Abstract
Background
In girls with Idiopathic Central Precocious Puberty (ICPP) concern has been raised by the potential impact of GnRH-analogues (GnRHa) treatment on body weight. We evaluated the effect of GnRHa on Body Mass Index (BMI) in girls with ICPP according to weight status at diagnosis.
Methods
One hundred seventeen ICPP girls were divided according to pretreatment weight status in: normal weight (NW), overweight (OW) and obese (OB). BMI at one and two years of treatment was assessed. BMI-SDS of 60 patients who reached adult height (AH) was compared to that of 33 ICPP untreated girls.
Results
NW girls significantly increased their baseline BMI-SDS at 1 and 2 years of treatment. OW girls only had a significant increment at one year of treatment while OB girls showed no BMI-SDS change. Patients evaluated at AH (at least four years after GnRHa withdrawal) showed a significant decrease on BMI compared to baseline and a significantly lower BMI than the untreated group.
Conclusion
In ICPP girls the BMI increase under GnRHa was inversely related to the pretreatment weight status. In the long term follow-up, no detrimental effect of GnRHa on body weight was observed. BMI-SDS was lower in treated than in untreated girls.
Keywords: Central Precocious Puberty, BMI, GnRH analogues
Background
Central Precocious Puberty (CPP) results from the premature activation of the hypothalamo-pituitary-ovarian axis. In girls, it is defined as the onset of secondary sexual development before the age of 8 with further progression, accompanied by increased growth velocity and bone age acceleration usually leading to adult height impairment.
Gonadotropin-releasing hormone analogues (GnRHa) are the treatment of choice in CPP and their effectiveness on adult height (AH) improvement has been widely recognized. However, concern has been raised by the potential impact of this treatment on body weight. While some studies have reported association between GnRHa treatment and Body Mass Index (BMI) increase [1–7], others have found no influence of GnRHa treatment on weight status [8–13] or even some decrease in BMI under therapy [14–16]. It has been demonstrated that girls with CPP are prone to developing obesity (8). A few reports have analyzed the influence of GnRHa on body weight according to BMI status at onset of treatment. However, in some of these studies follow-up was limited to a period under treatment or to a short time after withdrawal [17–19], whereas studies with a long-term follow-up did not categorize patients according to pre-treatment BMI status [20, 21].
The aim of the present study was to evaluate the impact of GnRHa therapy on BMI in girls with Idiopathic CPP (ICPP) according to weight status at diagnosis up to adult height (AH). The subgroup of patients who achieved AH was compared to a control group of untreated ICPP girls.
Patients and methods
Patients
This retrospective study was done reviewing the clinical charts of 117 girls with ICPP referred to the Division of Endocrinology of the Hospital de Niños Ricardo Gutiérrez (Buenos Aires, Argentina) from 1985 to 2010. All patients were treated for at least two years with intramuscular depot GnRHa (Triptorelin acetate), at a dose of 100–120 ug/kg, every 28 days for 2.8 ± 0.2 years; no patient received additional medications.
Inclusion criteria: patients with diagnosis of ICPP according to the following criteria: (1) onset of breast development before 8 years of chronological age (CA), (2) height velocity above the 97 centile for age and bone age (BA) advancement by at least one year over CA, (3) pubertal LH response to GnRH (≥ 6 mUI/ml), (4) uterine length ≥ to 35 mm. Exclusion criteria: organic central precocious puberty, congenital adrenal hyperplasia and any other underlying condition or medication that might affect body weight.
Patients were divided into three groups according to weight status at start of therapy: normal weight (NW); overweight (OW) and obese (OB). BMI at the end of first and second year of GnRH treatment was assessed. No specific life style intervention was recommended to any patient. At the time of reaching AH, auxological parameters in 60 patients were compared to those of a control group of 33 girls with ICPP who had not received treatment (age of menarche 9.5 ± 0.3).
Methods
Clinical and auxological features were assessed as follows: pubertal stage according to Marshall and Tanner criteria [22]; height in cm measured by a Harpenden stadiometer; weight using a calibrated scale; bone age according to Greulich and Pyle [23].
