Abstract
Targeted therapy for metastatic colorectal carcinoma consists of anti-EGFR therapy for patients with RAS/RAF wild-type tumors. However, the response rate remains low, suggesting the presence of alternative drivers possibly also representing potential therapeutic targets. We investigated receptor tyrosine kinase (RTK) alterations and MAP2K1 (MEK1) mutations in a large cohort of colorectal carcinoma patients studied by Memorial Sloan Kettering-Integrated Mutation Profiling of Actionable Cancer Targets and The Cancer Genome Atlas, focusing on amplifications, fusions, and hotspot mutations in RTK genes and MAP2K1. RTK gene amplifications were confirmed with FISH and immunohistochemical (IHC) staining. Among 751 colorectal carcinoma cases with next-generation sequencing data, 7% and 1% of colorectal carcinoma harbored RTK alterations and MAP2K1 hotspot mutations (n = 7), respectively. RTK-altered cases had fewer concurrent RAS/RAF mutations (P = 0.003) than RTK/MAP2K1 wild-type colorectal carcinoma. MAP2K1-mutated colorectal carcinoma showed no RAS/RAF mutations. ERBB2 (n = 32) and EGFR (n = 13) were the most frequently altered RTKs, both activated by amplification and/or hotspot mutations. Three RTK fusions were identified: NCOA4-RET, ERBB2-GRB7, and ETV6-NTRK3. Only 1 of 6 patients with an RTK or MAP2K1 alteration who received anti-EGFR and/or anti-ERBB2 therapy demonstrated stable disease; the rest progressed immediately. Overall, RTK alterations and MAP2K1 mutations occur in approximately 8% of colorectal carcinoma. In spite of the usual absence of RAS/RAF mutations, response to anti-EGFR and/or anti-ERBB2 therapy was poor in this limited group. Larger studies are warranted to further define these kinase alterations as novel therapeutic targets in colorectal carcinoma and as negative predictors of response to anti-EGFR therapy.
Introduction
Approximately 137,000 people are diagnosed with colorectal carcinoma, and about 50,000 die from the disease in the United States annually. Despite early-stage interventions, such as polypectomy and surgery, many patients develop metastatic disease requiring systemic therapy. To date, the only FDA-approved biomarker-driven targeted therapies for advanced colorectal carcinoma include anti-EGFR therapy such as cetuximab and panitumumab for RAS wild-type malignancies. Interestingly, DNA-level alterations activating EGFR are very rare in colorectal carcinoma and EGFR expression by IHC does not predict benefit from anti-EGFR therapy (1). The frequency and impact of other potentially targetable receptor tyrosine kinase (RTK) alterations and downstream MAP2K1 mutations have not been thoroughly studied in advanced colorectal carcinoma. Here, we report the incidence and clinicopathologic characteristics of kinase alterations in colorectal carcinoma, including many novel to colorectal carcinoma.
Methods
Case selection Cases were selected on the basis of RTK and MAP2K1 alterations known or presumed to be activating such as amplification, missense mutations at hotspots that have been previously characterized, and previously described in frame fusion events that preserve the kinase domain.
Patients with advanced colorectal carcinoma receiving treatment at Memorial Sloan Kettering Cancer Center from January 2014 through May 2015 were consented for molecular testing with Memorial Sloan Kettering-Integrated Mutation Profiling of Actionable Cancer Targets (MSK-IMPACT), a hybridization capture-based next-generation sequencing (NGS) assay that detects somatic single nucleotide mutations and small indels, copy number alterations, and select structural rearrangements in 341 cancer-related genes (2, 3). At least 50 ng of formalin-fixed paraffin-embedded tumor samples with greater than 10% tumor content and their matched blood normal were required for testing for MSK-IMPACT. A second subset of cases with molecular analysis were identified through the cBioPortal (www.cbioportal.org) by analysis of the provisional colorectal carcinoma dataset from The Cancer Genome Atlas (TCGA) for MAP2K1 missense and RTK alterations (4). To expand the set of MAP2K1-mutated colorectal carcinoma, a third subset of colorectal carcinoma consecutively submitted for clinical molecular testing to guide anti-EGFR antibody therapy at MSKCC from 2009 to 2013 that was analyzed with the MassARRAY system (Sequenom) that included MAP2K1 p. Q56P, K57N, or D67N mutations in an eight-gene panel was included (5). Missense mutations in hotspots including KRAS c.34, 35, 37 38, 181, 182, 183, 351, and 437; NRAS c. 34, 35, 37, 38, 181, 182, and 183; BRAF c. 1781, 1798 and 1799; PIK3CA c. 263, 1624, 1633, and 3140; and AKT1 c.49 were recorded for these MAP2K1-mutated cases detected by Sequenom genotyping.
