Abstract
Targeted drug decisions in metastatic renal cell carcinoma are exclusively made on the basis of clinical criteria. We investigated whether these biomarkers (HIF-1α, HIF-2α, CAIX, VEGF, VEGFR1, VEGFR2, VEGFR3, PDGFB, PDGFRA, PDGFRB, CD31, CD44, bcl-xL, KIT, p21, CXCR4, PTEN, (CSF)-1R, RET, and FLT-3) can predictive the different effects between sunitinib and sorafenib treatments and are available to guide targeted drug selection. We enrolled all patients who underwent nephrectomy with postoperative sunitinib- or sorafenib-treatment at our institution from 2007 to 2012. Immunohistochemical approach was applied to assess the potential differential effects of immunostainings between sunitinib- and sorafenib-treated groups. We found that patients with high HIF-2α, CD31 expression showed greater relative PFS and OS benefit and patients with high CAIX expression presented greater relative OS benefit from sunitinib than from sorafenib, patients with high VEGFR1 or PDGFRB expression levels exhibited worse relative PFS benefit from sunitinib than from sorafenib. Namely high HIF-2α, CD31, and CAIX expression levels along with low VEGFR1 and PDGFRB expression levels improved the benefit of sunitinib treatment compared with sorafenib treatment. These results can identify whether patients can benefit more from sunitinib or sorafenib for drug selection guidance, eventually with precision medicine.
Metastatic renal cell carcinoma (mRCC) is a chemotherapy-resistant malignancy. Recent advances in molecular biology have led to the development of several novel agents to treat mRCC. As a consequence, monotherapy with interferon (IFN)-α or high-dose bolus interleukin (IL)-2 should no longer be routinely recommended as first-line therapy in mRCC, except in certain circumstances (e.g. lung metastasis, renal clear cell carcinoma (ccRCC), and long interval)1. To date, seven targeted drugs have been approved in the USA and Europe for treating mRCC: four vascular endothelial growth factor receptor (VEGFR)-targeted tyrosine kinase inhibitors (TKIs), including sorafenib, sunitinib, pazopanib, and axitinib; one anti-vascular endothelial growth factor (VEGF) monoclonal antibody, bevacizumab; and two mammalian target of rapamycin (mTOR) inhibitors, temsirolimus and everolimus2. Molecular-targeted therapy improved prognosis of mRCC compared with cytokine therapy. However, the length of response and survival benefit of targeted therapy varies considerably among patients. Patients may demonstrate different targeted-therapy sensitivities even with same pathological classification, clinical stage, dose, and mode of clinical treatment3,4.
VEGF signalling pathway inhibitors have been associated with various toxicities, including an increased risk of fatal adverse events5. With the realisation that limited criteria exist for prediction of response to a particular drug and that many sequential treatments are likely to be pursued6, the economic burden and adverse events of several drugs must be globally considered to achieve the optimum potential risk-to-benefit ratio for each patient. Clinical and translational studies to identify the phenotypic predictors of response to each drug are urgent. Partial treatment-resistant patients still respond to other targeted drugs in clinical practice. The differential effects of patients treated with differential targeted drugs for mRCC do exist. Identifying the biomarkers for efficacy is necessary to select suitable patients for this therapeutic approach.
A number of approaches such as blood-based biomarker, tissue-based biomarker, SNP biomarker, and cellular biomarkers are currently under investigation2. However, these markers typically only provide clinicians with risk assessment for a patient based on multiple criteria and are prognostic (i.e. providing information about independent outcome of treatment). A fraction of these are known to be predictive (i.e. providing information about efficacy of a specific treatment intervention). Nevertheless, the differential effects indicated by these predictive markers are typically compared with placebo or cytokine treatment but not with other targeted drugs. Hence, whether these biomarkers signify similar traits of distinguished therapeutic effects of other targeted drugs remains unclear. No guideline is available for drug selection. The hope and interest lie in the identification of accurate markers that can predict the responses to existing effective but toxic target therapies7,8,9,10.
Sunitinib and sorafenib were the first approved vascular endothelial growth factor-targeted drugs as first-line treatment of mRCC in China, with everolimus being the second-line drug. Sunitinib and sorafenib are all oral multikinase inhibitors with effects on tumour–cell proliferation and tumour angiogenesis. Sunitinib was identified as platelet-derived growth factor receptors (PDGFRA, PDGFRB), VEGFR1, VEGFR2, VEGFR3, stem cell factor receptor (KIT), Fms-like tyrosine kinase-3 (FLT-3), colony stimulating factor receptor Type 1 (CSF-1R), and the glial cell-line derived neurotrophic factor receptor (RET) inhibitor9. Sorafenib was initially identified as a Raf kinase inhibitor,10 which also inhibits VEGFR1, VEGFR2, VEGFR3, PDGFRB, Flt-3, RET, and KIT11. Sunitinib or sorafenib inhibition of these receptor tyrosine kinases has been demonstrated in biochemical and cellular assays, and inhibition of function has been demonstrated in cell proliferation assays.
We collected prognostic and predictive tissue-based biomarkers related to mRCC patients treated with sunitinib or sorafenib to validate whether these biomarkers can predictive the different effects between sunitinib and sorafenib treatments and are available to guide targeted drug selection. The following biomarkers were included in the study: hypoxia inducible factor 1, alpha subunit (HIF-1α), hypoxia inducible factor 2, alpha subunit (HIF-2α), carbonic anhydrase IX (CAIX), VEGF, VEGFR1, VEGFR2, VEGFR3, platelet-derived growth factor beta polypeptide (PDGFB), PDGFRA, PDGFRB, differentiated microvascular density (MVD, assessed by CD31 staining), CD44, BCL2-like 1 (bcl-xL), KIT, cyclin-dependent kinase inhibitor 1A (p21), chemokine (C-X-C motif) receptor 4 (CXCR4), and phosphatase and tensin homolog (PTEN). All these biomarkers have been established these prognostic or predictive function of RCC benefit from sunitinib or sorafenib in existing research12,13,14,15,16,17,18,19,20,21. For instance,’ C. D’Alterio found that high CXCR4 expression correlates with poor response to sunitinib in metastatic renal cancer21. A study of J. Garcia-Donas regarding the outcome of sunitinib treatment in advanced ccRCC found that PDGFRB was associated with better response evaluation criteria in solid tumours (RECIST) objective response to sunitinib19. In addition, we included FLT-3, CSF-1R, and RET in the present analysis because these biomarkers were targets of sunitinib or sorafenib9,11.
