Abstract
Purpose: To investigate the effect of blocking renin-angiotensin system by direct renin inhibition using aliskiren on the renal dysfunction following reversible unilateral ureteral obstruction (UO). Methods: Wistar rats underwent reversible left UO for 72 hours. Group-Alsk (n=12) received aliskiren (30 mg/kg/day) dissolved in water starting one day before creating UO and continued until the terminal experiment five days post reversal when renal functions were measured using clearance techniques. Group-Vx (n=12) underwent similar protocol but had water only. Gene expression analysis of some markers of kidney injury was measured using PCR technique. Results: In Group-Vx, renal blood flow (RBF) and glomerular filtration rate (GFR) in the left kidney were significantly lower than the right kidney (1.82±0.12 vs. 3.19±0.40, P=0.001 and 0.81±0.08 vs. 1.44±0.09, P=0.004, respectively). However, left fractional excretion of sodium (FENa) was higher than the right FENa (0.80±0.15 vs. 0.55±0.04, P=0.05). Comparing the left obstructed kidney in Group-Alsk vs. Group-Vx, RBF and GFR were higher in Group-Alsk (2.44±0.30 vs. 1.82±0.12, P=0.049 and 1.02±0.11 vs. 0.81±0.08, P=0.07, respectively). The left renal FENa was lower in Group-Alsk but did not reach statistical significance (0.54±0.07 vs. 0.80±0.15, P=0.07). Aliskiren also decreased the gene expressions of NGAL, KIM-1 and p53. Conclusion: Direct renin inhibition by aliskiren appears to have protective effect on the renal dysfunction and on the markers of renal injury following UO indicating a potential clinical benefit of this agent. Further, this data and the previous studies indicate that blocking renin-angiotensin system at any level has a protective effect in obstructive nephropathy.
Keywords: Ureteral obstruction, aliskiren, renal function
Introduction
Ureteral obstruction (UO) is a relatively common clinical problem and usually cause pain and if prolonged might lead to renal impairment [1]. The renal damage is due to the interaction of various intra-renal systems leading to alterations in hemodynamic and tubular renal functions [2]. Renin-angiotensin system (RAS) plays a central role in orchestrating these alterations and the blockade of RAS at different levels has been shown to ameliorate these obstruction-induced changes [3-6].
The RAS consists of several peptides which interact in a cascade fashion to produce angiotensin-II. This cascade can be blocked at different levels by agents such as angiotensin converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs). Similarly, this blockade can be achieved by direct renin inhibition. Since renin catalyzes the first and rate-limiting step in the RAS cascade, direct renin inhibition is expected, at least theoretically, to be more effective than ACE inhibitors or ARBs in blocking the RAS. Moreover, ACE inhibitors and ARBs have been shown to result in a compensatory increase in renin release which might dampen the effects of these agents [7,8] as demonstrated by the larger effect of renin inhibitor enalkiren in achieving renal vasodilation compared to the ACE inhibitor captopril [9,10].
Historically, direct renin inhibition has not been used successfully in the clinical setup until recently due to the lack of potency or good bioavailability. Aliskiren is the first oral direct renin inhibitor which has been approved by the US Food and Drug Administration. Aliskiren has been shown to have beneficial effects in different renal conditions [11-13]. Few studies have investigated the role of direct renin inhibition by aliskiren on the renal dysfunction induced by UO [14-18]. For instance, aliskiren has been recently shown to ameliorate the renal inflammatory process induced by UO [14,16]. Aliskiren also reduced renal fibrosis in a mice [14] and rat [15,17] models of unilateral UO. However, the effect of direct renin inhibition by aliskiren on the alterations in renal hemodynamic and tubular functional parameters following UO has not been investigated yet. Thus, the aim of this study was to investigate this effect in a rat model of reversible unilateral UO.
Materials and methods
Studies were performed in male Wistar rats weighing 211-261 g at the time of UO. Rats were housed in standard cages and kept in a 12-hour light-dark cycle at 20°C. They were fed a standard rat chow and had free access to water. Animals were fasted for 12 hours before the experimental procedures but had water ad libitum. The experimental protocol was approved by the local animal research ethics committee.
Ureteral obstruction and reversal
The following procedures were carried out under aseptic conditions. Rats were placed on a heated table to maintain rectal temperature at 37-38°C. Each animal was anesthetized with kitamine hydrochloride (80 mg/kg, intraperitoneally, Pantex Holland B.V., Holland) and xylazine Hydrochloride (8 mg/kg, intraperitoneally, Troy Laboratory PTY Limited, NSW, Australia). The left ureter was exposed via a midline abdominal incision and obstructed by placing a 3 mm length of bisected PVC tubing (0.58 mm internal diameter) around the left midureter as described previously [19,20]. The ureter was then occluded by constricting the tubing with a 4-0 silk suture. At the end, the wound was closed in layers.