Histopathologic classification
World Health Organization definitions were used to classify histopathology: mucinous histology was defined as ≥50% of tumor volume comprised of extracellular mucin. Moderately differentiated colorectal carcinoma was defined as 50% to 95% of tumor volume comprised of glands. Poorly differentiated colorectal carcinoma was defined as <50% of tumor volume forming glands (6).
IHC and FISH
Immunohistochemical staining for EGFR and ERBB2 were performed to investigate protein expression for RTK amplifications on full sections of tumor that underwent MSK-IMPACT analysis. FISH was performed to confirm amplifications and fusions detected via MSK-IMPACT.
Microsatellite instability/mismatch repair protein analysis
In RTK-altered cases for which mismatch repair protein expression for MLH1, PMS2, MSH2, and MSH6 had not yet been performed, a C++ program called MSI-sensor (7) was used to analyze MSK-IMPACT sequencing data for microsatellite stable versus microsatellite instability–high (MSI-H) status.
Statistical analysis
Statistical analyses were performed with χ2 for 2 × 2 categorical analyses. Two-tailed P values less than 0.05 are reported in Table 1 and two-tailed P values less than 0.01 (to account for multiple hypothesis testing) are reported in the text.
Table 1.
Clinicopathologic characteristics of sequenced colorectal carcinoma cases
| RTK/MAP2K1 wild type (n = 229 of 694)a |
RTK Altered (n = 52: 26 TCGA+ 26 MSK-IMPACT) |
MAP2K1 Mutated (n = 10: 3 Sequenom, 3 MSK-IMPACT, 4 TCGA) |
|
|---|---|---|---|
| Age (mean, median) | 56, 57 | 61, 62 | 55, 57 |
| Sex (M:F) | 125:104 | 34:18 | 8:2 |
| KRAS/NRAS/BRAF Mutation | 137 (60%) | 19 (37%) (P = 0.003) | 0 (P = 0.0002) |
| APC/CTNNB1 Mutation | 178 (78%) | 33 (63%) | 5 of 7 tested (71%) |
| TP53 Mutation | 134 (59%) | 39 (75%) | 6 of 7 tested (86%) |
| PIK3CA/ PTEN alteration | 61 (27%) | 4 (8%) (P = 0.003) | 1 of 7 tested (14%) |
| Primary site (proximal, distal) | 84 (37%), 145 (63%) | 20 (38%), 32 (62%) | 3 (30%), 7 (70%) |
| MMR-Deficient/MSI-H | 12 (11%) of 114 tested | 1 (4%) of 25 tested | 0 of 6 tested |
| 20q Gain | 28 (12%) | 8 (15%) | 0 of 7 tested |
| Mucinous differentiation | 5 (2%) | 4 (8%) | 0 |
| Poor differentiation | 17 (7%) | 10 (19%) | 2 (20%) |
NOTE: P values <0.01 were considered significant and are listed in bold.
TCGA RTK/MAP2K1 wild-type cases were excluded from wild-type clinicopathologic analysis due to limited data.
Results
Mutation screening
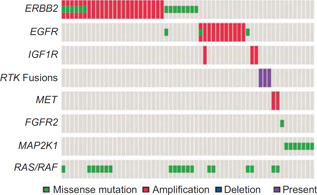
Genomic data from 255 MSK-IMPACT cases and 496 cases from the TCGA were reviewed for RTK alterations and hotspot MAP2K1 mutations (total n = 751). To identify additional MAP2K1-mutated cases, we analyzed data from additional 2631 cases screened for MAP2K1 hotspot mutations as part of a previous clinical mass spectrometry–based genotyping assay. In these additional 2,631 cases, there were 3 MAP2K1-mutated colorectal carcinoma cases. Among the 751 cases that underwent broad NGS screening (MSK-IMPACT and TCGA cases), 52 RTK alterations and 7 MAP2K1 hotspot mutations were identified (7% and 1% of colorectal carcinoma, respectively). RTK alteration and MAP2K1 mutation frequencies and case profiles are detailed in Fig. 1 and Supplementary Table S1, respectively. The most frequent alterations occurred as ERBB2 hotspot missense mutations and amplifications. Alterations novel to colorectal carcinoma were also identified in the MSK-IMPACT cohort and included EGFR hotspot mutations common in lung cancer (p. L858R and p. L861Q), an FGFR2-activating mutation p. R203C described in breast cancer (8), as well as fusions previously described in other entities (Fig. 2).
