Abstract
Introduction. Luteolin, a falconoid compound in many Chinese herbs and formula, plays important roles in cardiovascular diseases. The underlying mechanism of luteolin remains to be further elaborated. Methods. A model of hydrogen peroxide- (H2O2-) induced H9C2 cells apoptosis was established. Cell viabilities were examined with an MTT assay. 2′,7′-Dichlorofluorescin diacetate (DCFH-DA) and flow cytometry were used to detect ROS level and apoptosis rate, respectively. The expressions of signaling proteins related to apoptosis were analyzed by western blot and mRNA levels were detected by real-time polymerase chain reaction (PCR). Quercetin was applied as positive drug. Results. Incubation with various concentrations of H2O2 (0, 50, 100, and 200 μM) for 1 h caused dose-dependent loss of cell viability and 100 μM H2O2 reduced the cell viability to approximately 50%. Treatments with luteolin and quercetin protected cells from H2O2-induced cytotoxicity and reduced cellular ROS level and apoptosis rate. Moreover, luteolin could downregulate the expressions of Bax, caspase-8, cleaved-caspase-3, and p53 in apoptotic signaling pathway. Further study showed that the expressions of Akt, Bcl-2, and Mdm2 were upregulated by luteolin. Conclusion. Luteolin protects H9C2 cells from H2O2-induced apoptosis. The protective and antiapoptotic effects of luteolin could be mediated by regulating the Akt-P53/Mdm2 apoptotic pathway.
1. Introduction
Heart failure (HF) is the common condition resulting from a variety of cardiovascular diseases. The morbidity and mortality of HF remain high even though great efforts have been made to manage it [1]. The five-year survival rate of HF is only 35% [2]. One of the molecular mechanisms of HF is associated with apoptosis which occurs in various cardiovascular diseases, including myocardial infarction (MI), atherosclerosis (AS) and ischemia/reperfusion (IR) [3, 4]. Cardiomyocytes apoptosis is considered to be a key factor in the development of heart failure [5].
P53, known as the tumor suppressor gene, could induce apoptosis by blocking cellular DNA damage repair. Accumulating evidence shows that p53 plays a pivotal role in the progression of AS, MI, and IR [6, 7]. Expression of p53 is higher in the failing myocardium than normal cardiomyocytes [8]. By activating proapoptotic proteins, such as Bax and Bak, p53 induces apoptosis and accelerates progression of HF.
Murine double minute 2 (Mdm2), which is induced by p53, interacts with p53 and inhibits the apoptotic effect of p53. P53 level is regulated by the feedback between Mdm2 and p53 [9, 10]. A model of MI in mice [7] showed that a synthetic inhibitor targeting p53 could reduce cardiomyocytes apoptosis. Meanwhile, the area of MI was enlarged, indicating that the side effects of monotarget therapy should be taken seriously. Developing apoptotic inhibitors with minor side effects is an important potential strategy in the management of HF.
Traditional medicine has been making great contributions in treating varieties of human diseases including heart failure [11]. Luteolin, one of the flavonoids abundant in many vegetables and fruits [12], has been reported to provide protective effects against oxidative stress, inflammation, and cancer [13–15]. Plenty of evidence indicate that luteolin exerts multiple effects in cardiovascular diseases [16, 17]. In particular, luteolin has been shown to have antiapoptotic effects in cardiomyocytes [18, 19]. However, the antiapoptotic mechanism of luteolin is unclear and the effects of luteolin on Mdm2 and p53 remain to be investigated.
In this study, we explored the antiapoptotic effects of luteolin in H9C2 cell lines. Cellular apoptosis was induced by H2O2 culture and the effect of luteolin on p53/Mdm2 apoptotic signaling pathway was investigated.
2. Material and Methods
2.1. Material and Chemicals
MTT, DCFH-DA, and H2O2 (3 wt.%) were purchased from Sigma (USA). The Annexin V-PI kit was obtained from BD (USA). cDNA Synthesis kit, Green PCR Master Mix, penicillin, streptomycin, and 0.05% trypsin were purchased from Invitrogen (USA). Dulbecco's modified Eagle's medium (DMEM) was purchased from HyClone (USA). Fetal bovine serum (FBS) was purchased from Corning (USA). Luteolin and quercetin were purchased from National Institutes for Food and Drug Control (China). Luteolin and quercetin were dissolved in DMSO for in vitro assay as storage concentration (100 mM).
2.2. Cell Culture and Grouping
H9C2 cardiomyocyte cell line was obtained from China Infrastructure of Cell Line Resources (Beijing, China). The H9C2 cells were cultured in high glucose DMEM supplemented with 10% FBS and a mixture of penicillin (100 U/mL) and streptomycin (100 μg/mL) in a humidified incubator with 5% CO2 at 37°C. Cells were nearly 80–90% confluent and treated with 5, 10, and 20 μM luteolin and 10 μM quercetin for 6 h. Then cells were divided into several groups: normal control group, H2O2-induced model group, luteolin-treated group, and quercetin-treated positive control group. Cells in model group were cultured in DMEM for 6 h, followed by treatment with H2O2 for 1 h. Cells in luteolin and quercetin groups were pretreated with luteolin and quercetin for 6 h before exposed to H2O2.
