Abstract
The CRISPR-Cas9 system enables genome editing and somatic cell genetic screens in mammalian cells. We performed genome scale loss-of-function screens in 33 cancer cell lines to identify genes essential for proliferation/survival and found a strong correlation between increased gene copy number and decreased cell viability after genome editing. Within regions of copy number gain, CRISPR-Cas9 targeting of both expressed and unexpressed genes, as well as intergenic loci, led to significantly decreased cell proliferation through induction of a G2 cell cycle arrest. By examining single guide RNAs that map to multiple genomic sites, we found that this cell response to CRISPR-Cas9 editing correlated strongly with the number of target loci. These observations indicate that genome targeting by CRISPR-Cas9 elicits a gene-independent anti-proliferative cell response. This effect has important practical implications for interpretation of CRISPR-Cas9 screening data and confounds the use of this technology for identification of essential genes in amplified regions.
Introduction
Genome engineering using site-specific DNA endonucleases has operationalized functional somatic cell genetics, enabling precise perturbation of both coding and non-coding regions of the genome in cells from a range of different organisms. Zinc-finger nucleases (ZFNs) and transcription activator-like effector nucleases (TALENs) are custom-designed endonucleases that enable site-specific genome editing, but their widespread application has been limited by reagent complexity and cost (1, 2). The bacterial CRISPR-Cas9 (clustered regularly interspaced short palindromic repeats–CRISPR-associated 9) system, which serves as an adaptive immune mechanism, has been shown to serve as a versatile and highly effective technology for genome editing (3–8). CRISPR-Cas9 applications require introduction of two fundamental components into cells: (i) the RNA-guided CRISPR-associated Cas9 nuclease derived from Streptococcus pyogenes and (ii) a single guide RNA (sgRNA) that directs the Cas9 nuclease through complementarity with specific regions of the genome (3, 7–11).
Genome editing occurs through induction of double stranded breaks in DNA by the Cas9 endonuclease in an sgRNA-directed sequence-specific manner. These DNA breaks can be repaired by one of two mechanisms: non-homologous end joining (NHEJ) or homology-directed repair (HDR)(3, 12). CRISPR-Cas9-mediated gene knock-out results from a DNA break being repaired in an error-prone manner through NHEJ and introduction of an insertion/deletion (indel) mutation with subsequent disruption of the translational reading frame (11). Alternatively, HDR-mediated repair in the presence of an exogenously supplied nucleotide template can be utilized to generate specific point mutations or other precise sequence alterations. Furthermore, nuclease-dead versions of Cas9 (dCas9) can also be fused to transcriptional activator or repressor domains to modulate gene expression at specific sites in the genome (13–17). CRISPR-Cas9 technology has been effectively utilized in cultured cells from a myriad of organisms (12), and has also been successfully employed for in vivo modeling in the mouse germline (18, 19) as well as for somatic gene editing to generate novel mouse models of cancer (20–24).
Recent studies have shown that CRISPR-Cas9 can be effectively used for loss-of-function genome scale screening in human and mouse cells (9–11, 25–28). These approaches rely upon lentiviral delivery of the gene encoding the Cas9 nuclease and sgRNAs targeting annotated human or mouse genes. Multiple different CRISPR-Cas9 knock-out screening libraries have been developed, including both single-vector (Cas9 and the sgRNA on the same vector) and dual-vector systems (9, 25, 29). Pooled CRISPR-Cas9 screening is typically performed through massively parallel introduction of sgRNAs targeting all genes into Cas9-expressing cells, with a single sgRNA per cell. Positive- or negative-selection proliferation screens are performed and sgRNA enrichment or depletion is measured by next generation sequencing (9, 10).
To date, only a limited number of genome-scale CRISPR-Cas9 knock-out screens have been reported, and these screens have demonstrated a high rate of target gene validation (9–11, 25–28). Wang et al. recently reported an analysis of cell essential genes using CRISPR-Cas9-mediated loss-of-function screens in four leukemia and lymphoma cell lines (28). Hart et al. also reported identification of core and cell line-specific essential genes in five cancer cell lines of differing lineages (25). This approach has enabled the identification of known oncogene dependencies as well as many novel essential genes and pathways in individual cancer cell lines (25, 28). In addition to knock-out screens, proof-of-concept CRISPR-activator or inhibitor screens using dCas9 and genome-scale sgRNA libraries have also been successfully conducted (30, 31). Moreover, in vivo genome-scale screens with CRISPR-Cas9 have also been performed for cancer-relevant phenotypes (32).
To identify cancer cell vulnerabilities in a genotype- and phenotype-specific manner, we performed genome-scale loss-of-function genetic screens in 33 cancer cell lines representing a diversity of cancer types and genetic contexts of both adult and pediatric lineages (Table S1)(29). When we analyzed essential genes across the entire dataset, we unexpectedly found a robust correlation between apparent gene essentiality and genomic copy number, where the number of CRISPR-Cas9-induced DNA cuts predict the cellular response to genome editing.
Results
High-resolution CRISPR-Cas9 screening in cancer cell lines for gene dependencies
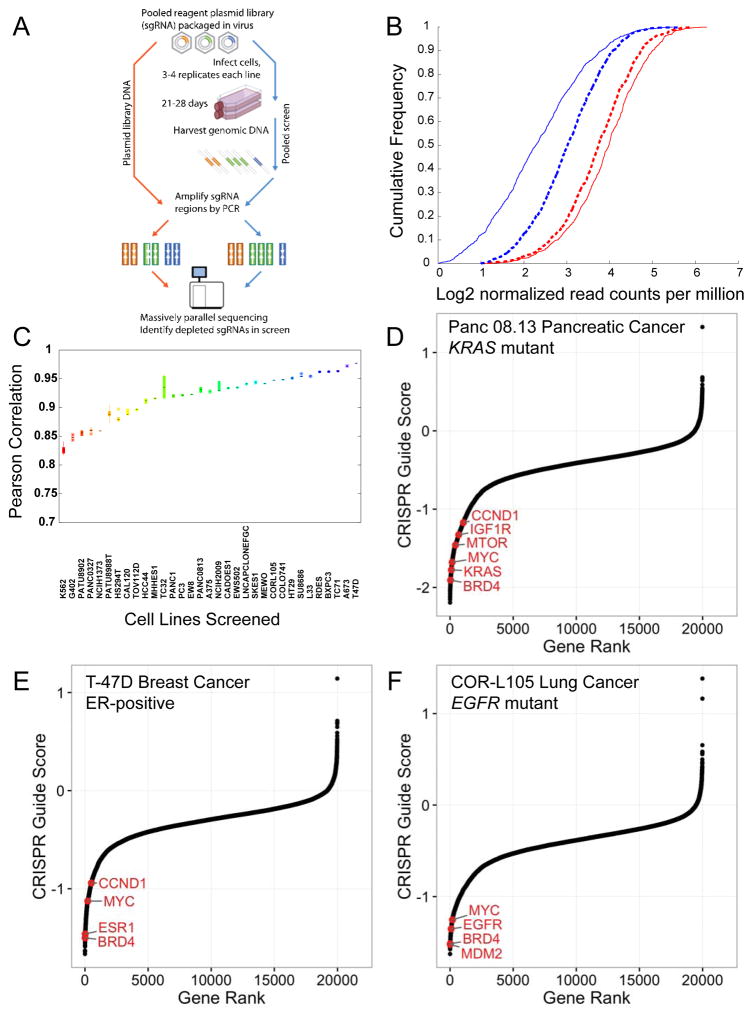
Using the dual-vector GeCKOv2 CRISPR-Cas9 system, we performed genome-scale pooled screening in 33 cancer cell lines representing a wide diversity of adult and pediatric cancer types (Table S1; Fig. 1A). Cancer cell lines were transduced with a lentiviral vector expressing the Cas9 nuclease under blasticidin selection. These stable cell lines were then infected in replicate (n = 3 or 4) at low multiplicity of infection (MOI<1) with a library of 123,411 unique sgRNAs targeting 19,050 genes (6 sgRNAs per gene), 1,864 miRNAs and 1,000 non-targeting negative control sgRNAs (29). Infected cells were purified by selection with puromycin and then passaged with an average representation of 500 cells per sgRNA until an endpoint of 21 or 28 days. At the endpoint, the abundance of sgRNAs in these cells was quantitated from genomic DNA by massively parallel sequencing and compared to the abundance in the plasmid pool used for virus production to define the relative drop-out or enrichment in the screen (Fig. 1A).
Figure 1.
Genome scale loss-of-function CRISPR-Cas9 screening in cancer cell lines. (A) Schematic of the pooled screening process. (B) Cumulative frequency of log2 normalized read counts per million to 1000 non-targeting sgRNA controls (red) and sgRNAs targeting 213 positive control genes (KEGG ribosome, proteasome and spliceosome subsets, Table S2) (blue) in both the initial DNA reference pool (dotted) and 28 d after transduction in the PANC-1 cell line (solid). (C) A boxplot of Pearson correlation between replicates (y-axis) plotted for each cell line (x-axis) shows the range of replicate-replicate correlations after quality control (Methods). (D, E, F) Rank ordered depiction of second-best CRISPR guide scores for each gene in the Panc 08.13 (D), T47D (E) and CORL105 (F) cell lines. Hallmark cancer-relevant oncogene and non-oncogene dependencies are depicted in red for each cell line.