Figure 1.
Distribution of RTK alterations and MAP2K1 hotspot mutations in colorectal carcinoma. Illustrated are cases with RTK or MAP2K1 oncogenic alterations. The most frequently altered genes within the 751 MSK-IMPACT and TCGA patients were ERBB2, EGFR, and MAP2K1. ERBB2 amplification and mutation often cooccurred.
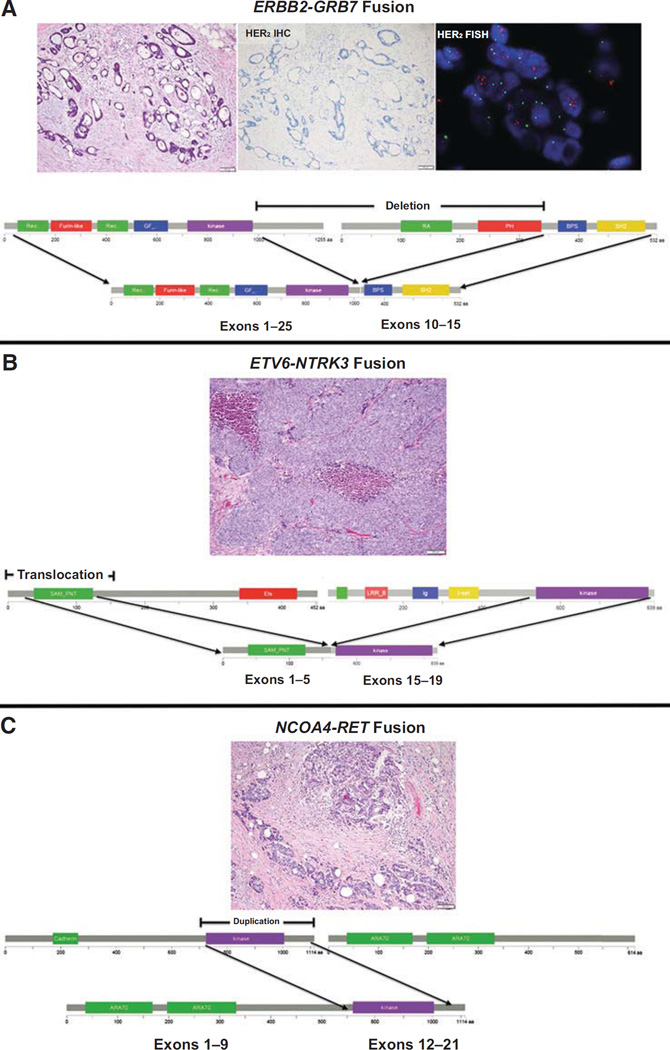
Figure 2.
Fusions detected in colorectal carcinoma. All fusions involved in frame events with the preservation of the kinase domain (depicted in fuschia). The three fusions detected among MSK-IMPACT cases included an ERBB2-GRB7 fusion, resulting in the deletion of the C-terminal of ERBB2 and the N-terminal of GRB7 (A), an ETV6-NTRK3 fusion, resulting in the translocation of the proximal portion of ETV6 to the kinase domain of NTRK3 (B), and an NCOA4-RET duplication (C).
The specimen genotyped was a recurrence or metastasis in 9 MSK-IMPACT cases with RTK alterations, and 3 cases with MAP2K1 mutations. The remaining MSKCC cases and all TCGA cases in which RTK alterations (n = 43) or MAP2K1 (n = 7) mutations were identified were primary tumors. Thus, RTK alterations and MAP2K1 mutations are seen in both primary tumors and metastases, although no paired samples were included.
Three (1%) of MSK-IMPACT cases harbored in-frame, kinase domain–preserving fusions that have been previously identified in other tumor types. These fusions included ETV6-NTRK3, RETNCOA4, and ERBB2-GRB7; and are detailed in Fig. 2. None of these 3 cases harbored RAS/RAF mutations. The allele harboring the ERBB2-GRB7 fusion was also amplified.
