Abstract
Objective
Evaluate the effects of VEGF165 gene transfer in the process of remodeling of the extracellular matrix after an acute myocardial infarct.
Methods
Wistar rats were submitted to myocardial infarction, after the ligation of the left descending artery, and the left ventricle ejection fraction was used to classify the infarcts into large and small. The animals were divided into groups of ten, according to the size of infarcted area (large or small), and received or not VEGF165 treatment. Evaluation of different markers was performed using immunohistochemistry and digital quantification. The primary antibodies used in the analysis were anti-fibronectin, anti-vimentin, anti-CD44, anti-E-cadherin, anti-CD24, anti-alpha-1-actin, and anti-PCNA. The results were expressed as mean and standard error, and analyzed by ANOVA, considering statistically significant if p≤0.05.
Results
There was a significant increase in the expression of undifferentiated cell markers, such as fibronectin (protein present in the extracellular matrix) and CD44 (glycoprotein present in the endothelial cells). However, there was decreased expression of vimentin and PCNA, indicating a possible decrease in the process of cell proliferation after treatment with VEGF165. Markers of differentiated cells, E-cadherin (adhesion protein between myocardial cells), CD24 (protein present in the blood vessels), and alpha-1-actin (specific myocyte marker), showed higher expression in the groups submitted to gene therapy, compared to non-treated group. The value obtained by the relation between alpha-1-actin and vimentin was approximately three times higher in the groups treated with VEGF165, suggesting greater tissue differentiation.
Conclusion
The results demonstrated the important role of myocytes in the process of tissue remodeling, confirming that VEGF165 seems to provide a protective effect in the treatment of acute myocardial infarct.
Keywords: Myocardial infarction, Genetic therapy, Angiogenesis inducing agents, Neovascularization physiologic, Vascular endothelial growth factors
INTRODUCTION
Cardiovascular disease represents one of the main causes of mortality in the world.( 1 ) Unfortunately, current pharmacological treatment for acute myocardial infarction is limited as to the process of ventricle remodeling, besides not preventing progression to cardiac failure.( 2 - 4 )
Gene therapy represents an alternative modality of treatment for cardiac diseases, since it intensifies the action of exogenous angiogenic factors, therefore inducing the formation of new arterial capillaries and promoting remodeling of existing vessels.( 5 ) The possible benefits of gene therapy within the context of acute myocardial infarct include, besides intensification of the action of angiogenic factors, the promotion of myocyte protection (by inhibiting cellular processes such as apoptosis and guaranteeing cell survival) and the alteration of the expression of components responsible for the integrity of the ventricular extracellular matrix.( 6 - 9 ) The vascular endothelial growth factor (VEGF) protein shows molecular heterogeneity and has five different isoforms with 121, 145, 165, 189, and 206 amino acids. Its most common form is a polypeptide homodimer with 165 or 121 amino acids (VEGF165 and VEGF121), and both are capable of increasing the collateral blood flow in experimental models. VEGF121 regulates vascular permeability, while VEGF165 regulates angiogenesis.( 10 )
VEGF165 is a potent mitogen of endothelial cells of arteries, veins, and lymphatic vessels, since besides stimulating the proliferation and migration of endothelial cells, it promotes the formation of new vessels and increases vascular permeability.( 10 - 12 ) The expansion of collateral vessels in the infarcted myocardial tissue, triggers invasion and proliferation of endothelial cells and smooth muscle cells, inducing responses such as mitosis, migration, cell differentiation, degradation, and remodeling of the extracellular matrix. In the heart of an adult individual, the genes that encode induction factors for formation of new vessels, as well as their receptors, are expressed at levels apparently insufficient in response to a chronic ischemia.( 13 , 14 )
VEGF165 presents hypoxia-induced expression and promotes the differentiation of mesenchymal stem-cells in various types of specialized cells, including myocytes, therefore allowing greater cardiac protection, followed by formation of new vessels in the ischemic myocardium.( 15 )
The overexpression of VEGF165 during survival of the myocytes under conditions of hypoxia, besides stimulating the formation of new blood vessels, increases the vascular density and the blood flow in the infarcted zone. Neovascularization determined by VEGF165 is responsible for the decrease in apoptosis of myocytes and for the increased contractility of the cardiac ventricles.( 15 , 16 ) In this way, gene therapy with VEGF165 can affect the fibrous remodeling in the ischemic area and impede the expansion of the scar.( 17 )
Recent studies have suggested a broader role for VEGF165 in the maintenance of the ventricular function, since the drop in number of myocytes associated with myocardial infarct leads to a relative state of deficiency of VEGF165 and this fact contributes towards an alteration of normal cardiac function and of the ventricular structure. Consequently, such discoveries make evident an autocrine and paracrine role of VEGF165 that seems to be able to confer a protective effect on myocardium and extracellular heart matrix, which extends to its role in the formation of new vessels.( 18 , 19 )
Alterations of molecules of the extracellular matrix and of the surface of myocytes after an acute myocardial infarction were evaluated by the antibodies that identify cell differentiation and tissue proliferation, such as an adhesive glycoprotein present in the soluble form in the plasma and insoluble in the extracellular matrix of most tissues responsible for the interaction between the cell surface and the component of the extracellular matrix (anti-fibronectin), cell surface receptor present in monocytes and vascular endothelium cells (anti-CD44), intermediate filament of fibroblasts and mesenchymal cells (anti-vimentin), adhesion protein present in cardiac cells that guarantee adhesion during systole and diastole (anti-E-cadherin), blood vessel marker (anti-CD24), myocyte-specific market (anti-alpha-1-actin), and the marker that identifies the process of cell proliferation, defined as proliferating cell nuclear antigen − anti-PCNA.( 19 )
OBJECTIVE
Evaluate the effects of VEGF165 gene transfer in the process of remodeling of the extracellular matrix after an acute myocardial infarct.