In this prospective analysis, we enrolled sunitinib- and sorafenib-treated groups. We aimed to use the IHC approach to identify predictive markers of drug selection in patients with mRCC (subgroups of patients receiving different degrees of relative benefit from sunitinib compared with sorafenib). This goal leads to selection of treatment with the optimum potential risk-to-benefit ratio for each patient.
Patients and Methods
Study design and patients
In this prospective cohort study, we consecutively enrolled adults (≥18 years) who underwent nephrectomy and had a pathologically confirmed diagnosis of renal clear cell carcinoma and distant organ metastasis. These patients were treated at the PLA General hospital from January 2007 to December 2012. Before the targeted treatment, all patients had a detailed history, physical examination, and laboratory parameters. Response to treatment was assessed by a treating doctor according to RECIST criteria trimestral22. Patients who were lost-to-follow-up are excluded from subsequent steps of analysis (three and 11 patients were excluded in the sunitinib and sorafenib groups, respectively). A total of 52 patients from the sunitinib-treated group and 55 from the sorafenib-treated group met the inclusion and exclusion criteria, had complete follow-up records, and were scheduled for therapy on a daily clinical practice setting. We aimed to use the IHC approach to identify predictive markers of drug selection in patients with mRCC (subgroups of patients receiving different degrees of relative benefit from sunitinib compared with sorafenib). This goal leads to selection of treatment with the optimum potential risk-to-benefit ratio for each patient. Written informed consent for a tumour-oriented study was obtained from all patients prior to sample collection. The study was approved by the Protection of Human Subjects Committee of Chinese People’s Liberation Army General Hospital, and the study was carried out in accordance with the Declaration of Helsinki.
Tissue microarray and Immunohistochemistry
The formalin-fixed paraffin-embedded primary tumour specimens were obtained from the Department of Pathology at the PLA General Hospital. Three core-tissue biopsies with 1.0 mm in diameter were taken from the selected morphologically representative regions of each paraffin-embedded RCC and precisely arrayed using a custom-built instrument (Quick-Ray UT-06, UNITMA). Commercially available antibodies were used for all IHC studies. The following antibodies were studied: HIF-1α (1:200), HIF-2α (1:200), CAIX (1:1000), VEGF (1:100), VEGFR1 (1:200), VEGFR2 (1:300), VEGFR3 (1:200), PDGFB (1:200), PDGFRA (1:150), PDGFRB (1:300), CD31 (1:200), CD44 (1:200), bcl-xL (1:200), KIT (1:300), p21 (1:100), CXCR4 (1:900), PTEN (1:300), (CSF)-1R (1:25), RET (1:75), and FLT-3 (1:75). The stained TMA sections were analysed by two dedicated urologic pathologists (DJ Wang and ZG Song) while being unaware of the sample origin and clinical outcomes.
IHC analysis
The immunostaining level was assessed by manual counting and was aided by analysis using Image-pro Plus 6.0 (IPP 6.0)23. The accuracy of this software has not only been verified by some authors but has also been recently applied to numerous aspects in biomedicine. The measurement parameter was integrated optical density (IOD). The function of irregular automated optical inspection (irregular AOI) was applied using the IPP 6.0 software to score and rule out non-target staining. Interesting targets included the tumour cell and adjacent fibroblasts. All images analysed using IPP 6.0 were verified by pathologists. Bcl-xL, which mainly stains the cell nuclei, was determined by counting 1,000 cells in 10 large graticules visible in the microscope. The results were semiquantitatively reported on a scale of 0–3 for intensity, where 0 was negative, 1 was weak, 2 was moderate, and 3 was strong. The percentage of tumour staining was reported as 0–100% in increments of 10%. A composite score was formed using the product of the intensity and percentage of tumour staining. Differentiated MVD was determined by IHC staining of CD31, and MVD was derived by counting each vessel identified within the selected areas, including any stained endothelial cell or endothelial-cell cluster that was separated from adjacent microvessels. Vessel lumens were not required for identifying a structure as a microvessel. Microvessels in necrotic or sclerotic areas within a tumour were not considered in vessel evaluations. Measurements for three cores per sample were averaged for the analysis. The mean value of the IOD or vessel counts in the selected ‘hotspots’ was retained as the final value.
Statistical analysis
We defined progression free survival (PFS) as the time between the first day of treatment and the date of radiological progressive disease (PD), clear clinical evidence of PD, or death. Patients who had not progressed at database closure were censored during the final follow-up. If the PD date was unknown, we censored PFS at the last tumour assessment. Overall survival (OS) was defined as the time between the first day of system treatment and the date of death.
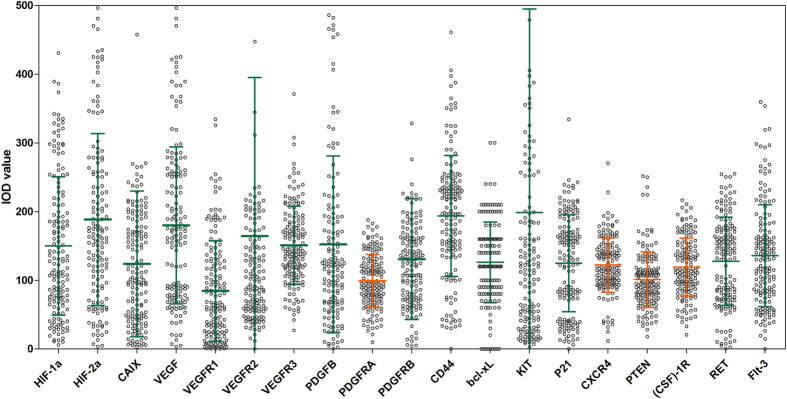
An ideal marker can reliably separate patients, and its expression should provide enough difference. Therefore, we created a scatter chart with IOD values or composite scores of all IHC markers (MVD was not included) to observe the distribution of IHC marker expression. Those with less coefficient of variation (CV) were excluded.
Cut-off estimation aims to assess potential differential effects of individual immunostainings among different treatment groups. The same standard was needed to define high- and low-expression subgroups in different treatment groups. (Fig. 1) Therefore, individual IHC markers were analysed on the basis of the results of median cut-off among all 107 patients.
