Abstract
Purpose
The development of a genetically modified live rabies vaccine applicable to wild raccoon dogs is necessary for the eradication of rabies in Korea. Thus, we constructed a recombinant rabies virus (RABV) called the ERAGS strain, using a reverse genetic system and evaluated its safety and efficacy in mice and its safety and immunogenicity in raccoon dogs.
Materials and Methods
ERAGS, which has Asn194Ser and Arg333Glu substitutions in the glycoprotein, was constructed using site-directed mutagenesis. Mice were inoculated with the ERAGS strain (either 105.0 or 107.0 FAID50/mL) via intramuscular (IM) or intracranial injections and then challenged with a virulent RABV. Raccoon dogs were administered the ERAGS strain (108.0 FAID50/mL) either orally or via the IM route and the immunogenicity of the strain was evaluated using fluorescent antibody virus neutralization tests.
Results
The ERAGS strain inoculated into murine neuroblastoma cells reached 107.8 FAID50/mL at 96-hour post-inoculation. The virus was not pathogenic and induced complete protection from virulent RABV in immunized 4- and 6-week-old mice. Korean raccoon dogs immunized with the ERAGS strain via IM or oral route were also safe from the virus and developed high titer levels (26.4-32.8 IU/mL) of virus-neutralizing antibody (VNA) at 4 weeks post-inoculation.
Conclusion
The ERAGS RABV strain was effectively protective against rabies in mice and produced a high VNA titer in raccoon dogs.
Keywords: Rabies virus, Mouth, Vaccines, Raccoon dogs
Introduction
Rabies is an important zoonosis that results in more than 60,000 human deaths around the world every year. Once its clinical symptoms are evident, death is almost inevitable. Mass vaccination of dogs, including stray dogs in high-risk areas, can reduce the incidence of rabies in both animals and humans [1]. In South Korea, the rabies virus (RABV) isolates are clustered into an Arctic-like lineage based on its molecular and biological characteristics and has been circulated by animals bitten by raccoon dogs (Nyctereutes procynoide korensis) and badgers (Meles meles) since the 1990s [2]. The distribution of oral bait vaccines with either live attenuated RABV (SAG2 strain), recombinant adenovirus (ONRAB), or vaccinia-rabies glycoprotein (V-RG) is the most effective method of eliminating rabies in wild animals [3,4,5]. The World Health Organization (WHO) has recommended that live rabies vaccines should not cause any adverse symptoms in either target or nontarget species including rodents, wild animals, or other domestic species [6,7].
Currently, an inactivated vaccine is used to prevent rabies in animals, and the use of this vaccine will soon be expanded in an attempt to completely eliminate the occurrence of dog-mediated human rabies. However, its high cost is threatening the achievement of 70% vaccination coverage in developing countries [6]. On the other hand, live attenuated rabies vaccines are more cost-effective and simpler to manufacture. For example, the Evelyn-Rokitnicki–Abelseth (ERA) strain propagated in primary porcine kidney cell cultures or Vero cells has been used to immunize dogs and cattle, and shows protective immunogenicity [8]. Although live rabies vaccine strains can also be given to wild animals via oral administration, there use is no longer recommended by world organizations involved in animal health. Furthermore, the ERA strain used in live vaccines cannot be administered to cats or wild carnivores in South Korea due to safety concerns [7,9].
RABV is a single-stranded negative sense RNA virus belonging to Lyssavirus of the family Rhabdoviridae. The genome consists of five genes that encode a nucleoprotein (N), phosphoprotein (P), matrix protein (M), glycoprotein (G), and large protein (L). Three of these proteins (N, P, and L) form a ribonucleoprotein complex that serves as a template for virus transcription and replication. When associated with the N and L proteins, the P protein acts as a non-catalytic cofactor of the viral RNA polymerase [10]. The G protein, which represents the major surface protein of the virion, is associated with an interaction with the receptor binding site, fusion to facilitate virus entry into host cells, and the induction of a neutralizing antibody [11].
The G protein is also responsible for virulence. Either the arginine (Arg) or lysine (Lys) residue at position 333 of the G protein determines RABV virulence [12]. When the Arg333 residue in glycoprotein RABVs is substituted with glutamic acid (Glu), isoleucine (Ile), glycine (Gly), methionine (Met), or serine (Ser), the variants are less pathogenic or avirulent in adult mice following intracranial (IC) inoculation [13,14]. However, after more than three back passages of these recombinant RABVs in suckling mice, a single mutation (asparagine: Asn to Lys) at position 194 of the G protein occurs and increases virulence [15]. Recombinant RABVs rescued with a single mutation at position 333 have the risk of reverting to a virulent virus. However, this risk can be circumvented by constructing an RABV with multiple mutations that involve Glu at position 333 and Asn at position 194, which would be more appropriate as a live vaccine because it cannot revert into a pathogenic virus and thus will be useful for animals [15].
