Abstract
Following binding to receptors in the arcuate nucleus (ArcN), insulin increases sympathetic nerve activity (SNA) and baroreflex control of SNA via a pathway that includes the paraventricular nucleus of the hypothalamus (PVN). Previous studies in males indicate that the sympathoexcitatory response is mediated by α-melanocyte stimulating hormone (α-MSH), which binds to PVN melanocortin type 3/4 receptors (MC3/4R). The present study was conducted in α-chloralose-anesthetized female rats to test the hypothesis that suppression of inhibitory neuropeptide Y (NPY) inputs to the PVN is also involved. In support of this, blockade of PVN NPY Y1 receptors with BIBO 3304 (NPY1x), ArcN insulin nanoinjections, and PVN NPY1x followed by ArcN insulin each increased lumbar SNA (LSNA) and its baroreflex regulation similarly. Moreover, prior PVN injections of NPY blocked the sympathoexcitatory effects of ArcN insulin. Finally, PVN nanoinjections of the MC3/4R inhibitor SHU9119 prevented both the acute (15 min) and longer, more slowly developing (60 min), increases in LSNA in response to ArcN insulin. In conclusion, in females, ArcN insulin increases LSNA, in part, by suppressing tonic PVN NPY inhibition, which unmasks excitatory α-MSH drive of LSNA. Moreover, the steadily increasing rise in LSNA induced by ArcN insulin is also dependent on PVN MC3/4R.
Keywords: PVN, NPY, SNA, α-MSH, arcuate nucleus
the pancreatic hormone, insulin, binds to receptors in the arcuate nucleus (ArcN) to increase basal and baroreflex control of lumbar sympathetic nerve activity (LSNA) via a neuronal pathway that includes the hypothalamic paraventricular nucleus (PVN) (3, 8, 20). Insulin activates at least two excitatory inputs into the PVN: α-melanocyte stimulating hormone (α-MSH), which binds to melanocortin type 3/4 receptors (MC3/4R) (20), and glutamate, which binds to NMDA receptors (19).
Another major ArcN neuronal population that projects to the PVN expresses neuropeptide Y (NPY)/agouti-related protein (AgRP). NPY tonically inhibits PVN presympathetic neurons that are also stimulated by α-MSH (4). This response is primarily mediated by the NPY receptor subtype 1 (Y1R) (4). Moreover, insulin has been shown to inhibit ArcN NPY neurons (10). However, whether insulin also increases LSNA via suppression of tonically inhibitory NPY inputs is unknown.
Therefore, we tested the hypothesis that insulin increases LSNA and its baroreflex regulation, at least in part, via inhibition of ArcN NPY neurons projecting to the PVN. To test this hypothesis, we determined whether prior nanoinjection into the PVN of BIBO 3304, a selective NPY Y1R antagonist, or NPY attenuates the increases in heart rate (HR), LSNA, and baroreflex control of LSNA induced by ArcN insulin in female rats. Because a role for PVN α-MSH inputs in the sympathoexcitatory effects of insulin has not yet been established in females, we also determined whether prior inhibition of PVN MC3/4R attenuates insulin-induced increases in HR, LSNA, and baroreflex control of LSNA.
METHODS
Animals
Female virgin Sprague-Dawley rats (250–290 g; Charles River) were used for all experiments. Female rats were chosen for study because of the contribution of PVN NPY and α-MSH inputs to elevated basal SNA during pregnancy (14). The sympathoexcitatory effects of insulin do not vary during the estrous cycle (13); therefore, the cycle stage was not established before experimentation. Rats were housed in a room with a 12:12-h light-dark cycle with free access to food (LabDiet 5001, Richmond, IN) and water. All procedures were conducted in accordance with the National Institutes of Health's Guide for the Health and Use of Laboratory Animals and approved by the Institutional (Oregon Health & Science University) Animal Care and Use Committee.
Surgery
All surgical procedures were performed under isoflurane anesthesia (2% in 100% O2) following induction (5%). Body temperature was monitored with a rectal thermistor and maintained at 37 ± 1°C using a heating pad. Femoral arterial (1) and venous (3) catheters were implanted for the measurement of arterial pressure (AP) and drug infusions, respectively. Rats were artificially ventilated via an endotracheal tube (with 100% O2), and expired CO2 was maintained at 30–40 mmHg for the duration of the experiment. The lumbar nerve was located after a midline incision and placed on bipolar stainless-steel electrodes. The nerve was secured around the electrode with lightweight silicone material (Kwik-Sil; World Precision Instruments, Sarasota, FL). Rats were placed in a stereotaxic frame (David Kopf Instruments, Tujunga, CA), and the top of the skull was exposed. An opening was burred through the skull on the midline caudal to bregma to expose the dorsal surface of the forebrain. After completion of surgery, isoflurane was withdrawn, and anesthesia transitioned to α-chloralose [40 mg/ml in 0.1 μM sodium tetraborate; loading dose: 50 mg/kg over 30 min (10–12.5 μl/min); maintenance dose: 25 mg·kg−1·h−1 (2.5–3.1 μl/min)] for the duration of the experiment. Rats were allowed to stabilize for at least 1 h before experimental protocols were performed.