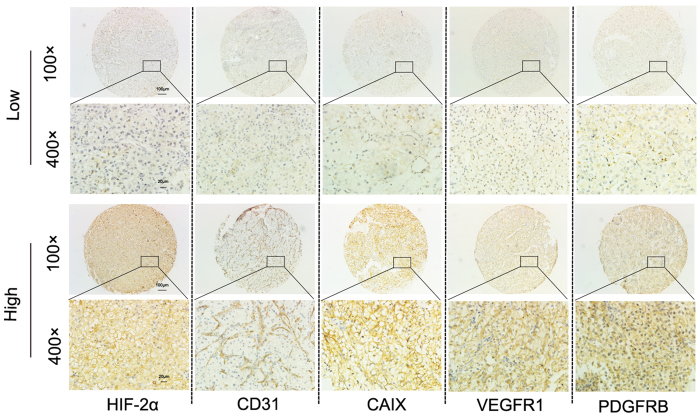
Figure 1. High expression and low expression of HIF-2α, CD31, CAIX, VEGFR1, and PDGFRB in metastatic renal-cell carcinoma.
Representative immunohistochemistry of tissue core array stained with HIF-2α, CD31, CAIX, VEGFR1, and PDGFRB antibody.
Ultimately, candidate predictive markers related to targeted drug selection were assessed. To establish any IHC marker as a prognostic marker for sunitinib- and/or sorafenib-treated groups, We used the Kaplan–Meier method to analyse PFS and OS, and the Cox regression model to verify significant differences noted in the Kaplan–Meier curves for sunitinib- and sorafenib-treated groups between high- and low-expression subgroups defined by the respective median IOD value. We used multivariable analysis by including age, sex, BMI, T stage, Fuhrman grade, and Memorial Sloan–Kettering Cancer Center (MSKCC) score as covariates (clinical factors that were associated with p < 0·05 with a specific variable were used as covariates for that specific variable). To establish any IHC marker as a predictive marker, we included a treatment versus immunostaining interaction term in the Cox model analysis for both PFS and OS to assess the potential differential effects of immunostainings between the two treatment groups, with treatment group and immunostainings as two other independent variables. IHC markers with a significant pinteraction value with treatment were regarded as predictor of drug selection. A two-sided p value of less than 0.05 was regarded as significant in all stages of this analysis. All the statistical analyses (apart from clustering) were conducted using SPSS version 19.0 and GraphPad 6.0.
Results
A total of 107 patients were enrolled in this study between January 2007 and December 2012, and the follow-up database was closed in July 2015. After a median follow-up of 47 months (range of 29–102 months, IQR33–54 months), the median PFS and OS of the 107 patients was 13.6 months (1.5– >102 months, 6.7– >35 months) and 30.5 months (1.5– >102 months, 18– >41 months), respectively. Common metastatic sites, risk factors, Fuhrman grades, and T stage between the sunitinib and sorafenib groups were approximate. Table 1 shows the clinical characteristics of the enrolled patients in the three groups.
Table 1. Clinical characteristics of patients.
| Sunitinib (N = 52) | Sorafenib (N = 55) | |
|---|---|---|
| Median age (years) | 51(18–81,44–62) | 58(29–79,47–64) |
| Sex (male) | 40(78%) | 41(75%) |
| Median follow-up (months) | 49(32–78, 33–59) | 46(29–102, 34–50) |
| Median PFS | 14.25(1.5– >78, 6.6– >35) | 13.4(3.7– >102, 6.7– >36) |
| Median OS | 30(1.5– >78, 17.1– >41.6) | 31.5(3.7– >102, 19.4– >41) |
| Common metastasis sites | ||
| Lung | 45(86%) | 43(78%) |
| Lymph nodes | 25(48%) | 23(42%) |
| Bone | 11(21%) | 13(24%) |
| Liver | 9(17%) | 6(11%) |
| Risk factors* | ||
| 0 (favourable) | 10(19%) | 12(22%) |
| 1–2 (intermediate) | 38(73%) | 39(71%) |
| ≥3 (poor) | 4(8%) | 4(7%) |
| Fuhrman grade | ||
| 1 | 1(2%) | 4(7%) |
| 2 | 28(54%) | 27(49%) |
| 3 | 19(36%) | 19(35%) |
| 4 | 4(8%) | 5(9%) |
| T stage | ||
| 1–2 | 28(54%) | 31(56%) |
| ≥3 | 24(46%) | 24(44%) |
Data are median (range, IQR) or n (%). *Risk groups according to Memorial Sloan-Kettering Cancer Center prognostic factors, T stage is pathological stage.
The scatter chart shows the expression distribution of 19 markers (Fig. 2). The expression levels of PDGFRA (CV = 0.39), PTEN (CV = 0.32), (CSF)-1R (CV = 0.35), and CXCR4 (CV = 0.32) in a fitted normal distribution, and this coefficient of variation is small, thus were excluded from the analysis.
Figure 2. Scatter chart with IOD values or composite scores of 19 IHC markers.
Line at Mean with Standard Deviation (SD), Coefficient of Variation(SD/Mean) as follows: CV(HIF-1α) = 0.67, CV(HIF-2α) = 0.66; CV(CAIX) = 0.86; CV(VEGF) = 0.64; CV(VEGFR1) = 0.86; CV(VEGFR2) = 1.40; CV(VEGFR3) = 0.48; CV(PDGFB) = 0.84; CV(PDGFRA) = 0.39; CV(PDGFRB) = 0.67; CV(CD44) = 0.45; CV(bcl-xL) = 0.46; CV(KIT) = 1.49; CV(P21) = 0.57; CV(CXCR4) = 0.32; CV(PTEN) = 0.32; CV((CSF)-1R) = 0.35; CV(RET) = 0.50; CV(Flt-3) = 0.55.
