Abstract
Kv11.1 channels are crucial in cardiac physiology, and there is increasing evidence of physiological roles of different Kv11 channels outside the heart. The HERG (human Kv11.1a) channel has previously been shown to carry substantially more current at elevated temperatures, and we have now comparably investigated the temperature dependence of neuronal Kv11.3 channels and the more ubiquitous heteromeric Kv11.1a/1b channels. Transiently expressed rat Kv11 channels were studied at 21°C, 30°C, and 35°C. At near-physiological temperature, the maximal sustained outward current density was almost three times the mean value obtained at room temperature for Kv11.1a/1b, and increased by ∼150% for Kv11.3. For both channels, reduced inactivation contributed to the current increase at higher temperature. Elevated temperature moved Kv11.1a/1b isochronal activation curves to more negative potentials, but shifted the potential of half-maximal Kv11.3 channel activation to more depolarized values and reduced its voltage sensitivity. Thus, increased temperature stabilized the open state over the closed state of Kv11.1a/1b channels and exerted the opposite effect on Kv11.3 channel activation. Both Kv11 channels exhibited an overall high temperature sensitivity of most gating parameters, with remarkably high Q10 factors of ∼5 for the rate of Kv11.1a/1b activation. The Q10 factors for Kv11.3 gating were more uniform, but still higher for activation than for inactivation kinetics. The results demonstrate that characteristic differences between Kv11.1a/1b and Kv11.3 determined at room temperature do not necessarily apply to physiological conditions. The data provided here can aid in the design of models that will enhance our understanding of the role of Kv11 currents in excitable cells.
Introduction
The ether-à-go-go-related gene (erg) or Kv11 K+ channel family consists of three family members. The three Kv11 channels differ in their voltage dependence and gating kinetics, but they also share unique properties such as their distinct gating behavior and their high sensitivity to class III antiarrhythmic drugs (1) (reviewed in (2)) and external K+ (3). HERG (human erg1; Kv11.1) channels are functionally important for timely repolarization of the human cardiac action potential, and the channels have been investigated in detail concerning their biophysical and pharmacological properties. In the human heart, impaired function of the repolarizing Kv11.1 current IKr due to genetic mutations or more commonly pharmacological block results in a prolongation of the ventricular action potential, possibly inducing long QT syndrome type 2, which increases the risk for life-threatening torsade de pointes arrhythmia (4). The use of specific blockers of Kv11 channels allowed the isolation of the endogenous Kv11.1 current IKr as a drug-sensitive current (5) and revealed gradual differences in the deactivation kinetics of endogenous IKr and heterologously expressed HERG channels (reviewed in (2)).
In 1997, an isoform of Kv11.1 (= Kv11.1a) named erg1b (Kv11.1b) was cloned from heart (6, 7). Kv11.1b lacks most of the Kv11.1a N-terminus and exhibits a much shorter and unique N-terminal sequence. Kv11.1a subunits have an EAG or PAS domain in the distal N-terminus, which is crucial for slow channel deactivation (8, 9, 10, 11). The absence of this region in Kv11.1b readily explains its much faster deactivation kinetics (6, 7, 12). Therefore, differences in Kv11.1a and IKr deactivation kinetics have been suggested to arise from heteromeric Kv11.1 channels formed by coexpressed Kv11.1a and 1b subunits in cardiac tissue (6, 7). In addition to their distinct deactivation kinetics, Kv11.1a and Kv11.1b channels exhibit different biophysical properties, including faster activation and a pronounced right shift in steady-state inactivation for Kv11.1b and heteromeric Kv11.1a/1b channels compared with Kv11.1a (6, 13, 14, 15). Furthermore, functional differences exist between the two Kv11.1 isoforms with respect to channel modulation (14, 16, 17) and the efficacy of some HERG channel-blocking or -activating agents (18, 19, 20) (for review, see (21)). The presence of native heteromeric Kv11.1a/1b channels in the heart was first demonstrated by immunochemical studies (22) and more recently was confirmed by functional studies (23). Coexpression of Kv11.1a and Kv11.1b transcripts also occurs in other tissues, including smooth muscle (24), pituitary, and brain (13). In adult mouse brain, Kv11.1a and 1b have been found to be widely coexpressed on the protein level, and the coassembly of both isoforms has been demonstrated (25). Another feature of Kv11.1 is its de novo expression in neoplastic tissue and its role in cell proliferation. Kv11.1b can be coexpressed with Kv11.1a in a subunit stoichiometry that varies during the cell cycle (for review, see (26)).
The other two Kv11 family members, erg2 (Kv11.2) and erg3 (Kv11.3), are mainly found in the central nervous system, and therefore they have been referred to as neuronal Kv11 channels (1). The different Kv11 channel subunits exhibit distinct but partially overlapping distributions in the brain, with Kv11.1 and Kv11.3 being considerably more abundant than Kv11.2 (25, 27, 28). Electrophysiological investigations of Kv11 channels outside the heart indicate that these channels play a functional role in modulating cellular excitability by contributing to the maintenance of the resting membrane potential or to the phenomenon of action potential frequency accommodation (e.g., (29, 30, 31, 32)) (reviewed in (4, 33)). In particular, it was suggested that the Kv11.3 channel, with its more negative activation threshold, considerably faster activation kinetics, and less steady-state inactivation than the other Kv11 channels (1), is able to contribute to both the resting potential and spike frequency adaptation. So far, Kv11.3 channels have not been characterized at physiological temperatures. Accordingly, all of the described differences in the biophysical properties of Kv11.3 and homo- or heteromeric Kv11.1 channels are based on data obtained at room temperature. Moreover, despite the emerging relevance of heteromeric Kv11.1a/1b channels, their temperature sensitivity has not been investigated in the same detail as that of Kv11.1a (34, 35). To assess the effect of temperature on Kv11.1a/1b and Kv11.3 channels, we comparably analyzed their voltage-dependent gating at room temperature and at constant elevated temperatures. Our data show an overall high temperature sensitivity of both channels, as well as unexpected differences, suggesting that some characteristics that distinguish the channels at room temperature do not hold true for physiological conditions.
Materials and Methods
Heterologous expression
Chinese hamster ovary (CHO) cells were injected or transfected with rat Kv11 cDNA cloned into the pcDNA3 vector as described previously (20, 36). EGFP-N1 (Clontech, Heidelberg, Germany) was coexpressed. For expression of heteromeric Kv11.1a/1b channels, CHO cells were always microinjected using equal concentrations of Kv11.1a and Kv11.1b cDNA (each 40 ng/μL). Kv11.3 cDNA was either injected (100 ng/μL) or used for transfection (1 ng/μL) according to the Lipofectamine 2000 method (Invitrogen, Karlsruhe, Germany).
Electrophysiology
Experiments were performed 8–48 h after CHO cell transfection or 5–48 h after microinjection in the conventional whole-cell configuration of the patch-clamp technique. The patch pipettes had resistances of 2–4 MΩ. Data were low-pass filtered at 3 kHz and compensated for both fast- and slow-capacity transients. The access resistance ranged from 2.5 to maximally 10 MΩ and series resistance was compensated for by 60–90%. Data were not corrected for the liquid junction potential error of ∼4 mV. An EPC-9 patch-clamp amplifier was used in combination with PULSE stimulation and data acquisition software (HEKA, Lamprecht, Germany). Experiments were performed at a constant bath temperature (21°C, 30°C, or 35°C) using a temperature-control system (WK 14; Colora Messtechnik, Lorch, Germany) combined with an in-house-made holder of the recording chamber, designed for temperature control by a circulating pump. The bath temperature was measured close to the recorded cell. Experiments at different temperatures were performed on different cells.
Solutions
The external Ringer solution contained 140 mM NaCl, 5 mM KCl, 0.8 mM MgCl2, 1 mM CaCl2, 10 mM HEPES, 5 mM glucose, pH adjusted to 7.4 with NaOH. The pipette solution contained 140 mM KCl, 2 mM MgCl2, 1 mM CaCl2, 10 mM HEPES, 2.5 mM EGTA, pH adjusted to 7.3 with KOH.
Data analysis and statistics
Experimental data are given as means ± SEM, with n representing the number of experiments with different cells. Student’s two-tailed unpaired t-test was used to test for significance if not mentioned otherwise. Data processing was performed with PulseFit 8.65 (HEKA, Lamprecht, Germany), Excel (Microsoft, Redmond, WA), and Sigmaplot 11.0 (Systat Software, San Jose, CA).
To assess the voltage dependence of Kv11 channel activation, normalized data were fitted with a Boltzmann equation, Y = 1/(1 + exp (−(V − V0.5)/k)), where V0.5 is the potential of half-maximal channel activation and k is the slope factor. For Kv11.1a/1b channels, the tail current amplitude represents a reasonable measure of the relative proportion of channels activated at the end of the preceding depolarization, as elaborated for Kv11.1a. Since this procedure is less practicable for Kv11.3 with its initial current transient, reduced inactivation, and fast deactivation, we used the total charge of the tail current as a more appropriate measure of preceding channel activation, as suggested previously (3). A comparison of both analysis methods yielded qualitatively similar results concerning the temperature sensitivity of Kv11.3 channel activation (data not shown). The equivalent thermodynamic form of the above Boltzmann equation is Y = 1/(1 + exp (−(zgFV − ΔG0)/RT)), where zg is the equivalent charge, F is the Faraday’s constant, ΔG0 is the Gibbs free energy of activation at 0 mV, R is the gas constant, and T is the temperature in Kelvin. zg and ΔG0 were calculated as follows: zg = RT/(F × k), and ΔG0 [kcal/mol] = 0.2389 zgFV0.5.
As a measure of steady-state inactivation, the relative conductance values for the single experiments were calculated from the fully activated current-voltage (I-V) relationship using values of the reversal potential estimated by interpolation. The resulting conductance-voltage data were fitted with a Boltzmann equation (see above), yielding inactivation curves with V0.5 as the potential of half-maximal channel inactivation and k as the slope factor.
We determined the time course of activation by using an envelope-of-tails protocol. Tail current amplitudes (for Kv11.1a/1b) or the charge of the tail current (for Kv11.3) were normalized with respect to the maximal and minimal values. Apart from the first data points (obtained with the shortest test pulse durations) where no significant tail current was elicited, the data were fitted with a single exponential function yielding the time constant of activation (τact) and a delay (d): Y(t) = 1 − exp ((−t + d)/τact), where Y is either the normalized tail current amplitude or the charge of the tail current, t is the time, and the delay d represents the time at which the function yields Y = 0. The time constant of activation (τact) characterizes the slowest and thus rate-limiting step in the activation pathway. The delay d is a measure of the sum of the time constants of all other forward steps in the activation pathway. Together, τact and d characterize the time taken by the Kv11 channels to traverse through the whole activation pathway, including multiple preopen closed states before the final opening.
The time constant of inactivation was obtained by fitting a single exponential function to the later part of the decaying current traces to minimize the influence of capacitive currents and insufficient voltage control due to large current amplitudes. Recovery from inactivation and deactivation kinetics was analyzed using fully activated Kv11 channels. The time course of recovery from inactivation was fitted in the more depolarized voltage range with a single exponential function. At more negative potentials, which induced considerable channel deactivation, the time constant of recovery from inactivation was obtained by fitting the sum of two exponential functions to the hook-like part of the current traces. The time constants of fast and slow deactivation were obtained by fitting the sum of two exponential functions to the exponentially decaying part of the tail currents. Weighted time constants were calculated as the sum of both time constants, each multiplied by its relative contribution to the tail current decay.