2.3. Establishment of Apoptosis Model Induced by H2O2
H9C2 cells were seeded in 96-well plates at a density of 104 cells per well. Following overnight adherence, cells were incubated with H2O2 (0, 50 μM, 100 μM, and 200 μM) in DMEM at 37°C for 1 h. Cells viabilities were determined by MTT assay. Cells were treated with MTT solution dissolved in DMEM (0.5 mg/mL) for 4 h. The supernatants were removed, followed by the addition of 150 μL DMSO to each well to dissolve the precipitate. The absorbance of each well was then measured with a microplate reader (Thermo, USA) at a wavelength of 492 nm.
2.4. Cell Viability Analysis
H9C2 cells were seeded as described above. Before being treated with H2O2 for 1 h, cells were incubated with luteolin (5 μM, 10 μM, and 20 μM) and quercetin (10 μM) in DMEM supplemented at 37°C for 6 h. After treatment, MTT assay was taken to measure cell viability.
2.5. Cell Morphological Analysis
H9C2 cells were seeded at a density of 105 cells/mL in 6-well plates. When the confluence reached 70–80%, cells were pretreated with/without luteolin (10 μM) and quercetin (10 μM) for 6 h and then exposed to H2O2 (100 μM) for another 1 h. After incubation, cell morphology was photographed under an inversion microscope (OLYMPUS, Japan).
2.6. Analysis of Reactive Oxygen Species (ROS)
The cellular production of ROS was measured by detecting the fluorescent intensity of DCFH-DA, an oxidant-sensitive probe. After incubations at different conditions, H9C2 cells were washed three times with PBS, followed by incubation with DCFH-DA (10 μM) at 37°C for half an hour. Fluorescent intensity was examined under a fluorescence microscope (OLYMPUS, Japan) with excitation at 485 nm and emission at 525 nm. Image J was used to analyze the data.
2.7. H9C2 Cell Apoptosis Assay
The Annexin V-PI method was used to detect the apoptosis rate of H9C2 cells by flow cytometry. Cells were harvested with 0.05% trypsin, washed three times with cold PBS (4°C), and collected by centrifugation at 1000 rpm for 5 minutes. After that cells were resuspended in 200 μL binding buffer and incubated with Annexin V (10 μg/mL) and PI (10 μg/mL) in the dark for 15 minutes at room temperature. Then cells were detected with a flow cytometer (BD, USA).
2.8. Quantitative Real-Time PCR Analysis
The total mRNA of cells was extracted using TRIzol (Thermo, USA) and cDNA was synthesized from 2 μg of total RNA using an RT kit (Thermo, USA) following the manufacturer's instructions. The mRNA quantity of Mdm2 gene was assessed by quantitative real-time PCR (ABI, USA). The primers used for amplification are as follows: β-actin F, CCCATCTATGAGGGTTACGC; β-actin R, TTTAATGTCACGCACGATTTC; Mdm2 F, TTGATGATGGCGTAAGTGA; Mdm2 R, AGGCTGTAATCTTCTGAGTC.
2.9. Western Blotting Analysis
The collected cells were prepared with RIPA buffer (PPLYGEN, China) and proteins were extracted according to the manufacture's instruction. Protein contents were measured with BCA Bradford protein assay (PPLYGEN, China). After addition of loading buffer, cell lysates were separated by 10% SDS-PAGE and transferred to NC membranes (Millipore, Germany). After being blocked with 5% nonfat dry milk for 2 h, the membrane was incubated with different primary antibodies (Abcam, USA) overnight at 4°C and washed with TBST three times. The membrane was then incubated with HRP-conjugated secondary antibodies (ZSGB-BIO, China) for 1 h at room temperature. After being washed with TBST for three times, the proteins were detected with an enhanced chemiluminescence agent (GE, USA) and quantified by densitometry using an image analyzer (Bio-Rad, USA). β-actin (ZSGB-BIO, China) served as an internal control.
The antibodies included rabbit monoclonal antibodies against cleaved-caspase-3 (1 : 1000), Bcl-2 (1 : 1000), Bax (1 : 1000), Akt (1 : 1000), rabbit multiclonal antibodies against caspase-8 (1 : 1000), mouse monoclonal antibody against p53 (1 : 1000), and secondary antibodies (goat anti-rabbit, 1 : 5000; goat anti-mouse, 1 : 5000).
2.10. Statistical Analyses
Data were expressed as the mean ± SD. Analysis was undertaken by one-way ANOVA. Differences between groups were considered as statistically significant when P < 0.05.
3. Result
3.1. Working Concentration of H2O2
H9C2 cell lines were incubated with different concentrations of H2O2 for 1 h. Cell viability was measured by MTT assay. The result showed that H2O2 induced dose-dependent decrease of cell viability (Figure 1). When cells were incubated with 100 μM and 200 μM H2O2, cell viabilities were reduced significantly compared with control group (P < 0.01). The cellular survival rate in 100 μM H2O2-treated group was 52.48 ± 0.0416% and these cells could be applied as apoptotic model to assess effects of luteolin. Therefore, 100 μM H2O2 was applied to cells to induce apoptosis in the subsequent experiments.
Figure 1.