METHODS
Animal Model
Wistar rates (adult females) were anesthetized and submitted to ligation of the left descending coronary artery to provoke an infarction, with subsequent gene transfer to the treated animal group, as previously described by Mataveli et al.( 13 ) Next, 250µg of recombinant DNA plasmid (cDNA) of VEGF165 (pVEGF165) were injected in 150µl of 0.1 M phosphate buffer immediately after the induced infarct. The intramyocardial injection was done at three equidistant points, around the area irrigated by the left portion of the descending coronary artery.
Six weeks after induction of acute myocardial infarction, all animals were submitted to echocardiography before being euthanized. The test had the objective of calculating the size of the infarction on the anterior wall of the left ventricle (LV) and measuring the ejection fraction (EF) as a dimension of cardiac function, as described in detail by Mataveli et al.( 13 )
The groups of animals were divided according to the size of the infarction, that is, infarctions that reached less than 30% of the anterior LV wall were considered small (SAMI), and infarcts that reached more than 30% of the anterior wall of the LV were considered large (LAMI). The group of animals that suffered infarction (LAMI or SAMI) received no treatment with pVEGF165, but served as controls. Each group was made up of ten animals.
This project was approved by the Ethics in Animal Research Committee of the Universidade Federal de São Paulo (UNIFESP), process number 0933/07. Infarct induction in animals was conducted at the animal house of the National Institute of Pharmacology of UNIFESP, during the period between 2008 and 2010. Nevertheless, the immunohistochemical analyses of the different markers were done at the Biochemistry Department Laboratory, at the Faculdade de Medicina do ABC, during the period between 2009 and 2012.
Immunohistochemistry reactions
The primary antibodies used were: anti-fibronectin (sc-6953, Santa Cruz Biotechnology, California, United States), anti-CD-44 (AM310-5M, BioGenex®, Andhra Pradesh, India), anti-vimentin (sc-7558, Santa Cruz Biotechnology, California, United States), anti-E-cadherin (number 4,065, Cell Signaling Technology Inc®, Beverly Hills, United States), anti-CD-24 (sc-7036, Santa Cruz Biotechnology, California, United States), anti-alpha-1-actin (M0851, Dako®, Glostrup, Denmark), and anti-PCNA (sc-56, Santa Cruz Biotechnology, California, United States). The secondary antibody used was LSAB-HRP (Large Streptavidin-Avidin-Biotin-; System Peroxidase; k-0690; Dako®, Glostrup, Denmark). The slides were developed with a chromogene (3-3’-diaminobenzidine (DAB), obtained from Sigma Diagnostics®, St. Louis, United States, and counterstained with Harris’s hematoxylin (Sigma Diagnostics®, St. Louis, United States).
Digital quantification of the immunohistochemistry reactions
For the quantitative analysis of the immunohistochemistry reactions, digital image processing software was used (ImageLab®, Rio de Janeiro, Brazil), following the instructions described by Matos et al.( 20 ) Digital quantification of each immunohistochemical reaction was determined as an expression index (EI), indicated in optic units/mm2 (ou/mm2).
Statistical analyses
The results were expressed as means and standard deviation or median and interquartile variation, assessed by Analysis of Variance (ANOVA), and was considered statistically significant if p≤0.05. For the statistical analyses, the Statistical Package for Social Science (SPSS) software was used, version 13.0 (SPSS Inc., Illinois, United States).
RESULTS
The results were presented according to the group of markers that determined cell undifferentiation, differentiation, and proliferation.
