Introduction
Etanercept, adalimumab, infliximab, and ustekinumab are all biologic therapies licensed for the treatment of chronic plaque psoriasis in the United Kingdom. Although these therapies have advanced the treatment of psoriasis, they can lose effectiveness with time.1
Three studies looked at drug survival in particular.2, 3, 4 Warren et al2 found that in biologic-naïve patients, negative predictors of drug survival were female sex, being a current smoker, having a higher baseline Dermatology Life Quality Index (DLQI), and taking etanercept or infliximab. Positive predictive factors were having psoriatic arthritis and taking ustekinumab. A model for discontinuation because of ineffectiveness found that one of the predictors for discontinuation is a body mass index greater than 35. Warren et al2 found that survival with biologic therapies decreases over time—77% in the first year, 63% in the second year, and 53% in the third year. Interestingly, ustekinumab had the highest survival rate compared with all the other anti–tumor necrosis factor inhibitors.
Gniadecki et al4 analyzed data derived from the Danish biologics national registry. Etanercept was found to have the shortest drug survival compared with ustekinumab, which had the longest long-term survival. A total of 81.9% of patients on ustekinumab, as a first biologic, still remained on this therapy 4 years later, and when all patients, biologic naïve and nonnaïve, were included, this percentage decreased to 70% at 4 years.3
One possible mechanism in which biologics lose their efficacy is the presence of antidrug antibodies (ADA).5 Biologic therapies include fusion proteins and monoclonal antibodies. Monoclonal antibodies can either be murine, chimeric, or fully human. Monoclonal antibodies generate variable immunogenic responses and ADA depending on their type with the fully humanized being the least immunogenic.6, 7
Methods
We undertook a retrospective survey of all patients who took concomitant immunosuppressive agents while taking ustekinumab for psoriasis from October 2009 to April 2015 in an attempt to improve the biologic drug survival at the first indication of loss of efficacy. A total of 76 patients were treated with ustekinumab during this period, and 7 patients were identified who required additional immunosuppression in the form of methotrexate, fumaric acid esters, azathioprine, hydroxyurea, and acitretin. All 7 patients had chronic plaque psoriasis, and 2 had psoriatic arthritis.
Results
Patients 1 through 7 are detailed in Table I. These patients had concomitant immunosuppressive agents with ustekinumab—2 women and 5 men. Two patients took methotrexate, 1 took fumaric acid esters, 3 took hydroxyurea, and 1 took azathioprine. Three patients were biologic naïve and 4 were non-naïve. The time between the initiation of ustekinumab and the addition of an immunosuppressive agent ranged between 8 months and 20 months, with an average of 14.1 months. The range of time that ustekinumab and the additional immunosuppressive agent controlled the disease ranged between 37 and 68 months. The disease remains well controlled in all patients on the combination to date except patients 6 and 7 who are discussed in detail. After performing a nonpaired 2-tail t test, there was a significant difference with a P value of .002 between the Psoriasis Area and Severity Index (PASI) before and after an immunosuppressive agent was added.
Table I.
Seven patients who required additional immunosuppression while taking ustekinumab
| Sex | Age, y | Body mass index | Previous systemic treatment | Previous biologic treatment | PASI/DLQI before ustekinumab | PASI and DLQI at wk 16 | Duration between start of ustekinumab and additional immunosuppressive agent added, mo | Immunosuppressive agent | PASI before immunosuppressive agent | PASI after immunosuppressive agent | Total survival of ustekinumab in months |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Female | 47 | 36.1 | Methotrexate, mycophenolate mofetil, hydroxyurea, fumaric acid esters, cyclosporine | None | 20/23 | 0/0 | 12 | Hydroxyurea, 500 mg 3 times a day | 6.0 | 3 | 49 |