Therefore, we constructed a novel recombinant RABV termed the ERAGS strain that contains two mutations at positions 194 and 333 of the G protein. Its safety and efficacy was evaluated in mice, and its safety and immunogenicity was assessed in raccoon dogs.
Materials and Methods
Cells and viruses
BHK/T7-9 cells were grown in Dulbecco's modified Eagle medium (DMEM) with 600 ng/mL hygromycin, 10% heat-inactivated fetal bovine serum (FBS), and several antibiotics (100 IU/mL penicillin, 10 µg/mL streptomycin, and 0.25 µg/mL amphotericin B) and then maintained in 3% FBS [16]. Murine neuroblastoma (NG108-15) cells were maintained in DMEM supplemented with 5% FBS and then placed in a 5% CO2 incubator at 37℃. The ERA strain of RABV that was introduced from Canada in 1974 was used as a safety comparison for the ERAGS strain. A challenge virus standard (CVS) strain, CVSN2c, which is a virulent RABV, was used to assess the efficacy of ERAGS in mice while another CVS strain, CVS11, which is a fixed RABV, was used for the fluorescent assay virus neutralizing (FAVN) tests in raccoon dogs.
Construction of the recombinant RABV
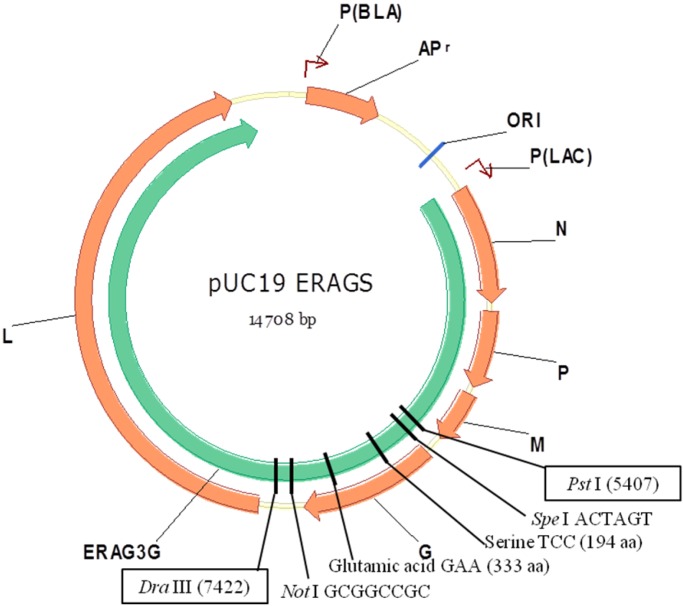
Full-length cDNA modified with Ser and Glu amino acid mutations at positions 194 and 333 of the G gene of the ERA strain was cloned into the pTM1 vector. Briefly, restriction enzyme sites of Pst I and Dra III were located at positions 5,407 and 7,422 in the full-length cDNA. After the digestion of cDNA with two restriction enzymes, the Asn (AAT) to Ser (TCC; Asn194Ser) and Arg (AGA) to Glu (GAA; Arg333Glu) mutations were conducted at positions 194 and 333 of the G gene in the DNA fragment via site-directed mutagenesis (Fig. 1). After the modified DNA fragment was inserted into the original plasmid, the modified full-length cDNA was constructed and designated the pUC19 ERAGS plasmid.
Fig. 1.
Full-length plasmid map for the construction of the ERAGS strain. The full-length cDNA plasmid was modified using site-directed mutagenesis at positions 194 and 333 of the glycoprotein in the ERA strain.
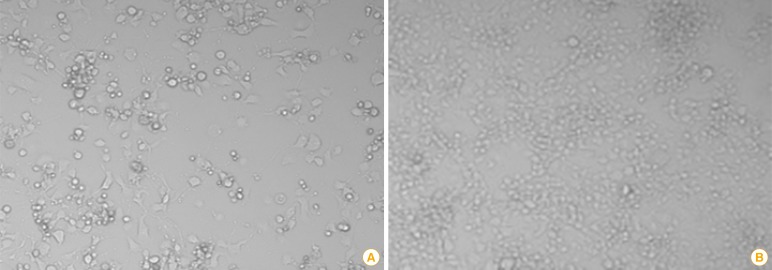
The N, P, and L genes obtained from the ERA strain were also cloned into the same vector and named EN, EP, and EL, respectively. To recover the recombinant RABV (the ERAGS strain) from cloned full-length cDNA and helper plasmids, the full-length modified genome plasmid (2 µg), EN (0.25 µg), EP (0.02 µg), and EL (0.25 µg) were mixed with 9 µL X-treme GENE HP DNA transfection reagent (Roche Diagnostic, Mannheim, Germany) and transfected into BHK/T7-9 cells grown in a 24-well tissue culture plate. Following transfection, the cells were screened for cytopathic effects for 5 days (Fig. 2).