The reversal of UO was performed 72 hours later using similar anesthesia as described above. Using a dissecting microscope, and through the same incision, the obstructing tube was identified and removed. Full release of the obstruction was confirmed by observation of a free flow of urine across the site of obstruction. The wound was then closed in layers.
Aliskiren/vehicle administration
Aliskiren (Novartis Pharma Stein AG, Stein, Basle, Switzerland) was dissolved in 0.5 ml of water (vehicle) and administered by gavage immediately after preparation as single daily dose of 30 mg/kg. Control animals received only 0.5 ml of water. In the two groups, treatment was commenced 24 hours before UO and continued daily throughout the period of obstruction and for 5 days thereafter until the time of the terminal experiment.
Experimental groups
Animals were divided into two groups:
Group-Vx (n=12): Rats which underwent left UO for 72 hours and received water only.
Group-Alsk (n=12): Rats which underwent left UO and received aliskiren.
Surgical procedure in the terminal experiment
All rats underwent terminal experiment five days following reversal of UO. Animals were anaesthetised with pentobarbital sodium (60-70 mg/kg, intraperitoneally; Sigma Life Science, St Louis, USA) and the trachea was cannulated. Following cannulation of a femoral vein with polyethylene tubing (PE-50), anaesthesia was maintained by a continuous infusion of pentobarbital sodium (12.5 mg/kg/hr) and a sustaining infusion of 0.9% saline was established at a rate of 50 µl/min using an infusion pump. A femoral artery was cannulated with similar tubing used in the femoral vein and the tip of the cannula was positioned just below the level of the left renal artery. The cannula was connected to a pressure transducer (Memscap, Skoppum, Norway). The blood pressure signal was amplified using a bridge Amp (ADInstruments, Castle Hill, Australia), digitised using Power Lab 4/30 and Lab Chart version-6 software (ADInstruments, Australia) and displayed on a computer screen. The arterial cannula was also used to obtain blood samples throughout the procedure as required. Both kidneys were exposed through a midline abdominal incision and the upper ureters were cannulated with polyethylene tubing (PE-10) for the collection of urine into pre-weighed micro-capped tubes. The urine volume was determined gravimetrically.
On completion of surgery, the sustaining infusion of 0.9% saline was replaced by one composed of Fluorescein isothiocyanate-inulin (FITC-inulin, Sigma-Aldrich, St Louis, USA) (2.5 mg/ml) and para-aminohippuric acid (PAH, Sigma-Aldrich, St Louis, USA) (0.2% w/v) in 0.9% saline. A priming dose of 2 ml of the same solution was infused over 2 minutes. Animals were allowed 2 hours to equilibrate before being subjected to the experimental protocol.
Experimental protocol and assays
The terminal experiment was performed to determine the hemodynamic functions including glomerular filtration rate (GFR) and renal blood flow (RBF) as well as the tubular functions (urine volume (UV), urinary sodium (UNaV) and fractional excretion of sodium (FENa) The experimental protocol consisted of two 30-minute clearance periods. Arterial blood samples (0.4 ml) taken at the beginning and end of the clearance periods were immediately centrifuged. Plasma samples (125 µl) were frozen to be assayed later. The plasma was replaced by an equal volume of saline and the erythrocytes were re-suspended by gentle vortexing and returned to the animal. The hematocrit was determined. Finally, after euthanizing the animals, the kidneys were removed, weighed and prepared for gene expression analysis (vide infra).
Urine and plasma samples were assayed for sodium level using a flame photometer (Corning, Halstead, Essex, England). GFR was estimated from the clearance of inulin. RBF was calculated using the formula [RBF=ERPF/(1-hematocrit)], where the PAH clearance was used to estimate ERPF (effective renal plasma flow). The values of GFR, RBF, UV, UNaV and FENa were calculated as the average of the two clearance periods and were corrected for kidney weight.
Gene expression analysis
The middle part of each kidney was excised, immediately snap-frozen in liquid nitrogen and stored at -80°C for a later measurement of gene expression of neutrophil gelatinase-associated lipocalin (NGAL) and kidney injury molecule-1 (KIM1) which were used as markers of renal tubular injury and the apoptotic protein p53 which was used as a marker of apoptotic response.