Weight status was determined as BMI and it was expressed as BMI-SDS using the software Auxology version 1.0 b 17 Copyright® 2003 Pfizer. Overweight was defined as BMI > 85th centile and obesity as BMI > 95th centile according to Cole criteria [24].
AH was defined when two successive height measurements six months apart were equal or less than 0.5 cm, and/or when BA was equal or greater than 15 years. This study fulfilled the requirements defined by the Ethical Committee of Hospital de Niños Ricardo Gutiérrez for retrospective studies. Reference number CEI 11.053.
Statistical analyses were performed using GraphPad Prism version 5.00 for Windows (GraphPad Software, San Diego, CA). Changes on BMI were analyzed by ANOVA for repeated measurements. Post hoc Tukey’s multiple comparison test was performed when differences were detected by ANOVA. Chi square test was used to compare the percentage of NW, OW and OB girls in the whole group at treatment start vs the percentage of NW, OW and OB girls in those who reached AH. The comparison between patients and control girls who reached AH was done using Mann Whitney test. P < 0.05 was considered statistically significant. Data is expressed as mean ± SD.
Results
A total of 117 girls with ICPP were studied. At treatment start all patients had breast development Grade II-III (Tanner) and three of them have had menarche. CA was 7.6 ± 1.3 years, BA 9.6 ± 0.16 years, height SDS 1.71 ± 0.08. The uterine length was 37.2 ± 0.7 mm.
Before treatment, BMI in the whole group was 18.5 ± 2.4 (SDS 1.1 ± 1). BMI distribution by groups showed: 48 % girls NW, 37 % OW and 15 % OB. No significant difference in chronological age was observed among groups, p: ns (Table 1).
Table 1.
Auxological characteristics of the whole group at start of treatment
| Age (years) | BMI at start (kg/m2) | BMI-SDS at start | |
|---|---|---|---|
| All patients (n = 117) | 7.6 ± 1.3 | 18.5 ± 2.4 | 1.1 ± 1 |
| Normal Weight (n = 56) | 7.53 ± 1.3 | 16.5 ± 1.3 | 0.3 ± 0.7 |
| Overweight (n = 43) | 7.33 ± 1.5 | 19.3 ± 4.7 | 1.5 ± 0.3 |
| Obese (n = 18) | 7.74 ± 0.8 | 22.4 ± 1.2 | 2.4 ± 0.3 |
The whole cohort of patients significantly increased their baseline BMI-SDS (1.1 ± 1) at year one (1.35 ± 0.95, p < 0.001) and year two (1.26 ± 0.1 p < 0.01) of treatment. When changes in BMI were analyzed according to the initial weight status, NW patients showed a significant increase in BMI-SDS from 0.3 ± 0.7 to 0.7 ± 0.8 at one year of treatment and 0.6 ± 0.8 at two years (p < 0.001). In OW girls a significant increase was only observed between baseline (1.5 ± 0.2) and one year of treatment (1.7 ± 0.5) (p < 0.05). OB girls showed no BMI-SDS changes during treatment (baseline 2.4 ± 0.3, year one 2.4 ± 0.4 and year two 2.3 ± 0.6) (Fig. 1).
Fig. 1.

BMI evolution of patients with ICPP during treatment with GnRHa. a All, b Normal weight, c Overweight, d Obese
A subset of 60 girls were followed up to AH [160.4 ± 0.7 cm (SDS −0.01 ± 0.14)], which was achieved at 4.4 ± 0.4 years after GnRHa withdrawal. BMI composition at beginning of treatment of the group of girls who attained AH was not different from that of the whole group (NW 47 % vs 48 %, OW 33 % vs 37 %, OB 20 % vs 15 % respectively, p = ns). All girls in the AH group showed a significant decrease on BMI-SDS at the moment of reaching AH (0.5 ± 1.1) compared to BMI-SDS at start (1.0 ± 1.1), p < 0.001. A similar pattern was observed when the BMI status was analyzed in each subgroup (Table 2) (Fig. 2). The BMI-SDS of the 60 treated patients who reached adult height was 0.5 ± 1.1, significantly lower than the BMI-SDS of the untreated control group, 1.3 ± 1.00 (p = 0.0004) (Fig. 3).