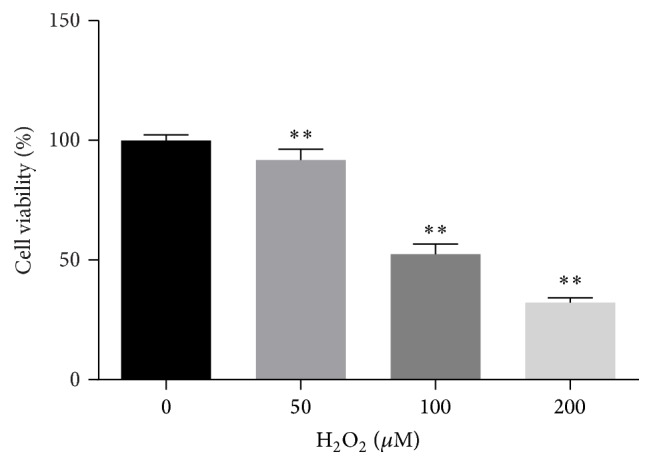
Cell viability in cells treated with different concentrations of H2O2 for 1 h. MTT assay was taken to measure cell viability. Data were expressed as mean ± SD. ∗∗ P < 0.01 versus control.
3.2. Effects of Luteolin/Quercetin on Cell Survival Rate and H2O2-Induced Cytotoxicity
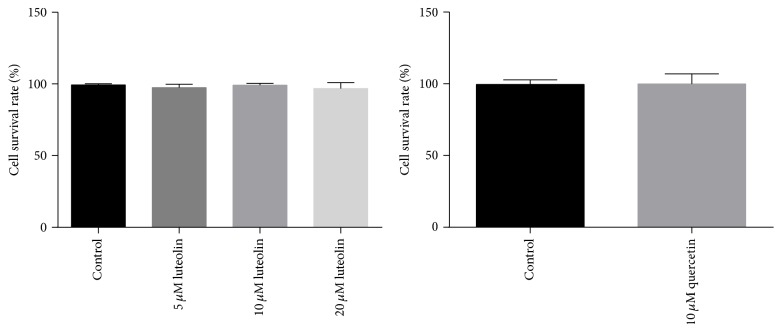
Cell viabilities were assessed by MTT assay. As shown in Figure 2, cell survival rates in luteolin (5, 10, and 20 μM) and quercetin (10 μM) groups were similar to that in control group, indicating that quercetin and luteolin have no cytotoxicity on H9C2 cells. The effects of luteolin/quercetin on H2O2-induced cytotoxicity were also detected. As shown in Figure 3, luteolin (5 μM, 10 μM, and 20 μM)/quercetin (10 μM) pretreatment provided significant protective effects on H2O2-mediated cytotoxicity in a dose-dependent manner (## P < 0.01). Cells treated with 10 μM luteolin had significant higher viability than those treated with 5 μM luteolin and cell viabilities were similar between cells treated with 10 μM and 20 μM luteolin. Therefore, 10 μM luteolin was applied to treat cells to assess its effects in the subsequent experiments.
Figure 2.
5, 10, and 20 μM luteolin and 10 μM quercetin had no effect on cell survival rate.
Figure 3.
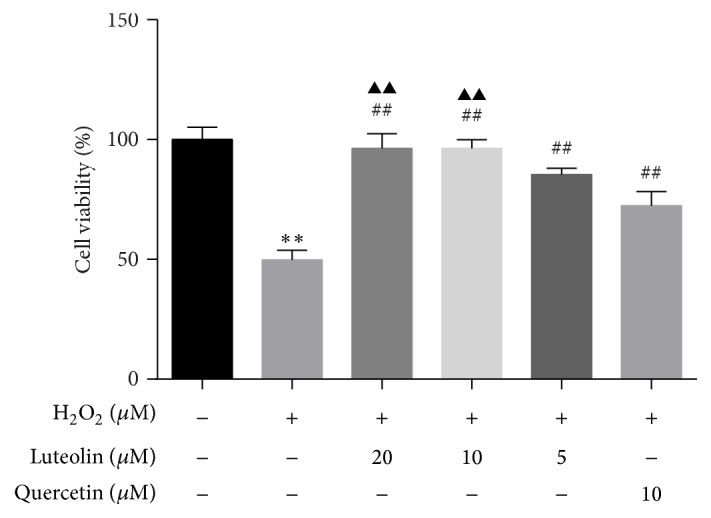
Luteolin rescued H2O2-induced loss of cell viability. H9C2 cells were preincubated with different concentrations of luteolin (5 μM, 10 μM, and 20 μM)/quercetin (10 μM) for 6 h and then treated with 100 μM H2O2 for 1 h. Cell viability was detected with MTT assay. Data were expressed as mean ± SD. ∗∗ P < 0.01 versus control; ## P < 0.01 versus model; ▲▲ P < 0.01 versus 5 μM luteolin.
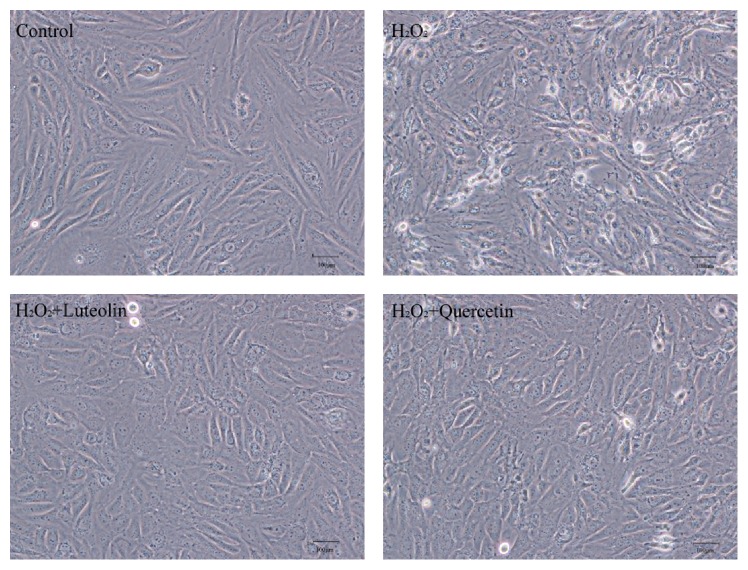
3.3. Effects of Luteolin on H2O2-Induced Morphologic Changes in H9C2 Cells
To further evaluate the protective effect of luteolin against H2O2-induced cellular injury, cell morphology was examined under an inversion microscope. Normal cells in control group showed long fusiform shapes with clear structure (Figure 4). Treating cells with H2O2 (100 μM) for 1 h resulted in cell shrinkage, alterations of cell shape, and wider intercellular gap. Pretreatment with luteolin and quercetin protected cells from the morphological changes induced by H2O2. These results demonstrated that luteolin pretreatment had significant protective effects against H2O2-induced cytotoxicity in H9C2 cells.