| Male | 55 | 25.6 | Hydroxyurea, cyclosporine, acitretin, methotrexate | None | 17.2/16 | 3.6/0 | 14 | Methotrexate, 10 mg weekly | 5.2 | 2.1 | 37 |
| Male | 32 | 31.5 | Methotrexate, cyclosporine | Adalimumab | 14.6/11 | 3/5 | 15 | Fumaric acid esters, 30 mg 3 times a day | 5.0 | 2.4 | 30 |
| Female | 64 | 47.5 | Methotrexate | Adalimumab and infliximab | 17.4/10 | 2.9/0 | 20 | Methotrexate, 7.5 mg weekly | 7.6 | 4.2 | 65 |
| Male | 40 | 20.4 | Methotrexate, hydroxyurea, cyclosporine | None | 16.1/13 | 2.4/1 | 18 | Hydroxyurea, 500 mg twice a day | 6.0 | 5 | 68 |
| Male | 47 | 32.2 | Methotrexate, cyclosporine, acitretin, fumaric acid esters | Raptiva, infliximab, etanercept, adalimumab | 20/30 | 10.8/11 | 12 | Hydroxyurea, 500 mg 3 times a day | 9.4 | 4.7 | 47 |
| Male | 65 | 30.1 | Methotrexate, systemic 5 fluorouracil, cyclosporine, hydroxyurea, fumaric acid esters, acitretin | Etanercept, raptiva, infliximab, adalimumab | 22.7/30 | 17.2/9 | 8 | Azathioprine, 150 mg daily | 6.9 | 4.6 | 54 |
Patients 6 and 7 had more complicated outcomes. Patient 6 did not respond to 4 biologic agents; he started ustekinumab in October 2009 with a PASI of 20 and DLQI 30 and initially did well for 12 months on ustekinumab alone, then he started losing disease control with a PASI of 7.6 and DLQI of 17. At this point, methotrexate, 15 mg weekly, was added, which led to a decrease in PASI to 5, but the methotrexate was stopped because of side effects. Hydroxurea, 500 mg twice a day, initially, then 3 times a day, was used in place of methotrexate, and this achieved disease control with a PASI of 4.7. In April 2012, ustekinumab was increased to 90 mg, as the patient's weight increased to more than 100 kg. This increase reduced the PASI from 6.4 to 3.3. In September 2014, disease control was lost, and enrollment in the secukinumab trial was considered. In this patient, the addition of an immunosuppressive agent to ustekinumab achieved 47 months of disease control.
Patient 7 also had no response to 4 biological agents. He started ustekinumab in April 2010 with a PASI of 22 and DLQI of 30 and up until December 2010. His disease was not under control; therefore, fumaric acid esters were added, initially at 120 mg daily increasing to thrice daily. At that time, the PASI decreased from 6.3 to 0.3. In January 2012, fumaric acid esters were stopped because of side effects, and hydroxyurea was added. However side effects developed, and in April 2012 azathioprine, 150 mg daily, was added with a PASI of 6.9 and DLQI of 3. The PASI decreased to 4.6 and the DLQI to 0. He was doing well until June 2015 when his disease flared, and acitretin, 25 mg daily, was added. Recently, the patient had a transient ischemic event, and his disease is flaring. He is now being considered for secukinumab. In this patient, the addition of an immunosuppressive agent to ustekinumab achieved 54 months of disease control.
Discussion
In our population of patients, we found that the addition of an immunosuppressive agent to ustekinumab seemed to restore response. Patients 1 through 6 had a good response at week 16 and subsequently efficacy dipped. At this point, an immunosuppressive agent was added, and efficacy was then recaptured. Because there is evidence that the survival of a biologic depreciates year after year, this combination could potentially prolong the survival of a biologic therapy, thereby delaying the inevitable decrease in efficacy when one is switched from one biologic to another.2 In patient 7, adequate disease control was only achieved by multiple switches between different immunosuppressive agents. The number of switches was significantly higher than for the rest of the patients.
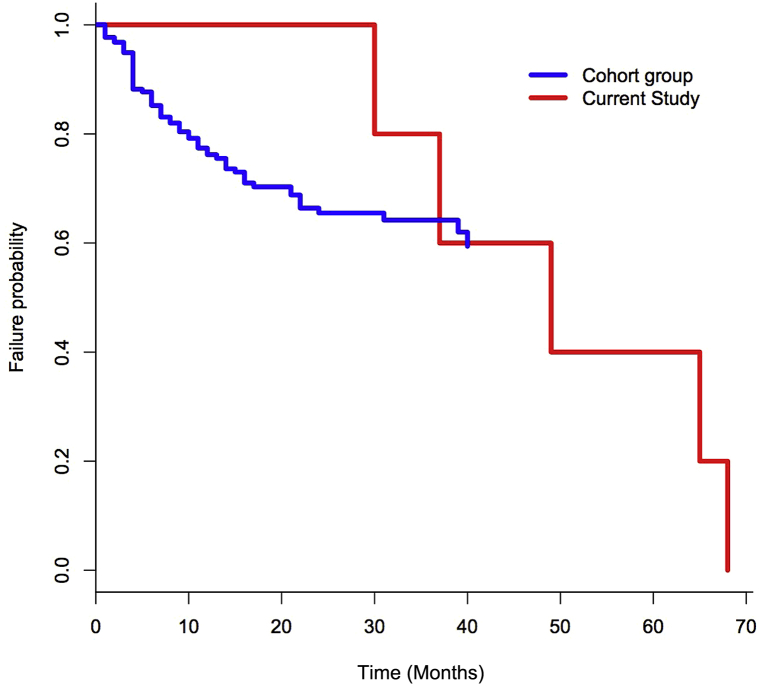
Comparing our data with data from Gniadecki et al4 shows a trend toward enhanced survival, although this was limited by our small numbers. Fig 1 shows a Kaplan-Meier plot comparing the time to drug failure for patients collected from our department labelled current study and Gniadecki's patients labelled cohort group.
