Abstract
A cohort of patients operated at the National Cancer Center Hospital in Tokyo for rectal carcinoma, at or below the peritoneal reflection, was reviewed retrospectively. The purpose was to study the risk factors for local relapse and the patterns of local recurrence. Three hundred fifty-one patients operated between 1993 and 2002 for rectal carcinoma, at or below the peritoneal reflection, were analyzed. One hundred forty-five patients, with preoperatively staged T1 or T2 tumors without suspected lymph nodes, underwent total mesorectal excision (TME). Lateral lymph node dissection (LLND) was performed in suspected T3 or T4 disease, or when positive lymph nodes were seen; 73 patients received unilateral LLND and 133 patients received bilateral LLND. Of the 351 patients 6.6% developed local recurrence after 5 years. TME only resulted in 0.8% 5-year local recurrence. In lymph-node-positive patients, 33% of the unilateral LLND group had local relapse, significantly more (p = 0.04) than in the bilateral LLND group with 14% local recurrence. Local recurrence in the lateral, presacral, perineal, and anastomotic subsites was lower in the bilateral LLND group as compared with in the unilateral LLND group. We conclude that, in selected patients, surgery without LLND has a very low local recurrence rate. Bilateral LLND is more effective in reducing the chance of local recurrence than unilateral LLND. Either surgical approach, with or without LLND, requires reliable imaging during work-up.
For rectal cancer, surgery is the principal treatment in order to cure. Total mesorectal excision (TME) removes the primary tumor with its surrounding mesorectum as an intact package, preventing residual tumor cells in the mesorectum from developing into local recurrence.1,2 In advanced lesions neoadjuvant (chemo)radiotherapy can downstage tumors, but good surgical quality is still essential in order to achieve total clearance of tumor cells.3
The Japanese concept of surgical treatment of rectal cancer has evolved from anatomical studies in which three lymphatic flow routes were identified.4,5 The upper route is along the superior rectal artery to the inferior mesenteric artery; the lateral route reaches from the middle rectal artery to the internal iliac and obturator basins; and the downward route extends to the inguinal lymph nodes. The upper and lateral routes were shown to be the main two routes of rectal cancer spread, with the peritoneal reflection as the limitation between the two lymphatic areas.6 Consequently, lateral lymph node dissection (LLND) was developed in Japan in order to resect the tumor with the primary locoregional lymph node basins beyond the mesorectal plane.7 LLND has resulted in better survival and lower recurrence rates than conventional surgery.8,9
A problem is that the lateral lymph node routes are anatomically close to the pelvic autonomic nerve plexus, requiring challenging surgery to preserve these during LLND.10 In order to prevent damage to autonomic nerves, nowadays case-oriented policy is practised in Japan, adopting LLND only in advanced disease at or below the peritoneal reflection.
The aim of this study is to evaluate the treatment of rectal cancer between 1993 and 2002 at the National Cancer Center Hospital (NCCH), looking at patterns of local recurrence and the risk factors for local recurrence.
Patients and Methods
Patients
From 1993 to 2002, 923 patients were operated for confirmed primary adenocarcinoma of the rectum at the National Cancer Center Hospital (NCCH) in Tokyo. Surgery was performed according to the guidelines of the Japanese Research Society for Cancer of the Colon and Rectum.11,12 The rectum was defined as located below the lower border of the second sacral vertebra. The peritoneal reflection is the most important landmark in defining the location of the tumor, and low rectal carcinoma is defined as a tumor of which the major part is located at or below the reflection.13
For this analysis the following patients were excluded: metastasis at the time of surgery (n = 134) and in situ carcinoma (n = 22). Of the remaining 767 patients, only patients with rectal carcinoma at or below the peritoneal refection were selected, resulting in 360 patients.
Neoadjuvant chemotherapy was given to some patients with suspicion of stage T4 disease (n = 3) in other hospitals, before referral to the NCCH. Neoadjuvant radiotherapy was not routinely given, so no patients received preoperative radiotherapy. Sometimes in the case of positive lymph nodes, adjuvant radiotherapy (n = 5) or chemoradiotherapy (n = 1) was given. The nine patients who received neoadjuvant chemotherapy and adjuvant (chemo)radiation were excluded, leaving 351 patients for analysis.
