Abstract
Background and objectives
Idiopathic hypercalciuria is a frequent defect in calcium kidney stone formers that is associated with high intestinal calcium absorption and osteopenia. Characteristics distinguishing hypercalciuric stone formers from hypercalciuric patients without kidney stone history (HNSFs) are unknown and were explored in our study.
Design, setting, participants, & measurements
We compared 172 hypercalciuric stone formers with 36 HNSFs retrospectively selected from patients referred to outpatient clinics of the San Raffaele Hospital in Milan from 1998 to 2003. Calcium metabolism and lumbar bone mineral density were analyzed in these patients. A strontium oral load test was performed: strontium was measured in 240-minute urine and serum 30, 60, and 240 minutes after strontium ingestion; serum strontium concentration-time curve and renal strontium clearance were evaluated to estimate absorption and excretion of divalent cations.
Results
Serum strontium concentration-time curve (P<0.001) and strontium clearance (4.9±1.3 versus 3.5±2.7 ml/min; P<0.001) were higher in hypercalciuric stone formers than HNSFs, respectively. The serum strontium-time curve was also higher in hypercalciuric stone formers with low bone mineral density (n=42) than in hypercalciuric stone formers with normal bone mineral density (n=130; P=0.03) and HNSFs with low (n=22; P=0.01) or normal bone mineral density (n=14; P=0.02). Strontium clearance was greater in hypercalciuric stone formers with normal bone mineral density (5.3±3.4 ml/min) than in hypercalciuric stone formers and HNSFs with low bone mineral density (3.6±2.5 and 3.1±2.5 ml/min, respectively; P=0.03). Multivariate regression analyses displayed that strontium absorption at 30 minutes was positively associated calcium excretion (P=0.03) and negatively associated with lumbar bone mineral density z score (P=0.001) in hypercalciuric stone formers; furthermore, hypercalciuric patients in the highest quartile of strontium absorption had increased stone production risk (odds ratio, 5.06; 95% confidence interval, 1.2 to 20.9; P=0.03).
Conclusions
High calcium absorption in duodenum and jejunum may expose hypercalciuric patients to the risk of stones because of increased postprandial calcium concentrations in urine and tubular fluid. High calcium absorption may identify patients at risk of bone loss among stone formers.
Keywords: hypercalciuria; calcium metabolism disorders; kidney stones; Bone Density; Bone Diseases, Metabolic; Calcium, Dietary; Humans; Intestinal Absorption; Regression Analysis; Strontium
Introduction
Idiopathic hypercalciuria is a defect detectable in 40% of calcium kidney stone formers and acknowledged as a risk factor for kidney stones (1,2). It has been mainly studied in patients producing kidney stones but was also found in 20% of osteoporotic women and 10% of the general population (2–4). Patients with idiopathic hypercalciuria are characterized by high intestinal absorption and low tubular reabsorption of calcium and may have decreased bone mass (3–5). A primary increase of enteral calcium absorption was hypothesized to cause hypercalciuria in a large portion of patients (6). It may be sustained by an increase of calcium carrier activity, vitamin D receptor sensitivity, or 25-hydroxyvitamin D [25(OH)D] hydroxylation activity (7,8). Defects of calcium reabsorption in both proximal and distal tubules were observed in hypercalciuric patients with or without stones (9,10). Decreased bone mineral density (BMD) may result from the negative balance between urine loss and enteral absorption of calcium (3,4). However, low BMD was associated with high calcium absorption and could be supported by a primary defect of calcium handling in intestinal, bony, and kidney tubular cells (7,11). Epidemiologic surveys also suggested that high serum levels of 25(OH)D and sodium intake may predispose to hypercalciuria (3,12), whereas serum levels of 1,25-dihydroxyvitamin D [1,25(OH)2D] were not found to be increased in hypercalciuric stone formers (HSFs) (13).