The log2 normalized read counts of the 1000 non-targeting sgRNAs show a slight enrichment in representation from the original plasmid DNA pool, indicating that on average non-targeting guides have no substantial detrimental effect on viability (Fig. 1B). As positive controls, we also compiled a list of 213 putative cell essential genes that are part of the ribosome, proteasome or spliceosome complexes (Table S2). In contrast to the non-targeting negative control guides, the read counts of these positive controls in late time point samples were substantially depleted compared to the initial reference pool (Fig. 1B). Replicate reproducibility after quality control for each cell line was consistently high (Fig. 1C).
We defined a CRISPR-Cas9 guide score for each sgRNA in the screen by first calculating the log2 fold-change in abundance from the screen endpoint compared to the pool of plasmid DNA, followed by subtraction of the median scores of the negative control sgRNAs (see Methods). Hence, in our dataset a guide score of zero equates to the median effect of negative control sgRNAs. Similarly, the second most depleted sgRNA for each gene was used to call a single “second best” CRISPR-Cas9 guide score and therefore allow the representation of gene level dependencies (33). Significant depletions of sgRNAs are denoted by negative CRISPR-Cas9 guide scores and correspond to decreased proliferation/survival after CRISPR-Cas9-mediated gene editing.
To identify genes essential for viability in each cell line across a variety of cancer contexts, we rank ordered genes by second best CRISPR-Cas9 guide score from most negative (most depleted) to positive (not depleted or enriched). For each cell line, we identified key vulnerabilities corresponding to both oncogenic driver lesions as well as non-oncogene dependencies (Fig. 1D–F). For instance, we observed that KRAS, ESR1 and EGFR were essential genes in KRAS mutant (Fig. 1D), estrogen-receptor positive (Fig. 1E) and EGFR mutant cell lines (Fig. 1F), respectively. Moreover, we observed strong dependency on a number of other cancer-relevant genes and therapeutic targets in each cell line, including BRD4, MTOR, IGF1R, CCND1 and MYC (Fig. 1D–F). Thus, our approach to CRISPR-Cas9 screening yields high quality reproducible data that enables identification of cancer gene dependencies across many different cellular contexts.
Genomic copy number variation predicts the response to CRISPR-Cas9 genome targeting independent of target gene expression
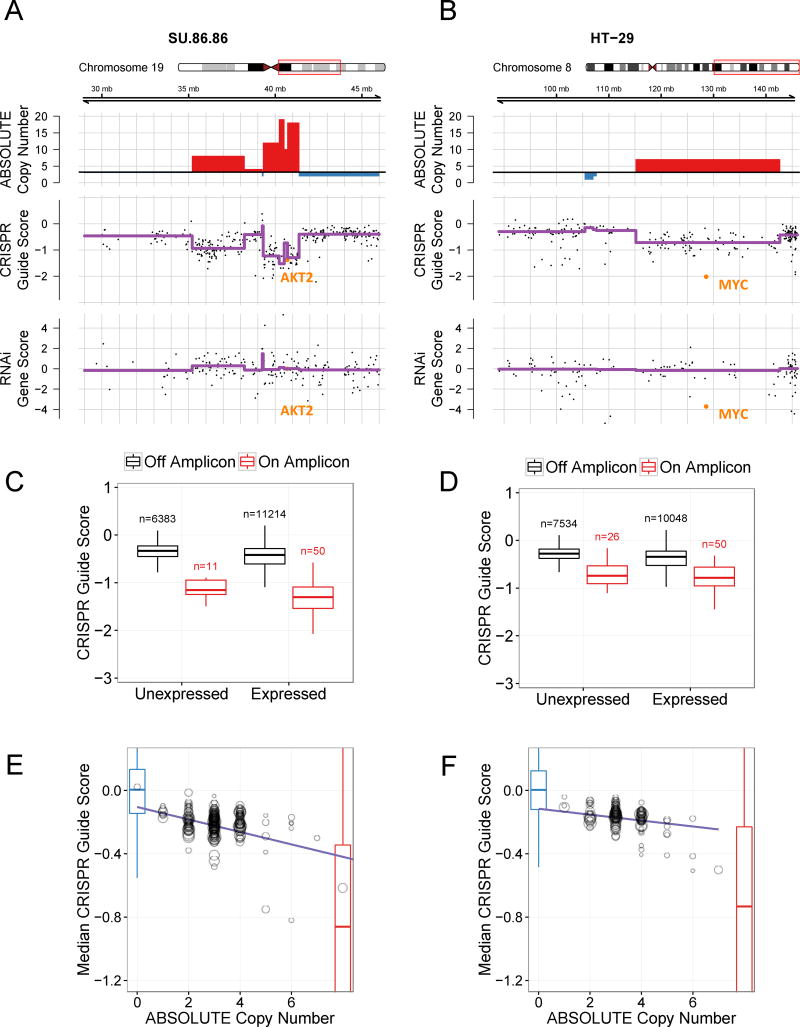
Copy number alterations (CNA’s) are the most common genetic alterations in human epithelial cancers (34) and lead to overexpression of driver oncogenes in cancer. To identify such driver oncogenes responsible for cancer cell proliferation and survival within regions of copy number amplification, we mapped sgRNAs in CRISPR-Cas9 screens of each cell line to genomic coordinates and investigated the relationship of apparent gene essentiality with ABSOLUTE DNA copy number (CN) data available from the Broad Institute-Novartis Cancer Cell Line Encyclopedia (CCLE) (Methods; Table S1) (35, 36). We observed a striking enrichment of negative CRISPR-Cas9 guide scores for genes that reside in copy number amplifications in several cancer cell lines (Fig. 2A–B; Fig. S1A–C). Specifically, CRISPR-Cas9 targeting of genes that reside in amplifications conferred decreased proliferation/survival as compared with targeting genes that mapped outside of these amplifications. As expected, we found that known oncogenes, such as AKT2, MYC, or CDK4, scored as essential in cell lines that harbored amplifications involving these genes. However, we also noted that sgRNAs targeting other genes in these same amplified regions appeared similarly detrimental to cell proliferation or survival (Fig. 2A–B; Fig. S1A–C).
Figure 2.
Genome scale CRISPR-Cas9 screening identifies a strong correlation between copy number and sensitivity to CRISPR-Cas9 genome editing. Two cell lines are shown: SU86.86 (A, C, E) and HT29 (B, D, F). (A) Chromosome 19q amplicon in SU86.86 and (B) chromosome 8q amplicon in HT29: Three tracks are plotted along genomic coordinates within the region defined the red box on the chromosome schematic. Top: ABSOLUTE genomic copy number from Cancer Cell Line Encyclopedia (CCLE) SNP arrays with red indicating copy number gain above average ploidy and blue indicating copy number loss below average ploidy; Middle: CRISPR-Cas9 guide scores plotted according to the 2nd most dependent sgRNA for each gene with purple trend line indicating the mean CRISPR guide score for each copy number segment defined from the above track; Bottom: RNAi gene dependency scores. AKT2 and MYC, known driver oncogenes at these loci, respectively, are highlighted in orange. For RNAi data, shRNAs targeting AKT2 used in Project Achilles were not effective in suppressing AKT2. (C, D) Boxplots of CRISPR guide scores for both expressed and not expressed genes located on (red) or off (black) of the chromosome 19q amplicon in SU86.86 (C) and the chromosome 8q amplicon in HT29 (D). For the SU86.86, the amplicon represented in panel C red box plots ranges from 39.3–41.4 Mb on the corresponding plot in panel A. The number of represented genes is noted above each box plot. (E, F) For each copy-number-defined genomic segment, median CRISPR-Cas9 guide score is plotted against copy number. Each circle represents a single genomic segment of defined copy number for the indicated cell line. The size of the circle corresponds to the number of sgRNAs targeting that segment. Non-targeting negative control sgRNAs are shown with a blue boxplot and known cell essential genes (defined as positive controls) are shown as a red boxplot embedded within the plot.
When we compared these observations to those derived from genome scale RNA interference (RNAi) screens performed in the same cell lines (37), we failed to observe enrichment of apparently essential genes within amplifications and instead identified a small number of genes in each region of copy number gain that scored as essential (Fig. 2A–B; Fig. 1B–C). Moreover, we found that sensitivity to CRISPR-Cas9 targeting within amplified genomic regions was also observed for genes that failed to show significant mRNA expression (Fig. 2C–D; Fig. S1D–F). These observations suggested that the observed dependency of cancer cells to CRISPR-Cas9 targeting of genes resident in amplifications was not the direct consequence of deleting the target gene.
We next sought to determine if this “CRISPR-CN relationship” also extends to lower levels of copy number alterations. For all 33 cancer cell lines screened, we defined genomic segments by their copy number and labeled those segments with their median CRISPR-Cas9 guide score across all sgRNAs targeting within the segment (Fig. 2E–F; Fig. S2). We found a striking correlation between copy number and median CRISPR-Cas9 guide score across even low ranges of copy number alterations. The 1000 “negative control” sgRNAs in the CRISPR-Cas9 library exhibited minimal effects on cell proliferation and viability, and the majority of other data points had lower CRISPR-Cas9 guide scores than the median of these negative controls (Fig. 2E–F, Fig. S2). Strikingly, targeting a locus with an ABSOLUTE copy number of 1, which corresponds to a single CRISPR-Cas9-induced DNA cut, also resulted in reduced proliferation/viability in comparison to the negative controls (Fig. 2E–F, Fig. S2), indicating that even a discrete instance of CRISPR-Cas9 genome modification significantly affects cell proliferation/viability. For each incremental increase in DNA copy number, we observed a progressive decrease in CRISPR-Cas9 guide scores in nearly all of the cell lines that we screened (Fig. 2E–F, Fig. S2). Moreover, we observed this CRISPR-CN correlation among both low-level copy number gains (e.g. 1–2 extra copies), as well as high-level amplifications, and both focal and arm-level copy number alterations (Fig. 2, S1, S2).