Fig 1.
A Kaplan-Meier plot compares our 7 patients (current study) with those of Gniadecki et al (cohort group).
The effectiveness of biologics in non–biologic-naïve patients is less than that of their biologic-naïve counterparts.2 Explanations for this phenomenon include: exposure to biologic agents may cause the production of ADA, which may then adversely affect the next biologic or differing rates of drug or antibody catabolism and immunologic reorchestration in which suppression of a single cytokine may induce other proinflammatory cytokines with the redundant function.4, 8
Immune complexes can form when ADAs are present; therefore, concomitant immunosuppressive therapy has been found to be associated with a lower frequency of ADA compared with monotherapy treatment.9 In order to reduce ADA, the concomitant use of immunosuppressive medications has been used particularly in inflammatory bowel disease and rheumatoid arthritis patients. Vermeire et al10 showed the incidence of ADA was 44% in patients treated with infliximab and methotrexate versus 73% in patients treated with solely infliximab. The presence of ADAs was associated with a shorter duration of response in patients not taking concomitant immunosuppressive medications compared with patients taking concomitant immunosuppressive medications. This difference was not statistically significant, and the authors felt it was because of low numbers.11 Similar results have been documented with adalimumab in rheumatoid arthritis patients.10
Methotrexate has been used in combination with biologics to prolong drug survival in patients with rheumatoid arthritis and seems to be efficient in reducing immunogenicity in a dose-dependent manner with adalimumab according to Krieckaert et al.9 Van der Heijde et al12 found statistically significant improvements in efficacy when methotrexate was combined with adalimumab in rheumatoid arthritis patients compared with either methotrexate or adalimumab monotherapy. The mechanisms of this effect remain unclear; however, it may act via an additional or synergistic effect as well as or modulation of the immune response.10, 11
Azathioprine has for some time been used in combination with infliximab to improve the efficacy of treatment for patients with ulcerative colitis and Crohn's disease. Panaccione et al13 and Lémann et al14 found that patients treated with azathioprine in addition to infliximab were more likely to remain corticosteroid free compared with patients who received either azathioprine or infliximab as monotherapy.
The addition of concomitant immunosuppressive therapy may avoid the effect of immunogenicity on the efficacy of biologic agents. There are only 2 reports documenting the use of additional immunosuppressive agents with biologic therapies in psoriasis, but neither of them are related to ustekinumab.15, 16
None of our patients responded to systemic immunosuppression on their own. These same immunosuppressants were subsequently introduced in addition to ustekinumab. We found that in most of the cases the doses of the additional immunosuppressive agents tended to be smaller than the standard dose of the same agents when used in the treatment of psoriasis.
One explanation is that the mechanism of their action is through limiting immunogenic neutralization of the biologic treatment and the other would be additional immunosuppression. The choice of the additional immunosuppressive agent depended on the occurrence of side effects with previous exposure and patient comorbidities.
The main limitations with this case series are the small number of patients and we did not have a control group; however, we feel that these results are promising and warrant a multicenter study/survey. Another limitation is that we did not measure, ADA as this test is currently not available in our trust.
Conclusion
Psoriasis is a chronic disease with limited treatment options, and there is a need to have strategies to prolong the longevity of treatment options. Treatment algorithms could include, first, the addition of immunosuppressive agents as soon as efficacy starts to dip or if ADAs are demonstrated in biologic-naïve patients and, second, adding an immunosuppressive agent from the start in biologic nonnaïve patients.
Footnotes
Funding sources: None.
Conflicts of interest: None declared.