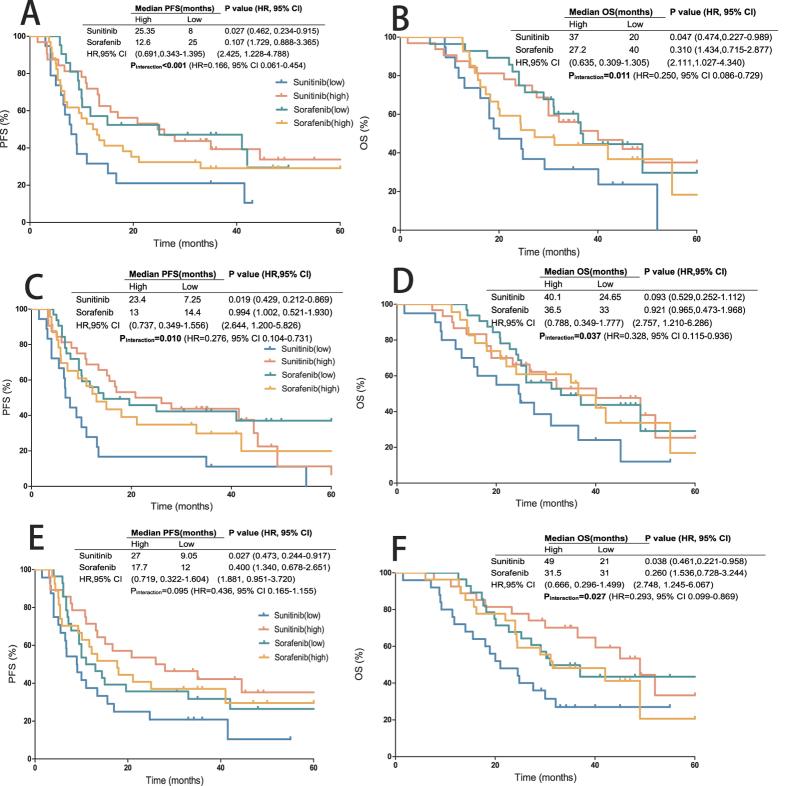
Tables 2 and 3 show the 16 IHC markers staining as a dichotomous variable correlated with PFS and OS. Multivariable analysis showed the prognostic function of each IHC marker for sunitinib- and sorafenib-treated groups (Tables 2 and 3). Then we compared the sunitinib group with the sorafenib group to identify the predictive markers related to targeted drug selection. HIF-2α, CD31 were the IHC markers with significant predictive value for both PFS and OS benefits. CAIX (pinteraction = 0.027) exhibited a significant predictive value for OS benefit, whereas VEGFR1 (pinteraction = 0.038) and PDGFRB (pinteraction = 0.017) showed significant predictive value for PFS benefit. In the sunitinib group, patients with high HIF-2α expression displayed a longer PFS than those with low HIF-2α expression (25.35 months vs 8 months; p = 0.027). An inverse effect was noted in the sorafenib group (12.6 vs 25 months; p = 0.107). Patients with high HIF-2α expression showed a greater relative PFS benefit from sunitinib than from sorafenib (hazard ratio: 0.691, 95% CI: 0.343–1.395). On the contrary, patients with low HIF-2α expression presented a worse relative PFS benefit from sunitinib than from sorafenib (2.425, 1.228–4.788). Equivalent difference was also noted in the OS benefit (pinteraction = 0.011). Patients with high HIF-2α expression displayed a longer OS than those with low HIF-2α expression in the sunitinib group (37 months vs 20 months; p = 0.047). And inverse effect was noted in the sorafenib group (27.2 vs 40 months; p = 0.310) (Fig. 3A,B). Approximate predictive value was also noted in CD31 for both PFS and OS benefits. Patients with high CD31 expression showed a greater relative PFS and OS benefits from sunitinib than from sorafenib, whereas those with low CD31 expression presented a worse relative PFS and OS benefits (hazard ratio and 95% CI on Fig. 3C,D and Tables 2 and 3). CAIX exhibited a significant predictive value for the OS benefit (pinteraction = 0.027) but did not attain enough significance in the PFS benefit (pinteraction = 0.095). Patients with high CAIX expression showed a greater relative OS benefit from sunitinib than from sorafenib (0.666, 0.296–1.499). Low concentrations of CAIX expression presented a worse relative OS as well (2.748, 1.245–6.067) (Fig. 3E,F). Inverse difference was noted for VEGFR1 (pinteraction = 0.038) and PDGFRB (pinteraction = 0.017) in PFS benefit. In the sorafenib group, patients with high VEGFR1 expression showed a much longer PFS than those with low VEGFR1 expression (41 months vs. 10.1 months; p = 0.032), whereas this effect was not noted in the sunitinib group (13.4 vs. 16.15 months; p = 0.508) (Fig. 4A,B). Equivalent predictive value was also noted in PDGFRB for the PFS benefit (hazard ratio and 95% CI on Fig. 4C,D and Tables 2 and 3). To compare the power of interaction action of each immunostainings, we analysed the HR values of each interaction term. HIF-2α showed the most notable HR value of interaction action with treatment for both PFS and OS benefits, and the hazard ratios of drug comparison in the lower HIF-2α group and higher HIF-2α group are in different direction.
Table 2. 16 IHC markers and PFS in the biomarker population.
