To the Editor:
Wiskott-Aldrich syndrome (WAS) is an X-linked disorder characterized by thrombocytopenia, eczema, and immunodeficiency. Up to 70% of patients with WAS present with at least 1 autoimmune or autoinflammatory episode, and many of them suffer from recurrent or multiple events.1, 2, 3 IL-1 new-generation blockers have been used in patients exhibiting clinical symptoms compatible with an autoinflammatory condition,4 but have not been reported in WAS. Here, we describe a patient with WAS with a peculiar large genomic inversion presenting with multiple manifestations of immune dysregulation, in whom autoinflammatory manifestations improved after the use of anakinra (IL-1 receptor antagonist, Kineret).
A 11.6-year-old boy was referred to our center for suspected immunodeficiency. The patient presented with a history of microthrombocytopenia since birth and eczema in the first years of life, suggestive of WAS. Analysis of WAS protein (WASp) expression was reported abnormal, but Sanger sequencing on DNA did not reveal mutations. From 1.5 years of age he underwent recurrent episodes of postinfectious vasculitis of the lower limbs and arthritis. At 7.5 years, he presented with a bilateral pneumonia that triggered Schonlein-Henoch purpura with fever and arthritis, managed with oral steroids. Subsequently, a nephritic-nephrotic syndrome was treated with antihypertensive treatment and high-dose corticosteroids (CCS), with partial response. Cyclosporin A (CyA) and CCS led to remission of renal disease, which relapsed after CyA was stopped. Intravenous high-dose CCS and anti-CD20 mAb did not lead to substantial improvement. CyA and low-dose prednisone were restarted with partial benefit. However, the patient experienced varicella zoster reactivation on his half-right-face, with sequelae to the right eye (anterior and posterior uveitis with acute retinitis) requiring a vitrectomy, and severe impairment of visual function. An anterior uveitis at the left eye was treated with steroids. At the age of 9.8 years, he developed clinical and histological features of pancolitic Crohn disease, managed with an increase in CCS, as well as arthritis and histologically confirmed vasculitis and eventually pyoderma gangrenosum (PG) on the hips, buttocks, and upper and lower limbs. Crohn disease was not responsive to infliximab, thalidomide, cyclophosphamide, or high-dose intravenous steroids, while adalimumab (Humira) resulted in an initial benefit (see Table E1 in this article's Online Repository at www.jacionline.org). The patient presented with fistulas and perianal abscesses when he was 10.7 years old and he underwent several fistulectomies and removal of granulation tissue in the perianal area by “cone-like technique.” For the poor control of the enterocolitis, a subtotal colectomy with terminal ileostomy was performed at age 11 years.