Clinicopathologic and molecular characteristics
Clinicopathologic characteristics for MSK-IMPACT patients are summarized in Table 1. RTK-altered colorectal carcinoma had a significantly lower prevalence of activating mutations in KRAS, NRAS, and BRAF than colorectal carcinoma wild type for RTK and MAP2K1 alterations (P = 0.003). PIK3CA mutations/amplification and PTEN mutations/losses were also less common in RTK-altered colorectal carcinoma in comparison with RTK wild-type colorectal carcinoma (P = 0.003). None of the 10 colorectal carcinoma (n = 3 Sequenom, n = 3 MSK-IMPACT, n = 4 TCGA) with MAP2K1 hotspot mutations harbored concurrent RAS/RAF mutations (P = 0.0002). There were no significant differences in mean and median age, sex, primary site in colon, or mismatch repair protein expression between patients with a MAP2K1 or RTK alteration and patients without a MAP2K1 or RTK alteration.
IHC and FISH results
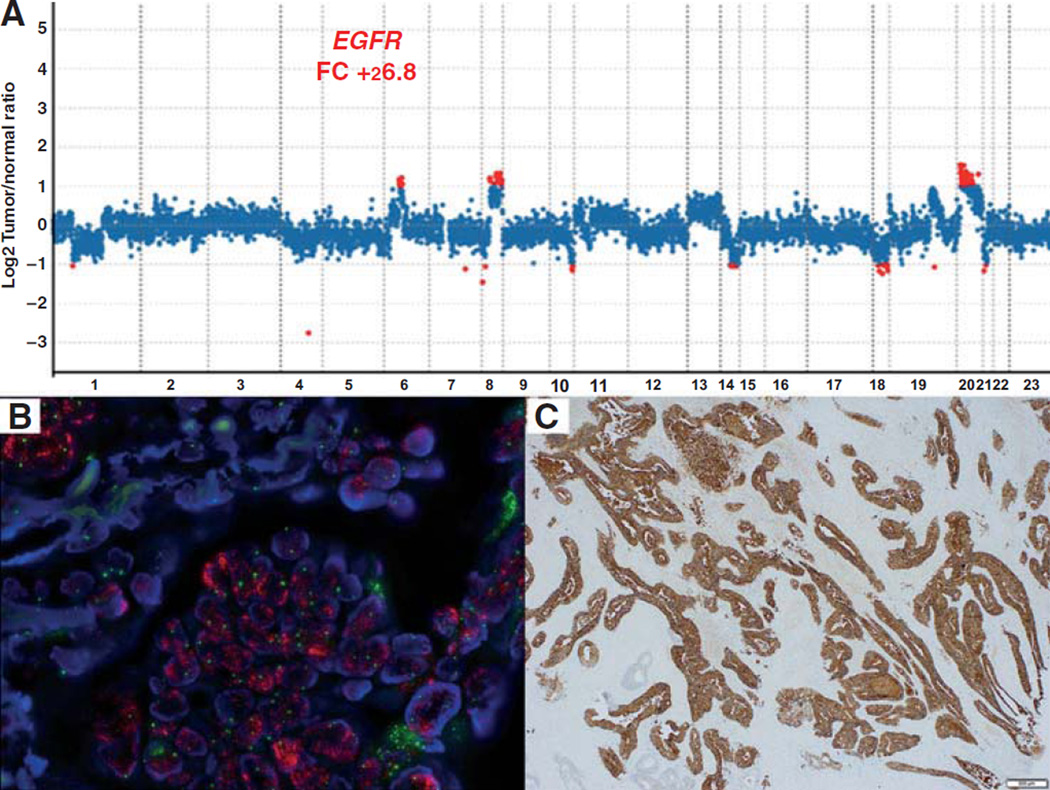
Using tumor tissue available from patients who underwent MSK-IMPACT testing, all amplifications of EGFR or ERBB2 called by deep sequencing of these tumors were confirmed with IHC and FISH testing. The results of IHC and FISH studies are detailed in Table 2. All cases with EGFR or ERBB2 amplification detected by MSK-IMPACT showed amplification by FISH. Of 6 cases with EGFR amplification by MSK-IMPACT and FISH, all showed strong (3+) membranous staining in a percentage of tumor cells ranging from 20% to 90% (Fig. 3). Of the 6 MSK-IMPACT cases with ERBB2 amplification, all showed moderate (2+) or strong (3+) membranous staining on IHC in 40% to 90% of tumor cells; with the exception of the case harboring an ERBB2-GRB7 fusion with amplification (Fig. 2), likely due to the fact that the ERBB2 antibody used (Ventana, 4B5) binds to an epitope in the Cterminal of the ERBB2 protein, which is excluded from the ERBB2-GRB7 fusion product.