| PFS (months) | P value (HR, 95% CI) | Pinteraction | ||||
|---|---|---|---|---|---|---|
| Sunitinib | Sorafenib | Sunitinib | Sorafenib | |||
| HIF-1α | High | 11 | 13 | 0.239 (1.496, 0.765–2.928) | 0.994 (1.002, 0.521–1.930) | 0.847 |
| Low | 18.9 | 14.4 | ||||
| HIF-2α | High | 25.35 | 12.6 | 0.027 (0.462, 0.234–0.915) | 0.107 (1.729, 0.888–3.365) | <0.001 |
| Low | 8 | 25 | ||||
| CAIX | High | 27 | 17.7 | 0.027 (0.473, 0.244–0.917) | 0.400 (1.340, 0.678–2.651) | 0.095 |
| Low | 9.05 | 12 | ||||
| VEGF | Low | 13.4 | 10 | 0.218 (0.659, 0.340–1.280) | 0.027 (0.423, 0.197–0.909) | 0.347 |
| High | 14.9 | undefined | ||||
| VEGFR1 | High | 13.4 | 41 | 0.508 (1.253, 0.647–2.430) | 0.032 (0.463, 0.229–0.934) | 0.038 |
| Low | 16.15 | 10.1 | ||||
| VEGFR2 | High | 15.6 | 13.4 | 0.908 (0.958, 0.465–1.976) | 0.638 (1.197, 0.567–2.528) | 0.187 |
| Low | 11 | 13.3 | ||||
| VEGFR3 | High | 11 | 9.75 | 0.303 (1.434, 0.722–2.845) | 0.325 (1.391, 0.721–2.683) | 0.263 |
| Low | 18.9 | 21.1 | ||||
| PDGFB | High | 17 | 15 | 0.599 (0.776, 0.259–1.930) | 0.384 (1.393, 0.660–2.939) | 0.087 |
| Low | 11 | 29.5 | ||||
| PDGFRB | High | 12.2 | 63.5 | 0.345 (1.373, 0.711–2.651) | 0.032 (0.479, 0.245–0.937) | 0.017 |
| Low | 15.6 | 10 | ||||
| CD31 | High | 23.4 | 13 | 0.019 (0.429, 0.212–0.869) | 0.994 (1.002, 0.521–1.930) | 0.010 |
| Low | 7.25 | 14.4 | ||||
| CD44 | High | 10.9 | 11 | 0.026 (2.259, 1.101–4.637) | 0.067 (1.949, 0.955–3.975) | 0.838 |
| Low | 41.5 | 25 | ||||
| bcl-xL | High | 6.8 | 14.4 | 0.001 (3.530, 1.739–7.168) | 0.868 (1.057, 0.551–2.028) | 0.084 |
| Low | 44.5 | 12.35 | ||||
| KIT | High | 13.4 | 9.4 | 0.8678 (0.867, 0.478–1.546) | 0.198 (1.558, 0.793–3.063) | 0.622 |
| Low | 16.7 | 18 | ||||
| p21 | High | 11 | 9.3 | 0.039 (2.120, 1.037–4.333) | 0.135 (1.674, 0.851–3.291) | 0.411 |
| Low | 45.3 | 41 | ||||
| RET | High | 12.05 | 9.5 | 0.377 (1.389, 0.670–2.876) | 0.065 (2.051, 0.955–4.405) | 0.090 |
| Low | 16.7 | undefined | ||||
| FLT-3 | High | 14.25 | 14.85 | 0.747 (0.895, 0.456–1.756) | 0.921 (1.034, 0.538–1.986) | 0.422 |
| Low | 14.5 | 13.4 | ||||
Survival data are median, p values and HRs for sunitinib- and sorafenib-treated groups are both come from multivariable analysis, they compare higher biomarker group to lower biomarker group; pinteraction values are come from multivariate analysis (Cox model analysis) to assess the potential differential effects of immunostainings between the two treatment groups.
Table 3. 16 IHC markers and OS in the biomarker population.
| OS (months) | P value | Pinteraction | ||||
|---|---|---|---|---|---|---|
| Sunitinib | Sorafenib | Sunitinib | Sorafenib | |||
| HIF-1α | High | 29.2 | 32.5 | 0.849 (1.073, 0.521–2.209) | 0.921 (0.965. 0.473–1.968) | 0.737 |
| Low | 36.5 | 38.5 | ||||
| HIF-2α | High | 37 | 27.2 | 0.047 (0.474, 0.227–0.989) | 0.310 (1.434, 0.715–2.877) | 0.011 |
| Low | 20 | 40 | ||||
| CAIX | High | 49 | 31.5 | 0.038 (0.461, 0.221–0.958) | 0.260 (1.536, 0.728–3.244) | 0.027 |
| Low | 21 | 31 | ||||
| VEGF | High | 36.5 | 55 | 0.248 (0.652, 0.315–1.348) | 0.024 (0.368, 0.154–0.879) | 0.215 |
| Low | 32.1 | 25.8 | ||||
| VEGFR1 | High | 36.5 | 49 | 0.282 (0.672, 0.325–1.386) | 0.103 (0.531, 0.249–1.136) | 0.243 |
| Low | 29.6 | 29.1 | ||||
| VEGFR2 | High | 36.5 | 42 | 0.897 (0.950, 0.435,2.073) | 0.515 (1.308, 0.582–2.940) | 0.138 |
| Low | 24.8 | 31.5 | ||||
| VEGFR3 | High | 29.4 | 28.15 | 0.989 (1.005, 0.478–2.114) | 0.765 (1.119, 0.535–2.341) | 0.127 |
| Low | 33.25 | 37 | ||||
| PDGFB | High | 36.5 | 37 | 0.373 (0.829, 0.352–1.912) | 0.490 (1.343, 0.581–3.105) | 0.121 |
| Low | 27.6 | 35 | ||||
| PDGFRB | High | 29.7 | 49 | 0.457 (1.314, 0.640–2.696) | 0.333 (0.700, 0.340–1.441) | 0.130 |
| Low | 32.1 | 31.1 | ||||
| CD31 | High | 40.1 | 36.5 | 0.093 (0.529, 0.252–1.112) | 0.921 (0.965, 0.473–1.968) | 0.037 |
| Low | 24.65 | 33 | ||||
| CD44 | High | 24.8 | 36 | 0.033 (2.380, 1.071–5.289) | 0.476 (1.302, 0.630–2.691) | 0.562 |
| Low | 52 | 49 | ||||
| bcl-xL | High | 21 | 36.5 | 0.011 (2.626, 1.244–5.543) | 0.477 (0.772, 0.377–1.577) | 0.465 |
| Low | 49 | 36 | ||||
| KIT | High | 49 | 19.15 | 0.7140 (0.835, 0.324–1.334) | 0.508 (1.280, 0.616–2.660) | 0.622 |
| Low | 28.4 | 37 | ||||
| p21 | High | 29.2 | 31 | 0.185 (1.690, 0.778–3.671) | 0.276 (1.494, 0.726–3.075) | 0.600 |
| Low | 40.1 | 46 | ||||
| RET | High | 28.05 | 25.2 | 0.236 (1.610, 0.732–3.540) | 0.039 (2.377, 1.044–5.411) | 0.452 |
| Low | 32.1 | 49 | ||||
| FLT-3 | High | 36.5 | 42 | 0.867 (0.939, 0.447–1.970) | 0.600 (0.828, 0.410–1.676) | 0.564 |
| Low | 29.4 | 30.6 | ||||
Survival data are median, p values and HRs for sunitinib- and sorafenib-treated groups are both come from multivariable analysis, they compare higher biomarker group to lower biomarker group; pinteraction values are come from multivariate analysis (Cox model analysis) to assess the potential differential effects of immunostainings between the two treatment groups.
Figure 3. HIF-2α, CD31 and CAIX associated with PFS and OS.
(A) HIF-2α with PFS; (B) HIF-2α with OS; (C) CD31 with PFS; (D) CD31 with OS; (E) CAIX with PFS; (F) CAIX with OS. HRs and p values are both come from Cox model analysis.
Figure 4. VEGFR1 and PDGFRB associated with PFS and OS.