Amplified genes rank among the top dependencies in genome scale negative-selection CRISPR-Cas9 screens
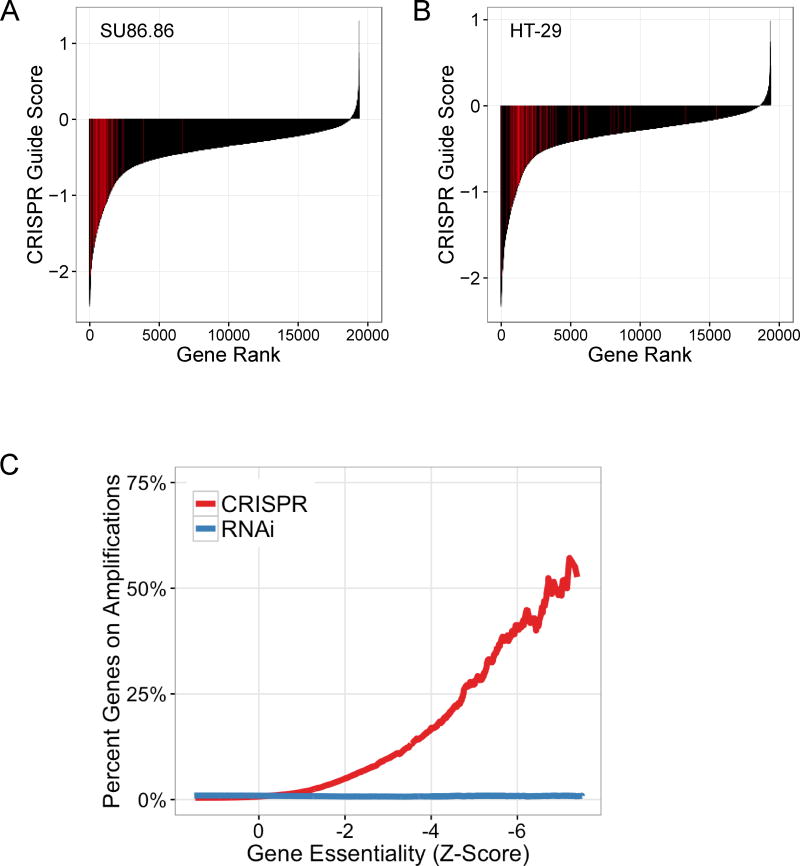
Given the profound impact of genomic copy number on apparent gene essentiality in CRISPR-Cas9 screens as well as the high rate of CNAs in cancer cells, we reasoned that this effect could result in a high false-positive rate for identification of essential genes. To characterize the impact of these false positives on CRISPR-Cas9 screening data, we compared the apparent essentiality of amplified genes with that of all other genes within each of the 33 cell lines. Specifically, we examined CRISPR guide scores for all genes and observed that genes residing in focal high-level amplifications consistently rank among the most highly essential genes identified for each cell line (Fig. 3A–B; Fig. S3A–C).
Figure 3.
Amplified genes represent the strongest perceived dependencies in pooled CRISPR-Cas9 screening data. (A, B) Rank ordered plots showing the second-best CRISPR-Cas9 guide score for each gene in the indicated cell lines. sgRNAs targeting genes within the amplicons represented in Figure 1 are highlighted in red for SU86.86 19q amplicon (A) and HT29 8q amplicon (B). These amplicon-targeting sgRNAs are significantly enriched as apparent dependencies relative to the other sgRNAs targeting genes outside these amplicons (one-sided Kolmogorov–Smirnov test: p = 1.04E-41, A; p = 5.57E-33, B). (C) The cumulative fraction of amplified genes at or below a given dependency score is shown for both CRISPR-Cas9 and RNAi pooled screening datasets. Amplified genes are defined as those genes with a copy number ratio > 2. Gene dependency scores are shown as global Z-scores for both CRISPR-Cas9 and RNAi screening datasets, with Z-scores representing standard deviations from the mean of all genes evaluated in all cell lines screened (CRISPR-Cas9, n = 33 cell lines; RNAi, n = 503 cell lines).
We then performed an aggregate analysis of apparent essentiality due to amplified genes across the entire CRISPR-Cas9 dataset. For this analysis of all genes and all cell lines, we accounted for differences in Cas9 activity/efficacy across cell lines using cell-line specific Z-score normalization (Methods). To investigate relative gene dependencies within the dataset, we calculated composite CRISPR-Cas9 gene scores using the ATARiS algorithm, as previously described (Methods) (38). We next calculated a global Z-score for gene dependency values, representing the number of standard deviations from the mean of the distribution. In parallel, we performed a similar analysis of an available RNAi dataset (Fig. 3C). Thus, this analysis enables a global examination of apparent relative gene dependencies and their relationship to genomic copy number amplification. Strikingly, we observed that increasingly essential genes (lower Z-scores) were more likely to reside on copy number amplifications in CRISPR-Cas9 data but not RNA-interference data (Fig. 3C). For genes with a Z-score of less than or equal to −5, 27.6% (81/294) of those genes reside within a copy number amplification, defined as a copy number ratio (ABSOLUTE/average sample ploidy) greater than two. Thus, copy number amplification is a strong determinant of apparent essentiality in CRISPR-Cas9 screening data, and if not properly accounted for, this CRISPR-CN relationship will likely contribute to a higher false-positive rate for calling gene dependency. When we inspected results from another recently published study that screened five human cancer cell lines with a different CRISPR-Cas9 library (25), we found that gene copy number also predicted essentiality (Fig. S4A–E), thus indicating that the CRISPR-CN correlation occurs independently of the specific sgRNA library used.
The CRISPR-CN relationship is observed across multiple different chromosome structural alterations
To investigate the CRISPR-CN relationship across a spectrum of different chromosomal structural alterations, we performed whole genome sequencing (WGS) on three cell lines harboring copy number gains and amplifications and showing a strong correlation between copy number and CRISPR-Cas9 guide scores (HT29, CAL120, PANC-1). We observed the CRISPR-CN relationship in the context of several different structural amplification patterns, including near arm-level copy number gain (Fig. S5A), simple tandem duplication (Fig. S5A), low-level copy gain from inter-chromosomal translocation (Fig. S5B), and complex amplicon structure involving breakage-fusion-bridge cycles and chromothripsis (Fig. S6). These observations suggest that the CRISPR-CN correlation occurs at both low and high-amplitude copy number changes and does not relate to specific types of chromosomal structural variation.
The response of cells to CRISPR-Cas9 genome targeting correlates with the total predicted number of DNA cuts at target loci
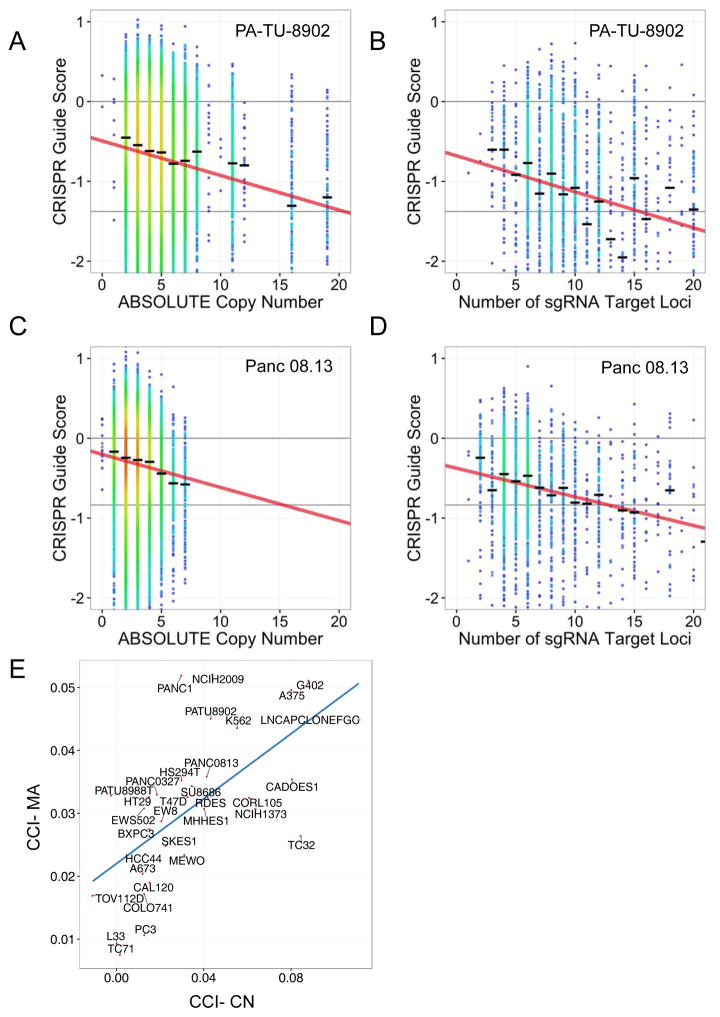
We have demonstrated that there is a gene-independent anti-proliferative effect of CRISPR-Cas9 targeting that occurs with even a single target locus, increases with increasing genomic copy number, and is independent of the type of structural alteration that leads to increased copy number. Thus, we hypothesized that this gene-independent response reflects the total number of CRISPR-Cas9-induced DNA cuts at target loci. The GeCKOv2 library contains 3593 sgRNAs that have multiple perfect match alignments along with a protospacer-adjacent motif (PAM) sequence within the hg19 reference genome. We typically remove these sgRNAs prior to analyzing cancer cell line dependencies. However, these promiscuous sgRNAs provided an opportunity to perform a comparative analysis of the response of cells to CRISPR-Cas9 editing and the relationship to the predicted number of CRISPR-Cas9-induced DNA cuts based on either copy number or number of perfect-match on- and off-target alignments (“multiple alignment analysis”). For the copy number analysis, we used only sgRNAs mapping to a single genomic locus. For the multiple alignment analysis, we reintroduced these multi-targeting sgRNAs and only used sgRNAs targeting non-amplified regions, thus allowing segregation of the impact of CRISPR-Cas9-induced DNA cuts due to copy-number or promiscuous multiple genome alignments.