Despite the large amount of data, it is unknown if a qualitative or quantitative defect of calcium absorption and tubular reabsorption may distinguish HSFs and hypercalciuric patients without kidney stone history (HNSFs). Therefore, this work retrospectively compares HSFs and HNSFs who were studied with a protocol including an oral strontium load test. This test used stable strontium as a surrogate marker to estimates intestinal calcium absorption and excretion during a 4-hour time period after an oral load (14). This test is practical, feasible, and because of its duration, estimates calcium absorption in duodenum and jejunum (14). Previous findings showed higher strontium excretion and absorption in HSFs than in normocalciuric stone formers (14,15), consistent with the known alteration of calcium metabolism in these patients (5,10). In addition, strontium absorption was negatively correlated with lumbar BMD values in hypercalciuric stone–forming women (11). In this study, strontium absorption and excretion were compared in HSFs and HNSFs to find differences potentially explaining stone formation. We also considered BMD values in this analysis because of the previously observed association of strontium absorption with BMD.
Materials and Methods
Patients
One hundred seventy-two HSFs were retrospectively selected from patients referring to the kidney stones outpatient clinic of the San Raffaele Hospital in Milan from 1998 to 2003 for their history of urinary emission or surgical removal of calcium oxalate or phosphate stones or a radiology examination positive for radio-opaque stones. In the same period, 36 HNSFs and 40 normocalciuric controls (NCs) were selected from patients referring to the nephrology, hypertension, and osteoporosis outpatient clinics of the San Raffaele Hospital because of their negative history of stones, neoplasia, diabetes, bowel diseases, endocrine disorders, and inflammatory disorders; a renal echography excluded kidney stones in each of them. All participants were selected on their first access to the mentioned outpatient clinics and studied during a free diet. They could suffer from osteoporosis/osteopenia or hypertension. They had to have an age between 17 and 67 years old and normal serum values of creatinine, potassium, and calcium. They could not be taking thiazide, vitamin D or calcium supplements, or other drugs for osteoporosis or affecting mineral and electrolyte metabolism. Stone formers were studied at least 3 months after urologic treatments for stones.
In all participants, we measured serum concentrations of calcium (total and ionized), creatinine, 25(OH)D, and intact parathyroid hormone as well as 24-hour urine excretion of calcium and sodium. eGFR was estimated with the Chronic Kidney Disease Epidemiology Collaboration equation (16). Intact parathyroid hormone plasma levels were determined by immunoradiometric assay (Nichols Institute, San Juan Capistrano, CA). Plasma 25(OH)D was measured by RIA method (Diasorin, Stillwater, MN). Idiopathic hypercalciuria was diagnosed as 24-hour calcium excretion >300 mg in men or 250 mg in women or 4 mg/kg body wt in both sexes.
All participants underwent a strontium absorption test (14). An oral load of strontium (2.65 mg/kg body wt) was administered as a chloride salt in water solution (1 g/L) after an overnight fast (17,18). Blood samples were drawn before and 30, 60, and 240 minutes after strontium ingestion. Urine was also collected during the test (240 minutes) (15). Strontium concentration was measured in plasma and urine with an atomic absorption spectrophotometer (PerkinElmer 4000; PerkinElmer, Waltham, MA). Strontium absorption was calculated as the incremental area under the plasma strontium concentration-time curve (AUCSr) at 30, 60, and 240 minutes after strontium ingestion determined by the trapezoid method (grams per deciliter per minute) (13,15). Renal strontium clearance (milliliters per minute) was calculated as the ratio of the strontium excreted in the 240-minute urine divided by AUCSr at 240 minutes (13,15). The variation coefficient of the method, calculated by repeated measurements of strontium concentration in a sera pool, was 6.8% (18). BMD was measured with a dual–energy x-ray absorptiometry (QDR4500W or 1000; Hologic, Bedford, MA) at the lumbar spine and expressed as z score, which is the number of SDs from the mean in a control population with the same sex and age. The use of z score to express BMD enabled us to compare patients having z score ≤−2 or >−2, keeping patients of different sex and age together. The variation coefficient of the instruments, calculated daily by quality control scans of the spine phantom, was <0.5%.
The study was part of a research project approved by the San Raffaele Hospital Ethical Committee. All participants gave informed consent to participate in the study.