Data Acquisition
Pulsatile and mean AP (MAP) were continuously recorded on a Grass polygraph via connection of the arterial catheter to a pressure transducer and Grass bridge amplifier. The pulsatile signal was directed to a Grass tachograph amplifier for determination and recording of HR. Data were collected using a Biopac MP100 data acquisition and analysis system (sampling rate 2 kHz). Raw data (MAP, HR, and LSNA) were averaged into 1-s bins. LSNA was band-pass filtered (100-3,000 Hz) and amplified (×10,000). The LSNA signal was then rectified and integrated in 1-s bins. Post mortem LSNA was subtracted from values of LSNA recorded during the experiment. LSNA was normalized to control LSNA before nanoinjections were initiated (% control).
Baroreflex Function
Baroreflex gain (BRG) was measured by lowering MAP to ∼50 mmHg by intravenous infusion of nitroprusside (1 mg/ml; 20 μl/min), then by slowly raising MAP to ∼175 mmHg over 3–5 min by infusing phenylephrine at increasing rates (1 mg/ml; 1–35 μl/min) and withdrawing nitroprusside. Curves were constructed from data obtained during the MAP rise from 50 to 175 mmHg. The sigmoidal baroreflex relationships between MAP and LSNA generated in each baroreflex test were fitted using the Boltzmann equation: LSNA = (P1 − P2)/[1 + exp (MAP − P3)/P4] + P2. P1 is the maximum LSNA, P2 is the minimum LSNA, P3 is the MAP associated with the LSNA value midway between the maximal and minimal LSNA (BP50; denotes position of the curve on the x-axis), and P4 is the width, the coefficient used to calculate maximum BRG: (P1 − P2)/(P4x4), which is an index of the maximum slope of the sigmoidal baroreflex curve. Absolute values of BRG, the maximum and minimum LSNA, and the BP50 are depicted in the figures.
PVN and ArcN Nanoinjections
The PVN and ArcN were generally functionally identified by observing changes in MAP, HR, and LSNA following unilateral nanoinjection of bicuculline (60 nl of 1 mM/l; Tocris, Bristol, UK) and NMDA (30 nl of 10 mM; Sigma, St. Louis, MO), respectively. The coordinates for PVN and ArcN (using bregma and the dorsal surface of the dura as 0) were as follows: for PVN, 1.8–1.9 mm caudal, 0.5 mm lateral, and 7.4 mm ventral; for ArcN: 3.5–3.6 mm caudal, 0.2–0.3 mm lateral, and 9.5–9.7 mm ventral. Drugs for all nanoinjections were dissolved in artificial cerebrospinal fluid (aCSF) containing (in mM): 128 NaCl, 2.6 KCl, 1.3 CaCl2, 0.9 MgCl2, 20 NaHCO3, and 1.3 Na2HPO4, at pH 7.4. Drug doses were as follows: NPY (60 nl of 0.1 mM in PVN; Tocris), NPY Y1R antagonist BIBO 3304 (60 nl of 1 mM in PVN; Tocris; NPY1x), MC3/4R blocker SHU9119 (60 nl of 0.5 mM in PVN; Tocris), and insulin (60 pU in 30 nl in ArcN; Eli Lilly, Indianapolis, IN). The dose of NPY is sufficient to inhibit food intake (17) and to produce a sustained suppression of LSNA (4) in previous dose-response studies. BIBO 3304 is a highly selective NPY Y1R antagonist, used at a dose that blocks the sympathoinhibitory effects of PVN NPY (4). At this dose, SHU9119 blocks the sympathoexcitatory effects of intravenous insulin (20) and reduces the increases in SNA in response to intracerebroventricular leptin (15). The dose of insulin is within the physiological range (3) and produces a threshold sympathoexcitatory response in female rats that is of similar magnitude to responses to PVN NPY1x. All nanoinjections were made bilaterally (∼2 min for both) and conducted over ∼5–10 s using a PicoPump (World Precision Instruments). At the end of each experiment, ∼60 nl of 2.5% Alcian blue in 0.5 M sodium acetate were injected into the PVN and ArcN. The brain was fixed in 4% paraformaldehyde (PFA) in 0.1 M PBS for 24 h, and the hypothalamus was subsequently cut into 25-μm sections using a cryostat and mounted on gelatin-coated glass microscope slides. Histological locations of PVN and ArcN nanoinjections are summarized in Figs. 1G, 3E, and 6C, using images adapted from Paxinos and Watson (9). We have recently published representative photomicrographs of PVN and ArcN injections (3).