We observed that sgRNAs that target multiple sites in the unamplified genome yield a strong anti-proliferative effect, similar to that observed for sgRNAs targeting genomic amplifications (Fig. 4A–D). We found that the number of predicted DNA cuts correlated strongly with the observed depletion of sgRNAs, whether mediated by copy number (Fig. 4A,C) or multiple alignments (Fig. 4B,D). To quantify this effect, we calculated the slope coefficient for a linear regression of CRISPR guide scores versus predicted number of cuts for both singly and multiply targeted sets of sgRNAs within each cell line. We term these coefficients the CRISPR-Cut Index (CCI) for single-targeting sgRNAs where the amount of cutting depends on copy number (CCI-CN, Fig. 4A,C), and for multiple-targeting sgRNAs where the amount of cutting depends on the number of multiple alignments (CCI-MA, Fig. 4B,D). We observed that the CCI-CN and the CCI-MA for each individual cell line are comparable, suggesting that the decreased proliferation/survival response of cells to increases in the number of loci targeted by CRISPR-Cas9 is similar whether the number of target loci is driven by copy number alteration of a single target locus or multiple different target loci within the genome (Fig. 4E).
Figure 4.
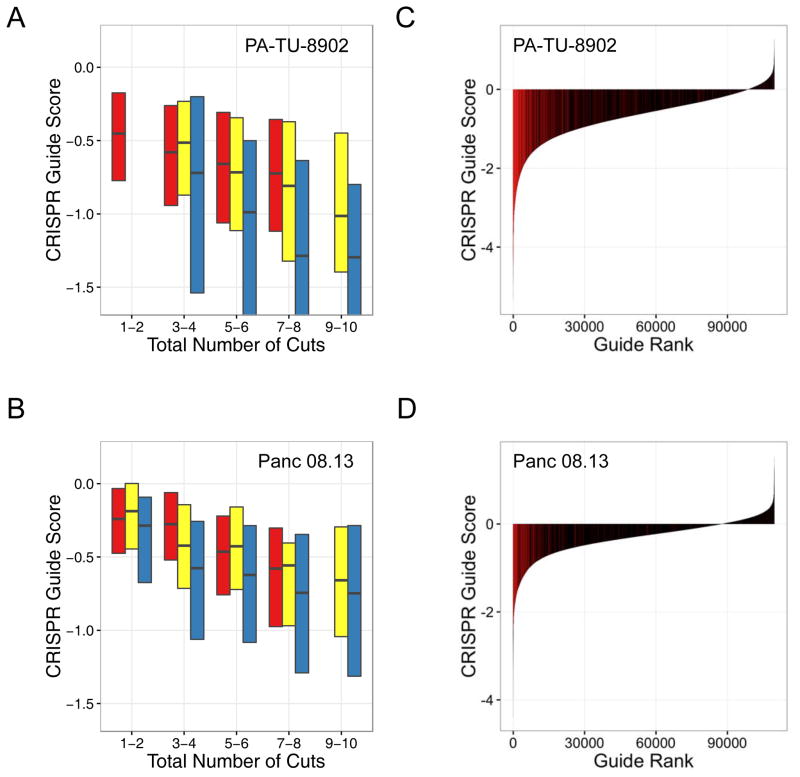
CRISPR-Cas9 sensitivity correlates with number of predicted cuts for both guides targeting single loci and multiple loci. Data from two representative cell lines are shown (PA-TU-8902, A–B; Panc 08.13, C–D) (A, C) CRISPR-Cas9 sensitivity for sgRNAs targeting only a single locus is plotted against copy number of that locus. The black hash marks represent the median CRISPR guide score for all guides targeting a locus at that copy number. The linear trendline is shown. (B, D) CRISPR-Cas9 guide scores for sgRNAs targeting multiple loci, are plotted against the predicted number of cuts for each sgRNA. Only sgRNAs targeting non-amplified regions are included, thus allowing segregation of the impact of multiple CRISPR-Cas9-induced DNA cuts due to either copy number or number of target loci. The influence of the number of predicted DNA cuts on CRISPR-Cas9 guide scores was modeled for each cell line as the slope of the trend line in A–D and termed the CRISPR-Cut Index (CCI). The CCI was determined for both copy number-driven (CCI-CN) (A, C) and multiple alignment-driven effects (CCI-MA) (B, D). (E) Scatter plot of CCI-MA versus CCI-CN showing strong correlation of the effect on CRISPR-Cas9 guide scores for either multiple alignment driven or copy number-driven DNA cuts across the cell lines.
We further investigated whether there was a difference in the cell response to CRISPR-Cas9-induced DNA cuts targeted to different chromosomes or multiple cuts within a single chromosome. Using the multiple alignment analysis described above, we further split multiple-targeting sgRNAs into sets that either targeted multiple chromosomes (inter-chromosomal) or targeted sites within only a single chromosome (intra-chromosomal). We observed, on average, lower guide scores for sgRNAs targeting multiple inter-chromosomal loci as compared to sgRNAs targeting a comparable number of intra-chromosomal loci (Fig. 5A–B; Fig. S7). Moreover, the most promiscuous sgRNAs targeting more than ten inter-chromosomal loci rank among most depleted sgRNAs in pooled screening data for each cell line (Fig. 5C–D). Thus, the response of cancer cells to multiple CRISPR-Cas9-induced DNA cuts is greater when multiple loci are targeted across several chromosomes. Beyond the effects of target gene disruption, these observations further suggest that CRISPR-Cas9 gene editing also yields an anti-proliferative response that is truly gene-independent.
Figure 5.
sgRNAs targeting multiple chromosomes show greater sensitivity to CRISPR-Cas9-induced cutting. Data from two representative cell lines are shown (PA-TU-8902, A, C; Panc 08.13, B, D) (A, B) Boxplots of CRISPR-Cas9 sensitivity to the predicted number of CRISPR-Cas9-induced DNA cuts. CRISPR-Cas9 guide scores are shown on the Y-axis and the predicted number of DNA cuts is shown on the X-axis. sgRNAs are divided into three groups. In red are sgRNAs that target a single locus, and therefore total number of predicted cuts is based on copy number. In yellow are sgRNAs that target multiple loci within a single chromosome (intra-chromosomal). In blue are sgRNAs that target multiple loci across multiple chromosomes (inter-chromosomal). The analysis demonstrates a more potent detrimental influence on cell viability for multiple CRISPR-Cas9-induced DNA cuts across multiple chromosomes (inter-chromosomal) as compared to those restricted to a single chromosome (intra-chromosomal). Multiple linear regression accounting for difference in total number of cuts for inter-chromosomal vs. intra-chromosomal: panel A, β = −0.27, p = 2.64e-22; panel C, β = −0.16, p = 4.97e-22. (C, D) Waterfall plots showing CRISPR guide scores for all sgRNAs in the pooled screens performed on the indicated cell lines. sgRNAs from the multiple alignment analysis targeting multiple chromosomes with >10 predicted target sites are shown in red and are significantly enriched with negative CRISPR-Cas9 guide scores relative to all other sgRNAs in the library (one-sided Kolmogorov–Smirnov test: p = 2.13E-159, C; p = 9.17E-88, D) These data highlight the potent detrimental effect that these sgRNAs have on cell proliferation and viability within the screen.
Variation in cell response to CRISPR-Cas9 targeting
Since CCI-CN and CCI-MA are correlated across cell lines, we next calculated a net index for each cell line by integrating the number of targeted sites and genomic copy number to predict the total number of cuts for all sgRNAs. We observed a plateau in CRISPR guide scores beyond a certain number of cuts for each cell line, typically ranging from 10–50 cuts, suggesting an important limitation in the resolution of sgRNA depletion for sgRNAs targeting many genomic sites (Fig. S8A, B). Informed by this observation, we fit a segmented least-squares model composed of a general linear regression below a breakpoint (estimated by the model) and a flat segment above this breakpoint. The slope coefficient of the first segment of the model is used as the net index (CCI-Total) reflecting the magnitude of the effect of cutting on CRISPR guide scores.