Q10 values describing the temperature dependence of the Kv11 channel kinetics were calculated according to the equation Q10 = (τ1/τ2) exp (10/(T2 − T1)), where τX is the mean time constant at the temperature TX. The analysis of temperature effects on Kv11.1a/1b channels was based on two sets of experiments that were conducted at different times and differed with respect to the Kv11.1 cDNA preparations and the passage number of CHO cells. The first set of experiments consisted of recordings at 21°C and 35°C (data set A) and the other set comprised experiments at 21°C and 30°C (data set B). There was a tendency toward faster kinetics at 21°C in set B compared with set A (see Table 1), which might be explained by a slightly higher relative contribution of Kv11.1b subunits to the functional Kv11.1 channels in set B. Due to the observed differences at 21°C, the kinetic analyses and Q10 values were based on either data set A or data set B.
Table 1.
Temperature Dependence of Kv11.1a/1b Channels
| 21°C A | 21°C B | 30°C B | 35°C A | p B/A | Q10 B/A | |
|---|---|---|---|---|---|---|
| Activation Parameters | ||||||
| V0.5 (mV) | −29.3 ± 1.1 | −29.4 ± 0.5 | −33.1 ± 0.9 | −39.5 ± 0.6 | ∗∗/∗∗∗ | n.a. |
| k (mV) | 7.4 ± 0.3 | 7.2 ± 0.3 | 6.5 ± 0.3 | 6.6 ± 0.2 | n.s./∗ | n.a. |
| zg (e−) | 3.5 ± 0.1 | 3.6 ± 0.1 | 4.1 ± 0.2 | 4.1 ± 0.2 | ∗/∗∗ | n.a. |
| ΔG0 (kcal/mol) | −2.4 ± 0.1 | −2.4 ± 0.1 | −3.1 ± 0.2 | −3.8 ± 0.2 | ∗/∗∗∗ | n.a. |
| n | 17 | 7 | 10 | 17 | ||
| Inactivation Parameters (mV) | ||||||
| V0.5 | −45.2 ± 2.5 | – | – | −19.4 ± 3.0 | ∗∗∗ | n.a. |
| k | −29.4 ± 1.2 | – | – | −24.0 ± 1.6 | ∗ | n.a. |
| n | 9 | 9 | ||||
| Activation Kinetics (ms) at 20 mV | ||||||
| τ act | 192 ± 10.7 | 147 ± 5.8## | 36.6 ± 2.3 | 23.1 ± 1.4 | ∗∗∗/∗∗∗ | 4.69/4.55 |
| Delay | 37.3 ± 2.8 | 39.1 ± 3.4 | 8.1 ± 0.8 | 3.2 ± 0.3 | ∗∗∗/∗∗∗ | 5.73/5.79 |
| n | 16 | 8 | 12 | 14 | ||
| τ Inact (ms) | ||||||
| At 40 mV | 9.2 ± 0.7 | 7.8 ± 2.1 | 3.1 ± 0.2 | 2.0 ± 0.3 | ∗∗/∗∗∗ | 2.82/2.94 |
| At 20 mV | 13.3 ± 0.7 | 11.0 ± 2.0 | 4.1 ± 0.2 | 2.9 ± 0.3 | ∗∗/∗∗∗ | 2.98/2.98 |
| At 0 mV | 16.9 ± 0.6 | 13.6 ± 1.4# | 4.9 ± 0.3 | 3.3 ± 0.2 | ∗∗/∗∗∗ | 3.11/3.22 |
| n | 15 | 3 | 4 | 13 | ||
| τ Recov (ms) | ||||||
| At 20 mV | 9.0 ± 0.6 | 9.2 ± 0.4 | 2.9 ± 0.2 | 1.4 ± 0.1 | ∗∗∗/∗∗∗ | 3.60/3.79 |
| At −20 mV | 13.6 ± 0.4 | 13.0 ± 0.5 | 4.1 ± 0.2 | 2.4 ± 0.1 | ∗∗∗/∗∗∗ | 3.59/3.50 |
| n | 13–14 | 6 | 5 | 12 | ||
| Deactivation kinetics (ms) at −100 mV | ||||||
| τ fast | 31.2 ± 2.5 | 20.4 ± 1.1## | 7.3 ± 0.6 | 6.6 ± 0.5 | ∗∗∗/∗∗∗ | 3.13/3.03 |
| τ slow | 148 ± 12 | 93 ± 11## | 34.7 ± 1.7 | 29.5 ± 3.2 | ∗∗∗/∗∗∗ | 3.00/3.17 |
| τ weighted | 56.1 ± 5.1 | 33.5 ± 2.8## | 11.6 ± 1.0 | 10.9 ± 1.1 | ∗∗∗/∗∗∗ | 3.26/3.22 |
| n | 12 | 7 | 7 | 11 | ||
| Deactivation Kinetics (ms) at −50 mV | ||||||
| τ fast | 122 ± 7.0 | 111 ± 8.8 | 46.4 ± 5.5 | 38.4 ± 1.5 | ∗∗∗/∗∗∗ | 2.65/2.29 |
| τ slow | 830 ± 91 | 547 ± 59 | 274 ± 42 | 278 ± 15 | ∗∗/∗∗∗ | 2.16/2.18 |
| τ weighted | 579 ± 77 | 348 ± 45 | 155 ± 26 | 155 ± 12 | ∗∗/∗∗∗ | 2.45/2.56 |
| n | 11 | 5 | 5 | 11 | ||
Q10 values were calculated using mean values obtained at 21°C and 30°C (data set B) or at 21°C and 35°C (data set A). Activation parameters correspond to 4-s isochronal activation and are explained in Materials and Methods. Data are means ± SEM. n, number of independent experiments; ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001, significantly different from the corresponding data at 21°C; n.s., not significant; n.a., not applicable; #p < 0.05 and ##p < 0.01, significant differences between 21°C data sets A and B.
Results
Expression of heteromeric Kv11.1a/1b channels
Kv11.1a/1b channels were transiently expressed in CHO cells by cDNA injection into single cells to yield constant ratios of Kv11.1a and Kv11.1b channel subunits. Patch-clamp experiments were performed on different cells at 21°C and 35°C (data set A) or at 21°C and 30°C (data set B). The Kv11.1 currents recorded at 21°C with relatively fast deactivating tail currents were typical for measurements of heteromeric Kv11.1a/1b channels performed at room temperature (Fig. 1 A). The most apparent difference between homomeric Kv11.1a and Kv11.1b channels is the much faster deactivation kinetics of Kv11.1b, and in heteromeric channels, the time constants of deactivation mirror the relative contribution of both isoforms to the functional channels (6, 7, 14). In our experiments, small differences in Kv11.1a/1b gating kinetics existed between the two data sets (see Table 1 and Materials and Methods), pointing to a slightly higher relative contribution of Kv11.1b subunits in data set B.
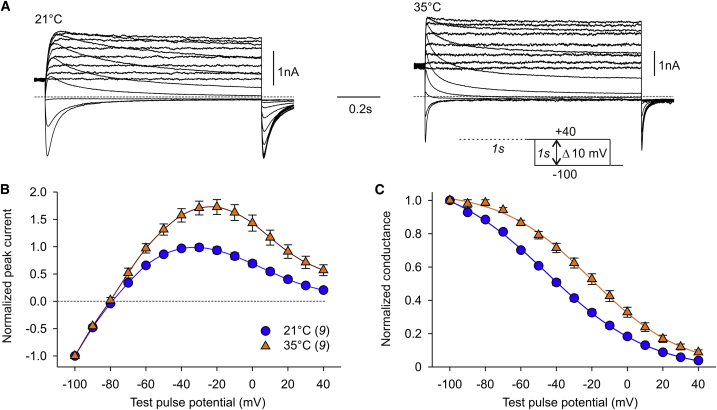
Figure 1.
Temperature effects on the voltage dependence of Kv11.1a/1b activation. Membrane currents were recorded in CHO cells transiently expressing Kv11.1a/1b channels. (A) Representative Kv11.1a/1b current traces recorded from different cells at 21°C and 35°C. From a holding potential of −80 mV, channel activation was assessed by 4-s test pulses between −90 mV and +40 mV with 10 mV increments, followed by a constant pulse to −110 mV (see pulse diagram). (B) I-V relationship of outward current amplitudes measured at the end of the depolarizing test pulses at 21°C, 30°C, and 35°C. Data are means ± SEM of absolute amplitudes (upper panel) or current amplitudes normalized to the individual maximum outward current (lower panel). The number of experiments is given in brackets. The inset shows mean maximal outward current densities determined in matched experiments (data set A) at 21°C (−10 mV; n = 17) and 35°C (−10 mV; n = 17); asterisks indicate significant differences between the unpaired data (∗∗∗p < 0.001). (C) Peak tail current amplitudes plotted against the potential of the preceding test pulse. Data were normalized to the maximum tail current amplitudes, averaged, and fitted with a Boltzmann function to yield activation curves. The fit parameters of the 4-s isochronal activation curves were V0.5 = −29.3 mV and k = 7.7 mV at 21°C, V0.5 = −33.1 mV and k = 6.7 mV at 30°C, and V0.5 = −39.6 mV and k = 6.6 mV at 35°C. In part of the experiments at the recording temperature of 21°C, an additional activation protocol was applied using depolarizing test pulses of 12 s duration (open circles and dashed fit line; V0.5 = −34.4 mV, k = 6.0 mV). To see this figure in color, go online.
Effects of temperature on the voltage dependence of Kv11.1a/1b activation and inactivation
Fig. 1 A shows representative Kv11.1a/1b membrane currents recorded in a CHO cell at 21°C and in another cell at 35°C using the indicated activation protocol. Kv11.1 currents were evoked at depolarizing test pulse potentials more positive than −60 mV, and current amplitudes increased up to potentials between −20 and 0 mV. Upon test pulses to more positive potentials, the outward current amplitude decreased, resulting in the bell-shaped I-V relationship that is typical for channels of the Kv11 family. The mean outward current amplitudes measured at the end of the variable 4-s test pulses were higher at the elevated temperature (Fig. 1 B, top). When we analyzed all experiments in data set A, we found a significantly higher maximal current density at −10 mV in the 35°C experiments compared with the 21°C experiments (21°C: 62.3 ± 23.6 pA/pF, n = 17; 35°C: 179.8 ± 21.7 pA/pF, n = 17; p = 0.0004). Normalization of the steady-state current amplitudes illustrates maximal current amplitudes close to −10 mV and a broadening of the bell-shaped I-V curves with higher temperature (Fig. 1 B, bottom).
The voltage dependence of Kv11.1a/1b channel activation was determined by plotting the peak tail current amplitudes against the preceding test pulse potential (Fig. 1 C). Boltzmann functions were fitted to the data points, yielding activation curves characterized by the potential of half-maximal channel activation (V0.5) and the slope factor (k). At higher temperatures, the activation curves were slightly steeper and significantly shifted to more negative potentials with V0.5 values of approximately −29 mV at 21°C, −33 mV at 30°C, and −39 mV at 35°C (see Table 1). All of these values describe 4-s isochronal activation curves. An inspection of the current traces recorded at 21°C shows that the membrane currents did not reach a steady amplitude at the end of the 4-s test pulses in the negative potential range. To assess the influence of the test pulse duration, we conducted additional activation protocols with 12-s test pulses in part of the 21°C experiments. The increase in test pulse duration led to a hyperpolarizing shift in the isochronal activation curves by −4.9 ± 0.8 mV (n = 5; p = 0.0015) and to an increased steepness of the curves (4-s: k = 6.9 ± 0.2 mV; 12-s: k = 5.8 ± 0.2 mV; p = 0.0037).