Figure 4.
Morphologic changes of H9C2 cells induced by H2O2 with/without luteolin. H9C2 cells were preincubated with/without luteolin (10 μM)/quercetin (10 μM) for 6 h and then treated with 100 μM H2O2 for 1 h. Cell morphology was examined under an inversion microscope.
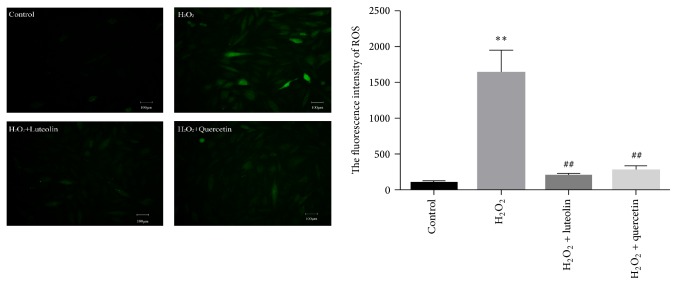
3.4. Luteolin Attenuated Intracellular ROS Generation
ROS generated by H2O2 could promote cellular damage. To determine whether pretreatment with luteolin could alleviate H2O2-induced early-stage oxidative stress, the treated cells were detected with DCFH-DA assay. Fluorescent intensity was examined under a fluorescence microscope. As shown in Figure 5, intracellular ROS significantly increased in H2O2-treated H9C2 cells compared with that in the control group. After pretreatment with luteolin (10 μM)/quercetin (10 μM), intracellular ROS was significantly reduced compared with that in H2O2-treated cells.
Figure 5.
Intracellular ROS of H9C2 cells induced by H2O2 with/without luteolin. H9C2 cells were preincubated with/without luteolin (10 μM)/quercetin (10 μM) for 6 h and then treated with 100 μM H2O2 for 1 h. Then cells were detected with DCFH-DA assay. Fluorescent intensity was examined under a fluorescence microscope. Image J was used to analyze the data. Data were expressed as mean ± SD. ∗∗ P < 0.01 versus control; ## P < 0.01 versus model.
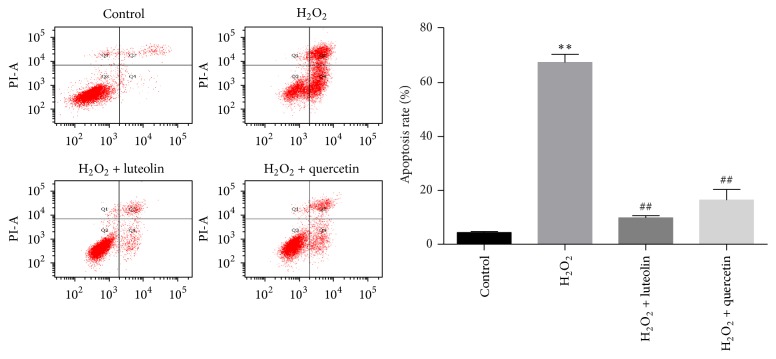
3.5. Luteolin Protected H9C2 Cells from H2O2-Induced Apoptosis
Apoptosis is considered to be one of the pathogenesis of H2O2-induced injury. To analyze the effects of luteolin on apoptosis in H9C2 cells, Annexin V-PI method was used to detect the apoptosis rate of H9C2 cells. As shown in Figure 6, the number of apoptotic cells significantly increased in H2O2 group compared to control group (67.47 ± 3.05% versus 4.33 ± 0.4%, P < 0.01). Pretreatment with luteolin/quercetin (10 μM) for 6 h significantly reduced the percentage of apoptotic cells to 9.8 ± 0.85%/16.4 ± 3.96% (## P < 0.01), suggesting that luteolin could protect H9C2 cells from apoptosis induced by H2O2.
Figure 6.
Luteolin protected H9C2 cells from H2O2-induced apoptosis. H9C2 cells were preincubated with/without luteolin (10 μM)/quercetin (10 μM) for 6 h and then treated with 100 μM H2O2 for 1 h. Cells were harvested and detected with a flow cytometer. Data were expressed as mean ± SD. ∗∗ P < 0.01 versus control; ## P < 0.01 versus model.
3.6. Effects of Luteolin on the mRNA Expression of Mdm2
Mdm2, an E3 ubiquitin ligase, can be induced by increased cellular level of p53 and in turn negatively regulate p53 stability. When cells are exposed to H2O2, apoptosis is induced and expression of p53 is enhanced [20], as well as Mdm2. As shown in Figure 7, mRNA level of Mdm2 in H2O2 group increased slightly compared to control group (P < 0.05). Pretreatment with luteolin could remarkably elevate expression of Mdm2 (P < 0.05).