(A) VEGFR1 with PFS; (B) VEGFR1 with OS; (C) PDGFRB with PFS; (D) PDGFRB with OS. HRs and p values are both come from Cox model analysis.
Discussion
In clinical practice, the patient’s genetic information used to lead the treatment decisions of personalized treatment increasingly. Oncology transited to accurate drug phase rapidly. Nowadays researchers recognize that tiny genotype alteration may cause distinguished drug responses for different cancers. A number of studies reported that how gene researches apply to personalized treatment recently. E.g. 2014 National Comprehensive Cancer Network (NCCN) Guideline Insights highlighted the important role of KIT or PDGFRA mutation status in treatment decisions of gastrointestinal stromal tumours with imatinib and/or sunitinib24; The antibody-drug conjugate trastuzumab emtansine has improved outcomes in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer compared with trastuzumab-based therapy, as demonstrated in phase III studies25; BRAF-positive non-small cell lung cancer (NSCLC) is sensitive to Dabrafenib. Anaplastic lymphoma kinase (ALK)-positive NSCLC is sensitive to treatment with an ALK-tyrosine kinase inhibitor26.
However, treatment decisions in mRCC are exclusively made on the basis of clinical criteria. Emergence of new targeted drugs options for mRCC has increased the need to prospectively identify populations of patients that are likely to benefit from the specific target treatment and predictive markers related to targeted drug selection. An ideal predictive biomarker is the one that is easily and unambiguously measured and reliably separates patients who will benefit from a specific approach to those who will benefit from an alternative approach27. IHC is a simple, inexpensive, and reliable assay. Given that renal cell tumours are usually treated by partial or radical nephrectomy, tumour tissue is routinely available for IHC. Sunitinib-treated patients showed longer PFS and OS than sorafenib-treated patients in multiple phases 2 and 3 trials9,10. In the current study, the sunitinib-treated group displayed similar median PFS and OS compared with the sorafenib-treated group. This finding may be ascribed to the exclusion of patients who were lost-to-follow-up (three in sunitinib group and 11 in sorafenib group). Those patients who were lost-to-follow-up usually demonstrate poor prognosis. The sunitinib- or sorafenib-treated group having slightly longer median PFS and OS than in phase 3 trial may come from two aspects. One is exclusion of those lost-to-follow-up patients; The other is that all the included patients underwent nephrectomy or cytoreductive nephrectomy (CN). In a meta-analysis of two randomised controlled trials comparing CN plus systemic treatment versus systemic treatment alone, a significant increase was observed in long-term survival of patients treated with CN28.
Five IHC markers (HIF-2α, CD31, CAIX, VEGFR1, and PDGFRB) were identified as predictive markers related to targeted drug selection. High HIF-2α, CD31, and CAIX expression levels along with low VEGFR1 and PDGFRB expression levels improved the benefit of sunitinib treatment compared with sorafenib treatment. On the contrary, low HIF-2α, CD31, and CAIX expression levels along with high VEGFR1 and PDGFRB expression predicted improved benefit relative to sorafenib. High RET and low bcl-xL expression demonstrated the predictive trend of improved benefit relative to sunitinib compared with sorafenib, but pinteraction was not significant. High levels of HIF-2α confer more favourable response to sunitinib therapy in two articles and agreed well with the present results13,14. We further discovered that this effect was not observed in the sorafenib therapy. But the inverse tendency was noted, patients with high HIF-2α expression displayed a shorter PFS and OS than those with low HIF-2α expression in the sorafenib group. High CAIX expression is associated with a better prognosis in localised RCC and mRCC12, similar to the tendency reflected in our trail, while high CAIX expression predicted more benefit relative to sunitinib treatment than sorafenib treatment. CD31 was associated with better prognosis in RCC in multiple studies15, we further found the differential effects of CD31 between sunitinib and sorafenib treatments. CD31 was associated with better sunitinib treatment than sorafenib treatment. The study by J. Garcia-Donas on the outcome of sunitinib treatment in advanced ccRCC found that PDGFRB was associated with better sunitinib RECIST objective response19. In the present study, this effect was not noted in the sunitinib group but was noted in the sorafenib group. This is possibly because of different evaluation methods. J. Garcia-Donas used objective response as effect indicator; that effect was not noted in PFS and OS. Moreover, the study of J. Garcia-Donas did not find VEGFR1 related to prognosis in sunitinib treatment. The present study reached the same conclusion, and found that VEGFR1 was associated with better sorafenib treatment effect furthermore.
The above five proteins have been identified as key factors of hypoxia and angiogenesis. They work cooperatively or function independently in different circumstance. As shown in Fig. 5, In clear cell RCC, the upregulation of VEGF mRNA levels is expected due to HIF-1α dysregulation as a result of VHL protein loss in addition to the hypoxic environment3. HIF-2α is one of the most investigated member of the HIF-α subunits, ccRCC has an inactivated VHL gene express either in the HIF-2α alone or in both HIF-1α and HIF-2α. VEGF–VEGFR signalling plays an important role in angiogenesis and vasculogenesis, VEGFR1 is expressed on tumour cells and binds to VEGFR-A, VEGFR-B, and placental growth factor. And that PDGFRB have been suggested to play crucial roles in tumour–vessel stability by recruiting pericytes to newly formed vessels29. CAIX is a HIF-1α-regulated transmembrane protein12. MVD gives important information on tumour vascularisation, which might be important for response to TKI treatment.
Figure 5. Biologic pathways in renal cell carcinoma and tyrosine kinase receptors targeted by VEGF signalling pathway inhibitors.