Table 2.
Response to biomarker-targeted therapy in patients with metastatic KRAS/BRAF wild-type, RTK/MAP2K1–altered colorectal carcinomas
| RTK/MAP2K1 Alteration | Targeted therapy | Duration of therapy |
Outcome | Notes |
|---|---|---|---|---|
| ERBB2 amplification (FC = +38.9) | Cetuximab + irinotecan | 5 | Stable disease for 5 months | Previously treated with FOLFIRI without progression |
|
ERBB2 amplification (FC = +8.5), ERBB2 p. V842I |
Irreversible inhibitor of EGFR/ ERBB2 |
2 | RECIST stable disease and mixed response |
|
| Panitumumab + irinotecan | 2 | Progression of disease | Irinotecan naïve | |
|
ERBB2 amplification, ERBB2 p. S310F |
Dual ERBB2/ EGFR inhibitor | 2 | Progression of disease | |
| ERBB2-GRB7 fusion, amplified | Cetuximab + irinotecan | 2 | Progression of disease | |
| Monoclonal anti-ERBB2 + capecitabine |
2 | Progression of disease | ||
| Irreversible inhibitor of EGFR/HER2 |
1.5 | Progression of disease | ||
| MAP2K1 p. K57E | Cetuximab + capecitabine + Avastin |
5 | Progression of disease | |
| Cetuximab + irinotecan | 2 | Progression of disease | Irinotecan naïve | |
| MAP2K1 p. K57N | Panitumumab + irinotecan | 2 | Progression of disease | Previously treated with irinotecan |
Figure 3.
Correlation of NGS copy number plot results, FISH, and IHC. A, EGFR amplification detected via copy number plot on MSK-IMPACT portal. Bottom left, high-level EGFR amplification (red signal) detected on FISH. B, strong membranous immunohistochemical staining is present in the majority of the tumor whereas rare glands are negative for EGFR expression (C).
Responses to targeted therapies
Clinical data were available for patients who underwent MSKIMPACT genotyping. Of the 29 MSK-IMPACT cases with RTK/MAP2K1 alterations identified, 4 patients with ERBB2 altered and 2 patients with MAP2K1--mutated colorectal carcinoma, all without concurrent RAS/RAF mutations, received anti-EGFR and/or anti-ERBB2 therapy. Of these 6 cases, 1 patient with high-level ERBB2-amplified colorectal carcinoma who had previously received irinotecan (FOLFIRI) achieved a period of stable disease on subsequent cetuximab in combination with irinotecan. All other patients experienced progression of disease on anti-EGFR or anti-ERBB2–based therapies (Table 2).
Discussion
In this analysis of a very large series of annotated colorectal carcinoma cases, we find that approximately 8% of colorectal carcinoma harbor activating alterations in RTK genes or MAP2K1, with ERBB2 amplifications and mutations being most frequent. These alternative drivers are enriched in the RAS/RAF wild-type cases. The majority of patients in the limited number of colorectal carcinomas with MAP2K1 mutations or ERBB2 alterations did not benefit from anti-EGFR therapy. The one patient with ERBB2 amplification who achieved stable disease on cetuximab and irinotecan had previously achieved stable disease on an irinotecan-based regimen (FOLFIRI), so the patient may have benefited from the irinotecan. Broad NGS for these alterations in patients with advanced colorectal carcinoma would be useful to identify patients who may demonstrate a suboptimal response to anti-EGFR therapy and who would therefore benefit from consideration of clinical trials with other therapeutic agents or combinations.
Previous studies have reported ERBB2 and EGFR amplifications to occur in 0% to 25% and 1% to 8% of colorectal carcinoma patients (9–11); with a degree of heterogeneity (15% of cases) between amplification status in different lesions (10). Our results are concordant with both general incidence in colorectal carcinoma patients and heterogeneity; however, our heterogeneity was intratumoral and demonstrated by percent of tumor cells staining with IHC.