Figure 7.
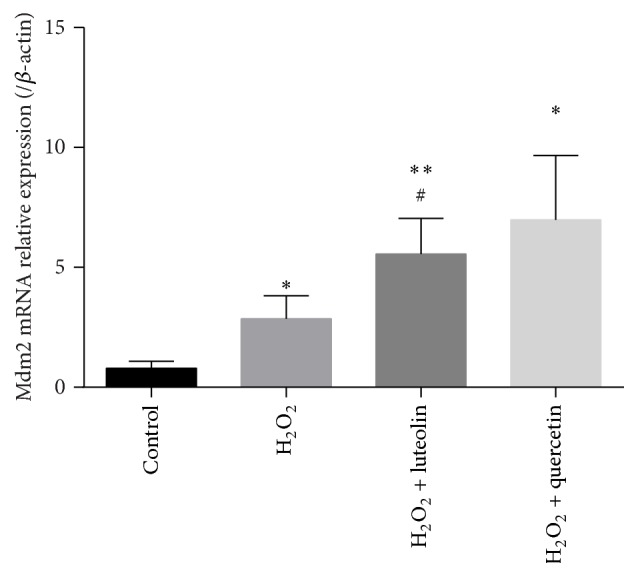
Luteolin increased the mRNA expression of Mdm2. H9C2 cells were preincubated with/without luteolin (10 μM)/quercetin (10 μM) for 6 h and then treated with 100 μM H2O2 for 1 h. Cells were extracted, cDNA was synthesized, and the Mdm2 gene mRNA amount was assessed by quantitative real-time PCR and normalized to the β-actin. Data were expressed as mean ± SD. ∗ P < 0.05 and ∗∗ P < 0.01 versus control; # P < 0.01 versus model.
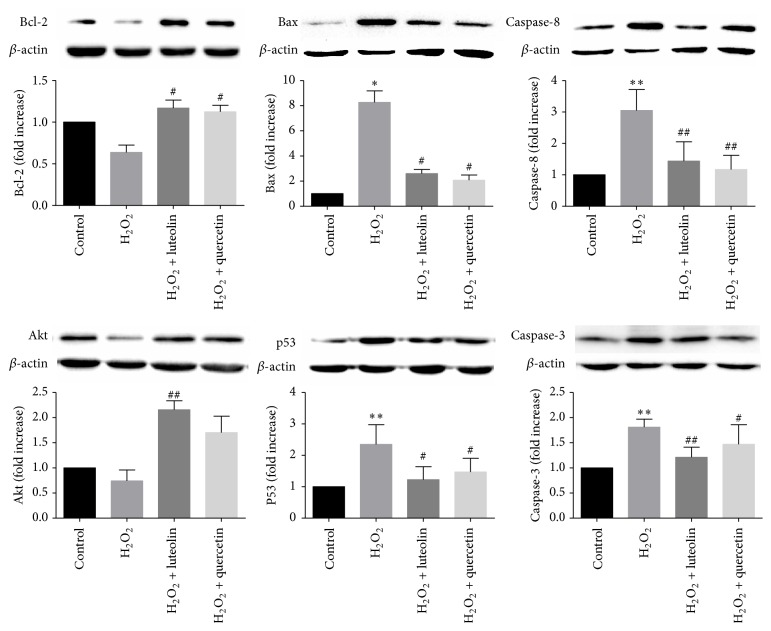
3.7. Effects of Luteolin on the Expression of Apoptosis-Related Proteins
We further investigated the mechanism of luteolin in protecting cells from H2O2-induced apoptosis. Expressions of key molecules in apoptotic signaling pathway were measured by western blot. Bax, caspase-8, cleaved-caspase-3, and p53 are proapoptotic proteins. Akt and Bcl-2 have antiapoptotic effects. As shown Figure 8, in H2O2 group, expressions of Bcl-2 and Akt decreased, whereas the expressions of Bax, caspase-8, cleaved-caspase-3, and p53 increased compared to control group, indicating that apoptotic signaling pathway was activated by H2O2. Pretreatment with luteolin reversed the expressions of these proteins back toward normal levels. The results showed that protection of luteolin against H2O2-induced cell apoptosis may be mediated through Akt-p53 apoptotic signaling pathway.
Figure 8.
Luteolin modulated apoptosis-related proteins. H9C2 cells were preincubated with/without luteolin (10 μM)/quercetin (10 μM) for 6 h and then treated with 100 μM H2O2 for 1 h. The expression of β-actin was measured as an internal control. Data were expressed as mean ± SD. ∗ P < 0.05 and ∗∗ P < 0.01 versus control; # P < 0.05 and ## P < 0.01 versus model.
4. Discussion
Luteolin is a plant flavonoid that exists in a variety of plants and has been shown to have antitumor, antioxidant, antiapoptotic, and anti-inflammatory effects [13–15]. Luteolin has protective effects against cardiovascular diseases, but the mechanism remains unclear. In this study, we investigated the effects of luteolin on apoptosis induced by H2O2 in H9C2 cardiomyocytes and explored the antiapoptotic mechanism. Our study showed that one of the antiapoptotic mechanisms of luteolin was mediated through Akt-p53/Mdm2 signaling pathway.
