Abstract
Background
Due to the concerns about the overtreatment of low-risk prostate cancer (PCa), active surveillance (AS) is now a recommended alternative to the active treatments (AT) of surgery and radiotherapy. However, AS is not widely utilized, partially due to psychological and decision-making factors associated with treatment preferences.
Methods
In a longitudinal cohort study, we conducted pretreatment telephone interviews (N=1,140, 69.3% participation) with newly diagnosed, low-risk PCa patients (PSA≤10, Gleason≤6) from Kaiser Permanente Northern California. We assessed psychological and decision-making variables, and treatment preference [AS, AT, No Preference (NP)].
Results
Men were 61.5 (SD=7.3) years old, 24 days (median) post-diagnosis, and 81.1% white. Treatment preferences were: 39.3% AS, 30.9% AT, and 29.7% NP. Multinomial logistic regression revealed that men preferring AS (vs. AT) were older (OR=1.64, CI 1.07-2.51), more educated (OR=2.05, CI 1.12-3.74), had greater PCa knowledge (OR=1.77, CI 1.43-2.18) and greater awareness of having low-risk cancer (OR=3.97, CI 1.96-8.06), but also were less certain about their treatment preference (OR=0.57, CI 0.41 - 0.8), had greater PCa anxiety (OR=1.22, CI 1.003-1.48), and preferred a shared treatment decision (OR=2.34, CI 1.37-3.99). Similarly, men preferring NP (vs. AT) were less certain about treatment preference, preferred a shared decision, and had greater knowledge.
Conclusions
Although a substantial proportion of men preferred AS, this was associated with anxiety and uncertainty, suggesting that this may be a difficult choice.
Impact
Increasing the appropriate use of AS for low-risk PCa will require additional reassurance and information, and reaching men almost immediately post-diagnosis while the decision-making is ongoing.
Keywords: low-risk prostate cancer, active surveillance, treatment decision making
In 2016, 180,890 new cases of prostate cancer are expected in the U.S. Approximately 35-40% of these cases will have a low risk of disease progression (1), meaning that treatment is unlikely to be beneficial and that men are more likely to die of causes other than their prostate cancer (2-6). However, historically, most men with low-risk prostate cancer receive an active treatment of either surgery or radiotherapy (7-15), which frequently leads to sexual, urinary, and bowel problems (16-20). Given concerns about overtreatment, active surveillance has become an increasingly important option for men with low-risk prostate cancer (21). Active surveillance protocols, using serial prostate-specific antigen (PSA) tests, digital rectal exams, and periodic biopsies, allow men to avoid treatment and its complications if the cancer does not progress. Several large observational studies have shown low rates of disease progression and mortality for men on active surveillance (15, 22-27) and one randomized trial is underway (28). Further, several organizations now recommend active surveillance for low-risk prostate cancer (29-31).
Despite the advantages of active surveillance, it continues to be underused for low-risk prostate cancer for many reasons, including lack of patient awareness of active surveillance, patient anxiety regarding living with untreated cancer, physician anxiety regarding not treating the cancer, the societal inclination for aggressively treating all cancers, and financial incentives for treating cancer (32-36). Salaried physicians in integrated health care systems can offer treatment options that are not influenced by financial incentives. Studying patients in these systems can provide a clearer assessment of the patient-related psychological and decision-making factors associated with treatment selection. Integrated systems, such as Health Maintenance Organizations (HMOs), represent a large and increasing proportion of U.S. healthcare delivery (37, 38). In 2007-2008, 74 million Americans were enrolled in a group model HMO (39). However, we are not aware of any large-scale studies assessing treatment decisions among low-risk prostate cancer patients in these settings. To address this gap, we have accrued a prospective cohort of men newly diagnosed with low-risk prostate cancer in an HMO setting to comprehensively assess factors associated with treatment decision-making for low-risk prostate cancer.
In developing our measures and our research questions, we were guided by Zafar et al.'s (40) model of treatment decision-making and quality of life (Figure 1), which postulates the role of several demographic, clinical, and decision-making factors in reaching a satisfactory treatment decision. We have adapted the model to include events specific to low-risk PCa, in order to recognize the impact of disease monitoring, the potential need for subsequent treatment decisions, that treatment preferences may change over time, and quality of life outcomes. This model postulates the importance of shared decision making in reaching a satisfactory decision, the need to consider the opinions of both patients and physicians, and the discordance that can occur between patient and physician treatment preferences. Further, the model recognizes that both patient and physician treatment preferences may change over time, dependent on changes in disease status, experience with treatments, and previous QOL. The measures included in Figure 1, under Baseline Patient Characteristics, Baseline Decisional and Psychological Characteristics, and Baseline Treatment Decision Resources, include most of the measures that were recommended for inclusion by Zafar's model. These will be used, along with subsequent measures of patient preference and physician recommendation, in a multivariate model to predict treatment decision(s) and long-term quality of life.
Figure 1.
Treatment Decision Making and Quality of Life Among Men with Low-Risk Prostate Cancer
Utilizing this model we hypothesized that men's initial preference for active surveillance (vs. active treatment) would be associated with demographic and clinical characteristics (i.e., older age, lower-risk disease characteristics, more comorbid conditions), and with psychological and decisional factors (i.e., lower prostate cancer anxiety, less decisional uncertainty, greater prostate cancer knowledge, and the inclination for a shared decision). Understanding the factors associated with men's treatment preferences, following a physician consultation, provides important information for developing decision support strategies during this critical period. Given the unique methodological strengths of the prospective, pre-treatment assessment, the large sample in an integrated health system, and the comparative effectiveness design, this study addresses critical gaps in our understanding of the patient-related factors that contribute to the overtreatment of low-risk PCa.
Materials and Methods
Participants
We enrolled subjects from Kaiser Permanente Northern California (KPNC) from May 2012 to May 2014. Inclusion criteria were: 1) a new diagnosis of low-risk prostate cancer (defined as stage T2a or less, PSA ≤ 10 ng/mL and Gleason ≤ 6); 2) within 30 days of diagnosis when staff attempted first phone contact; 3) able to provide informed consent; and 4) English speaking. Exclusion criteria were: 1) already started prostate cancer treatment; 2) diagnosis via transurethral resection of the prostate, with no subsequent biopsy; and 3) physician refusal.
Procedures
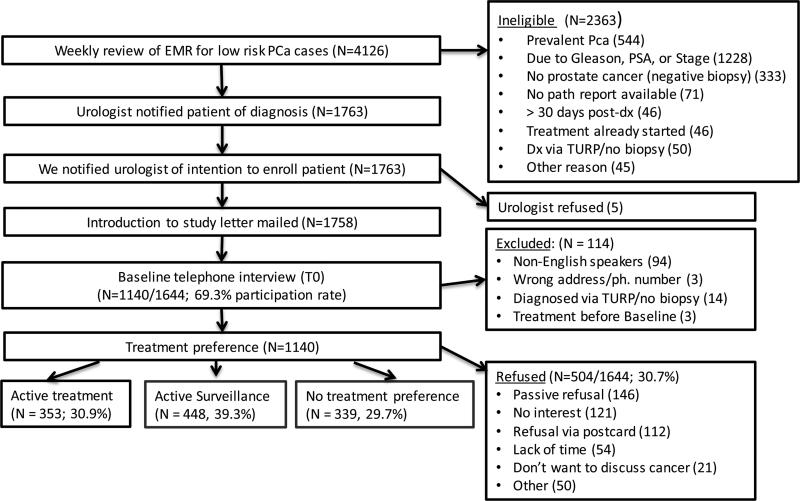
We identified new prostate cancer cases by reviewing electronic medical records (EMRs) of prostate biopsies and pathology reports (Figure 2), and confirmed that a clinician had informed the patient of the diagnosis and that the patient met eligibility criteria. All cases were subsequently linked with the KPNC Cancer Registry to remove prevalent cases. We notified urologists to give them the option of excluding patients for clinical or psychological reasons. We then mailed an invitation letter to eligible men with a return postcard for them to decline further contact. We sought to conduct the baseline telephone assessment within 30 days of the patient being notified of his diagnosis, but called up to 90 days post-notification for difficult to reach patients. The baseline assessment required 30-40 minutes and men received a $20 gift card following completion. We are presently conducting two follow-up telephone assessments, at 6-months and 24-months following the baseline assessment (Figure 1). The study was approved by the Kaiser Foundation Research Institute IRB and the Georgetown University IRB.