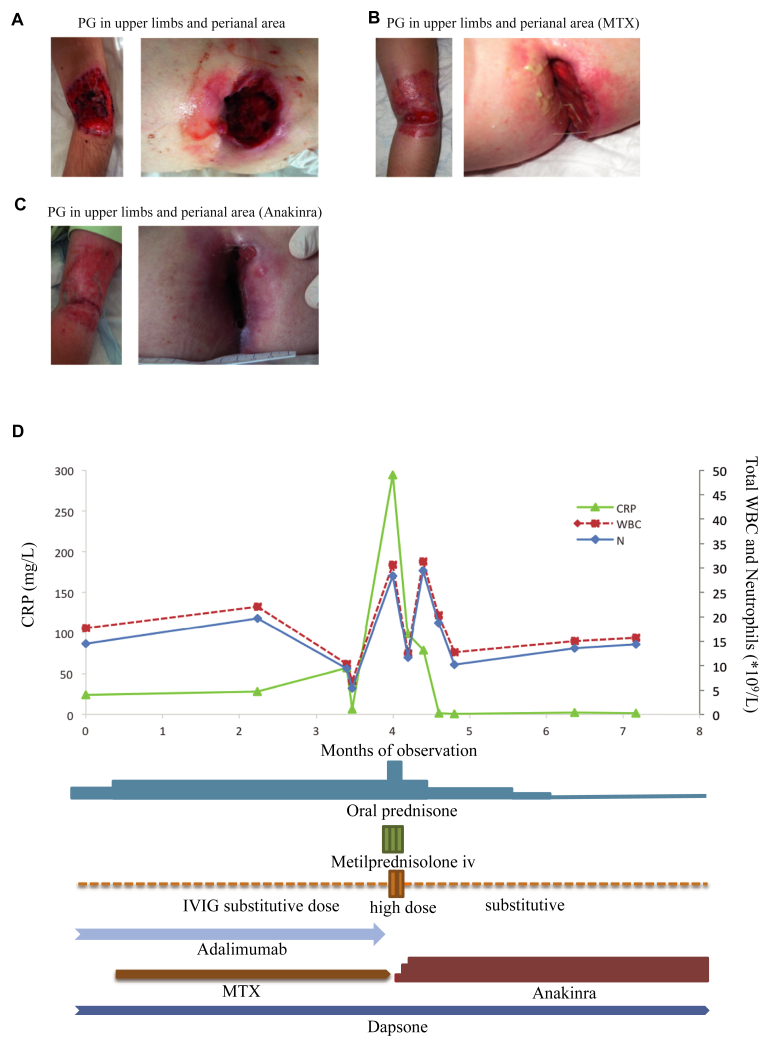
When the patient was referred to our center, he was on adalimumab and low-dose CCS with a good control of bowel disease, but still showed severe manifestations of PG on the upper limbs and in the perianal area (Fig 1, A; see Table E2 in this article's Online Repository at www.jacionline.org). His parents signed informed consent for research investigations (protocol Tiget06).
Fig 1.
Skin lesions and biochemical markers in a patient with WAS with autoinflammatory manifestations. A, Patient at the time of WAS diagnosis. B, Patient after 3 months of treatment with MTX. C, Patient after 5 months of treatment with anakinra. D, Inflammation pattern during several immunosuppressive and antinflammatory treatments administered. Increase in CRP, WBC, and neutrophils (N) at month 4 and amelioration after treatment with anakinra. CRP, C-Reactive protein; IV, intravenous; IVIG, intravenous immunoglobulin; MTX, methotrexate; WBC, white blood cell.
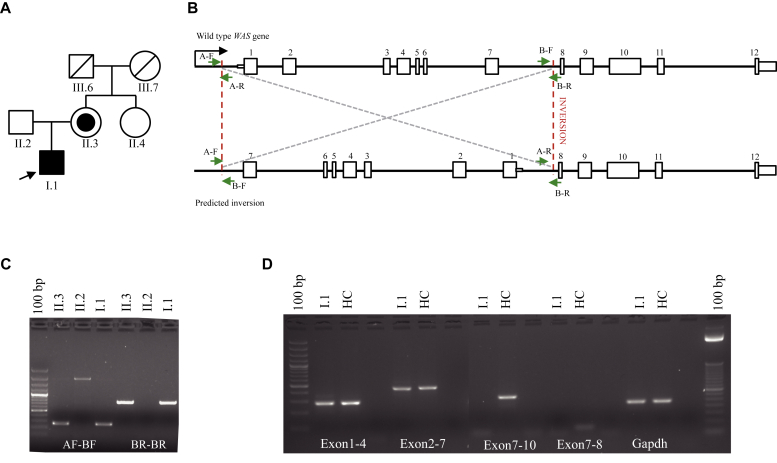
Because of the strong suspicion of WAS, whole-genome sequencing was performed and an inversion of 6kb spanning from the promoter to the intronic region between exons 7 and 8 was detected (see Fig E1 in this article's Online Repository at www.jacionline.org). Specific primers in this region identified the precise breaking points (see Tables E3 and E4 in this article's Online Repository at www.jacionline.org; Fig 2, B). The rearranged allele was present in the patient and his mother, whereas the patient's aunt was unaffected (data not shown and Fig 2, A-C).
Fig E1.
Graphical representation of WGS results. WAS gene and coverage are indicated. Primers R1 and R2 for Illumina sequencing that pair correctly are represented in gray. The red lines in the patient indicate the pairing in the region spanning the inversion and their specific orientation. HC, Healthy control; WGS, whole-genome sequencing.
Fig 2.
Identification of inversion in the WAS gene. A, Pedigree of the family. Proband is indicated by arrow. B, Graphical representation of predicted effects of inversion in the WAS gene. Primer design in the sites of inversion. C, DNA amplification with primers AF/BF and AR/BR in the family. Aspecific band in sample II.2. D, cDNA amplification with indicated primers.
