Abstract
Approximately 600 million people worldwide practise the carcinogenic habit of betel nut/quid chewing. Carcinogenic N-nitroso compounds have been identified in saliva or urine of betel chewers and the betel alkaloid arecoline in hair from habitual betel quid chewers. However, the pharmacokinetic parameters of these compounds have been little explored. Assessment of betel use by biomarkers is urgently needed to evaluate the effectiveness of cessation programmes aimed at reducing betel consumption to decrease the burden of cancers in regions of high betel consumption. In the search for biomarkers of betel consumption, we measured by liquid chromatography-mass spectrometry (LC-MS) the appearance and disappearance of betel alkaloids (characteristic for betel nuts), N-nitroso compounds, and chavibetol (characteristic for Piper Betle leaves) in saliva (n=4), hair (n=2), and urine (n=1) of occasional betel nut/quid chewers. The betel alkaloids arecoline, guvacoline, guvacine, and arecaidine were detected in saliva of all four participants and peaked within the first 2 h post-chewing before returning to baseline levels after 8 h. Salivary chavibetol was detected in participants consuming Piper Betle leaves in their quid and peaked ~1 h post-chewing. Urinary arecoline, guvacoline, and arecaidine excretion paralleled saliva almost exactly while chavibetol glucuronide excretion paralleled salivary chavibetol. No betel nut related compounds were detected in the tested hair samples using various extraction methods. From these preliminary results, we conclude that betel exposure can only be followed on a short-term basis (≤8 h post-chewing) using the applied biomarkers from urine and saliva while the feasibility of using hair has yet to be validated.
Keywords: betel, betel quid, alkaloids, saliva, urine
Introduction
Worldwide, approximately 600 million people chew betel in a wide variety of preparations[1,2] despite its known carcinogenicity.[3,4] Therefore, betel cessation programmes are urgently needed which require compliance measures for validation preferably based on reliable biomarkers to avoid self-reporting problems (e.g. recall bias, under-reporting).
Urine and saliva are widely used for monitoring compliance owing to their non-invasiveness and ease of collection. Hair analysis is advantageous because analyte exposure can be measured over defined and long periods of time that plasma and urine cannot.[5]
Studies with habitual betel quid (BQ) chewers have found carcinogenic N-nitroso compounds in saliva and urine[6] and arecoline and nicotine in hair.[7] We suggested using the main betel alkaloids and chavibetol as biomarkers for betel nut and quid consumption, respectively.[8] The pharmacokinetics of arecoline, one of the major betel alkaloids was followed in a few patients after i.v. administration,[9] and only three betel-related alkaloids were followed over time.[10] However, the absorption, distribution, metabolism, and excretion (ADME) parameters of betel alkaloids and chavibetol after betel chewing using various matrices have hitherto not been explored. Therefore, we aimed to determine the ADME parameters of betel biomarkers and betel-nut specific nitrosamines using urine, saliva, and hair of occasional betel chewers after one controlled chewing episode.
Experimental
Chemicals
Arecoline hydrobromide, guvacine hydrochloride, nicotine, cotinine, hydroxycotinine, N′-nitrosonornicotine (NNN), N′-nitrosoanatabine (NAT), NNK (4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone), NNK-d3, nicotine-d4, cotinine-d3 and eugenol and o-eugenol were purchased from Sigma-Aldrich (St Louis, MO, USA), Guvacoline hydrobromide, N-nitrosoguvacoline, Arecaidine Hydrobromide, Arecodine-d5, hydrobromide salt were purchased from Medical Isotopes (Pelham, NH). Proteinase K was purchased from Sigma-Aldrich (St Louis, MO, USA). All solvents were of LC-MS grade.
Standards
An 8-point calibration curve (0.1~5000 ng/mL) was prepared from a 10μg/mL stock in 0.1% formic acid in MeOH/H2O (1/1 v/v). 100 μL of each calibrator was mixed with 10 μL of the internal standard (IS) mixture (1 μg/mL of each arecoline-d5, nicotine-d4, cotinine-d3, and NNK-d3,) and 10 μL was injected for LC-MS analysis. The lower limit of detection was determined using a signal-to-noise ratio of 3.