Table 2.
Auxological characteristics in girls who reached adult height (AH)
| BMI at start (SDS) | BMI at AH (SDS) | |
|---|---|---|
| All patients (n = 60) | 1.0 ± 1.1 | 0.5 ± 1.1 |
| Normal Weight (n = 28) | 0.25 ± 0.8 | −0.19 ± 0.7 |
| Overweight (n = 20) | 1.4 ± 0.2 | 0.7 ± 0.7 b,c |
| Obese (n = 12) | 2.4 ± 0.4 | 1.57 ± 1.1 a |
ANOVA a < 0.0001 vs NW, b 0.001 vs NW, c 0.03 vs OB. Unpaired t Test start vs at AH: All patients p 0.004, NW p 0.07, OW p 0.0004, OB p 0.03
Fig. 2.

BMI evolution during treatment in 60 patients at the time of reaching Adult Height. a All, b Normal weight, c Overweight, d Obese
Fig. 3.
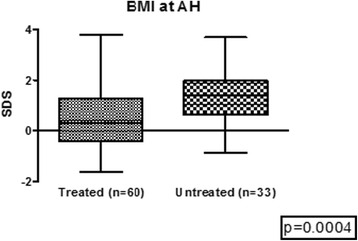
BMI at adult height treated vs. untreated patients
The evolution of the weight status from start to AH in each category is shown in Table 3.
Table 3.
Evolution of BMI category during follow-up in each subgroup
| At start | At Adult Height (N° of patients) |
|---|---|
| NW n = 28 | NW 27 |
| OW 1 | |
| OB 0 | |
| OW n = 20 | NW 14 |
| OW 5 | |
| OB 1 | |
| OB n = 12 | NW 4 |
| OW 4 | |
| OB 4 |
Discussion
The present study evaluates the long-term effect of a monthly depot GnRHa therapy on BMI in a large cohort of 117 girls with ICPP from a single endocrine pediatric center in relation to weight status at diagnosis, along treatment and at AH in a subset of 60 patients.
Girls with ICPP at the moment of diagnosis usually have a high prevalence of overweight and obesity [17–19, 25], although if early puberty is the cause or the consequence of the increased body fat remains unclear. In agreement with the literature, before treatment our patients had an elevated proportion of overweight and obesity, 37 and 15 % respectively, these rates are higher than those of average Argentinian girls of the same age, 20.8 and 5.4 % [26].
The impact of GnRHa treatment on body weight in our patients was clearly related to the pretreatment condition. Although the whole cohort of ICPP patients showed a BMI increase along treatment, the negative effect of the GnRHa seemed to decrease as the pretreatment weight was higher. Girls in the NW subgroup showed an increase of BMI at one and two years of treatment while OW girls had a higher BMI only at year one, while no changes were observed in OB girls along the treatment.
The inconsistency in the results about the effect of GnRHa on body weight found in the literature could be related to differences in the studied populations, i.e., mixed early and precocious puberty, idiopathic and organic CPP, and/or comparison with heterogeneous control groups.
While some authors found no changes in body weight on ICPP patients under GnRHa [8–13], few studies categorized patients according to pretreatment BMI to analyze the effect of treatment. Some reports showed that OW/OB girls at baseline remained unchanged or increased at the end of treatment, without any reference to NW patients [27, 28].
Lee et al. studied a group of girls with CPP (including 3 with organic CPP) during 18 months on treatment with GnRHa, and showed a significant BMI increase in the whole group, higher although not significant, in NW than in OW patients [18].
Wolters et al. evaluated a mixed group of girls with early or precocious puberty treated with GnRHa during one year according to pretreatment weight status and compared the OW subgroup to an OW untreated control group. In agreement with our study, their whole population of girls with CPP had a significant BMI increase along treatment, as well as the NW subset patients, whereas OW girls showed no changes in BMI while the OW control group showed a BMI increase [17].