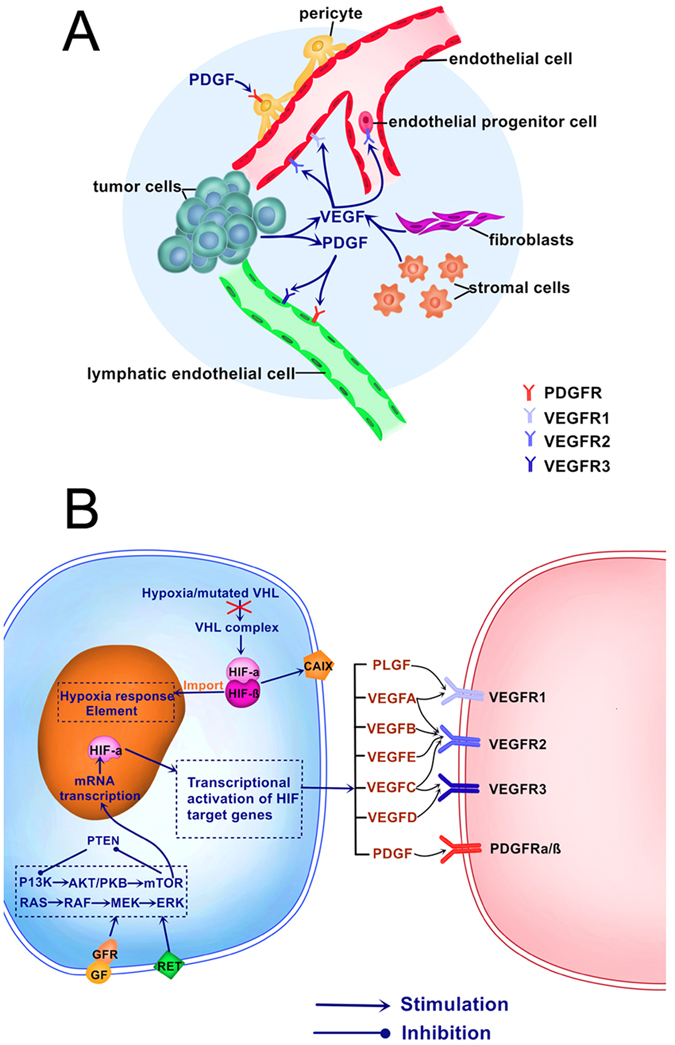
(A) Tyrosine kinase receptors involved in angiogenesis and lymphangiogenesis targeted by VEGF signalling pathway inhibitors. Multiple cellular subtypes, including endothelial cells, pericytes, tumour cells, fibroblasts and endothelial progenitor cells, are implicated in tumour angiogenesis. Signalling through vascular endothelial growth factor receptors and platelet-derived growth factor receptors leads to endothelial cell growth, migration and survival. Tumour lymphangiogenesis is mainly driven through VEGFC/VEGFR3 and PDGF/PDGFR signalling in lymphatic endothelial cells. (B) Biologic pathways and markers in renal cell carcinoma: AKT/PKB = akt/protein kinase B (gene); ERK = extracellular signal-regulated kinase; GF = growth factor; GFR = growth factor receptor; MEK = methyl ethyl ketone.
The mechanisms by which HIF-2α, CD31, CAIX, VEGFR1, and PDGFRB may predict drug-specific benefit remains unclear. The cause may be the different inhibiting effects to several kinases between sunitinib and sorafenib. In vitro studies found that differential regulation of sunitinib or sorafenib targets predicts its tumour-type-specific effect on endothelial and/or tumour cell apoptosis, and showed that molecular targets could be used as biomarkers capable of assessment of therapeutic response30. We need to dig deeper to find out these mechanisms. Molecular events that can unveil the biologic heterogeneity underlying the varied clinical behaviour of RCC may help improve individualised prognostication and risk-stratified clinical decision-making.
Protein translation is a biological process occurring in the cytoplasm and a transcription process occurring in the nucleus. Therefore, the absolute expression and sub-cellular localisation staining of IHC markers may predict clinicopathological features and outcome in ccRCC. We did not analyse sub-cellular localisation staining of IHC markers, but this method should be involved in future research. Comparative study between other targeted drugs such as pazopanib, bevacizumab, and temsirolimus should be conducted as well, and others kinds of markers (e.g. blood-based biomarker, cellular biomarkers, even circulating tumour DNA) should be involved in future research.
Finally, this study did not include an external validation. Small sample sizes, long time spans, and single-centred study decreased the power. Because the patients were all from the yellow race, the relevance of these predictive roles needs to be assessed in other ethnic groups. However, we only included patients with clear-cell cancer component tumours to ensure homogeneity of our data and excluded patients who were lost-to-follow-up. Response to treatment was assessed by a treating doctor. These factors are probably the major contributors to the robustness of the results, with statistically significant outcomes that persisted after adjustment for multiple testing.
Conclusion
High HIF-2α, CD31, and CAIX expression levels along with low VEGFR1 and PDGFRB expression levels improved the benefit of sunitinib treatment compared with sorafenib treatment. On the contrary, low HIF-2α, CD31, and CAIX expression levels along with high VEGFR1 and PDGFRB expression levels improved the benefit of sorafenib treatment compared with sunitinib treatment. If confirmed, these results can identify drug resistant patients early, and identify whether a patient may benefit from a specific targeted therapy and serve as a guide for drug selection, eventually with precision medicine.
Take home message
We enrolled sunitinib-treated and sorafenib-treated groups meanwhile, to identify predictive markers of drug selection in patients with mRCC (subgroups of patients receiving different degrees of relative benefit from sunitinib compared with sorafenib).
Additional Information
How to cite this article: Ma, X. et al. Predictive Immunohistochemical Markers Related to Drug Selection for Patients Treated with Sunitinib or Sorafenib for Metastatic Renal Cell Cancer. Sci. Rep. 6, 30886; doi: 10.1038/srep30886 (2016).
Acknowledgments
We thank the investigators and the patients and their families who made this work possible. This work was supported by the People’s Republic of China and the National High Technology Research and Development Program (“863”Program) of China: the screening and clinical validation of characteristic protein biomarkers in renal cancer based on a large-scale biobank (2014AA020607); People’s Republic of China and the National High Technology Research and Development Program (“863”Program) of China: clinical exploratory application of LESS and NOTES in urology (2012AA02101).
Footnotes
Author Contributions X.M., L.W., X.Z. and H.L. contributed to study conception, participated in the study design, data collection and assembly, data analysis and interpretation, and interpretation, writing of the report, and decision to submit for publication; Y.Z. and G.G. contributed to provision of material and patients, response assess to treatment, and decision to submit for publication. D.W. and Z.S. contributed to assessment of immunostaining level; X.M., L.W. and Y.G. contributed to the data analysis and the writing of the report; K.L. contributed to drawing; K.L., Q.M. and C.Z. participated in the data collection, analysis and interpretation.