Studies have shown that apoptosis of cardiac myocytes is an essential process in the progression of heart failure [5]. A study using transgenic mice that expressed a conditionally active caspase demonstrated that low level of apoptosis is sufficient to induce lethal cardiomyopathy and inhibition of cell death can largely prevent the development of HF [21]. Antiapoptotic treatment is likely to become an important form of HF therapy [22].
Previous study showed that H2O2 could induce apoptosis in H9C2 cells [23]. In this study, we also demonstrated that H2O2 has proapoptotic effect on H9C2 cells. Pretreatment with luteolin could increase cell viability and reduce intracellular ROS level and apoptosis rate suggesting that luteolin possesses antioxidant and antiapoptotic properties in cardiomyocytes [24, 25]. Quercetin was applied as positive control drug in this study and it showed similar protective effect as that of luteolin. The cell survival rate in quercetin group was significantly higher than that in H2O2-treated group, indicating that quercetin also has antiapoptotic effects. We further explored the mechanisms by which luteolin exerts antiapoptotic effects.
Extrinsic and intrinsic signaling pathways can both lead to apoptosis. In the extrinsic pathway, caspase-8 activates caspase-3 after it is activated by intracellular and extracellular stimuli [26]. In the intrinsic pathway, permeability of mitochondrial membrane increases and proapoptotic factors, such as cytochrome C, are released to cytoplasm, activating signaling cascades and leading to activation of caspase-3 [27, 28]. The Bcl-2 family proteins, consisting of death antagonists (Bcl-2 and Bcl-xL) and death agonists (Bax and Bak), are also pivotal regulatory components of the cellular apoptosis process [29]. Their primary functions are to protect or disrupt the integrity of mitochondrial membrane and control the release of (pro)apoptotic proteins [5]. Our study showed that H2O2 induced apoptosis by downregulating Bcl-2 and upregulating expressions of proapoptotic proteins, such as Bax, caspase-8, and cleaved-caspase-3. In cells that were pretreated with luteolin/quercetin, expression of Bcl-2 was upregulated and expressions of Bax, caspase-8, and cleaved-caspase-3 were downregulated compared to apoptotic model cells, indicating that luteolin protects cells from apoptosis by modulating anti-(pro)apoptotic proteins.
Apoptosis is an orderly regulated process and p53 is one of the most important modulators in apoptotic signaling pathways [6, 30, 31]. Apoptosis induced by p53 can be divided into two types: the transcription-dependent apoptosis and transcription-independent apoptosis [32]. P53 interacts with transcription coactivator CBP/P300, induces apoptotic mRNA expression in the nucleus, and leads to the transcription-dependent apoptosis. In addition, p53 can bind to antiapoptotic Bcl-2 family proteins, such as Bcl-XL and Bcl-2, and release the proapoptotic effectors Bak/Bax from the complex, thereby inducing transcription-independent apoptosis. Regulating the level of p53 may be an effective target in the prevention of HF.
The activity and stability of p53 are regulated by a variety of enzymes and Mdm2 is one of the most important regulators. Mdm2 inhibits the transactivation ability of p53 by interacting with p53 transactivation domain and decreases the stability of p53 by promoting p53 ubiquitination [32]. Overexpression of Mdm2 exerts antiapoptotic effect and alleviates cardiomyocytes hypertrophy by inhibiting activity of p53 [10]. Ubiquitination activity of Mdm2 can be enhanced when Mdm2 is phosphorylated by Akt [33]. Therefore, activation of Akt could inhibit apoptosis by promoting degradation of p53. In the meantime, elevated cellular level of p53 can induce expression of Mdm2, which in turn suppresses activation of p53. The level of p53 is thus strictly controlled by this feedback loop between p53 and Mdm2 [9, 10]. Studies showed that, in tumor cells, luteolin exerts anticancer effects and promotes cell death by upregulating p53 expression [15, 34]. Luteolin can also exert antiapoptotic properties in cardiovascular diseases. However, the regulatory effect of luteolin on p53/Mdm2 in cardiomyocytes has rarely been reported. Our study showed that p53/Mdm2 pathway was altered in H2O2-treated cells. Expression of p53 was upregulated and expression of Akt was downregulated by stimulus of H2O2, leading to cellular apoptosis. Pretreatment with luteolin rescued cells from apoptosis by increasing expressions of Mdm2 and Akt and suppressing expression of p53. Pretreatment with quercetin only decreased the expression of p53 and had no significant effects on the expressions of Mdm2 and Akt. The results indicated that the antiapoptotic effect of luteolin is mediated by regulating Akt/Mdm2/p53 signaling pathway in H9C2 cells.
Treatment of H9C2 cells with H2O2 could induce cellular apoptosis. ROS level was increased and Akt-p53/Mdm2 apoptotic signaling pathway was activated in H2O2-treated cells. Pretreatment with luteolin could protect H9C2 cells from H2O2-induced apoptosis. The antiapoptotic effect of luteolin was mediated by upregulating expression of Bcl-2 and downregulating apoptotic proteins, including Bax, caspase-8, and cleaved-caspase-3. Furthermore, luteolin could suppress expression of p53 by activating Mdm2 and Akt. Our results illustrate that Akt-p53/Mdm2 signaling pathway is a potential target of luteolin in the prevention of HF.
Acknowledgments
This study was financially supported, in part, by the Grants from the National Science & Technology Pillar Program (no. 2012BAI29B07), the National Natural Science Foundation of China (nos. 81202788, 81473456, 81470191, 81302908, and 81530100), Beijing Natural Science Foundation (no. 7142099), and Excellent Young Scientist Foundation of BUCM (2015-JYB-XYQ001).