Colmenares et al. analyzed 37 girls with ICPP and 34 girls with early puberty under GnRHa treatment, compared to a heterogeneous control group of untreated girls (3 with CPP who refused treatment, 2 with slowly progressive CPP and 20 with early slowly progressive puberty). In this study 72.9 % of the girls with ICPP were OW or OB, a percentage significantly higher than that of OW/OB girls with early puberty (35.3 %). The entire group of treated ICPP girls showed no changes in BMI at one and two years of treatment, whereas an increase at year three was observed compared to the control group. However, no individual analysis was performed in relation to the weight status at baseline [19].
The few reports evaluating body weight status in ICPP patients after GnRHa treatment showed return to pretreatment values [27] or no differences in BMI when compared to untreated control girls [20, 29]. Lazar et al. analyzed the evolution of a large cohort of CPP treated patients at the third to fifth decades of life and found that although the BMI was increased at start and during early treatment, it normalized thereafter [30]. None of the studies with a long-term follow-up analyzed BMI in relation to the weight status at the beginning of treatment.
Our subgroup of 60 patients evaluated at least four years after GnRHa withdrawal, when they had reached AH, showed a significant decrease in BMI compared to pretreatment BMI. The same pattern was observed when BMI status was analyzed for each subgroup. Approximately 2/3 of OW and 2/3 of OB patients showed a decrease in weight status, whereas only 1 /28 NW girls had become OW at AH. Furthermore, this subgroup with a long-term follow-up had a significantly lower BMI than the control group of untreated CPP girls, both compared at AH.
Categorizing our patients according to their weight status at start, along and after treatment allowed us to observe that only NW girls increased their BMI during treatment, while OW and OB girls did not, despite no changes in dietary habits or exercise were prescribed. Wolters et al. found a similar BMI behavior in CPP treated patients. The BMI of their OW girls (authors did not categorize between OW and OB) remained stable while a control group of untreated girls showed a significant BMI increase during follow-up, and they suggested a positive effect of GnRHa on weight status in OW children [17]. We did not have a control group of untreated patients evaluated as from the moment of diagnosis, but the comparison between our treated CPP girls with our group of untreated patients at AH status showed a significantly lower BMI among treated girls, thus seeming to confirm Wolters´s suggestion.
A limitation in our study was that we were able to analyze the body composition as a marker of nutritional status only on a small group of girls. There are a few reports evaluating the changes in body composition in CPP under GnRHa treatment. Van der Sluis et al. observed an increase of fat mass (estimated by DEXA) during treatment and a normalization after withdrawal. Chiocca et al. analyzed a cohort of CPP girls after GnRHa treatment at near final height and found no changes in BMI but an increase of total fat mass compared to a control group. Both authors suggested that the body fat accumulation was related to the estrogen depletion produced by GnRHa, as a “menopausal effect” [5, 14].
At present, there is no clear explanation of the different effects produced by GnRHa treatment on lean and overweight ICPP girls. Although no specific prescription on life style was given to our patients, it is not possible to rule out that the fact that OW/OB girls did not increase their BMI under treatment could have been related to a family concern about weight gain leading to changes in dietary or physical activity.
Although in the present study, we did not systematically analyze the metabolic profile of our patients, Colmenares et al. found no differences for glucose and lipids at baseline and during follow-up among treated children [19].
Conclusions
In conclusion, in our study, although a moderate increase in BMI was observed throughout the first and second year on GnRHa treatment, mainly in NW girls, such increase did not lead to further obesity since a significant improvement on BMI was observed when girls achieved adult height. Furthermore, the fact that treated girls had a lower BMI than untreated girls at adult height allowed us to rule out a long-term detrimental effect of GnRHa treatment on body mass in girls with Central Precocious Puberty. Further studies analyzing changes in body composition are desired.