Evaluation of the cell undifferentiation markers
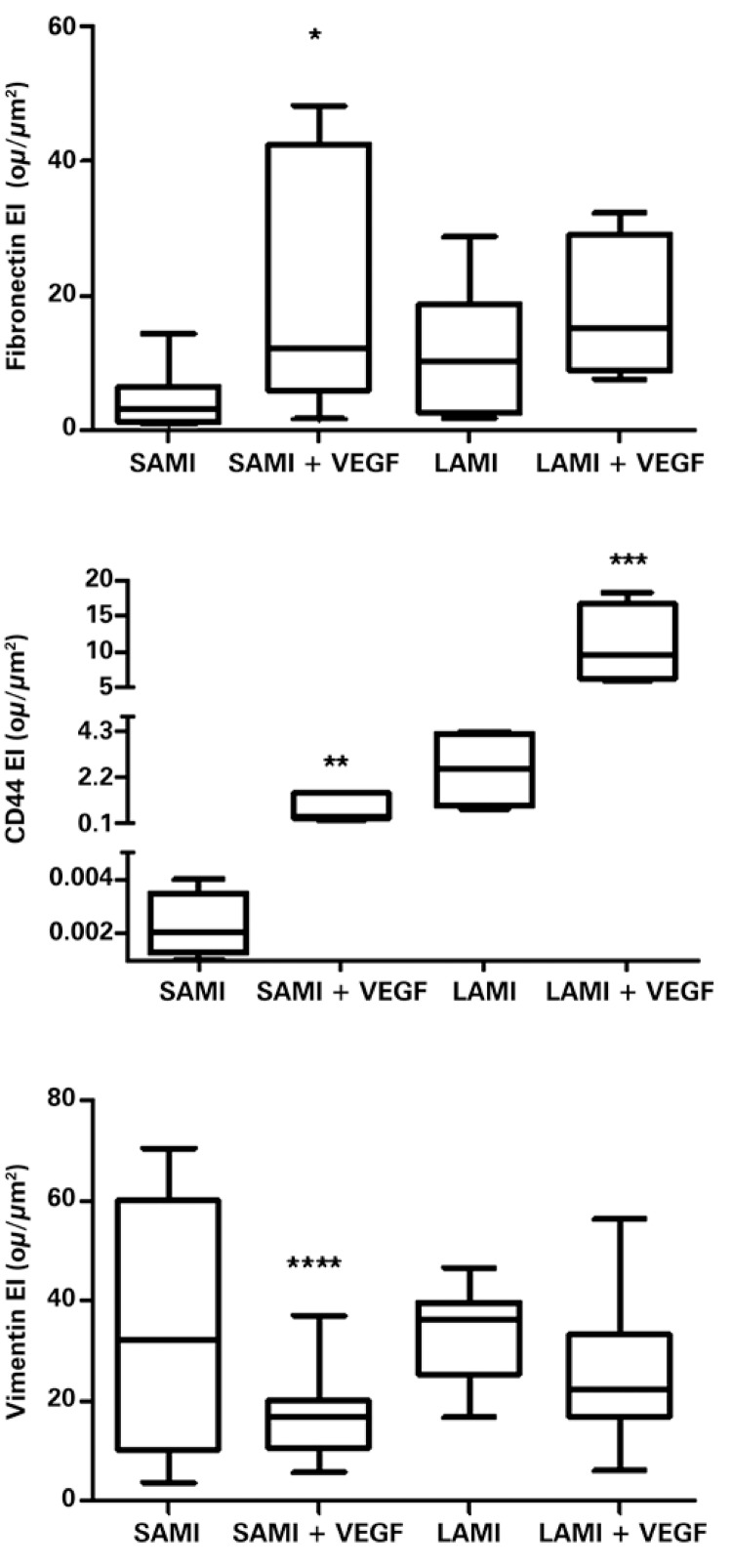
The gene transfer of VEGF165 increased the expression of specific markers that identified undifferentiated cells, such as fibronectin, CD44, and vimentin, as is shown in figure 1.
Figure 1. Expression of fibronectin, CD44, and vimentin in infarcted animals, submitted or not to treatment with VEGF165. The values indicate the median and the interquartile variation, obtained by statistical analysis after determining the expression index (EI), by digital quantification of the immunohistochemical reactions, as was described in details in Methods. There were ten (n=10) animals in each group. Asterisks indicate the p-values obtained for each marker, comparing the results obtidos from animals treated with VEGF versus the non-treated group. *p=0.0125; **p=0.0358; ***p=0.0299; ****p=0.0120. SAMI: group of animals submitted to acute myocardial infarction (small infarction) without treatment; SAMI + VEGF: group of animals submitted to acute myocardial infarction (small infarction) treated with gene transfer containing VEGF165; LAMI: group of animals submitted to acute myocardial infarction (large infarction) without treatment; LAMI + VEGF: group of animals submitted to acute myocardial infarction (large infarction) treated with gene transfer containing VEGF165.
An increase in the expression of fibronectin and of CD44 was noted in the infarcted tissues treated with VEGF165 comparatively with the respective tissues of infarcted groups and not treated with VEGF165. However, vimentin, showed a decrease in expression in the infarcted groups, after treatment with gene therapy, as can be observed in figure 1.
Figure 1 also shows that there was a significant increase in the expression of fibronectin for the group of animals with SAMI, treated with VEGF165, comparatively with the non-treated group – respectively 21.74±6.00ou/µm2 and 4.62±1.43ou/µm2, with p=0.0125. Nevertheless, there was no statistically significant difference in the expression of fibronectin when comparing the group of animals who suffered a treated or non-treated LAMI: 18.2±4.72ou/µm2 and 11.45±2.61ou/µm2, respectively, with p=0.2108.
The significant increase of CD44 for the groups of animals treated with gene transfer, containing the VEGF165 factor, is also demonstrated in figure 1. Values obtained for the non-treated and treated SAMI groups were, respectively, 0.0023±0.01ou/µm2 and 0.82±0.27ou/µm2, with p=0.0358). In the LAMI group, the values obtained for CD44 expression were 2.57±0.91ou/µm2 for the group without treatment and 10.83±2.76ou/µm2 for the treated with VEGF165 (p=0.0299), as is shown in figure 1.
There was greater expression of vimentin in the groups of animals not treated with VEGF165, as can be observed in figure 1, both for LAMI and for SAMI. One can clearly see the significant decrease in the expression of vimentin, when comparing, respectively, the SAMI groups non-treated and treated with VEGF165: 34.18±6.81ou/µm2 and 17.11±2.02ou/µm2, with p=0.0120. However, there was no statistical difference for the non-treated LAMI group (34.00±3.45ou/µm2) and that treated with VEGF165 (25.58±3.025ou/µm2), with p=0.1122.