The CCI-Total showed considerable variability across cell lines. While the limited sample size of this CRISPR-Cas9 screening dataset restricts the power for a full multi-variate analysis of the genetic and biologic influences on the CCI-Total, we found two variables that impact this index. Investigating the median CRISPR-Cas9 guide score for “positive control” cell essential genes as a surrogate for CRISPR-Cas9 efficacy in the screens, we identified a strong correlation of this metric with the CCI-Total (Fig. S8C), suggesting that Cas9 efficacy influences the strength of the CRISPR-CN relationship. We also identified that TP53 mutation status also correlates with the CCI-Total (Fig. S8D). While both TP53 mutant and wild-type cells clearly demonstrate the CRISPR-CN relationship, wild-type cells on average show a more pronounced effect, therefore suggesting that the p53 pathway may play a role in mediating the gene-independent response of cells to CRISPR-Cas9 targeting.
CRISPR-Cas9 targeting of amplified regions induces DNA damage and a G2 cell cycle arrest
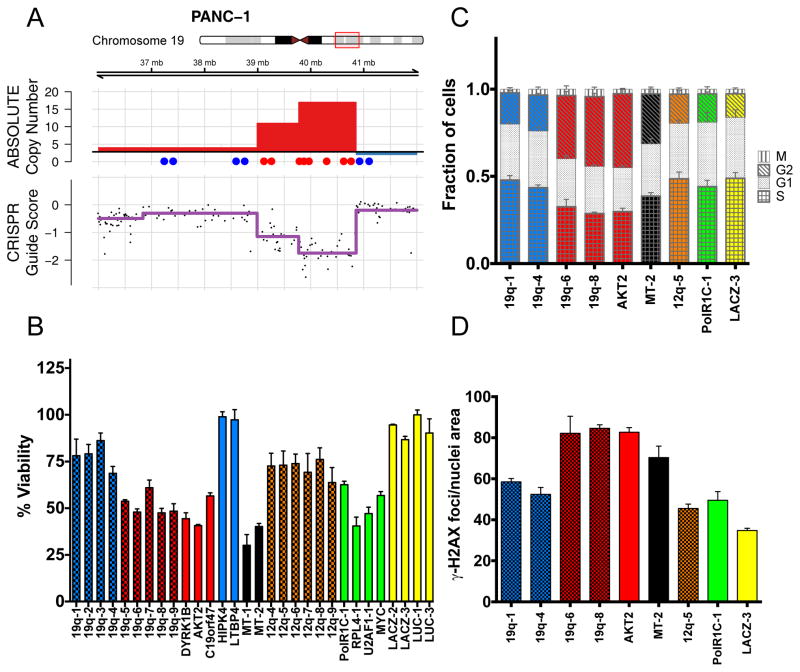
To interrogate a specific amplification example, we introduced sgRNAs targeting genes and intergenic regions inside and outside of the 19q13 amplicon in the PANC-1 pancreatic cancer cell line (Fig. 6A) and measured viable cell number in a short-term, arrayed format Cell-Titer-Glo luminescent assay (Fig. 6B, S9A). We observed a significant reduction in cell proliferation for sgRNAs targeting loci inside the amplicon as compared to outside the amplicon at 6 days after expression of each sgRNA. We noted that the observed effect was equally strong for sgRNAs targeting both amplified genes and intergenic regions and was at least as potent as those sgRNAs targeting non-amplified known essential genes, such as RPL4, U2AF1 and MYC (Fig. 6B). Furthermore, we noted that CRISPR-Cas9 targeting of other loci that are not highly amplified resulted in decreased cell proliferation compared to Lac-Z and Luciferase targeting negative controls. In addition to interrogating sgRNAs targeting amplified regions, we also investigated the effect of two multi-targeted sgRNAs on cell proliferation in this 6-day assay, including one sgRNA with multiple perfect match alignments as well as an sgRNA previously shown to target the genome at 151 different loci (Fig. 6B) (39). Here, we also observed a potent reduction in cell proliferation with these multi-targeted sgRNAs.
Figure 6.
CRISPR-Cas9 targeting of amplified regions or multiple genomic loci induces DNA damage and a G2 cell cycle arrest. (A) Schematic of the PANC-1 19q13 amplicon demonstrating ABSOLUTE DNA copy number (top panel) and CRISPR guide scores (middle panel) mapped by genomic position. Schematic and color scheme are similar to that detailed in Fig. 2. (B) In vitro validation experiment measuring arrayed proliferation and viability response of PANC-1 cells at 6 d post-infection with sgRNAs targeting regions inside (red) and outside (blue) of the demonstrated amplicon. sgRNAs targeting intergenic regions are labeled by chromosomal locus and columns are given a checkered pattern. Multi-targeted sgRNA’s (MT-1 and MT-2) are indicated by black bars. sgRNAs targeting an alternative unamplified locus (12q, orange) and known essential genes (green) are also shown. Non-targeting negative control sgRNAs are shown in yellow. Dots placed below the copy number panel correspond to the validation sgRNAs targeting the indicated genes or intergenic regions on the locus, and are matched by color and left-to-right genomic position. Cell-Titer-Glo was performed at 6-days post-infection. Error bars indicate SD of biologic replicates (n=3). p < 0.0001 for two-tailed T-test comparing sgRNAs inside (red) vs outside (blue and orange) the amplicon. (C) Plot of the percentage of PANC-1 cells in each phase of the cell cycle at 48 hours post-infection with the indicated sgRNAs targeting inside (red) or outside (blue) the amplicon. Data for a multi-targeted sgRNA (MT-2) and a control sgRNA targeting an alternative locus (12q-5), as well as for control genes are also shown. Fraction of cells in each phase of the cell cycle is indicated by a unique pattern within the column corresponding to each cell cycle phase. Colors scheme is as indicated above, with coloration of the G2 and S phases for emphasis. Error bars represent the standard deviation for the mean of three replicates. (D) Plot of the number of γ-H2AX foci present in PANC-1 cells at 48 hours post-infection with the indicated sgRNAs. Color scheme is as indicated above, with checkered pattern corresponding to sgRNAs targeting intergenic regions.
To investigate the mechanism of decreased cell proliferation observed with sgRNAs targeting amplified regions or multiple genomic loci, we utilized a high-content imaging assay to interrogate cell cycle kinetics in multiple sgRNAs in parallel (40). At 48 hours after expression of these sgRNAs, we observed decreased incorporation of the modified thymidine analogue EdU, with diminished S-phase suggestive of decreased DNA synthesis (Fig. 6C). We also observed an accumulation of cells in the G2 phase of the cell cycle with sgRNAs targeting amplified regions or multiple genomic loci (Fig. 6C). Moreover, we observed an increased number of γ-H2AX foci in cells infected with these amplicon-targeting or multi-targeted sgRNAs as compared to control sgRNAs, suggesting that increased DNA damage leads to a G2 cell cycle arrest in these cells (Fig. 6C–D). Notably, we did not observe significant levels of apoptosis at this same time point by measuring cleaved PARP by immunoblotting (Fig. S9B). We have performed similar experiments with the chromosome 12 amplicon in the CAL120 breast cancer cell line and confirmed that these observations are not restricted to the chromosome 19 amplicon in PANC-1 (Fig. S10A–E).
Overall, these observations suggest that CRISPR-Cas9 genome targeting of amplified regions induces a potent early DNA damage response and cell cycle arrest that is proportional to the number of target loci. Notably, this anti-proliferative effect is independent of targeting expressed protein coding genes and does not depend on target gene disruption and protein loss, which typically occurs on a longer timescale (10).
Increased genomic copy number of cell essential genes may protect from complete gene knock-out
Although we found that an increased number of target loci for each sgRNA generally leads to increased gene-independent CRISPR-Cas9-mediated cytotoxicity, we reasoned that since CRISPR-Cas9 genome editing is often incomplete within a cell population, more copies of a target gene could also make a cell resistant to complete gene disruption and protein loss through CRISPR-Cas9 targeting of that locus. Therefore, we hypothesized that certain cell essential gene sets may show the opposite correlation with DNA copy number in pooled negative-selection screening. When we examined the CRISPR-CN correlation across all genes screened in all cell lines from the dataset, we first found an overall negative correlation as expected. However, we also observed that cell essential genes from the KEGG gene sets for the proteasome, ribosome and spliceosome exhibit a CRISPR-CN correlation significantly shifted in the positive direction relative to the rest of the genes in the genome, i.e. higher copy number correlated with higher CRISPR gene scores and less observed gene essentiality (Fig. 7). These observations suggest that increased DNA copy number for target genes with strong underlying essentiality may protect cells from complete CRISPR-induced knock-out of these genes, and thus manifest as relatively less apparent essentiality compared with other essential genes in copy number normal regions of the genome. Together, these data further highlight the importance of considering target gene copy number and gene function in the interpretation of negative selection pooled screening data.
Figure 7.