Abbreviations
AH, adult height; BA, bone age; BMI, Body Mass Index; CA, chronological age; CPP, Central Precocious Puberty; GnRHa, GnRH-analogues; ICPP, Idiopathic Central Precocious Puberty; NW, normal weight; OB, obese; OW, overweight
Acknowledgements
Not applicable.
Funding
This research has no sources of funding.
Availability of data and materials
Data will not be shared unless Editor-in-Chief or reviewers require it.
Authors’ contributions
All authors made substantial contributions to the conception and design of this data. All authors read and approved the final manuscript.
Authors’ information
All authors work at the Endocrinology Division of Hospital de Niños Ricardo Gutiérrez.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
This study fulfilled the requirements defined by the Ethical Committee of Hospital de Niños Ricardo Gutiérrez for retrospective studies. The reference number is CEI 11.053.
Contributor Information
A. J. Arcari, Phone: (+54-11) 4963-5931, Email: aarcari@cedie.org.ar, Email: ajarcari@hotmail.com
M. G. Gryngarten, Email: mgryngarten@cedie.org.ar
A. V. Freire, Email: freireav@yahoo.com.ar
M. G. Ballerini, Email: mgballerini@cedie.org.ar
M. G. Ropelato, Email: gropelato@cedie.org.ar
I. Bergadá, Email: ibergada@cedie.org.ar
M. E. Escobar, Email: meugeniaescobar@cedie.org.ar
References
- 1.Boot AM, De Muinck K-SS, Pols HA, Krenning EP, Drop SL. Bone mineral density and body composition before and during treatment with gonadotropin-releasing hormone agonist in children with central precocious and early puberty. J Clin Endocrinol Metab. 1998;83(2):370–373. doi: 10.1210/jcem.83.2.4573. [DOI] [PubMed] [Google Scholar]
- 2.Feuillan P, Jones J, Barnes K, Oerter-Klein K, Cutler G., Jr Reproductive axis after discontinuation of gonadotropin-releasing hormone analog treatment of girls with precocious puberty: long term follow-up comparing girls with hypothalamic hamartoma to those with idiopathic precocious puberty. J Clin Endocrinol Metab. 1999;84(1):44–49. doi: 10.1210/jcem.84.1.5409. [DOI] [PubMed] [Google Scholar]
- 3.Chiumello G, Brambilla P, Guarneri MP, Russo G, Manzoni P, Sgaramella P. Precocious puberty and body composition: effects of GnRH analog treatment. J Pediatr Endocrinol Metab. 2000;13(Suppl 1):791–794. doi: 10.1515/JPEM.2000.13.s1.791. [DOI] [PubMed] [Google Scholar]
- 4.Traggiai C, Perucchin P, Zerbini K, Gastaldi R, De Biasio P, Lorini R. Outcome after depot gonadotrophin-releasing hormone agonist treatment for central precocious puberty: effects on body mass index and final height. Eur J Endocrinol. 2005;153(3):463–464. doi: 10.1530/eje.1.01975. [DOI] [PubMed] [Google Scholar]
- 5.Chiocca E, Dati E, Baroncelli GI, Mora S, Parrini D, Erba P, Bertelloni S. Body mass index and body composition in adolescents treated with gonadotropin-releasing hormone analogue triptorelin depot for central precocious puberty: data at near final height. Neuroendocrinology. 2009;89(4):441–447. doi: 10.1159/000197862. [DOI] [PubMed] [Google Scholar]
- 6.Taşcilar ME, Bilir P, Akinci A, Köse K, Akçora D, Inceoğlu D, Fitöz SO. The effect of gonadotropin-releasing hormone analog treatment (leuprolide) on body fat distribution in idiopathic central precocious puberty. Turk J Pediatr. 2011;53(1):27–33. [PubMed] [Google Scholar]