Sample collection
Four male occasional betel chewers (51–64 y/o) chewed one serving of their typically consumed betel preparation for their usual amount of time (BQ consisting of 1/2 or 1 nut+Piper betle leaf+lime -participants 1, 3, and 4; 5–6 min) or ¼ nut without leaf (participant 2, 2.25 h). Saliva was collected at: t=0 (baseline) and 1, 2, 3, 4, 8, 24, and 48 h post-chewing and immediately centrifuged, aliquoted, then kept at −80°C until analysis. Participants 1 and 2 provided baseline scalp hair samples (200 mg of proximal 2 cm and 200 mg of distal hair) that were stored at room temperature until analysis. Participant 1 provided a 48-h urine sample during saliva collection that was centrifuged, aliquoted, then kept at −80°C until analysis.
All betel material was obtained in Hawai’i from one store at the beginning of the study and stored in a cooled environment until study completion. The Western IRB (Puyallup, WA, USA) approved this study protocol and all participants signed an informed consent before study commencement.
Saliva and urine preparation and analysis
One hundred μL thawed saliva or urine was mixed with 10 μL IS solution and 100 μL acetonitrile then vortexed to precipitate proteins. The mixture was diluted with 800 μL 0.1% formic acid in MeOH/H2O (v/v, 1/1) and centrifuged at 13 500g for 5 min. The supernatant was transferred to HPLC vials and 10 μL were injected for LC-MS analysis.
Hair hydrolysis with 12N NaOH
According to Marchei et al. [7] hair samples (part 1= root to 1 cm; part 2= 1~ 5 distal cm) were pulverized with a ball mill (SPEX 8000 mixer/mill, Metuchen, NJ, USA) for 40 min. To 50 mg of the resultant powder were added 10 μL IS (arecoline-d5, NNK-d3, nicotine-d4 and cotininine-d4: 1 μg/mL in MeOH) and 2 mL 12N NaOH followed by incubation at 40°C for 20 h and extraction with 5 mL CHCl3/i-PrOH (95/5 v/v), vortexing, and centrifugation. The organic layer was vortexed with 2.5 mL 0.5M HCl for 2 min. After centrifugation, the aqueous layer was neutralized with 1 mL 1N NaOH then made alkaline with 2 mL saturated NH4Cl buffer (pH 9.5, buffered with ammonia hydroxide). The resulting solution was extracted with 5 mL CHCl3/i-PrOH (95/5 v/v) then dried under nitrogen and reconstituted in 150 μL 0.1% formic acid in MeOH/H2O (1/1 v/v). 10 μL were injected for LC-MS analysis.
Hair hydrolysis with 1N NaOH
Modified from Tzatzarakis[11] 30 mg pulverized hair and 10 μL IS were incubated with 2 mL 1N NaOH at 60°C for 1 h then extracted twice with 3 mL dichloromethane. The organic layers were combined, acidified to pH 2 using 50 μL 1N HCl, dried under nitrogen, then reconstituted in 150 μL 0.1% formic acid in MeOH/H2O (1/1 v/v). 10 μL were injected for LC-MS analysis.
Extraction of pulverized hair samples with MeOH
Modified from Baumgartner[8] pulverized hair and IS were incubated with MeOH at room temperature for 2 h then centrifuged after which the MeOH supernatant extract was removed and the remaining hair powder was re-extracted with CHCl3/i-PrOH (95/5) at 50°C for 2 h. After incubation, the latter extract was centrifuged at 13 500g for 5 min and the supernatant was combined with the MeOH extract, dried under nitrogen and redissolved in 150 μL 0.1% formic acid in MeOH/H2O (1/1 v/v). 10 μL were injected for LC-MS analysis.
Stability study of Betel alkaloid standards
Guvacine, guvacoline, arecaidine, and arecoline standards and arecoline-d5 IS were tested in three solvents at 45°C: 1) MeOH; 2) 0.1N/ 1N HCl; and 3) 1N NaOH over 24 h. The changes were followed by LC-MS periodically.