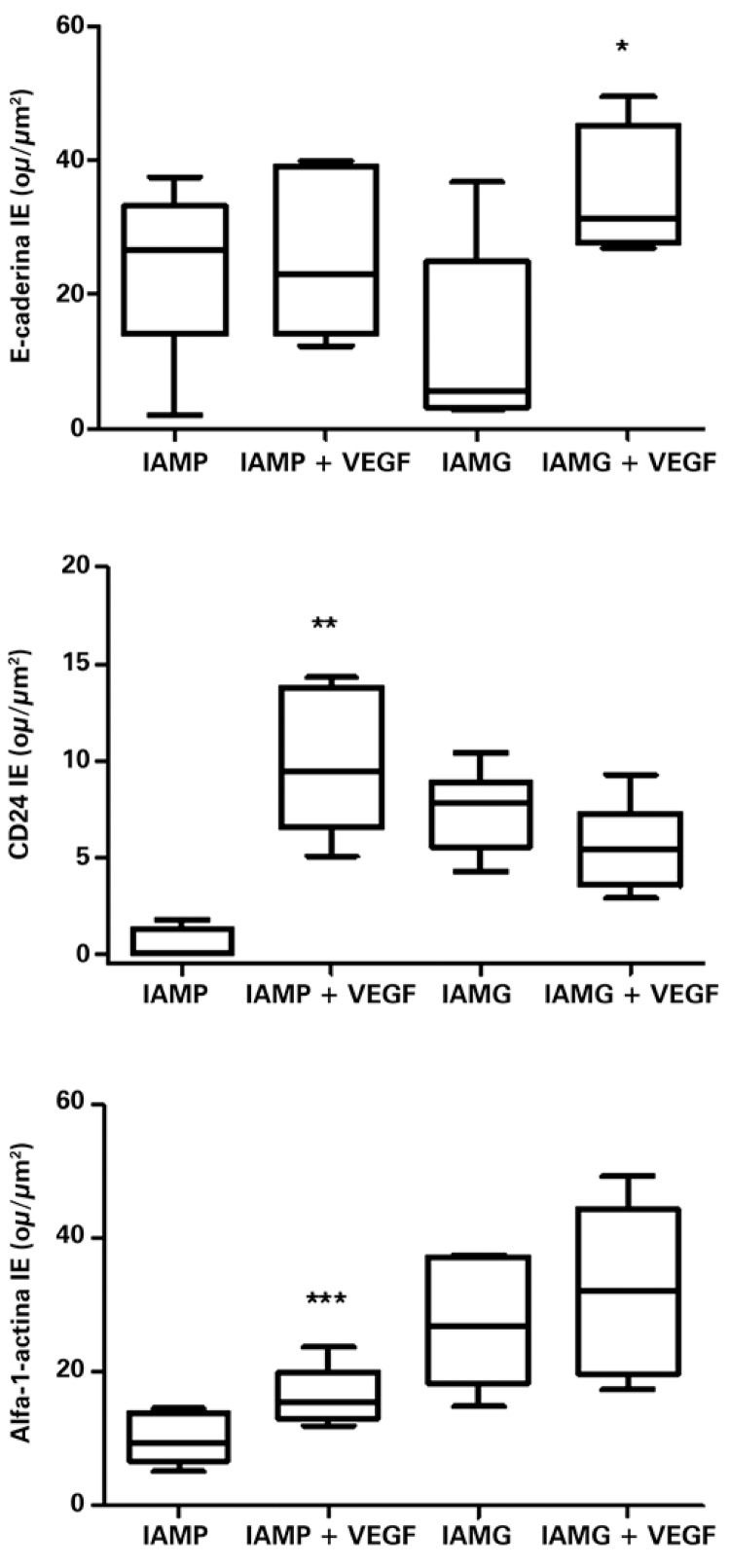
Evaluation of the cell differentiation markers
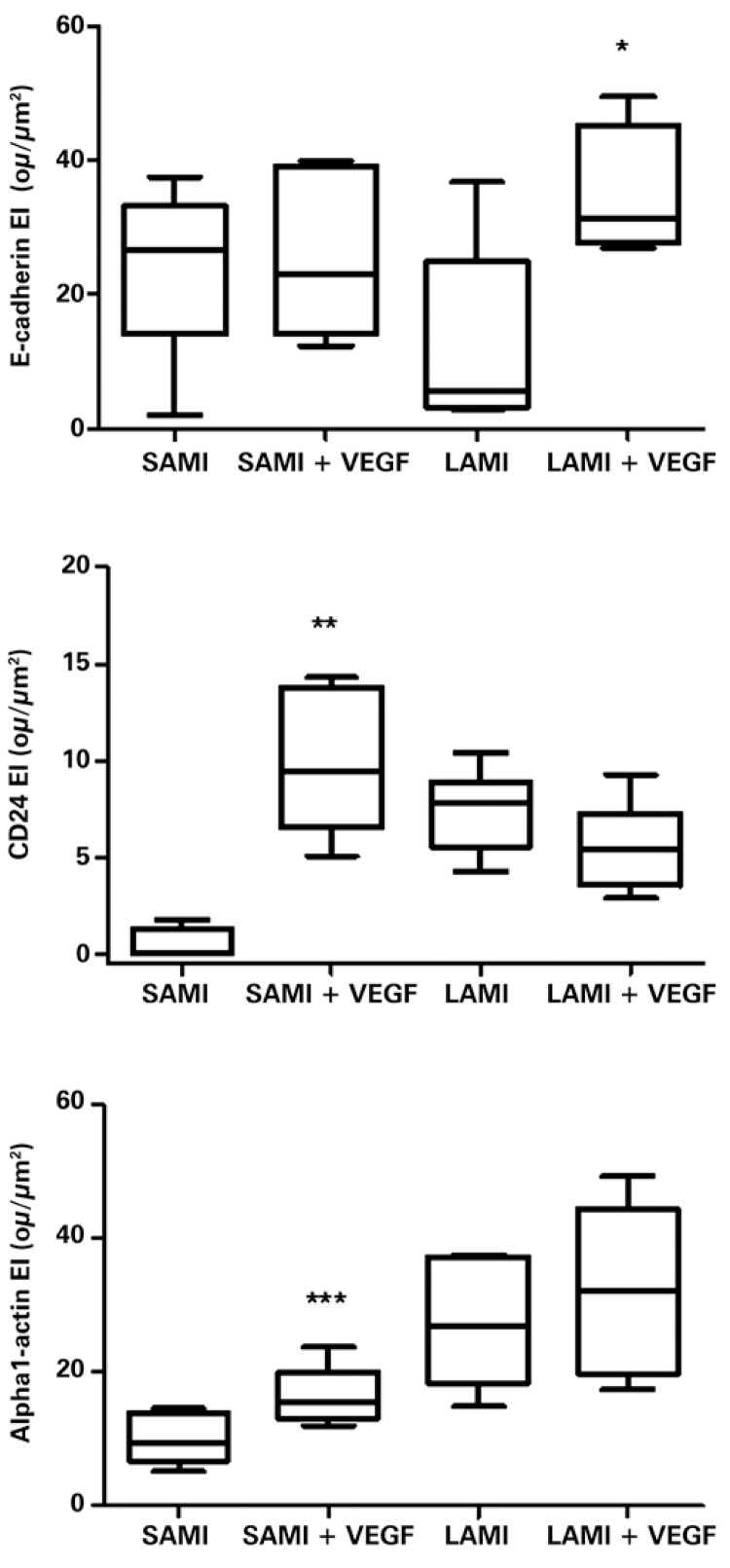
The markers of cellular differentiation were increased in the infarcted tissues that received the recombinant plasmid containing VEGF165, compared to the tissues from the non-treated group.
The E-cadherin showed a significant increase in expression in the LAMI group that received gene therapy, in comparison with the non-treated group (respectively, 34.08±5.03ou/µm2 and 12.40±6.37ou/µm2; p=0.0331), as shown in figure 2. However, no significant difference was seen between the treated group of SAMI and the non-treated with VEGF165 (respectively, 26.87±5.52ou/µm2 and 25.35±4.93ou/µm2; p=0.8419), as can be seen in figure 2.
Figure 2. Expression of E-cadherin, CD24, and alpha-1-actin in infarcted animals submitted or not to treatment with VEGF165. The values indicate the median and interquartile range, obtained by statistical analysis after obtaining the expression index (EI), by digital quantification of the immunohistochemistry reactions, as described in detail in Methods. The number of animals in each group was ten (n=10). Asterisks (*) indicate the p-values obtained for each marker, comparing the results acquired of animals treated with VEGF versus the non-treated group. *p=0.0331; **p=0.0018; ***p=0.0490. SAMI: group of animals submitted to acute myocardial infarction (small infarction) without treatment; SAMI + VEGF: group of animals submitted to acute myocardial infarction (small infarction) treated with gene transfer containing VEGF165; LAMI: group of animals submitted to acute myocardial infarction (large infarction) without treatment; LAMI + VEGF: group of animals submitted to acute myocardial infarction (large infarction) treated with gene transfer containing VEGF165.
