Abstract
A nanoparticle-based assay for detection and quantification of Plasmodium falciparum histidine-rich protein 2 (HRP2) in urine and serum is reported. The assay uses magnetic beads conjugated with anti-HRP2 antibody for protein capture and concentration, and antibody-conjugated quantum dots for optical detection. Western blot analysis demonstrated that magnetic beads allow the concentration of HRP2 protein in urine by 20-fold. The concentration effect was achieved because large volume of urine can be incubated with beads, and magnetic separation can be easily performed in minutes to isolate beads containing HRP2 protein. Magnetic beads and quantum dots conjugated to anti-HRP2 antibodies allows the detection of low concentrations of HRP2 protein (0.5 ng/mL), and quantification in the range of 33–2,000 ng/mL corresponding to the range associated with non-severe to severe malaria. This assay can be easily adapted to a noninvasive point-of-care test for classification of severe malaria.
Diagnosis of severe malaria is particularly important in highly endemic regions since most patients are positive for parasitemia and treatment differs from non-severe malaria. Diagnosis can be challenging due to the prevalence of diseases with similar symptoms. Accurate diagnosis is increasingly important to avoid overprescribing antimalarial drugs, minimize drug resistance, and minimize costs.1,2
Methods for malaria diagnosis include direct microscope measurement of the parasite burden in blood samples, antigen detection, or nucleic acid detection.3,4 Microscopy analysis of circulating parasites in blood does not accurately represent the real parasite burden because in cases of severe malaria, the parasites are sequestered in venules and capillaries in various stages of development (e.g., trophozoites and schizont stage).5 In addition, the complexity and logistic difficulties limit the applicability of microscopy analysis in developing countries.4
Plasmodium falciparum histidine-rich protein 2 (HRP2) is a 37 kDa water-soluble protein that can be measured in serum, saliva, or urine.6 HRP2 is released from free and sequestered parasites and hence is a potential biomarker for the total parasite burden in the host.7,8 HRP2 levels associated with severe malaria are typically greater than 100 ng mL−1.3,8,9 Rapid antigen detection tests (RDTs) based on immunochromatography are used to detect HRP2 protein in endemic areas. However, these tests are qualitative and suffer of low analytical sensitivity (mean: 11.7 ng/mL, minimum: 0.62 ng/mL, maximum: 62.5 ng/mL).10 Detection of HRP2 by enzyme-linked immunosorbent assay (ELISA) is only used to quantify HRP2 levels for research proposes. Recently, an immuno-polymerase chain reaction (PCR) assay has been reported to detect low concentrations of HRP2 protein (down to 0.2 ng/mL).11 However, both ELISA and immuno-PCR are not practical for highly endemic areas.
Here we report on a magnetic bead-quantum dot (MB-QD) assay for measurement of HRP2 antigen in serum and urine. The antibody-based assay combines magnetic beads for capture and concentration of the target protein, with quantum dots for efficient quantitative detection.
Superparamagnetic beads (Dynabeads® M-270; Life Technologies, Carlsbad, CA), 2.7 μm in diameter and functionalized with surface epoxy groups, were covalently coupled to primary amine and sulfhydryl groups of mouse IgG monoclonal anti-HRP2 antibodies (clone 3A4) at a concentration of 2 μg antibodies/mg beads. The conjugation was performed in 1.5 M ammonium sulfate overnight at 37°C, and then stored at 4°C prior to use. From the density of beads (6.7 × 107 beads/mg) and assuming that all antibodies are conjugated to the beads, approximately 6 × 105 antibodies are estimated to be conjugated to each bead.