Statistical Analyses
Quantitative variables were reported in the text as means±SDs and compared between groups by the Kruskal–Wallis test. Serum strontium concentration-time curve was analyzed by ANOVA for repeated measures and compared in subject groups with Tukey post hoc tests. Multivariable linear stepwise regression was used to evaluate variables associated with calcium excretion and lumbar spine BMD z score; the analysis included age; strontium clearance; body weight; calcium excretion (if lumbar z score is the dependent variable); AUCSr at 30, 60, and 240 minutes; serum concentrations of parathyroid hormone; 25(OH)D; and excretion of calcium and sodium as independent variables. Stone risk sustained by strontium absorption was expressed as odds ratio with 95% confidence interval calculated by multinomial logistic regression, including stones as the dependent variable and age (>40 or <40 years old), quartiles of calcium excretion, sodium excretion, and AUCSr at 30 or 60 minutes as confounding factors. Statistical analysis was two tailed and conducted at the α=0.05 level. It was performed using the SPSS statistical package (IBM SPSS, Chicago, IL).
Results
Table 1 displays characteristics of HSFs, HNSFs, and NCs participating in the study. Sodium excretion was higher in both hypercalciuric groups compared with n NCs. Lumbar spine BMD z score was lower in HNSFs than in HSFs. AUCSr values were higher in HSFs than in NCs at each time point. Strontium clearance was higher in HSFs than in NCs and HNSFs.
Table 1.
Variables in hypercalciuric stone formers, hypercalciuric patients, and normocalciuric participants without stone history
| Characteristics | Hypercalciuric Stone Formers | Hypercalciuric Patients without Stones | Normocalciuric Controls |
|---|---|---|---|
| N (men/women) | 172 (105/67) | 36 (20/16) | 40 (24/16) |
| Age, yr | 44±12 | 47±10 | 41±15 |
| Body weight, kg | 68±14 | 66±14 | 69±17 |
| Serum calcium, mg/dl | 9.5±0.4 | 9.6±0.4 | 9.5±0.5 |
| Serum ionized calcium, mg/dl | 4.9±0.2 | 4.9±0.2 | 4.9±0.2 |
| eGFR, ml/min per 1.73m2 | 93.6±15.2 | 98.4±12.3 | 92.1±16.3 |
| Serum PTH, pg/ml | 36±18 | 40±18 | 43±20 |
| Serum 25(OH)D, ng/ml | 21±16 | 18±9 | 20±6 |
| Urine calcium, mg/d | 365±105 | 378±102 | 166±54 |
| Urine sodium, mEq/d | 180±70 | 206±69 | 139±64a |
| AUCSr at 30 min, g⋅dl−1⋅min | 2.96±1.66b | 2.24±1.40 | 2.02±0.88 |
| AUCSr at 60 min, g⋅dl−1⋅min | 10.56±4.76c | 8.59±3.94 | 7.59±2.51 |
| AUCSr at 240 min, g⋅dl−1⋅min | 76.10±25.99d | 65.81±21.3 | 58.15±16.64 |
| Strontium clearance, ml/min | 4.9±3.2e | 3.5±2.7 | 2.3±1.9 |
| Lumbar spine BMD z score | −1.33±1.27f | −2.11±0.82 | −1.67±0.92 |
Variables are reported as mean±SD. Kruskal–Wallis test was used for multiple comparisons between groups, except for sex distribution, which was compared with the chi-squared test. PTH, parathyroid hormone; 25(OH)D, 25-hydroxyvitamin D; AUCSr, area under the plasma strontium concentration-time curve; BMD, bone mineral density.
P<0.01 versus hypercalciuric stone formers and P<0.001 versus hypercalciuric patients without stones.
P=0.003 versus normocalciuric controls and P=0.03 versus hypercalciuric patients without stones.
P<0.001 versus normocalciuric controls and P=0.05 versus hypercalciuric patients without stones.
P<0.001 versus normocalciuric controls.
P<0.001 versus normocalciuric controls and P=0.04 versus hypercalciuric patients without stones.
P=0.001 versus hypercalciuric patients without stones.