Stability study of hair samples treated with 0.1N HCl
Pulverized hair samples (30 mg) and IS were treated with 2 mL 0.1N HCl at 45°C overnight. The samples remained undissolved after 20 h of incubation. After centrifugation, the supernatant was directly injected for LC-MS analysis.
Hair hydrolysis with protease K
Modified from Welch [9] pulverized hair and IS were treated with 1 mL proteinase K solution (3 mg proteinase K, 20 mg DTT, 100 μL triton X-100 in 10 mL Tris buffer (0.01M, pH 6.4) at 37°C for 3 days. This procedure only slightly dissolved hair. After centrifugation, the supernatant was mixed with 0.5 mL saturated NH4Cl (pH 9.5), and extracted twice with 2 mL CHCl3/i-PrOH (95/5). The organic phase was dried under nitrogen then reconstituted in 150 μL 0.1% formic acid in MeOH/H2O (1/1 v/v). 10 μL was injected for LC-MS analysis.
LC-MS analysis
LC-MS analysis was carried out on a model Accela ultra HPLC system coupled to a Q Exactive Orbitrap Mass Spectrometer and a CTC PAL autosampler (all from ThermoFisher, San José, CA, USA). Mixtures were injected onto a Kinetex C18 column (150 × 3 mm, 2.6 μm, Phenomenex, Torrance, CA, USA) with a Phenomenex UHPLC C18 pre-column (3.0 mm i.d.). Gradient elution was performed at a flow rate of 300 μL/minute using a linear gradient of 10 mM NH4OH in H2O to 10 mM NH4OH in MeOH. Mass analysis was performed under positive and negative ESI in full scan mode. Quantitation was performed with Xcalibur™ software by extracting the protonated analyte masses within 5 ppm of the respective monoisotopic masses except for chavibetol which was monitored in negative mode at its deprotonated monoisotopic mass. We found that the sensitivity using tSIM was similar to that using full scan mode and since we intended to search for potential unknown metabolites, the only possible way to achieve that was to use full scan mode.
Results and discussion
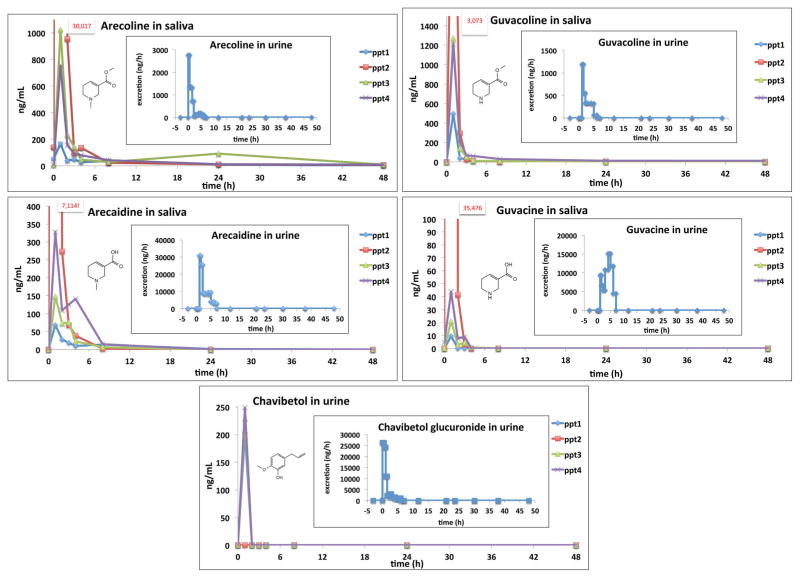
Phytochemicals that were found most diagnostically valuable for betel nut and BQ exposure have been identified by us and others previously and include arecoline and guvacoline, their hydrolysis products arecaidine and guvacine, and chavibetol and its glucuronide.[8,12] In the current study we detected the diagnostically valuable alkaloids arecoline, guvacoline, guvacine, and arecaidine in the saliva of all participants well above the limit of detection (0.1 ng/mL for all betel alkaloids and 200 ng/mL for chavibetol) up to 8 h after chewing with peaks appearing within 2h post-chewing (Figure 1). Chavibetol was detected in saliva of participants 1, 3, and 4 and peaked 0.5–1 hour post-chewing before returning to baseline levels after 2 h.
