Abstract
This study aimed to assess safety and therapeutic potential of gene electrotransfer as a method for delivery of plasmid encoding vascular endothelial growth factor A to ischemic myocardium in a porcine model. Myocardial ischemia was induced by surgically occluding the left anterior descending coronary artery in swine. Gene electrotransfer following plasmid encoding vascular endothelial growth factor A injection was performed at four sites in the ischemic region. Control groups either received injections of the plasmid without electrotransfer or injections of saline vehicle. Animals were monitored for seven weeks and hearts were evaluated for angiogenesis, myocardial infarct size, and left ventricular contractility. Arteriograms suggest growth of new arteries as early as two weeks post treatment in electrotransfer animals. There is a significant reduction of infarct area and left ventricular contractility is improved in gene electrotransfer treated group compared to controls. There was no significant difference in mortality of animals treated with gene electrotransfer of plasmid encoding vascular endothelial growth factor A from control groups. Gene delivery of plasmid encoding vascular endothelial growth factor A to ischemic myocardium in a porcine model can be accomplished safely with potential for myocardial repair and regeneration.
Keywords: gene therapy, gene electro transfer, angiogenesis, myocardial infarct, vascular endothelial growth factor A
Introduction
Heart disease is the leading cause of death in the United States accounting for a quarter of all deaths 1, 2. Approximately 22% of men and 46% of women surviving myocardial infarction will develop congestive heart failure within six years post MI 1-3. Of those diagnosed with heart failure, the five year survival rate is only 50% 1-3. Extensive atherosclerosis of the coronary arteries can lead to sufficient blockage of the coronary vessels to cause downstream ischemia in the ventricles of the heart 4. Therefore, current clinical therapies aim at direct revascularization to improve the survival rates for patients post MI 5. However, the myocardium does not regenerate to its former electrical, histological, and functional pumping potential 1, 6. Rather, ischemic myocardium remodels with fibroblast proliferation, collagen deposition replacing cardiomyocytes, resulting in thin, non-contracting patches in the ventricle, limiting cardiac output 7. There is little clinical evidence of return of viable cardiomyoctes in patients surviving MI 6-8.
Due to atherosclerosis, angiogenesis in ischemic myocardium is likely impaired. There are many pre-clinical therapies aimed at inducing angiogenesis in ischemic myocardium. Vascular endothelial growth factor A (VEGF-A) is a commonly used gene for therapeutic angiogenesis in coronary artery disease models, with intentions of collateral vessel development in hypoxic areas 6. The primary role of VEGF-A is induction of angiogenesis by stimulation of endothelial cells to migrate and proliferate in hypoxic areas 9-11. VEGF-A also plays an important role in stem cell mobilization and differentiation to cardiomyocytes as well as cardiomyocyte proliferation 12-15. VEGF-A promotes embryonic stem cell differentiation into cardiomyocytes in a mouse model 14 and has been shown to promote cardiac stem/progenitor cell mobilization and cardiomyocyte differentiation 12 and cardiomyocyte proliferation 13. In addition to the use of VEGF-A delivered in the form of recombinant protein, several gene therapy approaches for VEGF-A delivery to the myocardium have also been evaluated 6.
Primary methods of gene delivery are viral mediated transfer and plasmid-based transfer. Adenoviral vectors and adeno-associated (AAV) vectors are the most commonly used vectors for delivery to the heart with immunogenicity and toxicity as major side effects. Plasmid DNA delivery is associated with fewer side effects, low immune response, low toxicity, and reduced cost but with lower efficiency 6, 16, 17.
There are several studies reported on electroporation aka gene electrotransfer (GET) mediated gene delivery to the beating heart in a small animal model 18, 19. In our previous studies we reported on plasmid DNA delivery via GET to cardiac muscle in vivo in a porcine model 20, 21. GET of plasmid DNA can be applied safely directly to the ischemic and non-ischemic myocardium and gene expression can be modulated by pulsing conditions 20, 21. We have also determined that there may be a therapeutic benefit to local delivery of pVEGF-A to ischemic myocardium, with improved myocardial perfusion two weeks after treatment 21.
Our current study is focused on long-term outcome of GET delivery of pVEGF-A to ischemic myocardium. We demonstrate that seven weeks post treatment new arteries can be observed in arteriograms in the majority of treated ischemic porcine hearts. We also observed a reduction of myocardial damage from ischemic injury at treatment sites, with improved contractility and relaxation of the left ventricular myocardium under chemically induced stress. Overall, our results indicate a potential long-term therapeutic benefit of exogenous VEGF-A gene delivery mediated by electrotransfer to ischemic myocardium, with reduced progression to pathological cardiac remodeling at treatment sites in a porcine model of myocardial infarction.
Results
Occlusion of Left Anterior Descending Coronary Artery leads to Myocardial Infarction of Anterior Left Ventricle
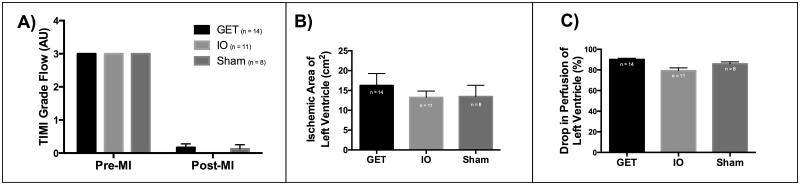
To study the effects of an angiogenic therapy for myocardial infarction, we first confirmed the presence of myocardial infarction of the anterior left ventricle after occlusion of the left anterior descending (LAD) coronary artery. Thrombosis in myocardial infarction (TIMI) scoring of the arterial tree, confirmed successful permanent occlusion of the LAD coronary artery (Figure 1A). There were two animals that were excluded from the study, based on a TIMI score of 3, indicating insufficient occlusion, and lack of ischemia (which was confirmed at 7 weeks, with lack of MI detectable by triphenyltetrazolium (TTC) staining). SPY® System analysis indicates a presence of a consistent ischemic region in terms of size and drop in perfusion (Figure 1B and 1C). Further analysis of plasma indicated a sharp increase of cardiac troponin-I 24 hours after the induction of ischemia indicating presence of myocardial infarction (Supplemental Table 1). There is a sharp increase of plasma creatine kinase muscle-isoform (CK-MM), indicating muscle cell damage and also consistent with myocardial infarction in all groups (Supplemental Table 1).