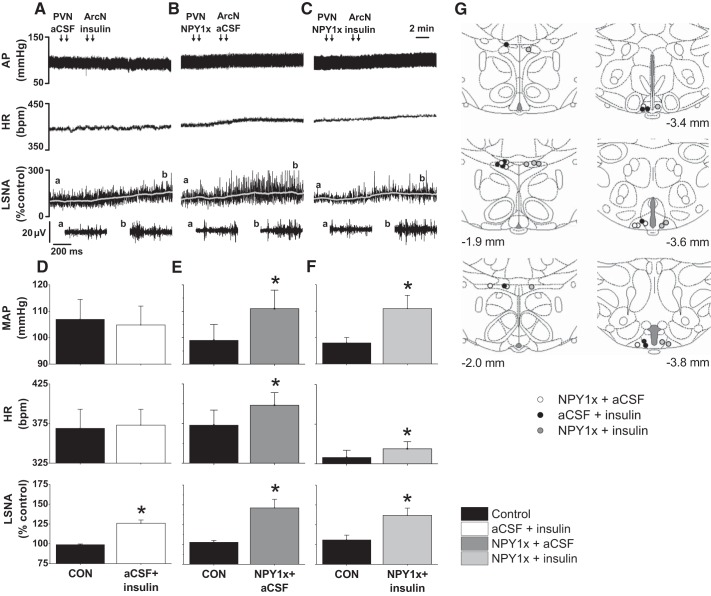
Fig. 1.
Blockade of paraventricular nucleus (PVN) neuropeptide Y (NPY) Y1R (NPY1x) prevents further increases in lumbar sympathetic nerve activity (LSNA) induced by arcuate nucleus (ArcN) insulin. Representative experiments (A–C; the a and b above the top LSNA tracing show where the expanded tracings below were taken.) and group data (D–F) showing that PVN NPY1x (B and E), ArcN insulin (A and D), and PVN NPY1x followed by ArcN insulin (C and F) similarly increased LSNA (bottom). While ArcN insulin did not alter mean arterial pressure (MAP) or heart rate (HR; D), PVN NPY1x followed by ArcN insulin (F) increased AP and HR similarly to PVN NPY1x followed by ArcN artificial cerebrospinal fluid (aCSF) (E). G: illustration of PVN and ArcN injection sites. *P < 0.05 compared with control (CON) values. No between-group differences were observed.
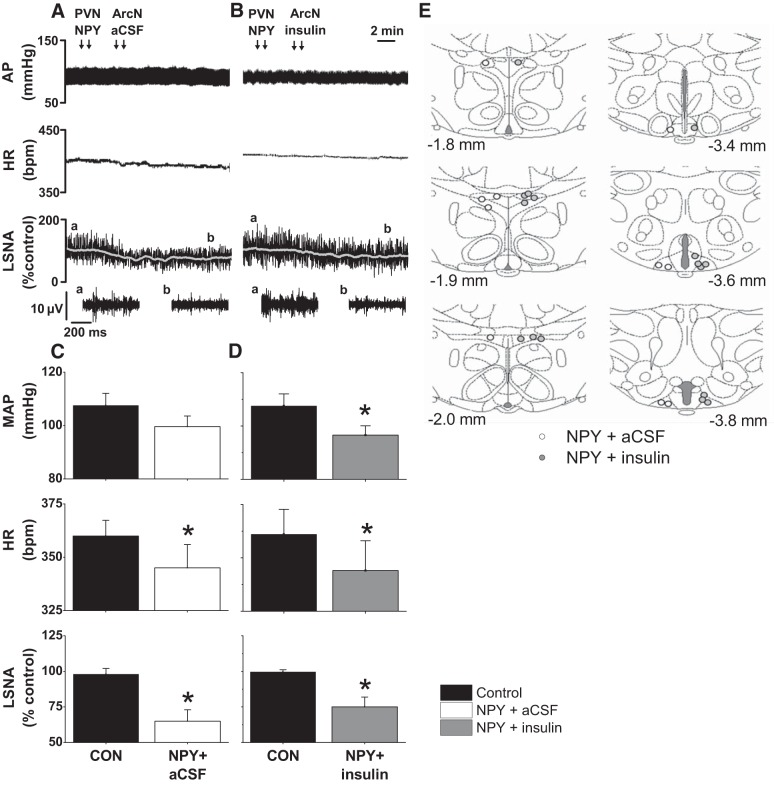
Fig. 3.