Competing Interests
The authors deny any competing interests related to this study.
Authors' Contributions
Hong Chang, Chun Li, and Kuiyuan Huo contribute equally as first coauthors to this paper.
References
- 1.Silva D., Gomez-Sanchez M. A. Heart failure: a new epidemic of cardiovascular disease. Acta Medica Portuguesa. 2016;29(1):10–11. doi: 10.20344/acta20med20port.v29i1.5909. [DOI] [PubMed] [Google Scholar]
- 2.Bleumink G. S., Knetsch A. M., Sturkenboom M. C. J. M., et al. Quantifying the heart failure epidemic: prevalence, incidence rate, lifetime risk and prognosis of heart failure—the Rotterdam Study. European Heart Journal. 2004;25(18):1614–1619. doi: 10.1016/j.ehj.2004.06.038. [DOI] [PubMed] [Google Scholar]
- 3.Nicholson D. W. From bench to clinic with apoptosis-based therapeutic agents. Nature. 2000;407(6805):810–816. doi: 10.1038/35037747. [DOI] [PubMed] [Google Scholar]
- 4.Gill C., Mestril R., Samali A. Losing heart: the role of apoptosis in heart disease—a novel therapeutic target? The FASEB Journal. 2002;16(2):135–146. doi: 10.1096/fj.01-0629com. [DOI] [PubMed] [Google Scholar]
- 5.Van Empel V. P. M., Bertrand A. T. A., Hofstra L., Crijns H. J., Doevendans P. A., De Windt L. J. Myocyte apoptosis in heart failure. Cardiovascular Research. 2005;67(1):21–29. doi: 10.1016/j.cardiores.2005.04.012. [DOI] [PubMed] [Google Scholar]
- 6.Gogiraju R., Xu X., Bochenek M. L., et al. Endothelial p53 deletion improves angiogenesis and prevents cardiac fibrosis and heart failure induced by pressure overload in mice. Journal of the American Heart Association. 2015;4(2) doi: 10.1161/jaha.115.001770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhang Y., Köhler K., Xu J., et al. Inhibition of p53 after acute myocardial infarction: reduction of apoptosis is counteracted by disturbed scar formation and cardiac rupture. Journal of Molecular and Cellular Cardiology. 2011;50(3):471–478. doi: 10.1016/j.yjmcc.2010.11.006. [DOI] [PubMed] [Google Scholar]
- 8.Song H., Conte J. V., Jr., Foster A. H., McLaughlin J. S., Wei C. Increased p53 protein expression in human failing myocardium. The Journal of Heart and Lung Transplantation. 1999;18(8):744–749. doi: 10.1016/s1053-2498(98)00039-4. [DOI] [PubMed] [Google Scholar]
- 9.Schwartzbauer G., Robbins J. The tumor suppressor gene PTEN can regulate cardiac hypertrophy and survival. The Journal of Biological Chemistry. 2001;276(38):35786–35793. doi: 10.1074/jbc.m102479200. [DOI] [PubMed] [Google Scholar]
- 10.Toth A., Nickson P., Liu L. Q., Erhardt P. Differential regulation of cardiomyocyte survival and hypertrophy by MDM2, an E3 ubiquitin ligase. Journal of Biological Chemistry. 2006;281(6):3679–3689. doi: 10.1074/jbc.M509630200. [DOI] [PubMed] [Google Scholar]
- 11.Li X., Zhang J., Huang J., et al. A multicenter, randomized, double-blind, parallel-group, placebo-controlled study of the effects of qili qiangxin capsules in patients with chronic heart failure. Journal of the American College of Cardiology. 2013;62(12):1065–1072. doi: 10.1016/j.jacc.2013.05.035. [DOI] [PubMed] [Google Scholar]
- 12.López-Lázaro M. Distribution and biological activities of the flavonoid luteolin. Mini-Reviews in Medicinal Chemistry. 2009;9(1):31–59. doi: 10.2174/138955709787001712. [DOI] [PubMed] [Google Scholar]
- 13.Xu Y., Zhang J., Liu J., et al. Luteolin attenuate the D-galactose-induced renal damage by attenuation of oxidative stress and inflammation. Natural Product Research. 2016;29(11):1078–1082. doi: 10.1080/14786419.2014.981181. [DOI] [PubMed] [Google Scholar]
- 14.Sung J., Lee J. Anti-inflammatory activity of butein and luteolin through suppression of NFκB activation and induction of heme oxygenase-1. Journal of Medicinal Food. 2015;18(5):557–564. doi: 10.1089/jmf.2014.3262. [DOI] [PubMed] [Google Scholar]
- 15.Xu H., Yang T., Liu X., et al. Luteolin synergizes the antitumor effects of 5-fluorouracil against human hepatocellular carcinoma cells through apoptosis induction and metabolism. Life Sciences. 2016;144:138–147. doi: 10.1016/j.lfs.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 16.Hu J., Man W., Shen M., et al. Luteolin alleviates post-infarction cardiac dysfunction by up-regulating autophagy through Mst1 inhibition. Journal of Cellular and Molecular Medicine. 2016;20(1):147–156. doi: 10.1111/jcmm.12714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nai C., Xuan H., Zhang Y., et al. Luteolin exerts cardioprotective effects through improving sarcoplasmic reticulum Ca2+-ATPase activity in rats during ischemia/reperfusion in vivo. Evidence-Based Complementary and Alternative Medicine. 2015;2015:12. doi: 10.1155/2015/365854.365854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Qi L., Pan H., Li D., Fang F., Chen D., Sun H. Luteolin improves contractile function and attenuates apoptosis following ischemia-reperfusion in adult rat cardiomyocytes. European Journal of Pharmacology. 2011;668(1-2):201–207. doi: 10.1016/j.ejphar.2011.06.020. [DOI] [PubMed] [Google Scholar]