Figure 1. Permanent ischemia was induced via LAD occlusion.
A) TIMI flow index drops significantly after occlusion, with no significant difference in TIMI between treatment groups (Two-way ANOVA and Tukey’s multiple comparisons test, alpha=0.05, P = <0.0001). B) SPY based measurement of ischemic surface area of the left ventricle indicates consistent occlusion between treatment groups, with no significant difference in ischemic area (One-way ANOVA, alpha = 0.05, P=0.6388). C) SPY based measurement of drop in perfusion in the ischemic area of the left ventricle indicates a consistent reduction in perfusion, with GET group receiving a slightly larger drop in perfusion than the IO group (One-way ANOVA, Tukey’s multiple comparisons test alpha=0.05, P=0.0012).
GET of pVEGF-A to the ischemic myocardium has no deleterious effects on survival or cardiac output in a porcine model
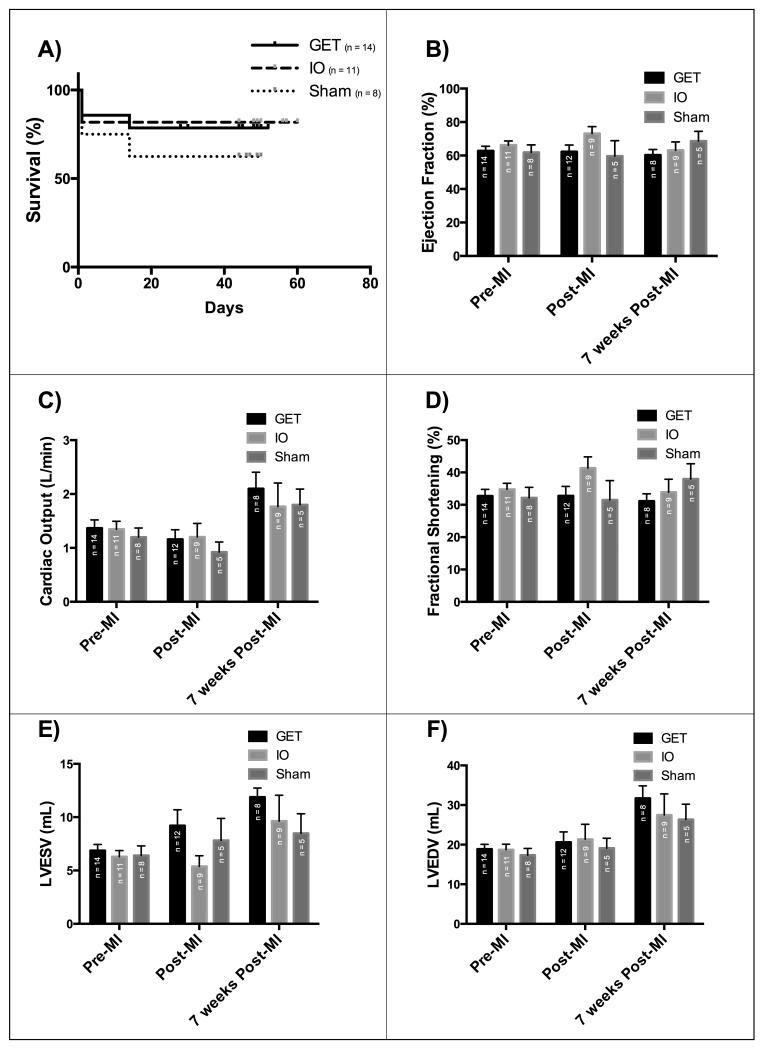
Kaplan-Meier log ranks survival analysis was performed on all animals surviving the onset of acute ischemia and receiving GET with pVEGF-A, pVEGF-A injections without electrotransfer, or saline injections. Two animals were excluded from the study due to irreversible ventricular fibrillation following the occlusion of the left anterior descending coronary artery prior to any experimental treatment. Two additional animals were excluded due to lack of sufficient occlusion (TIMI score of 3), resulting in unabated flow through the coronary artery, thus insufficient ischemia and no myocardial infarction. There was no significant difference in survival between treated groups and untreated controls (Figure 2A), indicating no significant increase of mortality due to electrotransfer mediated gene delivery of VEGF-A encoding plasmid DNA to ischemic myocardium seven weeks post induction of ischemia.
Figure 2. GET of VEGF-A has no deleterious effects on survival or heart performance over 7 weeks post MI and gene delivery.