Figure 2.
Study Flow Chart
Measures
Demographic and clinical characteristics
We elicited demographic characteristics from participants (Table 1). We abstracted EMR-based clinical information, including diagnosis date, PSA level at diagnosis, clinical stage, Gleason score, number and percent of positive biopsy cores, and comorbid illnesses. We used the Elixhauser Comorbidity Index (41) to calculate a comorbidity score, which is based on the compilation of 30 individual health conditions noted in the EMR, from one year pre-diagnosis to 60 days post-diagnosis of prostate cancer.
Table 1.
Associations of Demographic and Clinical Characteristics with Treatment Preference
| Variable | Categories | All (n=1140) | Active Treatment (AT) (n=353, 31%) | Active Surveillance (AS) (n=448, 39%) | No Preference (NP) (n=339, 30%) | Significance AS / AT / NPa AS and ATb AT and NPc AS and NPd |
|---|---|---|---|---|---|---|
| Age at Diagnosis Mean (SD) |
continuous | 61.46 (7.3) | 60.06 (7.7) | 62.6 (7) | 61.42 (7) | <.0001a |
| <.0001b | ||||||
| 0.015c | ||||||
| 0.019d | ||||||
| Age at Diagnosis (N, %) |
< 60 | 450 (39.5) | 166 (47) | 147 (32.8) | 137 (40.4) | 0.0002a |
| 60-69 | 566 (49.6) | 159 (45) | 236 (52.7) | 171 (50.4) | <.0001b | |
| 0.214c | ||||||
| 70+ | 124 (10.9) | 28 (7.9) | 65 (14.5) | 31 (9.1) | 0.020d | |
| Race (N, %) |
White | 924 (81.1) | 284 (80.5) | 371 (82.8) | 269 (79.4) | 0.218a |
| Black | 140 (12.3) | 49 (13.9) | 43 (9.6) | 48 (14.2) | 0.112b | |
| 0.892c | ||||||
| Other | 76 (6.7) | 20 (5.7) | 34 (7.6) | 22 (6.5) | 0.130d | |
| Ethnicity (N, %) |
Hispanic | 123 (10.9) | 47 (13.4) | 48 (10.8) | 28 (8.3) | 0.093a |
| 0.264b | ||||||
| 0.029c | ||||||
| 0.228d | ||||||
| Marital Status (N, %) |
Married | 927 (81.4) | 297 (84.1) | 358 (80.1) | 272 (80.2) | 0.279a |
| 0.140b | ||||||
| 0.180c | ||||||
| 0.959d | ||||||
| Education (N, %) |
≤ High School | 219 (19.3) | 83 (23.5) | 87 (19.6) | 49 (14.5) | 0.004a |
| 1-3 Yrs College | 357 (31.4) | 117 (33.1) | 144 (32.4) | 96 (28.4) | 0.102b | |
| 4 Year College | 266 (23.4) | 83 (23.5) | 94 (21.1) | 89 (26.3) | 0.001c | |
| ≥ Grad School | 294 (25.9) | 70 (19.8) | 120 (27) | 104 (30.8) | 0.072d | |
| Employment (N, %) |
Employed | 675 (59.7) | 200 (56.7) | 255 (58.1) | 220 (65.1) | 0.052a |
| 0.686b | ||||||
| 0.023c | ||||||
| 0.047d | ||||||
| Income (N, %) |
</= to $75,000 | 377 (35.6) | 127 (37.9) | 151 (36.6) | 99 (31.8) | 0.289a |
| $75,001-125,000 | 356 (33.6) | 104 (31) | 146 (35.4) | 106 (34.1) | 0.435b | |
| 0.270c | ||||||
| $125,001+ | 326 (30.8) | 104 (31) | 116 (28.1) | 106 (34.1) | 0.191d | |
| Days from diagnosis to Baseline Survey (Mean, SD) Median |
continuous | 28.84 (14.8) | 28.25 (13.9) | 30.58 (15.9) | 27.16 (14.0) | 0.004a |
| 0.031b | ||||||
| 0.301c | ||||||
| 24 | 24 | 25.5 | 23 | 0.002d | ||
| PSA at Baseline (N, %) |
<= 4 | 126 (11.1) | 46 (13) | 38 (8.5) | 42 (12.4) | 0.130a |
| 4.1 to 7.9 | 832 (73) | 247 (70) | 345 (77) | 240 (70.8) | 0.050b | |
| 0.963c | ||||||
| 8.0 to 10.0 | 182 (16) | 60 (17) | 65 (14.5) | 57 (16.8) | 0.102d | |
| Gleason at Baseline (N, %) |
<= 5 | 5 (0.4) | 0 (0) | 4 (0.9) | 1 (0.3) | 0.147a |
| 6 | 1135 (99.6) | 353 (100) | 444 (99.1) | 338 (99.7) | 0.075b | |
| 0.307c | ||||||
| 0.296d | ||||||
| Number of positive cores Mean (SD) |
continuous | 2.67 (2.13) | 3.39 (2.57) | 1.93 (1.38) | 2.91 (2.13) | <.0001a |
| <.0001b | ||||||
| 0.007c | ||||||
| <.0001d | ||||||
| Elixhauser Index (N, %) |
0 | 384 (33.7) | 125 (35.4) | 148 (33) | 111 (32.7) | 0.206a |
| 1 | 314 (27.5) | 91 (25.8) | 123 (27.5) | 100 (29.5) | 0.628b | |
| 2 | 185 (16.2) | 57 (16.1) | 63 (14.1) | 65 (19.2) | 0.318c | |
| 3+ | 257 (22.5) | 80 (22.7) | 114 (25.4) | 63 (18.6) | 0.059d | |
| Treatment Discussion with Urologist (N, %) |
Yes | 997 (87.5) | 343 (97.2) | 410 (91.5) | 244 (72) | <.0001a |
| 0.0008b | ||||||
| <.0001c | ||||||
| <.0001d | ||||||
| Treatment Discussion with Radiation Oncologist (N, %) |
Yes | 213 (18.7) | 105 (29.8) | 44 (9.8) | 64 (18.9) | <.0001a |
| <.0001b | ||||||
| 0.001c | ||||||
| 0.0003d | ||||||
| Treatment Discussion with primary care physician (N, %) | Yes | 108 (9.5) | 39 (11) | 44 (9.8) | 25 (7.4) | 0.244a |
| 0.572b | ||||||
| 0.095c | ||||||
| 0.229d | ||||||
| Treatment Recommendation from Urologist* (N, %) | No rec/Pt Decide | 469 (47.1) | 181 (52.8) | 148 (36.2) | 140 (57.4) | <.0001a |
| AS | 253 (25.4) | 11 (3.2) | 214 (52.3) | 28 (11.5) | <.0001b | |
| AT | 274 (27.5) | 151 (44) | 47 (11.5) | 76 (31.1) | <.0001c | |
| Missing (did not see urologist) | 144 | 10 | 39 | 95 | <.0001d | |
| First Degree Relative with prostate cancer (N, %) |
Yes | 332 (29.1) | 130 (36.8) | 114 (25.4) | 88 (26) | 0.001a |
| 0.001b | ||||||
| 0.002c | ||||||
| 0.871d | ||||||
| Prior Cancer (N, %) |
Yes | 80 (7) | 31 (8.8) | 28 (6.3) | 21 (6.2) | 0.295a |
| 0.173b | ||||||
| 0.197c | ||||||
| 0.975d | ||||||
We have not presented the treatment recommendations given by radiation oncologists given that only a small percentage of participants (18.7%) had seen a radiation oncologist prior to the baseline assessment.
Men's Treatment Preference
We assessed whether men preferred a particular treatment option by asking, Have you decided on which treatment or management option you will choose? (yes/no), and if ‘yes,’ we asked: What is the treatment or management option? We listed each potential option, eliciting a ‘yes’ or ‘no’ for each. These included: a) a monitoring strategy such as active surveillance, watchful waiting, or expectant management; b) surgery (radical prostatectomy), c) external beam radiation therapy, d) brachytherapy (seeds), or e) hormone therapy (no one endorsed hormone therapy). For these analyses, we collapsed men preferring surgery or either form of radiation into the ‘active treatment’ group. Those who did not yet have a treatment preference were categorized in the ‘no preference’ group.