We assessed the effect of temperature on Kv11.1a/1b steady-state inactivation by means of availability protocols using the fully activated I-V relationship (Fig. 2). Since the majority of Kv11.1a/1b channels were inactivated during the depolarizing prepulse to +40 mV, the variable test pulses induced time- and voltage-dependent recovery from inactivation, followed by deactivation at more negative potentials. Experiments were carried out at 21°C and 35°C. Fig. 2 B shows that the normalized, fully activated I-V plots for the two temperatures differed in the depolarized potential range, with more outward current at the higher temperature. Individual normalized peak current values were used to calculate the relative Kv11.1a/1b conductance (Fig. 2 C). Boltzmann functions were fitted to the relative conductance values of each experiment, yielding inactivation curves with ∼25 mV less negative inflection potentials at the elevated temperature (Table 1). In addition, the slope factor k was significantly smaller at 35°C (Table 1), indicating an increased voltage sensitivity of channel inactivation at higher temperature.
Figure 2.
Temperature effects on fully activated Kv11.1a/1b currents and steady-state inactivation. (A) Representative families of fully activated Kv11.1a/1b currents recorded from different cells at 21°C and 35°C. Membrane currents were elicited by 1-s depolarizations to +40 mV, followed by variable 1-s test pulses from +40 to −120 mV with 10 mV decrements and a final step to −100 mV (see pulse protocol). (B) To obtain the fully activated I-V relationship, maximal current amplitudes during the test pulses were plotted as a function of the test pulse potential. Data from individual experiments were normalized to the inward current amplitude obtained upon the test pulse to −100 mV before averaging. Data points are means ± SEM, and the number of experiments is given in brackets. (C) Normalized current amplitudes of the experiments shown in (B) were used to calculate the relative conductance as a measure of the voltage dependence of Kv11.1a/1b steady-state inactivation. Data were normalized to the conductance value at −100 mV before averaging. Lines denote fits of a Boltzmann equation to the averaged conductance-voltage data (21°C: V0.5 = −45.4 mV, k = −29.2 mV; 35°C: V0.5 = −20.1 mV, k = −25.5 mV). To see this figure in color, go online.
Effect of temperature on Kv11.1a/1b gating kinetics
We assessed the Kv11.1a/1b activation kinetics by conducting envelope-of-tails protocols at 21°C and 30°C, using four different potentials of the depolarizing pulse (Fig. 3). Test pulse durations were adjusted for the different temperatures. Representative current traces with an overlay of the recordings obtained with the test pulse potentials of 20 and 60 mV are illustrated in Fig. 3 A. At both temperatures, longer depolarizations to +60 mV induced obvious inactivation of the macroscopic Kv11.1a/1b current, whereas the depolarizations to 20 mV elicited slowly increasing outward currents. The presence of concomitant channel inactivation can be deduced from the diverging time courses of the increase in outward current and the increase in tail current. To illustrate the latency of channel opening due to closed-closed transitions of the channels, tail current amplitudes were plotted against the duration of the preceding test pulse as a semilogarithmic graph (Fig. 3 B). In the range of increasing tail current amplitudes, the data were fitted with a single exponential function, yielding values for the time constant of activation (τact) and the delay (see Materials and Methods). Both the delay and τact decreased steeply with more depolarized test pulse potentials (Fig. 3 C). The parallel course of the data obtained at 21°C and 30°C indicates that the voltage dependence of the time course of Kv11.1a/1b channel activation was not significantly influenced by changes in temperature. The voltage dependence of τact was well fitted by an exponential declining to an asymptotic value of ∼45 ms at 21°C and to 9 ms at 30°C (Fig. S1, A and B, in the Supporting Material). Increasing temperature strongly accelerated channel activation. Temperature had a greater effect on the delay than on the time constant of activation, with mean Q10 values over the tested voltage range of 5.56 ± 0.12 and 4.95 ± 0.16 (p = 0.025), respectively. Data set A of the Kv11.1a/1b experiments included recordings obtained with envelope-of-tails protocols at 21°C and 35°C, using depolarizing pulses to +20 mV. Despite the difference in τact at 21°C compared with data set B, these experiments demonstrated the same high temperature sensitivities of the delay and the time constant of activation (see Table 1).
Figure 3.
Effect of temperature and voltage on the time course of Kv11.1a/1b channel activation. (A) Representative Kv11.1a/1b currents recorded from different cells at 21°C and 30°C using envelope-of-tails protocols. Please note the different timescales. The pulse protocols consisted of depolarizing pulses of increasing duration (2–2300 ms at 21°C, and 1–440 ms at 30°C), followed by a hyperpolarization to −100 mV to elicit tail currents. Four different potentials were used for the depolarizing pulse in every experiment, and the examples show an overlay of current traces recorded with test pulses to 20 mV (black traces) and 60 mV (gray traces). (B) Normalized peak tail current amplitudes were averaged and plotted against the duration of the preceding depolarizing pulse. Data points obtained with longer pulse durations were fitted with single exponential functions, yielding values for the time constant of activation and a delay. Data are means ± SEM from experiments performed at 21°C (n = 8) or 30°C (n = 12). (C) Mean values (± SEM) of the delay and the time constant of activation as a function of the test pulse potential. To see this figure in color, go online.
The effect of temperature on the time course of inactivation of Kv11.1a/1b channels was analyzed by means of triple pulse protocols, as shown in Fig. 4 A for experiments in data set A. The reduced steady-state inactivation at higher temperature (see above) was apparent from the higher relative current amplitudes at the end of the P3 pulse at 35°C compared with 21°C (ratio Isteady-state to Ipeak at −20 mV: 21.6% ± 1.4% at 21°C, n = 7, and 44.4% ± 2.7% at 35°C, n = 5; p < 0.001). The current decay during the P3 test pulses was fitted by single exponential functions to achieve the time constants of inactivation (τinact). Fig. 4 B shows the voltage dependence of τinact obtained from recordings at 21°C and 35°C, demonstrating faster inactivation upon stronger depolarization. In the voltage range between +60 and −20 mV, the mean values of τinact increased continuously with more negative P3 potentials at 21°C, whereas maximal values of τinact were observed at 0 mV at 35°C (Fig. 4 B). Due to this divergent voltage dependence, the calculated Q10 values for the time course of inactivation were higher in the negative voltage range than at positive potentials with a mean Q10 of 3.11 ± 0.07 for the nine P3 potentials. Similar temperature effects were observed in the independent experiments of data set B at 21°C and 30°C (see Table 1), yielding a mean Q10 of 3.02 ± 0.08 over the same voltage range.
Figure 4.
Temperature effects on the time course of Kv11.1a/1b channel inactivation, recovery from inactivation, and deactivation. (A) Typical examples of Kv11.1a/1b current traces recorded at 21°C and 35°C with triple pulse protocols designed to study inactivation rates. Please note the different timescales. Starting from a holding potential of −80 mV, the potential was stepped for 500 ms to +80 mV (P1) to maximally activate and inactivate the channels. Subsequent short P2 pulses to −140 mV (10 ms at 21°C and 2 ms at 35°C) to open the Kv11.1 channels due to fast recovery from inactivation were followed by variable 300-ms test pulses to potentials between +60 and −20 mV (P3) to reinduce inactivation (see pulse protocol). (B) Means (± SEM) of time constants of inactivation (open symbols) and time constants of recovery from inactivation (solid symbols) were plotted versus the test pulse potential; the number of experiments is given in brackets. Time constants of inactivation were obtained from monoexponential fits to the decay phase of the current traces during the P3 pulse as shown in (A). Time constants of recovery from inactivation and deactivation were obtained from experiments with fully activated Kv11.1a/1b channels as shown in Fig. 2A. (C) Voltage and temperature dependence of Kv11.1a/1b channel deactivation. Time constants of fast and slow deactivation, and the relative contribution of the fast deactivating current component as a function of the test pulse potential are shown. The number of experiments is given in brackets; asterisks indicate significant differences (∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001). To see this figure in color, go online.
The temperature dependence of the recovery and deactivation kinetics was analyzed from experiments performed with the availability protocol at 21°C and 35°C (see Fig. 2 A). Fig. 4 B shows the time constants of recovery from inactivation (τrecov). The mean τrecov values exhibited a bell-shaped voltage dependence, with maximal values at potentials between −40 and −20 mV at 21°C and approximately −15 mV at 35°C. In the voltage range between −20 and +20 mV, where deactivation was absent or marginal, the differences in the mean τrecov values yielded a mean Q10 value of 3.48 ± 0.09 for the time course of recovery from inactivation. The respective Q10 value at −100 mV was 3.81. Similar effects of temperature were found in the experiments of data set B at 21°C and 30°C (see Table 1), yielding a mean Q10 of 3.69 ± 0.04 in the voltage range between −20 and +20 mV, and a Q10 value of 4.08 at −100 mV.
Fig. 4 C illustrates the effects of temperature and voltage on the Kv11.1a/1b channel deactivation kinetics. The time course of current decay was fitted with the sum of two exponential functions, yielding a fast (τdeact,fast) and a slow (τdeact,slow) time constant of deactivation as well as the ratio of both current components. With more negative membrane potentials, both time constants decreased steeply and the relative contribution of the fast deactivating current component became dominant. The acceleration of the deactivation time course at 35°C compared with 21°C was voltage dependent: the decrease in both time constants was clearly more pronounced for Kv11.1a/1b inward currents at potentials negative to −80 mV than for outward currents positive to −80 mV. The voltage dependence of the temperature effect on deactivation kinetics is mirrored in the higher Q10 values calculated for the mean time constants at −100 mV compared with −50 mV (Table 1). On the other hand, the contribution of the fast deactivating current component increased at higher temperature in the less negative voltage range (Fig. 4 C). To account for this difference, we calculated weighted deactivation time constants. The corresponding Q10 values were also higher at −100 mV than at −50 mV (Table 1). These findings were confirmed by the analysis of data set B recorded at 21°C and 30°C. Despite a significantly faster time course of deactivation at 21°C compared with data set A, the Q10 values of the Kv11.1a/1b deactivation kinetics for both data sets were similar (Table 1).
Effects of temperature on the voltage dependence of Kv11.3 activation and inactivation
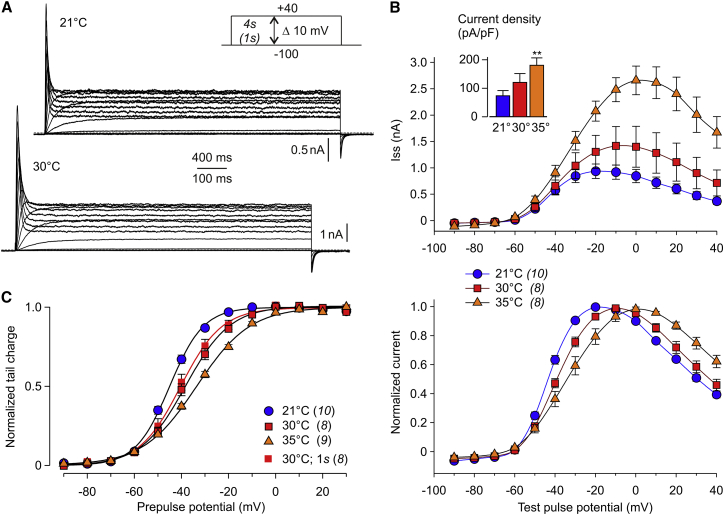
Kv11.3 channel activation was studied at 21°C, 30°C, and 35°C. Fig. 5 A shows Kv11.3 membrane currents recorded at 21°C with 4-s test pulses and, for comparison, Kv11.3 currents recorded at 30°C with 1-s test pulses plotted on a fourfold expanded timescale. Kv11.3 currents became visible close to −60 mV and current amplitudes increased upon stronger depolarizations. As is typical for Kv11.3 channels, test pulses more positive than approximately −20 mV induced increasing initial current transients followed by lower sustained currents, resulting in a bell-shaped I-V relationship of the Kv11.3 steady-state current. Fig. 5 B shows the results of experiments with 4-s test pulses. At 21°C, the steady-state current was maximal at −20 mV. With higher temperature, the potential for maximal sustained current shifted to −10 mV at 30°C and to 0 mV at 35°C. The mean Kv11.3 current amplitudes tended to be higher in the experiments at elevated temperature, with a more pronounced difference at depolarized potentials. The maximal steady-state current density was significantly higher in the 35°C experiments (163.2 ± 29.9 pA/pF at 0 mV, n = 9) than in the 21°C experiments (73.6 ± 23.3 pA/pF at −20 mV, n = 10; p = 0.009; see inset in Fig. 5 B). Normalization of the steady-state current amplitudes illustrates a right shift of the bell-shaped I-V curves with higher temperature (Fig. 5 B, bottom). The effect of temperature on the voltage dependence of Kv11.3 channel activation was determined by plotting the normalized integral of the tail currents against the prepulse potential (Fig. 5 C). Boltzmann functions fitted to the data points yielded 4-s isochronal activation curves. In contrast to the results for Kv11.1a/1b, the potential of half-maximal Kv11.3 channel activation shifted significantly to more positive potentials with higher temperature, and the slope factor k increased, reflecting a decrease in the voltage sensitivity of Kv11.3 channel activation at higher temperature (Table 2). In additional experiments, 1-s isochronal activation curves were obtained at 30°C, and they did not significantly differ (V0.5 = −40.1 ± 2.0 mV, k = 8.5 ± 0.6 mV, n = 8; p = 0.48 and p = 0.41; Fig. 5, A and C) from the 4-s isochronal activation curves at the same temperature, demonstrating that these activation curves closely mirror the steady-state conditions of the Kv11.3 channels.