Methods
Until 2002 preoperative evaluation at the NCCH consisted of computed tomography (CT) imaging and endoscopic ultrasonography for all patients. Based on preoperative imaging and intraoperative findings, standard total mesorectal excision (TME) was performed in T1 or T2 stage disease without suspected lymph nodes. Lateral lymph node dissection (LLND) was added to TME in stage T3 or T4 rectal cancer at or below the peritoneal reflection, or when positive mesorectal lymph nodes were suspected. Unilateral LLND was performed when the tumor was located lateral in the low rectum, bilateral LLND when the tumor was located centrally. When the lateral lymph nodes were 1 cm or larger on preoperative imaging or intraoperative findings, bilateral extended lymph node dissection was performed, consisting of dissection of the complete internal iliac artery and the autonomic nerve system. When there was no suspicion on positive lateral lymph nodes, autonomic nerve preservation (ANP) was carried out.
Accurate documentation of lymph node status and localization is obtained because all lymph nodes are harvested and recorded from the fresh specimen. The definition of mesorectal lymph nodes is pararectal location or in the direction of the mesentery. Lateral lymph nodes are located along the iliac or obturator arteries.
Follow-up of all patients consisted of thorax, abdominal, and pelvic CT imaging every 6 months. Median follow-up of patients alive was 7.9 years.
All patients who developed local recurrence, defined as any recurrence of rectal cancer in the lesser pelvis, were identified. Local recurrence was diagnosed clinically, radiologically or histologically.
For all locally recurrent patients the available preoperative images and the images at the time of discovery of the local recurrence were retrieved. A specialized oncologic radiologist (R.G.H.B.-T.) reviewed the images. Examining the images, the site of the local recurrence was determined. The sites were classified into the following regions: lateral, presacral, perineal, anterior or anastomotic. The same borders for the respective sites were used as defined by Roels et al.14 When no images were available, the location of recurrence was classified using the radiology reports and clinical data. In one patient insufficient information was provided to determine the location of recurrence with certainty.
Statistical Analysis
Statistical analysis was performed using the SPSS package (SPSS 12.0 for Windows; SPSS Inc., Chicago, IL) and R version 2.5.1. T-tests and chi-square tests were used to compare individual variables. Survival and cumulative recurrence incidences were estimated using the Kaplan–Meier method. Differences between the groups were assessed using the log-rank test. All p-values were two-sided and considered statistically significant at 0.05 or less. For local recurrence, cumulative incidences were calculated accounting for death as competing risk.15 Similarly, cumulative incidences were calculated for subsite of local recurrence, with death and other types of local recurrence as competing risks, and for cancer-specific survival, with death due to other causes as competing risk. Multivariate analyses of local recurrence and overall survival were performed by first testing the effect of covariates in a univariate Cox regression. Covariates with trend-significant effects (p-value < 0.10) were then selected for multivariate Cox regression. The following variables were studied for local recurrence and overall survival: age, sex, operative procedure, degree of lateral lymphadenectomy, T-stage, mesorectal lymph node N-stage, lateral lymph node positivity, maximum tumor diameter, differentiation, and autonomic nerve preservation.
Results
Clinicopathology
Patient characteristics and treatment details are listed in Table 1. Of the 351 studied patients, 145 had standard TME surgery without LLND, 73 underwent unilateral LLND, and 133 patients received bilateral LLND. LLND was performed in significantly younger patients and more often in combination with a non-sphincter-saving procedure, compared with patients who had not undergone an LLND. The tumors in the LLND patients had higher T- and N-stages and were significantly larger. Comparing the clinicopathological characteristics between the unilateral and the bilateral LLND, no significant differences were found, except that unilateral LLND was more often combined with autonomic nerve preservation (ANP).
Table 1.