Urologist's Treatment Recommendation
We assessed men's self-report of their urologist's treatment recommendation, which was classified as: active surveillance, active treatment (surgery or radiation therapy), or don't know/no recommendation was made. For men who had not yet had an appointment with their urologist (N = 144), we included this group as a fourth category in this variable in order to maintain the full sample size for analyses. We also assessed men's self-report of their radiation oncologist's treatment recommendation, but as only 18.7% had seen a radiation oncologist prior to the baseline interview, we do not describe this variable further.
Educational Resources Used for Treatment Decision-Making
We assessed the educational resources (Table 2) patients reported using to learn about treatment options and whether each resource was helpful (‘not at all,’ ‘somewhat,’ or ‘very helpful’). The number of resources used was summed for a total score.
Table 2.
Associations of Educational Resources Used for Treatment Decisions with Treatment Preference
| Variable Have you... |
Categories | All (n=1140) | Active Treatment (AT) (n=353, 31%) | Active Surveillance (AS) (n=448, 39%) | No Preference (NP) (n=339, 30%) | Significance AS / AT /NP a AS and AT b AT and NP c AS and NP d |
|---|---|---|---|---|---|---|
| Attended a KPNC class about prostate cancer treatment? (N, %) |
Yes | 228 (20.2) | 102 (29) | 53 (12) | 73 (21.7) | <.0001 a |
| <.0001 b | ||||||
| 0.029 c | ||||||
| 0.0003 d | ||||||
|
*If YES: How helpful was it? (N, %) |
Not at all/somewhat | 39 (17.1) | 16 (15.7) | 10 (18.9) | 13 (17.8) | 0.867 a |
| 0.615 b | ||||||
| Very | 189 (82.9) | 86 (84.3) | 43 (81.1) | 60 (82.2) | 0.710 c | |
| 0.879 d | ||||||
| Had an additional discussion with a nurse? N, % |
Yes | 94 (8.3) | 43 (12.3) | 31 (7) | 20 (6) | 0.005 a |
| 0.013 b | ||||||
| 0.004 c | ||||||
| 0.543 d | ||||||
|
*If YES: How helpful was it? (N, %) |
Not at all/somewhat | 28 (29.8) | 11 (25.6) | 10 (32.3) | 7 (35) | 0.700 a |
| 0.530 b | ||||||
| Very | 66 (70.2) | 32 (74.4) | 21 (67.7) | 13 (65) | 0.441 c | |
| 0.839 d | ||||||
| Received a second opinion from a doctor? N, % |
Yes | 241 (21.4) | 107 (30.5) | 72 (16.4) | 62 (18.5) | <.0001 a |
| <.0001 b | ||||||
| 0.0003 c | ||||||
| 0.446 d | ||||||
|
*If YES: How helpful was it? (N, %) |
Not at all/somewhat | 35 (14.6) | 15 (14) | 11 (15.5) | 9 (14.5) | 0.963 a |
| 0.785b | ||||||
| Very | 205 (85.4) | 92 (86) | 60 (84.5) | 53 (85.5) | 0.929 c | |
| 0.875 d | ||||||
| Had discussion(s) with prostate cancer patients who have been treated? N, % |
Yes | 596 (52.9) | 222 (63.1) | 214 (48.7) | 160 (47.6) | <.0001 a |
| <.0001 b | ||||||
| <.0001 c | ||||||
| 0.756 d | ||||||
|
*If YES: How helpful was it? (N, %) |
Not at all/somewhat | 209 (35.1) | 58 (26.2) | 88 (41.1) | 63 (39.4) | 0.002 a |
| 0.001 b | ||||||
| Very | 386 (64.9) | 163 (73.8) | 126 (58.9) | 97 (60.6) | 0.007 c | |
| 0.733 d | ||||||
| Read any booklets about prostate cancer treatment? N, % |
Yes | 721 (64) | 231 (65.6) | 281 (64) | 209 (62.2) | 0.646 a |
| 0.636 b | ||||||
| 0.350 c | ||||||
| 0.605 d | ||||||
|
*If YES: How helpful was it? (N, %) |
Not at all/somewhat | 235 (32.7) | 63 (27.4) | 104 (37.1) | 68 (32.5) | 0.065 a |
| 0.020 b | ||||||
| Very | 484 (67.3) | 167 (72.6) | 176 (62.9) | 141 (67.5) | 0.239 c | |
| 0.291 d | ||||||
| Watched any DVDs or videos about prostate cancer treatment? (N, %) |
Yes | 201 (17.8) | 66 (18.7) | 76 (17.3) | 59 (17.6) | 0.870 a |
| 0.614 b | ||||||
| 0.699 c | ||||||
| 0.928 d | ||||||
|
*lf YES: How helpful was it? (N, %) |
Not at all/somewhat | 56 (27.9) | 28 (42.4) | 14 (18.4) | 14 (23.7) | 0.004 a |
| 0.002 b | ||||||
| Very | 145 (72.1) | 38 (57.6) | 62 (81.6) | 45 (76.3) | 0.027 c | |
| 0.451 d | ||||||
| Visited any websites about prostate cancer treatment? (N, %) |
Yes | 806 (71.5) | 256 (72.5) | 312 (71.2) | 238 (70.6) | 0.851 a |
| 0.689 b | ||||||
| 0.581 c | ||||||
| 0.853 d | ||||||
|
*If YES: How helpful was it? (N, %) |
Not at all/somewhat | 310 (38.5) | 100 (39.1) | 106 (34) | 104 (43.7) | 0.066 a |
| 0.210 b | ||||||
| Very | 496 (61.5) | 156 (60.9) | 206 (66) | 134 (56.3) | 0.296 c | |
| 0.020 d | ||||||
| Total number of educational resources used (N, %) |
0/1 | 251 (22.2) | 49 (13.9) | 115 (26.1) | 87 (25.7) | <.0001a |
| 2 | 318 (28.1) | 90 (25.5) | 136 (30.9) | 92 (27.2) | <.0001b | |
| 3 | 304 (26.9) | 102 (28.9) | 110 (25) | 92 (27.2) | <.0001c | |
| 4+ | 258 (22.8) | 112 (31.7) | 79 (18) | 67 (19.8) | 0.655d | |
Percentages may not add up to 100 due to missing values
Decisional, Psychological, and Knowledge Variables
We used the SURE Test (42), a 4-item version of the Decisional Conflict Scale (43), to measure decisional certainty (Cronbach's alpha = 0.71; Table 3). Sample items include: ‘Do you feel sure about the best choice for you?’ and, ‘Do you know the risks and benefits of each option?’ Response categories are ‘yes’ and ‘no.’ We assessed prostate cancer-related anxiety with five items from the Cancer Control Subscale of the Health Worry Scale (Cronbach's alpha = 0.76) (44). Sample items include, ‘I worry about what my doctor will find next’ and ‘I am confident that my cancer can be kept under control’ (reverse coded). The 5-point response categories range from ‘not at all’ to ‘very much.’ We assessed men's preference for making a shared treatment decision with the Degner Control Preference Scale (45). Because only 21 men (1.8%) selected either of the two ‘doctor-dependent’ categories, we included only three preference categories for decision-making: a shared decision, an independent decision after considering the doctor's opinion, and an independent decision.
Table 3.