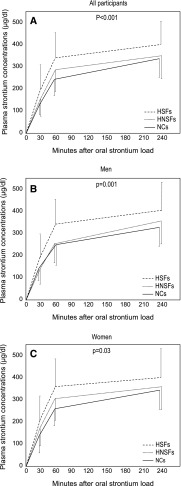
The plasma strontium concentration-time curve during the test was significantly higher in HSFs than in HNSFs and NCs both when using crude data and when using adjusted data for age, sex, and body weight (Figure 1A). Considering men or women separately, the curve was higher in HSFs than in NCs in both sexes and significantly higher in HSFs than in HNSFs in men (Figure 1, B and C).
Figure 1.
Serum strontium concentration-time curve in hypercalciuric patients and controls. Data were compared with ANOVA for repeated measures and Tukey post hoc test for multiple comparisons. (A) The curve was significantly faster in hypercalciuric stone formers (HSFs; n=172; men/women: 105/67) than hypercalciuric patients without kidney stone history (HNSFs; n=36; men/women: 20/16; P=0.02) and normocalciuric controls (NCs; n=40; men/women: 24/16; P<0.001) using crude data (F=10.1; P<0.001) and after adjustment for age, sex, and body weight (F=9.9; P<0.001). (B) Considering only men, the curve was higher in HSFs (n=105) than in NCs (n=24; P=0.03) and HNSFs (n=20; P<0.01) with analysis of crude data (F=6.8; P=0.001) or adjusted data for weight and age (F=6.3; P=0.003). (C) In women, the curve was higher in HSFs (n=67) than in NCs (n=16; P=0.03) using crude data (F=3.6; P=0.03) or adjusted data (F=4; P=0.02).
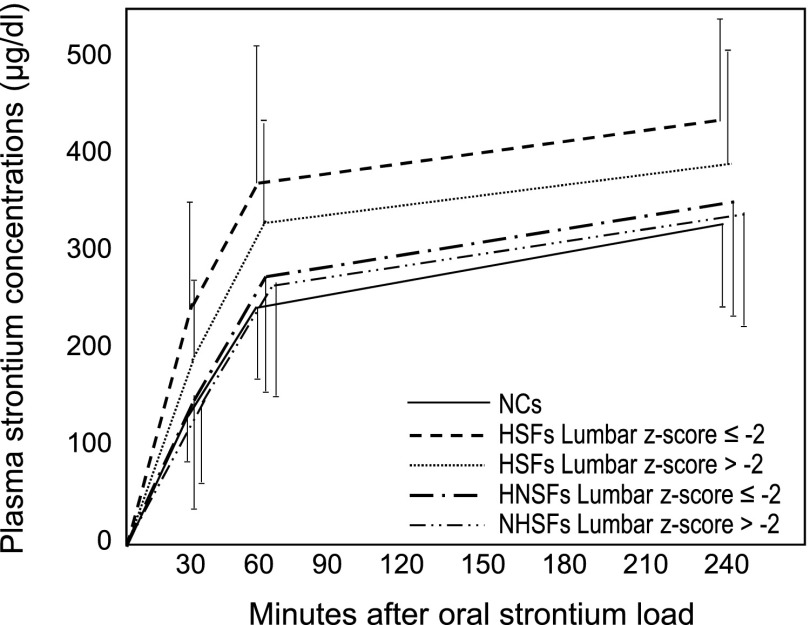
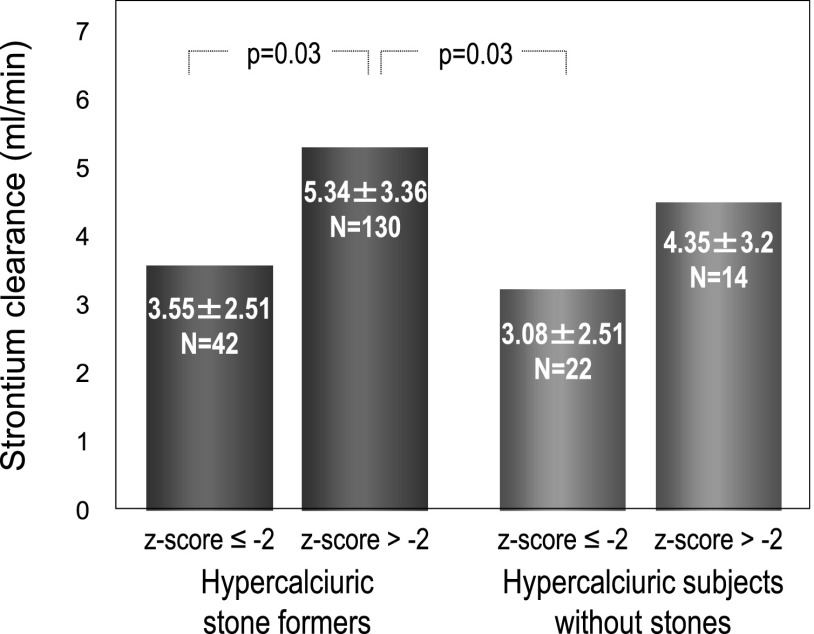
The association of strontium absorption with stones was compared in HSFs and HNSFs having lumbar BMD z score ≤−2 or >−2 (Figure 2). The plasma strontium concentration-time curve was significantly higher in HSFs with z score ≤−2 (n=42) than in HNSFs having z score either ≤−2 (n=22) or >−2 (n=14) and also, in HSFs with z score >−2 (n=130). These findings were observed with the crude data analysis and after their adjustment for age and body weight. Strontium clearance was higher in HSFs with lumbar BMD z score >−2 than in HSFs and HNSFs having z score ≤−2 (Figure 3).
Figure 2.
Serum strontium concentration-time curve in hypercalciuric patients as a function of lumbar bone mineral density. Data were compared with ANOVA for repeated measures and Tukey post hoc test for multiple comparisons. The curve was higher in HSFs with z scores ≤−2 (n=42) than in HNSFs with z scores ≤−2 (n=22; P=0.01) or >−2 (n=14; P=0.02) and HSFs with z score >−2 (n=130; P=0.03) when we considered crude data (F=3.6; P=0.02) or data adjusted for weight and age (F=3.8; P=0.01). NC, normocalciuric control.