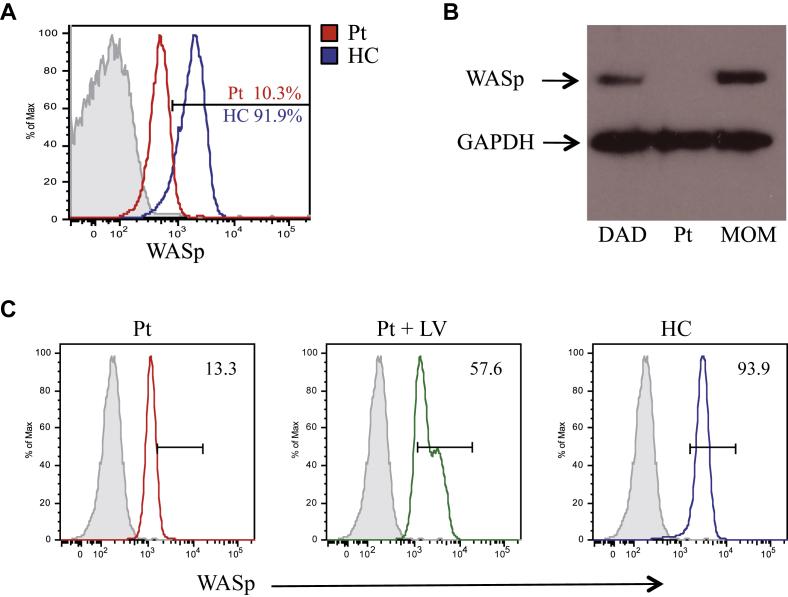
RNA analyses showed an aberrant transcript produced from the inverted region (Fig 2, D). WASp expression, analyzed by flow cytometry (see Fig E2, A, in this article's Online Repository at www.jacionline.org), was deeply reduced in peripheral blood T-, B-, and natural killer lymphocytes and monocytes (data not shown) while it was undetectable by Western blot performed with an antibody recognizing the N-terminal portion of WASp including exons 7 and 8 (Fig E2, B). WASp expression was restored in the patient's T-cell line transduced with a lentiviral vector expressing WASp under the control of the autologous 1.6-kb long promoter5 (Fig E2, C).
Fig E2.
WASp expression. A, Flow cytometry characterization of WASp expression in patient and HC lymphocytes. Percentage of WASp+ cells is reported on histograms. Detection of WASp was performed after permeabilization (Cytofix/Cytoperm kit; BD Biosciences, San Jose, Calif) by a noncommercial rabbit polyclonal antibody (“EFOB,” generated against a WASp peptide), kindly provided by GSK. Gray line: negative control. B, Western blot performed on untransformed T-cell line generated in vitro from patient and his parents. Antibody for detection: polyclonal H250 (BD). C, Restoration of WASp expression in a patient's T-cell line after transduction with LV. GAPDH, Glyceraldehyde 3-phosphate dehydrogenase; HC, healthy control; LV, lentiviral vector; Pt, patient.
The start of low-dose methotrexate (Reumaflex) and the increase in prednisone led to a moderate improvement in the PG after 3 months (Fig 1, B), but shortly after the patient underwent a reactivation of vasculitis and arthritis with systemic inflammation that was not controlled by multiple immunosuppressive and anti-inflammatory drugs.
On the basis of the reported efficacy of IL-1 blockers in the treatment of autoinflammatory manifestations and of PG,4 anakinra was started as an off-label drug titrating the dose from 1 up to 3 mg/kg/day. This led to a resolution of vasculitis and arthritis and to a decrease in the inflammation indexes within few days (Fig 1, D) with dramatic improvement in the PG skin lesions during the following 5 months (Fig 1, C).
The patient was enrolled in a gene therapy trial based on autologous gene-corrected hematopoietic stem cells (clinicaltrials.gov #NCT01515462), mobilized with G-CSF and plerixafor. Treatment with anakinra was discontinued 48 hours before mobilization, but was soon restarted because of the increase in white blood cells and inflammation indexes with exacerbation of skin lesions, arthralgia, and hematuria, and led again to a rapid laboratory and clinical remission (data not shown). Notably, the use of anakinra allowed a successful mobilization with G-CSF without the occurrence of other autoinflammatory manifestations. To our knowledge, this is the first reported case of use of IL-1R blocker in a patient with WAS, with clinical benefit.