Associations of Psychological and Decision-Making Variables with Treatment Preference
| Variable | Categories | All (n=1140) | Active Treatment (AT) (n=353, 31%) | Active Surveillance (AS) (n=448, 39%) | No Preference (NP) (n=339, 30%) | Significance AS/ AT/NP a AS and AT b AT and NP c AS and NP d |
|---|---|---|---|---|---|---|
|
Knowledge Scale Mean (SD) (higher = more knowledge) |
continuous | 3.51 (0.84) | 3.39 (0.84) | 3.61 (0.81) | 3.49 (0.86) | 0.001 a |
| 0.0002 b | ||||||
| 0.123 c | ||||||
| 0.048 d | ||||||
|
SURE scale: Certainty of treatment preference (higher = more certain) Mean (SD) |
continuous | 3.17 (1.18) | 3.76 (0.63) | 3.54 (0.87) | 2.07 (1.23) | <.0001 a |
| <.0001 b | ||||||
| <.0001 c | ||||||
| <.0001 d | ||||||
|
Health Worry Scale: Prostate-related anxiety (higher = more anxious) Mean (SD) |
continuous | 10.62 (4.44) | 10.17 (4.4) | 10.77 (4.39) | 10.91(4.52) | 0.060 a |
| 0.054 b | ||||||
| 0.029 c | ||||||
| 0.663 d | ||||||
| Degner Control Preference Scale (N, %) | Prefers shared decision | 243 (21.7) | 37 (10.5) | 115 (26.3) | 91 (27.6) | <.0001 a |
| Prefers to make decision after considering doctor's opinion | 718 (64.2) | 237 (67.5) | 262 (60) | 219 (66.4) | <.0001 b | |
| <.0001 c | ||||||
| Prefers to make an independent decision | 157 (14) | 77 (21.9) | 60 (13.7) | 20 (6.1) | 0.003 d | |
|
Aware of low risk status
(N, %) |
Incorrect/don't know | 117 (10.4) | 52 (14.9) | 25 (5.7) | 40 (12) | <.0001 a |
| <.0001 b | ||||||
| Correct (low risk) | 1004 (89.6) | 296 (85.1) | 416 (94.3) | 292 (88) | 0.270 c | |
| 0.002 d | ||||||
|
Numeracy (N, %) |
0 correct | 221 (20.5) | 63 (18.3) | 90 (21.5) | 68 (21.4) | 0.409 a |
| 1 correct | 337 (31.2) | 119 (34.6) | 129 (30.9) | 89 (28) | 0.409 b 0.173 c |
|
| 2 correct | 522 (48.3) | 162 (47.1) | 199 (47.6) | 161 (50.6) | 0.659 d | |
* Percentages may not add up to 100 due to missing values
Based on our previously developed scales, we included 5 items to assess participants’ knowledge of the natural history of prostate cancer (e.g., ‘Most men diagnosed with prostate cancer die of something other than prostate cancer’), the treatment side effects (e.g., ‘ Loss of sexual function is a common side effect of prostate cancer treatment.’), and the treatment options for low-risk prostate cancer (‘Men with low-risk prostate cancer can choose to be monitored closely by their doctors, rather than receive surgery or radiation.’) (46-48). Response choices were ‘true,’ ‘false,’ or ‘don't know,’ with ‘don't know’ scored as incorrect. Correct items were summed to form a total score, with a higher score indicating greater knowledge. Internal consistency was low (Cronbach's α = .36), most likely due to using only 5 items to assess multiple aspects of the disease. As we were interested in the relationship of the entire scale to the outcome, we did not assess the association of individual items with treatment preference. We also assessed men's knowledge of their prostate cancer risk category with a single item (‘low,’ ‘intermediate,’ ‘high,’ or ‘don't know’). Responses of ‘low-risk’ were counted as correct. Finally, we assessed men's understanding of numerical concepts (numeracy) concerning disease risk using two multiple choice items (49) on percentages and fractions (e.g., ‘Which of the following numbers represent the biggest risk of getting a disease?’ Response options include: ‘1 in 100,’ ‘1 in 1000,’ or ‘1 in 10.’). The total score ranged from 0 (neither correct) to 2 (both correct).
Statistical Analysis
We assessed differences between the three treatment preference groups (active surveillance, active treatment, or no preference) across demographic and clinical characteristics using two-sided chi square (χ2) tests and ANOVAs (Table 1). Further, we assessed group differences on the educational resources used (Table 2), as well as on decision-making, psychological, and knowledge variables (Table 3). We conducted two multinomial logistic regression models (Table 4), with treatment preference as the outcome, to evaluate 1) the associations of demographics, clinical variables, and urologist's treatment recommendation with treatment preference, and 2) whether educational resources, knowledge, decisional, and psychological factors were independently associated with treatment preference (active surveillance vs. active treatment, and no preference vs. active treatment), controlling for the demographic, clinical, and urologist's treatment recommendation variables. We included all demographic and clinical variables that had a bivariate association (p< 0.10) with treatment preference; race, Elixhauser Index, and baseline PSA were also included. For each of the continuous variables, we reported ORs and 95% CIs for a one standard deviation increase. Group differences were evaluated using Wald tests from the multinomial logistic regression models (p≤0.05).
Table 4.
Multinomial Logistic Regression Models for Treatment Preference
| Model 1-Demographic and clinical variables (n=1136) | Model 2 - All Variables (n=1090)# | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| AS vs AT (ref) | No Preference vs AT (ref) | Overall p value | AS vs AT (ref) | No Preference vs AT (ref) | Overall p value | |||||
| OR | 95% CI | OR | 95% CI | OR | 95% CI | OR | 95% CI | |||
| Age | ||||||||||
| less than 60 (ref) | 1 | 1 | 1 | 1 | ||||||
| 60-69 | 1.49 | 1.02 - 2.19 | 1.17 | 0.82 - 1.68 | 1.64 | 1.07 - 2.51 | 1.34 | 0.84 - 2.14 | ||
| 70+ | 1.79 | 0.94 - 3.39 | 1.05 | 0.55 - 2 | 2 | 0.95 - 4.18 | 1.02 | 0.43 - 2.42 | ||
| Education | * | |||||||||
| < High School (ref) | 1 | 1 | 1 | 1 | ||||||
| 1-3 Years College | 1.13 | 0.7 - 1.84 | 1.26 | 0.78 - 2.03 | 1.37 | 0.8 - 2.34 | 1.63 | 0.85 - 3.15 | ||
| 4 Year College | 1.29 | 0.77 - 2.17 | 1.73 | 1.05 - 2.85 | 1.61 | 0.9 - 2.89 | 2.19 | 1.1 - 4.36 | ||
| > Grad School | 1.71 | 1.02 - 2.87 | 2.29 | 1.38 - 3.8 | 2.05 | 1.12 - 3.74 | 2.84 | 1.42 - 5.69 | ||
| Race | ||||||||||
| Black(ref) | 1 | 1 | 1 | 1 | ||||||
| White | 1.44 | 0.83 - 2.51 | 0.85 | 0.52 - 1.4 | 1.28 | 0.71 - 2.3 | 0.71 | 0.38 - 1.33 | ||
| Other | 2.1 | 0.92 - 4.79 | 0.99 | 0.45 - 2.18 | 1.59 | 0.64 - 3.94 | 0.68 | 0.24 - 1.91 | ||
| Elixhauser Index | ||||||||||
| 0 (ref) | 1 | 1 | 1 | 1 | ||||||
| 1 | 0.9 | 0.58 - 1.4 | 1.15 | 0.76 - 1.75 | 0.87 | 0.54 - 1.4 | 1.09 | 0.64 - 1.86 | ||
| 2 | 0.65 | 0.38 - 1.12 | 1.13 | 0.69 - 1.83 | 0.7 | 0.39 - 1.27 | 1.36 | 0.73 - 2.55 | ||
| 3+ | 1.14 | 0.71 - 1.82 | 0.84 | 0.53 - 1.35 | 1.19 | 0.7 - 2.01 | 0.72 | 0.38 - 1.35 | ||
| PSA at diagnosis | ||||||||||
| <= 4 (ref) | 1 | 1 | 1 | 1 | ||||||
| 4.1 to 7.9 | 1.32 | 0.74 - 2.37 | 0.87 | 0.52 - 1.45 | 1.14 | 0.6 - 2.15 | 0.88 | 0.44 - 1.76 | ||
| 8.0 to 10.0 | 0.79 | 0.39 - 1.61 | 0.73 | 0.38 - 1.38 | 0.56 | 0.26 - 1.23 | 0.75 | 0.32 - 1.72 | ||
| Days from Diagnosis to Baseline | ||||||||||
| (one-unit increase = 1 SD) | 1.18 | 0.99 - 1.4 | 1.03 | 0.87 - 1.23 | 1.19 | 0.98 - 1.44 | 0.97 | 0.77 - 1.21 | ||
| Number of positive cores | 0.73 | 0.66 - 0.81 | 0.96 | 0.9 - 1.03 | *** | 0.74 | 0.67 - 0.82 | 0.95 | 0.86 - 1.04 | *** |
| First Degree relative with prostate cancer | *** | ** | ||||||||
| No (ref) | 1 | 1 | 1 | 1 | ||||||
| Yes | 0.61 | 0.42 - 0.88 | 0.55 | 0.38 - 0.78 | 0.58 | 0.38 - 0.87 | 0.56 | 0.35 - 0.88 | ||
| Urologist Recommendation | *** | *** | ||||||||
| No recommendation/Patient Decide (ref) | ||||||||||
| AS/WW | 20.02 | 10.36 - 38.7 | 3.15 | 1.5 - 6.61 | 17.89 | 8.98 - 35.66 | 2.58 | 1.09 - 6.08 | ||
| AT | 0.43 | 0.29 - 0.66 | 0.64 | 0.45 - 0.93 | 0.41 | 0.26 - 0.65 | 0.56 | 0.34 - 0.92 | ||