Figure 3.
Strontium clearance in hypercalciuric patients as a function of lumbar bone mineral density. It was higher in hypercalciuric stone formers with lumbar bone mineral density z score >−2 (n=130) than in hypercalciuric stone formers and hypercalciuric patients without kidney stone history having lumbar bone mineral density z score ≤−2 (n=42 and n=22, respectively; Kruskal–Wallis test for multiple comparisons).
Table 2 shows findings of the regression analysis. In hypercalciuric patients, calcium excretion was associated with AUCSr at 30 minutes, sodium excretion, and body weight in HSFs. It was associated with sodium excretion in HNSFs. In HSFs, AUCSr at 30 minutes and strontium clearance were negatively and positively, respectively, associated with lumbar spine z score (Table 2).
Table 2.
Variables associated with calcium excretion and lumbar spine bone mineral density z score in hypercalciuric patients with or without stones
| Sample, Dependent Variable, and Independent Variables | r2 Change | Standardized Regression Coefficient | P Value |
|---|---|---|---|
| Hypercalciuric stone formers | |||
| Calcium excretion, mg/d | |||
| Sodium excretion, mEq/d | 0.32 | 0.44 | <0.001 |
| AUCSr at 30 min, g⋅dl−1⋅min | 0.04 | 0.23 | 0.03 |
| Body weight, kg | 0.04 | 0.25 | 0.02 |
| Model cumulative r2 | 0.40 | <0.001 | |
| Hypercalciuric patients without stones | |||
| Calcium excretion, mg/d | |||
| Sodium excretion, mEq/24 h | 0.56 | 0.75 | 0.001 |
| Model cumulative r2 | 0.56 | 0.001 | |
| Hypercalciuric stone formers | |||
| Lumbar spine BMD z score | |||
| AUCSr at 30 min, g⋅dl−1⋅min | 0.11 | −0.33 | 0.001 |
| Strontium clearance, ml/min | 0.06 | 0.25 | 0.01 |
| Model cumulative r2 | 0.17 | <0.001 |
Multivariable linear regression analysis was performed to find associations in hypercalciuric stone formers (n=172) and hypercalciuric patients without stones (n=36). Variables included in the model are reported in the text. AUCSr, area under the plasma strontium concentration-time curve; BMD, bone mineral density.
