et al. Chronic exposure to tumor necrosis factor (TNF) in vitro impairs the activation of T cells through the T cell receptor/CD3 complex; reversal in vivo by anti-TNF antibodies in patients with rheumatoid arthritis. J Clin Invest. 1994;94:749–760. doi: 10.1172/JCI117394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Braun J, Sieper J. Biological therapies in the spondyloarthritides: the current state. Rheumatology (Oxford) 2004;43:1072–1084. doi: 10.1093/rheumatology/keh205. [DOI] [PubMed] [Google Scholar]
- 16.Ji YW, Byun YJ, Choi W, et al. Neutralization of ocular surface TNF-α reduces ocular surface and lacrimal gland inflammation induced by in vivo dry eye. Invest Ophthalmol Vis Sci. 2013;54:7557–7566. doi: 10.1167/iovs.12-11515. [DOI] [PubMed] [Google Scholar]
- 17.Barabino S, Shen L, Chen L, et al. The controlled-environment chamber: a new mouse model of dry eye. Invest Ophthalmol Vis Sci. 2005;46:2766–2771. doi: 10.1167/iovs.04-1326. [DOI] [PubMed] [Google Scholar]
- 18.Moutsopoulos NM, Katsifis GE, Angelov N, et al. Lack of efficacy of etanercept in Sjogren syndrome correlates with failed suppression of tumour necrosis factor alpha and systemic immune activation. Ann Rheum Dis. 2008;67:1437–1443. doi: 10.1136/ard.2007.077891. [DOI] [PubMed] [Google Scholar]
- 19.Ramos-Casals M, Tzioufas AG, Stone JH, et al. Treatment of primary Sjogren syndrome: a systematic review. JAMA. 2010;304:452–460. doi: 10.1001/jama.2010.1014. [DOI] [PubMed] [Google Scholar]
- 20.Sankar V, Brennan MT, Kok MR, et al. Etanercept in Sjogren' syndrome: a twelve-week randomized, double-blind, placebo-controlled pilot clinical trial. Arthritis Rheum. 2004;50:2240–2245. doi: 10.1002/art.20299. [DOI] [PubMed] [Google Scholar]
- 21.Mah F, Milner M, Yiu S, et al. PERSIST: physician's Evaluation of Restasis(R) Satisfaction in Second Trial of topical cyclosporine ophthalmic emulsion 0.05% for dry eye: a retrospective review. Clin Ophthalmol. 2012;6:1971–1976. doi: 10.2147/OPTH.S30261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Brănişteanu DE, Voicu CM, Cretu A, et al. Adverse reactions of biological therapy for psoriasis. Rev Med Chir Soc Med Nat Iasi. 2015;119:38–44. [PubMed] [Google Scholar]
- 23.Ramos-Casals M, Roberto-Perez-Alvarez, Diaz-Lagares C, et al. Autoimmune diseases induced by biological agents: a double-edged sword? Autoimmun Rev. 2010;9:188–193. doi: 10.1016/j.autrev.2009.10.003. [DOI] [PubMed] [Google Scholar]
- 24.Matsuda S, Koyasu S. Mechanisms of action of cyclosporine. Immunopharmacology. 2000;47:119–125. doi: 10.1016/s0162-3109(00)00192-2. [DOI] [PubMed] [Google Scholar]