Evaluation of the ability of magnetic bead-antibody (MB-Ab) conjugates to capture and concentrate recombinant HRP2 protein11 was performed by Western blot. These experiments were performed using urine to demonstrate antigen capture in a physiologically relevant sample while avoiding the nonspecific binding associated with serum samples. Collection of urine samples does not require trained personnel and is more acceptable for patients. However, urine is not used for detection of HRP2 because of the low concentration of proteins in urine. Using MB-Ab conjugates, relatively large volumes of urine can be used to concentrate the target protein, and the beads can be easily separated from the sample solution by magnetic separation without the need of centrifugation steps. Magnetic separation of beads from the supernatants is fast (< 1 minute) making this test feasible to be performed in endemic areas.
For Western blot analysis, MB-Ab conjugates were blocked with 6% bovine serum albumin (BSA) in phosphate buffered saline (PBS) for 1 hour at room temperature. One milliliter of antigen in urine (at concentrations up to 500 ng/mL) was incubated with 250 μg MB-Ab conjugates (1.7 × 107 MB-Abs) at room temperature for 1 hour. After incubation and washing steps, the magnetic beads were separated from solution using a magnet (Magnetic Separator Stand; Promega, Fitchburg, WI). The antigens were eluted from the beads using a reducing buffer (10 μL of 50 mM glycine, pH 2.5; 5 μL of sodium dodecyl sulfate (SDS) sample buffer; 3 μL of sample reducing agent), and incubated at 90°C for 15 minutes. The beads were removed by magnetic separation and the elution buffer containing the antigens was loaded onto 4–15% precast gels to perform SDS polyacrylamide gel electrophoresis analysis and electrotransference to a polyvinylidene fluoride membrane. After blocking steps, the membranes were incubated with the same mouse monoclonal antibody (3A4) at a dilution of 1/1,000. Goat anti-mouse horseradish peroxidase conjugated antibodies (Bio-Rad, Hercules, CA) were added at a dilution of 1/10,000. Visualization of the bands was performed using an enhanced chemiluminescence system (Supersignal West Pico; Thermo Fisher Scientific, Waltham, MA).
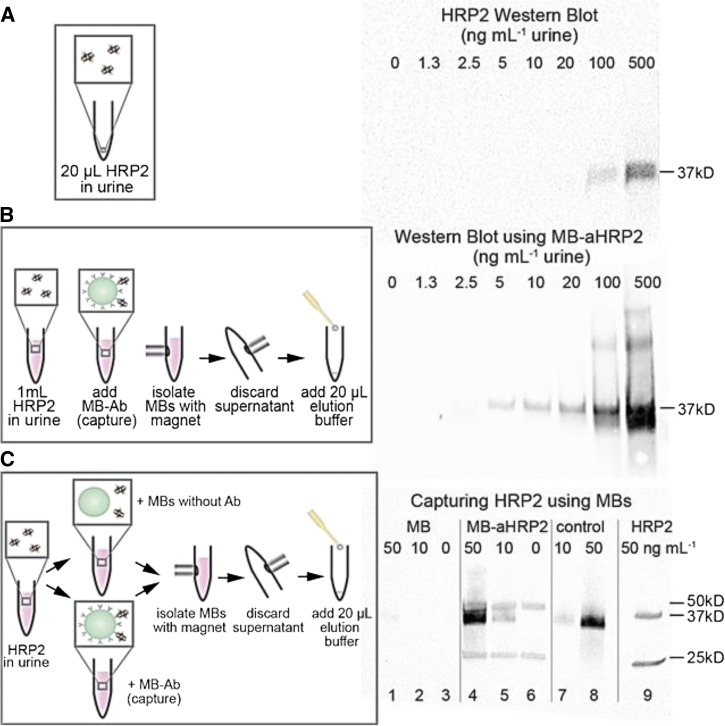
The Western blot shows detection of the captured protein down to a concentration of 5 ng/mL. Without concentration, the detection limit for free HRP2 in urine using Western blot is about 100 ng/mL (Figure 1A ). Comparison of the detection limits suggests a protein concentration of about 20-fold (Figure 1B). These results demonstrate efficient capture and concentration of HRP2 in urine samples by magnetic beads.
Figure 1.