Total RNA was extracted using TRI Reagent® Solution (Life Technologies Corporation, NY, USA) according to the manufacturer protocol. Quality and quantity of the extracted RNA was estimated using NanoDrop instrument (Thermo Fisher Scientific Inc., DE, USA). First-strand cDNAs were prepared in duplicates from 2.0 µg of the extracted RNA with GoScript™ Reverse Transcriptase (Promega Corporation, Wisconsin, USA) in the presence of RNasin® Plus RNase inhibitor (Promega Corporation, Wisconsin, USA) according to manufacturer protocols. Prepared cDNA was used as a template for the relative gene expression analysis by real time PCR using TaqMan® chemistry on QuantStudio™ 7 Flex Real-Time PCR system (Applied Biosystems, CA, USA). The reaction mixture consisted of 75 ng cDNA, TaqMan® Universal Master Mix (Applied Biosystems, CA, USA), 0.6 μM of forward and reverse primers and 0.25 μM of the fluorescent probes (Biosearch Technologies, Inc., CA, USA). Sequences of primers and fluorogenic probes are listed in Table 1. Primers and probes were designed using the online RealTimeDesign™ software (Biosearch Technologies, Inc., CA, USA) in a way that at least one of the primers was spanning an exon-exon junction within their respective gene. Peptidylprolyl isomerase A (ppia) (cyclophilin A) was used as the endogenous control gene for normalization between samples and it was multiplexed within each PCR reaction.
Table 1.
Forward and reverse primers and fluorogenic probe sequences used for real time quantitative PCR analysis
| Gene | Gene Bank Reference | 5’-3’ Sequence | Size | |
|---|---|---|---|---|
| NGAL | NM_130741.1 | Forward | CTGTTCCCACCGACCAATGC | 69 |
| Reverse | CCACTGCACATCCCAGTCA | |||
| Probe | d FAM-TGACAACTGAACAGACGGTGAGCG-BHQ-1 | |||
| kIM-1 | NM_173149.2 | Forward | GCCTGGAATAATCACACTGTAAG | 75 |
| Reverse | GCAACGGACATGCCAACATAG | |||
| Probe | d FAM-TCCCTTTGAGGAAGCCGCAGA-BHQ-1 | |||
| p53 | NM_030989.3 | Forward | CGAGATGTTCCGAGAGCTGAATG | 86 |
| Reverse | GTCTTCGGGTAGCTGGAGTG | |||
| Probe | d FAM-CCTTGGAATTAAAGGATGCCCGTGC-BHQ-1 | |||
Calculated CT values were used to estimate changes in gene expression of target genes using delta-delta CT formula.
The results are expressed as the mean fold change of gene expression in the left obstructed kidney compared to right control kidney in the respective group.
Statistical analysis
Statistical analysis was performed using SPSS V16.0. Results were expressed as means ± SEM. One-way factorial ANOVA was used for comparison of variables between the two groups and between the control and obstructed kidneys within each group. P value of less than 0.05 was considered statistically significant.
Results
The mean arterial blood pressure and heart rate in Group-Vx and Group-Alsk were similar (121±3 vs. 119±4, P=0.8 and 461±7 vs. 463±17, P=0.5, respectively).
Glomerular and tubular functions
In Group-Vx which had left UO but did not receive aliskiren, left RBF, five days following reversal of UO, was 57% of the right RBF (1.82±0.12 vs. 3.19±0.40, P=0.005). Similarly, left GFR was 55% that of the right GFR (0.81±0.08 vs. 1.44±0.09, P=0.0001) (Figure 1). With the decrease in both RBF and GFR, there was an increase in the FENa in the left kidney compared to the right kidney (0.80±0.15 vs. 0.55±0.04, P=0.05). This was associated with a decrease in both the UV and UNaV in the left kidney but both did not also reach statistical significance (8.4±1.6 vs. 15.5±4.7 and 1.1±0.3 vs. 2.1±0.6, respectively, P=0.08 for both) (Figure 2).
Figure 1.