Clinicopathological characteristics
| No LLND (n = 145) | Unilateral LLND (n = 73) | Bilateral LLND (n = 133) | p* | p** | |
|---|---|---|---|---|---|
| Sex ratio (M:F) | 96:49 (66:34) | 47:26 (64:36) | 86:47 (65:35) | 0.95 | 0.97 |
| Mean age (years) | 61 | 57 | 57 | 0.03 | 0.98 |
| Operation | |||||
| Sphincter-saving | 112 (77) | 36 (49) | 63 (47) | ||
| Not sphincter-saving | 33 (23) | 37 (51) | 70 (53) | <0.001 | 0.79 |
| Adjuvant chemotherapy | |||||
| No | 139 (96) | 67 (92) | 121 (91) | ||
| Yes | 6 (4) | 6 (8) | 12 (9) | 0.24 | 0.85 |
| T-stage | |||||
| T1 | 52 (36) | 3 (4) | 3 (2) | ||
| T2 | 47 (32) | 27 (37) | 37 (28) | ||
| T3 | 46 (32) | 40 (55) | 83 (62) | ||
| T4 | 0 (0) | 3 (4) | 10 (8) | <0.001 | 0.37 |
| Meso LN positive | |||||
| 0 | 102 (70) | 44 (60) | 64 (48) | ||
| 1–3 | 30 (21) | 19 (26) | 39 (29) | ||
| >4 | 13 (9) | 10 (14) | 30 (23) | 0.003 | 0.28 |
| Lat LN positive | |||||
| No | – | 62 (85) | 109 (82) | ||
| Yes | – | 11 (15) | 24 (18) | – | 0.59 |
| ANP | |||||
| No | 3 (2) | 2 (3) | 17 (13) | ||
| Yes | 142 (98) | 71 (97) | 116 (87) | <0.001 | 0.02 |
| Differentiation | |||||
| Well | 75 (52) | 27 (37) | 50 (38) | ||
| Moderate | 67 (46) | 44 (60) | 75 (56) | ||
| Poor | 2 (2) | 2 (3) | 8 (6) | 0.18 | 0.29 |
| Tumor size | |||||
| 0–4 cm | 106 (73) | 31 (42) | 42 (32) | ||
| >4 cm | 39 (27) | 42 (58) | 91 (68) | <0.001 | 0.12 |
| Diss. LN (mean) | 21 | 38 | 45 | <0.001 | 0.004 |
Values in parentheses are percentages
* p value between no LLND, unilateral LLND, and bilateral LLND
** p value between unilateral LLND and bilateral LLND
Meso mesorectal; Lat lateral; LN lymph node; ANP autonomic nerve preservation
Mean lymph node harvest was 21 LNs in standard TME (Table 1). After unilateral LLND the mean number of recovered LNs was 38, and after bilateral LLND this was 45 (p = 0.004).
Table 2 shows the outcomes of lymph node involvement for all 351 patients, stratified by T-stage. Overall lymph node involvement was 42%, and lateral lymph node involvement was 10%. Jump metastases (mesorectal lymph nodes negative and lateral lymph nodes positive) occurred in 3% (7/207) of the patients with LLND.
Table 2.
Lateral lymph node dissection and lymph node status, stratified by T-stage
| Stage | LLND | LNI | LNI | LLNI | ||
|---|---|---|---|---|---|---|
| T1: 58 | No LLND | 52 (90%) | N0 | 47 | 8/58 = 14% | 1/58 = 2% |
| Upper pos | 5 | |||||
| LLND | 6 (10%) | N0 | 3 | |||
| Upper pos, lat neg | 2 | |||||
| Upper neg, lat pos | 0 | |||||
| Upper pos, lat pos | 1 | |||||
| T2: 111 | No LLND | 47 (42%) | N0 | 33 | 32/111 = 29% | 7/111 = 6% |
| Upper pos | 14 | |||||
| LLND | 64 (58%) | N0 | 46 | |||
| Upper pos, lat neg | 11 | |||||
| Upper neg, lat pos | 2 | |||||
| Upper pos, lat pos | 5 | |||||
| T3: 169 | No LLND | 46 (27%) | N0 | 22 | 97/169 = 57% | 19/169 = 11% |
| Upper pos | 24 | |||||
| LLND | 123 (73%) | N0 | 50 | |||
| Upper pos, lat neg | 54 | |||||
| Upper neg, lat pos | 5 | |||||
| Upper pos, lat pos | 14 | |||||
| T4: 14 | No LLND | 0 (0%) | N0 | – | 12/14 = 86% | 8/14 = 57% |
| Upper pos | – | |||||
| LLND | 14 (100%) | N0 | 1 | |||
| Upper pos, lat neg | 4 | |||||
| Upper neg, lat pos | 0 | |||||
| Upper pos, lat pos | 8 | |||||
| Total: 351 | 207/351 = 59%* | 149/351 = 42% | 35/351 = 10% | |||