Figure 5.
Temperature effects on Kv11.3 channel activation. Membrane currents were recorded in CHO cells transiently expressing Kv11.3 channels. (A) Examples of Kv11.3 current traces from two different cells using 4-s (21°C) or 1-s (30°C) depolarizing test pulses. Please note the different timescales. Test pulses varied between −100 mV and +40 mV with 10 mV increments and were followed by a constant pulse to −100 mV to elicit tail currents (see pulse diagram). (B) I-V relationship of outward current amplitudes measured at the end of 4-s depolarizing test pulses at 21°C, 30°C, and 35°C. Data are means ± SEM of absolute amplitudes (upper panel) or current amplitudes normalized to the individual maximum outward current (lower panel). The number of experiments is given in brackets in the symbol legend. The inset shows the mean maximal outward current densities determined in these experiments. Asterisks indicate a significant difference between the unpaired data obtained at 35°C and 21°C (∗∗p < 0.01). (C) Charge of the tail current plotted against the potential of the preceding test pulse. Charge values of individual experiments were normalized to the maximum tail charge, averaged, and fitted with a Boltzmann function. Symbols with a black margin represent means ± SEM from experiments with 4-s test pulses for the three different temperatures. The fit parameters of the 4-s isochronal activation curves are V0.5 = −45.0 mV and k = 7.2 mV at 21°C, V0.5 = −38.2 mV and k = 9.5 mV at 30°C, and V0.5 = −33.5 mV and k = 11.4 mV at 35°C. At the recording temperature of 30°C, additional experiments were performed with 1-s test pulses (squares without margin; V0.5 = −40.4 mV, k = 8.8 mV). The number of experiments is given in brackets. To see this figure in color, go online.
Table 2.
Temperature Dependence of Kv11.3 Channels
| 21°C | 30°C | 35°C | P Δ9°/ Δ14° | Q10 Δ9°/ Δ14° | |
|---|---|---|---|---|---|
| Activation Parameters | |||||
| V0.5 (mV) | −44.9 ± 0.8 | −38.3 ± 1.5 | −33.5 ± 0.8 | ∗∗∗/∗∗∗ | n.a. |
| k (mV) | 7.1 ± 0.3 | 9.1 ± 0.3 | 11.3 ± 0.3 | ∗∗∗/∗∗∗ | n.a. |
| zg (e−) | 3.6 ± 0.1 | 2.9 ± 0.1 | 2.4 ± 0.1 | ∗∗/∗∗∗ | n.a. |
| ΔG0 (kcal/mol) | −3.8 ± 0.2 | −2.6 ± 0.2 | −1.8 ± 0.1 | ∗∗∗/∗∗∗ | n.a. |
| n | 10 | 8 | 9 | ||
| Inactivation Parameters (mV) | |||||
| V0.5a | −16.5 ± 1.4 | −2.4 ± 2.2 | – | ∗∗∗ | n.a. |
| ka | −22.0 ± 0.7 | −21.0 ± 1.7 | – | n.s. | n.a. |
| na | 12 | 7 | – | ||
| V0.5b | −11.5 ± 2.5 | – | 14.9 ± 2.4 | ∗∗∗ | n.a. |
| kb | −23.9 ± 0.8 | – | −17.6 ± 1.2 | n.a. | |
| nb | 8 | – | 8 | ∗∗∗ | |
| Activation Kinetics (ms) at 20 mV | |||||
| τ act | 32.4 ± 2.4 | 9.8 ± 1.0 | – | ∗∗∗ | 3.76 |
| Delay | 7.9 ± 0.6 | 2.7 ± 0.2 | – | ∗∗∗ | 3.38 |
| n | 12 | 8 | – | ||
| τ Inact (ms) | |||||
| At 40 mV | 10.0 ± 0.4 | 4.3 ± 0.5 | 2.2 ± 0.2 | ∗∗∗/∗∗∗ | 2.58/2.93 |
| At 20 mV | 14.0 ± 0.5 | 5.8 ± 0.6 | 3.3 ± 0.2 | ∗∗∗/∗∗∗ | 2.65/2.80 |
| At 0 mV | 18.2 ± 0.8 | 6.9 ± 0.4 | 4.4 ± 0.2 | ∗∗∗/∗∗∗ | 2.91/2.77 |
| n | 10 | 9 | 9 | ||
| τ Recov (ms) | |||||
| At 20 mV | 13.3 ± 0.8 | 5.0 ± 0.5 | 3.0 ± 0.2 | ∗∗∗/∗∗∗ | 2.96/2.92 |
| At −20 mV | 17.9 ± 0.7 | 5.2 ± 0.2 | 2.8 ± 0.1 | ∗∗∗/∗∗∗ | 3.94/3.74 |
| n | 10 | 10 | 9 | ||
| Deactivation Kinetics (ms) at −100 mV | |||||
| τ fast | 15.1 ± 1.0 | 4.3 ± 0.2 | 2.4 ± 0.1 | ∗∗∗/∗∗∗ | 4.04/3.73 |
| τ slow | 77 ± 6.6 | 26.0 ± 1.4 | 13.1 ± 0.5 | ∗∗∗/∗∗∗ | 3.35/3.56 |
| n | 10 | 9 | 7 | ||
Kv11.3 steady-state and kinetic parameters were obtained at different temperatures. Activation parameters correspond to 4-s isochronal activation; all parameters are explained in Materials and Methods. Statistical p-values and Q10 values correspond to differences in values obtained at 21°C and 30°C (Δ9°C) or at 21°C and 35°C (Δ14°C); ∗∗∗p < 0.001, ∗∗p < 0.01; n.s., not significant (Bonferroni correction for multiple testing was used where appropriate); n, number of independent experiments; all experimental data are means ± SEM.
The listed inactivation parameters result from experiments as shown in Fig. 6 using a prepulse potential of +80 mV.
The listed inactivation parameters result from experiments as shown in Fig. 6 using a prepulse potential of+40 mV.
Kv11.3 steady-state inactivation was investigated using the fully activated I-V relationship. Experiments were carried out at 21°C and 30°C in the potential range between −120 and +80 mV, as well as at 21°C and 35°C in the potential range between −120 and +40 mV (Fig. 6). Both fully activated I-V plots (Fig. 6, B and D) showed less pronounced inward rectification at higher temperature, with maximal outward current amplitudes shifted to more depolarized values. Individual normalized peak current values were used to calculate the relative Kv11.3 conductance for each experiment. Fig. 6, C and E, show that the resulting conductance values deviate from a sigmoidal course at test potentials negative to about −20 mV, which is most probably due to concomitant deactivation. Therefore, only data in the potential range with no apparent or only marginal deactivation were considered for fits with a Boltzmann function reflecting the Kv11.3 inactivation curve. In experiments with +80 mV as the prepulse potential, the inflection potential significantly differed between the measurements obtained at 21°C and 30°C without changes in the slope of the inactivation curve (Fig. 6 C; Table 2). The experiments with +40 mV as the prepulse potential yielded similar results, with significantly reduced inactivation at the near-physiological temperature of 35°C compared with 21°C. In addition, the slope of the inactivation curve was steeper at the elevated temperature (Fig. 6 E; Table 2).
Figure 6.
Temperature effects on fully activated Kv11.3 currents and steady-state inactivation. (A) Representative families of fully activated Kv11.3 currents recorded in two different cells at 21°C and 30°C. Membrane currents were elicited by 500-ms depolarizing pulses to +80 mV followed by variable 250-ms test pulses between +80 and −120 mV with 10 mV decrements, and a final step to +80 mV (see pulse protocol). (B) To obtain the fully activated I-V relationship, maximal current amplitudes during the variable test pulses were plotted as a function of the test pulse potential. Data from individual experiments were normalized to the inward current amplitude obtained upon the test pulse to −100 mV before averaging. Data points are means ± SEM, and the number of experiments is given in brackets. (C) Normalized current amplitudes of the experiments shown in (B) were used to calculate the relative maximal conductance as a measure of the voltage dependence of Kv11.3 steady-state inactivation. Lines denote fits of a Boltzmann equation to the conductance data in the voltage range between −30 mV and +80 mV (21°C: V0.5 = −17.7 mV, k = −22.4 mV) or between −20 mV and +80 mV (30°C: V0.5 = −4.4 mV, k = −21.3 mV). (D and E) With a recording temperature of 35°C, Kv11.3 steady-state inactivation was studied in the voltage range between −120 mV and +40 mV (see pulse protocol in D), and experiments were performed at 21°C with the same pulse protocol for comparison. (D) Fully activated I-V relationship. Current amplitudes were normalized to the inward current amplitude obtained upon the test pulse to −100 mV before averaging. Data points are means ± SEM, and the number of experiments is given in brackets. (E) Relative maximal conductance (means ± SEM) plotted as a function of the test pulse potential. Individual data were normalized to the value at −100 mV. Lines denote fits of a Boltzmann equation to the mean conductance data in the voltage range between −30 mV and +40 mV (21°C: V0.5 = −13.6 mV, k = −24.6 mV) or between −20 mV and +40 mV (35°C: V0.5 = 14.7 mV, k = −18.0 mV). To see this figure in color, go online.
Effect of temperature on Kv11.3 gating kinetics
We studied the Kv11.3 activation kinetics by means of envelope-of-tails protocols at 21°C and 30°C using different test pulse potentials (Fig. 7). The total charge of the inward tail currents was assumed to mirror the fraction of Kv11.3 channels that were activated during the course of the depolarizing pulse. Representative current traces with an overlay of the recordings obtained with test pulse potentials of 0 and 40 mV are illustrated in Fig. 7 A. Similarly to Kv11.1, the faster-activating Kv11.3 channels exhibited a sigmoidal time course of channel opening that was described by a delay and the time constant of activation. Both the delay and τact decreased with more positive potentials and elevated temperature (Fig. 7, B and C). For the four tested potentials, the resulting Q10 values amounted to 3.18 ± 0.10 for the delay and 3.73 ± 0.20 for τact. The difference in the temperature sensitivity of the two activation parameters was not significant (p = 0.053). The voltage dependence of τact could be well described by an exponential function declining to a mean asymptotic value of 19 ms at 21°C and to ∼4 ms at 30°C (Fig. S1, C and D).
Figure 7.