Fig. 2.
Cytopathic effects (×200) of BHKT7-9 cells (A) infected with the ERAGS strain and normal cells (B). BHKT7-9 cells infected with the virus were detached from the plate.
Identification of the recombinant RABV
The second-passaged recombinant ERAGS strain was propagated in BHK/T7-9 cells and infected cells were fixed using 80% chilled acetone for 15 minutes. To stain the cells, they were reacted with a specific monoclonal antibody (Median Diagnostics, Chuncheon, Korea) against the N protein of the RABV for 45 minutes and then stained with fluorescence isothiocyanate (FITC)-conjugated goat anti-mouse IgG+IgM (KPL, Gaithersburg, MD, USA). After being washed in phosphate buffered saline (PBS, pH 7.2), the cells were examined via fluorescent microscopy (Nikon, Tokyo, Japan) and a commercial rapid immunodiagnostic assay (RIDA) kit was used according to the manufacturer's instruction as a supplementary identification measure (Bionote, Hwaseong, Korea). Briefly, a mixture of 50 µL supernatant of the ERAGS strain and 50 µL proprietary buffer was applied to the sample well of the RIDA kit. The appearance of two lines was considered a positive result.
Propagation of the ERAGS strain in NG108-15 cells
A monolayer of NG108-15 cells in a 25 cm2 tissue culture flask was infected with either the ERA or ERAGS strain at a multiplicity of infection of 0.01. The flasks were incubated at 37℃ and frozen at 12-, 24-, 36-, 48-, 72-, 96-, and 120-hour post-infection. After being thawed and frozen three times, the ERA and ERAGS strains were titered in a 96-well microplate (tenfold dilution) and the viral titer was determined by an indirect fluorescent assay calculated using the method of Reed and Muench.
Safety and potency of RABVs in mice
Mice (4- and 6-week-old BALB/c type) were divided into seven groups consisting of either four or eight animals each. Groups 1 and 2 received intramuscular (IM) inoculations with the ERAGS strain (105.0 and 107.0 FAID50/mL, respectively), group 3 received an IM inoculation with the ERA strain (107.0 FAID50/mL), groups 4 and 5 received IC inoculations with the ERAGS strain (105.0 and 107.0 FAID50/mL, respectively), group 6 received an IC inoculation with the ERA strain (107.0 FAID50/mL), and group 7 was the control group and did not receive an inoculation. Each mouse was inoculated with either 30 µL (IC) or 100 µL (IM) of the virus and mice showing central nervous system symptoms were humanely euthanized. The body weights of the mice were measured after inoculation and their survival rates were observed for 16 days after the inoculation. To determine the efficacy of the ERAGS strain, mice that survived after immunization were challenged with a lethal 100 µL IM dose of 25 LD50/0.1 mL CVSN2c strain in the right leg. Changes in the body weights of the mice were measured and the survival rates of the mice were observed for 14 days after the challenge.
Safety and immunogenicity of the ERAGS strain in raccoon dogs
Raccoon dogs (4 months old) that were sero-negative against RABV were divided into three groups. Group 1 included six raccoon dogs that received oral administration of the ERAGS strain (1 mL, 108.0 FAID50/mL), group 2 included four animals that received IM inoculations with the same strain and dose, and group 3 included three animals that did not receive any treatment as a control group. All raccoon dogs were monitored daily for clinical signs of rabies such as abnormal behavior, nervous prostration, anxiety, agitation, aggression, and paralysis. Two and four weeks after inoculation, the body weight of each raccoon dog was measured and blood was collected from each subject, including controls. The titers of the serum samples are expressed in international units per milliliter (IU/mL) and were compared to the titers of a rabies-positive standard serum (WHO). The minimum protective titer has been determined to be 0.5 IU/mL FAVN [6,17].
Serological assay
A virus neutralizing antibody (VNA) test was determined using the FAVN test [17] with a positive reference serum sample obtained from the WHO (adjusted to 0.5 IU/mL) used as a positive control. Briefly, 50 µL volumes of serum and positive and negative controls were distributed in four consecutive wells and then serially diluted threefold. Then the CVS11 RABV strain (approximately 100 FAID50/50 µL) was added to each well and incubated for 1 hour at 37℃. Next, 50 µL BHK-21 cell suspension (4×105 cells/mL) was added to each well and the microplates were incubated for 72 hours in a humidified incubator with 5% CO2 at 37℃. The cells within the microplates were fixed with 100 µL cold acetone (-20℃) for 20 minutes, successively washed three times with PBS, reacted with a specific monoclonal antibody against the N protein of RABV for 45 minutes at 37℃, and then stained with FITC-conjugated goat/anti-mouse IgG+IgM. Following a wash in PBS, the microplates were air-dried and examined at 200× using a fluorescent microscope (Nikon). The titers of the serum samples were expressed in IU/mL by comparing the results to those of the positive standard.
