Abstract
Physiological and degenerative changes affecting human standing balance are major contributors to falls with ageing. During imbalance, stepping is a powerful protective action for preserving balance that may be voluntarily initiated in recognition of a balance threat, or be induced by an externally imposed mechanical or sensory perturbation. Paradoxically, with ageing and falls, initiation slowing of voluntary stepping is observed together with perturbation‐induced steps that are triggered as fast as or faster than for younger adults. While age‐associated changes in sensorimotor conduction, central neuronal processing and cognitive functions are linked to delayed voluntary stepping, alterations in the coupling of posture and locomotion may also prolong step triggering. It is less clear, however, how these factors may explain the accelerated triggering of induced stepping. We present a conceptual model that addresses this issue. For voluntary stepping, a disruption in the normal coupling between posture and locomotion may underlie step‐triggering delays through suppression of the locomotion network based on an estimation of the evolving mechanical state conditions for stability. During induced stepping, accelerated step initiation may represent an event‐triggering process whereby stepping is released according to the occurrence of a perturbation rather than to the specific sensorimotor information reflecting the evolving instability. In this case, errors in the parametric control of induced stepping and its effectiveness in stabilizing balance would be likely to occur. We further suggest that there is a residual adaptive capacity with ageing that could be exploited to improve paradoxical triggering and other changes in protective stepping to impact fall risk.
Abbreviations
- APA
anticipatory postural adjustment
- BoS
base of support
- CoM
centre of mass
- CoP
centre of pressure
- CRT
choice reaction time
- M‐L
medio‐lateral
- RT
reaction time
- WMH
white matter hyperintensity
Introduction
The inherently unstable nature of human standing requires postural motor actions for body orientation and balance stabilization that must be effectively integrated with other goal‐directed actions to allow safe and efficient movement function. During actual or anticipated loss of balance, multi‐segmental postural movements of the limbs such as stepping are commonly executed, and represent powerful protective actions for preserving balance by reconfiguring the body centre of mass (CoM)–base of support (BoS) relationship. This requires that the motion of stepping limb be appropriately controlled to match the ongoing motion of the CoM. Stepping may be voluntarily initiated proactively in recognition of an impending threat to balance, or externally induced by a mechanical or sensory perturbation. Importantly, the ability to step quickly is a critical factor in arresting a fall (van den Bogert et al. 2002).
With older age, physiological and degenerative changes involving multiple systems contributing to standing balance are major risk factors for falls and mobility disability. These changes are manifested as impairments in several motor control functions that compromise protective stepping behaviour, effective stabilization of balance, and fall avoidance (Tinetti et al. 1988; Rogers & Mille, 2003; Lord et al. 2007). In this symposium review, we will address ageing deficits in only one of these control functions, the triggering of protective stepping.
Triggering voluntary stepping
A pedestrian standing at ease or walking on urban terrain may quickly sidestep to avoid an anticipated collision from oncoming pedestrian traffic that may destabilize balance or cause injury; one may step to extend their base of support and guard against an expected threat to balance; another may suddenly back step onto a curb when the crosswalk stop signal flashes to pre‐empt being flattened by vehicular traffic. Whereas voluntary step initiation or gait initiation may be viewed as actions that accomplish locomotion transport, a voluntary ‘protective step’ is internally triggered in order to avoid a perturbation of balance by altering the CoM–BoS relationship to increase stability, or to avoid injury that might be imminent or is anticipated. The initiation of such proactive volitional stepping often involves motor planning and includes active changes in the CoM–BoS configuration that precede stepping through a sequence of muscle activations, joint moments and corresponding changes in the ground reaction forces and centre of pressure (CoP). These motor actions propel the CoM in the direction of stepping and towards the single stance limb for weight transfer and medio‐lateral (M‐L) stability prior to stepping (Carlsoo, 1966; Breniere et al. 1987; Mille et al. 2003). Hence, anticipatory postural adjustments (APAs) precede and accompany the initiation of stepping and are adaptively scaled in amplitude and duration in accordance with the distance and speed of the first step (Breniere et al. 1987).