Figure 1.
Pattern of detected areca alkaloids and chavibetol in saliva and urine of betel chewers. Participant 1 chewed 1 nut + Piper Betle leaves + lime for 5 min, participant 2 chewed ¼ nut alone for 2.25 h without a piper leaf or lime, participant 3 chewed ½ nut + Piper Betle leaves + lime for 6 min, and participant 4 chewed 1 nut + 1 Piper Betle leaves + lime for 6 min. Participant 1 also provided urine samples during and up to 48 h after chewing.
The appearance patterns of arecoline, guvacoline, arecaidine, and guvacine in urine paralleled those in saliva (Figure 1) while urinary excretion of chavibetol glucuronide and salivary chavibetol paralleled each other (Figure 1); in each case peaks occurred approximately 2 h post-chewing before returning to baseline levels after approximately 8 h.
On average we found the salivary arecoline/arecaidine ratio to be circa 4:1 but in urine it was the opposite (circa 1:10) indicating that arecoline is metabolized to arecaidine after ingestion, which is in agreement with previous findings.[10] Hu et al. also reported the time course of arecoline, arecaidine, and N-methylnipecotic acid (NMNA) in human urine with arecoline having the shortest elimination half-life (t1/2= 0.97 h) followed by its hydrolyzed product arecaidine (t1/2 =4.3 h), and its succeeding reduced (dihydro) compound NMNA (t1/2 = 7.9 h).[10] The increasing t1/2 with each succeeding metabolite indicates a metabolic pathway similar to that suggested in the mouse[13] in which the parent compound changes rapidly making the metabolic fate of parent alkaloids (e.g. arecoline) difficult to track for long-term determinations.
Our attempts to detect NMNA in our saliva and urine samples failed although this was not the case for the other free acids arecaidine and guvacine which eluted at different retention times from the NMNA standard. In contrast, Hu et al. reported urinary NMNA excretion of 1298 ng/mg creatinine.[10] The subjects in that study, however, consumed water extracts of areca nuts that were claimed to contain NMNA whereas our subject chewed the nut directly. We did not analyze our nuts for NMNA and therefore, the absence of NMNA in our nuts would be a possible explanation for the discrepant findings. Also, the urinary excretion of NMNA was reported to be only <25% that of arecaidine suggesting that NMNA falls easily below detection limits and, therefore, may not be a better marker of betel nut exposure than arecaidine or the other alkaloids that we measured in our study. Most importantly, many isobaric co-eluting biochemicals can prevent the accurate quantitation of NMNA, particularly proline betaine, a physiologic component that serves as an osmoprotectant for the kidney. Proline betaine is also present in many citrus foods and, consequently, elevated levels are found in humans after citrus fruit/juice consumption. Therefore, we conclude that NMNA is not a good marker for betel exposure determinations.
LC-MS analysis could not detect any of the above betel related compounds including NMNA in hair samples from participants who chewed betel nuts at least once daily for ½-2 h. The traditional hair analyses by Marchei[7] using a strong base (12N NaOH) for extraction completely destroyed all four above mentioned betel alkaloids. Treatment with a lower concentration (NaOH, 1N)[11] did not lead to the recovery of IS or betel alkaloids while using MeOH followed by CHCl3/i-PrOH for extraction [14] allowed the spiked IS to be recovered but none of the betel alkaloids were detected; in contrast, a hair sample from a tobacco consumer showed high levels of nicotine and cotinine.
The stability studies revealed that alkaloids treated: (1) in MeOH (condition #1) or 0.1N HCl (condition #2) remained stable during 24 h of incubation; (2) in 1N HCl at 45°C (condition #2) remained stable up to 1 hour incubation, however, longer incubation resulted in conversions to multiple products; (3) in basic conditions (condition #3:1N NaOH) betel alkaloid esters arecoline and guvacoline were not stable and were hydrolyzed to their carboxylic acids (arecaidine and guvacine, respectively) within 1 hour of incubation then decomposed to multiple unknowns and both acids disappeared after 24 h. Hair samples treated with 12N NaOH completely destroyed all spiked IS while treatment with 0.1N HCl did not; however, no betel alkaloids were found in hair using either method although one hair sample from a tobacco consumer showed high levels of nicotine and cotinine.