LLND lateral lymph node dissection; LNI lymph node involvement (upper and lateral lymph nodes); LLNI lateral lymph node involvement; Upper, upper lymph nodes; Lat lateral lymph nodes; pos positive; neg negative
* Percentage of patients submitted to LLND
Local Recurrence
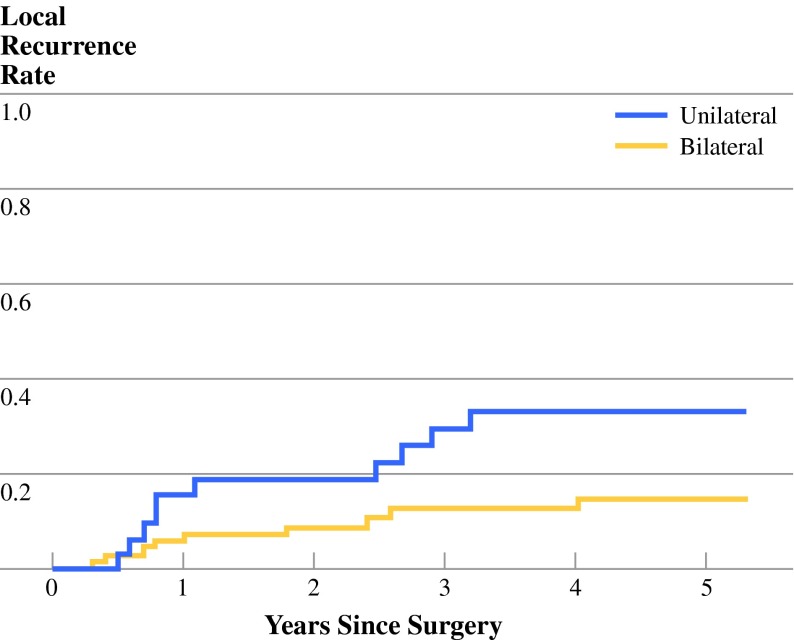
At time of last follow-up 23 of the total of 351 patients had developed local recurrence (6.6% 5-year local recurrence rate). In the patients who had not undergone LLND, only one patient (0.8%) had local recurrence at the site of the anastomosis. In the unilateral LLND group, 12 of the 73 patients (5-year 15.4%) had local relapse. This was more than in the bilateral LLND group, with 10 of 133 local recurrences (5-year 8.3%). In N+ patients (Fig. 1), the difference between the uni- and bilateral LLND (32.8% versus 14.2%, respectively) was significant (p = 0.04).
Fig. 1.
Local recurrence in N+ patients
In multivariate analysis (Table 3) including uni- and bilateral LLND patients, lateral lymphadenectomy, mesorectal lymph node N-stage, and lateral lymph node positivity were independent risk factors for local recurrence. Compared with patients with bilateral LLND the relative risk for local recurrence was 4.0 for unilateral LLND patients.
Table 3.
Multivariate analysis for local recurrence
| Variable | HR | 95% CI | p |
|---|---|---|---|
| Lateral dissection | 0.003 | ||
| Unilateral | 1.00 | ||
| Bilateral | 0.25 | 0.10–0.64 | |
| T-stage | 0.09 | ||
| T1 + T2 | 1.00 | ||
| T3 + T4 | 2.99 | 0.84–10.73 | |
| N-stage mesorectal LN | 0.008 | ||
| 0 pos | 1.00 | ||
| 1–3 pos | 2.71 | 0.75–9.85 | |
| > 4 pos | 7.22 | 2.01–25.94 | |
| Lateral LN status | 0.007 | ||
| Negative | 1.00 | ||
| Positive | 3.53 | 1.41–8.85 |
Table 4 reports the sites of the local recurrences for the uni- and bilateral LLND groups. The rate of lateral recurrence in the unilateral LLND patients was 5.6%, and in the bilateral LLND patients was 3.3%. It was noticed that the three patients who developed lateral local recurrence on the ipsilateral side after unilateral LLND had lower lymph node harvest (mean 28 LNs) than the patients who developed no lateral recurrence after unilateral LLND (mean 38 LNs). However, the number of patients is too low to draw any firm conclusion from this finding.
Table 4.
Sites of local recurrence
| All patients | Only N+ patients | |||||
|---|---|---|---|---|---|---|
| Site of local recurrence | Unilateral LLND (n = 73) | Bilateral LLND (n = 133) | p | Unilateral LLND (n = 32) | Bilateral LLND (n = 74) | p |
| Lateral | 5 (5.6) | 4 (3.3) | 4 (13.2) | 3 (4.6) | ||
| Ipsilateral | 3 (3.4) | 3 (9.9) | ||||
| Contralateral | 2 (2.2) | 1 (3.3) | ||||
| Presacral | 2 (2.8) | 0 (0) | 2 (6.7) | 0 (0) | ||
| Perineal | 2 (2.8) | 2 (1.7) | 1 (3.1) | 2 (3.4) | ||
| Anterior | 0 (0) | 1 (0.9) | 0 (0) | 1 (1.8) | ||
| Anastomotic | 3 (4.2) | 2 (1.6) | 3 (9.8) | 2 (3.0) | ||
| Unknown | 0 (0) | 1 (0.8) | 0 (0) | 1 (1.4) | ||
| Total | 12 | 10 | 10 | 9 | ||
| 5-Year LR rate | 15.4% | 8.3% | 0.06 | 32.8% | 14.2% | 0.04 |
Values in parentheses are the 5-year local recurrence rates per subsite
Distant Recurrence and Survival
At local recurrence diagnosis 40% of the unilateral LLND patients and 60% of the bilateral LLND patients had distant metastases. One year after local recurrence diagnoses these figures were 70% and 80% in the uni- and bilateral LLND patients, respectively.