A) Kaplan-Meier Survival curve with no significant difference between the groups (Logrank test for trend, P=0.4639), B) Ejection fraction (P=0.9240), C) Cardiac Output (P=0.5603), D) Fractional Shortening(P=0.3775), E) Left ventricular end systolic volume (P=0.5603), F) Left Ventricular end diastolic volume(P=0.92), with no significant difference between treatment groups based on echocardiographic analysis (Two-way ANOVA and Tukey’s multiple comparison test, alpha=0.05)
Measurements of left ventricular diameters via echocardiography indicate no significant difference in calculated end-systolic and end-diastolic measures of volumes, ejection fraction, fractional shortening and cardiac output between GET treated, pVEGF-A injection without electrotransfer and sham control groups at any time point throughout the study (Figure 2B-F), indicating no additional damage caused by GET mediated pVEGF-A to ischemic myocardium of the left ventricle. Other cardiac ultrasound parameters are also consistent with this observation (Supplemental Figure 1).
GET of VEGF-A induces angiogenesis in ischemic myocardium in a porcine model
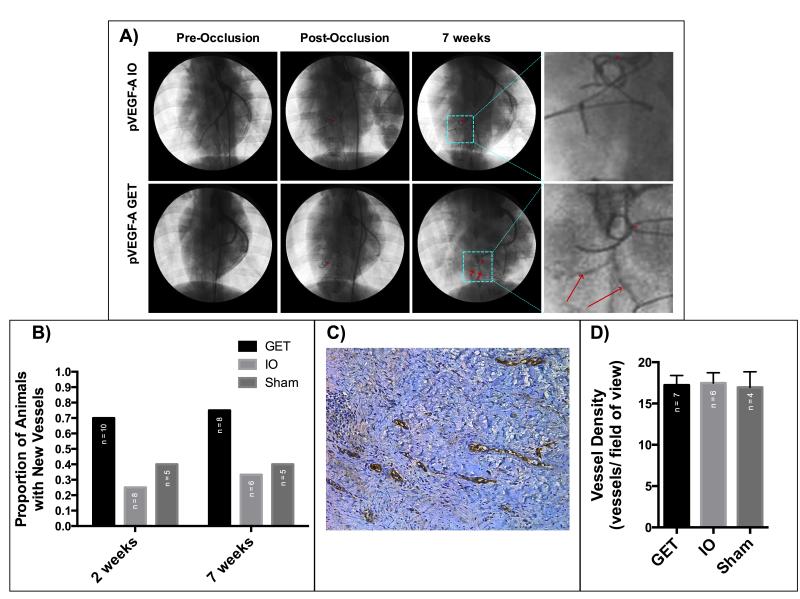
Arteriograms taken prior to LAD coronary artery occlusion were compared to arteriograms taken two and seven weeks after LAD coronary artery occlusion and gene therapy treatment. More than 70% of GET treated animals developed new arteries large enough to be visible via arteriogram of the heart, while only 25% and 40% of the pVEGF-A injection only and saline injection controls, respectively, developed new vessels (Figure 3A and 3B). The average length of new vessels as visible in arteriograms was not significantly different between treatment groups for animals that did have new vessel growth (Supplemental Figure 2).
Figure 3. GET of pVEGF-A induces angiogenesis in ischemic myocardium at treatment sites.
A) Arteriograms of GET and IO animals, before occlusion, immediately after occlusion and 7 weeks after occlusion, with new arteries visible in the GET heart. *LAD occlusion site, red arrows point to new vessels. B) The proportion of hearts with new arteries visible via arteriogram 2 (P=0.152313) and 7 weeks (P=0.243917) after treatment is higher in hearts treated with GET of pVEGF-A (Chi-square test). C) vWF staining for blood vessels at a treatment site of GET of pVEGF-A seven weeks after MI and treatment. D) Blood vessel density at treatment sites seven weeks after MI and treatment is not significantly different between treatment groups on a microscopic level (One-way ANOVA, alpha=0.05, P=0.9704).
Histological comparison of vessel density in ten randomly selected fields of view from treatment sites indicate no significant difference in blood vessel density between the groups seven weeks after induction of myocardial infarction and gene therapy treatment (Figure 3C and 3D).
GET of pVEGF-A reduces myocardial damage from ischemic injury at treatment sites of the left ventricle
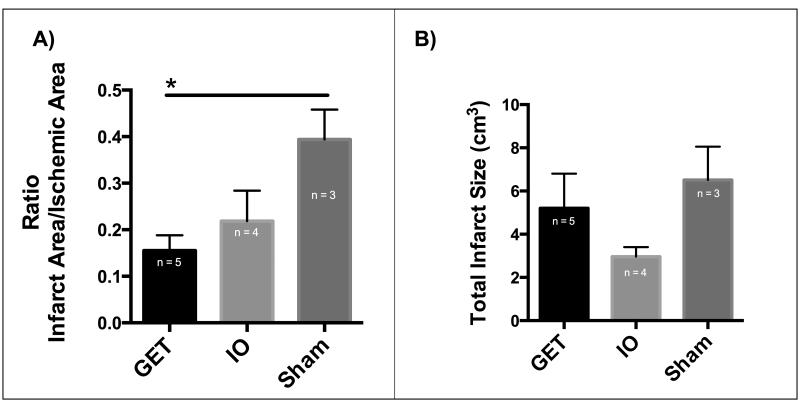
Myocardial infarction caused by occlusion of the LAD coronary artery extended through the anterior left ventricle, septum and right ventricle in most animals, based on TTC staining performed on harvested hearts seven weeks post LAD coronary artery occlusion and gene therapy treatment (Supplemental Figure 3). The infarct/ischemic area in the left ventricle post LAD occlusion is significantly lower in GET of pVEGF-A group than the saline injection group and is lower than the pVEGF-A injection without electrotransfer (4B). Therefore, the infarct size in GET with pVEGF-A group at the treatment sites is smaller than the infarct size of the control groups.