Recently, ERBB2 and dual EGFR/ERBB2 inhibitors have been found to have modest activity in colorectal carcinoma with a range of activating ERBB2 mutations, yet response and need for single-agent or multiple-agent drugs differed depending on the mutation present (12). Furthermore, the clinical response of these drugs when ERBB2 mutations cooccur with ERBB2 amplification needs to be assessed because half of ERBB2 mutations detected in this study cooccurred with ERBB2 amplification. ERBB2 amplification was the most frequent RTK alteration identified, and this alteration has been associated with resistance to cetuximab (13), yet tumors from patients with advanced colorectal carcinoma are not typically screened for ERBB2 amplification before the start of anti-EGFR therapy.
Prior to this report, kinase domain hotspot mutations in EGFR in colorectal carcinoma have only been described at codon 719 (14). We identified 2 cases with EGFR hotspot mutations commonly seen in lung adenocarcinoma including EGFR p.L858R and EGFR p.L861Q. Neither mutation was subclonal in comparison with tumor percent or variant frequency of other mutations. Interestingly, the EGFR p.L861Q mutation cooccurred with a KRAS p. G13D mutation, in contrast to lung adenocarcinomas where EGFR and KRAS mutations are mutually exclusive (15). These rare EGFR hotspot mutations may be targetable in colorectal carcinoma as they are in lung adenocarcinoma, at least in the absence of downstream mutations that may cause primary resistance to EGFR TKIs. We also identified a hotspot EGFR extracellular domain mutation that occurs in gliomas (16, 17), EGFR p.R222C, in a case with EGFR amplification.
Fusions were relatively rare (1% of MSK-IMPACT cases), although a possible limitation of the study is that only select recurrently rearranged introns are interrogated by the MSKIMPACT assay. All fusions detected in this study occurred in frame with preservation of the kinase domain and in mutual exclusivity of RAS/RAF mutation. The NCOA4-RET fusion detected in this study is similar to the recently described fusion by Le Rolle and colleagues in colorectal carcinoma (18). The ETV6-NTRK3 fusion seen here is the first one described in colorectal carcinoma to our best knowledge. It is similar to those described in mammary analogue secretory carcinoma and congenital infantile fibrosarcoma (19, 20). The ERBB2-GRB7 fusion described here has been described previously with similar structure (21). This fusion has several clinically important characteristics including (i) absence of 4B5 monoclonal ERBB2 staining due to an IHC epitope at the Cterminal, which is deleted in the fusion product, (ii) discordant ERBB2 IHC (negative) and FISH (positive) results in cases with amplification of the fusion, and (iii) resistance to anti-HER2 therapy. We did not detect any ALK or ROS1 rearrangements that have been previously described in colorectal carcinoma (22), which suggests that these fusions may be exceedingly rare.
We also report, to our knowledge for the first time, MAP2K1 hotspot mutations in colorectal carcinoma. Like the recently described cases in lung cancer (23), these rare mutations are mutually exclusive with RAS/RAF mutations, suggesting similar effects on activation of ERK signaling. These mutations were associated with primary resistance to anti-EGFR therapy in both MAP2K1-mutated colorectal carcinoma patients with available treatment response data, further supporting an analogous function to RAS/RAF–activating mutations. The potential use of MAP2K1 mutation as a negative predictor for anti-EGFR therapy is supported by a recent report of an acquired MAP2K1 p. K57N mutation at resistance to anti-EGFR therapy (13).
In summary, RTK alterations and MAP2K1 mutations occur in approximately 8% of colorectal carcinoma, and most frequently occur without comutations in RAS/RAF, suggesting that broad molecular profiling may identify a substantial number of patients eligible for clinical trials. Although our numbers for RTK/MAP2K1–altered colorectal carcinoma patients who received anti- EGFR therapy are limited, our preliminary findings suggest a low rate of response and warrant further studies.
Supplementary Material
Implications.
Targetable kinase alterations were identified in a subset of advanced colorectal carcinoma patients, preferentially associated with wild-type RAS/RAF, and may predict poor response to standard anti-EGFR therapy.
Acknowledgments
Grant Support
The MSK-IMPACT program is supported in part by the Marie-Josée and Henry R. Kravis Center for Molecular Oncology. The MSKCC Sequenom facility was supported by the Anbinder Fund.