References
- Ljungberg B. et al. EAU guidelines on renal cell carcinoma: 2014 update. Eur Urol 67, 913–924 (2015). [DOI] [PubMed] [Google Scholar]
- Patard J. J., Rioux-Leclercq N. & Fergelot P. Understanding the importance of smart drugs in renal cell carcinoma. Eur Urol 49, 633–643 (2006). [DOI] [PubMed] [Google Scholar]
- Sun M. et al. Prognostic factors and predictive models in renal cell carcinoma: a contemporary review. Eur Urol 60, 644–661 (2011). [DOI] [PubMed] [Google Scholar]
- Funakoshi T., Lee C. H. & Hsieh J. J. A systematic review of predictive and prognostic biomarkers for VEGF-targeted therapy in renal cell carcinoma. Cancer Treat Rev 40, 533–547 (2014). [DOI] [PubMed] [Google Scholar]
- Schutz F. A., Je Y., Richards C. J. & Choueiri T. K. Meta-analysis of randomized controlled trials for the incidence and risk of treatment-related mortality in patients with cancer treated with vascular endothelial growth factor tyrosine kinase inhibitors. J Clin Oncol 30, 871–877 (2012). [DOI] [PubMed] [Google Scholar]
- Eichelberg C. et al. SWITCH: A Randomised, Sequential, Open-label Study to Evaluate the Efficacy and Safety of Sorafenib-sunitinib Versus Sunitinib-sorafenib in the Treatment of Metastatic Renal Cell Cancer. Eur Urol (2015). [DOI] [PubMed] [Google Scholar]
- Escudier B. et al. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet 370, 2103–2111 (2007). [DOI] [PubMed] [Google Scholar]
- Hudes G. et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med 356, 2271–2281 (2007). [DOI] [PubMed] [Google Scholar]
- Motzer R. J. et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 356, 115–124 (2007). [DOI] [PubMed] [Google Scholar]
- Escudier B. et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 356, 125–134 (2007). [DOI] [PubMed] [Google Scholar]
- Wilhelm S. et al. Discovery and development of sorafenib: a multikinase inhibitor for treating cancer. Nat Rev Drug Discov 5, 835–844 (2006). [DOI] [PubMed] [Google Scholar]
- Bui M. H. et al. Carbonic anhydrase IX is an independent predictor of survival in advanced renal clear cell carcinoma: implications for prognosis and therapy. Clin Cancer Res 9, 802–811 (2003). [PubMed] [Google Scholar]
- Choueiri T. K. et al. von Hippel-Lindau gene status and response to vascular endothelial growth factor targeted therapy for metastatic clear cell renal cell carcinoma. J Urol 180, 860–865; discussion 865–866 (2008). [DOI] [PubMed] [Google Scholar]
- Escudier B. et al. Sorafenib for treatment of renal cell carcinoma: Final efficacy and safety results of the phase III treatment approaches in renal cancer global evaluation trial. J Clin Oncol 27, 3312–3318 (2009). [DOI] [PubMed] [Google Scholar]
- Dornbusch J. et al. Analyses of potential predictive markers and survival data for a response to sunitinib in patients with metastatic renal cell carcinoma. PLoS One 8, e76386 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H. L. et al. c-KIT: potential predictive factor for the efficacy of sorafenib in metastatic renal cell carcinoma with sarcomatoid feature. Clin Genitourin Cancer 11, 134–140 (2013). [DOI] [PubMed] [Google Scholar]
- Mikami S. et al. Expression of TNF-alpha and CD44 is implicated in poor prognosis, cancer cell invasion, metastasis and resistance to the sunitinib treatment in clear cell renal cell carcinomas. Int J Cancer 136, 1504–1514 (2015). [DOI] [PubMed] [Google Scholar]
- del Puerto-Nevado L. et al. Active angiogenesis in metastatic renal cell carcinoma predicts clinical benefit to sunitinib-based therapy. Br J Cancer 110, 2700–2707 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Donas J. et al. Prospective study assessing hypoxia-related proteins as markers for the outcome of treatment with sunitinib in advanced clear-cell renal cell carcinoma. Ann Oncol 24, 2409–2414 (2013). [DOI] [PubMed] [Google Scholar]
- Terakawa T., Miyake H., Kusuda Y. & Fujisawa M. Expression level of vascular endothelial growth factor receptor-2 in radical nephrectomy specimens as a prognostic predictor in patients with metastatic renal cell carcinoma treated with sunitinib. Urol Oncol 31, 493–498 (2013). [DOI] [PubMed] [Google Scholar]
- D.A. C. et al. High CXCR4 expression correlates with sunitinib poor response in metastatic renal cancer. Curr Cancer Drug Targets 12, 693–702 (2012). [DOI] [PubMed] [Google Scholar]
- Taylor P. T. & Haverstick D. Re: New guidelines to evaluate the response to treatment in solid tumors (ovarian cancer). J Natl Cancer Inst 97, 151; author reply 152 (2005). [DOI] [PubMed] [Google Scholar]
- Wang C. J. et al. Survivin expression quantified by Image Pro-Plus compared with visual assessment. Appl Immunohistochem Mol Morphol 17, 530–535 (2009). [DOI] [PubMed] [Google Scholar]
- von Mehren M. et al. Gastrointestinal stromal tumors, version 2. 2014. J Natl Compr Canc Netw 12, 853–862 (2014). [DOI] [PubMed] [Google Scholar]
- Verma S. et al. Trastuzumab emtansine for HER2-positive advanced breast cancer. N Engl J Med 367, 1783–1791 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soda M. et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature 448, 561–566 (2007). [DOI] [PubMed] [Google Scholar]
- Ahern T. P. & Hankinson S. E. Re: Use of archived specimens in evaluation of prognostic and predictive biomarkers. J Natl Cancer Inst 103, 1558–1559; author reply 1559–1560 (2011). [DOI] [PubMed] [Google Scholar]
- Osborn J. R., Chodak G. W., Kommu S. & Persad R. A. Re: Cytoreductive nephrectomy in patients with metastatic renal cancer: a combined analysis. J Urol 174, 396; author reply 397 (2005). [DOI] [PubMed] [Google Scholar]
- Erber R. et al. Combined inhibition of VEGF and PDGF signaling enforces tumor vessel regression by interfering with pericyte-mediated endothelial cell survival mechanisms. FASEB J 18, 338–340 (2004). [DOI] [PubMed] [Google Scholar]
- Bousquet G. et al. Differential regulation of sunitinib targets predicts its tumor-type-specific effect on endothelial and/or tumor cell apoptosis. Cancer Chemother Pharmacol 72, 1183–1193 (2013). [DOI] [PubMed] [Google Scholar]