- 19.Fang F., Li D., Pan H., et al. Luteolin inhibits apoptosis and improves cardiomyocyte contractile function through the PI3K/Akt pathway in simulated ischemia/reperfusion. Pharmacology. 2011;88(3-4):149–158. doi: 10.1159/000330068. [DOI] [PubMed] [Google Scholar]
- 20.Diestel A., Drescher C., Miera O., Berger F., Schmitt K. R. L. Hypothermia protects H9c2 cardiomyocytes from H2O2 induced apoptosis. Cryobiology. 2011;62(1):53–61. doi: 10.1016/j.cryobiol.2010.12.003. [DOI] [PubMed] [Google Scholar]
- 21.Wencker D., Chandra M., Nguyen K., et al. A mechanistic role for cardiac myocyte apoptosis in heart failure. The Journal of Clinical Investigation. 2003;111(10):1497–1504. doi: 10.1172/jci200317664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Abbate A., Narula J. Role of apoptosis in adverse ventricular remodeling. Heart Failure Clinics. 2012;8(1):79–86. doi: 10.1016/j.hfc.2011.08.010. [DOI] [PubMed] [Google Scholar]
- 23.Zhang F., Huang B., Zhao Y., et al. BNC protects H9c2 cardiomyoblasts from H2O2-induced oxidative injury through ERK1/2 signaling pathway. Evidence-Based Complementary and Alternative Medicine. 2013;2013:12. doi: 10.1155/2013/802784.802784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yu D., Li M., Tian Y., Liu J., Shang J. Luteolin inhibits ROS-activated MAPK pathway in myocardial ischemia/reperfusion injury. Life Sciences. 2015;122:15–25. doi: 10.1016/j.lfs.2014.11.014. [DOI] [PubMed] [Google Scholar]
- 25.Zhou F., Chen S., Xiong J., Li Y., Qu L. Luteolin reduces zinc-induced tau phosphorylation at Ser262/356 in an ROS-dependent manner in SH-SY5Y cells. Biological Trace Element Research. 2012;149(2):273–279. doi: 10.1007/s12011-012-9411-z. [DOI] [PubMed] [Google Scholar]
- 26.Kerr J. F., Wyllie A. H., Currie A. R. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. British Journal of Cancer. 1972;26(4):239–257. doi: 10.1038/bjc.1972.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Patterson S. D., Spahr C. S., Daugas E., et al. Mass spectrometric identification of proteins released from mitochondria undergoing permeability transition. Cell Death and Differentiation. 2000;7(2):137–144. doi: 10.1038/sj.cdd.4400640. [DOI] [PubMed] [Google Scholar]
- 28.Jiang X., Wang X. Cytochrome c promotes caspase-9 activation by inducing nucleotide binding to Apaf-1. The Journal of Biological Chemistry. 2000;275(40):31199–31203. doi: 10.1074/jbc.c000405200. [DOI] [PubMed] [Google Scholar]
- 29.Volkmann N., Marassi F. M., Newmeyer D. D., Hanein D. The rheostat in the membrane: BCL-2 family proteins and apoptosis. Cell Death and Differentiation. 2014;21(2):206–215. doi: 10.1038/cdd.2013.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jiang X., Guo C.-X., Zeng X.-J., Li H.-H., Chen B.-X., Du F.-H. A soluble receptor for advanced glycation end-products inhibits myocardial apoptosis induced by ischemia/reperfusion via the JAK2/STAT3 pathway. Apoptosis. 2015;20(8):1033–1047. doi: 10.1007/s10495-015-1130-4. [DOI] [PubMed] [Google Scholar]
- 31.Zhou S. F., Yuan J., Liao M. Y., et al. IL-17A promotes ventricular remodeling after myocardial infarction. Journal of Molecular Medicine. 2014;92(10):1105–1116. doi: 10.1007/s00109-014-1176-8. [DOI] [PubMed] [Google Scholar]
- 32.Chi S.-W. Structural insights into the transcription-independent apoptotic pathway of p53. BMB Reports. 2014;47(3):167–172. doi: 10.5483/BMBRep.2014.47.3.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Morisco C., Zebrowski D., Condorelli G., Tsichlis P., Vatner S. F., Sadoshima J. The Akt-glycogen synthase kinase 3β pathway regulates transcription of atrial natriuretic factor induced by β-adrenergic receptor stimulation in cardiac myocytes. The Journal of Biological Chemistry. 2000;275(19):14466–14475. doi: 10.1074/jbc.275.19.14466. [DOI] [PubMed] [Google Scholar]
- 34.Wang T.-T., Wang S.-K., Huang G.-L., Sun G.-J. Luteolin Induced-growth inhibition and apoptosis of human esophageal squamous carcinoma cell line Eca109 cells in vitro. Asian Pacific Journal of Cancer Prevention. 2012;13(11):5455–5461. doi: 10.7314/apjcp.2012.13.11.5455. [DOI] [PubMed] [Google Scholar]