Marking of differentiated cells with specific antibodies targeted at proteins present in blood vessels (CD24) and in myocytes (alpha-1-actin) was also analyzed.
The number of vessels, identified by antibody anti-CD24, showed a significant increase (p=0.0018) in the tissues collected from the SAMI group that received treatment with VEGF165 relative to the non-treated animals (respectively, 10.04±1.69ou/µm2 and 0.46±0.44ou/µm2), as can be seen in figure 2.
No significant difference was observed in the expression of CD24 (p=0.1626), when compared to the LAMI group, submitted or not to transfer with VEGF165 (respectively, 5.56±0.91ou/µm2 and 7.45±0.86ou/µm2).
The anti-alpha-1-actin antibody also showed an expressive increase (p=0.0490) in the tissues of animals that presented with SAMI treated with VEGF165 (16.26±1.99ou/µm2) compared with tissues of non-treated animals (10.20±1.69ou/µm2), as can be seen in figure 2 .
There was no significant difference (p=0.5534) in the expression of alpha-1-actin among the groups of animals affected with LAMI, treated with VEGF165 (27.54±4.38ou/µm2) and non-treated (32.02±5.70ou/µm2), as is shown in figure 2.
Relation between differentiation and undifferentiation markerstion
The mean values of EI, obtained by digital quantification of the immunohistochemical reactions, utilizing the different antibodies that mark undifferentiated cells (fibronectin, vimentin, and CD44) and differentiated cells (E-cadherin, CD24, and alpha-1-actin), are summarized on table 1.