Statistical analysis
All data are expressed as means±standard deviations and all statistical analyses of the VNA titers and body weights were conducted with IBM SPSS version 19.0 for Windows (IBM Corp., Armonk, NY, USA). Differences between the groups were analyzed with a one-way analysis of variance (ANOVA) followed by Tukey's post hoc tests, and for more detailed investigations, unpaired Student's t-tests were performed for each individual time point. p-values of <.05 were considered to indicate statistical significance.
Results
Construction of recombinant RABV
After transfection with a mixture of the full-length genome mutated at positions 194 and 333 in the G gene of the ERA strain and three helper plasmids (EN, EP, and EL), the recombinant RABV (ERAGS strain) was successfully rescued in BHK/T7-9 cells. The BHK/T7-9 cells inoculated with the ERAGS strain were fixed with cold acetone and strained using a specific antibody against the N protein of RABV. RABV-specific fluorescence appeared in the cytoplasm of the cells infected with the rescued virus indicating that the specific fluorescence in cytoplasm was RABV (Fig. 3). Additional tests, including the use of the RIDA kit, confirmed that the rescued virus was RABV (Fig. 4).
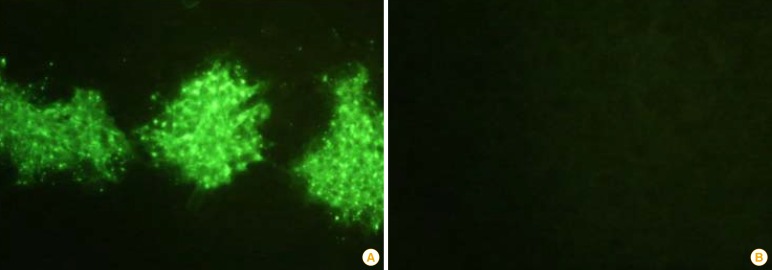
Fig. 3.
Immunofluorescence (×200) of the ERAGS strain using an indirect fluorescent assay test with monoclonal antibodies against the nucleoprotein of the rabies virus (A) and normal cells (B). Recombinant rabies virus-specific fluorescent sites appeared in the cytoplasm of the infected BHK/T7-9 cells.
Fig. 4.
Results of the rapid immunodiagnostic assay (RIDA) kit following the application of the ERAGS strain supernatant. The "C"and "T"in the RIDA kit stand refer to the "control"and "test"lines. The appearance of two lines was considered to be a positive result.
Growth properties and genetic stability
After three successive passages with the rescued ERAGS strain, the kinetics of the growth properties of the ERA and ERAGS strains were analyzed to determine whether the two mutations affected the replication ability of the ERAGS strain in NG108-15 cells. The growth curve of the ERAGS strain was similar to that of ERA strain but it had a slightly higher titer (107.8 FAID50/mL vs. 107.3 FAID50/mL) (Fig. 5). Both strains had the highest titer at 96-hour post-inoculation, which indicated that the mutations at positions 194 and 333 in the G protein of the ERA strain did not influence the growth properties.
Fig. 5.
Multistep growth curves of the ERA and ERAGS strains in NG108-15 cells. Both strains revealed the highest viral titer at 96-hour post-inoculation.
Safety and efficacy of the ERAGS strain in mice
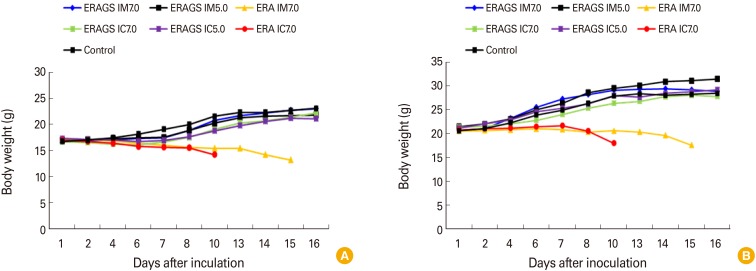
The body weights of mice inoculated with either the ERA or ERAGS strain via either the IM or IC route were measured on designated days after the inoculation. The average body weight of mice that received ERA IM slowly decreased until 10 days after inoculation (Fig. 6) and the average weight of those that received ERA IC showed a similar trend until 15 days. In contrast, the weights of the IM and IC groups inoculated with different titers of the ERAGS strain increased without any drastic weight loss.