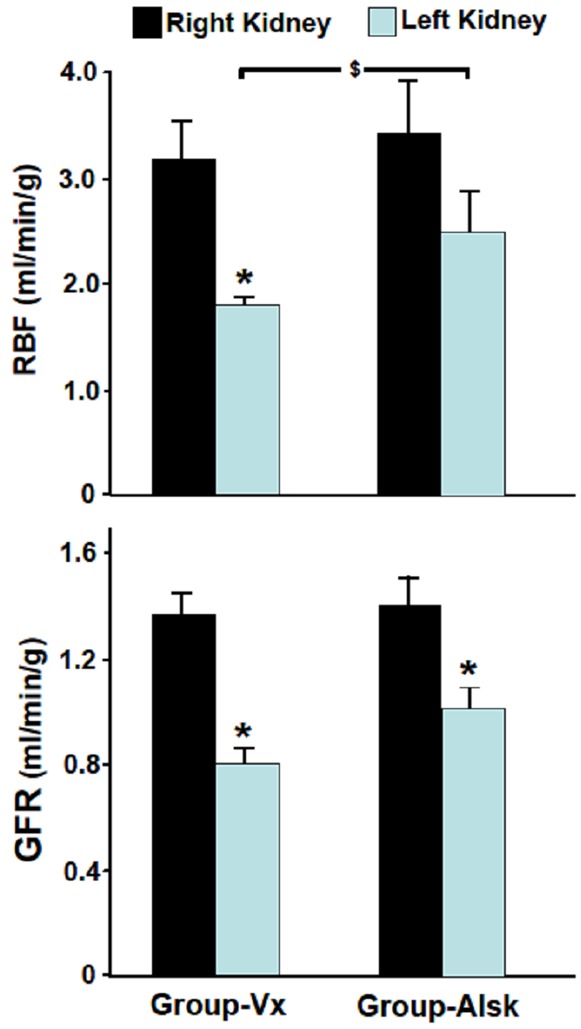
The glomerular filtration rate (GFR) and renal blood flow (RBF) in the right and left kidneys in Group-Vx and Group-Alsk following reversal of unilateral UO. Values represent mean ± SEM. *indicates statistical significance between the right and left kidney within the same group whereas $indicates statistical significance between the left kidneys in both groups.
Figure 2.
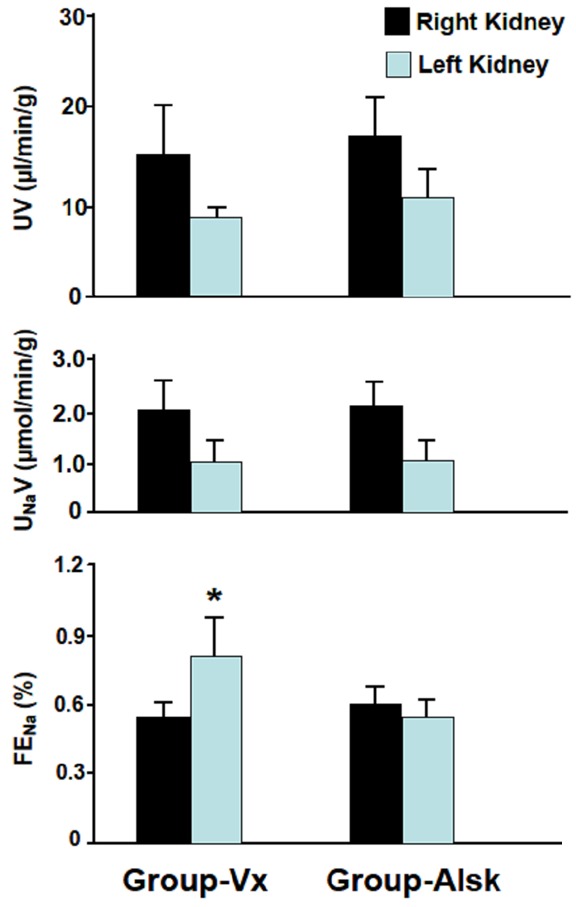
The tubular functional parameters including urine volume (UV), urinary sodium (UNaV) and fractional excretion of sodium (FENa) in both kidneys in Group-Vx and Group-Alsk following reversal of unilateral UO. Values represent mean ± SEM. *indicates statistical significance between the right and left kidney within the same group.
In Group-Alsk which received aliskiren, the left RBF was 71% of the right RBF (2.44±0.34 vs. 3.42±0.45, P=0.09) and the left renal GFR was 72% of the right GFR (1.02±0.11 vs. 1.41±0.14, P=0.04) (Figure 1). As shown in Figure 2, the UV, UNaV and FENa of the left kidney were not significantly different from those of the right kidney (11.0±2.9 vs. 17.3±4.1 (P=0.2), 1.09±0.26 vs. 2.16±0.47 (P=0.07) and 0.54±0.07 vs. 0.59±0.07 (P=0.6).
When Group-Alsk was compared to Group-Vx, all variables in the right kidneys in both groups were similar (P>0.05 for all variables). However, when the left obstructed kidneys in the two groups were compared, the left RBF was higher in Group-Alsk (2.44±0.34 vs. 1.82±0.12, P=0.05). The left GFR was also higher in Group-Alsk but did not reach statistical significance (1.02±0.11 vs. 0.81±0.08, P=0.07) (Figure 1). As shown in Figure 2, left renal FENa was lower in Group-Alsk but also did not reach statistical significance (0.54±0.07 vs. 0.80±0.15, P=0.07) (Figure 2). However, the UV and UNaV were similar in both groups (P>0.05 for both variables). All variables in the right kidneys in both groups were similar (P>0.05 for all variables).