Table 1. Expression index of markers of differentiated and undifferentiated cells.
| Fibronectin | Vimentin | CD44 | E-cadherin | CD24 | Alpha-1-actin | |
|---|---|---|---|---|---|---|
| SAMI | 4.62 | 34.18 | 0 | 26.87 | 0.46 | 10.20 |
| SAMI + VEGF | 21.74 | 17.10 | 0.82 | 25.35 | 10.04 | 16.26 |
| LAMI | 11.45 | 34.00 | 2.57 | 12.40 | 7.45 | 27.54 |
| LAMI + VEGF | 18.12 | 25.58 | 10.83 | 34.80 | 5.56 | 32.02 |
SAMI: small infarciont; LAMI: large infarction; VEGF: animals treated with injection of the recombinant plasmid containing VEGF165.
Table 2 shows the value that expresses the relation between the EI of alpha-1-actin (differentiation marker) and vimentin (undifferentiation marker). The greatest value obtained by the alpha-1-actin/vimentin relation indicated that there was a greater degree of cellular differentiation of the tissue analyzed. The choice of alpha-1-actin and vimentin as markers of cell differentiation and undifferentiation was due to the fact that both markers are present in the interior of differentiated (myocytes) and undifferentiated cells, as was mentioned before.
Table 2. Alpha-1-actin and vimentin relation. Note the evidence of the myocardial differentiation level.
| Groups | Alpha-1-actin/vimentin |
|---|---|
| SAMI | 0.3 |
| SAMI + VEGF | 0.95 |
| LAMI | 0.81 |
| LAMI + VEGF | 1.03 |
SAMI: small infarction; LAMI: large infarction; VEGF: animals treated with injection of recombinant plasmid containing VEGF165.
Tissues of animals from the group submitted to treatment with VEGF165 presented with approximately three times greater proportion of cell differentiation markers, comparatively with their respective non-treated groups. This relation shows the increase in proportion of myocytes, after treatment with VEGF165, which suggests that such differentiated cells are important in tissue remodeling.
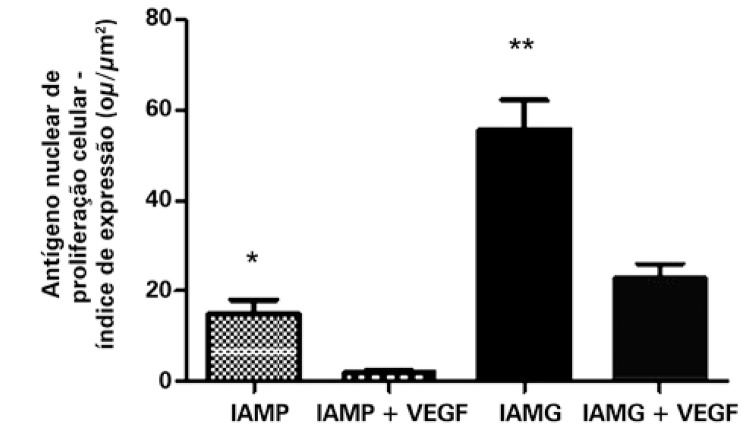
Evaluation of cell proliferation
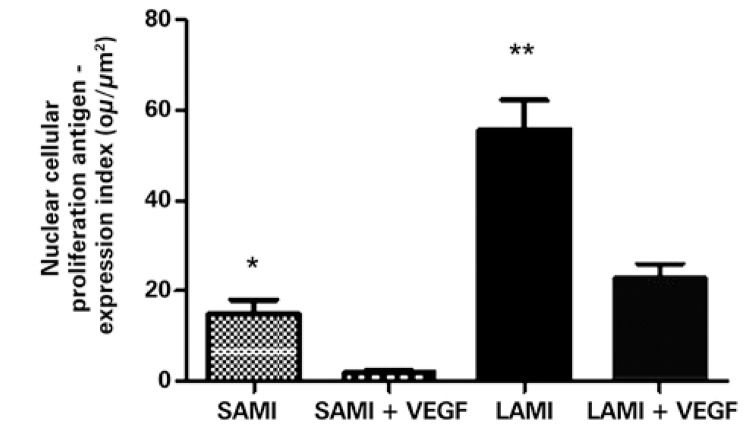
PCNA was significantly diminished in the animals submitted to treatment with VEGF165. Relative to the group that suffered a small infarction, the result was, respectively, for those treated with VEGF165 and not treated: 1.92±0.66ou/µm2 and 14.75±3.36ou/µm2, with p=0.0078. The group that suffered LAMI presented with the result 22.82±3.23ou/µm2 for the treated group and 55.70±6.54ou/µm2 for the non-treated (p=0.0006), as can be seen in figure 3.
Figure 3. Expression of the nuclear cellular proliferation antigen in infarcted animals submitted or not to treatment with VEGF165. The values indicate the mean and standard deviation by analysis of the expression index, after digital quantification of the immunohistochemical reactions, as is described in methods. The number of animals in each group was 10 (n=10). Asterisks (*) indicated the p values obtained for each marker, comparing the results acquired of animals treated with VEGF versus the non-treated group. *p=0.0078; **p=0.0006. SAMI: group of animals submitted to acute myocardial infarction (small infarction) without treatment; SAMI + VEGF: group of animals submitted to acute myocardial infarction (small infarction) treated with gene transfer containing VEGF165; LAMI: group of animals submitted to acute myocardial infarction (large infarction) without treatment; LAMI + VEGF: group of animals submitted to acute myocardial infarction (large infarction) treated with gene transfer containing VEGF165.