Fig. 6.
Changes in body weights in 4-week-old (A) and 6-week-old (B) mice inoculated with either the ERA or ERAGS strains via either the intramuscular (IM) or intracranial (IC) route. The weights of mice inoculated with the ERAGS strain continued to increase whereas those of mice inoculated with ERA progressively decreased, and these mice eventually died.
Regarding survival, all mice inoculated with the ERA strain, whether IM or IC, succumbed to rabies and died 10 or 15 days post-inoculation, respectively. In contrast, all mice inoculated with ERAGS (both IM and IC) and all control mice survived (Figs. 6, 7). All mice in the ERAGS group were subsequently challenged with a highly virulent RABV strain, CVSN2c, on day 15 post-immunization. The average body weights of the mice slightly increased and their survival rates did not change for 14 days after the challenge (Fig. 8) but the average weight of control mice inoculated with virulent RABV began to decrease rapidly 7-8 days post-challenge and all mice died 9-10 days post-challenge. Thus, the ERA strain was pathogenic whereas the ERAGS strain was non-pathogenic and safe for adult mice.
Fig. 7.
Survival in 4-week-old (A) and 6-week-old (B) mice inoculated with either the ERA or ERAGS strain via either the intramuscular (IM) or intracranial (IC) route. Mice inoculated with ERA (both IM and IC) died 10 and 16 days post-inoculation, respectively. In contrast, 100% of mice inoculated with ERAGS (both IM and IC) survived, as did controls.
Fig. 8.
Changes in body weights (A, B) and survival rates (C, D) in 4- and 6-week-old mice that were immunized with the ERAGS strains via either the intramuscular (IM) or intracranial (IC) route and then challenged with a highly pathogenic rabies virus strain (CVSN2c). The average body weight increased for 14 days after the challenge and the survival rate did not change for 14 days after the challenge.
Safety and immunogenicity of the ERAGS strain in raccoon dogs
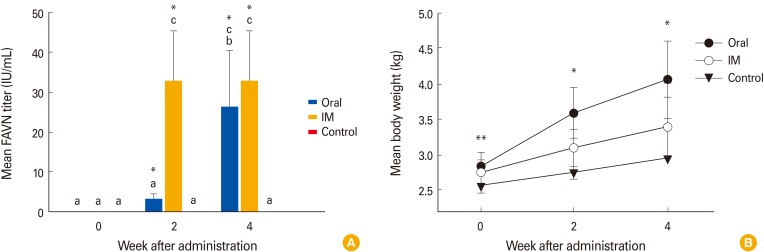
All raccoon dogs were sero-negative for rabies VNA prior to inoculation, and sero-converted at 2-4 weeks after its administration. Neither vaccinated nor non-vaccinated raccoon dogs exhibited any clinical signs of rabies during the test period. At 2 weeks post-inoculation, animals that received IM inoculations of the ERAGS strain developed a higher VNA titer (32.8 IU/mL) against rabies than those that were orally inoculated (3.27 IU/mL). However, all raccoon dogs that were immunized with the ERAGS strain (either orally or IM) exhibited similar mean VNA titers that ranged from 26.4 to 32.8 IU/mL at 4 weeks after inoculation (Fig. 9). Three animals in group 3 remained sero-negative against RABV throughout the test, which confirmed that no contact transmission occurred between the vaccinated groups and the control group (Fig. 9A). The body weights of those that received oral inoculations with the ERAGS were slightly heavier than those that received IM inoculations. The average body weight of group 3 increased during the same period (Fig. 9B).
Fig. 9.
The immune responses and body weights of raccoon dogs that received the ERAGS strain via either the oral or intramuscular (IM) route (26.4 and 32.8 IU/mL, respectively). At 4 weeks post-inoculation, high virus-neutralizing antibody titers were induced in all immunized raccoon dogs (A). The body weights of those that received oral administrations significantly increased whereas those that received IM administrations did not (B). Each bar represents the mean±standard error of the mean of three independent samples. Different lowercase letters indicate significant differences (p < 0.05, Tukey's post hoc test). Asterisks (*p < 0.05) and double asterisks (**p < 0.01) in each panel indicate significant differences from corresponding control groups without any treatment (unpaired t-test). FAVN, fluorescent antibody virus neutralization.