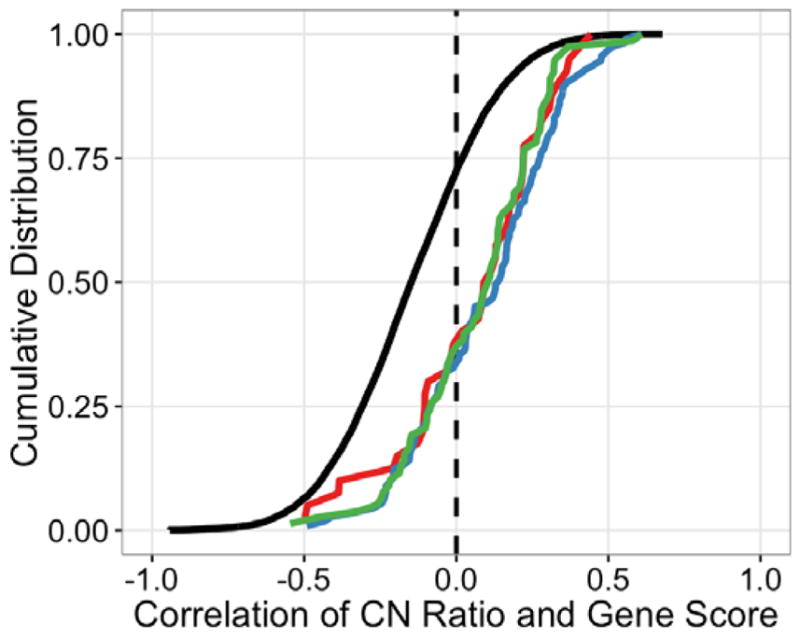
Cell essential genes and copy number. Cumulative distribution function (CDF) of the correlation coefficient between ABSOLUTE CN and CRISPR-Cas9 sensitivity for the indicated gene sets across all 33 cell lines screened with pooled CRISPR-Cas9. Known cell essential KEGG gene sets are displayed separately (proteasome, red; ribosome, blue; spliceosome, green, Table S2) from all other genes in the screen (black). Cell essential genes show a positive shift in CRISPR-CN correlation relative to the overall distribution (two-sided K-S statistic: spliceosome, p = 2.22e-16; proteasome, p = 2.067e-06; ribosome, p = 5.402e-11).
Discussion
Using data from the genome scale interrogation of essential genes in 33 cancer cell lines by CRISPR-Cas9, we report that the number of CRISPR-Cas9-induced DNA cuts strongly influences the proliferation/survival response of cells to CRISPR-Cas9 genome editing in a gene-independent manner. We report that targeting sequences within copy number amplifications with the CRISPR-Cas9 endonuclease induces decreased cell proliferation/viability that is independent of target gene expression or the structure of the targeted amplicon. The magnitude of the effect increases with the amplitude of copy number amplification, and CRISPR-Cas9 targeting within high-level amplifications shows some of the most profound anti-proliferative effects observed in the screens. Moreover, analysis of sgRNAs targeting multiple genomic sites also revealed a strong correlation of cell proliferation/viability with the number of predicted CRISPR-Cas9 DNA cuts. Thus, we propose that there are two types of responses to CRISPR-Cas9 targeting in cancer cell lines: (i) an early anti-proliferative effect of CRISPR-Cas9-induced DNA cuts that increases with the number of cuts conferred by each sgRNA and that is independent of the target gene, and (ii) the gene essentiality resulting from CRISPR-Cas9-induced knock-out of the target gene and subsequent loss of normal protein expression.
The mechanism of the early anti-proliferative response to CRISPR-Cas9-mediated gene editing likely relates to induction of multiple double-strand DNA breaks and subsequent G2 cell cycle arrest. Wang et al. also recently reported an analysis of cell essential genes using CRISPR-Cas9-mediated loss of function screens in 4 leukemia and lymphoma cell lines (28). They found that CRISPR-Cas9-mediated targeting of several genes within the BCR-ABL amplification in the K562 leukemia cell line and JAK2 amplification in the HEL erythroleukemia cell line induced decreased cell viability associated with increased levels of phosphorylated histone H2AX, a marker of DNA damage. Hart et al. also recently reported that guide RNAs targeting greater than 20 sites appear similar to known essential genes (25). Here, we present a comprehensive global analysis of this CRISPR-CN correlation in a large and diverse array of cancer cell lines and demonstrate that this phenomenon is pervasive across many different genetic and phenotypic contexts. Moreover, we provide the first evidence that this CRISPR-CN correlation occurs across a wide range of copy number alterations and chromosome structures, including those with low-level copy number gain. Importantly, we demonstrate that targeting sequences within regions of high-level copy number gain induces among the strongest observed viability phenotypes of all sgRNAs in the screen. Since this effect is not related to specific genes, these observations have important practical implications for utilizing CRISPR-Cas9 technology for cancer dependency profiling and for studying gene essentiality in general.
When we analyzed the effects on cell proliferation/viability induced by increased numbers of cuts, we noted that even a single CRISPR-Cas9-induced DNA cut resulted in decreased cell proliferation when compared to sgRNAs that do not target any human sequence. Thus, choice of negative controls for CRISPR-Cas9 experiments is critically important to interpret the consequences of CRISPR-Cas9-mediated genome editing. While non-targeting sgRNAs may best represent truly neutral negative controls, it may be more appropriate to use a targeting sgRNA directed at a non-genic and copy number normal region of the genome to better model the baseline impact of non-specific DNA targeting with CRISPR-Cas9. The observation that off-target CRISPR-Cas9 cuts likely also cooperate with on-target cuts to effect a cumulative toll on the cell highlights the paramount importance of optimal library design for better on-target and less off-target activity. Improved sgRNA libraries would thus allow better prediction of the total number of CRISPR-Cas9-induced DNA cuts according to baseline copy number and therefore enable enhanced resolution of actual gene-based dependencies within the data.
Moreover, the observation that targeting the CRISPR-Cas9 endonuclease to even a single locus induced decreased proliferation/viability indicates that this approach to targeting genes induces a cellular response in the majority of cases. As such, the effects of this response should be considered in the interpretation of any phenotype observed after targeting a specific gene. Indeed, this observation may also affect efforts to use the CRISPR-Cas9 approach to perform genome editing for therapeutic purposes.
We also observed that for high-level genomic amplifications, the cellular responses to CRISPR-Cas9 cutting toxicity overwhelm the signal from underlying gene essentiality, thus complicating efforts to use CRISPR-Cas9 for identification of essential genes in amplified regions. Hence, it may be most prudent in individual cell line screening data to exclude certain reagents from consideration for identification of essential genes, including sgRNAs targeting genomic amplifications as well as those predicted to confer multiple CRISPR-Cas9 DNA cuts. Failure to properly account for copy number alterations may lead to confounding effects and a higher rate of false positive identification of cell essential genes. Since copy number alterations are the most common genetic alteration found in human epithelial cancers, these observations have practical implications on both individual experiments as well as systematic efforts to interrogate the consequences of gene depletion. These observations also highlight the need to perform CRISPR-Cas9 screens across a large collection of diverse cancer cell lines to represent a variety of cancer gene dependencies while accounting for specific confounding genomic structural alterations within individual cell lines.
We propose that this observation extends beyond merely a confounding artifact of CRISPR-Cas9 technology and uncovers an important underlying biologic concept that cancer cells are vulnerable to induction of site-specific double-stranded DNA breaks within regions of genomic amplification. Genome-scale CRISPR-Cas9 screening has provided an unprecedented resolution of the degree of DNA damage necessary to effect an anti-proliferative or cytotoxic response in cancer cells, revealing an unappreciated susceptibility to even a small number of site-specific DNA breaks. Our observations support the notion that CRISPR-Cas9 targeting of amplified regions of the genome leads to increased DNA-damage and a significant consequent anti-proliferative response. Although these findings complicate the study of amplified regions with CRISPR-Cas9-based approaches, this early anti-proliferative cell response may enable sequence specific therapeutic approaches to target cancer. Many chemotherapy agents (e.g. cisplatin), as well as ionizing radiation, achieve their effects by inducing DNA-damage that is not adequately repaired by cancer cells (41, 42). While many cancer cells are more susceptible than normal cells to chemotherapy and radiation, a major limitation of these treatment approaches is the non-specific nature of these modalities and the narrow therapeutic window for preferential killing of cancer cells versus normal cells. Our observations suggest that targeting non-essential genes or even non-coding, intergenic regions of amplified DNA with CRISPR-Cas9 technology may unveil critical vulnerabilities in cancer cells that could be harnessed for cancer-specific therapy. A precision medicine approach employing simultaneous combination of CRISPR-Cas9 reagents to target multiple amplified loci or tumor-specific mutated sequences within a cancer cell, may enable development of cancer-specific treatments with an optimal therapeutic window.
Materials and Methods
CRISPR-Cas9 screening
Cancer cell lines were transduced with a lentiviral vector expressing the Cas9 nuclease under blasticidin selection (pXPR-311Cas9). Each Cas9 expressing cell line was subjected to a Cas9 activity assay (see below) to characterize the efficacy of CRISPR-Cas9 in these cell lines (Table S1). Cell lines with less than 45% measured Cas9 activity were considered ineligible for screening. Stable polyclonal Cas9+ cell lines were then infected in replicate (n = 3) at low multiplicity of infection (MOI<1) with a library of 123,411 unique sgRNAs targeting 19,050 genes (6 sgRNAs per gene), 1,864 miRNAs and 1,000 non-targeting control sgRNAs (GeCKO v2), selected in puromycin and blasticidin for 7 days and then passaged without selection while maintaining a representation of 500 cells per sgRNA until a defined time point. Genomic DNA was purified from end cell pellets and the guide sequence PCR amplified with sufficient gDNA to maintain representation, and quantified using massively parallel sequencing.