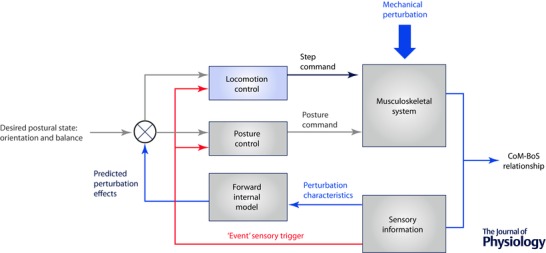
The anticipatory nature of the postural‐step coupling appears to involve a role for motor prediction in coordination. In an attempt to explain this coupling, we formulated a conceptual framework based on a forward internal model (Fig. 1) involving neural processes that predict (estimate) the future state of the system given the current (actual) state and the sensorimotor control signals (Davidson & Wolpert, 2005). To trigger voluntary proactive stepping, the integrated networks for posture and locomotion are activated in parallel to generate a postural command that reconfigures the CoM–BoS relationship, and a locomotion command for stepping. Using internal and external feedback information, the forward model would determine if the APAs have achieved a sufficiently stable postural‐balance state by estimating the position‐velocity relationship between the COM and BOS before initiating the step and completing the postural phase. Hence, the neural circuits for triggering stepping would normally be actively delayed until the APAs that generate the weight transfer from bipedal to single leg stance support have achieved single stance limb loading (Massion et al. 2004; Mille et al. 2014).
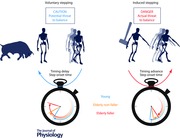
Figure 1. Conceptual model of posture and locomotion coupling during the initiation of stepping .
To initiate voluntary stepping, posture and locomotion networks are activated in parallel to generate motor commands (black arrows) where the posture network acts on the stepping controller. These motor outputs modify the body centre of mass (CoM)–base of support (BoS) relationship. Efferent copies of the motor commands and sensory information about the actual state of the body can be used by the CNS (internal feedback loops – green arrows) to estimate the future state of instability produced by the evolving movement, and modify in advance the two commands based on an internal representation of the body and external environment (forward model). On‐line sensory information can also modulate posture and locomotion commands via external feedback information (grey arrows). For perturbation‐induced stepping, an external mechanical perturbation (red arrows) modifies the CoM–BoS relationship. Multi‐sensory systems transmit information about the evolving state of instability that can be used by the CNS to estimate its consequences using the internal representation of the body, and to activate posture and locomotion networks to initiate a suitably directed, timed and scaled induced step to compensate for the perturbation. Again, these commands can be modified via internal or external feedback processes.
With older age, the time to initiate voluntary stepping under reaction time (RT) conditions generally increases (Luchies et al. 2002; Rogers et al. 2003 a; Melzer & Oddsson, 2004), and this initiation delay is greater for older adults with impaired balance and higher fall risk (Medell & Alexander, 2000; Lord & Fitzpatrick, 2001; St George et al. 2007). The time to perform a rapid step increases more than for younger adults when the direction of stepping is uncertain during choice reaction time (CRT) conditions, or when a dual task activity is performed concurrently suggesting that increased cognitive demands worsen the differences in step triggering linked with older age (Lord & Fitzpatrick, 2001; Luchies et al. 2002; Rogers et al. 2003 a; Melzer & Oddsson, 2004; St George et al. 2007). Such RT delays are often attributed to age‐related changes in sensorimotor speed and central neural processing. In this regard, associations between slowed RT stepping performance and white matter hyperintensities (WMHs) affecting the integrity and function of neural networks linked with sensorimotor processing and cognitive executive processes have been shown (Sparto et al. 2008; Zheng et al. 2012). With an increased WMH volume, CRT slowing appeared to be mediated through slower cognitive processing and not through reduced sensorimotor functioning (Zheng et al. 2012). An association between greater levels of WMHs and prospective falls also appeared to be independent of diminished cognitive executive function and sensorimotor performance indicating a role for vascular health in preserving balance performance and preventing falls with ageing (Zheng et al. 2011).