Digesting pulverized hair with protease K[9] only dissolved the hair samples partially and resulted in the supernatant turning a shade of brown but no betel alkaloids were found by LC-MS analysis.
The need to objectively confirm self-reports during cessation studies is especially vital for highly addictive substances such as betel. Since it is not always possible or desirable to obtain blood samples, the use of other biospecimens such as urine, saliva, and hair become attractive alternatives.
Our previous study using saliva from habitual betel consumers[8] indicated promising candidate compounds to identify betel nut and BQ consumers; thus, in this preliminary study we investigated the urinary and salivary appearance and disappearance patterns of these compounds. Results showed identical patterns of arecoline, guvacoline, arecaidine, and guvacine in urine and saliva, which indicates that saliva patterns reflect systemic exposure accurately and that the measured alkaloids in saliva are derived from secretion and not from the leftover extraction of betel material that occurred during betel chewing. Since the measured betel alkaloids disappeared very quickly from saliva and urine, similar to plasma arecoline (≥15 min) after infusion, [15] betel exposure can be followed only on a short-term basis (<8 h post chewing) using the four mentioned betel alkaloids (Figure 1). The carcinogenic N-nitroso alkaloids were not found in any of our specimens and are, therefore, not usable as biomarkers.
Not surprisingly, chavibetol was detected in the saliva of participants 1, 3, and 4, who consumed Piper betle leaves in their BQ. Our previous study[8] found chavibetol exclusively in the Piper betle leaf and not betel nut; thus the absence of chavibetol in the saliva of participant 2, who chewed exclusively betel nut strengthens our previous findings. Unfortunately, the quick disappearance of chavibetol (within ~2 h post chewing) indicates that it is not an ideal biomarker for BQ consumption.
We did not find any areca alkaloids in our hair samples although they were collected from subjects with high and frequent betel consumption. Since typical tobacco related compounds were detected in some of our hair samples, we feel confident that the appropriate methods were applied for the intended purpose. Marchei[7] found arecoline in hair of chronic male and female betel consumers by digesting samples in 12N NaOH, however, we were not successful applying this method. It is possible that their participants consumed more betel nut/BQ than ours, the betel nuts consumed were more concentrated in alkaloids, or the method of chewing affected the amount of juice extracted from the betel nut thereby releasing more arecoline during chewing as previously suggested.[7] Other factors such as betel nut variety, diet, and smoking/tobacco habits may also help explain the difference.
Conclusion
Results from this preliminary pilot study suggest that betel alkaloids arecoline, guvacoline, arecaidine, and guvacine and also chavibetol (if BQ is consumed), characteristic for betel chewing[8], can be followed only up to ~8 h post exposure using urine or saliva due to their quick disappearance. No betel related compounds were detectable with our methods in hair, implying that areca alkaloids or their metabolites may not get accumulated in this matrix possibly because of their extremely short half-lives in contrast to tobacco alkaloids. The number of analyzed samples from this study does not permit any definitive conclusions; thus, further studies are warranted to investigate the possibility and feasibility of hair as a matrix to find biomarkers for betel use.
Acknowledgments
The authors would like to thank Brunhild M. Halm for her support in designing, executing, and publishing results of this study and Yukiko Morimoto for her assistance with saliva collection.
Funding
National Cancer Institute awards U54CA143727 and P30 CA71789.