DISCUSSION
In results previously published by our group, it was described that the maximal expression of VEGF165 occurs 14 days after gene transfer. Consequently, the evaluation of the samples was done within the period of 6 weeks after induction of the infarction and posterior treatment with gene transfer. Therefore, the late response of the effect promoted by treatment with VEGF165 was evaluated, as had been previously described by Mataveli et al.( 13 )
Literature data suggest that the increase in expression of molecules that determine undifferentiation might be related to the increased degree in cell proliferation.( 10 ) Results obtained from the present study suggest that the increased expression of fibronectin in the infarcted area is related to greater cell proliferation, which occurs in the myocardium after treatment with VEGF165 (specifically in the areas of small infarction).
The significant increase of CD44 after treatment with VEGF165 indicated the formation and regeneration of blood vessels in the myocardium after the infarct, regardless of the size of the infarct.
The decrease in expression of vimentin, which corresponds to an intermediate filament of the cytoskeleton, characteristic of fibroblasts and mesenchymal cells, demonstrated that possibly such cells were not responsible for the processes of cardiac tissue remodeling after the myocardial infarction.
It is important to point out that E-cadherin represents an adhesion protein present in cardiac cells and serves to maintain systole and diastole.( 16 , 19 ) The increased expression of E-cadherin after treatment with VEGF165 indicates that it is likely that this marker has a fundamental role in the protection and maintenance of cardiac contractility, stimulated by gene transfer with VEGF165, mainly in areas of a large infarction.
The increased expression of CD24 in the SAMI group treated with gene transfer suggested that VEGF165 could stimulate the proliferation and migration of endothelial cells, hence promoting the formation of new vessels and greater ease in myocardial tissue regeneration.
An increase in number of blood vessels would be expected after treatment with VEGF165 in both groups of animals, with a small or large infarcted area. However, no significant increase was observed in CD24 in the areas of large infarction. Possibly, the VEGF165 demonstrated a more effective role in treating small myocardial lesions.
The increased expression of the antibody anti-alpha-1-actin in the groups submitted to gene therapy indicated that such a growth factor likely promoted the induction of myocyte proliferation. Therefore, the data obtained demonstrated that myocytes were possibly involved in the process of myocardial extracellular matrix remodeling after the infarction.
The fact that no difference was observed in marking of the myocytes in the tissues of animals of the large infarct group made evident, once again, that possibly VEGF165 was not effective in inducing the proliferation of myocytes, when the area affected by the infarction was very large.
The groups that received treatment with VEGF165 presented with lower expression of PCNA and, therefore, less cell proliferation. The decrease in rate of tissue proliferation after gene transfer of VEGF165 corroborates the data already mentioned that evidenced an intense process of cell differentiation.
Therefore, six weeks after the treatment with VEGF165, it was possible to demonstrate that VEGF165 was related to alterations in the infarction area, responsible for the increased expression of markers specific for cell differentiation. Additionally, the results demonstrated that myocytes were the differentiated cells responsible for such a process. Data obtained in the present study clarified that the damaged tissue after infarction was under intense remodeling and did not present with only scar tissue, as suggested in literature.
CONCLUSIONS
Treatment with VEGF165 represented a fundamental alternative for extracellular matrix remodeling after acute myocardial infarction, promoting stimulation of cell differentiation. Myocytes actively participated in the remodeling process of the extracellular matrix after an acute myocardial infarction.
ACKNOWLEDGMENTS
We wish to thank the financing obtained for conducting this study from the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) [State of São Paulo Research Foundation] with Regular Research Grant, Process number 2011/188638-300, of the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) [National Council of Scientific and Technological Development], Process number 127212/2010-8, and of Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) [Coordination for the Improvement of Higher Education Personnel].