The slower voluntary stepping with ageing is frequently accompanied by deficits in initial postural adjustments such as errors in directional selection, multiple postural movements, reduced peak force, longer time to peak force, and prolonged duration especially for individuals at higher fall risk under dual‐task conditions (Melzer et al., 2007, 2010; Cohen et al. 2011; Sparto et al. 2013). These postural changes that precede step initiation suggest that a disruption in the normal coupling between posture and locomotion elements of stepping may contribute to step triggering delays through suppression of the locomotion network based on an estimation of the evolving state conditions for stability (see Fig. 1) (Mille et al. 2012). In support of this postulate, recent work has shown that the onset of rapid voluntary stepping in older and younger adults can be made faster by externally assisting with M‐L APAs (Mille et al. 2007), while impeding the performance of APAs can delay the onset timing (Mille et al. 2014) (Fig. 2). Additionally, during CRT stepping, older adults make more errors in postural preparation that further delay step onset timing with greater forward excursion of the body in association with response inhibition deficit (Cohen et al. 2011). Furthermore, incongruous stimulus–response cues caused older adults to make more postural adjustments than they typically would with stimulus–response compatibility, indicating errors in postural preparation selection (Sparto et al. 2013). During these erroneous trials, the onset of postural adjustments was earlier than during compatible cueing trials, indicating a failure to inhibit an inappropriate initial postural adjustment. Consequently, with additional postural adjustments, step onset was delayed compared with steps where postural errors did not occur. Overall, these findings suggested that deficits in inhibitory function involving response preparation may detrimentally affect proactive stepping by interfering with posture–locomotion coupling and delaying stepping.
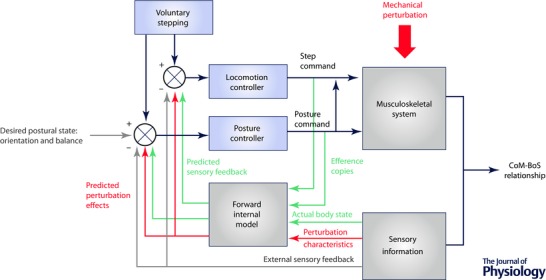
Figure 2. Posture and locomotion coupling during voluntary stepping .
A, during voluntary stepping, single limb stance support is normally achieved prior to stepping. If anticipatory postural adjustments (APAs) are unexpectedly modified by external assistance (data presented in black, group mean and SD of 8 older subjects; adapted from Mille et al. 2007) or resistance (data presented in green, group mean and SD of 12 young women; adapted from Mille et al. 2014), step onset timing is then either advanced (Assisted) or delayed (Resisted) compared with the control condition (Normal) so that step onset occurs when the estimated conditions for stability are achieved. When APAs are assisted, older individuals initiated volitional stepping as fast as younger adults’ performance in the unassisted baseline condition. B, the averaged time profiles from a representative younger adult during baseline rapid step initiation (in blue, average of 10 trials) and during mechanically resisted step initiation (in red, average of 15 trials). All records were synchronized with the onset of the net CoP displacement (vertical dotted line) at time zero. The shaded regions represent ± 1 SD. The vertical displacement of the stepping ankle marker indicates a delay in the first step onset time and increased step height. The mean EMG activity of stepping leg muscles shows a delay of the step motor command (illustrated by the rectus femoris (RF) and the tensor fasciae latae (TFL) activity), and a modification of the step motor command with the appearance of a phasic activation bursts in soleus (SOL) and gastrocnemius medialis (GAST) when a postural perturbation is randomly applied, whereas no modification in the onset of the tiabialis anterior (TA) activity was observed.