- 7.Anık A, Çatlı G, Abacı A, Böber E. Effect of gonadotropin‑releasing hormone agonist therapy on body mass index and growth in girls with idiopathic central precocious puberty. Indian J Endocrinol Metab. 2015;19(2):267–271. doi: 10.4103/2230-8210.131770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Palmert MR, Mansfield MJ, Crowley WF, Jr, Crigler JF, Jr, Crawford JD, Boepple PA. Is obesity an outcome of gonadotropin-releasing hormone agonist administration? Analysis of growth and body composition in 110 patients with central precocious puberty. J Clin Endocrinol Metab. 1999;84(12):4480–4488. doi: 10.1210/jcem.84.12.6204. [DOI] [PubMed] [Google Scholar]
- 9.Heger S, Partsch CJ, Sippell WG. Long-term outcome after depot gonadotropin-releasing hormone agonist treatment of central precocious puberty: final height, body proportions, body composition, bone mineral density, and reproductive function. J Clin Endocrinol Metab. 1999;84(12):4583–4590. doi: 10.1210/jcem.84.12.6203. [DOI] [PubMed] [Google Scholar]
- 10.Lazar L, Kauli R, Pertzelan A, Phillip M. Gonadotropin-suppressive therapy in girls with early and fast puberty affects the pace of puberty but not total pubertal growth or final height. J Clin Endocrinol Metab. 2002;87(5):2090–2094. doi: 10.1210/jcem.87.5.8481. [DOI] [PubMed] [Google Scholar]
- 11.Glab E, Barg E, Wikiera B, Grabowski M, Noczyńska A. Influence of GnRH analog therapy on body mass in central precocious puberty. Pediatr Endocrinol Diabetes Metab. 2009;15(1):7–11. [PubMed] [Google Scholar]
- 12.Ko JH, Lee HS, Lim JS, Kim SM, Hwang JS. Changes in bone mineral density and body composition in children with central precocious puberty and early puberty before and after one year of treatment with GnRH agonist. Horm Res Paediatr. 2011;75:174–179. doi: 10.1159/000320039. [DOI] [PubMed] [Google Scholar]
- 13.Arani KS, Heidari F. Gonadotropin-releasing hormone agonist therapy and obesity in girls. Int J Endocrinol Metab. 2015;13(3):e23085. doi: 10.5812/ijem.23085v2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.van der Sluis IM, Boot AM, Krenning EP, Drop SL, de Muinck Keizer-Schrama SM. Longitudinal follow-up of bone density and body composition in children with precocious or early puberty before, during and after cessation of GnRH agonist therapy. J Clin Endocrinol Metab. 2002;87(2):506–512. doi: 10.1210/jcem.87.2.8202. [DOI] [PubMed] [Google Scholar]
- 15.Arrigo T, De Luca F, Antoniazzi F, Galluzzi F, Segni M, Rosano M, Messina MF, Lombardo F. Reduction of baseline body mass index under gonadotropin-suppressive therapy in girls with idiopathic precocious puberty. Eur J Endocrinol. 2004;150:533–537. doi: 10.1530/eje.0.1500533. [DOI] [PubMed] [Google Scholar]
- 16.Poomthavorn P, Suphasit R, Mahachoklertwattana P. Adult height, body mass index and time of menarche of girls with idiopathic central precocious puberty after gonadotropin-releasing hormone analogue treatment. Gynecol Endocrinol. 2011;27:524–528. doi: 10.3109/09513590.2010.507289. [DOI] [PubMed] [Google Scholar]
- 17.Wolters B, Lass N, Reinehr T. Treatment with gonadotropin-releasing hormone analogues: different impact on body weight in normal-weight and overweight children. Horm Res Paediatr. 2012;78(5–6):304–311. doi: 10.1159/000346145. [DOI] [PubMed] [Google Scholar]
- 18.Lee SJ, Yang EM, Seo JY, Jong KC. Effects of gonadotropin-releasing hormone agonist therapy on body mass index and height in girls with central precocious puberty. Chonnam Med J. 2012;48:27–31. doi: 10.4068/cmj.2012.48.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Colmenares A, Gunczler P, Lanes R. Higher prevalence of obesity and overweight without an adverse metabolic profile in girls with central precocious puberty compared to girls with early puberty, regardless of GnRH analogue treatment. Int J Pediatr Endocrinol. 