| no discussion with urologist | 4.58 | 2.17 - 9.69 | 12.27 | 6.08 - 24.76 | 3.38 | 1.51 - 7.57 | 5.62 | 2.41 - 13.11 | ||
| # of decision making resources used | *** | |||||||||
| 0/1 (ref) | -- | -- | -- | -- | 1 | 1 | ||||
| 2 | -- | -- | -- | -- | 0.64 | 0.36 - 1.14 | 0.81 | 0.41 - 1.6 | ||
| 3 | -- | -- | -- | -- | 0.39 | 0.21 - 0.71 | 1.01 | 0.5 - 2 | ||
| 4+ | -- | -- | -- | -- | 0.28 | 0.15 - 0.52 | 0.81 | 0.4 - 1.66 | ||
| Sure Scale (certainty about treatment preference) | *** | |||||||||
| (one-unit increase = 1 SD) | -- | -- | -- | -- | 0.57 | 0.41 - 0.8 | 0.13 | 0.09 - 0.17 | ||
| Health Worry Scale (prostate cancer anxiety) | ||||||||||
| (one-unit increase = 1 SD) | -- | -- | -- | -- | 1.22 | 1.003 - 1.48 | 1.05 | 0.84 - 1.31 | ||
| Degner Control Preference Scale | *** | |||||||||
| Decision with Doctor or Independent (ref) | -- | -- | -- | -- | 1 | 1 | ||||
| Shared decision | -- | -- | -- | -- | 2.34 | 1.37 - 3.99 | 3.09 | 1.73 - 5.52 | ||
| Knowledge Scale | *** | |||||||||
| (one-unit increase = 1 SD) | -- | -- | -- | -- | 1.77 | 1.43 - 2.18 | 1.63 | 1.28 - 2.06 | ||
| Awareness of Low Risk Level | *** | |||||||||
| Incorrect/DK (ref) | -- | -- | -- | -- | 1 | 1 | ||||
| Correct (low risk) | 3.97 | 1.96 - 8.06 | 1.74 | 0.87 - 3.48 | ||||||
Notes: AS (active surveillance), AT (active treatment), NP (no preference).
Model 2 has fewer subjects than Model 1 due to missing data in the psychological and decisional variables included in Model 2.
p value <0.05
p value <0.01
p value <0.001
Using continuous measures of the psychological and decisional predictors, after adjusting for demographic and clinical variables, we have 80% power to detect true ORs of 1.26 (or 0.79 for inverse associations) at a significance level of 0.05, for one standard deviation increase in the continuous predictors for the active surveillance vs. active treatment comparison, and for the no preference vs. active treatment comparison. SAS version 9.3 was used for all analyses.
Results
Participation rate
Of 1644 eligible men, 1140 (69.3%) agreed to participate (Figure 2). Compared to those who declined/could not be reached, participants were more likely to be white (p< 0.0001). There were no significant differences on age, ethnicity, comorbidities, PSA, or Gleason score (data not shown).
Baseline characteristics
Demographic and clinical characteristics are presented in Table 1, with the overall statistical comparison and comparisons between treatment preferences: active treatment (N = 353, 30.9%), active surveillance (N = 448, 39.3%), and no preference (N = 339; 29.7%). Thus, over two-thirds (70.2%) had a treatment preference by the baseline interview, which was conducted a median of 24 days post-diagnosis (interquartile range = 13). Among the active treatment group, 57% (n = 202) preferred surgery, 20.5% (n = 72) preferred external beam radiation, and 22.5% (n = 79) preferred brachytherapy.
In bivariate analyses (Table 1), compared to men preferring active treatment, those preferring active surveillance were older, were less likely to have a first-degree relative with prostate cancer, were interviewed further from the time of diagnosis, had fewer positive cores, and were less likely to have discussed treatment with a urologist or with a radiation oncologist. Compared to men preferring active treatment, men in the no preference group were older, less likely to be Hispanic, more educated, more likely to be employed, were less likely to have a first-degree relative with prostate cancer, had fewer positive cores, and were less likely to have discussed treatment with a urologist or a radiation oncologist. There were 133 men (11.7%) who reported not yet having discussed treatment with a physician by the baseline assessment (data not shown).
Use of Educational Resources (Table 2)
Compared to men preferring active treatment, both the active surveillance and no preference groups were less likely to have used the ‘face-to-face’ educational resources—attending the KPNC educational class, talking to a nurse, getting a second opinion from a doctor, and talking with other prostate cancer patients. We found no group differences on using booklets, DVDs, or the Internet. Overall, the most frequently used resources included the Internet (71.5%), booklets (64%), and talking with other prostate cancer patients (52.9%). The majority of users reported that all resources were ‘very helpful.’ Based on the total number of resources used, the active treatment group used significantly more resources than either of the other two groups (Table 2).
Knowledge, psychological, and decisional factors (Table 3)
Compared to men preferring active treatment, the active surveillance group had greater prostate cancer knowledge and was more likely to correctly report having low-risk prostate cancer. Both the active surveillance and no preference groups had greater decisional uncertainty, greater prostate-specific anxiety, and a greater preference for shared decision-making, compared to active treatment. There were no group differences on the numeracy items, and almost one-half of each group responded correctly to both numeracy items.
Modeling treatment preference (Table 4)
In Model 1 of the multinomial logistic regression analysis, with demographics, clinical factors, and urologist's treatment recommendation as covariates, we found that men expressing a preference for active surveillance (vs. active treatment, reference group) were older (OR=1.49, CI 1.02 - 2.19), more educated (OR=1.71, CI 1.02 - 2.87), had fewer positive cores (OR=0.73, CI 0.66 - 0.81), were less likely to have a first-degree relative with prostate cancer (OR=0.61, CI 0.42 - 0.88), were more likely to have received a recommendation for AS (OR=20.02, CI 10.36 - 38.7) and were less likely to have received a recommendation for AT (OR=0.43, CI 0.29 - 0.66). Next, men in the no preference group (vs. active treatment, reference group), were more educated (OR 2.29, CI 1.38 - 3.8), were less likely to have a first-degree relative with prostate cancer (OR=0.55, CI 0.38 - 0.78). Further, compared to those who had not received any treatment recommendation, men with no preference were more likely to have received a recommendation for AS (OR=3.15, CI 1.5 - 6.61) and less likely to have received a recommendation for AT (OR=0.64, CI 0.45 - 0.93),
In Model 2, in which we added the resources, psychological, and decisional variables, the associations between demographic and clinical factors with treatment preferences were virtually unchanged, with the exception that the association with urologist's treatment recommendation was slightly attenuated (although still significant). We found that men preferring active surveillance (vs. active treatment) reported using fewer resources for decision-making (OR=0.39, CI 0.21 - 0.71), were less certain about their treatment preference (OR=0.57, CI 0.41 - 0.8), had greater PCa-related anxiety (OR=1.22, CI 1.003 - 1.48), and were more likely to prefer to make a shared treatment decision (OR=2.34, CI 1.37 - 3.99), but also had greater prostate cancer knowledge (OR=1.77, CI 1.43 - 2.18) and greater awareness of having a low-risk cancer (OR=3.97, CI 1.96 - 8.06). Comparing the no preference group to the active treatment group, the no preference group was less certain about their treatment preference (OR=0.13, CI 0.09 - 0.17), preferred to make a shared treatment decision (OR=3.09, CI 1.73 - 5.52), and had greater prostate cancer knowledge (OR=1.63, CI 1.28 - 2.06).