The total volume of myocardial infarction was not significantly different between the experimental groups (Figure 4A). There is notable variability in the extent of the infarction into the right ventricle and septum, which were untreated affecting the total size of the infarct (Supplemental Figure 3).
Figure 4. Myocardial infarct size is reduced at GET of pVEGF treatment sites 7 weeks post treatment.
A) Infarct area to ischemic area ratio is significantly lower in GET treated left ventricles than in Sham control group, (One-way ANOVA, and Tukey’s multiple comparison test, alpha=0.05, p=0.0349), B) Total myocardial infarct size including untreated septum and untreated right ventricle, with no significant differences between the groups (One-way ANOVA, alpha=0.05, P=0.2028).
GET of pVEGF-A improves left ventricular contractility and relaxation under dobutamine stress
While the heart rate of all animals irrespective of the treatment group increased dramatically in response to dobutamine administration, the change in intra-ventricular pressure correlated inversely with total size of infarct in the left ventricle. Higher ventricular compliance was observed for GET (n=2) treated hearts than controls (n=3), based on higher ±dP/dtmax measurements as indicators of contractility and relaxation in response to increasing dobutamine dose (Supplemental Figure 4).
Discussion
Atherosclerosis of coronary arteries with stable or unstable atherosclerotic plaques often leads to ischemia of the myocardium or acute myocardial infarction respectively. Sufficient hypoxic conditions result in loss of cardiomyocytes and remodeling of the myocardium into non-contractile scar tissue. Sufficient loss of contractile function of the heart leads to heart failure and reduced long-term survival. Restoration of blood flow is a common strategy for clinical treatments such as coronary artery bypass grafting (CABG), however such procedures do not address the problem of atherosclerosis. Autologous blood vessels (ex. saphenous vein), often used for bypassing blocked arteries suffer the same atherosclerotic disease as the original vessels, can and often do develop atherosclerotic plaques. An angiogenic therapy is an attractive option due to the potential to alleviate ischemia in short term and to cultivate new autologous vessels, which are not yet effected by atherosclerosis, similar to blood vessels of younger individuals, who are not likely to suffer from atherosclerosis. Here, we demonstrate an angiogenic gene therapy approach that results in development of new arteries in ischemic myocardium with minimal adverse effects.
Our gene delivery approach is non-viral and non-integrating with temporary elevation of local gene expression up to two weeks 21. We injected naked plasmid DNA encoding human VEGF-A to four sites in the ischemic myocardium in a porcine animal model, followed by electrotransfer. In our previous studies we have shown that GET greatly enhances gene expression of VEGF-A compared to plasmid DNA injection without electrotransfer in a porcine model of myocardial infarction. Here, we evaluated long term impact of GET mediated delivery of pVEGF-A on ischemic myocardium.
The occlusion of the LAD coronary artery resulted in consistent ischemia of the left ventricle in terms of the area affected and drop in perfusion observed, both measured with SPY perfusion analysis system. There were no significant differences between ischemia of the left ventricle between the groups of animals. Ischemia of the septum and the right ventricle were not assessed, due to limitations of the SPY imaging system. Only the apical surface of the left ventricle is visible via the SPY system, thus only perfusion through that area can be assessed before and after the occlusion. Elevated plasma levels of cardiac troponin I and CK-MM one day after LAD coronary artery occlusion indicated that induction of ischemia also resulted in myocardial infarction. Therefore, the occlusion of the LAD coronary artery resulted in consistent ischemia and acute myocardial infarction for all treatment groups.
Seven-week survival was evaluated with the Kaplan-Meier log-rank evaluation. Mortality was not significantly different between the treatment groups, indicating that GET of VEGF-A did not adversely effect survival, or increase mortality compared to sham in DNA injection only groups. This finding is consistent with our previous studies, indicating that proper administration of electric pulses for gene therapy during the rise of the R-wave, does not increase mortality, neither does overexpression of pVEGF-A at treatment sites of ischemic myocardium. Therefore, the GET of pVEGF-A can be safely administered to ischemic myocardium as a potential therapeutic.
Echocardiography evaluation of ejection fraction, fractional shortening, end-systolic and end diastolic volumes, cardiac output, and hypertrophy, indicates no significant difference between treatment groups at any time points of the study. The occlusion distal to the second diagonal branch, resulted in a relatively mild form of ischemia leading to low mortality and allowing for evaluation of treatment effects. Variability in ischemia to the septum and the right ventricle, may play a role in lack of significant differences in end-systolic and end-diastolic diameters and calculated volumes, ejection fraction and cardiac output measures between the groups. Increase in cardiac output, end-systolic and end-diastolic volume at seven weeks compared to the pre myocardial infarction values can also be caused by growth of the entire animal and the heart, since most animals grew throughout the duration of the study. Significantly, however, there were no adverse effects observed via echocardiography in GET of VEGF-A treated animals compared to the control animals, indicating safety of the treatment.
While damage to the myocardium from ischemia occurs within hours of the onset of ischemia, with some indication that necrosis of cardiomyocytes can also start within hours after onset of ischemia, angiogenesis is not induced in the remodeling myocardium until about 2-3 weeks from MI onset, with maturation of the new vessels taking even longer 26. An angiogenic therapy with an onset earlier than what happens during the endogenous course of remodeling of ischemic myocardium, may prove to be extremely beneficial therapeutically. Here, we note the presence of new arteries in the distal regions of the ischemic myocardium as early as 2 weeks after occlusion of the LAD coronary artery (Figure 3). Arteriogram analysis (Figure 3A-B) indicates presence of arteries distal to the location of the occlusion in the left ventricle. These vessels were not present in the arterial tree of the same hearts prior to occlusion and treatment, indicating angiogenesis in more than 70% of the hearts treated with GET of pVEGF-A at seven weeks. In comparison only ~25% and ~40% of the hearts treated with injection of pVEGF-A or saline, respectively, developed new arteries in the same time frame (Figure 3B). Arteriograms can only detect macroscopically sized vessels with radiopaque dye, thus onset of angiogenesis is actually earlier than two weeks, in order to observe functional arteries via arteriogram analysis (Figure 3A-B). Also, due to the size (approximately 0.5-1mm inner diameter) and abundant perfusion of the detected arteries it is likely that these new collateral arteries are mature and efficient.