J.F. Hechtman is a consultant/advisory board member for Navigant. R.D. Yaeger reports receiving commercial research grant from Genentech, GlaxoSmithKline, and Novartis and is a consultant/advisory board member for GlaxoSmithKline. D.B. Solit is a consultant/advisory board member for Pfizer. M. Ladanyi is a consultant/advisory board member for NCCNBI Afatinib Targeted Therapy Advisory Committee.
Footnotes
Supplementary data for this article are available at Molecular Cancer Research Online (http://mcr.aacrjournals.org/).
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed by the other authors.
Authors' Contributions
Conception and design: J.F. Hechtman, D. Solit, M.E. Arcila, M. Ladanyi Development of methodology: J.F. Hechtman, L. Wang, M.E. Arcila, J. Shia, M. Ladanyi
Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): J.F. Hechtman, A. Zehir, R.D. Yaeger, L. Wang, D.M. Hyman, D.B. Solit, M.E. Arcila, L. Borsu, E. Vakiani, L. Saltz, M. Ladanyi
Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): J.F. Hechtman, A. Zehir, R.D. Yaeger, S. Middha, D.B. Solit, M.E. Arcila, J. Shia, M. Ladanyi
Writing, review, and/or revision of the manuscript: J.F. Hechtman, A. Zehir, R.D. Yaeger, L. Wang, S. Middha, D.M. Hyman, D.B. Solit, M.E. Arcila, J. Shia, E. Vakiani, L.B. Saltz, M. Ladanyi
Administrative, technical, or material support (i.e., reporting or organizing data, constructing databases): J.F. Hechtman, D.M. Hyman, D.B. Solit, M.E. Arcila
Study supervision: J.F. Hechtman, D.B. Solit, M. Ladanyi
Other: T. Zheng
References
- 1.Chung KY, Shia J, Kemeny NE, Shah M, Schwartz GK, Tse A, et al. Cetuximab shows activity in colorectal cancer patients with tumors that do not express the epidermal growth factor receptor by immunohistochemistry. J Clin Oncol. 2005;23:1803–1810. doi: 10.1200/JCO.2005.08.037. [DOI] [PubMed] [Google Scholar]
- 2.Cheng DT, Mitchell TN, Zehir A, Shah RH, Benayed R, Syed A, et al. Memorial Sloan Kettering-integrated Mutation Profiling of Actionable Cancer Targets (MSK-IMPACT): a hybridization capture-based next-generation sequencing clinical assay for solid tumor molecular oncology. J Mol Diagn. 2015;17:251–264. doi: 10.1016/j.jmoldx.2014.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hyman DM, Solit DB, Arcila ME, Cheng D, Sabbatini P, Baselga J, et al. Precision medicine at Memorial Sloan Kettering Cancer Center: clinical next-generation sequencing enabling next-generation targeted therapy trials. Drug Discov Today. 2015;20:1422–1428. doi: 10.1016/j.drudis.2015.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012;2:401–404. doi: 10.1158/2159-8290.CD-12-0095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Arcila M, Lau C, Nafa K, Ladanyi M. Detection of KRAS and BRAF mutations in colorectal carcinoma: roles for high-sensitivity locked nucleic acid-PCR sequencing and broad-spectrum mass spectrometry genotyping. J Mol Diagn. 2011;13:64–73. doi: 10.1016/j.jmoldx.2010.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bosman F, Carneiro F. World Health Organization classification of tumours, pathology and genetics of tumours of the digestive system. Lyon, France: IARC Press; 2010. [Google Scholar]
- 7.Niu B, Ye K, Zhang Q, Lu C, Xie M, McLellan MD, et al. MSIsensor: microsatellite instability detection using paired tumor-normal sequence data. Bioinformatics. 2014;30:1015–1016. doi: 10.1093/bioinformatics/btt755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Reintjes N, Li Y, Becker A, Rohmann E, Schmutzler R, Wollnik B. Activating somatic FGFR2 mutations in breast cancer. PLoS One. 2013;8:e60264. doi: 10.1371/journal.pone.0060264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Al-Kuraya K, Novotny H, Bavi P, Siraj AK, Uddin S, Ezzat A, et al. HER2, TOP2A, CCND1, EGFR and C-MYC oncogene amplification in colorectal cancer. J Clin Pathol. 