PVN NPY prevents the increases in LSNA induced by ArcN insulin. Representative experiments (A and B; the a and b above the top LSNA tracing show where the expanded tracings below were taken.) and group data (C and D) showing that PVN NPY blocks the increases in LSNA (B and D) induced by ArcN insulin. PVN NPY (followed by ArcN aCSF) decreased AP and HR (C) similarly to PVN NPY followed by ArcN insulin (D). *P < 0.05 compared with control (CON) values. E: illustration of PVN and ArcN injection sites. No between-group differences were observed.
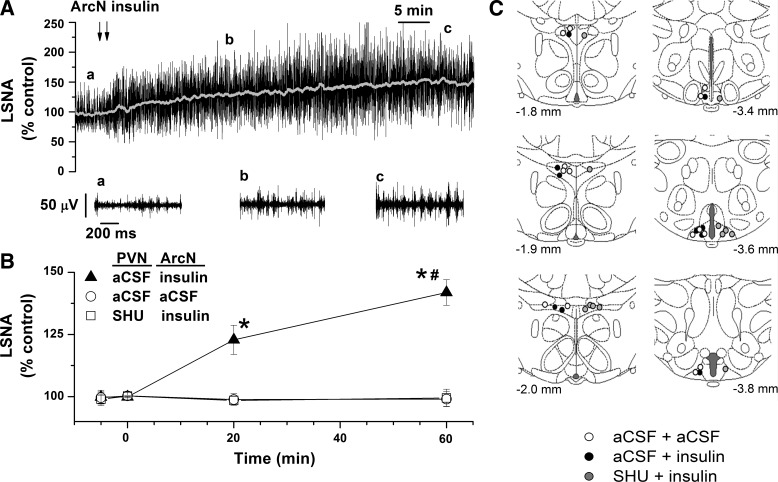
Fig. 6.
ArcN insulin elicits a slowly developing increase in LSNA, which is dependent on PVN MC3/4R. Representative experiment (A; the a and b above the top LSNA tracing show where the expanded tracings below were taken.) and grouped data (B) showing that a single bilateral ArcN nanoinjection of insulin produces a slowly developing sympathoexcitatory response. Prior blockade of PVN MC3/4R with SHU9119 prevents ArcN insulin's effect to increase LSNA (B). C: illustration of PVN and ArcN injection sites. *P < 0.05 compared with control (time zero). #P < 0.05 compared with 20 min.
Experimental Protocols
Experiment 1. Does PVN nanoinjection of NPY1x prevent the effects of ArcN insulin?
MAP, HR, and LSNA and baroreflex control of LSNA were measured before and 15 min after one of three sets of nanoinjections: 1) PVN NPY1x followed 5 min later by ArcN aCSF (n = 5); 2) PVN aCSF followed 5 min later by ArcN insulin (n = 5); and 3) PVN NPY1x followed 5 min later by ArcN insulin (n = 5). We have shown previously that responses to PVN NPY1x reach steady state within 10–15 min (4, 15).
Experiment 2. Does PVN nanoinjection of NPY prevent the effects of ArcN insulin?
MAP, HR, LSNA, and baroreflex control of LSNA were measured before and 15 min after: 1) PVN NPY followed 5 min later by ArcN aCSF (n = 5); 2) PVN aCSF followed 5 min later by ArcN insulin (n = 5; same rats as protocol 1); and 3) PVN NPY followed 5 min later by ArcN insulin (n = 8). Previously, we documented that the responses to PVN NPY stabilize within 15–20 min (4).
Experiment 3. Does PVN nanoinjection of SHU9119 prevent the initial effects of ArcN insulin?
MAP, HR, LSNA, and baroreflex control of LSNA were measured before and 15 min after 1) PVN aCSF followed 5 min later by ArcN insulin (n = 5; same as Protocol 1); and 2) PVN SHU9119 followed 5 min later by ArcN insulin (n = 5). We did not test whether PVN SHU9119 plus ArcN aCSF alters basal values or baroreflex control of LSNA, since neither PVN SHU9119 (4, 13) nor ArcN aCSF [see (3) and below] alters these parameters.