This case is very emblematic for several reasons. Whole-genome sequencing complemented by specific breakpoint sequencing allowed the identification of the inversion with intact exon sequences, elucidating the previous normal genetic analysis. Complex genomic rearrangements involving inversions are generally noncanonical gene conversion events6 and could have occurred in an ancestor allele in the family through a de novo mutation occurring in the mother.
Autoimmune and autoinflammatory manifestations in patients with WAS typically present early in life, are often refractory to therapy, and are associated with a worse clinical prognosis and an increased risk of developing a malignancy.3, 7 Our patient's autoinflammatory manifestations were resistant to several immunosuppressive drugs and the use of CyA was associated with a severe viral complication. Anakinra dramatically improved PG, vasculitis, and arthritis, showed a good safety profile, and allowed stabilization of the patient for definitive treatment. The response to anakinra suggests that the dysregulation of the innate immune system is involved in the genesis of autoinflammatory manifestations in patients with WAS and shows that IL-1 may serve in selected cases as a target for therapy, avoiding the use of other classes of immunosuppressors that can increase the risk for severe infections.
It has been hypothesized that defects in chemotaxis and podosomes formation in WASp-deficient cells may favor the onset of autoinflammatory manifestations. In addition, a recent study in a patient with aggressive PG showed a critical role for proline-serine-threonine phosphatase interacting protein 1, which is involved in cytoskeletal regulatory functions through interaction with WASp, in the Pyogenic Arthritis, Pyoderma gangrenosum, and Acne syndrome.8 A greater understanding of the role of WASp in inflammation and of potential pathways that may be targeted therapeutically to modulate immunity in WAS is desirable to improve the management of the affected patients while waiting for definitive treatment by stem cell transplantation or gene therapy.
Footnotes
This work was supported by Fondazione Telethon and FP7-EU grant n° HEALTH-F5-2010-261387 (CELL-PID) to A.A. GSK has licensed gene therapy for WAS from Telethon and San Raffaele and in 2014 became the financial sponsor of the clinical trial.
Disclosure of potential conflict of interest: A. Aiuti declares grants from Fondazione Telethon and the European Commission and is the Principal Investigator of the TIGET-WAS clinical trial sponsored by GSK. The rest of the authors declare that they have no relevant conflicts of interest.
Appendix
Table E1.
Summary of treatment of possible autoimmune and autoinflammatory manifestations
| Treatment | Response |
|---|---|
| Nephritic/nephrotic syndrome | |
| Steroids | Partial |
| Steroids + CyA | Remission, then relapse when CyA stopped |
| Steroids + anti-CD20 mAb | No |
| Steroids + CyA | Partial; infectious complications |
| Crohn disease | |
| Steroids + CyA | No |
| Steroids + CyA + IVIG | No |
| Steroids + CyA + infliximab | Partial, then relapse |
| Steroids + CyA + talidomide | No |
| Steroids + cyclophosphamide | No |
| Steroids + adalimumab | Yes |
| PG | |
| Steroids + dapsone + adalimumab | No |
| Steroids + dapsone + adalimumab + MTX | Partial |
| Steroids + dapsone + anakinra | Yes |
| Arthritis and vasculitis | |
| Steroids + CyA | Remission, then relapse |
| Steroids + CyA + infliximab | No |
| Steroids + dapsone + adalimumab + MTX | No |
| Steroids + dapsone + anakinra | Yes |
IVIG, Intravenous immunoglobulin; MTX, methotrexate.
Table E2.