Histological analysis for blood vessel density was only performed at seven weeks, for observation of long-term effects of GET of pVEGF-A on the myocardium (Figure 3C). By seven weeks vessel density at treated sites is indistinguishable between the gene therapy group and corresponding controls (Figure 3C-D). Based on our previous data 21, exogenous gene expression is expected to persist for up to two weeks, therefore homeostatic processes are expected to take over after exogenous gene expression subsides. Blood vessel density generally depends on oxygen requirements of the tissue 27, therefore once hypoxic conditions and/or exogenous growth factors no longer drive angiogenesis, it is likely that the tissue no longer requires angiogenic processes. This is consistent with our results of similar blood vessel density on the microscopic level at 7 weeks after LAD coronary artery occlusion and gene therapy application.
VEGF-A has a well-known role in angiogenesis in response to hypoxic conditions, however there are also multiple reports of VEGF-A promotion of cardiomyocyte proliferation as well as induction of differentiation of cardiac progenitor cells or cardiac stem cells into cardiomyocytes. Evaluation of the total size of the infarct including the myocardium of the right ventricle, septum and left ventricle yielded no significant differences between treatment groups, however there was considerable variability in the degree to which the right ventricle and the septum were effected by LAD coronary artery occlusion (Supplemental Figure 3), with some hearts having larger or smaller infarcts in those regions. Neither the right ventricle nor septum was evaluated for ischemia prior to treatment with SPY analysis. These regions contained no treatment sites, thus the variability in myocardial infarct size in these regions is not due to treatment group (Figure 4A). This variability may also account for little distinction between treatment groups via seven-week echocardiography evaluation (Figure 2B-2F and Supplemental Figure 1). Therefore, we evaluated ischemia and infarct size of the left ventricle separately from other infarcted regions in order to assess the effect of GET of VEGF-A at the treatment sites. Treatment sites were only present in the left ventricle. There was a significant decrease in infarct size in terms of surface area of infarct at seven weeks relative to the initial area of ischemia of the left ventricle, which is the site of GET treatment (Figure 4B). The reduction of myocardial infarct area of the left ventricle was larger in the GET of VEGF-A group than the pVEGF-A injection only, or the saline injection (sham) control groups. The difference between GET and sham group is statistically significant (p=0.0349), implicating a therapeutic potential for GET of pVEGF-A treatment.
A strong correlation between the presence of new arteries and reduction of infarct area was not observed, suggesting another role for exogenous VEGF-A in repair of the myocardium after ischemic injury. VEGF-A may preserve viability of resident cardiomyocytes, induce cardiomyocyte proliferation in adjacent normoxic regions, promote migration and differentiation of cardiac stem/progenitor cells to cardiomyocytes, or a combination of these three. It has been noted that VEGF-A is capable of promoting proliferation of cardiomyocytes 13, and induction of differentiation of cardiac stem cells or progenitor cells to cardiomyocytes 15. This role of VEGF-A may account for the reduction of myocardial infarct size in vivo with recruitment of new cardiomyocytes to the hypoxic myocardium in addition to the traditional angiogenic role.
Ventricular contractility and relaxation are extremely important to quality of life. The ability of the heart to respond to increased demand in oxygen due to stress such as exercise determines patients’ daily physical abilities. Therefore, we evaluated contractility and relaxation of infarcted hearts under increasing dobutamine stress seven weeks after treatment. GET treated animals had a higher contractility and relaxation in response to increased stress than the controls (Supplemental Figure 4A-B). While our sample size is very limited, these results are consistent with our other findings, that there are more contractile cells present in the treated left ventricles, leading to improved contractility of the left ventricle in the treated groups, compared to control groups.
In summary, gene electrotransfer of VEGF-A encoding plasmid applied to left ventricular ischemic myocardium has no deleterious effects on long term survival compared to injection of VEGF-A encoding plasmid without electrotransfer, or injection of saline. There is no significant difference in echocardiographic measures between treatment groups at any time point, possibly due to initial variation in ischemia to the septum and the right ventricle. The proportion of animals with new arteries is much higher in GET group than in any of the control groups as early as two weeks and persisting through seven weeks post treatment. By seven weeks, there is no difference in blood vessel density between treatment groups at the sites of treatment on the microscopic level. Overall myocardial infarct size is not significantly different between treatment groups, however there is a difference in reduction of left ventricular area of infarct. Myocardial infarct area reduction in GET group is significantly larger, indicating a potential therapeutic benefit for this gene therapy approach. Improved left ventricular contractility and relaxation under dobutamine stress further support therapeutic application potential of GET of pVEGF-A to ischemic myocardium for increasing angiogenesis and myocardial repair and regeneration. This study supports safety and therapeutic potential for using gene electrotransfer to deliver plasmid DNA encoding VEGF-A to ischemic myocardium in a porcine model.