References
- 1.Carrascosa J.M., van Doorn M.B., Lahfa M., Nestle F.O., Jullien D., Prinz J.C. Clinical relevance of immunogenicity of biologics in psoriasis: implications for treatment strategies. J Eur Acad Dermatol Venereol. 2014;28(11):1424–1430. doi: 10.1111/jdv.12549. [DOI] [PubMed] [Google Scholar]
- 2.Warren R.B., Smith C.H., Yiu Z.Z. Differential Drug Survival of Biologic Therapies for the Treatment of Psoriasis: A Prospective Observational Cohort Study from the British Association of Dermatologists Biologic Interventions Register (BADBIR) J Invest Dermatol. 2015;135(11):2632–2640. doi: 10.1038/jid.2015.208. [DOI] [PubMed] [Google Scholar]
- 3.Esposito M., Gisondi P., Cassano N. Survival rate of antitumour necrosis factor-α treatments for psoriasis in routine dermatological practice: a multicentre observational study. Br J Dermatol. 2013;169(3):666–672. doi: 10.1111/bjd.12422. [DOI] [PubMed] [Google Scholar]
- 4.Gniadecki R., Bang B., Bryld L.E., Iversen L., Lasthein S., Skov L. Comparison of long-term drug survival and safety of biologic agents in patients with psoriasis vulgaris. Br J Dermatol. 2015;172(1):244–252. doi: 10.1111/bjd.13343. [DOI] [PubMed] [Google Scholar]
- 5.Hsu L., Snodgrass B.T., Armstrong A.W. Antidrug antibodies in psoriasis: a systematic review. Br J Dermatol. 2014;170(2):261–273. doi: 10.1111/bjd.12654. [DOI] [PubMed] [Google Scholar]
- 6.Saravolatz L.D., Wherry J.C., Spooner C. Clinical safety, tolerability, and pharmacokinetics of murine monoclonal antibody to human tumor necrosis factor-alpha. J Infect Dis. 1994;169(1):214–217. doi: 10.1093/infdis/169.1.214. [DOI] [PubMed] [Google Scholar]
- 7.Boulianne G.L., Hozumi N., Shulman M.J. Production of functional chimaeric mouse/human antibody. Nature. 1984;312(5995):643–646. doi: 10.1038/312643a0. [DOI] [PubMed] [Google Scholar]
- 8.Krieckaert C.L., Bartelds G.M., Lems W.F., Wolbink G.J. The effect of immunomodulators on the immunogenicity of TNF-blocking therapeutic monoclonal antibodies: a review. Arthritis Res Ther. 2010;12(5):217. doi: 10.1186/ar3147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Krieckaert C.L., Nurmohamed M.T., Wolbink G.J. Methotrexate reduces immunogenicity in adalimumab treated rheumatoid arthritis patients in a dose dependent manner. Ann Rheum Dis. 2012;71(11):1914–1915. doi: 10.1136/annrheumdis-2012-201544. [DOI] [PubMed] [Google Scholar]
- 10.Vermeire S., Noman M., Van Assche G., Baert F., D'Haens G., Rutgeerts P. Effectiveness of concomitant immunosuppressive therapy in suppressing the formation of antibodies to infliximab in Crohn's disease. Gut. 2007;56(9):1226–1231. doi: 10.1136/gut.2006.099978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bartelds G.M., Wijbrandts C.A., Nurmohamed M.T. Clinical response to adalimumab: relationship to anti-adalimumab antibodies and serumadalimumab concentrations in rheumatoid arthritis. Ann Rheum Dis. 2007;66(7):921–926. doi: 10.1136/ard.2006.065615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Van der Heijde D., Breedveld F.C., Kavanaugh A. Disease activity, physical function, and radiographic progression after longterm therapy with adalimumab plus methotrexate: 5-year results of PREMIER. J Rheumatol. 2010;37(11):2237–2246. doi: 10.3899/jrheum.100208. [DOI] [PubMed] [Google Scholar]
- 13.Panaccione R., Ghosh S., Middleton S. Combination therapy with infliximab and azathioprine is superior to monotherapy with either agent in ulcerative colitis. Gastroenterology. 2014;146(2):392–400. doi: 10.1053/j.gastro.2013.10.052. [DOI] [PubMed] [Google Scholar]
- 14.Lémann M., Mary J.Y., Duclos B. Infliximab plus azathioprine for steroid-dependent Crohn's disease patients: a randomized placebo-controlled trial. Gastroenterology. 2006;130(4):1054–1056. doi: 10.1053/j.gastro.2006.02.014. [DOI] [PubMed] [Google Scholar]
- 15.Vincent T., Best M., Plawecki M., Girard C. Complete reversion of anti-infliximab immunization by methotrexate combination in a patient with psoriasis. J Dermatol. 2015;42(10):1030–1031. doi: 10.1111/1346-8138.13010. [DOI] [PubMed] [Google Scholar]
- 16.Kawakami H., Maeda T., Abe N. Efficacy of adalimumab and methotrexate combination therapy on generalized pustular psoriasispatients unresponsive to infliximab monotherapy due to anti-infliximab antibody development. J Dermatol. 2015;42(1):94–95. doi: 10.1111/1346-8138.12704. [DOI] [PubMed] [Google Scholar]