Stones risk was calculated with multinomial logistic regression in hypercalciuric patients and associated with the highest quartile of AUCSr at 30 minutes and age ≤40 years old (Table 3).
Table 3.
Factors associated with kidney stone risk in hypercalciuric patients
| Sample and Factor | n | Stone Formers, n (%) | OR | 95% CI | P Value |
|---|---|---|---|---|---|
| Hypercalciuric patients | |||||
| AUCSr at 30 min, g⋅dl−1⋅min | |||||
| Quartile 1 | 48 | 34 (70.8) | 1.00 | ||
| Quartile 2 | 49 | 41 (83.7) | 1.30 | 0.4 to 4.1 | 0.65 |
| Quartile 3 | 53 | 42 (79.2) | 1.28 | 0.4 to 3.8 | 0.66 |
| Quartile 4 | 58 | 55 (94.8) | 5.06 | 1.2 to 20.9 | 0.03 |
| Age, yr | |||||
| >40 | 136 | 107 (78.7) | 1.00 | ||
| ≤40 | 72 | 65 (90.3) | 3.28 | 1.1 to 9.5 | 0.03 |
Multinomial logistic regression was used to identify factors associated with kidney stone risk in hypercalciuric patients (n=208; n=172 stone formers; n=36 patients without stones). OR, odds ratio; 95% CI, 95% confidence interval; AUCSr, area under the plasma strontium concentration-time curve.
Discussion
This study compared calcium metabolism in HSFs and HNSFs by using strontium as a surrogate marker to estimate calcium absorption and excretion (13–15,17,18). As expected, strontium absorption and excretion were higher in HSFs than in NCs and HNSFs, whereas their values were not significantly increased in HNSFs. The higher strontium absorption was more marked in HSFs with low BMD than in HSFs with normal BMD, whereas no differences related to BMD were observed in HNSFs. Strontium clearance was higher in HSFs with normal BMD than in HSFs and HNSFs having low BMD.
Multivariate analyses displayed that strontium absorption was associated with calcium excretion and kidney stone production, thus suggesting that enteral absorption was more relevant than tubular reabsorption of calcium in determining hypercalciuria in our patients (5,10). Conversely, both defects might contribute to explain bone mass in HSFs (7,19). These characteristics of strontium handling suggest that HSFs could represent a specific group of hypercalciuric patients having a disorder of cellular calcium transport predisposing to hypercalciuria, stone production, and low bone mass. This defect might sustain a greater efficiency of intestinal calcium absorption (7,10) and also, explain the lower strontium clearance observed in patients with low BMD after an acute load.
The test used in this study estimates calcium absorption in duodenum and jejunum that could be crucial to the postprandial urine calcium peak (5,10) and the supersaturation of tubular fluid and urine with respect to calcium salts (9). Accordingly, the fact that AUCSr at 30 or 60 minutes was recognized as a correlate of calcium excretion and kidney stones confirms the relevance of early calcium absorption for stone risk. The lithogenic effect of calcium absorption in duodenum and jejunum may be additive to that of other nutrients, like sodium, which may stimulate calcium excretion (12,20). These observations lead to the conclusion that intestinal absorption may be a key factor in inducing stone risk, because it may determine tubular load of calcium and other lithogenic substances after a meal (21).
Strontium absorption was not related with serum 25(OH)D levels in HSFs. Also, serum levels of 1,25(OH)2D, measured by us in previous studies, were not correlated with strontium absorption and were not different in hypercalciuric and normocalciuric stone formers (13,15). Furthermore, calcium intake, which may influence calcium absorption in intestine, was not different in hypercalciuric and normocalciuric stone formers in two Italian studies (3,12). Considered together, findings of these studies suggest that a vitamin D–independent disorder of calcium transport in intestinal cells may sustain hypercalciuria in stone formers. However, calcium intake and serum levels of 1,25(OH)2D were not assessed in this study, and this limitation does not allow a definitive conclusion about the cause of calcium hyperabsorption in HSFs.