Methods
Animals
Thirty-seven adult Yorkshire pigs were purchased from Bellview Farms, Smithville, Virginia at an approximate weight of 31-47kg. All experimental studies followed an approved Old Dominion University’s Institutional Animal Care and Use Committee protocol, in accordance with the Guide for the Care and Use of Laboratory Animals at an AAALAC-accredited facility, including a 12:12-hour light cycle. An ACLAM board certified veterinarian determined all animals were free of disease through the use of health examinations. Animals were quarantined and acclimated for a 7-day period before any procedures were conducted. In addition, an echocardiography exam was conducted on each animal to obtain baseline measures of cardiac health.
Plasmid
A commercially prepared plasmid, pVax1-hVEGF165(pVEGF-A), was used for these studies (Aldevron, Fargo, ND). DNA was suspended in sterile saline at 2mg/ml. Endotoxin levels were <0.1 EU/μg plasmid, confirmed by Aldevron via a Limulus Amebocyte Lysate assay.
Myocardial Infarction
Surgical procedure and induction of acute ischemia were performed as previously described 21, with more details available in supplemental methods. After induction of anesthesia by ketamine (20mg/kg) and diazepam (3-5mg/kg), pigs were anesthetized with isoflurane via a nose mask and intubated, with respiration maintained by a volume-controlled BonAir mechanical ventilator. A medial sternotomy incision was made for exposing the heart. The sternotomy was chosen less invasive methods, to allow adequate view of the entire ischemic surface area for SPY measurements. The pericardium was removed to expose LAD coronary artery. The LAD was ligated with a 4-0 silk suture below the second diagonal branch, inducing downstream ischemia, with the suture left in place following treatment. A radiopaque thread was tied to the silk ligature for marking the LAD occlusion site in x-ray imaging. Ischemic areas of the heart wall were identified by perfusion assessment after the ligation of the LAD by SPY and arteriograms.
Perfusion Assessment
Tissue perfusion through the left ventricular wall was assessed with the SPY® System (LifeCell Corporation, Branchburg, NJ) prior and immediately after the occlusion of the LAD. Indocyanine green (IC-Green) fluorescent dye was injected intravenously followed by SPY scanning of the anterior left ventricular wall for the presence of the dye. Higher fluorescence intensity was interpreted as higher perfusion. The images collected prior to the LAD ligation and immediately after the ligation were used to determine the relative loss in perfusion, the precise location and size of ischemic area in the left ventricle (ischemia of the septum or the right ventricle are not visible in the field of view of the SPY).
Arteriograms
X-ray fluoroscopy (Advantx® Systems Fluoroscope, GE Medical Systems, Wauwatosa, WI) was utilized to view the coronary arteries of the heart prior to LAD occlusion, after the occlusion, 2 and 7 weeks after myocardial infarction. The femoral artery was accessed via a cut-down followed by a modified Seldinger technique with a J-tipped angiographic guidewire (Bard Medical, Covington, GA). A 6-French catheter (Boston Scientific, Marborough, MA) was guided to the LAD. Arteries were visualized with injection of Isovue-300 iodine contrast agent (Bracco Diagnostics Inc., Monroe Township, NJ) into the catheter. A TIMI score of 0, 1, 2 or 3 was assigned to assess flow through the LAD immediately after the occlusion, at 2 weeks and at 7 weeks. A score of 0 indicated no flow past occlusion site, a score of 1 indicates slow flow past the occlusion site, but no filling of the distal regions of the arteries, a score of 2 indicates slow flow past the occlusion with filling of the distal arteries, and a score of 3 indicates unimpeded flow 22. Arteriograms performed prior to occlusion and after 7 weeks were compared to assess angiogenesis at the macroscopic level. The number of animals with new vessels was recorded along with the vessel length and compared among different experimental groups.
Gene Electrotransfer
Gene electrotransfer, plasmid DNA injection only, or sham injection of saline were administered following SPY perfusion assessment, within an hour of LAD occlusion. This time point was chosen to minimize stress to the animals, by performing only one invasive, open-heart procedure and avoiding separate surgeries for LAD occlusion and GET treatment. Four sites bordering ischemia were selected for GET. A 7mm, 4 needle (5 mm electrode gap) penetrating electrode with an injection port was used for DNA delivery to the heart as previously described 20, 21. For each treatment site, the electrode was placed on the surface of the myocardium, with electrode needles penetrating into the myocardium to 7mm. A hypodermic needle was used to inject 100μl of plasmid DNA encoding VEGF-A (pVEGF-A) in saline at 2mg/ml, centrally between the electrode needles through the injection port. The hypodermic needle was withdrawn, to prevent electrical interference. An electric field was established between and around the electrode needles with applied voltage to accomplish electroporation and DNA delivery at the treatment site. Our custom-built pulse generator and software continuously captured the echocardiogram (ECG) of the animal and enabled synchronizing pulsing with the R-wave of the ECG (Accusync Medical Research Company, Milford, CT). Each site was immediately pulsed eight times with 20ms pulses at an amplitude of 60V. Animals randomly assigned to the negative control groups received either saline injections or plasmid DNA injection without electrotransfer.
Immunofluorescence Staining
Seven weeks post myocardial infarction, animals were euthanized and tissue samples were collected, fixed in 10% formalin, paraffin embedded and sectioned. Slides were stained for von Willebrand Factor (vWF) to identify microvasculature with a blood vessel staining kit (Millipore, Billerica, MA). The number of vessels per field of view was counted and averaged over 10 randomly selected fields of view within each treatment region. Experimental groups were then compared based on the average number of vessels for vessel density.