Experiment 4. Does PVN nanoinjection of SHU9119 prevent the longer-term effects of ArcN insulin?
Preliminary experiments revealed that LSNA continues to rise after the 15-min postinsulin time point. Therefore, to determine whether PVN SHU9119 also blocks the later increases in LSNA, we measured LSNA before, 20 min, and 60 min after 1) PVN aCSF and ArcN insulin (n = 5), 2) PVN aCSF and ArcN aCSF (n = 7); and 3) PVN SHU9119 and ArcN insulin (n = 6).
Data Analysis
All data are presented as means ± SE. Thirty-second data segments were collected immediately prior and for experiments 1–3 at the maximum response following nanoinjections into the PVN and ArcN. Data were compared using two-way repeated-measures ANOVA with Newman-Keuls post hoc test. P < 0.05 was considered statistically significant.
RESULTS
PVN NPY1x Blocks the Sympathoexcitatory Effects of ArcN Insulin
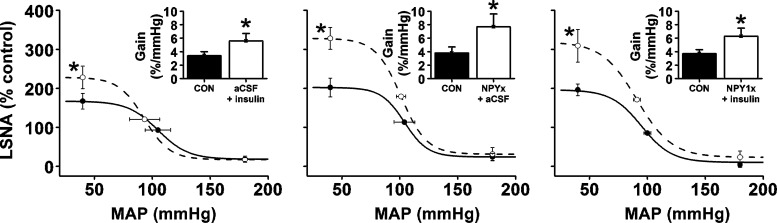
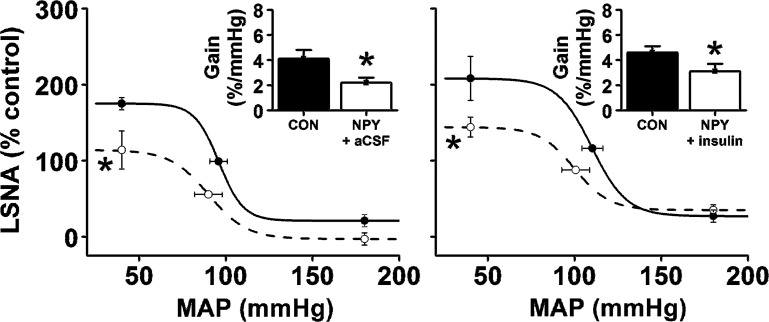
After PVN nanoinjection of aCSF, bilateral nanoinjection of insulin into the ArcN increased (P < 0.05) LSNA, without affecting MAP or HR (Fig. 1, A and D). Insulin also enhanced baroreflex control of LSNA (Fig. 2), by increasing BRG, maximum LSNA, and the baroreflex range (149 ± 25 to 228 ± 26%; P < 0.05). Blockade of PVN NPY Y1 receptors with BIBO 3340 (NPY1x) (followed by ArcN aCSF) increased (P < 0.05) MAP, HR, and LSNA (Fig. 1, B and E). PVN NPY1x also increased gain of baroreflex control of LSNA (Fig. 4), as well as the baroreflex maximum (Fig. 2) and range (178 ± 27 to 298 ± 42%; P < 0.05). Finally, the increases in LSNA and HR and baroreflex control of LSNA induced by PVN NPY1x followed by nanoinjection of insulin into the ArcN were not additive, but similar to the increases produced by PVN NPY1x or ArcN insulin alone (Figs. 1, C and F, and 2).
Fig. 2.
PVN NPY1x, ArcN insulin, and PVN NPY1x followed by ArcN insulin similarly enhance baroreflex control of LSNA (increased baroreflex maximum and gain). *P < 0.05 compared with control (CON) values. No between-group differences were observed.
Fig. 4.
PVN NPY followed by ArcN aCSF suppresses baroreflex control of LSNA (decreased baroreflex maximum and gain) similarly to PVN NPY followed by ArcN insulin. *P < 0.05 compared with control (CON) values. No between-group differences were observed.
PVN NPY Blocks the Sympathoexcitatory Effects of ArcN Insulin
Bilateral nanoinjection of NPY into the PVN followed by ArcN aCSF lowered (P < 0.05) basal MAP, HR, and LSNA (Fig. 3, A and C). PVN NPY also reduced (P < 0.05) LSNA baroreflex gain and the LSNA baroreflex maximum, without significantly affecting other baroreflex parameters (Fig. 4). Nanoinjection of insulin into the ArcN after PVN NPY failed to increase or reverse the effect of NPY on basal LSNA; as a result, the responses to NPY+insulin were similar to NPY alone and significantly lower (P < 0.05) than the effects of PVN aCSF followed by ArcN insulin. More specifically, NPY+insulin decreased (P < 0.05) basal LSNA, MAP, HR, LSNA baroreflex gain, and the LSNA baroreflex maximum below control and below the responses elicited by ArcN insulin (Figs. 3, B and D, and 4).