Clinical data and significant laboratory findings at WAS diagnosis
| Age of onset/diagnosis of disease | Neonatal/11.6 y |
| Genetic analyses (whole-genome sequencing) | inv(X)g.5721-11840 |
| Clinical manifestations | Microthrombocytopenia Mild eczema, severe infections, arthritis, vasculitis Multiple Henoch-Schonlein purpura episodes Nephritic-nephrotic syndrome, panuveitis Steroid-resistant Chron enterocolitis Perianal fistulas and abscesses PG |
| Zhu score | 5AE1 |
| WBC (×109/L) (normal value, 4.8-10.8 × 109/L) | 14.4 |
| Neutrophils (×109/L) (normal value, 1.8-7.7 × 109/L) | 12.7 |
| Lymphocytes (×109/L) | 1 |
| Platelets range (×109/L) | 20-40 |
| MPV (fl) (normal value, 9.1-12.5) | 8 |
| CD3+ (cells/μL) (normal value, 1000-2100)E2 | 498 |
| CD3+CD4+ (cells/μL) (normal value, 500-1300)E2 | 364 |
| CD3+CD8+ (cells/μL) (normal value, 220-950)E2 | 91 |
| CD19+ (cells/μL) (normal value, 180-600)E2 | 101 |
| CD16+CD56+ (cells/μL) (normal value, 170-670)E2 | 338 |
| CD4+CD45RA+ (cells/μL) (normal value, 320-1000)E3 | 152 |
| CD4+CD45RO+ (cells/μL) (normal value, 230-630)E3 | 201 |
| CD8+CD45RA+ (cells/μL) (normal value, 310-900)E3 | 159 |
| CD8+CD45RO+ (cells/μL) (normal value, 70-390)E3 | 40 |
| Vβ repertoire | Polyclonal on CD3+ |
| IgG (g/L) (normal value, 7.07-19.19)E4 | 7.69 (on IVIG supplementation) |
| IgA (g/L) (normal value, 0.60-2.7)E4 | 6.68 |
| IgM (g/L) (normal value, 0.61-2.76)E4 | 0.33 |
| Autoimmunity screening∗ | Negative |
| Antiplatelets antibody (indirect test) | Positive |
| LLAC | Negative |
ACA, Anticardiolipin antibody; AMA, antimitochondrial antibody; ANA, antinuclear antibody; ASCA, anti–Saccharomyces cerevisiae antibody; ASMA, anti–smooth muscle antibody; cANCA, cytoplasmic antineutrophilic cytoplasmic antibodies; ds-DNA, double-strand DNA antibody; ENA, extractable nuclear antibodies; IVIG, intravenous immunoglobulin; LLAC, lupus-like anticoagulant; LKM1, liver kidney microsomal type 1 antibody; pANCA, protoplasmic antineutrophilic cytoplasmic antibodies; WBC, white blood cell.
Including Coombs test, ANA, ENA, AMA, ASMA, anti–LKM1 antibody, anti–dS-DNA antibody, ASCA, cANCA, pANCA, ACA, and anti–β2 glicoprotein antibody.
Table E3.
Primers for the detection of DNA inversion
| Primer | Orientation | Sequence | PCR cycles |
|---|---|---|---|
| A | FOR | CTGGAGATACGCCTAGCACA | 94°C 3 min, 94°C 30 s, 55°C 30 s, 72°C 1 min × 40 cycles, 72°C 10 min |
| REV | TCTGAGGGCTGTAGGGTTTG | ||
| B | FOR | AAGGTGGGCACATGGGTAG | 94°C 3 min, 94°C 30 s, 55°C 30 s, 72°C 1 min × 40 cycles, 72°C 10 min |
| REV | TCTCAGCAGTGGTCTTTGGA |
Table E4.