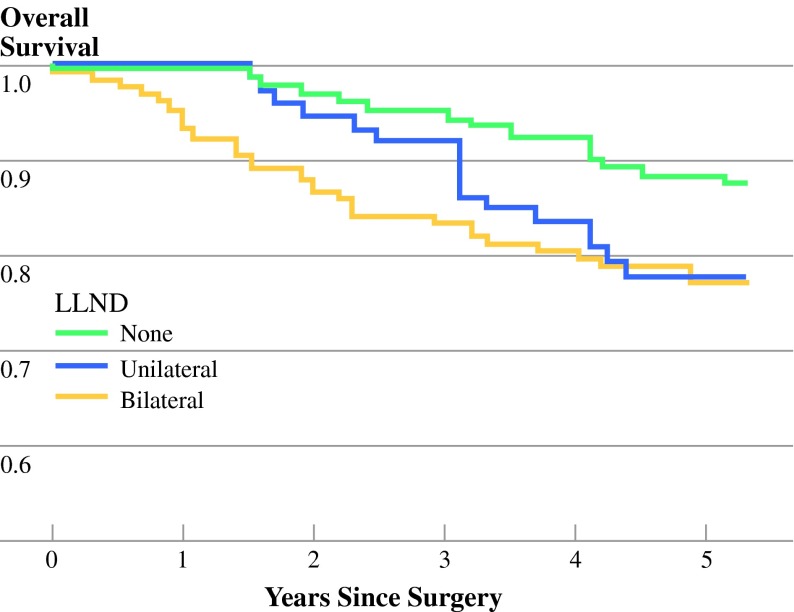
Figure 2 shows the survival curves of the TME-only, and uni- and bilateral LLND patients. Overall 5-year survival was 89% for patients who had standard TME. Five-year overall survival in the unilateral LLND group was 78%, which did not differ significantly from the bilateral LLND group (77%) (p = 0.37).
Fig. 2.
Overall survival in all patients
The multivariate Cox regression analysis, when including the uni- and bilateral LLND groups, identified T-stage, mesorectal lymph node N-stage and lateral lymph node positivity as independent factors for death risk.
Two years after local recurrence diagnosis 37% of the unilateral LLND patients was still alive, as compared with 60% of the bilateral LLND patients. The number of patients is however too low to conclude significant better survival for bilateral LLND patients.
Discussion
Lateral lymph node dissection (LLND) was introduced in Japan in the 1970s and results in good survival and low local recurrence rates.7–9 Since approximately 1984 several forms of nerve-sparing techniques, combined with LLND, have been developed. Bilateral and even unilateral complete autonomic nerve preservation (ANP) combined with LLND often maintains urinary function, but reports vary about the results in sexual function.16–20 In the many decades of LLND surgery in Japan constant evaluation has taken place with the purpose of preventing overtreatment and minimizing morbidity.21 Nowadays the policy in many Japanese hospitals is highly case-oriented, adapting the degree of surgical resection and ANP to the extent of cancer spread.22 Whereas in the 1970s and 1980s in the National Cancer Center Hospital (NCCH) in Tokyo the standard procedure was to perform bilateral LLND in case of advanced rectal cancer, lately also unilateral LLND has been performed. The purpose of this study was to evaluate the treatment between 1993 and 2002 at the National Cancer Center Hospital for rectal carcinoma, at or below the peritoneal reflection, looking at the patterns of local recurrence and the risk factors for local recurrence. To our knowledge, there are no published results of unilateral lymph node dissection in rectal carcinoma.
The results of this study show 5-year local recurrence rate of 6.6% in rectal cancer at or below the peritoneal reflection by Japanese surgery. This primarily surgical approach compares favorably with results in Western countries, where neoadjuvant treatment is adopted as the standard in order to reduce local recurrence rates. Therefore, the Japanese concept of removing the lateral basins of lymph nodes spread can be considered successful. However, some questions still remain to be answered. The etiology of locally recurrent disease is not completely understood yet.