Discussion
Men with low-risk prostate cancer face the decision of immediate active treatment vs active surveillance, which includes the option to select curative treatment at a later time. The prostate cancer mortality risk is low with either option. Active treatments are frequently associated with complications that adversely affect quality of life, but men are often uncomfortable with forgoing immediate treatment. Given these tradeoffs, the treatment decision is very sensitive to patient preferences. We are conducting a prospective study to better understand men's decision-making processes for managing low-risk prostate cancer. We found that over two-thirds of men already had a treatment preference by the baseline interview, which occurred in a median of 24-days post-diagnosis. These are rapid decisions, given the indolent nature of the cancer. The proportion of men preferring active surveillance (39.3%) is somewhat greater than in previous reports (8, 12, 15), although our findings are more consistent with recent reports of apparently increasing rates of active surveillance (28, 50-53).
We found that men preferring active surveillance (vs. active treatment) were older, had fewer positive cores, were less likely to have a first degree relative with prostate cancer, and were more likely to understand that their prostate cancer was low-risk. This suggests that, following a physician consultation about treatment, these men interpreted the clinical information to suggest that their cancer was unlikely to require immediate treatment. As has been found in prior studies (34, 54), physician treatment recommendation was significantly associated with men's treatment preferences. In addition, prior studies have shown that physician specialty plays a role in patients’ treatment preferences (34, 55, 56). Due to the low percentage of men who had consulted with physicians other than urologists at this early point in the decision process, we were unable to investigate the association of physician specialty with treatment preference in the current study.
Importantly, after controlling for the urologist's recommendation and the clinical and demographic variables, we found that decisional and psychological factors were independently associated with men's initial treatment preferences. Compared to the active treatment group, the active surveillance group was more knowledgeable about prostate cancer and had more education, but used fewer resources to learn about treatment options, and preferred a shared decision over an independent treatment decision. These results suggest an opportunity to support active surveillance decisions through more physician engagement and by providing educational resources to patients. Contrary to our prediction, men preferring active surveillance (vs. active treatment) reported more prostate cancer-related anxiety and less certainty about their treatment preference. As we cannot determine causality from the available data, further research will be necessary to ascertain whether the active surveillance group's greater anxiety and uncertainty triggered their treatment preference, or whether it was a result of their treatment preference. Also, we speculate that a preference for active treatment may serve to reduce men's anxiety and uncertainty, as this is the more familiar and perhaps more understood choice. Further, the anxiety finding may be due to the fact that 4 of the 5 items on the Health Worry scale concern anxiety regarding disease monitoring and disease progression, which may be more salient for men considering AS than for those considering AT. Although some studies report that men recall that the active surveillance decision was not difficult (57, 58), our data present a somewhat different picture.
Regarding men preferring an active treatment, they may perceive the treatment decision as more straightforward. Alternatively, they may not be fully considering the decision, making this group an appropriate target for early decision support, to communicate that the treatment decision is not urgent (59). Men who did not yet have a treatment preference appeared more similar to the active surveillance group in terms of education, family history, knowledge of cancer, uncertainty about their decision, and preference for making a shared decision.
Regardless of treatment preference, the most frequently reported educational resources used were printed booklets and the Internet (60). These resources are likely the easiest to access and the most familiar to patients. However, it was the in-person resources of a second opinion with a doctor and the KPNC class that were more likely to be rated as ‘very’ helpful. Notably, men preferring active treatment were significantly more likely to utilize resources overall, especially the face-to-face resources, including the class, discussions with nurses, doctors, and other prostate cancer patients. We cannot tell whether different resources provided different treatment messages, or whether men with different treatment inclinations sought different resources. These are important areas for further study.
The study limitations include the under-representation of non-white participants in the sample, although race was not associated with treatment preference. Secondly, although the knowledge scale was associated with treatment preference, it had low internal consistency. A more comprehensive scale with greater internal consistency might provide a more nuanced interpretation of the relationship between knowledge and treatment preferences. Finally, men's initial treatment preferences may change, particularly among those selecting active surveillance, as they have the option of selecting active treatment later. Despite these limitations, this study contributes to an understanding of the factors that play a role in men's early treatment decision-making. This period is a crucial point for ultimately providing decision support, as this is when men are gathering information, forming their views about treatment, and using educational resources.
Strengths of this study include using the electronic medical records of an integrated health care system to rapidly identify and contact a large patient sample shortly following diagnosis. Integrated health systems are growing in the U.S., making this an increasingly important clinical setting to study, as it facilitates assessments of treatment decision-making in the absence of physicians’ financial incentives. Another strength is assessing factors associated with men's initial treatment preferences, which provides important information for developing decision support tools for assisting men in making informed treatment decisions. Finally, although several smaller studies have assessed treatment decisions among men choosing active surveillance, our study's novel comparative effectiveness framework has implications for improving decision-making and quality of life for all treatment modalities.
In sum, although a substantial proportion of men preferred active surveillance during the early stages of decision-making, this was associated with increased anxiety and uncertainty compared to men who preferred active treatment, suggesting that this is not an easy choice. Increasing the appropriate use of active surveillance among men with low-risk prostate cancer may require that men receive additional reassurance and information almost immediately post-diagnosis, while the decision-making is ongoing. Our future studies will address the predictors of the final treatment decision, of remaining on active surveillance, and the long-term quality of life associated with the treatment decision. We plan to use this information to develop decision support strategies to help men understand all management options, reduce the anxiety associated with the decision, and ultimately, address the overtreatment of low-risk prostate cancer. Existing prostate cancer treatment decision tools do not specifically address issues relevant to low-risk prostate cancer, and none have found an impact on the treatment decision, suggesting that additional work is needed in this area (61).
Acknowledgements
The authors gratefully acknowledge the men who participated in the study, the KPNC research staff (Socorro Caglia, Carol Rabello, and Erica Kerezsi), and Susan Marx, for her administrative assistance.
Financial Support: NCI R01 CA 155578-01 (K.L. Taylor and S. Van Den Eeden, Multiple PIs)
Footnotes
The authors have no conflicts of interest to disclose.