Triggering induced stepping
Stepping can also be induced by an externally applied mechanical or sensory perturbations. The initiation timing of externally induced reflex‐like stepping can be more rapid than voluntary stepping (McIlroy & Maki, 1996; Luchies et al., 1999, 2002; Rogers et al. 2003 a). As shown in Fig. 1, an externally applied mechanical perturbation modifies the CoM–BoS relationship. This postural disturbance is detected by multi‐sensory systems that transmit information about the evolving state of balance instability that can be used by the central nervous system (CNS) to predict the consequences to stability based on the internal representation of the body. In turn, posture and locomotion networks are activated to trigger induced stepping with the required direction, timing and amplitude needed to compensate for the perturbation. Unlike voluntary stepping, APAs are normally either absent or diminished in magnitude and effectiveness during induced stepping, and this appears to compromise M‐L stability (McIlroy & Maki, 1999; Rogers et al. 2001).
Based on a hierarchical model of strategy selection according to the amplitude of the postural challenge, induced stepping reactions have been traditionally thought to occur after relatively large postural disturbances where postural movements that maintain stationary foot–ground contact such as ankle and hip strategies fail to prevent the CoM from exceeding the mechanical limits of the BoS (Shumway‐Cook & Woollacott, 2001). This perspective has been refuted by more recent work showing that reactive stepping often normally occurs well before the limits of stability are exceeded even for smaller intensity perturbations as determined by the instantaneous position and velocity of the CoM relative to the physical limits of the BoS (Maki & McIlroy, 1997; Pai & Patton, 1997; Pai et al. 1998; Hof et al. 2005; Patton et al. 2006; Carty et al. 2011). Thus, stepping does not depend solely on the mechanical constraints of the position of the CoM exceeding the BoS as many studies have shown that stepping occurs more frequently than predicted by this model (McIlroy & Maki, 1993; Pai et al., 1998, 2000).
Consistent findings across studies have shown that older individuals step more often and with smaller perturbations than younger adults (McIlroy & Maki, 1993; Pai et al. 1998; Rogers et al. 2001; Mille et al. 2003). This suggests that physiological factors rather than solely mechanical reasons determine step triggering. Recent observations have indicated that, at first step lift‐off following lateral postural disturbances, the area used to move the net CoP for decelerating CoM motion was reduced for older fallers compared with non‐fallers indicating reduced functional limits of dynamic stability (Fujimoto et al. 2015). This finding implied that although the CoM motion state for fallers is located farther from the BoS boundary than for non‐fallers indicating a greater stability margin in relation to their BoS, they might have approached their functional limit for CoP movement requiring them to step. Diminished excursion of the CoP, likely to be through impaired neuromechanical control of hip abduction–adduction torques, precipitated greater instability for at‐risk individuals when stepping was triggered. Thus, the functional limit might represent a more sensitive estimation of lateral balance stability.
Experimental determination of the threshold for triggering induced stepping in response to forward waist‐pull perturbations (Mille et al. 2003) showed that the threshold boundary had a characteristic sigmoidal relationship between position and velocity of the pelvis relative to the BoS, with distinct position thresholds for both low‐ and high‐velocity waist‐pulls (Fig. 3). The boundary had a forward limit (T L) that, when exceeded, always made subjects step no matter how slowly they were pulled. As velocity increased, the threshold position that triggered a step shifted nearer to the ankle joint centre. Eventually a pull velocity was reached above which velocity had no further effect and a position threshold (T H) was identified behind which subjects never stepped (Fig. 3). Older subjects stepped more often than the young (69% vs. 40% of trials). For the older subjects, T L and T H were closer to the ankles, and the transition between T L and T H occurred at lower velocities (Fig. 3). The physiological factors that could explain this threshold were investigated by studying the association of the boundary characteristics with a range of sensorimotor physiological tests (Mille et al. 2003). Overall, many sensorimotor factors were associated with T L and T H. However, these associations were not present when age was removed as a factor. Thus, although the older subjects stepped more often, this cannot be attributed directly to the sensorimotor factors tested. It can be explained, however, by stepping as a triggered response to the perturbation event rather than to later sensory input about body movement reflecting the evolving state of instability.