Mechanisms of hypercalciuria may develop with a different involvement of calcium absorption, tubular reabsorption, and bone metabolism in HNSFs related to HSFs. They did not have an increased absorption of calcium in duodenum and jejunum but could have an increased calcium absorption in ileum and colon that are not explored by the strontium oral load test (16). Therefore, the kinetics of enteral calcium absorption could be different in hypercalciuric patients with or without stones: HSFs may have an early high tubular calcium load after a meal mediated by the active duodenal absorption, whereas HNSFs may have a tubular load diluted in a longer time period that could explain their normal strontium excretion during the load test. According to this hypothesis, the defect of calcium transport explaining hypercalciuria could be different in the two populations of hypercalciuric patients.
To our knowledge, this study is the first one comparing HSFs and HNSFs. Participants without stones were selected from patients referring to the outpatient clinics of one hospital, including the osteoporosis clinic; this explains the low bone mass of nonstone formers but may be a source of bias. Therefore, we stratified participants according to their BMD and found that strontium absorption was not related to BMD in HNSFs, whereas their negative relation indicated that low calcium absorption did not cause bone mass loss in HSFs.
In conclusion, findings of our study suggest that high calcium absorption in duodenum and jejunum may expose hypercalciuric patients to the risk of stones because of an increased postprandial calcium load for tubular fluid. The increased calcium absorption may identify patients at risk of bone loss among stone formers.
Disclosures
None.
Acknowledgments
We thank Simone Pappacena (Vita Salute University, San Raffaele Scientific Institute) for his help in editing the manuscript.
The study was supported by grants from the Italian Ministry of University and Scientific Research and from the San Raffaele Scientific Institute.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.Hodgkinson A, Pyrah LN: The urinary excretion of calcium and inorganic phosphate in 344 patients with calcium stone of renal origin. Br J Surg 46: 10–18, 1958 [DOI] [PubMed] [Google Scholar]
- 2.Curhan GC, Willett WC, Speizer FE, Stampfer MJ: Twenty-four-hour urine chemistries and the risk of kidney stones among women and men. Kidney Int 59: 2290–2298, 2001 [DOI] [PubMed] [Google Scholar]
- 3.Vezzoli G, Soldati L, Arcidiacono T, Terranegra A, Biasion R, Russo CR, Lauretani F, Bandinelli S, Bartali B, Cherubini A, Cusi D, Ferrucci L: Urinary calcium is a determinant of bone mineral density in elderly men participating in the InCHIANTI study. Kidney Int 67: 2006–2014, 2005 [DOI] [PubMed] [Google Scholar]
- 4.Giannini S, Nobile M, Dalle Carbonare L, Lodetti MG, Sella S, Vittadello G, Minicuci N, Crepaldi G: Hypercalciuria is a common and important finding in postmenopausal women with osteoporosis. Eur J Endocrinol 149: 209–213, 2003 [DOI] [PubMed] [Google Scholar]
- 5.Pak CY, Oata M, Lawrence EC, Snyder W: The hypercalciurias. Causes, parathyroid functions, and diagnostic criteria. J Clin Invest 54: 387–400, 1974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Reed BY, Gitomer WL, Heller HJ, Hsu MC, Lemke M, Padalino P, Pak CY: Identification and characterization of a gene with base substitutions associated with the absorptive hypercalciuria phenotype and low spinal bone density. J Clin Endocrinol Metab 87: 1476–1485, 2002 [DOI] [PubMed] [Google Scholar]