This study, although retrospective, provides further evidence of disease outside the TME envelope in higher-stage tumors. Bilateral LLND (5-year local recurrence rate 14%) resulted in better local control than unilateral LLND (5-year LR rate 33%) in N+ patients. Persistent disease in lateral lymph nodes that is left behind may account for some of the local recurrences, as would occur in standard TME surgery. However in that case, it would be expected that most of the recurrences would occur originating in this lateral basin. In this study we noted that only a part of the local recurrences was present in the lateral side walls. Most of the recurrences could not be explained by the anatomical position of the lateral lymph nodes. One can only speculate about other mechanisms of how tumor cells seed into the surgical resection volume. Maybe removal of the lateral lymph nodes also removes (microscopic) tumor cells which are in transit in the lateral lymph flow route, which could otherwise leak back into the surgical wound. This would explain why unilateral dissection is inferior to bilateral dissection, having more local recurrence in also the presacral, perineal, and anastomotic subsite, not only the lateral.
The rationale behind the unilateral LLND is that the contralateral autonomic nervous system stays untouched, decreasing the chance of autonomic nerve injury. Studies report that, after LLND with nerve-sparing surgery, urinary function is maintained. Between 50% and 100% of males are sexually active, however with compromised ejaculation.16,18,19,23 This is ascribed to traction and injury to nerves during the mobilization and electrocautery required for LLND.18 Unfortunately we have no data on urinary and sexual function of this cohort, being unable to report on the results after unilateral LLND with nerve preservation. Therefore, the question of whether functional results are truly better remains unanswered.
The tumors of the patients who had TME without LLND were smaller and less advanced compared with those of LLND patients. This better staging is reflected in better survival. That only one patient who had standard TME surgery had local relapse (5-year local recurrence 0.8%) is striking. The selection for low-risk disease by pre- and intraoperative evaluation has obviously been accurate. Interesting however, is that pathology (Tables 1 and 2) showed that about 30% of the patients operated by TME had T3-stage or N-positive disease. Pathology seems to filter out more metastatic lymph nodes than preoperative imaging, but these (micro)metastases obviously have no oncologic consequences. Jump metastases (mesorectal negative, lateral positive) occurred in only 3% of the LLND patients, thus when mesorectal lymph nodes are unsuspected, risk for lateral lymph node recurrence is very low.
Preoperative evaluation in advanced disease is difficult. In this study local recurrence developed on the contralateral side after unilateral lymph node dissection, while these contralateral lymph node metastases were not suspicious on preoperative CT imaging. Meta-analysis report that assessment of lymph node status by CT is unreliable for clinical decision making, because the radiologist can only look at lymph node size.24,25 Since 2002 in the NCCH magnetic resonance imaging (MRI) has been used, which is reported to be superior to CT because it can rely on additional morphological criteria, such as signal intensity and border contour.26–28 Furthermore, lymph-node-specific contrast agents or molecular imaging might play a role in detecting micrometastases in the near future.29
In the West, (chemo)radiation is used instead of LLND. There are no (randomized) studies comparing preoperative (chemo)radiotherapy and TME with LLND in similar patients, making it difficult to make a statement about which regimen is preferred in advanced rectal carcinoma. Western surgeons are hesitant to do lateral lymph node dissections for three reasons. First, in Western patients with a higher body mass index, nerve-sparing techniques are more difficult and the fear of excess morbidity is realistic. Further, it is well known that lateral lymph node status is reflective of overall mesenteric lymph node status and lateral lymph node positivity results in poor prognosis.13,30 Lastly, although LLND has improved oncologic results in Japanese patients in historical studies and also the current study suggests that LLND is able to prevent residual tumor cells from developing into local recurrence, the clinical effectiveness of LLND has not been proved in a randomized fashion. Currently, the National Cancer Center Hospital is coordinating a multicenter randomized clinical trial comparing conventional TME with bilateral LLND in patients with rectal carcinoma. The results are awaited with anticipation, but it is questionable whether they will be applicable to Western patients.
Concluding, in this study patterns of local recurrence were evaluated in the treatment of rectal cancer, at or below the peritoneal reflection, with selective LLND. Overall local recurrence was 6.6% at 5 years. Local recurrence rate after standard TME was 0.8% in low-stage disease. In lymph-node-positive patients, 33% of the unilateral LLND patients had local relapse, significantly more than in the bilateral LLND group with 14% local recurrence. Either surgical approach, with or without LLND, requires reliable imaging during work-up.
Acknowledgements
The authors would like to express their gratitude to the Japan Prizewinners Program of the Dutch Government who financed the stay of M. Kusters in Tokyo and the Japan–Netherlands Institute (Executive Director Dr. W. G. J. Remmelink) for practical support.
The authors would also like to thank Drs. S. Ishiguro and Y. Kobayashi, from the Colorectal Surgery Department of the National Cancer Center Hospital, for help with obtaining data; as well as Drs. Y. Arai and M. Takahashi from the Department of Radiology, National Cancer Center Hospital, for assistance with imaging.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Footnotes
An erratum to this article is available at http://dx.doi.org/10.1245/s10434-009-0320-0.