Effect of temperature and voltage on the time course of Kv11.3 channel activation. (A) Representative Kv11.3 currents recorded from different cells at 21°C and 30°C with envelope-of tails protocols. Please note the different timescales. The pulse protocol consisted of depolarizing pulses of increasing duration (2–354 ms at 21°C and 1–88 ms at 30°C), followed by a hyperpolarization to −100 mV to elicit tail currents. Four different potentials were used for the depolarizing pulse in every experiment, and the examples show an overlay of current traces recorded with test pulses to 0 mV (black traces) and 40 mV (gray traces). (B) Normalized tail charge values were averaged and plotted against the duration of the preceding depolarizing pulse. Data points obtained with longer pulse durations were fitted with single exponential functions, yielding values for the time constant of activation and a delay. Data are means ± SEM from experiments performed at 21°C (n = 12) or 30°C (n = 8). (C) Mean values (± SEM) of the delay and the time constant of activation as a function of the test pulse potential. To see this figure in color, go online.
We studied the time course of inactivation of Kv11.3 channels at 21°C, 30°C, and 35°C using a triple pulse protocol as shown in Fig. 8 A. In the potential range between 60 and 0 mV, the current decay during the P3 test pulses was fitted by single exponential functions to achieve the time constants of inactivation (τinact). Fig. 8 B shows the voltage dependence of τinact, demonstrating faster inactivation upon stronger depolarization at each temperature. Q10 values were calculated from the mean τinact values obtained for seven different P3 potentials at 21°C and 30°C, as well as at 21°C and 35°C. The Q10 values were similar for temperature differences of 9°C (Q10 = 2.73 ± 0.07) and 14°C (Q10 = 2.88 ± 0.04; p = 0.078).
Figure 8.
Temperature effects on the time course of Kv11.3 channel inactivation, recovery from inactivation, and deactivation. (A) Typical examples of Kv11.3 current traces recorded at 21°C and 30°C with triple pulse protocols designed to study inactivation rates. Starting from a holding potential of −80 mV, the potential was stepped for 500 ms to +80 mV (P1) to maximally activate and inactivate the channels. Subsequent 2-ms P2 pulses to −100 mV to open the Kv11.3 channels were followed by variable 300-ms test pulses to potentials between +60 and −20 mV (P3) to reinduce inactivation (see pulse protocol). (B) Means (± SEM) of time constants of inactivation (open symbols) and time constants of recovery from inactivation (solid symbols) plotted versus test pulse potential; the number of experiments is given in brackets. Time constants of inactivation were obtained from monoexponential fits to the decay phase of the current traces during the P3 pulse using current recordings as shown in (A). Time constants of recovery from inactivation and deactivation were obtained from experiments with fully activated Kv11.3 channels as shown in Fig. 6A. (C) Voltage and temperature dependence of Kv11.3 channel deactivation. Time constants of fast and slow deactivation, and the relative contribution of the fast deactivating current component as a function of the test pulse potential are shown. The number of experiments is given in brackets. To see this figure in color, go online.
The effect of temperature on recovery and deactivation kinetics was analyzed from experiments on fully activated Kv11.3 channels as shown in Fig. 6. Time constants of recovery from inactivation (τrecov) were obtained between 30 and −100 mV. Negative to −100 mV, recovery at higher temperature was too fast to be resolved from capacitive transients. τrecov exhibited a bell-shaped voltage dependence, with maximal values close to −20 mV at 21°C and ∼0 mV at 30°C and 35°C (Fig. 8 B). Due to the shift in the maximum of the bell-shaped curves, the Q10 values for τrecov were voltage dependent, with values close to 3 around +20 mV and well above 4 around −50 mV. For the voltage range between +30 and −70 mV, the mean Q10 value amounted to 4.00 ± 0.26 for the 9°C temperature difference. The mean Q10 value for τrecov calculated for the 14°C temperature difference was slightly smaller (3.73 ± 0.20, p = 0.002).
Fig. 8 C demonstrates the effect of temperature on Kv11.3 deactivation kinetics. At each temperature, the fast (τdeact,fast) and slow (τdeact,slow) time constants of deactivation decreased continuously with more negative membrane potentials. The fast deactivating current component clearly dominated in the analyzed voltage range and its relative contribution did not change systematically with temperature. The mean Q10 value for τdeact,fast amounted to 4.08 ± 0.07 (between −90 and −130 mV) for the 9°C temperature difference, and to 3.80 ± 0.07 (between −90 and −120 mV) for the 14°C temperature difference. For τdeact,slow, the mean Q10 value amounted to 3.43 ± 0.03 (between −90 and −130 mV) for the 9°C temperature variation, and to 3.74 ± 0.10 (between −90 and −120 mV) for the 14°C temperature variation. Thus, there was a significant difference between the Q10 values for τdeact,fast and τdeact,slow for the 9°C temperature difference (p < 0.001), but not for the 14°C difference (p = 0.266). Kv11.3 time constants obtained at different temperatures are listed in Table 2.
Comparison of temperature effects on the steady-state parameters and gating kinetics of Kv11.1a/1b and Kv11.3 channels
To illustrate the distinct effects of temperature on the open probability of Kv11.1a/1b and Kv11.3 channels, we calculated the voltage dependence of the steady-state open probability as a product of Kv11 activation and inactivation curves (Fig. 9, A and C). The maximal relative open probability of Kv11.1a/1b channels increased substantially from 0.233 (−18 mV) at room temperature to 0.503 (−26 mV) at near-physiological temperature. The steady-state open probability increased over the whole voltage range, as indicated by a broadening of the bell-shaped curve (Fig. 9 A). In contrast, the maximal steady-state open probability of Kv11.3 channels was already high at room temperature (0.578 at −30 mV) and increased more moderately with higher temperatures (30°C: 0.617 at −21 mV; 35°C: 0.715 at −11 mV). The concomitant shift in the voltage for peak open probability to more depolarized values with increased temperature resulted in slightly decreased open probabilities at potentials between −60 and −30 mV, but higher levels of occupancy of the open state in the voltage range between −20 and 60 mV.
Figure 9.
Comparison of temperature effects on the steady-state and kinetic parameters of Kv11.1a/1b and Kv11.3 channels. (A) Kv11.1a/1b steady-state open probability at 21°C and 35°C as a function of the membrane potential was calculated from mean parameters of the Boltzmann functions describing channel activation and inactivation (see Table 1). (B) Theoretical Kv11.1a/1b window current amplitudes normalized to the maximum current value at 21°C were calculated from the open probability data in (A) without (open symbols) or with (continuous lines) adjustment for a low temperature sensitivity of the single-channel conductance (using a Q10 of 1.3). Solid symbols are the experimentally determined values of the maximal steady-state current density normalized to the mean value at 21°C (means ± SEM; for absolute values and number of experiments, see Fig. 1B, inset). (C) Kv11.3 steady-state open probability at 21°C, 30°C, and 35°C as a function of the membrane potential was calculated from mean parameters of the Boltzmann functions describing channel activation and inactivation (see Table 2). (D) Theoretical Kv11.3 window current amplitudes normalized to the maximum current value at 21°C were calculated from the open probability data in (C) without (open symbols) or with (continuous lines) adjustment for a low temperature sensitivity of the single-channel conductance (using a Q10 of 1.3). Solid symbols are the experimentally determined values of the maximal steady-state current density normalized to the mean value at 21°C (means ± SEM; for absolute values and number of experiments, see Fig. 5B, inset). (E) Comparison of Q10 values determined for the Kv11.1a/1b and Kv11.3 gating parameters. Except for Kv11.3 channel activation, the average of the Q10 values obtained for the 9°C and 14°C temperature differences is given (see Tables 1 and 2). (F) Temperature dependence of the Gibbs free energy of activation at 0 mV (ΔG0) for Kv11.1a/1b and Kv11.3 channels (means ± SEM; values and number of experiments are given in Tables 1 and 2; the data point for Kv11.1a/1b at 21°C represents combined experiments of data sets A and B). To see this figure in color, go online.
The steady-state open probabilities were used to assess the window currents for Kv11.1a/1b (Fig. 9 B) and Kv11.3 channels (Fig. 9 D) with and without consideration of the effect of temperature on the single-channel conductance. To estimate the Q10 of Kv11 single-channel conductance, we compared theoretical current values with experimentally determined sustained current density values. At 35°C, the maximal theoretical Kv11.1a/1b current increased to 197% of the value at room temperature, but the experimentally determined increase in maximal current density amounted to 288% ± 35%. The use of a Q10 factor of 1.3 for single-channel conductance, corresponding to the Q10 of aqueous diffusion in this temperature range (37), adjusted the theoretical values to the experimentally determined data (Fig. 9 B). For Kv11.3, the maximal theoretical current values averaged 128% (30°C) and 175% (35°C) of the value at 21°C, whereas the measured maximal current density was increased to 164% ± 42% (30°C) and to 246% ± 37% (35°C) of the mean value at 21°C (Fig. 9 D). Considering the same Q10 factor of 1.3 for the single-channel conductance, the theoretical values again closely mirrored the experimentally observed increase in maximal current density with elevated temperature.
In contrast to the low temperature sensitivity of the single-channel conductance, the Kv11.1a/1b and Kv11.3 channel kinetics strongly depended on temperature (Fig. 9 E). A comparison of the respective Q10 values illustrates the extraordinary high temperature sensitivity of Kv11.1a/1b activation kinetics and the more uniform Q10 factors of Kv11.3 gating transitions. Nevertheless, with Q10 factors slightly below 3, the effect of temperature on the rate of Kv11.3 inactivation was also smaller than the temperature effect on Kv11.3 activation kinetics.
The differential effect of temperature on Kv11.1a/1b and Kv11.3 channel activation was further analyzed by determining the thermodynamic parameter ΔG0, the Gibbs free energy between the closed state and the open state at 0 mV (Tables 1 and 2). More negative values of ΔG0 are associated with leftward-shifted or steeper activation curves, whereas rightward shifts and shallower slopes of activation curves cause less negative ΔG0 values. As illustrated in Fig. 9 F, increases in temperature resulted in significantly more negative values of ΔG0 for Kv11.1a/1b, but significantly less negative values of ΔG0 for the Kv11.3 channel. These data indicate that an increase in temperature stabilizes the open state over the closed state of Kv11.1a/1b channels, and in contrast, higher temperature stabilizes the closed state over the open state of Kv11.3 channels.
Discussion
Until now, the temperature dependence of Kv11 channels had only been studied for homomeric Kv11.1a channels, which respond to a rise in temperature with an impressive increase in current density (34, 35). In this work, we comparably investigated the temperature sensitivity of heteromeric Kv11.1a/1b and homomeric Kv11.3 channels. The data obtained at room temperature and physiological temperature revealed similarities as well as unexpected distinct features in the thermal dependence of these two Kv11 channels. Both channels showed an overall high temperature sensitivity, with an increase in maximal current density and reduced inactivation at higher temperature. Compared with Kv11.3 channels, however, Kv11.1a/1b exhibited more pronounced variations in the Q10 temperature coefficients of the different gating kinetics. The Kv11.1a/1b and Kv11.3 channels showed a striking difference with regard to the effects of temperature on channel activation.
Kv11.1a/1b and Kv11.3 channels
The data we obtained for rat Kv11.1a/1b channels at 21°C, 30°C, and 35°C agree well with previous experiments performed on rat heteromeric Kv11.1a/1b channels at room temperature (14), as well as on human Kv11.1a/1b channels at more physiological temperatures (15). In accordance with previously published data on rat Kv11.3 currents (1, 3, 36, 38, 39, 40), our experiments demonstrate that Kv11.3 channels activate at room temperature in a more negative potential range than Kv11.1a/1b channels, and they exhibit considerably faster activation kinetics and deactivation kinetics compared with Kv11.1a/1b. The analysis of Kv11 channels is complicated by the fact that neither kinetic nor steady-state properties are directly accessible in the entire physiologically important potential range. Therefore, a number of different and specially adapted pulse protocols are used to assess the biophysical channel properties, and the resulting values strongly depend on the recording and evaluation conditions (4). In this study, we applied similar pulse protocols and analysis methods as far as possible to enable a direct comparison of the temperature sensitivities of Kv11.1a/1b and Kv11.3 channels.