Gene expression analysis results
As demonstrated in Figure 3, in Group-Vx, there was 5.5±1.3 fold increase in the expression of NGAL in the left obstructed kidney compared to the right control kidney, whereas in Group-Alsk, there was only 2.7±0.26 fold increase (P=0.03). Similarly, the left to right kidney expression of KIM-1 was lower in Group-Alsk but this did not reach statistical significance (115±46 vs. 52±9, P=0.09). The left to right kidney expression of p53 was also significantly lower in the Group-Alsk (1.34±0.05 vs. 1.10±0.02, P=0.01).
Figure 3.
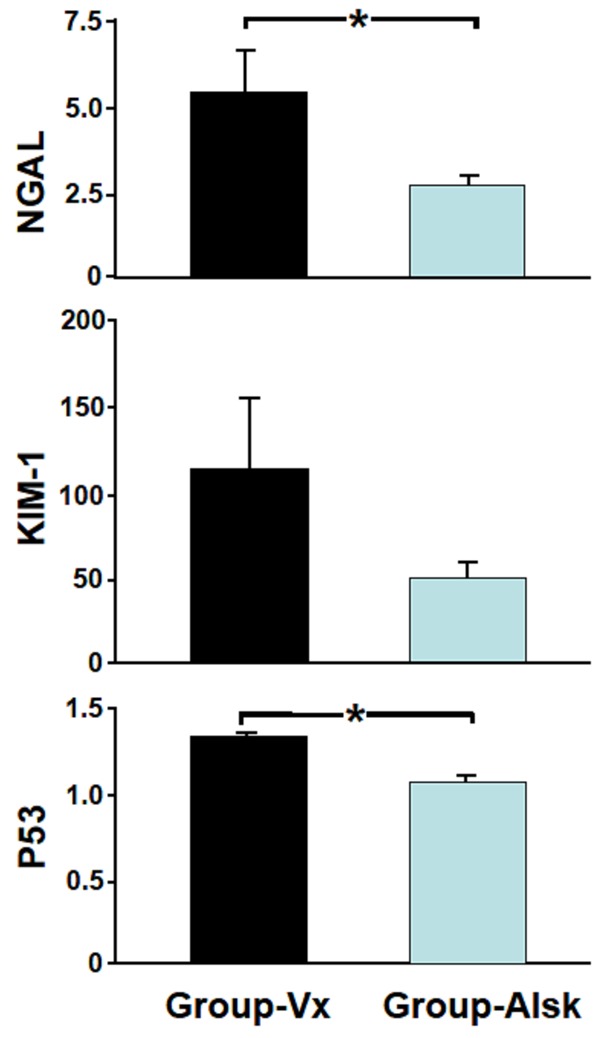
The expression of neutrophil gelatinase-associated lipocalin (NGAL), kidney injury molecule-1 (KIM-1) p53 in both groups. The results are expressed as the mean fold change of gene expression in the left obstructed kidney compared to right control kidney in each group. Values represent mean ± SEM. *indicates statistical significance between groups.
Discussion
In the current study, we have demonstrated for the first time that direct renin inhibition by aliskiren prior to, during and following reversal of unilateral UO has resulted in a significant improvement in the hemodynamic and tubular renal functional parameters as well as an attenuation of the gene expression of some of markers of renal injury.
RAS plays an important role in the development of UO-induced renal dysfunction. Angiotensin-II is synthetized de novo in the kidney and the level of intrarenal angiotensin-II has been shown to increase in the obstructed kidney [4,21]. This increase appears to mediate the decrease of GFR and RBF following UO as shown by the ability of both ACE inhibitors or ARBs to partially prevent the reductions in GFR and RBF observed in UO [3]. Further, ARBs increased the level of several important sodium transporters [22], indicating a potential role of RAS in the renal tubular dysfunction observed in obstructed kidneys. These effects might not be merely due to decreased production of angiotensin-II but also could be due to the associated decrease in the level of several other substances and cytokines which were shown to play an important role in the UO-induced renal impairment [23,24]. Collectively, these data and the data from the current study indicate that blocking RAS at any level results in an improvement of the UO-induced renal dysfunction.
Whether direct renin inhibition by aliskiren is more superior to ACE inhibitors or ARBs in attenuating the UO-induced renal alterations would require a direct comparative study using the three types of medications in the same model. However, it is expected that the effect might depend on the drug dose, species and model of obstruction including the period, laterality and reversibility of obstruction. In general, direct renin inhibition might be more effective than other methods of blocking RAS because it does not only decreases angiotensin-II production but also decreases plasma renin activity, an effect which is not observed in ACE inhibitors or ARBs [7-10]. Regardless of the superiority of any agent, blocking RAS at more than one level has been shown to be more beneficial in preventing the renal alterations following UO [17].