References
- 1.American Cancer Society [Internet] Cancer Facts & Figures 2016. American Cancer Society; Atlanta: 2016. [2016 Apr 10]. Available from: http://www.cancer.org/acs/groups/content/@research/documents/document/acspc-047079.pdf. [Google Scholar]
- 2.Draisma G, Etzioni R, Tsodikov A, Mariotto A, Wever E, Gulati R, et al. Lead time and overdiagnosis in prostate-specific antigen screening: importance of methods and context. J Natl Cancer Inst. 2009;101:374–83. doi: 10.1093/jnci/djp001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Epstein MM, Edgren G, Rider JR, Mucci LA, Adami HO. Temporal trends in cause of death among Swedish and US men with prostate cancer. J Natl Cancer Inst. 2012;104:1335–42. doi: 10.1093/jnci/djs299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Etzioni R, Penson DF, Legler JM, di TD, Boer R, Gann PH, et al. Overdiagnosis due to prostate-specific antigen screening: lessons from U.S. prostate cancer incidence trends. J Natl Cancer Inst. 2002;94:981–90. doi: 10.1093/jnci/94.13.981. [DOI] [PubMed] [Google Scholar]
- 5.Sakr WA, Grignon DJ, Crissman JD, Heilbrun LK, Cassin BJ, Pontes JJ, et al. High grade prostatic intraepithelial neoplasia (HGPIN) and prostatic adenocarcinoma between the ages of 20-69: an autopsy study of 249 cases. In Vivo. 1994;8:439–43. [PubMed] [Google Scholar]
- 6.Yao SL, Lu-Yao G. Understanding and appreciating overdiagnosis in the PSA era. J Natl Cancer Inst. 2002;94:958–60. doi: 10.1093/jnci/94.13.958. [DOI] [PubMed] [Google Scholar]
- 7.Chamie K, Williams SB, Hu JC. Population-based assessment of determining treatments for prostate cancer. JAMA Oncol. 2015;1:60–7. doi: 10.1001/jamaoncol.2014.192. [DOI] [PubMed] [Google Scholar]
- 8.Cooperberg MR, Broering JM, Carroll PR. Time trends and local variation in primary treatment of localized prostate cancer. J Clin Oncol. 2010;28:1117–23. doi: 10.1200/JCO.2009.26.0133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Filson CP, Schroeck FR, Ye Z, Wei JT, Hollenbeck BK, Miller DC. Variation in use of active surveillance among men undergoing expectant treatment for early stage prostate cancer. J Urol. 2014;192:75–81. doi: 10.1016/j.juro.2014.01.105. [DOI] [PubMed] [Google Scholar]
- 10.Hoffman KE, Niu J, Shen Y, Jiang J, Davis JW, Kim J, et al. Physician variation in management of low-risk prostate cancer: a population-based cohort study. JAMA Intern Med. 2014;174:1450–9. doi: 10.1001/jamainternmed.2014.3021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jacobs BL, Zhang Y, Schroeck FR, Skolarus TA, Wei JT, Montie JE, et al. Use of advanced treatment technologies among men at low risk of dying from prostate cancer. JAMA. 2013;309:2587–95. doi: 10.1001/jama.2013.6882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jang TL, Bekelman JE, Liu Y, Bach PB, Basch EM, Elkin EB, et al. Physician visits prior to treatment for clinically localized prostate cancer. Arch Intern Med. 2010;170:440–50. doi: 10.1001/archinternmed.2010.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mishra MV, Shen X, Den RB, Champ CE, Trabulsi EJ, Lallas CD, et al. Patterns of care for elderly men diagnosed with favorable-risk prostate cancer from 2004 to 2008: a population-based analysis. Am J Clin Oncol. 2013;36:606–11. doi: 10.1097/COC.0b013e318261056c. [DOI] [PubMed] [Google Scholar]
- 14.Shao YH, Albertsen PC, Roberts CB, Lin Y, Mehta AR, Stein MN, et al. Risk profiles and treatment patterns among men diagnosed as having prostate cancer and a prostate-specific antigen level below 4.0 ng/ml. Arch Intern Med. 2010;170:1256–61. doi: 10.1001/archinternmed.2010.221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shappley WV, III, Kenfield SA, Kasperzyk JL, Qiu W, Stampfer MJ, Sanda MG, et al. Prospective study of determinants and outcomes of deferred treatment or watchful waiting among men with prostate cancer in a nationwide cohort. J Clin Oncol. 2009;27:4980–5. doi: 10.1200/JCO.2008.21.2613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huang GJ, Sadetsky N, Penson DF. Health related quality of life for men treated for localized prostate cancer with long-term followup. J Urol. 2010;183:2206–12. doi: 10.1016/j.juro.2010.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Miller DC, Sanda MG, Dunn RL, Montie JE, Pimentel H, Sandler HM, et al. Long-term outcomes among localized prostate cancer survivors: health-related quality-of-life changes after radical prostatectomy, external radiation, and brachytherapy. J Clin Oncol. 2005;23:2772–80. doi: 10.1200/JCO.2005.07.116. [DOI] [PubMed] [Google Scholar]
- 18.Mols F, Korfage IJ, Vingerhoets AJ, Kil PJ, Coebergh JW, Essink-Bot ML, et al. Bowel, urinary, and sexual problems among long-term prostate cancer survivors: a population-based study. Int J Radiat Oncol Biol Phys. 2009;73:30–8. doi: 10.1016/j.ijrobp.2008.04.004. [DOI] [PubMed] [Google Scholar]
- 19.Sanda MG, Dunn RL, Michalski J, Sandler HM, Northouse L, Hembroff L, et al. Quality of life and satisfaction with outcome among prostate-cancer survivors. N Engl J Med. 2008;358:1250–61. doi: 10.1056/NEJMoa074311. [DOI] [PubMed] [Google Scholar]
- 20.Taylor KL, Luta G, Miller AB, Church TR, Kelly SP, Muenz LR, et al. Long-term disease-specific functioning among prostate cancer survivors and noncancer controls in the prostate, lung, colorectal, and ovarian cancer screening trial. J Clin Oncol. 2012;30:2768–75. doi: 10.1200/JCO.2011.41.2767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ganz PA, Barry JM, Burke W, Col NF, Corso PS, Dodson E, et al. National Institutes of Health State-of-the-Science Conference: role of active surveillance in the management of men with localized prostate cancer. Ann Intern Med. 2012;156:591–5. doi: 10.7326/0003-4819-156-8-201204170-00401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dall'Era MA, Albertsen PC, Bangma C, Carroll PR, Carter HB, Cooperberg MR, et al. Active surveillance for prostate cancer: a systematic review of the literature. Eur Urol. 2012;62:976–83. doi: 10.1016/j.eururo.2012.05.072. [DOI] [PubMed] [Google Scholar]
- 23.Eggener SE, Mueller A, Berglund RK, Ayyathurai R, Soloway C, Soloway MS, et al. A multi-institutional evaluation of active surveillance for low risk prostate cancer. J Urol. 2013;189:S19–S25. doi: 10.1016/j.juro.2012.11.023. [DOI] [PubMed] [Google Scholar]
- 24.Klotz L, Zhang L, Lam A, Nam R, Mamedov A, Loblaw A. Clinical results of long-term follow-up of a large, active surveillance cohort with localized prostate cancer. J Clin Oncol. 2010;28:126–31. doi: 10.1200/JCO.2009.24.2180. [DOI] [PubMed] [Google Scholar]
- 25.Klotz L, Vesprini D, Sethukavalan P, Jethava V, Zhang L, Jain S, et al. Long-term follow-up of a large active surveillance cohort of patients with prostate cancer. J Clin Oncol. 2015;33:272–7. doi: 10.1200/JCO.2014.55.1192. [DOI] [PubMed] [Google Scholar]
- 26.Tosoian JJ, Mamawala M, Epstein JI, Landis P, Wolf S, Trock BJ, et al. Intermediate and longer-term outcomes from a prospective active-surveillance program for favorable-risk prostate cancer. J Clin Oncol. 2015;33:3379–85. doi: 10.1200/JCO.2015.62.5764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.van den Bergh RC, Roemeling S, Roobol MJ, Aus G, Hugosson J, Rannikko AS, et al. Outcomes of men with screen-detected prostate cancer eligible for active surveillance who were managed expectantly. Eur Urol. 2009;55:1–8. doi: 10.1016/j.eururo.2008.09.007. [DOI] [PubMed] [Google Scholar]
- 28.Lane JA, Donovan JL, Davis M, Walsh E, Dedman D, Down L, et al. Active monitoring, radical prostatectomy, or radiotherapy for localised prostate cancer: study design and diagnostic and baseline results of the ProtecT randomised phase 3 trial. Lancet Oncol. 2014;15:1109–18. doi: 10.1016/S1470-2045(14)70361-4. [DOI] [PubMed] [Google Scholar]
- 29.American Urological Association [Internet] Guideline for the Management of Clinically Localized Prostate Cancer. American Urological Association Education and Research, Inc.; Linthicum (MD): 2007. [2015 Sep 28]. 2007. Available from: http://www.auanet.org/education/guidelines/prostate-cancer.cfm. [Google Scholar]
- 30.National Comprehensive Cancer Network [Internet] NCCN Clinical Practice Guidelines in Oncology: Prostate Cancer. Version 2.2014. NCCN; Fort Washington (PA): 2014. [2015 Sep 28]. Available from: http://www.tri-kobe.org/nccn/guideline/urological/english/prostate.pdf. [Google Scholar]
- 31.National Institutes of Health [Internet] NIH State-of-the-Science Conference: Role of Active Surveillance in the Management of Men with Localized Prostate Cancer. NIH; Bethesda, Maryland. Bethesda (MD): Dec 5-7, 2011. [2015 Sep 28]. 2011. Available from: http://consensus.nih.gov/2011/prostatefinalstatement.htm. [Google Scholar]
- 32.Carter HB. Active surveillance for prostate cancer: an underutilized opportunity for reducing harm. J Natl Cancer Inst Monogr. 2012;2012:175–83. doi: 10.1093/jncimonographs/lgs036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chapple A, Ziebland S, Herxheimer A, McPherson A, Shepperd S, Miller R. Is ‘watchful waiting’ a real choice for men with prostate cancer? A qualitative study. BJU Int. 2002;90:257–64. doi: 10.1046/j.1464-410x.2002.02846.x. [DOI] [PubMed] [Google Scholar]
- 34.Davison BJ, Breckon E. Factors influencing treatment decision making and information preferences of prostate cancer patients on active surveillance. Patient Educ Couns. 2012;87:369–74. doi: 10.1016/j.pec.2011.11.009. [DOI] [PubMed] [Google Scholar]
- 35.van den Bergh RC, Korfage IJ, Bangma CH. Psychological aspects of active surveillance. Curr Opin Urol. 2012;22:237–42. doi: 10.1097/MOU.0b013e328351dcb1. [DOI] [PubMed] [Google Scholar]
- 36.Xu J, Neale AV, Dailey RK, Eggly S, Schwartz KL. Patient perspective on watchful waiting/active surveillance for localized prostate cancer. J Am Board Fam Med. 2012;25:763–70. doi: 10.3122/jabfm.2012.06.120128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ritchie A, Marbury D, Verdon DR, Mazzolini C, Boyles S. Shifting reimbursement models: The risks and rewards for primary care. [2015 Sep 28];Medical Economics [Internet] 2014 Apr 8; [about 13 p.]. Available from: http://medicaleconomics.modernmedicine.com/medical-economics/content/tags/aca/shifting-reimbursement-models-risks-and-rewards-primary-care?page=full.