Temperature effects on Kv11.1a/1b and Kv11.3 steady-state properties
Effects on Kv11 channel activation
The most impressive observation regarding the thermal dependence of Kv11.1a/1b and Kv11.3 channels was that an increase in temperature shifted their activation curves to opposite directions. At higher temperature, the Kv11.1a/1b activation curve was significantly shifted to the left and the slope factor tended to be smaller. These findings are similar to results obtained for homomeric Kv11.1a channels in studies covering a comparable temperature range (34, 35, 41, 42).
We determined just the opposite effects of temperature on Kv11.3 channels for both the midpoint and slope of the activation curve. At 35°C, the significant right shift in the voltage dependence of Kv11.3 channel activation resulted in less negative V0.5 values compared with Kv11.1a/1b. Thus, the characteristic difference in the voltage dependence of activation between rat Kv11.3 and Kv11.1 measured at room temperature (where Kv11.3 activates at more negative potentials compared with Kv11.1) disappeared and even became inverted with increasing temperature. In addition, the slope factor of the Kv11.3 activation curve significantly increased, pointing to a reduced voltage sensitivity of Kv11.3 activation at physiological temperature compared with 21°C. Despite the shift in the midpoint of the activation curve, elevated temperature had no considerable effect on Kv11.3 currents in the more negative potential range due to the decreased slope of the activation curve, compensating for the right shift at more negative potentials.
The inflection potentials of isochronal activation curves of Kv11 channels depend on the test pulse duration in relation to the activation kinetics of the relevant Kv11 channel (43). Therefore, accelerated activation can result in a left shift of the isochronal activation curve if test pulses are too short to allow channel activation to approach a steady state. Despite the faster activation kinetics of Kv11.1a/1b channels compared with Kv11.1a, the 4-s isochronal activation curves obtained at room temperature probably do not represent true steady-state properties, because a longer test pulse duration led to a significant left shift of the Kv11.1a/1b activation curve. Thus, it appears that part of the observed left shift in the voltage dependence of Kv11.1a/1b channel activation at increased temperature is due to the concomitant acceleration of activation kinetics. This interpretation could apply also for Kv11.1a channels, since temperature-induced shifts in Kv11.1a isochronal activation curves were more pronounced with 5-s test pulses (35, 41) than with 30-s test pulses (34). In our experiments with Kv11.3, we found no significant differences between 1-s and 4-s isochronal activation curves obtained at 30°C, due to the faster activation kinetics of Kv11.3 (1, 3, 38, 39). Thus, the observed right shift in the 4-s isochronal activation curves associated with higher temperature is suggested to represent a similar change in the true steady-state I-V relationship of Kv11.3 channel activation.
Temperature effects on Kv11 channel inactivation
In this study, we assessed the voltage dependence of steady-state inactivation by means of the fully activated I-V relationship. The conversion of current into relative conductance by dividing through the driving force requires a constant single channel conductance over the investigated voltage range which is strongly suggested for Kv11 channels by the linear I-V relationship of instantaneous Kv11 currents when studied with a triple pulse protocol to assess the time course of inactivation (3, 34, 44). This method did not yield practicable conductance values for Kv11.3 at more negative potentials due to intermingling deactivation. Nevertheless, the remaining conductance data covered a voltage range in which the degree of channel inactivation changed substantially, allowing convincing fits to the data. The validity of the Kv11.3 steady-state inactivation parameters determined in this way was confirmed by a comparison of calculated and measured maximal steady-state outward currents (see below and Fig. 9 D).
Both Kv11.1a/1b channels and Kv11.3 channels exhibit reduced inactivation compared with Kv11.1a (3, 14, 38). Results obtained with N-terminal deletion mutants show that the Kv11.1a N-terminus can not only slow deactivation but also promote C-type inactivation (11), explaining the differences between the heteromeric and homomeric Kv11.1 channels. The mechanism of Kv11.3 channel inactivation, however, has not been studied in detail. Our results show that a rise in temperature markedly reduced steady-state inactivation of both Kv11.1a/1b and Kv11.3 channels. The significant shift of the inactivation curves to more positive potentials amounted to ∼25 mV for Kv11.1a/1b and to 30 mV for Kv11.3, considering data obtained at 35°C versus 21°C. In addition, the slope factors were slightly reduced for both channel types, indicating an overall similar thermal dependence of Kv11.1a/1b and Kv11.3 inactivation. These data agree well with the effects of temperature on Kv11.1a channels, where significantly less inactivation has been found at higher temperatures (34).
Effect of temperature on window currents and current density
The characteristic bell-shaped I-V relationship of Kv11 channels results from voltage-dependent activation and inactivation of the channels. The product of the level of steady-state activation and inactivation can serve as a measure of the relative steady-state open probability of the channels at a given voltage (Fig. 9, A and C). At room temperature, the calculated maximal steady-state open probability was much higher for Kv11.3 than for Kv11.1a/1b, in agreement with the view of Kv11.3 as the weakest functional inward rectifier in the family of Kv11 channels (1, 2). At elevated temperature, the maximal channel open probability markedly increased for Kv11.1a/1b and moderately increased for Kv11.3, resulting in a more similar window conductance of the two channels at physiological temperature. The theoretical values of the window currents, calculated as the product of the activation curve, inactivation curve, and driving force, should mirror the voltage dependence of the measured sustained outward currents. Indeed, the voltage values for the maxima of the calculated and measured bell-shaped I-V curves are in almost perfect agreement for Kv11.3, including the shift to more depolarized values with elevated temperature, and differed by less than 10 mV for Kv11.1a/1b, suggesting that the parameters determined for channel activation and inactivation adequately mirror the voltage dependence of both Kv11 channels. The theoretical I-V curves also demonstrate that the effect of temperature on the voltage dependence of steady-state activation and inactivation can explain the enhanced Kv11.1a/1b current density at physiological temperature over the whole voltage range with concomitant broadening of the bell-shaped I-V curve as well as the enhancement of Kv11.3 current density only at more depolarized potentials. Using the same low Q10 factor of 1.3 for Kv11.1a/1b and Kv11.3 single-channel conductance, the theoretical data fit to the experimentally observed higher maximal current densities found for the Kv11.1a/1b and Kv11.3 channels at higher temperature (Fig. 9, B and D). A similar Q10 factor of 1.4 was determined by Vandenberg et al. (34) for Kv11.1a channel conductance. In general, the thermal dependence of ion channel conductance with Q10 temperature coefficients in the range of 1.2–1.5 is very low compared with the temperature sensitivity of channel gating kinetics (37).
Temperature sensitivity of Kv11 channel kinetics
The Q10 temperature coefficient can be used to quantify the thermal dependence of channel gating kinetics. In this work, we compared data recorded at room temperature (21°C) with data recorded at more physiological temperatures (30°C and/or 35°C). The Q10 values calculated for temperature differences of 9°C and 14°C were overall similar (see Tables 1 and 2), with no indications of a heterogeneous thermal dependence in this temperature range.
By far the most temperature-sensitive gating process was the activation of Kv11.1a/1b channels (see Fig. 9 E). The pronounced differences between the Q10 factors for Kv11.1a/1b and Kv11.3 activation kinetics agree with the divergent effect of temperature on the voltage dependence of activation of these channels. For Kv11.1a/1b, the next most sensitive gating step was the rate of recovery from inactivation, followed by slightly lower values for inactivation and deactivation kinetics. Kv11.3 channel gating also exhibited a high, but more uniform, temperature sensitivity. Nonetheless, the rate of inactivation had a lower thermal dependence than the other gating steps of Kv11.3. We used two different parameters to describe the channel activation kinetics and account for the sigmoidal time course of the Kv11 tail currents. The observed latency in channel activation has been attributed to the passage through multiple closed states before the final channel opening (e.g., (34, 45)). The time constant of activation (τact) characterizes the slowest step in a linear activation pathway, whereas the delay d reflects the transit through the other parts of the activation pathway (46). Interestingly, a Kv11.1a/1b gating model assigned the slowest forward step to the final closed-open transition (15). Different Kv11.1 gating models implied (at least) three closed states before the open state and a voltage-insensitive step between two adjacent closed states (15, 45, 47). The existence of a voltage-insensitive step was suggested by the observation that values of τact exponentially decrease with more positive test potentials to an asymptotic value that clearly differs from zero (45, 47). This asymptotic value is assumed to be a measure of the forward time constant of the voltage-insensitive closed-closed transition, which becomes the rate-limiting step at extremely positive potentials. Our results (Fig. S1) agree well with the existence of such a voltage-insensitive step in the activation pathway of both Kv11.1a/1b and Kv11.3 channels, and they also hint at a very high temperature sensitivity of this voltage-insensitive transition.
Our data regarding the thermal dependence of Kv11.1a/1b gating transitions are largely in line with the temperature effects on homomeric human Kv11.1a channels reported by Zhou et al. (35). The Q10 factors calculated from the listed values for 23°C and 35°C are 6.25 (activation; 0 mV), 3.55 (inactivation; 0 mV), 3.65 (recovery from inactivation; −50 mV), 1.36 (fast deactivation; −50 mV), and 1.25 (slow deactivation). Thus, by far the greatest effect of temperature was found for the rate of Kv11.1a current activation. In contrast, the temperature sensitivities of the deactivation processes differed strikingly between the two Kv11.1 channels. Our data show an increasing effect of temperature on Kv11.1a/1b deactivation kinetics with more negative potentials. Since the voltage dependence of the relative contribution of the fast deactivating current component changed with temperature, we also determined weighted time constants. Considering the Q10 values at −50 mV instead of −100 mV, the rate of Kv11.1a/1b channel deactivation exhibited the smallest temperature sensitivity of all gating transitions. Nevertheless, Q10 factors well above 2 for the Kv11.1a/1b deactivation process (Table 1) reveal a distinction compared with homomeric Kv11.1a channels. The most remarkable difference between the two Kv11.1 isoforms (1a and 1b) is found in their deactivation kinetics, and it is tempting to speculate that the presence of the four N-termini of the homomeric Kv11.1a channel causes the low thermal dependence of its rate of deactivation. At more negative potentials, Kv11.3 channels exhibit faster deactivation kinetics with a higher temperature sensitivity than Kv11.1a/b channels. This might indicate that the Kv11.3 N-terminus plays a different role in channel deactivation despite its homology to the Kv11.1a N-terminus (1). As observed previously (38), the relative contribution of the fast deactivating Kv11.3 current component was uniformly high in the analyzed negative voltage range. This ratio did not change with temperature, but we cannot exclude effects of temperature at less negative potentials.
Differences in the temperature dependence of ion channels
Temperature-sensitive ion channels have been classified into heat- or cold-sensing channels depending on whether a rise or a decrease in temperature stabilizes the open over the closed state of the channel (48, 49). Surprisingly, Kv11.1a/1b and Kv11.3 exhibited a qualitatively similar difference concerning channel activation. The temperature sensitivity of an ion channel can be based on the channel itself or on the temperature sensitivity of another factor that regulates the activity of the channel (48). A contribution of the latter mechanism is described for the cardiac delayed rectifier current IKs. Similarly to Kv11.1 channels underlying IKr, IKs has a high temperature sensitivity of the rate of current activation (50). Subsequent work showed that the β-subunit KCNE confers this high temperature sensitivity to the Kv7.1-KCNE1 channel complex (51). In this study, we investigated Kv11.1a/1b and Kv11.3 channels in the same expression system under identical experimental conditions. This might indicate that the observed difference in the effect of temperature on channel activation is directly based on the two channel proteins.