PVN SHU9119 Blocks the Acute Sympathoexcitatory Effects of ArcN Insulin
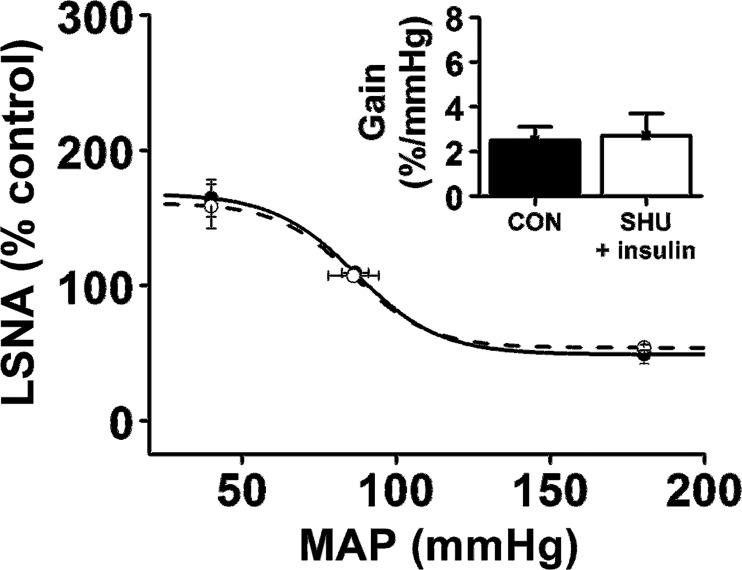
Prior bilateral nanoninjection of SHU9119 into the PVN completely prevented the ability of ArcN insulin to increase LSNA (from 100 ± 1 to 99 ± 3% control) and its baroreflex regulation (Fig. 5). As a result, these responses were significantly lower (P < 0.05) than the effects of ArcN insulin (following PVN aCSF; Figs. 1 and 2). Similarly, as with ArcN insulin (and PVN aCSF), insulin failed to alter MAP (104 ± 2 to 100 ± 2 mmHg), and HR (345 ± 15 to 344 ± 14 beats/min) when administered after PVN SHU91119.
Fig. 5.
Blockade of PVN MC3/4R with SHU9119 prevents ArcN insulin's effect to enhance baroreflex control of LSNA.
Progressively Increasing Sympathoexcitatory Response to ArcN Insulin Is Blocked by Prior PVN SHU9119
As shown in the representative tracing (Fig. 6A) and the grouped data (Fig. 6B), bilateral injection of insulin produced a gradually developing sympathoexcitatory response, in contrast to the effect of the aCSF vehicle, which did not alter LSNA. As a result, LSNA reached higher (P < 0.05) levels in rats that received insulin. However, if the insulin injections were preceded by nanoinjections of SHU9119 into the PVN, LSNA failed to rise even after 1 h (Fig. 6B).
DISCUSSION
The purpose of this study was to test the hypothesis that ArcN insulin increases LSNA, in part, by suppressing tonic inhibitory NPY inputs into the PVN. The major new findings are 1) while both PVN NPY1x and ArcN insulin increased LSNA and its baroreflex regulation, the responses were not additive; 2) PVN NPY prevented the sympathoexcitatory responses to ArcN insulin; 3) prior PVN SHU9119 blocked the acute effects of insulin to increase LSNA and its baroreflex regulation; and 4) the sympathoexcitatory response to ArcN insulin is slowly developing, and PVN SHU9119 also prevented the longer-term sympathoexcitatory effects of ArcN insulin. Therefore, we conclude that ArcN insulin increases LSNA, in part, by suppressing tonic PVN NPY inhibition, which unmasks an excitatory α-MSH drive of LSNA. Moreover, the steadily increasing rise in LSNA induced by insulin is also dependent on PVN MC3/4 receptors.