Discussion
The use of oral immunizations with a rabies bait vaccine has greatly contributed to the elimination of rabies in wild animals. For example, a live attenuated rabies vaccine (the SAG2 strain), which was isolated by selecting a monoclonal antibody, has been shown to be safe and immunogenic based on the absence of adverse clinical signs and salivary excretion in animals [18,19,20]. Thus, this vaccine has been registered as an oral rabies vaccine for wild animals in Europe and dogs in India. However, highly attenuated RABVs, such as the HEP-Flury and SPBNGA strains, revert to virulence following a single amino acid substitution to Arg at position 333 in the G protein after multiple passages in suckling mice [21,22]. This can lead to potential safety issues. The V-RG oral rabies vaccine meets the WHO safety requirements for animals and has played a key role in controlling rabies in wild animals but it has also raised safety concerns after two cases of human vaccinia infection [23].
Thus, because safer candidates are necessary, several recombinant RABVs constructed using reverse genetic systems have been developed under names such as the rERAG333E, rERAG3G, SPBNGA, Ni-CE (G333Glu), and ERAG333 strains [15,24,25,26,27]. These recombinant RABVs are based on the concept that the Arg at position 333 of the G protein of RABV is responsible for virulence. If the Arg is substituted with other amino acids, including Glu, Leu, and Gly, then its viral virulence is greatly decreased in adult mice [14,27]. It has also been reported that single mutated recombinant RABV strains containing a Glu at position 333 do not exhibit pathogenicity in mice older than 7 days [27]. A previous study from our research group found that the ERAG3G strain, which was developed based on the above mentioned concepts, is safe for several types of animals, including 4-week-old mice, and induces a high VNA titer against RABV in cats, dogs, and raccoon dogs [25]. However, because it is possible that the single mutated RABV strain can revert to pathogenic RABV strains, safer RABV vaccine strains are required to implement rabies eradication programs for wild animals.
Thus, we generated a novel recombinant RABV strain (called ERAGS), which is mutated at positions 194 and 333 of the G protein of the ERA strain. The ERA and ERAGS strains grew at similar rates in NG108-15 cells and reached over 107.5 FAID50/mL at 96-hour post-inoculation. The growth property of the ERAGS strain seems to be similar to those of other recombinant RABVs, such as the rERAG333E and Ni-CE (G333Glu) strains [15,24,26].
The pathogenicity of RABV has been measured in mice inoculated via a variety of routes. The important factors involved in RABV virulence include the age of the mice, the inoculation route, and the virus strain [28]. To determine whether the ERAGS strain was safe for mice, 4- and 6-week-old mice were inoculated with different titers of either the ERA or ERAGS strains via either the IM or IC routes. Mice that received the ERA strain with a titer of 107.0 FAID50/mL via either route exhibited virulence 6 days after inoculation. On the other hand, mice inoculated with the ERAGS strain containing either 105.0 or 107.0 FAID50/mL via either route were observed for 16 days and did not exhibit any clinical signs. Thus, the safety of the ERAGS strain is similar to that of recombinant RABVs [24,25,26]. In addition, the mean body weight of the mice did not decrease after the inoculation indicating that the ERAGS strain is non-pathogenic in 4- and 6-week-old mice.
The efficacy of the ERAGS strain was also evaluated in immunized mice using a virulent RABV challenge. The mice inoculated with the ERAGS strain were challenged with a 25 LD50/0.03 mL dose of rabies IC and all mice survived for 14 days. Furthermore, there were no clinical signs or decreases in body weight in these mice, indicating that a single immunization with different titers of the ERAGS strain may confer complete protection against an RABV challenge.
Raccoon dogs are primarily responsible for rabies transmission in South Korea and the safety and immunogenicity of any oral vaccine candidate should be evaluated in the main target animals. VNA is a critical protective component against lethal RABV infections [29] and the WHO considers a titer of 0.5 IU/mL rabies VNA to be the minimum protective antibody titer in carnivores [6]. Mucosal-related lymphoid tissues, including the tonsils, are a primary site of infection and this is replicated after the administration of an oral vaccine strain in dogs [30,31]. The present study showed that all raccoon dogs that received a 108.0 FAID50/mL dose via the oral route did not exhibit any adverse behavioral effects and developed high VNA titers that ranged from 13.7 to 41.6 IU/mL at 4 weeks post-administration. These findings suggest that raccoon dogs treated with the ERAGS strain can be protected from a challenge inoculation with virulent RABV, which provides a basis for the use of the rabies bait vaccine strain. Future studies from our research group will extend these findings to dogs, cats, and cattle to assess whether the ERA strain that has been used for prevention of rabies in South Korea since the 1980s can be replaced. In addition, back passages of the ERAGS strain in suckling mice will be conducted to elucidate the likelihood that this strain will revert to pathogenic RABV.
In conclusion, the ERAGS strain, which is mutated at positions 194 and 333 of the G protein, was safe and efficacious for use in 4- and 6-week-old mice. In addition, a single immunization with the ERAGS strain via both the IM and oral routes induced a high VNA titer against RABV in raccoon dogs. Therefore, the ERAGS strain may be sufficiently safe and effective for use as a live vaccine or oral bait vaccine in raccoon dogs.