Data quality control
Quality control measures were used to remove cell line replicate samples where (1) the SNP genotype fingerprint failed to match the reference cell line as previously described (37), (2) the reproducibility between replicates was less than 80% and (3) principal component analysis showed a replicate or cell line to be an outlier.
Data processing
Data were processed in a reproducible GenePattern pipeline and are provided on the Project Achilles portal (43). A fold change was calculated per sgRNA and the median of non-targeting controls (n=1000) in the GeCKOv2 library were subtracted from each sgRNA to generate a CRISPR guide score. Given the gene-independent effect of CRISPR-Cas9 described in this manuscript, we chose to use the second-best CRISPR-Cas9 guide score for the purpose of ranking gene-level dependencies in individual cell lines. See supplemental methods for further details.
Cancer cell lines and Cas9 activity assay
Cancer cell lines were obtained primarily from the Cancer Cell Line Encyclopedia, which obtained each line from the original source (Table S1) (35). All cell lines were mycoplasma negative and identity was confirmed through fingerprinting prior to screening using an Affymetrix single nucleotide polymorphism (SNP) array as previously described (37). Prior to screening, cell lines were engineered to stably express Cas9 under Blasticidin selection and Cas9 activity was assayed using a lentivirus with an EF1a driven puromycin-2A-GFP cassette, and a U6 driven sgRNA targeting GFP (pXPR_011) (44). The initial level of GFP is measured with FACs and monitored over time as a measure of cells harboring modified alleles. Cells with GFP remaining are due to either modifications that do not inactivate GFP florescence or inactive Cas9.
Essential gene controls
Genes from the KEGG gene sets for ribosome, proteasome and spliceosome subunits (Table S2) were used as cell essential (positive) controls in the analysis of negative selection CRISPR-Cas9 screening data. Guide sequences that were a perfect match to sgRNAs targeting any other gene or noncoding sequence were removed, except when specifically utilized in described analyses.
Copy number analysis
DNA copy number data were derived from single-nucleotide polymorphism (SNP) microarrays and ABSOLUTE copy number calls were made as previously described (35, 36). CRISPR-Cas9 screening data were mapped according to genomic position of sgRNA sequence (guide-level data) or target gene (by ATARiS algorithm) to the human genome version 19 (hg19). CRISPR-Cas9 screening data were plotted in parallel to Project Achilles shRNA dependency data (43) or Cancer Cell Line Encyclopedia copy number or gene expression data (35, 37).
Whole genome sequencing and analysis
Whole genome sequencing was performed through the Broad Institute-Novartis CCLE, as previously described (45). Whole-genome DNA sequencing data of the cancer cell lines are aligned by Burrows-Wheeler Aligner (46) to the human genome reference 19. The aligned reads are filtered for PCR duplicates by MarkDuplicates from Picard. Read depth coverage was computed and normalized using the previously described approach (47). Briefly, the number of aligned reads were counted for non-overlapping 1kb bins and then normalized for GC-content and mappability biases using the HMMcopy R/Biocondutor package. The normalization was applied to both the cancer cell line and pseudo-normal sample, independently, and then used to generate a log2 ratio (tumor:normal) of GC-corrected coverage. The GC-corrected coverage is then smoothed over 20 kb bins and plotted in Fig. S6 and S7. Chromosomal rearrangements are detected by dRanger (48) from clusters of discordant pairs. Rearrangements at the breakpoint boundaries are manually reviewed and plotted. The relative order of breakage-fusion-bridge cycles and chromothripsis in PANC-1 is inferred based on the criteria in Li et al. (49).
Analysis of published CRISPR-Cas9 screening data
Bayes Factor (BF) values were derived from Hart et al. (25), and fitness genes were determined per cell line according to the thresholds described therein. Gene level copy number data for HCT116, A375 and DLD1 were downloaded from the Cancer Cell Line Encyclopedia. Gene level copy number data for HeLa were downloaded from the Gene Expression Omnibus (GEO) database, accession number GSE8605. Further details of the analysis are provided in the supplemental methods.
sgRNA design and cloning
sgRNAs for validation experiments were designed utilizing the Broad Institute Avana sgRNA design tool (44). sgRNA sequences and characteristics are provided in the supplement (Table S3).
Cell viability assay
The PANC-1 and CAL120 cell lines stably expressing Cas9 were plated in a 96-well plate at 1000 cells/well. One day after plating, cells were infected at a high multiplicity of infection with virus harboring each of the indicated sgRNAs. Cells were cultured +/− puromycin and infection efficiency was calculated from comparison of the puromycin selected and unselected wells. At 6 days post-infection, cell viability was read out using Cell-Titer-Glo. Data is presented using unselected wells and calculating fold-change relative to the non-targeting negative control sgRNA. Error bars are the result of three biological replicates.
Immunoblots
Cells were infected at high MOI in 6 well plates and protein was extracted at 48 hours post-infection. Immunoblotting was performed using antibodies for PARP (Cell Signaling, 46D11, #9532) and beta-actin (Sigma Aldrich A5316).
High-content imaging assay and analysis
PANC-1 and CAL120 cells constitutively expressing Cas9 protein were plated at a density of 4000 cells per well, infected in replicate in 96-well paltes at high MOI and analyzed at an endpoint of 48 hours post-infection. Cells were labeled with EdU and fixed with paraformaldehyde and then labeled with anti-pHH3 (S10) primary antibody (Rabbit: #9701, Cell Signaling, 1:800), anti-phospho-histone H2A.X (Ser139, Mouse: 05-636, END Millipore, 1:1250) and Hoechst 33342 (H3570, Thermo Fisher Scientific, 1ug/mL). Imaging was perfomed with the OperaPhenix imaging system on 20× magnification and data was analyzed using the PerkinElmer Harmony software (40). See supplemental methods for additional details.
Supplementary Material
Significance.
We found that the number of CRISPR-Cas9-induced DNA breaks dictates a gene-independent anti-proliferative response in cells. These observations have practical implications for using CRISPR-Cas9 to interrogate cancer gene function and illustrate that cancer cells are highly sensitive to site-specific DNA damage, which may provide a path to novel therapeutic strategies.
Acknowledgments
We thank Eejung Kim, Joseph Rosenbluh, Srivatsan Raghavan and Belinda Wang for helpful discussions.
Financial Support
This project was supported by R01 CA130988 (W.C.H.), U01 CA199253 (W.C.H.), U01 CA176058 (W.C.H.), P01 CA154303 (W.C.H), and P50 CA12700323 (W.C.H. and A.J.A.). A.J.A. was supported by the Pancreatic Cancer Action Network Samuel Stroum Fellowship, Hope Funds for Cancer Postdoctoral Fellowship, American Society of Clinical Oncology Young Investigator Award, Dana Farber Cancer Institute Hale Center for Pancreatic Cancer, Perry S. Levy Endowed Fellowship, and the Harvard Catalyst and Harvard Clinical and Translational Science Center (UL1 TR001102). This work was conducted as part of the Slim Initiative for Genomic Medicine, a project funded by the Carlos Slim Foundation in Mexico.
Footnotes
Conflict of Interest Disclosure: Levi A. Garraway and William C. Hahn are consultants for Novartis. Matthew Meyerson receives research support from Bayer.