Recent data suggest that the ArcN can inhibit SNA via a NPY input to the PVN. More specifically, NPY injections into the PVN decrease SNA via binding to Y1R and, to a lesser extent, Y5R (4). In addition, in male rats, nonspecific activation of the ArcN with NMDA decreases AP partly via PVN NPY1R activation (6), and ArcN leptin increases SNA, in part, via suppression of a tonic NPY input to the PVN (15). In male mice, ArcN NPY neurons decrease sympathetically mediated brown adipose tissue thermogenesis, through a neuronal pathway that includes the PVN (12). The present study further demonstrates that ArcN insulin also increases LSNA and its baroreflex regulation, in part, by withdrawal of tonic PVN NPY sympathoinhibition, since PVN NPY1x prevented further increases in LSNA in response to ArcN insulin. A key feature of our approach was that a low dose of insulin was used. Importantly, higher doses of ArcN insulin elicit significantly larger LSNA responses (3); therefore, the ability of NPY1x to prevent further insulin-induced increases in LSNA was not due to a ceiling effect. Moreover, PVN NPY similarly prevented the increases in LSNA.
The present experiments were conducted in female rats, in which, as previously reported (2, 13), insulin [like leptin (13)], increased LSNA without increasing MAP, in contrast to males (13). Extensive studies in humans suggest that this sex difference may be explained by vasodilatory β-adrenergic effects counteracting α-adrenergic vasoconstriction in females (5). Interestingly, we have also recently shown that, unlike insulin, leptin increases LSNA and renal SNA in female rats only during proestrus, due to actions of elevated estrogen levels and to activation of α-MSH inputs to the PVN (13). Future experiments are required to test the role of PVN Y1R in the sympathoexcitatory effects of leptin in proestrus females, as well as in the sympathoexcitatory effects of insulin in males.
In males, insulin and leptin have both been shown to increase SNA via α-MSH inputs to the PVN and PVN MC3/4R (15, 20); however, whether PVN MC3/4R contribute to insulin-induced increases in SNA in females had not been determined. In support of such a role, we found that PVN SHU9119 completely prevented the initial increases in LSNA and baroreflex control of LSNA by ArcN insulin. Moreover, we observed that LSNA continues to rise after a single ArcN insulin injection for up to 1–2 h, as has been shown previously in males (18), and that prior PVN SHU9119 completely prevented these later increases. These results reinforce the notion that insulin increases SNA, in part, via slowly developing signaling events (11). Moreover, the data indicate that the mechanism involves ArcN POMC neurons that project to the PVN.
It is noteworthy that NPY1x, NPY, and SHU9119 all prevented the initial (first 15 min) increases in LSNA. Coupled with the previous finding that SHU9119 blocks the sympathoexcitatory effects of PVN NPY1x (4), these data suggest that the initial increases in LSNA may be mediated completely by withdrawal of tonic NPY inhibition, unveiling α-MSH excitatory drive. With time, without the tonic brake of NPY inputs, α-MSH-mediated excitation is allowed to grow unrestrained.
Because insulin inhibits ArcN NPY neurons (10) and the PVN receives a dense NPY projection from the ArcN (6), the most likely circuitry that subserves NPY actions in the sympathoexcitatory response to ArcN insulin is this subpopulation. Nevertheless, since the ArcN projects to the DMH, which houses NPY PVN-projecting neurons (7), this alternative neuropathway remains possible. Future experiments are required to specifically identify the neuronal pathway involved.
In conclusion, in female rats, ArcN insulin increases LSNA by suppressing tonic NPY inhibition of PVN presympathetic neurons and by activating PVN MC3/4R. Initially, the increases in LSNA can be explained by elimination of PVN NPY inhibition to unveil basal α-MSH-induced excitation. However, with time, the slowly developing sympathoexcitatory response is dependent on PVN MC3/4R.
Perspectives and Significance
We have recently shown that pregnancy decreases tonic NPY inhibition of PVN presympathetic neurons (14). Because pregnancy produces insulin resistance, which increases plasma insulin levels (1), the present results suggest that elevated insulin levels may contribute to the reduced PVN NPY tone exhibited by pregnant rats. Future experiments are required to test this possibility.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
P.A.C., Z.S., and V.L.B. conception and design of research; P.A.C. and Z.S. performed experiments; P.A.C., Z.S., and V.L.B. analyzed data; P.A.C., Z.S., and V.L.B. interpreted results of experiments; P.A.C., Z.S., and V.L.B. prepared figures; P.A.C. and V.L.B. drafted manuscript; P.A.C., Z.S., and V.L.B. approved final version of manuscript; V.L.B. edited and revised manuscript.
ACKNOWLEDGMENTS
This work was supported in part by National Institutes of Health (NIH) Grants HL-088552 and HL-128181, American Heart Association Grants 12GRNT11550018 and AHA15POST23040042, as well as a grant from the Medical Research Foundation of Oregon.