2007;60:768–772. doi: 10.1136/jcp.2006.038281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lee WS, Park YH, Lee JN, Baek JH, Lee TH, Ha SY. Comparison of HER2 expression between primary colorectal cancer and their corresponding metastases. Cancer Med. 2014;3:674–680. doi: 10.1002/cam4.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ooi A, Takehana T, Li X, Suzuki S, Kunitomo K, Iino H, et al. Protein overexpression and gene amplification of HER-2 and EGFR in colorectal cancers: an immunohistochemical and fluorescent in situ hybridization study. Mod Pathol. 2004;17:895–904. doi: 10.1038/modpathol.3800137. [DOI] [PubMed] [Google Scholar]
- 12.Kavuri SM, Jain N, Galimi F, Cottino F, Leto SM, Migliardi G, et al. HER2 activating mutations are targets for colorectal cancer treatment. Cancer Discov. 2015;5:832–841. doi: 10.1158/2159-8290.CD-14-1211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Siravegna G, Mussolin B, Buscarino M, Corti G, Cassingena A, Crisafulli G, et al. Clonal evolution and resistance to EGFR blockade in the blood of colorectal cancer patients. Nat Med. 2015;21:795–801. doi: 10.1038/nm.3870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barber TD, Vogelstein B, Kinzler KW, Velculescu VE. Somatic mutations of EGFR in colorectal cancers and glioblastomas. N Engl J Med. 2004;351:2883. doi: 10.1056/NEJM200412303512724. [DOI] [PubMed] [Google Scholar]
- 15.Stella GM, Scabini R, Inghilleri S, Cemmi F, Corso S, Pozzi E, et al. EGFR and KRAS mutational profiling in fresh non-small cell lung cancer (NSCLC) cells. J Cancer Res Clin Oncol. 2013;139:1327–1335. doi: 10.1007/s00432-013-1444-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P, et al. An integrated genomic analysis of human glioblastoma multiforme. Science. 2008;321:1807–1812. doi: 10.1126/science.1164382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wu G, Diaz AK, Paugh BS, Rankin SL, Ju B, Li Y, et al. The genomic landscape of diffuse intrinsic pontine glioma and pediatric non-brainstem high-grade glioma. Nat Genet. 2014;46:444–450. doi: 10.1038/ng.2938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Le Rolle AF, Klempner SJ, Garrett CR, Seery T, Sanford EM, Balasubramanian S, et al. Identification and characterization of RET fusions in advanced colorectal cancer. Oncotarget. 2015;6:28929–28937. doi: 10.18632/oncotarget.4325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Skálová A, Vanecek T, Sima R, Laco J, Weinreb I, Perez-Ordonez B, et al. Mammary analogue secretory carcinoma of salivary glands, containing the ETV6-NTRK3 fusion gene: a hitherto undescribed salivary gland tumor entity. Am J Surg Pathol. 2010;34:599–608. doi: 10.1097/PAS.0b013e3181d9efcc. [DOI] [PubMed] [Google Scholar]
- 20.Knezevich SR, McFadden DE, Tao W, Lim JF, Sorensen PH. A novel ETV6-NTRK3 gene fusion in congenital fibrosarcoma. Nat Genet. 1998;18:184–187. doi: 10.1038/ng0298-184. [DOI] [PubMed] [Google Scholar]
- 21.Ross JS, Wang K, Sheehan CE, Boguniewicz AB, Otto G, Downing SR, et al. Relapsed classic E-cadherin (CDH1)-mutated invasive lobular breast cancer shows a high frequency of HER2 (ERBB2) gene mutations. Clin Cancer Res. 2013;19:2668–2676. doi: 10.1158/1078-0432.CCR-13-0295. [DOI] [PubMed] [Google Scholar]
- 22.Aisner DL, Nguyen TT, Paskulin DD, Le AT, Haney J, Schulte N, et al. ROS1 and ALK fusions in colorectal cancer, with evidence of intratumoral heterogeneity for molecular drivers. Mol Cancer Res. 2014;12:111–118. doi: 10.1158/1541-7786.MCR-13-0479-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Arcila ME, Drilon A, Sylvester BE, Lovly CM, Borsu L, Reva B, et al. MAP2K1 (MEK1) mutations define a distinct subset of lung adenocarcinoma associated with smoking. Clin Cancer Res. 2015;21:1935–1943. doi: 10.1158/1078-0432.CCR-14-2124. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.