A number of ion channels display some temperature sensitivity (49), but thermosensitive TRP channels exhibit an unusually large temperature dependence. Several attempts have been made to identify thermosensitive domains in TRP channels. Candidate regions so far identified are spread over most parts of the channel, and it has been suggested that thermosensing might involve several small rearrangements in various parts of the channel (reviewed in (48)). This assumption could well apply to Kv11.1a/1b and Kv11.3 channels, and could explain the differences in their temperature dependence despite the high homology of structurally important subunit regions (1). Interestingly, the closely related Kv10.1 channel seems to be almost temperature insensitive, since in one study, current amplitudes were unaffected by changes in temperature and the Q10 of the activation kinetics amounted to 1.4 (52). In line with the idea that thermosensation is not mediated by a specialized channel region, a recently proposed model suggests that temperature sensitivity arises from changes in heat capacity during channel gating (53). Heat capacity changes are normally associated with changes in the solvation states of hydrophobic residues. Thus, altering the hydrophobicity of a few residues may influence the temperature dependence of an ion channel or even confer thermosensitivity to an otherwise thermo-insensitive channel, as demonstrated in a recent study (49). That study suggested that temperature dependence relies on channel-gating-associated movements of hydrophobic residues between buried and solvent-exposed environments. Kv11.1a (and thus Kv11.1b) and Kv11.3 subunits exhibit highly conserved transmembrane segments without significant differences in hydrophobicity. Nevertheless, cytoplasmic regions participate in Kv11 channel gating (54) and might mediate a divergent temperature dependence.
The temperature sensitivity of an ionic current is mainly reflected by the temperature effects on channel gating. Nevertheless, whereas elevated temperature strongly favors the open state of Kv11.1 channels, it can exert the opposite effect on Kv11.1 channel trafficking (55). Therefore, the net effect of temperature on macroscopic Kv11 current amplitudes might well change during a longer period of altered temperature.
Another aspect of the temperature dependence of an ion channel is its potential impact on the efficacy of drugs that preferentially bind to a certain channel state. For Kv11.1a, it is already known that the channel-blocking activity of some drugs varies with temperature (56, 57). Our results suggest that changes in temperature can also influence the susceptibility to pharmacological manipulation of Kv11.1a/1b channels and Kv11.3 channels.
Physiological context
An abnormally high or low body temperature can lead to possibly life-threatening neurologic dysfunction (58) and can also affect the length and shape of cardiac action potentials. Interestingly, a Kv11.1a missense mutation associated with fever-induced ventricular arrhythmias has been described (41), suggesting that cardiac Kv11.1 channels normally mediate a QTc shortening with elevated temperatures. This requires a high temperature sensitivity and an increase in the occupancy of the open state of the Kv11.1 channels at elevated temperature. This was previously shown for Kv11.1a channels and has now been demonstrated for heteromeric Kv11.1a/1b channels. Jones et al. (22) showed that the contribution of the 1b subunit to functional Kv11.1 channels substantially increased IKr current amplitudes during human cardiac action potentials. These results were obtained at physiological temperature, and our data strongly suggest that the high temperature sensitivity of the rate of Kv11.1a/1b channel activation, as well as the reduced steady-state inactivation, contributes to this observed current increase. We previously showed that Kv11.1a/1b channels are able to contribute even to the much shorter rat cardiac action potentials when studied at elevated temperatures (17). In the human heart, Kv11.1b is less abundant than Kv11.1a, and the relative Kv11.1b expression is lower in ventricles than in atria (59). Nevertheless, the impact of ventricular 1b subunits might increase in disease, as suggested by the recently reported relative upregulation of Kv11.1b protein in failing human hearts (60). In this context, the higher thermal dependence of the rate of deactivation of heteromeric Kv11.1a/1b channels might be unfavorable at elevated temperatures because it further reduces the time period in which the channels can serve as a repolarization reserve to counteract early afterdepolarizations.
The physiological role of the different Kv11 channels in the nervous system is still a matter of investigation. There are a few hints regarding their functional role in the human brain, as KCNH2 (encoding Kv11.1) and KCNH7 (encoding Kv11.3) have been suggested as candidate genes in schizophrenia (61) and bipolar disorders (62), respectively. Our current knowledge about a more detailed function of Kv11 channels in the nervous system is based entirely on animal experiments, which suggest that they play a role in the control and modulation of electrical excitability (for reviews, see (4, 33, 63)). The apparent expansion of Kv11.1a/1b and Kv11.3 window conductance in the depolarized voltage range due to reduced inactivation at elevated temperatures should favor a contribution of native Kv11 channels to the phenomenon of frequency accommodation that is observed in many neurons (e.g., (30)). Specific block of Kv11 channels can result in membrane depolarization, indicating that neuronal Kv11 channels are already activated in the subthreshold voltage range (e.g., (31)). Our results suggest that both Kv11.1a/1b and Kv11.3 channels are able to contribute to the resting membrane potential at physiological temperatures. Similarly to Kv11.1a (34), Kv11.1a/1b channels attain and boost this property with increased temperature. In contrast, Kv11.3 channels mediate steady-state outward current in the negative voltage range at room temperature and just retain a comparable current level at elevated temperatures. In the rat brain, some regions exhibit a dominant expression of one Kv11 subunit, whereas other regions show a strong overlapping distribution (27). Different Kv11 channel subunits may be present in a single cell (13), enabling the formation of heteromeric Kv11 channels with mainly intermediate properties (39, 64). Our results indicate that the effect of temperature on native Kv11 channels and their contribution to excitability might differ with the ratio of the expressed Kv11 channel subunits. In the heart and even more so in the brain, it is challenging to determine the molecular composition of native Kv11 channels, and it is difficult to record endogenous Kv11 currents under physiological conditions. Our data on heterologously expressed Kv11.1a/1b and Kv11.3 channels can aid in assessing the physiological role of native Kv11 currents using data recorded at room temperature.
Conclusions
We have shown that Kv11.1a/1b and Kv11.3 current amplitudes and the relevant voltage windows for sustained Kv11 current critically depend on temperature. Our analysis of the underlying changes in the biophysical properties of the Kv11 channels revealed differences in the thermal sensitivity of the different gating transitions of each channel, as well as unexpected differences between the two Kv11 channels concerning the effect of temperature on channel activation. The results show the difficulty of deducing a physiological channel function from recordings obtained at room temperature. The data provided here can be used to design models that will help elucidate the role of Kv11 channels in excitable cells.
Author Contributions
C.K.B. designed the study and wrote the manuscript. M.M. and C.K.B. performed and analyzed the experiments.
Acknowledgments
We thank Günter Glassmeier, Annett Hasse, Margrit Hölzel, and Telse Kock for their valuable support in the lab, and Robert Bähring for discussion.
Editor: Chris Lingle.
Footnotes
One figure is available at http://www.biophysj.org/biophysj/supplemental/S0006-3495(16)30516-1.
Supporting Material
References
- 1.Shi W., Wymore R.S., Dixon J.E. Identification of two nervous system-specific members of the erg potassium channel gene family. J. Neurosci. 1997;17:9423–9432. doi: 10.1523/JNEUROSCI.17-24-09423.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bauer C.K., Schwarz J.R. Physiology of EAG K+ channels. J. Membr. Biol. 2001;182:1–15. doi: 10.1007/s00232-001-0031-3. [DOI] [PubMed] [Google Scholar]
- 3.Sturm P., Wimmers S., Bauer C.K. Extracellular potassium effects are conserved within the rat erg K+ channel family. J. Physiol. 2005;564:329–345. doi: 10.1113/jphysiol.2004.078840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vandenberg J.I., Perry M.D., Hill A.P. hERG K+ channels: structure, function, and clinical significance. Physiol. Rev. 2012;92:1393–1478. doi: 10.1152/physrev.00036.2011. [DOI] [PubMed] [Google Scholar]
- 5.Sanguinetti M.C., Jurkiewicz N.K. Two components of cardiac delayed rectifier K+ current. Differential sensitivity to block by class III antiarrhythmic agents. J. Gen. Physiol. 1990;96:195–215. doi: 10.1085/jgp.96.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lees-Miller J.P., Kondo C., Duff H.J. Electrophysiological characterization of an alternatively processed ERG K+ channel in mouse and human hearts. Circ. Res. 1997;81:719–726. doi: 10.1161/01.res.81.5.719. [DOI] [PubMed] [Google Scholar]
- 7.London B., Trudeau M.C., Robertson G.A. Two isoforms of the mouse ether-a-go-go-related gene coassemble to form channels with properties similar to the rapidly activating component of the cardiac delayed rectifier K+ current. Circ. Res. 1997;81:870–878. doi: 10.1161/01.res.81.5.870. [DOI] [PubMed] [Google Scholar]
- 8.Schönherr R., Heinemann S.H. Molecular determinants for activation and inactivation of HERG, a human inward rectifier potassium channel. J. Physiol. 1996;493:635–642. doi: 10.1113/jphysiol.1996.sp021410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Spector P.S., Curran M.E., Sanguinetti M.C. Fast inactivation causes rectification of the IKr channel. J. Gen. Physiol. 1996;107:611–619. doi: 10.1085/jgp.107.5.611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Viloria C.G., Barros F., de la Peña P. Differential effects of amino-terminal distal and proximal domains in the regulation of human erg K+ channel gating. Biophys. J. 2000;79:231–246. doi: 10.1016/S0006-3495(00)76286-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang J., Trudeau M.C., Robertson G.A. Regulation of deactivation by an amino terminal domain in human ether-à-go-go-related gene potassium channels. J. Gen. Physiol. 1998;112:637–647. doi: 10.1085/jgp.112.5.637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Trudeau M.C., Leung L.M., Robertson G.A. hERG1a N-terminal eag domain-containing polypeptides regulate homomeric hERG1b and heteromeric hERG1a/hERG1b channels: a possible mechanism for long QT syndrome. J. Gen. Physiol. 2011;138:581–592. doi: 10.1085/jgp.201110683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hirdes W., Schweizer M., Schwarz J.R. Fast erg K+ currents in rat embryonic serotonergic neurones. J. Physiol. 2005;564:33–49. doi: 10.1113/jphysiol.2004.082123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kirchberger N.M., Wulfsen I., Bauer C.K. Effects of TRH on heteromeric rat erg1a/1b K+ channels are dominated by the rerg1b subunit. J. Physiol. 2006;571:27–42. doi: 10.1113/jphysiol.2005.101667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sale H., Wang J., Robertson G.A. Physiological properties of hERG 1a/1b heteromeric currents and a hERG 1b-specific mutation associated with Long-QT syndrome. Circ. Res. 2008;103:e81–e95. doi: 10.1161/CIRCRESAHA.108.185249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Du C.Y., El Harchi A., Hancox J.C. Enhanced inhibitory effect of acidosis on hERG potassium channels that incorporate the hERG1b isoform. Biochem. Biophys. Res. Commun. 2011;405:222–227. doi: 10.1016/j.bbrc.2011.01.014. [DOI] [PubMed] [Google Scholar]