Echocardiography
Cardiac ultrasound was performed with a Sonosite 180 Plus SonoHeart Ellite Ultrasound (Bothell, WA) 21, 23 prior to the occlusion of the LAD, immediately after myocardial infarction, two weeks after MI and 7 weeks after MI. Two dimensional and M-mode echocardiography was used to measure left ventricular end-systolic diameter, left ventricular end-diastolic diameter, left ventricular wall (LVPW) thickness, and intraventricular septum during systole and diastole. The Teicholz method was used to calculate left ventricular end-systolic volume (LVESV), left ventricular end-diastolic volume (LVEDV), cardiac output (CO), fractional shortening (FS), stroke volume and ejection fraction (EF) 24.
Enzyme-linked Immunosorbent Assays
Blood samples were collected prior to MI, 24 hours post occlusion and at 7 weeks, for analysis of changes cardiac protein levels. Plasma samples were tested for porcine cardiac troponin-1 (Kamiya Biomedical, Seattle, WA) and creatine kinase MM (Kamiya Biomedical, Seattle, WA) according to the ELISA kit manufacturer’s instructions, for indications of initial myocardial damage and presence of heart failure.
Myocardial Infarct Size Measurements
At seven weeks, hearts were harvested for infarct size assessment and histological analysis. Harvested hearts were sectioned into 1cm coronal slices (perpendicular to long axis of the heart). The slices were incubated with 1% triphenyltetrazolium (TTC) (Sigma-Aldrich, St Louis, MO) at 37°C for 30 minutes. Each side of each slide was then photographed and the infarct size was measured using ImageJ software to trace the infarct in each image.
In order to calculate the total volume of the infarct, the area measurement from each side of individual slices was averaged and multiplied by the thickness of the slice. The total infarct volume is the sum of individual infarct volumes from each heart slice 25. The total myocardial infarct volume included measurements from both ventricles and the intraventricular septum.
Left ventricular MI surface area was also measured in order to compare the area of left ventricular ischemia induced by occlusion of the LAD to the area of MI after treatment of the left ventricle. ImageJ software was used to outline the perimeter of the MI on the pericardial surface of the left ventricle from each slice. The average from each side of the slice was multiplied by the thickness of the slice. The sum of areas from each slice was then used to determine the area of left ventricular MI.
End of Study Euthanasia
On final day of study, animals were anesthetized as follows. After induction of anesthesia by ketamine (20mg/kg) and diazepam (3-5mg/kg), pigs were anesthetized with isoflurane via a nose mask and intubated, with respiration maintained by a volume-controlled BonAir mechanical ventilator. A final arteriogram was performed followed by sternotomy as described above. Following final assessment of perfusion by SPY procedure, animal was euthanized by an intracardiac injection of 5 ml of fatal plus.
Statistical Analyses
All quantitative data with calculated means were compared using a one-way or two-way analysis of variance (ANOVA) with p<0.05 considered to be statistically significant. For ANOVA analysis yielding statistically significant differences, the Tukey’s multiple comparisons test was performed to determine which data sets were significantly different. All quantitative data was reported with the standard error of the mean (SEM). All proportions were compared with the Chi-Square test for proportions, with p<0.05 considered significant.
Supplementary Material
Acknowledgements
The authors would like to thank Dr. Mark Jaroszeski of University of South Florida, Tampa, FL for design and construction of the electrode arrays used for gene electrotransfer. We would also like to thank RaShae Cooke, Danielle Tajirian, Ben Cuffee and Ann Nix for providing excellent veterinary care to the animals throughout this study. We would also like to thank Dr. Robert Strange Jr. of Portsmouth Naval Facility, Portsmouth, VA and Dr. Paul D. Mahoney of Sentara Heart Hospital, Norfolk, VA for surgical and arteriogram acquisition training.
Funding for this work came from the National Institutes of Health R33 HL005441. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Footnotes
Conflict of Interest The authors declare no conflict of interest
Supplementary Information Supplementary information is available at Gene Therapy’s website
References
- 1.Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Borden WB, et al. Heart disease and stroke statistics--2013 update: a report from the American Heart Association. Circulation. 2013;127:e6–e245. doi: 10.1161/CIR.0b013e31828124ad. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.2012 NHLBI Morbidity and Mortality Chart Book. 2013;2014 [Google Scholar]
- 3.Krumholz HM, Merrill AR, Schone EM, Schreiner GC, Chen J, Bradley EH, et al. Patterns of hospital performance in acute myocardial infarction and heart failure 30-day mortality and readmission. Circ Cardiovasc Qual Outcomes. 2009;2:407–413. doi: 10.1161/CIRCOUTCOMES.109.883256. [DOI] [PubMed] [Google Scholar]
- 4.Ross R. The pathogenesis of atherosclerosis: a perspective for the 1990s. Nature. 1993;362:801–809. doi: 10.1038/362801a0. [DOI] [PubMed] [Google Scholar]