(A) Detection of 0–500 ng/mL of free Plasmodium falciparum histidine-rich protein 2 (HRP2) in urine using Western blot. (B) Schematic illustration of HRP2 capture using magnetic beads. Western blots of eluted HRP2 show detection down to 2.5 ng/mL in urine. (C) Western blot comparing capture on HRP2 using magnetic beads with and without antibodies. 0, 10, and 50 ng of HRP2 were incubated with magnetic beads without antibodies and showed no capture (lanes 1–3), but were captured by magnetic beads conjugated with antibodies (lanes 4–6). Lanes 7 and 8: 10 and 50 ng of free HRP2 (control). Lane 9 is the same as lane 4 (magnetic bead-antibody [MB-Ab] conjugates incubated with 50 ng HRP2), but blotted without primary antibody to HRP2 to confirm that the two bands at 25 and 50 kDa correspond to antibody (heavy and light chain) fragments eluted from the MB-Ab conjugates.
To verify that the HRP2 protein is captured by the antibodies, magnetic beads with and without antibodies were incubated with 0, 10, and 50 ng of HRP2 in 1 mL of urine. After washing and resuspension in elution buffer, magnetic beads without antibodies show no protein capture (Figure 1C). In contrast, magnetic beads with antibodies show a concentration-dependent band at 37 kDa, very similar to the bands for free protein (Figure 1C).
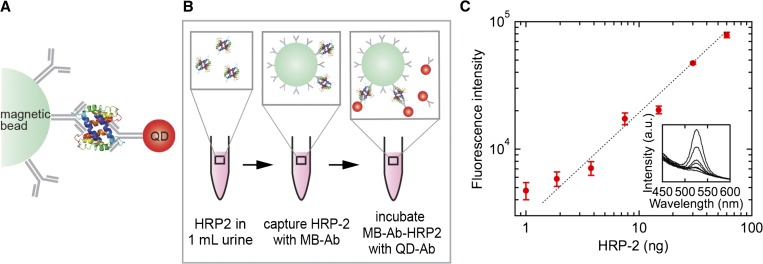
In the MB-QD assay for HRP2 detection (Figure 2A and B), the antibody to HRP2 is conjugated to magnetic beads for capture and concentration of the target protein, and quantum dots conjugated to the HRP2 antibody are used for optical detection. HRP2 antibodies were conjugated to quantum dots (Qdot 525; Life Technologies) using the SiteClick™ Antibody Conjugation Kit (Life Technologies). The same mouse IgG monoclonal antibody to HRP2 was used for capture and detection since HRP2 has repetitive B-cell epitopes. The final concentration of quantum dot–antibody (QD-Ab) conjugates was 2.6 μM.
Figure 2.
(A) Magnetic bead-quantum dot (MB-QD) conjugate. (B) Magnetic bead-quantum dot (MB-QD) assay for capture and detection of Plasmodium falciparum histidine-rich protein 2 (HRP2). The MB-Ab conjugates are incubated with HRP2 antigens for capture and after washing are then incubated with QD-Ab conjugates for detection. (C) MB-QD assay for detection of HRP2. Fluorescence intensity for detection of 1–60 ng (33–2,000 ng/mL) HRP2 in 30 μL of normal human serum. Similar results were obtained using 1 mL of human urine. The insert shows the photoluminescence spectra for one experiment. All measurements were performed in triplicate. Error bars represent standard deviation.
To demonstrate the complete capture-detection sandwich assay, magnetic beads conjugated with antibody were incubated in serum spiked with HRP2 to give protein concentrations from 33 to 2,000 ng/mL, corresponding to the range associated with non-severe to severe malaria (non-severe malaria: < 314 ng/mL, moderate severe malaria: 373 ng/mL, and severe malaria: > 1,103 ng/mL).3,7–9 Similar analyses were performed in urine.