- 17.Mewe M., Mauerhöfer M., Bauer C.K. Modulation of cardiac ERG1 K+ channels by cGMP signaling. J. Mol. Cell. Cardiol. 2010;49:48–57. doi: 10.1016/j.yjmcc.2010.02.015. [DOI] [PubMed] [Google Scholar]
- 18.Abi-Gerges N., Holkham H., Robertson G.A. hERG subunit composition determines differential drug sensitivity. Br. J. Pharmacol. 2011;164(2b):419–432. doi: 10.1111/j.1476-5381.2011.01378.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Larsen A.P., Bentzen B.H., Grunnet M. Differential effects of Kv11.1 activators on Kv11.1a, Kv11.1b and Kv11.1a/Kv11.1b channels. Br. J. Pharmacol. 2010;161:614–628. doi: 10.1111/j.1476-5381.2010.00897.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schuster A.M., Glassmeier G., Bauer C.K. Strong activation of ether-à-go-go-related gene 1 K+ channel isoforms by NS1643 in human embryonic kidney 293 and Chinese hamster ovary cells. Mol. Pharmacol. 2011;80:930–942. doi: 10.1124/mol.111.071621. [DOI] [PubMed] [Google Scholar]
- 21.Jonsson M.K., van der Heyden M.A., van Veen T.A. Deciphering hERG channels: molecular basis of the rapid component of the delayed rectifier potassium current. J. Mol. Cell. Cardiol. 2012;53:369–374. doi: 10.1016/j.yjmcc.2012.06.011. [DOI] [PubMed] [Google Scholar]
- 22.Jones E.M., Roti Roti E.C., Robertson G.A. Cardiac IKr channels minimally comprise hERG 1a and 1b subunits. J. Biol. Chem. 2004;279:44690–44694. doi: 10.1074/jbc.M408344200. [DOI] [PubMed] [Google Scholar]
- 23.Jones D.K., Liu F., Robertson G.A. hERG 1b is critical for human cardiac repolarization. Proc. Natl. Acad. Sci. USA. 2014;111:18073–18077. doi: 10.1073/pnas.1414945111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ohya S., Horowitz B., Greenwood I.A. Functional and molecular identification of ERG channels in murine portal vein myocytes. Am. J. Physiol. Cell Physiol. 2002;283:C866–C877. doi: 10.1152/ajpcell.00099.2002. [DOI] [PubMed] [Google Scholar]
- 25.Guasti L., Cilia E., Arcangeli A. Expression pattern of the ether-a-go-go-related (ERG) family proteins in the adult mouse central nervous system: evidence for coassembly of different subunits. J. Comp. Neurol. 2005;491:157–174. doi: 10.1002/cne.20721. [DOI] [PubMed] [Google Scholar]
- 26.Arcangeli A. Expression and role of hERG channels in cancer cells. Novartis Found. Symp. 2005;266:225–232. discussion 232–234. [PubMed] [Google Scholar]
- 27.Saganich M.J., Machado E., Rudy B. Differential expression of genes encoding subthreshold-operating voltage-gated K+ channels in brain. J. Neurosci. 2001;21:4609–4624. doi: 10.1523/JNEUROSCI.21-13-04609.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Papa M., Boscia F., Taglialatela M. Expression pattern of the ether-a-gogo-related (ERG) K+ channel-encoding genes ERG1, ERG2, and ERG3 in the adult rat central nervous system. J. Comp. Neurol. 2003;466:119–135. doi: 10.1002/cne.10886. [DOI] [PubMed] [Google Scholar]
- 29.Barros F., del Camino D., de la Peña P. Caffeine enhancement of electrical activity through direct blockade of inward rectifying K+ currents in GH3 rat anterior pituitary cells. Pflugers Arch. 1996;431:443–451. doi: 10.1007/BF02207284. [DOI] [PubMed] [Google Scholar]
- 30.Chiesa N., Rosati B., Wanke E. A novel role for HERG K+ channels: spike-frequency adaptation. J. Physiol. 1997;501:313–318. doi: 10.1111/j.1469-7793.1997.313bn.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hirdes W., Napp N., Bauer C.K. Erg K+ currents modulate excitability in mouse mitral/tufted neurons. Pflugers Arch. 2009;459:55–70. doi: 10.1007/s00424-009-0709-4. [DOI] [PubMed] [Google Scholar]
- 32.Niculescu D., Hirdes W., Schwarz J.R. Erg potassium currents of neonatal mouse Purkinje cells exhibit fast gating kinetics and are inhibited by mGluR1 activation. J. Neurosci. 2013;33:16729–16740. doi: 10.1523/JNEUROSCI.5523-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schwarz J.R., Bauer C.K. Functions of erg K+ channels in excitable cells. J. Cell. Mol. Med. 2004;8:22–30. doi: 10.1111/j.1582-4934.2004.tb00256.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vandenberg J.I., Varghese A., Huang C.L. Temperature dependence of human ether-a-go-go-related gene K+ currents. Am. J. Physiol. Cell Physiol. 2006;291:C165–C175. doi: 10.1152/ajpcell.00596.2005. [DOI] [PubMed] [Google Scholar]
- 35.Zhou Z., Gong Q., January C.T. Properties of HERG channels stably expressed in HEK 293 cells studied at physiological temperature. Biophys. J. 1998;74:230–241. doi: 10.1016/S0006-3495(98)77782-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bilet A., Bauer C.K. Effects of the small molecule HERG activator NS1643 on Kv11.3 channels. PLoS One. 2012;7:e50886. doi: 10.1371/journal.pone.0050886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hille B. Sinauer Associates; Sunderland, MA: 2001. Ion Channels of Excitable Membranes. [Google Scholar]
- 38.Schledermann W., Wulfsen I., Bauer C.K. Modulation of rat erg1, erg2, erg3 and HERG K+ currents by thyrotropin-releasing hormone in anterior pituitary cells via the native signal cascade. J. Physiol. 2001;532:143–163. doi: 10.1111/j.1469-7793.2001.0143g.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wimmers S., Bauer C.K., Schwarz J.R. Biophysical properties of heteromultimeric erg K+ channels. Pflugers Arch. 2002;445:423–430. doi: 10.1007/s00424-002-0936-4. [DOI] [PubMed] [Google Scholar]
- 40.Restano-Cassulini R., Korolkova Y.V., Wanke E. Species diversity and peptide toxins blocking selectivity of ether-a-go-go-related gene subfamily K+ channels in the central nervous system. Mol. Pharmacol. 2006;69:1673–1683. doi: 10.1124/mol.105.019729. [DOI] [PubMed] [Google Scholar]
- 41.Amin A.S., Herfst L.J., Wilde A.A. Fever-induced QTc prolongation and ventricular arrhythmias in individuals with type 2 congenital long QT syndrome. J. Clin. Invest. 2008;118:2552–2561. doi: 10.1172/JCI35337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang J., Della Penna K., Salata J.J. Functional and pharmacological properties of canine ERG potassium channels. Am. J. Physiol. Heart Circ. Physiol. 2003;284:H256–H267. doi: 10.1152/ajpheart.00220.2002. [DOI] [PubMed] [Google Scholar]
- 43.Schönherr R., Rosati B., Wanke E. Functional role of the slow activation property of ERG K+ channels. Eur. J. Neurosci. 1999;11:753–760. doi: 10.1046/j.1460-9568.1999.00493.x. [DOI] [PubMed] [Google Scholar]
- 44.Smith P.L., Baukrowitz T., Yellen G. The inward rectification mechanism of the HERG cardiac potassium channel. Nature. 1996;379:833–836. doi: 10.1038/379833a0. [DOI] [PubMed] [Google Scholar]
- 45.Wang S., Liu S., Rasmusson R.L. A quantitative analysis of the activation and inactivation kinetics of HERG expressed in Xenopus oocytes. J. Physiol. 1997;502:45–60. doi: 10.1111/j.1469-7793.1997.045bl.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Schoppa N.E., Sigworth F.J. Activation of shaker potassium channels. I. Characterization of voltage-dependent transitions. J. Gen. Physiol. 1998;111:271–294. doi: 10.1085/jgp.111.2.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gómez-Varela D., de la Peña P., Barros F. Influence of amino-terminal structures on kinetic transitions between several closed and open states in human erg K+ channels. J. Membr. Biol. 2002;187:117–133. doi: 10.1007/s00232-001-0156-4. [DOI] [PubMed] [Google Scholar]
- 48.Voets T. TRP channels and thermosensation. Handbook Exp. Pharmacol. 2014;223:729–741. doi: 10.1007/978-3-319-05161-1_1. [DOI] [PubMed] [Google Scholar]
- 49.Chowdhury S., Jarecki B.W., Chanda B. A molecular framework for temperature-dependent gating of ion channels. Cell. 2014;158:1148–1158. doi: 10.1016/j.cell.2014.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Busch A.E., Maylie J. Effects of calcium buffers and temperature on the β-receptor-mediated regulation of the slowly activating potassium current (IKs) in guinea pig cardiac myocytes. Cell. Physiol. Biochem. 1994;4:1–9. [Google Scholar]
- 51.Unsöld B., Kerst G., Bleich M. KCNE1 reverses the response of the human K+ channel KCNQ1 to cytosolic pH changes and alters its pharmacology and sensitivity to temperature. Pflugers Arch. 2000;441:368–378. doi: 10.1007/s004240000434. [DOI] [PubMed] [Google Scholar]
- 52.Mortensen L.S., Schmidt H., Pardo L.A. KV 10.1 opposes activity-dependent increase in Ca2+ influx into the presynaptic terminal of the parallel fibre-Purkinje cell synapse. J. Physiol. 2015;593:181–196. doi: 10.1113/jphysiol.2014.281600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Clapham D.E., Miller C. A thermodynamic framework for understanding temperature sensing by transient receptor potential (TRP) channels. Proc. Natl. Acad. Sci. USA. 2011;108:19492–19497. doi: 10.1073/pnas.1117485108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Morais-Cabral J.H., Robertson G.A. The enigmatic cytoplasmic regions of KCNH channels. J. Mol. Biol. 2015;427:67–76. doi: 10.1016/j.jmb.2014.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Chen M.X., Sandow S.L., Clare J.J. Improved functional expression of recombinant human ether-a-go-go (hERG) K+ channels by cultivation at reduced temperature. BMC Biotechnol. 2007;7:93. doi: 10.1186/1472-6750-7-93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kirsch G.E., Trepakova E.S., Brown A.M. Variability in the measurement of hERG potassium channel inhibition: effects of temperature and stimulus pattern. J. Pharmacol. Toxicol. Methods. 2004;50:93–101. doi: 10.1016/j.vascn.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 57.Yao J.A., Du X., Atterson P. Estimation of potency of HERG channel blockers: impact of voltage protocol and temperature. J. Pharmacol. Toxicol. Methods. 2005;52:146–153. doi: 10.1016/j.vascn.2005.04.008. [DOI] [PubMed] [Google Scholar]
- 58.Gomez C.R. Disorders of body temperature. Handb. Clin. Neurol. 2014;120:947–957. doi: 10.1016/B978-0-7020-4087-0.00062-0. [DOI] [PubMed] [Google Scholar]
- 59.Larsen A.P., Olesen S.P., Jespersen T. Characterization of hERG1a and hERG1b potassium channels-a possible role for hERG1b in the IKr current. Pflugers Arch. 2008;456:1137–1148. doi: 10.1007/s00424-008-0476-7. [DOI] [PubMed] [Google Scholar]
- 60.Holzem K.M., Gomez J.F., Efimov I.R. Reduced response to IKr blockade and altered hERG1a/1b stoichiometry in human heart failure. J. Mol. Cell. Cardiol. 2016;96:82–92. doi: 10.1016/j.yjmcc.2015.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Huffaker S.J., Chen J., Weinberger D.R. A primate-specific, brain isoform of KCNH2 affects cortical physiology, cognition, neuronal repolarization and risk of schizophrenia. Nat. Med. 2009;15:509–518. doi: 10.1038/nm.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Strauss K.A., Markx S., Puffenberger E.G. A population-based study of KCNH7 p.Arg394His and bipolar spectrum disorder. Hum. Mol. Genet. 2014;23:6395–6406. doi: 10.1093/hmg/ddu335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Babcock J.J., Li M. hERG channel function: beyond long QT. Acta Pharmacol. Sin. 2013;34:329–335. doi: 10.1038/aps.2013.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wimmers S., Wulfsen I., Schwarz J.R. Erg1, erg2 and erg3 K channel subunits are able to form heteromultimers. Pflugers Arch. 2001;441:450–455. doi: 10.1007/s004240000467. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.