2014;2014:5. doi: 10.1186/1687-9856-2014-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Alessandri SB, Pereira Fde A, Villela RA, Antonini SR, Elias PC, Martinelli CE, Jr, Castro MD, Moreira AC, Paula FJ. Bone mineral density and body composition in girls with idiopathic central precocious puberty before and after treatment with a gonadotropin-releasing hormone agonist. Clinics (Sao Paulo) 2012;67(6):591–596. doi: 10.6061/clinics/2012(06)08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Magiakou MA, Manousaki D, Papadaki M, Hadjidakis D, Levidou G, Vakaki M, Papaefstathiou A, Lalioti N, Kanaka-Gantenbein C, Piaditis G, Chrousos G, Dacou-Voutetakis C. The efficacy and safety of gonadotropin-releasing hormone analog treatment in childhood and adolescence: a single center, long-term follow-up study. J Clin Endocrinol Metab. 2010;95(1):109–117. doi: 10.1210/jc.2009-0793. [DOI] [PubMed] [Google Scholar]
- 22.Marshall WA, Tanner JM. Variations in the pattern of pubertal changes in girls. Arch Dis Child. 1969;44:291–303. doi: 10.1136/adc.44.235.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Greulich WW. Radiographic atlas of skeletal development of the hand and wrist, Pyle SI. 2. Palo Alto: Standford University Press; 1959. [Google Scholar]
- 24.Cole TJ, Bellizi MC, Fiegal KM, Dietz WH. Establishing a standard definition for child overweight and obesity worldwide: international survey. Brit Med J. 2000;320:1240–1243. doi: 10.1136/bmj.320.7244.1240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Carel J-C, Eugster E, Mogol A, Ghizzoni L, Palmert MR, on behalf of the members of the ESPE-LWPES GnRH Analogs Consensus Conference Group. on behalf of the members of the ESPE-LWPES GnRH Analogs Consensus Conference Group Consensus statement on the use of gonadotropin- releasing hormone analogs in children. Pediatrics. 2009;123:752–762. doi: 10.1542/peds.2008-1783. [DOI] [PubMed] [Google Scholar]
- 26.Consenso sobre factores de riesgo de enfermedad cardiovascular en Pediatría Obesidad. Subcomisión de Epidemiología y Comité de Nutrición. Coordinadores: Durán P, Piazza N y Trifone L. Arch Argent Pediatr. 2005;103(3):262–281. [Google Scholar]
- 27.Paterson WF, McNeill E, Young D, Donaldson MDC. Auxological outcome and time to menarche following long-acting goserelin therapy in girls with central precocious or early puberty. Clin Endocrinol. 2004;61:626–634. doi: 10.1111/j.1365-2265.2004.02146.x. [DOI] [PubMed] [Google Scholar]
- 28.Pasquino AM, Pucarelli I, Accardo F, Demiraj V, Segni M, Di Nardo R. Long-term observation of 87 girls with idiopathic central precocious puberty treated with gonadotropin-releasing hormone analogs: impact on adult height, body mass index, bone mineral content, and reproductive function. J Clin Endocrinol Metab. 2008;93(1):190–195. doi: 10.1210/jc.2007-1216. [DOI] [PubMed] [Google Scholar]
- 29.Cassio A, Bal MO, Orsini LF, Balsamo A, Sansavini S, Gennari M, De Cristofaro E, Cicognani A. Reproductive outcome in patients treated and not treated for idiopathic early puberty: long-term results of a randomized trial in adults. J Pediatr. 2006;149:532–536. doi: 10.1016/j.jpeds.2006.05.026. [DOI] [PubMed] [Google Scholar]
- 30.Lazar L, Lebenthal Y, Yackobovitch-Gavan M, Shalitin S, de Vries L, Phillip M, Meyerovitch J. Treated and untreated women with idiopathic precocious puberty: BMI evolution, metabolic outcome, and general health between third and fifth decades. J Clin Endocrinol Metab. 2015;100(4):1445–1451. doi: 10.1210/jc.2014-3748. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will not be shared unless Editor-in-Chief or reviewers require it.