In the current study, aliskiren improved the RBF and to some extent the GFR in the previously obstructed kidney. These effects are similar to the effect of ACE inhibitors or ARBs as shown previously [3]. In this study, there was some improvement in the GFR although it did not reach statistical significance. The exact reason is difficult to ascertain but it could be due to the possible selective effect of angiotensin-II on efferent as compared to afferent glomerular arterioles. Angiotensin-II was shown to have preferential effect on the efferent arterioles which results in the maintaining of GFR especially in some pathological condition [25]. Therefore, decreasing the level of angiotensin-II by aliskiren might have resulted in eliminating this selective effect on efferent arterioles and hence a lower effect on GFR compared to its effect on RBF.
The current data indicates that aliskiren does not only affect the hemodynamic functions but also the tubular renal functions. So aliskiren significantly reduced the increase in FENa observed in the obstructed kidney and hence its ability to concentrate urine. This is consistent with the previous findings of the effect of RAS blockade by ACE inhibitors or ARBs [22,26]. The lack of significant effect of aliskiren on total UV and UNaV is probably due to the fact that these parameters are not determined only by the FENa which has decreased by aliskiren but also by GFR which has increased somewhat by aliskiren resulting in a lack of significant change in these two parameters as a net result.
Aliskiren has been shown to have a higher specificity for human and mouse renin compared to rat renin [27]. In this study, the dose of aliskiren was similar to that used in other studies in rat models [11]. The significant effect of aliskiren on hemodynamic and tubular parameters indicates that bioavailability was not a major issue in this study.
In addition to ameliorating the alterations in renal functional parameters, aliskiren decreased the expression of some of the markers of renal tubular injury such as NGAL and KIM-1. Each of the markers is heavily expressed by different parts of the renal tubules and hence, it was possible to determine the exact site of aliskiren effect. KIM-1 has been shown to be strongly expressed and released by injured proximal tubular epithelial cells [28] whereas NGAL is synthesized in the thick ascending limb of Henle’s loop and collecting ducts [29]. In this study, the effect of aliskiren administration on NGAL was more pronounced than the effect on KIM-1. Although, it is difficult to determine the reason for this differential effect from this study, the results might indicate a weaker effect of aliskiren on the proximal tubules where the majority of the filtered sodium gets reabsorbed. This is consistent with the weak effect of aliskiren on the FENa demonstrated in the current study.
In addition to its effect on NGAL and KIM-1, aliskiren decreased the expression of p53 which is a marker of apoptotic response. Renal p53 expression has been shown previously to increase following ureteric obstruction [23] and the mechanism of this rise remains unclear, although increased renin production, ischemia, and elevated intra-tubular pressure has been suggested to be relevant [30]. The results of this study lend support to the role of RAS in this rise. Regardless of the exact mechanism, the observed effect on p53 in the current study indicates a protective effect on the ongoing renal fibrosis observed in obstructive nephropathy [17,23] and is consistent with the previous findings [14].
The obstruction model used in this study is similar to the clinical scenario of a transiently obstructing ureteral calculus. The beneficial effect of aliskiren on the kidney might be of clinical interest in patients with obstructing ureteral calculus especially those with a single kidney or those with compromised renal functions; however, further clinical studies are required to extrapolate the results to humans.
In conclusion, direct renin inhibition by aliskiren before, during and after release of unilateral UO appears to have a protective effect on the hemodynamic and tubular renal functional alterations as well as on the markers of renal injury. This indicates a potential benefit of this agent in the clinical set-up. Further, this data and the previous findings indicate that blocking RAS at any level has a protective effect in obstructive nephropathy.
Acknowledgements
This study was funded by an individual grant from the College of Medicine & Health Sciences, United Arab Emirates University.
Disclosure of conflict of interest
None.