References
- 1.Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet. 1986;1:1479–82. doi: 10.1016/S0140-6736(86)91510-2. [DOI] [PubMed] [Google Scholar]
- 2.Quirke P, Durdey P, Dixon MF, Williams NS. Local recurrence of rectal adenocarcinoma due to inadequate surgical resection. Histopathological study of lateral tumour spread and surgical excision. Lancet. 1986;2:996–9. doi: 10.1016/S0140-6736(86)92612-7. [DOI] [PubMed] [Google Scholar]
- 3.Den Dulk M, Collette L, van d, V, Marijnen CA, Calais G, Mineur L, et al. Quality of surgery in T3–4 rectal cancer: involvement of circumferential resection margin not influenced by preoperative treatment. Results from EORTC trial 22921. Eur J Cancer. 2007;43:1821–8. [DOI] [PubMed]
- 4.Senba Y. An anatomical study of the lymphatic system of the rectum. In Japanese. J Fukuoka Med Coll. 1927;20:1213–68. [Google Scholar]
- 5.Gerota D. Die lymphgefasse des rectums und des anus. Arch Anat Physiol. 1895;240.
- 6.Kuru M. Cancer of the rectum. In Japanese. J Jpn Surg Soc. 1940;41:832–77. [Google Scholar]
- 7.Hojo K, Sawada T, Moriya Y. An analysis of survival and voiding, sexual function after wide iliopelvic lymphadenectomy in patients with carcinoma of the rectum, compared with conventional lymphadenectomy. Dis Colon Rectum. 1989;32:128–33. doi: 10.1007/BF02553825. [DOI] [PubMed] [Google Scholar]
- 8.Moriya Y, Hojo K, Sawada T, Koyama Y. Significance of lateral node dissection for advanced rectal carcinoma at or below the peritoneal reflection. Dis Colon Rectum. 1989;32:307–15. doi: 10.1007/BF02553486. [DOI] [PubMed] [Google Scholar]
- 9.Suzuki K, Muto T, Sawada T. Prevention of local recurrence by extended lymphadenectomy for rectal cancer. Surg Today. 1995;25:795–801. doi: 10.1007/BF00311455. [DOI] [PubMed] [Google Scholar]
- 10.Moriya Y, Sugihara K, Akasu T, Fujita S. Patterns of recurrence after nerve-sparing surgery for rectal adenocarcinoma with special reference to loco-regional recurrence. Dis Colon Rectum. 1995;38:1162–8. doi: 10.1007/BF02048331. [DOI] [PubMed] [Google Scholar]
- 11.General rules for clinical and pathological studies on cancer of the colon, rectum and anus. Part I. Clinical classification. Japanese Research Society for Cancer of the Colon and Rectum. Jpn J Surg. 1983;13:557–73. [DOI] [PubMed]
- 12.General rules for clinical and pathological studies on cancer of the colon, rectum and anus, 7th ed. Japanese Research Society for Cancer of the Colon and Rectum; 2006.
- 13.Steup WH, Moriya Y, van de Velde CJ. Patterns of lymphatic spread in rectal cancer. A topographical analysis on lymph node metastases. Eur J Cancer. 2002;38:911–8. doi: 10.1016/S0959-8049(02)00046-1. [DOI] [PubMed] [Google Scholar]
- 14.Roels S, Duthoy W, Haustermans K, Penninckx F, Vandecaveye V, Boterberg T, et al. Definition and delineation of the clinical target volume for rectal cancer. Int J Radiat Oncol Biol Phys. 2006;65:1129–42. doi: 10.1016/j.ijrobp.2006.02.050. [DOI] [PubMed] [Google Scholar]
- 15.Putter H, Fiocco M, Geskus RB. Tutorial in biostatistics: competing risks and multi-state models. Stat Med. 2007;26:2389–430. doi: 10.1002/sim.2712. [DOI] [PubMed] [Google Scholar]
- 16.Mori T, Takahashi K, Yasuno M. Radical resection with autonomic nerve preservation and lymph node dissection techniques in lower rectal cancer surgery and its results: the impact of lateral lymph node dissection. Langenbecks Arch Surg. 1998;383:409–15. doi: 10.1007/s004230050153. [DOI] [PubMed] [Google Scholar]
- 17.Hojo K, Vernava AM, III, Sugihara K, Katumata K. Preservation of urine voiding and sexual function after rectal cancer surgery. Dis Colon Rectum. 1991;34:532–9. doi: 10.1007/BF02049890. [DOI] [PubMed] [Google Scholar]