References
- 1.Paulino Y. Areca (Betel) Nut Chewing Practices in Micronesian Populations. Hawaii J Public Health. 2011;3:19. [PMC free article] [PubMed] [Google Scholar]
- 2.Jeng JH, Chang MC, Hahn LJ. Role of areca nut in betel quid-associated chemical carcinogenesis: current awareness and future perspectives. Oral Oncol. 2001;37:477. doi: 10.1016/s1368-8375(01)00003-3. [DOI] [PubMed] [Google Scholar]
- 3.IARC. IARC Monographs on the evaluation of carcinogenic risks to humans. World Health Organization; Lyon: 2007. [Google Scholar]
- 4.Franke AA, Lai JF, Kawamoto CT, Pokhrel P, Herzog TA. Areca (Betel) Nut Consumption: An Underappreciated Cause of Cancer. Hawaii J Med Public Health. 2014;73:400. [PMC free article] [PubMed] [Google Scholar]
- 5.Al-Delaimy WK. Hair as a biomarker for exposure to tobacco smoke. Tob Control. 2002;11:176. doi: 10.1136/tc.11.3.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nair J, Ohshima H, Friesen M, Croisy A, Bhide SV, Bartsch H. Tobacco-specific and betel nut-specific N-nitroso compounds: occurrence in saliva and urine of betel quid chewers and formation in vitro by nitrosation of betel quid. Carcinogenesis. 1985;6:295. doi: 10.1093/carcin/6.2.295. [DOI] [PubMed] [Google Scholar]
- 7.Marchei E, Durgbanshi A, Rossi S, Garcia-Algar O, Zuccaro P, Pichini S. Determination of arecoline (areca nut alkaloid) and nicotine in hair by high-performance liquid chromatography/electrospray quadrupole mass spectrometry. Rapid Commun Mass Spectrom. 2005;19:3416. doi: 10.1002/rcm.2183. [DOI] [PubMed] [Google Scholar]
- 8.Franke AA, Mendez AJ, Lai JF, Arat-Cabading C, Li X, Custer LJ. Composition of betel specific chemicals in saliva during betel chewing for the identification of biomarkers. Food Chem Toxicol. 2015;80:241. doi: 10.1016/j.fct.2015.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Welch MJ, Sniegoski LT, Allgood CC, Habram M. Hair analysis for drugs of abuse: evaluation of analytical methods, environmental issues, and development of reference materials. J Anal Toxicol. 1993;17:389. doi: 10.1093/jat/17.7.389. [DOI] [PubMed] [Google Scholar]
- 10.Hu CW, Chang YZ, Wang HW, Chao MR. High-throughput simultaneous analysis of five urinary metabolites of areca nut and tobacco alkaloids by isotope-dilution liquid chromatography-tandem mass spectrometry with on-line solid-phase extraction. Cancer Epidemiol Biomarkers Prev. 2010;19:2570. doi: 10.1158/1055-9965.EPI-10-0483. [DOI] [PubMed] [Google Scholar]
- 11.Tzatzarakis MN, Vardavas CI, Terzi I, Kavalakis M, Kokkinakis M, Liesivuori J, Tsatsakis AM. Hair nicotine/cotinine concentrations as a method of monitoring exposure to tobacco smoke among infants and adults. Hum Exp Toxicol. 2012;31:258. doi: 10.1177/0960327111422401. [DOI] [PubMed] [Google Scholar]
- 12.Arjungi KN. Areca nut: a review. Arzneimittelforschung. 1976;26:951. [PubMed] [Google Scholar]
- 13.Giri S, Idle JR, Chen C, Zabriskie TM, Krausz KW, Gonzalez FJ. A metabolomic approach to the metabolism of the areca nut alkaloids arecoline and arecaidine in the mouse. Chem Res Toxicol. 2006;19:818. doi: 10.1021/tx0600402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Baumgartner AM, Jones PF, Baumgartner WA, Black CT. Radioimmunoassay of hair for determining opiate-abuse histories. J Nucl Med. 1979;20:748. [PubMed] [Google Scholar]
- 15.Asthana S, Greig NH, Holloway HW, Raffaele KC, Berardi A, Schapiro MB, Rapoport SI, Soncrant TT. Clinical pharmacokinetics of arecoline in subjects with Alzheimer’s disease. Clin Pharmacol Ther. 1996;60:276. doi: 10.1016/S0009-9236(96)90054-5. [DOI] [PubMed] [Google Scholar]