References
- 1.Klahr S, Harris K, Purkerson ML. Effects of obstruction on renal functions. Pediatr Nephrol. 1988;2:34–42. doi: 10.1007/BF00870378. [DOI] [PubMed] [Google Scholar]
- 2.Klahr S. Pathophysiology of obstructive nephropathy. Kidney Int. 1983;23:414–26. doi: 10.1038/ki.1983.36. [DOI] [PubMed] [Google Scholar]
- 3.Frøkiaer J, Djurhuus JC, Nielsen M, Pedersen EB. Renal hemodynamic response to ureteral obstruction during converting enzyme inhibition. Urol Res. 1996;24:217–27. doi: 10.1007/BF00295895. [DOI] [PubMed] [Google Scholar]
- 4.Frøkiaer J, Knudsen L, Nielsen AS, Pedersen EB, Djurhuus JC. Enhanced intrarenal angiotensin II generation in response to obstruction of the pig ureter. Am J Physiol. 1992;263:F527–33. doi: 10.1152/ajprenal.1992.263.3.F527. [DOI] [PubMed] [Google Scholar]
- 5.Yanagisawa H, Morrissey J, Morrison AR, Purkerson ML, Klahr S. Role of ANG II in eicosanoid production by isolated glomeruli from rats with bilateral ureteral obstruction. Am J Physiol. 1990;258:F85–93. doi: 10.1152/ajprenal.1990.258.1.F85. [DOI] [PubMed] [Google Scholar]
- 6.Yarger WE, Schocken DD, Harris RH. Obstructive nephropathy in the rat: possible roles for the renin-angiotensin system, prostaglandins, and thromboxanes in postobstructive renal function. J Clin Invest. 1980;65:400–12. doi: 10.1172/JCI109683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Johnston CI, Millar JA, McGrath BP, Matthews PG. Long-term effects of captopril (SQ14 225) on blood-pressure and hormone levels in essential hypertension. Lancet. 1979;2:493–6. doi: 10.1016/s0140-6736(79)91552-6. [DOI] [PubMed] [Google Scholar]
- 8.Jones MR, Sealey JE, Laragh JH. Effects of angiotensin receptor blockers on ambulatory plasma Renin activity in healthy, normal subjects during unrestricted sodium intake. Am J Hypertens. 2007;20:907–16. doi: 10.1016/j.amjhyper.2007.04.009. [DOI] [PubMed] [Google Scholar]
- 9.Fisher ND, Allan D, Kifor I, Gaboury CL, Williams GH, Moore TJ, Hollenberg NK. Responses to converting enzyme and renin inhibition. Role of angiotensin II in humans. Hypertension. 1994;23:44–51. doi: 10.1161/01.hyp.23.1.44. [DOI] [PubMed] [Google Scholar]
- 10.Cordero P, Fisher ND, Moore TJ, Gleason R, Williams GH, Hollenberg NK. Renal and endocrine responses to a renin inhibitor, enalkiren, in normal humans. Hypertension. 1991;17:510–6. doi: 10.1161/01.hyp.17.4.510. [DOI] [PubMed] [Google Scholar]
- 11.Hammad FT, Al-Salam S, Lubbad L. Does aliskiren protect the kidney following ischemia reperfusion injury? Physiol Res. 2013;62:681–90. doi: 10.33549/physiolres.932485. [DOI] [PubMed] [Google Scholar]
- 12.Whaley-Connell A, Nistala R, Habibi J, Hayden MR, Schneider RI, Johnson MS, Tilmon R, Rehmer N, Ferrario CM, Sowers JR. Comparative effect of direct renin inhibition and AT1R blockade on glomerular filtration barrier injury in the transgenic Ren2 rat. Am J Physiol Renal Physiol. 2010;298:F655–61. doi: 10.1152/ajprenal.00373.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhou G, Liu X, Cheung AK, Huang Y. Efficacy of aliskiren, compared with angiotensin II blockade, in slowing the progression of diabetic nephropathy in db/db mice: should the combination therapy be a focus? Am J Transl Res. 2015;7:825–40. [PMC free article] [PubMed] [Google Scholar]
- 14.Choi DE, Jeong JY, Lim BJ, Chang YK, Na KR, Shin YT, Lee KW. Aliskiren ameliorates renal inflammation and fibrosis induced by unilateral ureteral obstruction in mice. J Urol. 2011;186:694–701. doi: 10.1016/j.juro.2011.03.122. [DOI] [PubMed] [Google Scholar]
- 15.Sakuraya K, Endo A, Someya T, Hirano D, Murano Y, Fujinaga S, Ohtomo Y, Shimizu T. The synergistic effect of mizoribine and a direct renin inhibitor, aliskiren, on unilateral ureteral obstruction induced renal fibrosis in rats. J Urol. 2014;191:1139–46. doi: 10.1016/j.juro.2013.10.053. [DOI] [PubMed] [Google Scholar]