Different quantities of HRP2 (0.01–60 ng) were added to a vial containing 30 μL of normal human serum (or 1 mL of urine). MB-Ab conjugates (2.5 mg) were blocked in 1 mL of 6% BSA and 10% fetal bovine serum (FBS) overnight at room temperature. PBS (with 6% BSA and 1% FBS) and 250 μg of the blocked MB-Ab conjugates (1.7 × 107 MB-Abs) were incubated with serum and urine samples for 2 hours. After incubation, the MB-Ab conjugates were washed three times with phosphate buffered saline with 0.05% Tween® 20 and twice with PBS. For detection, the stock solution of QD-Ab conjugates (2.6 μM) was diluted 80-fold in PBS and 6% BSA. After washing steps and magnetic separation, the QD-Ab solution (10 pmoles, 6 × 1012 QD-Ab conjugates) was added to the HRP2 solution and incubated for 2 hours. The QD-Ab concentration corresponds to about 105 QDs per magnetic bead. The MB-Ab conjugates with captured protein and labeled with QD-Ab conjugates were suspended in 500 μL PBS for photoluminescence measurements (Figure 2C). The photoluminescence intensity was read at 525 nm (Spectramax M3 multi-mode microplate reader; Molecular Devices, Sunnyvale, CA) and was subtracted from the background intensity.
Quantum dots have tunable size-dependent emission, a broad excitation spectrum, high quantum yield, and do not photobleach. The high quantum yield and resistance to photobleaching allow quantitative analysis over a wide range of exposure times, providing a significant advantage over organic fluorophores. The limit of detection of the assay was 0.5 ng/mL in serum and urine, which is comparable to the reported for RDTs and ELISA (11 and 0.7 ng/mL, respectively). The correlation between intensity and HRP2 concentration is linear at higher concentrations (4–1,000 ng/mL) with a slope of 1.0.
This study demonstrates detection of HRP2 over the range of concentration required for classification of severe malaria from a serum and urine. The photoluminescence measurements were performed on magnetic beads labeled with QD-Ab conjugates to minimize the number of steps in the protocol. Scattering from the magnetic beads increases the background signal at shorter wavelengths and the signal for 0.5 ng/mL of HRP2 is 10% above the background. This technique has the potential to be developed as a biosensor for semiquantitative analysis and can be used as a point-of-care test in highly endemic areas in low-income countries where there is a lack of infrastructure and professional and trained workers. In summary, we have demonstrated an assay using magnetic beads for capture and concentration of HRP2 antigen, and quantum dots for detection, which provides an alternative for classification for the severity of malaria caused by P. falciparum.
Footnotes
Authors' addresses: Yagahira E. Castro-Sesquen and Robert H. Gilman, Department of International Health, Johns Hopkins Bloomberg School of Public Health, Baltimore, MD, E-mails: ycastro1@jhu.edu and gilmanbob@gmail.com. Chloe Kim and Peter C. Searson, Department of Materials Science and Engineering, Institute for Nanobiotechnology (INBT), Johns Hopkins University, Baltimore, MD, E-mails: ykim86@jhu.edu and searson@jhu.edu. David J. Sullivan, Department of Molecular Microbiology and Immunology, Johns Hopkins Bloomberg School of Public Health, Baltimore, MD, E-mail: dsulliv7@jhmi.edu.