- 18.Kyo K, Sameshima S, Takahashi M, Furugori T, Sawada T. Impact of autonomic nerve preservation and lateral node dissection on male urogenital function after total mesorectal excision for lower rectal cancer. World J Surg. 2006;30:1014–9. doi: 10.1007/s00268-005-0050-9. [DOI] [PubMed] [Google Scholar]
- 19.Maeda K, Maruta M, Utsumi T, Sato H, Toyama K, Matsuoka H. Bladder and male sexual functions after autonomic nerve-sparing TME with or without lateral node dissection for rectal cancer. Tech Coloproctol. 2003;7:29–33. doi: 10.1007/s101510300005. [DOI] [PubMed] [Google Scholar]
- 20.Maas CP, Moriya Y, Steup WH, Kiebert GM, Kranenbarg WM, van de Velde CJ. Radical and nerve-preserving surgery for rectal cancer in The Netherlands: a prospective study on morbidity and functional outcome. Br J Surg. 1998;85:92–7. doi: 10.1046/j.1365-2168.1998.00530.x. [DOI] [PubMed] [Google Scholar]
- 21.Yano H, Moran BJ. The incidence of lateral pelvic side-wall nodal involvement in low rectal cancer may be similar in Japan and the West. Br J Surg. 2008;95:33–49. doi: 10.1002/bjs.6061. [DOI] [PubMed] [Google Scholar]
- 22.Moriya Y, Sugihara K, Akasu T, Fujita S. Importance of extended lymphadenectomy with lateral node dissection for advanced lower rectal cancer. World J Surg. 1997;21:728–32. doi: 10.1007/s002689900298. [DOI] [PubMed] [Google Scholar]
- 23.Moriya Y, Sugihara K, Akasu T, Fujita S. Nerve-sparing surgery with lateral node dissection for advanced lower rectal cancer. Eur J Cancer. 1995;31A:1229–32. doi: 10.1016/0959-8049(95)00164-E. [DOI] [PubMed] [Google Scholar]
- 24.Bipat S, Glas AS, Slors FJ, Zwinderman AH, Bossuyt PM, Stoker J. Rectal cancer: local staging and assessment of lymph node involvement with endoluminal US, CT, and MR imaging–a meta-analysis. Radiology. 2004;232:773–83. doi: 10.1148/radiol.2323031368. [DOI] [PubMed] [Google Scholar]
- 25.Lahaye MJ, Engelen SM, Nelemans PJ, Beets GL, van de Velde CJ, van Engelshoven JM, et al. Imaging for predicting the risk factors–the circumferential resection margin and nodal disease–of local recurrence in rectal cancer: a meta-analysis. Semin Ultrasound CT MR. 2005;26:259–68. doi: 10.1053/j.sult.2005.04.005. [DOI] [PubMed] [Google Scholar]
- 26.Arii K, Takifuji K, Yokoyama S, Matsuda K, Higashiguchi T, Tominaga T, et al. Preoperative evaluation of pelvic lateral lymph node of patients with lower rectal cancer: comparison study of MR imaging and CT in 53 patients. Langenbecks Arch Surg. 2006;391:449–54. doi: 10.1007/s00423-006-0066-0. [DOI] [PubMed] [Google Scholar]
- 27.Brown G. Thin section MRI in multidisciplinary pre-operative decision making for patients with rectal cancer. Br J Radiol. 2005;78 Spec no 2:S117–27. doi: 10.1259/bjr/15128198. [DOI] [PubMed] [Google Scholar]
- 28.Kim JH, Beets GL, Kim MJ, Kessels AG, Beets-Tan RG. High-resolution MR imaging for nodal staging in rectal cancer: are there any criteria in addition to the size? Eur J Radiol. 2004;52:78–83. doi: 10.1016/j.ejrad.2003.12.005. [DOI] [PubMed] [Google Scholar]
- 29.Lahaye MJ, Engelen SM, Kessels AG, de Bruine AP, von Meyenfeldt MF, van Engelshoven JM, et al. USPIO-enhanced MR imaging for nodal staging in patients with primary rectal cancer: predictive criteria. Radiology. 2008;246:804–11. doi: 10.1148/radiol.2463070221. [DOI] [PubMed] [Google Scholar]
- 30.Ueno M, Oya M, Azekura K, Yamaguchi T, Muto T. Incidence and prognostic significance of lateral lymph node metastasis in patients with advanced low rectal cancer. Br J Surg. 2005;92:756–63. doi: 10.1002/bjs.4975. [DOI] [PubMed] [Google Scholar]