- 16.Wang W, Luo R, Lin Y, Wang F, Zheng P, Levi M, Yang T, Li C. Aliskiren restores renal AQP2 expression during unilateral ureteral obstruction by inhibiting the inflammasome. Am J Physiol Renal Physiol. 2015;308:F910–22. doi: 10.1152/ajprenal.00649.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wu WP, Chang CH, Chiu YT, Ku CL, Wen MC, Shu KH, Wu MJ. A reduction of unilateral ureteral obstruction-induced renal fibrosis by a therapy combining valsartan with aliskiren. Am J Physiol Renal Physiol. 2010;299:F929–41. doi: 10.1152/ajprenal.00192.2010. [DOI] [PubMed] [Google Scholar]
- 18.Zhou X, Fukuda N, Matsuda H, Endo M, Wang X, Saito K, Ueno T, Matsumoto T, Matsumoto K, Soma M, Kobayashi N, Nishiyama A. Complement 3 activates the renal renin-angiotensin system by induction of epithelial-to-mesenchymal transition of the nephrotubulus in mice. Am J Physiol Renal Physiol. 2013;305:F957–67. doi: 10.1152/ajprenal.00344.2013. [DOI] [PubMed] [Google Scholar]
- 19.Hammad FT, Lubbad L. The effect of diclofenac sodium on renal function in reversible unilateral ureteric obstruction. Urol Res. 2011;39:351–6. doi: 10.1007/s00240-010-0348-4. [DOI] [PubMed] [Google Scholar]
- 20.Hammad FT, Wheatley AM, Davis G. Long-term renal effects of unilateral ureteral obstruction and the role of endothelin. Kidney Int. 2000;58:242–50. doi: 10.1046/j.1523-1755.2000.00159.x. [DOI] [PubMed] [Google Scholar]
- 21.el-Dahr SS, Gee J, Dipp S, Hanss BG, Vari RC, Chao J. Upregulation of renin-angiotensin system and downregulation of kallikrein in obstructive nephropathy. Am J Physiol. 1993;264:F874–81. doi: 10.1152/ajprenal.1993.264.5.F874. [DOI] [PubMed] [Google Scholar]
- 22.Jensen AM, Li C, Praetorius HA, Nørregaard R, Frische S, Knepper MA, Nielsen S, Frøkiaer J. Angiotensin II mediates downregulation of aquaporin water channels and key renal sodium transporters in response to urinary tract obstruction. Am J Physiol Renal Physiol. 2006;291:F1021–32. doi: 10.1152/ajprenal.00387.2005. [DOI] [PubMed] [Google Scholar]
- 23.Choi YJ, Mendoza L, Rha SJ, Sheikh-Hamad D, Baranowska-Daca E, Nguyen V, Smith CW, Nassar G, Suki WN, Truong LD. Role of p53-dependent activation of caspases in chronic obstructive uropathy: evidence from p53 null mutant mice. J Am Soc Nephrol. 2001;12:983–92. doi: 10.1681/ASN.V125983. [DOI] [PubMed] [Google Scholar]
- 24.Ishidoya S, Morrissey J, McCracken R, Reyes A, Klahr S. Angiotensin II receptor antagonist ameliorates renal tubulointerstitial fibrosis caused by unilateral ureteral obstruction. Kidney Int. 1995;47:1285–94. doi: 10.1038/ki.1995.183. [DOI] [PubMed] [Google Scholar]
- 25.Edwards RM. Segmental effects of norepinephrine and angiotensin II on isolated renal microvessels. Am J Physiol. 1983;244:F526–34. doi: 10.1152/ajprenal.1983.244.5.F526. [DOI] [PubMed] [Google Scholar]
- 26.Topcu SO, Pedersen M, Nørregaard R, Wang G, Knepper M, Djurhuus JC, Nielsen S, Jørgensen TM, Frøkiaer J. Candesartan prevents long-term impairment of renal function in response to neonatal partial unilateral ureteral obstruction. Am J Physiol Renal Physiol. 2007;292:F736–48. doi: 10.1152/ajprenal.00241.2006. [DOI] [PubMed] [Google Scholar]
- 27.Wood JM, Stanton JL, Hofbauer KG. Inhibitors of renin as potential therapeutic agents. J Enzyme Inhib. 1987;1:169–85. doi: 10.3109/14756368709020115. [DOI] [PubMed] [Google Scholar]
- 28.Lim AI, Tang SC, Lai KN, Leung JC. Kidney injury molecule-1: more than just an injury marker of tubular epithelial cells? J Cell Physiol. 2013;228:917–24. doi: 10.1002/jcp.24267. [DOI] [PubMed] [Google Scholar]
- 29.Mishra J, Ma Q, Prada A, Mitsnefes M, Zahedi K, Yang J, Barasch J, Devarajan P. Identification of neutrophil gelatinase-associated lipocalin as a novel early urinary biomarker for ischemic renal injury. J Am Soc Nephrol. 2003;14:2534–43. doi: 10.1097/01.asn.0000088027.54400.c6. [DOI] [PubMed] [Google Scholar]
- 30.Morrissey JJ, Ishidoya S, McCracken R, Klahr S. Control of p53 and p21 (WAF1) expression during unilateral ureteral obstruction. Kidney Int Suppl. 1996;57:S84–92. [PubMed] [Google Scholar]