References
- 1.WHO . World Malaria Report 2012. Geneva, Switzerland: World Health Organization; 2012. WHO Global Malaria Programme, 2012. [Google Scholar]
- 2.Dondorp AM, Fanello CI, Hendriksen IC, Gomes E, Seni A, Chhaganlal KD, Bojang K, Olaosebikan R, Anunobi N, Maitland K, Kivaya E, Agbenyega T, Nguah SB, Evans J, Gesase S, Kahabuka C, Mtove G, Nadjm B, Deen J, Mwanga-Amumpaire J, Nansumba M, Karema C, Umulisa N, Uwimana A, Mokuolu OA, Adedoyin OT, Johnson WB, Tshefu AK, Onyamboko MA, Sakulthaew T, Ngum WP, Silamut K, Stepniewska K, Woodrow CJ, Bethell D, Wills B, Oneko M, Peto TE, von Seidlein L, Day NP, White NJ. Artesunate versus quinine in the treatment of severe falciparum malaria in African children (AQUAMAT): an open-label, randomised trial. Lancet. 2010;376:1647–1657. doi: 10.1016/S0140-6736(10)61924-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hendriksen IC, Mwanga-Amumpaire J, von Seidlein L, Mtove G, White LJ, Olaosebikan R, Lee SJ, Tshefu AK, Woodrow C, Amos B, Karema C, Saiwaew S, Maitland K, Gomes E, Pan-Ngum W, Gesase S, Silamut K, Reyburn H, Joseph S, Chotivanich K, Fanello CI, Day NP, White NJ, Dondorp AM. Diagnosing severe falciparum malaria in parasitaemic African children: a prospective evaluation of plasma PfHRP2 measurement. PLoS Med. 2012;9:e1001297. doi: 10.1371/journal.pmed.1001297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Usdin M, Guillerm M, Chirac P. Neglected tests for neglected patients. Nature. 2006;441:283–284. doi: 10.1038/441283a. [DOI] [PubMed] [Google Scholar]
- 5.White NJ, Pukrittayakamee S, Hien TT, Faiz MA, Mokuolu OA, Dondorp AM. Malaria. Lancet. 2014;383:723–735. doi: 10.1016/S0140-6736(13)60024-0. [DOI] [PubMed] [Google Scholar]
- 6.Rubach MP, Mukemba J, Florence S, John B, Crookston B, Lopansri BK, Yeo TW, Piera KA, Alder SC, Weinberg JB, Anstey NM, Granger DL, Mwaikambo ED. Plasma Plasmodium falciparum histidine-rich protein-2 concentrations are associated with malaria severity and mortality in Tanzanian children. PLoS One. 2012;7:e35985. doi: 10.1371/journal.pone.0035985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Manning L, Davis TM. The mechanistic, diagnostic and prognostic utility of biomarkers in severe malaria. Biomarkers Med. 2013;7:363–380. doi: 10.2217/bmm.13.50. [DOI] [PubMed] [Google Scholar]
- 8.Dondorp AM, Desakorn V, Pongtavornpinyo W, Sahassananda D, Silamut K, Chotivanich K, Newton PN, Pitisuttithum P, Smithyman AM, White NJ, Day NP. Estimation of the total parasite biomass in acute falciparum malaria from plasma PfHRP2. PLoS Med. 2005;2:e204. doi: 10.1371/journal.pmed.0020204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hendriksen IC, White LJ, Veenemans J, Mtove G, Woodrow C, Amos B, Saiwaew S, Gesase S, Nadjm B, Silamut K, Joseph S, Chotivanich K, Day NP, von Seidlein L, Verhoef H, Reyburn H, White NJ, Dondorp AM. Defining falciparum-malaria-attributable severe febrile illness in moderate-to-high transmission settings on the basis of plasma PfHRP2 concentration. J Infect Dis. 2013;207:351–361. doi: 10.1093/infdis/jis675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.WHO, FIND, CDC . Malaria Rapid Diagnostic Test Performance. Results of WHO Product Testing of Malaria RDTs: Round 5, 2013. Geneva, Switzerland: World Health Organization; 2014. [Google Scholar]
- 11.Mikita K, Thakur K, Anstey NM, Piera KA, Pardo CA, Weinberg JB, Mukemba J, Florence S, Mwaikambo ED, Granger DL, Sullivan DJ. Quantification of Plasmodium falciparum histidine-rich protein-2 in cerebrospinal spinal fluid from cerebral malaria patients. Am J Trop Med Hyg. 2014;91:486–492. doi: 10.4269/ajtmh.14-0210. [DOI] [PMC free article] [PubMed] [Google Scholar]