- 5.Mukherjee D, Bhatt DL, Roe MT, Patel V, Ellis SG. Direct myocardial revascularization and angiogenesis--how many patients might be eligible? Am J Cardiol. 1999;84:598–600. A8. doi: 10.1016/s0002-9149(99)00387-2. [DOI] [PubMed] [Google Scholar]
- 6.Wolfram JA, Donahue JK. Gene therapy to treat cardiovascular disease. J Am Heart Assoc. 2013;2:e000119. doi: 10.1161/JAHA.113.000119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kocher AA, Schuster MD, Szabolcs MJ, Takuma S, Burkhoff D, Wang J, et al. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med. 2001;7:430–436. doi: 10.1038/86498. [DOI] [PubMed] [Google Scholar]
- 8.Mercola M, Ruiz-Lozano P, Schneider MD. Cardiac muscle regeneration: lessons from development. Genes Dev. 2011;25:299–309. doi: 10.1101/gad.2018411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9:669–676. doi: 10.1038/nm0603-669. [DOI] [PubMed] [Google Scholar]
- 10.Liu W, Xu J, Wang M, Wang Q, Bi Y, Han M. Tumor-derived vascular endothelial growth factor (VEGF)-A facilitates tumor metastasis through the VEGF-VEGFR1 signaling pathway. Int J Oncol. 2011 doi: 10.3892/ijo.2011.1138. [DOI] [PubMed] [Google Scholar]
- 11.Folkman J. Tumor angiogenesis: therapeutic implications. N Engl J Med. 1971;285:1182–1186. doi: 10.1056/NEJM197111182852108. [DOI] [PubMed] [Google Scholar]
- 12.Tang JM, Wang JN, Zhang L, Zheng F, Yang JY, Kong X, et al. VEGF/SDF-1 promotes cardiac stem cell mobilization and myocardial repair in the infarcted heart. Cardiovasc Res. 2011;91:402–411. doi: 10.1093/cvr/cvr053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tao Z, Chen B, Tan X, Zhao Y, Wang L, Zhu T, et al. Coexpression of VEGF and angiopoietin-1 promotes angiogenesis and cardiomyocyte proliferation reduces apoptosis in porcine myocardial infarction (MI) heart. Proc Natl Acad Sci U S A. 2011;108:2064–2069. doi: 10.1073/pnas.1018925108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen Y, Amende I, Hampton TG, Yang Y, Ke Q, Min JY, et al. Vascular endothelial growth factor promotes cardiomyocyte differentiation of embryonic stem cells. Am J Physiol Heart Circ Physiol. 2006;291:H1653–8. doi: 10.1152/ajpheart.00363.2005. [DOI] [PubMed] [Google Scholar]
- 15.Urbich C, Aicher A, Heeschen C, Dernbach E, Hofmann WK, Zeiher AM, et al. Soluble factors released by endothelial progenitor cells promote migration of endothelial cells and cardiac resident progenitor cells. J Mol Cell Cardiol. 2005;39:733–742. doi: 10.1016/j.yjmcc.2005.07.003. [DOI] [PubMed] [Google Scholar]
- 16.Muller OJ, Katus HA, Bekeredjian R. Targeting the heart with gene therapy-optimized gene delivery methods. Cardiovasc Res. 2007;73:453–462. doi: 10.1016/j.cardiores.2006.09.021. [DOI] [PubMed] [Google Scholar]
- 17.Yla-Herttuala S, Alitalo K. Gene transfer as a tool to induce therapeutic vascular growth. Nat Med. 2003;9:694–701. doi: 10.1038/nm0603-694. [DOI] [PubMed] [Google Scholar]
- 18.Ayuni EL, Gazdhar A, Giraud MN, Kadner A, Gugger M, Cecchini M, et al. In vivo electroporation mediated gene delivery to the beating heart. PLoS One. 2010;5:e14467. doi: 10.1371/journal.pone.0014467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tevaearai HT, Gazdhar A, Giraud MN, Fluck M. In vivo electroporation-mediated gene delivery to the beating heart. Methods Mol Biol. 2014;1121:223–229. doi: 10.1007/978-1-4614-9632-8_19. [DOI] [PubMed] [Google Scholar]
- 20.Hargrave B, Downey H, Strange R, Jr, Murray L, Cinnamond C, Lundberg C, et al. Electroporation-mediated gene transfer directly to the swine heart. Gene Ther. 2013;20:151–157. doi: 10.1038/gt.2012.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hargrave B, Strange R, Jr, Navare S, Stratton M, Burcus N, Murray L, et al. Gene electro transfer of plasmid encoding vascular endothelial growth factor for enhanced expression and perfusion in the ischemic Swine heart. PLoS One. 2014;9:e115235. doi: 10.1371/journal.pone.0115235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Anonymous The Thrombolysis in Myocardial Infarction (TIMI) trial. Phase I findings. TIMI Study Group. N Engl J Med. 1985;312:932–936. doi: 10.1056/NEJM198504043121437. [DOI] [PubMed] [Google Scholar]
- 23.Abraham J, Abraham TP. The role of echocardiography in hemodynamic assessment in heart failure. Heart Fail Clin. 2009;5:191–208. doi: 10.1016/j.hfc.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 24.Teichholz LE, Kreulen T, Herman MV, Gorlin R. Problems in echocardiographic volume determinations: echocardiographic-angiographic correlations in the presence of absence of asynergy. Am J Cardiol. 1976;37:7–11. doi: 10.1016/0002-9149(76)90491-4. [DOI] [PubMed] [Google Scholar]
- 25.Dib N, Diethrich EB, Campbell A, Gahremanpour A, McGarry M, Opie SR. A percutaneous swine model of myocardial infarction. J Pharmacol Toxicol Methods. 2006;53:256–263. doi: 10.1016/j.vascn.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 26.Turillazzi E, Di Paolo M, Neri M, Riezzo I, Fineschi V. A theoretical timeline for myocardial infarction: immunohistochemical evaluation and western blot quantification for Interleukin-15 and Monocyte chemotactic protein-1 as very early markers. J Transl Med. 2014;12 doi: 10.1186/1479-5876-12-188. 188-5876-12-188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Logsdon EA, Finley SD, Popel AS, Mac Gabhann F. A systems biology view of blood vessel growth and remodelling. J Cell Mol Med. 2014;18:1491–1508. doi: 10.1111/jcmm.12164. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.