Footnotes
No potential conflict of interest relevant to this article was reported.
This work was supported financially by a grant (B1543083-2016-18-01) from Animal, and Plant Quarantine Agency, Ministry of Agriculture, Food and Rural Affairs (MAFRA), Republic of Korea.
References
- 1.Seneschall C, Luna-Farro M. Controlling rabies through a multidisciplinary, public health system in Trujillo, La Libertad, Peru. Pathog Glob Health. 2013;107:361–366. doi: 10.1179/2047773213Y.0000000123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hyun BH, Lee KK, Kim IJ, et al. Molecular epidemiology of rabies virus isolates from South Korea. Virus Res. 2005;114:113–125. doi: 10.1016/j.virusres.2005.06.004. [DOI] [PubMed] [Google Scholar]
- 3.Mahl P, Cliquet F, Guiot AL, et al. Twenty year experience of the oral rabies vaccine SAG2 in wildlife: a global review. Vet Res. 2014;45:77. doi: 10.1186/s13567-014-0077-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brown LJ, Rosatte RC, Fehlner-Gardiner C, et al. Oral vaccination and protection of red foxes (Vulpes vulpes) against rabies using ONRAB, an adenovirus-rabies recombinant vaccine. Vaccine. 2014;32:984–989. doi: 10.1016/j.vaccine.2013.12.015. [DOI] [PubMed] [Google Scholar]
- 5.Fehlner-Gardiner C, Rudd R, Donovan D, Slate D, Kempf L, Badcock J. Comparing ONRAB(R) AND RABORAL V-RG(R) oral rabies vaccine field performance in raccoons and striped skunks, New Brunswick, Canada, and Maine, USA. J Wildl Dis. 2012;48:157–167. doi: 10.7589/0090-3558-48.1.157. [DOI] [PubMed] [Google Scholar]
- 6.World Health Organization. WHO Expert Consultation on Rabies. World Health Organ Tech Rep Ser. 2005;931:1–88. [PubMed] [Google Scholar]
- 7.Faber M, Dietzschold B, Li J. Immunogenicity and safety of recombinant rabies viruses used for oral vaccination of stray dogs and wildlife. Zoonoses Public Health. 2009;56:262–269. doi: 10.1111/j.1863-2378.2008.01215.x. [DOI] [PubMed] [Google Scholar]
- 8.Lawson KF, Chiu H, Crosgrey SJ, Matson M, Casey GA, Campbell JB. Duration of immunity in foxes vaccinated orally with ERA vaccine in a bait. Can J Vet Res. 1997;61:39–42. [PMC free article] [PubMed] [Google Scholar]
- 9.Yang DK, Go TO, Nam YH, et al. Antibody response in Korean raccoon dogs inoculated with inactivated rabies vaccines. J Bacteriol Virol. 2012;42:242–246. [Google Scholar]
- 10.Albertini AA, Schoehn G, Weissenhorn W, Ruigrok RW. Structural aspects of rabies virus replication. Cell Mol Life Sci. 2008;65:282–294. doi: 10.1007/s00018-007-7298-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Raux H, Flamand A, Blondel D. Interaction of the rabies virus P protein with the LC8 dynein light chain. J Virol. 2000;74:10212–10216. doi: 10.1128/jvi.74.21.10212-10216.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dietzschold B, Wunner WH, Wiktor TJ, et al. Characterization of an antigenic determinant of the glycoprotein that correlates with pathogenicity of rabies virus. Proc Natl Acad Sci U S A. 1983;80:70–74. doi: 10.1073/pnas.80.1.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Seif I, Coulon P, Rollin PE, Flamand A. Rabies virulence: effect on pathogenicity and sequence characterization of rabies virus mutations affecting antigenic site III of the glycoprotein. J Virol. 1985;53:926–934. doi: 10.1128/jvi.53.3.926-934.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tuffereau C, Leblois H, Benejean J, Coulon P, Lafay F, Flamand A. Arginine or lysine in position 333 of ERA and CVS glycoprotein is necessary for rabies virulence in adult mice. Virology. 1989;172:206–212. doi: 10.1016/0042-6822(89)90122-0. [DOI] [PubMed] [Google Scholar]
- 15.Faber M, Faber ML, Papaneri A, et al. A single amino acid change in rabies virus glycoprotein increases virus spread and enhances virus pathogenicity. J Virol. 2005;79:14141–14148. doi: 10.1128/JVI.79.22.14141-14148.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ito N, Takayama-Ito M, Yamada K, Hosokawa J, Sugiyama M, Minamoto N. Improved recovery of rabies virus from cloned cDNA using a vaccinia virus-free reverse genetics system. Microbiol Immunol. 2003;47:613–617. doi: 10.1111/j.1348-0421.2003.tb03424.x. [DOI] [PubMed] [Google Scholar]