Primers for amplification of cDNA
| Region | Orientation | Sequence | PCR cycles |
|---|---|---|---|
| EXON1-4 (279bp) | FOR | CGAAAATGCTTGACGCTGGC | 94°C 3 min, 94°C 30 s, 54.6°C 30 s, 72°C 1 min × 40 cycles, 72°C 10 min |
| REV | CTCGTCCTCGTCTGCAAAGT | ||
| EXON7-10 (303bp) | FOR | ACCCAGTGGATTCAAGCATGT | 94°C 3 min, 94°C 30 s, 54.6°C 30 s, 72°C 1 min × 40 cycles, 72°C 10 min |
| REV | GGACCAGAACGACCCTTGTT | ||
| EXON2-7 (449bp) | FOR | GTCCTACTTCATCCGCCTTTAC | 94°C 3 min, 94°C 30 s, 55.8°C 30 s, 72°C 1 min × 40 cycles, 72°C 10 min |
| REV | TCTTCCCTGAGCGTTTCTTATC | ||
| EXON7-8 (73bp) | FOR | GTGGATTCAAGCATGTCAGCC | 94°C 3 min, 94°C 30 s, 55.8°C 30 s, 72°C 1 min × 40 cycles, 72°C 10 min |
| REV | TCTGGGTCGAGGTTGTTCAC |
References
- 1.Dupuis-Girod S., Medioni J., Haddad E., Quartier P., Cavazzana-Calvo M., Le Deist F. Autoimmunity in Wiskott-Aldrich syndrome: risk factors, clinical features, and outcome in a single-center cohort of 55 patients. Pediatrics. 2003;111:e622–e627. doi: 10.1542/peds.111.5.e622. [DOI] [PubMed] [Google Scholar]
- 2.Chen N., Zhang Z.Y., Liu D.W., Liu W., Tang X.M., Zhao X.D. The clinical features of autoimmunity in 53 patients with Wiskott-Aldrich syndrome in China: a single-center study. Eur J Pediatr. 2015;174:1311–1318. doi: 10.1007/s00431-015-2527-3. [DOI] [PubMed] [Google Scholar]
- 3.Imai K., Morio T., Zhu Y., Jin Y., Itoh S., Kajiwara M. Clinical course of patients with WASP gene mutations. Blood. 2004;103:456–464. doi: 10.1182/blood-2003-05-1480. [DOI] [PubMed] [Google Scholar]
- 4.Geusau A., Mothes-Luksch N., Nahavandi H., Pickl W.F., Wise C.A., Pourpak Z. Identification of a homozygous PSTPIP1 mutation in a patient with a PAPA-like syndrome responding to canakinumab treatment. JAMA Dermatol. 2013;149:209–215. doi: 10.1001/2013.jamadermatol.717. [DOI] [PubMed] [Google Scholar]
- 5.Scaramuzza S., Biasco L., Ripamonti A., Castiello M.C., Loperfido M., Draghici E. Preclinical safety and efficacy of human CD34(+) cells transduced with lentiviral vector for the treatment of Wiskott-Aldrich syndrome. Mol Ther. 2013;21:175–184. doi: 10.1038/mt.2012.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen J.M., Chuzhanova N., Stenson P.D., Férec C., Cooper D.N. Intrachromosomal serial replication slippage in trans gives rise to diverse genomic rearrangements involving inversions. Hum Mutat. 2005;26:362–373. doi: 10.1002/humu.20230. [DOI] [PubMed] [Google Scholar]
- 7.Moratto D., Giliani S., Bonfim C., Mazzolari E., Fischer A., Ochs H.D. Long-term outcome and lineage-specific chimerism in 194 patients with Wiskott-Aldrich syndrome treated by hematopoietic cell transplantation in the period 1980-2009: an international collaborative study. Blood. 2011;118:1675–1684. doi: 10.1182/blood-2010-11-319376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Starnes T.W., Bennin D.A., Bing X., Eickhoff J.C., Grahf D.C., Bellak J.M. The F-BAR protein PSTPIP1 controls extracellular matrix degradation and filopodia formation in macrophages. Blood. 2014;123:2703–2714. doi: 10.1182/blood-2013-07-516948. [DOI] [PMC free article] [PubMed] [Google Scholar]
References
- Zhu Q., Watanabe C., Liu T., Hollenbaugh D., Blaese R.M., Kanner S.B. Wiskott-Aldrich syndrome/X-linked thrombocytopenia: WASP gene mutations, protein expression, and phenotype. Blood. 1997;90:2680–2689. [PubMed] [Google Scholar]
- Comans-Bitter W.M., de Groot R., van den Beemd R., Neijens H.J., Hop W.C., Groeneveld K. Immunophenotyping of blood lymphocytes in childhood: reference values for lymphocyte subpopulations. J Pediatr. 1997;130:388–393. doi: 10.1016/s0022-3476(97)70200-2. [DOI] [PubMed] [Google Scholar]
- Shearer W.T., Rosenblatt H.M., Gelman R.S., Oyomopito R., Plaeger S., Stiehm R. Lymphocyte subsets in healthy children from birth through 18 years of age: the Pediatric AIDS Clinical Trials Group P1009 study. J Allergy Clin Immunol. 2003;112:973–980. doi: 10.1016/j.jaci.2003.07.003. [DOI] [PubMed] [Google Scholar]
- Burgio GR, Perinotto G, Ugazio AG. Pediatria essenziale. UTET editore. 1991.