REFERENCES
- 1.Sanderson JE, Mayosi B, Yusuf S, Reddy S, Hu S, Chen Z, et al. Global burden of cardiovascular disease. 1175Heart. 2007;93(10) doi: 10.1136/hrt.2007.131060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.McGinn AN, Nam HY, Ou M, Hu N, Straub CM, Yockman JW, et al. Bioreducible polymer-transfected skeletal myoblasts for VEGF delivery to acutely ischemic myocardium. Biomaterials. 2011;32(3):942–949. doi: 10.1016/j.biomaterials.2010.09.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Katare RG, Kakinuma Y, Arikawa M, Yamasaki F, Sato T. Chronic intermittent fasting improves the survival following large myocardial ischemia by activation of BDNF/VEGF/PI3K signaling pathway. J Mol Cell Cardiol. 2009;46(3):405–412. doi: 10.1016/j.yjmcc.2008.10.027. [DOI] [PubMed] [Google Scholar]
- 4.Novotny NM, Ray R, Markel TA, Crisostomo PR, Wang M, Wang Y, et al. Stem cell therapy in myocardial repair and remodeling. J Am Coll Surg. 2008;207(3):423–434. doi: 10.1016/j.jamcollsurg.2008.04.013. Review. [DOI] [PubMed] [Google Scholar]
- 5.Furlani AP, Kalil RA, Castro I, Cañedo-Delgado A, Barra M, Prates PR, et al. Effects of therapeutic angiogenesis with plasmid VEGF165 on ventricular function in a canine model of chronic myocardial infarction. Rev Bras Cir Cardiovasc. 2009;24(2):143–149. doi: 10.1590/s0102-76382009000200009. [DOI] [PubMed] [Google Scholar]
- 6.Wang R, Crystal RG, Hackett NR. Identification of an exonic splicing silencer in exon 6A of the human VEGF gene. 103BMC Mol Biol. 2009;10 doi: 10.1186/1471-2199-10-103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ward MR, Stewart DJ. Progenitor cell therapy for cardiac regeneration following acute myocardial infarction: So far, so good? Can J Cardiol. 2008;24(C):5C–10C. [Google Scholar]
- 8.Payne TR, Oshima H, Okada M, Momoi N, Tobita K, Keller BB, et al. A relationship between vascular endothelial growth factor, angiogenesis, and cardiac repair after muscle stem cell transplantation into ischemic hearts. J Am Coll Cardiol. 2007;50(17):1677–1684. doi: 10.1016/j.jacc.2007.04.100. [DOI] [PubMed] [Google Scholar]
- 9.Kivelä R, Bry M, Robciuc MR, Räsänen M, Taavitsainen M, Silvola JM, et al. VEGF-B-induced vascular growth leads to metabolic reprogramming and ischemia resistance in the heart. EMBO Mol Med. 2014 Mar;6(3):307–321. doi: 10.1002/emmm.201303147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Levashova Z, Backer M, Backer JM, Blankenberg FG. Imaging vascular endothelial growth factor (VEGF) receptors in turpentine-induced sterile thigh abscesses with radiolabeled single-chain VEGF. J Nucl Med. 2009;50(12):2058–2063. doi: 10.2967/jnumed.109.068023. [DOI] [PubMed] [Google Scholar]
- 11.Yan D, Wang X, Li D, Liu W, Li M, Qu Z, et al. Macrophages overexpressing VEGF target to infarcted myocardium and improve neovascularization and cardiac function. Int J Cardiol. 2013;164(3):334–338. doi: 10.1016/j.ijcard.2011.07.026. [DOI] [PubMed] [Google Scholar]
- 12.Sato D, Otani H, Enoki C, Fujita M, Minato N, Iwasaka T. Phenotypic modulation and turnover of bone marrow-derived cells after myocardial infarction in rats. Cardiovasc Pathol. 2011;20(3):146–155. doi: 10.1016/j.carpath.2010.04.001. [DOI] [PubMed] [Google Scholar]
- 13.Mataveli FD, Han SW, Nader HB, Mendes A, Kanishiro R, Tucci P, et al. Long-term effects for acute phase myocardial infarct VEGF165 gene transfer cardiac extracellular matrix remodeling. Growth Factors. 2009;27(1):22–31. doi: 10.1080/08977190802574765. [DOI] [PubMed] [Google Scholar]
- 14.Jujo K, Li M, Losordo DW. Endothelial progenitor cells in neovascularization of infarcted myocardium. J Mol Cell Cardiol. 2008;45(4):530–544. doi: 10.1016/j.yjmcc.2008.08.003. Review. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ye L, Zhang W, Su LP, Haider HK, Poh KK, Galupo MJ, et al. Nanoparticle based delivery of hypoxia-regulated VEGF transgene system combined with myoblast engraftment for myocardial repair. Biomaterials. 2011;32(9):2424–2431. doi: 10.1016/j.biomaterials.2010.12.008. [DOI] [PubMed] [Google Scholar]
- 16.Zhang Y, Furumura M, Morita E. Distinct signaling pathways confer different vascular responses to VEGF 121 and VEGF 165. Growth Factors. 2008;26(3):125–131. doi: 10.1080/08977190802105909. [DOI] [PubMed] [Google Scholar]
- 17.Hagikura K, Fukuda N, Yokoyama S, Yuxin L, Kusumi Y, Matsumoto T, et al. Low invasive angiogenic therapy for myocardial infarction by retrograde transplantation of mononuclear cells expressing the VEGF gene. Int J Cardiol. 2010;142(1):56–64. doi: 10.1016/j.ijcard.2008.12.108. [DOI] [PubMed] [Google Scholar]
- 18.Vilahur G, Juan-Babot O, Peña E, Oñate B, Casaní L, Badimon L. Molecular and cellular mechanisms involved in cardiac remodeling after acute myocardial infarction. J Mol Cell Cardiol. 2011;50(3):522–533. doi: 10.1016/j.yjmcc.2010.12.021. [DOI] [PubMed] [Google Scholar]
- 19.Xu H, Shi BM, Lu XF, Liang F, Jin X, Wu TH, et al. Vascular endothelial growth factor attenuates hepatic sinusoidal capillarization in thioacetamide-induced cirrhotic rats. World J Gastroenterol. 2008;14(15):2349–2357. doi: 10.3748/wjg.14.2349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Matos LL, Stabenow E, Tavares MR, Ferraz AR, Capelozzi VL, Pinhal MA. Immunohistochemistry quantification by a digital computer-assisted method compared to semiquantitative analysis. Clinics (São Paulo) 2006;61(5):417–424. doi: 10.1590/s1807-59322006000500008. [DOI] [PubMed] [Google Scholar]