References
- 1.Urnov FD, Rebar EJ, Holmes MC, Zhang HS, Gregory PD. Genome editing with engineered zinc finger nucleases. Nature reviews Genetics. 2010;11:636–46. doi: 10.1038/nrg2842. [DOI] [PubMed] [Google Scholar]
- 2.Joung JK, Sander JD. TALENs: a widely applicable technology for targeted genome editing. Nature reviews Molecular cell biology. 2013;14:49–55. doi: 10.1038/nrm3486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hsu PD, Lander ES, Zhang F. Development and applications of CRISPR-Cas9 for genome engineering. Cell. 2014;157:1262–78. doi: 10.1016/j.cell.2014.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012;337:816–21. doi: 10.1126/science.1225829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jinek M, East A, Cheng A, Lin S, Ma E, Doudna J. RNA-programmed genome editing in human cells. eLife. 2013;2:e00471. doi: 10.7554/eLife.00471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cho SW, Kim S, Kim JM, Kim JS. Targeted genome engineering in human cells with the Cas9 RNA-guided endonuclease. Nat Biotechnol. 2013;31:230–2. doi: 10.1038/nbt.2507. [DOI] [PubMed] [Google Scholar]
- 7.Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 2013;339:819–23. doi: 10.1126/science.1231143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mali P, Yang L, Esvelt KM, Aach J, Guell M, DiCarlo JE, et al. RNA-guided human genome engineering via Cas9. Science. 2013;339:823–6. doi: 10.1126/science.1232033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang T, Wei JJ, Sabatini DM, Lander ES. Genetic screens in human cells using the CRISPR-Cas9 system. Science. 2014;343:80–4. doi: 10.1126/science.1246981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shalem O, Sanjana NE, Hartenian E, Shi X, Scott DA, Mikkelsen TS, et al. Genome-scale CRISPR-Cas9 knockout screening in human cells. Science. 2014;343:84–7. doi: 10.1126/science.1247005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shalem O, Sanjana NE, Zhang F. High-throughput functional genomics using CRISPR-Cas9. Nature reviews Genetics. 2015;16:299–311. doi: 10.1038/nrg3899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sander JD, Joung JK. CRISPR-Cas systems for editing, regulating and targeting genomes. Nat Biotechnol. 2014;32:347–55. doi: 10.1038/nbt.2842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gilbert LA, Larson MH, Morsut L, Liu Z, Brar GA, Torres SE, et al. CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell. 2013;154:442–51. doi: 10.1016/j.cell.2013.06.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maeder ML, Linder SJ, Cascio VM, Fu Y, Ho QH, Joung JK. CRISPR RNA-guided activation of endogenous human genes. Nature methods. 2013;10:977–9. doi: 10.1038/nmeth.2598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Perez-Pinera P, Kocak DD, Vockley CM, Adler AF, Kabadi AM, Polstein LR, et al. RNA-guided gene activation by CRISPR-Cas9-based transcription factors. Nature methods. 2013;10:973–6. doi: 10.1038/nmeth.2600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Konermann S, Brigham MD, Trevino AE, Hsu PD, Heidenreich M, Cong L, et al. Optical control of mammalian endogenous transcription and epigenetic states. Nature. 2013;500:472–6. doi: 10.1038/nature12466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cheng AW, Wang H, Yang H, Shi L, Katz Y, Theunissen TW, et al. Multiplexed activation of endogenous genes by CRISPR-on, an RNA-guided transcriptional activator system. Cell research. 2013;23:1163–71. doi: 10.1038/cr.2013.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang H, Yang H, Shivalila CS, Dawlaty MM, Cheng AW, Zhang F, et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell. 2013;153:910–8. doi: 10.1016/j.cell.2013.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yang H, Wang H, Shivalila CS, Cheng AW, Shi L, Jaenisch R. One-step generation of mice carrying reporter and conditional alleles by CRISPR/Cas-mediated genome engineering. Cell. 2013;154:1370–9. doi: 10.1016/j.cell.2013.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Platt RJ, Chen S, Zhou Y, Yim MJ, Swiech L, Kempton HR, et al. CRISPR-Cas9 knockin mice for genome editing and cancer modeling. Cell. 2014;159:440–55. doi: 10.1016/j.cell.2014.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sanchez-Rivera FJ, Papagiannakopoulos T, Romero R, Tammela T, Bauer MR, Bhutkar A, et al. Rapid modelling of cooperating genetic events in cancer through somatic genome editing. Nature. 2014;516:428–31. doi: 10.1038/nature13906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chiou SH, Winters IP, Wang J, Naranjo S, Dudgeon C, Tamburini FB, et al. Pancreatic cancer modeling using retrograde viral vector delivery and in vivo CRISPR/Cas9-mediated somatic genome editing. Genes & development. 2015;29:1576–85. doi: 10.1101/gad.264861.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xue W, Chen S, Yin H, Tammela T, Papagiannakopoulos T, Joshi NS, et al. CRISPR-mediated direct mutation of cancer genes in the mouse liver. Nature. 2014;514:380–4. doi: 10.1038/nature13589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Maddalo D, Manchado E, Concepcion CP, Bonetti C, Vidigal JA, Han YC, et al. In vivo engineering of oncogenic chromosomal rearrangements with the CRISPR/Cas9 system. Nature. 2014;516:423–7. doi: 10.1038/nature13902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hart T, Chandrashekhar M, Aregger M, Steinhart Z, Brown KR, MacLeod G, et al. High-Resolution CRISPR Screens Reveal Fitness Genes and Genotype-Specific Cancer Liabilities. Cell. 2015;163:1515–26. doi: 10.1016/j.cell.2015.11.015. [DOI] [PubMed] [Google Scholar]
- 26.Koike-Yusa H, Li Y, Tan EP, del Velasco-Herrera MC, Yusa K. Genome-wide recessive genetic screening in mammalian cells with a lentiviral CRISPR-guide RNA library. Nat Biotechnol. 2014;32:267–73. doi: 10.1038/nbt.2800. [DOI] [PubMed] [Google Scholar]
- 27.Parnas O, Jovanovic M, Eisenhaure TM, Herbst RH, Dixit A, Ye CJ, et al. A Genome-wide CRISPR Screen in Primary Immune Cells to Dissect Regulatory Networks. Cell. 2015;162:675–86. doi: 10.1016/j.cell.2015.06.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang T, Birsoy K, Hughes NW, Krupczak KM, Post Y, Wei JJ, et al. Identification and characterization of essential genes in the human genome. Science. 2015;350:1096–101. doi: 10.1126/science.aac7041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sanjana NE, Shalem O, Zhang F. Improved vectors and genome-wide libraries for CRISPR screening. Nature methods. 2014;11:783–4. doi: 10.1038/nmeth.3047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Konermann S, Brigham MD, Trevino AE, Joung J, Abudayyeh OO, Barcena C, et al. Genome-scale transcriptional activation by an engineered CRISPR-Cas9 complex. Nature. 2015;517:583–8. doi: 10.1038/nature14136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gilbert LA, Horlbeck MA, Adamson B, Villalta JE, Chen Y, Whitehead EH, et al. Genome-Scale CRISPR-Mediated Control of Gene Repression and Activation. Cell. 2014;159:647–61. doi: 10.1016/j.cell.2014.09.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen S, Sanjana NE, Zheng K, Shalem O, Lee K, Shi X, et al. Genome-wide CRISPR Screen in a Mouse Model of Tumor Growth and Metastasis. Cell. 2015;160:1246–60. doi: 10.1016/j.cell.2015.02.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Barbie DA, Tamayo P, Boehm JS, Kim SY, Moody SE, Dunn IF, et al. Systematic RNA interference reveals that oncogenic KRAS-driven cancers require TBK1. Nature. 2009;462:108–12. doi: 10.1038/nature08460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Beroukhim R, Mermel CH, Porter D, Wei G, Raychaudhuri S, Donovan J, et al. The landscape of somatic copy-number alteration across human cancers. Nature. 2010;463:899–905. doi: 10.1038/nature08822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Barretina J, Caponigro G, Stransky N, Venkatesan K, Margolin AA, Kim S, et al. The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature. 2012;483:603–7. doi: 10.1038/nature11003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Carter SL, Cibulskis K, Helman E, McKenna A, Shen H, Zack T, et al. Absolute quantification of somatic DNA alterations in human cancer. Nat Biotechnol. 2012;30:413–21. doi: 10.1038/nbt.2203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cowley GS, Weir BA, Vazquez F, Tamayo P, Scott JA, Rusin S, et al. Parallel genome-scale loss of function screens in 216 cancer cell lines for the identification of context-specific genetic dependencies. Scientific data. 2014;1:140035. doi: 10.1038/sdata.2014.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shao DD, Tsherniak A, Gopal S, Weir BA, Tamayo P, Stransky N, et al. ATARiS: computational quantification of gene suppression phenotypes from multisample RNAi screens. Genome research. 2013;23:665–78. doi: 10.1101/gr.143586.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tsai SQ, Zheng Z, Nguyen NT, Liebers M, Topkar VV, Thapar V, et al. GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR-Cas nucleases. Nat Biotechnol. 2015;33:187–97. doi: 10.1038/nbt.3117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Massey AJ. Multiparametric Cell Cycle Analysis Using the Operetta High-Content Imager and Harmony Software with PhenoLOGIC. PloS one. 2015;10:e0134306. doi: 10.1371/journal.pone.0134306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Begg AC, Stewart FA, Vens C. Strategies to improve radiotherapy with targeted drugs. Nat Rev Cancer. 2011;11:239–53. doi: 10.1038/nrc3007. [DOI] [PubMed] [Google Scholar]
- 42.Chabner BA, Roberts TG., Jr Timeline: Chemotherapy and the war on cancer. Nat Rev Cancer. 2005;5:65–72. doi: 10.1038/nrc1529. [DOI] [PubMed] [Google Scholar]
- 43.Project Achilles. Broad Institute of Harvard and MIT; http://wwwbroadinstituteorg/achilles. [Google Scholar]
- 44.Doench JG, Hartenian E, Graham DB, Tothova Z, Hegde M, Smith I, et al. Rational design of highly active sgRNAs for CRISPR-Cas9-mediated gene inactivation. Nat Biotechnol. 2014;32:1262–7. doi: 10.1038/nbt.3026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Huang FW, Hodis E, Xu MJ, Kryukov GV, Chin L, Garraway LA. Highly recurrent TERT promoter mutations in human melanoma. Science. 2013;339:957–9. doi: 10.1126/science.1229259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25:1754–60. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ha G, Roth A, Khattra J, Ho J, Yap D, Prentice LM, et al. TITAN: inference of copy number architectures in clonal cell populations from tumor whole-genome sequence data. Genome research. 2014;24:1881–93. doi: 10.1101/gr.180281.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Drier Y, Lawrence MS, Carter SL, Stewart C, Gabriel SB, Lander ES, et al. Somatic rearrangements across cancer reveal classes of samples with distinct patterns of DNA breakage and rearrangement-induced hypermutability. Genome research. 2013;23:228–35. doi: 10.1101/gr.141382.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Li Y, Schwab C, Ryan SL, Papaemmanuil E, Robinson HM, Jacobs P, et al. Constitutional and somatic rearrangement of chromosome 21 in acute lymphoblastic leukaemia. Nature. 2014;508:98–102. doi: 10.1038/nature13115. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.