- 38.U.S.Department of Health and Human Services [Internet] HHS.gov. Health Care. U.S. Department of Health and Human Services; Washington: [2016 Apr 10]. Available from: http://www.hhs.gov/healthcare/ [Google Scholar]
- 39.U.S.Census Bureau [Internet] Statistical Abstract of the United States: 2012. Health and Nutrition. U.S. Census Bureau; Washington: 2012. [2015 Sep 28]. Available from: https://www.census.gov/library/publications/2011/compendia/statab/131ed/health-nutrition.html. [Google Scholar]
- 40.Zafar SY, Alexander SC, Weinfurt KP, Schulman KA, Abernethy AP. Decision making and quality of life in the treatment of cancer: a review. Support Care Cancer. 2009;17:117–27. doi: 10.1007/s00520-008-0505-2. [DOI] [PubMed] [Google Scholar]
- 41.Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care. 1998;36:8–27. doi: 10.1097/00005650-199801000-00004. [DOI] [PubMed] [Google Scholar]
- 42.Legare F, Kearing S, Clay K, Gagnon S, D'Amours D, Rousseau M, et al. Are you SURE?: Assessing patient decisional conflict with a 4-item screening test. Can Fam Physician. 2010;56:e308–e314. [PMC free article] [PubMed] [Google Scholar]
- 43.O'Connor AM. User Manual - Decisional Conflict Scale. [Internet] Ottawa Hospital Research Institute; Ottawa: 1993. [2015 Sep 28]. [updated 2010]. 16 p. Available from: http://decisionaid.ohri.ca/docs/develop/User_Manuals/UM_Decisional_Conflict.pdf. [Google Scholar]
- 44.Clark JA, Inui TS, Silliman RA, Bokhour BG, Krasnow SH, Robinson RA, et al. Patients' perceptions of quality of life after treatment for early prostate cancer. J Clin Oncol. 2003;21:3777–84. doi: 10.1200/JCO.2003.02.115. [DOI] [PubMed] [Google Scholar]
- 45.Degner L. Patient participation in treatment decision making. Axone. 1992;14:13–4. [PubMed] [Google Scholar]
- 46.Taylor KL, Davis JL, III, Turner RO, Johnson L, Schwartz MD, Kerner JF, et al. Educating African American men about the prostate cancer screening dilemma: a randomized intervention. Cancer Epidemiol Biomarkers Prev. 2006;15:2179–88. doi: 10.1158/1055-9965.EPI-05-0417. [DOI] [PubMed] [Google Scholar]
- 47.Taylor KL, Williams RM, Davis K, Luta G, Penek S, Barry S, et al. Decision making in prostate cancer screening using decision aids vs usual care: a randomized clinical trial. JAMA Intern Med. 2013;173:1704–12. doi: 10.1001/jamainternmed.2013.9253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Williams RM, Davis KM, Luta G, Edmond SN, Dorfman CS, Schwartz MD, et al. Fostering informed decisions: a randomized controlled trial assessing the impact of a decision aid among men registered to undergo mass screening for prostate cancer. Patient Educ Couns. 2013;91:329–36. doi: 10.1016/j.pec.2012.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lipkus IM, Samsa G, Rimer BK. General performance on a numeracy scale among highly educated samples. Med Decis Making. 2001;21:37–44. doi: 10.1177/0272989X0102100105. [DOI] [PubMed] [Google Scholar]
- 50.Cooperberg MR, Carroll PR. Trends in management for patients with localized prostate cancer, 1990-2013. JAMA. 2015;314:80–2. doi: 10.1001/jama.2015.6036. [DOI] [PubMed] [Google Scholar]
- 51.Ritch CR, Graves AJ, Keegan KA, Ni S, Bassett JC, Chang SS, et al. Increasing use of observation among men at low risk for prostate cancer mortality. J Urol. 2015;193:801–6. doi: 10.1016/j.juro.2014.08.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Weiner AB, Patel SG, Etzioni R, Eggener SE. National trends in the management of low and intermediate risk prostate cancer in the United States. J Urol. 2015;193:95–102. doi: 10.1016/j.juro.2014.07.111. [DOI] [PubMed] [Google Scholar]
- 53.Womble PR, Montie JE, Ye Z, Linsell SM, Lane BR, Miller DC. Contemporary use of initial active surveillance among men in Michigan with low-risk prostate cancer. Eur Urol. 2015;67:44–50. doi: 10.1016/j.eururo.2014.08.024. [DOI] [PubMed] [Google Scholar]
- 54.Xu J, Dailey RK, Eggly S, Neale AV, Schwartz KL. Men's perspectives on selecting their prostate cancer treatment. J Natl Med Assoc. 2011;103:468–78. doi: 10.1016/s0027-9684(15)30359-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Fowler FJ, Jr., McNaughton CM, Albertsen PC, Zietman A, Elliott DB, Barry MJ. Comparison of recommendations by urologists and radiation oncologists for treatment of clinically localized prostate cancer. JAMA. 2000;283:3217–22. doi: 10.1001/jama.283.24.3217. [DOI] [PubMed] [Google Scholar]
- 56.Underwood W, III, Orom H, Poch M, West BT, Lantz PM, Chang SS, et al. Multiple physician recommendations for prostate cancer treatment: a Pandora's box for patients? Can J Urol. 2010;17:5346–54. [PubMed] [Google Scholar]
- 57.Davison BJ, Oliffe JL, Pickles T, Mroz L. Factors influencing men undertaking active surveillance for the management of low-risk prostate cancer. Oncol Nurs Forum. 2009;36:89–96. doi: 10.1188/09.ONF.89-96. [DOI] [PubMed] [Google Scholar]
- 58.Davison BJ, Goldenberg SL. Patient acceptance of active surveillance as a treatment option for low-risk prostate cancer. BJU Int. 2011;108:1787–93. doi: 10.1111/j.1464-410X.2011.10200.x. [DOI] [PubMed] [Google Scholar]
- 59.Henrikson NB, Ellis WJ, Berry DL. “It's not like I can change my mind later”: reversibility and decision timing in prostate cancer treatment decision-making. Patient Educ Couns. 2009;77:302–7. doi: 10.1016/j.pec.2009.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Silk KJ, Perrault EK, Nazione S, Pace K, Hager P, Springer S. Localized prostate cancer treatment decision-making information online: improving its effectiveness and dissemination for nonprofit and government-supported organizations. J Cancer Educ. 2013;28:709–16. doi: 10.1007/s13187-013-0515-8. [DOI] [PubMed] [Google Scholar]
- 61.Violette PD, Agoritsas T, Alexander P, Riikonen J, Santti H, Agarwal A, et al. Decision aids for localized prostate cancer treatment choice: Systematic review and meta-analysis. CA Cancer J Clin. 2015;65:239–51. doi: 10.3322/caac.21272. [DOI] [PubMed] [Google Scholar]