- 7.Karnauskas AJ, van Leeuwen JPTM, van den Bemd GJCM, Kathpalia PP, DeLuca HF, Bushinsky DA, Favus MJ: Mechanism and function of high vitamin D receptor levels in genetic hypercalciuric stone-forming rats. J Bone Miner Res 20: 447–454, 2005 [DOI] [PubMed] [Google Scholar]
- 8.Hess B, Ackermann D, Essig M, Takkinen R, Jaeger P: Renal mass and serum calcitriol in male idiopathic calcium renal stone formers: Role of protein intake. J Clin Endocrinol Metab 80: 1916–1921, 1995 [DOI] [PubMed] [Google Scholar]
- 9.Worcester EM, Bergsland KJ, Gillen DL, Coe FL: Evidence for increased renal tubule and parathyroid gland sensitivity to serum calcium in human idiopathic hypercalciuria. Am J Physiol Renal Physiol 305: F853–F860, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pak CY, Britton F, Peterson R, Ward D, Northcutt C, Breslau NA, McGuire J, Sakhaee K, Bush S, Nicar M, Norman DA, Peters P: Ambulatory evaluation of nephrolithiasis. Classification, clinical presentation and diagnostic criteria. Am J Med 69: 19–30, 1980 [DOI] [PubMed] [Google Scholar]
- 11.Vezzoli G, Rubinacci A, Bianchin C, Arcidiacono T, Giambona S, Mignogna G, Fochesato E, Terranegra A, Cusi D, Soldati L: Intestinal calcium absorption is associated with bone mass in stone-forming women with idiopathic hypercalciuria. Am J Kidney Dis 42: 1177–1183, 2003 [DOI] [PubMed] [Google Scholar]
- 12.Vezzoli G, Dogliotti E, Terranegra A, Arcidiacono T, Macrina L, Tavecchia M, Pivari F, Mingione A, Brasacchio C, Nouvenne A, Meschi T, Cusi D, Spotti D, Montanari E, Soldati L: Dietary style and acid load in an Italian population of calcium kidney stone formers. Nutr Metab Cardiovasc Dis 25: 588–593, 2015 [DOI] [PubMed] [Google Scholar]
- 13.Vezzoli G, Caumo A, Baragetti I, Zerbi S, Bellinzoni P, Centemero A, Rubinacci A, Moro G, Adamo D, Bianchi G, Soldati L: Study of calcium metabolism in idiopathic hypercalciuria by strontium oral load test. Clin Chem 45: 257–261, 1999 [PubMed] [Google Scholar]
- 14.Milsom S, Ibbertson K, Hannan S, Shaw D, Pybus J: Simple test of intestinal calcium absorption measured by stable strontium. Br Med J (Clin Res Ed) 295: 231–234, 1987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vezzoli G, Baragetti I, Zerbi S, Caumo A, Soldati L, Bellinzoni P, Centemero A, Rubinacci A, Moro G, Bianchi G: Strontium absorption and excretion in normocalciuric subjects: Relation to calcium metabolism. Clin Chem 44: 586–590, 1998 [PubMed] [Google Scholar]
- 16.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J; CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) : A new equation to estimate glomerular filtration rate. Ann Intern Med 150: 604–612, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sips AJ, van der Vijgh WJ, Barto R, Netelenbos JC: Intestinal strontium absorption: From bioavailability to validation of a simple test representative for intestinal calcium absorption. Clin Chem 41: 1446–1450, 1995 [PubMed] [Google Scholar]
- 18.Reid IR, Pybus J, Lim TMT, Hannon S, Ibbertson HK: The assessment of intestinal calcium absorption using stable strontium. Calcif Tissue Int 38: 303–305, 1986 [DOI] [PubMed] [Google Scholar]
- 19.Liberman UA, Sperling O, Atsmon A, Frank M, Modan M, Vries AD: Metabolic and calcium kinetic studies in idiopathic hypercalciuria. J Clin Invest 47: 2580–2590, 1968 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sakhaee K, Harvey JA, Padalino PK, Whitson P, Pak CY: The potential role of salt abuse on the risk for kidney stone formation. J Urol 150: 310–312, 1993 [DOI] [PubMed] [Google Scholar]
- 21.Bushinsky DA: Nephrolithiasis: Site of the initial solid phase. J Clin Invest 111: 602–605, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]