REFERENCES
- 1.Brooks VL, Dampney RA, Heesch CM. Pregnancy and the endocrine regulation of the baroreceptor reflex. Am J Physiol Regul Integr Comp Physiol 299: R439–R451, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brooks VL, Shi Z, Holwerda SW, Fadel PJ. Obesity-induced increases in sympathetic nerve activity: Sex matters. Auton Neurosci 187: 18–26, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cassaglia PA, Hermes SM, Aicher SA, Brooks VL. Insulin acts in the arcuate nucleus to increase lumbar sympathetic nerve activity and baroreflex function in rats. J Physiol 589: 1643–1662, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cassaglia PA, Shi Z, Li B, Reis WL, Clute-Reinig NM, Stern JE, Brooks VL. Neuropeptide Y acts in the paraventricular nucleus to suppress sympathetic nerve activity and its baroreflex regulation. J Physiol 592: 1655–1675, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Charkoudian N, Wallin BG. Sympathetic neural activity to the cardiovascular system: integrator of systemic physiology and interindividual characteristics. Compr Physiol 4: 825–850, 2014. [DOI] [PubMed] [Google Scholar]
- 6.Kawabe T, Kawabe K, Sapru HN. Cardiovascular responses to chemical stimulation of the hypothalamic arcuate nucleus in the rat: role of the hypothalamic paraventricular nucleus. PLoS One 7: e45180, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lee SJ, Kirigiti M, Lindsley SR, Loche A, Madden CJ, Morrison SF, Smith MS, Grove KL. Efferent projections of neuropeptide Y-expressing neurons of the dorsomedial hypothalamus in chronic hyperphagic models. J Comp Neurol 521: 1891–1914, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Luckett BS, Frielle JL, Wolfgang L, Stocker SD. Arcuate nucleus injection of an anti-insulin affibody prevents the sympathetic response to insulin. Am J Physiol Heart Circ Physiol 304: H1538–H1546, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Paxinos G, Watson C. The Rat Brain in Sterotaxic Coordinates. San Diego: Academic Press, 2007. [Google Scholar]
- 10.Qiu J, Zhang C, Borgquist A, Nestor CC, Smith AW, Bosch MA, Ku S, Wagner EJ, Ronnekleiv OK, Kelly MJ. Insulin excites anorexigenic proopiomelanocortin neurons via activation of canonical transient receptor potential channels. Cell Metab 19: 682–693, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rahmouni K, Morgan DA, Morgan GM, Liu X, Sigmund CD, Mark AL, Haynes WG. Hypothalamic PI3K and MAPK differentially mediate regional sympathetic activation to insulin. J Clin Invest 114: 652–658, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shi YC, Lau J, Lin Z, Zhang H, Zhai L, Sperk G, Heilbronn R, Mietzsch M, Weger S, Huang XF, Enriquez RF, Baldock PA, Zhang L, Sainsbury A, Herzog H, Lin S. Arcuate NPY controls sympathetic output and BAT function via a relay of tyrosine hydroxylase neurons in the PVN. Cell Metab 17: 236–248, 2013. [DOI] [PubMed] [Google Scholar]
- 13.Shi Z, Brooks VL. Leptin differentially increases sympathetic nerve activity and its baroreflex regulation in female rats: role of oestrogen. J Physiol 593: 1633–1647, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shi Z, Cassaglia PA, Gotthardt LC, Brooks VL. Hypothalamic paraventricular and arcuate nuclei contribute to elevated sympathetic nerve activity in pregnant rats: roles of neuropeptide Y and α-melanocyte-stimulating hormone. Hypertension 66: 1191–1198, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shi Z, Li B, Brooks VL. Role of the paraventricular nucleus of the hypothalamus in the sympathoexcitatory effects of leptin. Hypertension 66: 1034–1041, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stanley BG, Leibowitz SF. Neuropeptide Y injected in the paraventricular hypothalamus: a powerful stimulant of feeding behavior. Proc Natl Acad Sci USA 82: 3940–3943, 1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Steiner JL, Bardgett ME, Wolfgang L, Lang CH, Stocker SD. Glucocorticoids attenuate the central sympathoexcitatory actions of insulin. J Neurophysiol 112: 2597–2604, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stocker SD, Gordon KW. Glutamate receptors in the hypothalamic paraventricular nucleus contribute to insulin-induced sympathoexcitation. J Neurophysiol 113: 1302–1309, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ward KR, Bardgett JF, Wolfgang L, Stocker SD. Sympathetic response to insulin is mediated by melanocortin 3/4 receptors in the hypothalamic paraventricular nucleus. Hypertension 57: 435–441, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]