- 17.Cliquet F, Aubert M, Sagne L. Development of a fluorescent antibody virus neutralisation test (FAVN test) for the quantitation of rabies-neutralising antibody. J Immunol Methods. 1998;212:79–87. doi: 10.1016/s0022-1759(97)00212-3. [DOI] [PubMed] [Google Scholar]
- 18.Cliquet F, Gurbuxani JP, Pradhan HK, et al. The safety and efficacy of the oral rabies vaccine SAG2 in Indian stray dogs. Vaccine. 2007;25:3409–3418. doi: 10.1016/j.vaccine.2006.12.054. [DOI] [PubMed] [Google Scholar]
- 19.Cliquet F, Robardet E, Must K, et al. Eliminating rabies in Estonia. PLoS Negl Trop Dis. 2012;6:e1535. doi: 10.1371/journal.pntd.0001535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lafay F, Benejean J, Tuffereau C, Flamand A, Coulon P. Vaccination against rabies: construction and characterization of SAG2, a double avirulent derivative of SADBern. Vaccine. 1994;12:317–320. doi: 10.1016/0264-410x(94)90095-7. [DOI] [PubMed] [Google Scholar]
- 21.Takayama-Ito M, Inoue K, Shoji Y, et al. A highly attenuated rabies virus HEP-Flury strain reverts to virulent by single amino acid substitution to arginine at position 333 in glycoprotein. Virus Res. 2006;119:208–215. doi: 10.1016/j.virusres.2006.01.014. [DOI] [PubMed] [Google Scholar]
- 22.Dietzschold ML, Faber M, Mattis JA, Pak KY, Schnell MJ, Dietzschold B. In vitro growth and stability of recombinant rabies viruses designed for vaccination of wildlife. Vaccine. 2004;23:518–524. doi: 10.1016/j.vaccine.2004.06.031. [DOI] [PubMed] [Google Scholar]
- 23.Rupprecht CE, Blass L, Smith K, et al. Human infection due to recombinant vaccinia-rabies glycoprotein virus. N Engl J Med. 2001;345:582–586. doi: 10.1056/NEJMoa010560. [DOI] [PubMed] [Google Scholar]
- 24.Shuai L, Feng N, Wang X, et al. Genetically modified rabies virus ERA strain is safe and induces long-lasting protective immune response in dogs after oral vaccination. Antiviral Res. 2015;121:9–15. doi: 10.1016/j.antiviral.2015.06.011. [DOI] [PubMed] [Google Scholar]
- 25.Yang DK, Nakagawa K, Ito N, et al. A single immunization with recombinant rabies virus (ERAG3G) confers complete protection against rabies in mice. Clin Exp Vaccine Res. 2014;3:176–184. doi: 10.7774/cevr.2014.3.2.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nakagawa K, Ito N, Masatani T, et al. Generation of a live rabies vaccine strain attenuated by multiple mutations and evaluation of its safety and efficacy. Vaccine. 2012;30:3610–3617. doi: 10.1016/j.vaccine.2012.03.044. [DOI] [PubMed] [Google Scholar]
- 27.Bankovskiy D, Safonov G, Kurilchuk Y. Immunogenicity of the ERA G 333 rabies virus strain in foxes and raccoon dogs. Dev Biol (Basel) 2008;131:461–466. [PubMed] [Google Scholar]
- 28.Mebatsion T. Extensive attenuation of rabies virus by simultaneously modifying the dynein light chain binding site in the P protein and replacing Arg333 in the G protein. J Virol. 2001;75:11496–11502. doi: 10.1128/JVI.75.23.11496-11502.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Franka R, Wu X, Jackson FR, et al. Rabies virus pathogenesis in relationship to intervention with inactivated and attenuated rabies vaccines. Vaccine. 2009;27:7149–7155. doi: 10.1016/j.vaccine.2009.09.034. [DOI] [PubMed] [Google Scholar]
- 30.Orciari LA, Niezgoda M, Hanlon CA, et al. Rapid clearance of SAG-2 rabies virus from dogs after oral vaccination. Vaccine. 2001;19:4511–4518. doi: 10.1016/s0264-410x(01)00186-4. [DOI] [PubMed] [Google Scholar]
- 31.Ogra PL, Faden H, Welliver RC. Vaccination strategies for mucosal immune responses. Clin Microbiol Rev. 2001;14:430–445. doi: 10.1128/CMR.14.2.430-445.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]