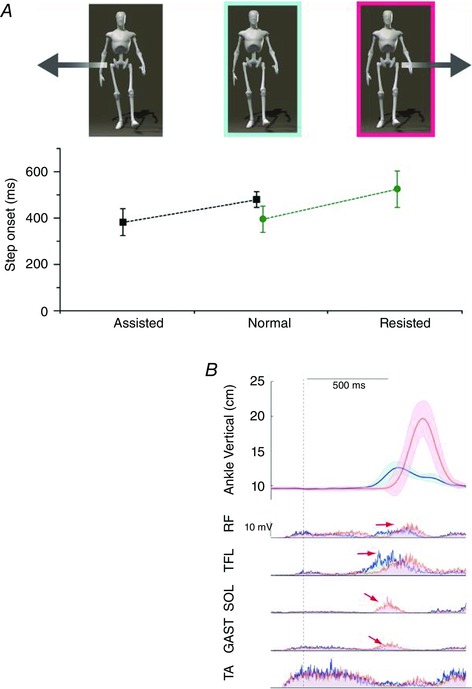
Figure 3. Experimental determination of perturbation‐induced stepping threshold .
Younger and older adults received 124 randomly applied displacement‐velocity forward waist‐pull perturbations to determine the threshold for triggering induced stepping. A, the threshold for forward‐induced stepping was determined using a linear‐motor pulling the subject forward via a flexible cable attached around the upper pelvis. The pulls started at an unexpected time and proceeded at the test velocity until, on reaching the test displacement (D), the cable tension is released and the subject can lean back if a step has not already been initiated. This is illustrated in the pull displacement profile (D) as a function of time. The initial position of the subject was estimated from the initial location of the centre of pressure (CoPi) relative to the ankle joint centre. D was added to the CoPi to estimate the final position of the pelvis at the end of the pull relative to the ankle (P). This reflected how far in front of the axis of the ankles the pelvis moved at the end of the pull. B, the threshold boundary was then described using a modified Boltzmann function, which is a negative, inverse exponential of velocity. Four parameters, represented in the graph, described the low‐velocity plateau (T L) and the high‐velocity plateau (T H) levels, the velocity that produces the mid‐range response (V 50), and the slope (S). The inverse of the slope is directly proportional to the maximal steepness of the function, which occurs at mid‐range. C, typical stepping records for a young and an older subject. Each circle corresponds to a pull trial, marked as either a step (yellow) or no step (white). At each velocity, a threshold position (red diamond) was identified half way between the most anterior position (largest value) that did not produce a step and the most posterior position (smallest value) that produced a step. The Boltzmann function was fitted (Gauss–Newton non‐linear least‐mean‐squares algorithm) to these threshold position points to describe the threshold boundary (continuous line). (Adapted from Mille et al. 2003.)
Timing paradox of step triggering with ageing and falls
As reviewed above, older adults are slower initiating voluntary stepping and this increase in RT has been associated with a greater risk of falls. Paradoxically, the initiation timing of induced protective stepping is as fast for older persons as for younger adults or even faster for those at greater risk of falls (Pai et al. 1998; Luchies et al. 1999; Rogers et al. 2001, 2003 a; Mille et al. 2013). When considered together, these observations suggest that induced stepping among older adults is pre‐selected as a strategy based on an internal representation estimate of system dynamics wherein it is recognized from experience that pre‐stepping compensatory responses (e.g. ankle torque strategy) are inadequate (Woollacott, 2000; Okada et al. 2001), and predetermines that a step will be needed when an input trigger arrives even before the step may actually be necessary.
The foregoing proposition has been further investigated by introducing experimental conditions involving contextual uncertainty about balance stability that alter the initiation timing of stepping. If a pre‐selection strategy were used, then combining an imperative voluntary RT instruction set with the potential for perturbation would delay the triggering of reactive stepping in the presence of uncertainty about the impending state of stability that could accompany stepping (Rogers et al. 2003 a). With this paradigm, the premature triggering of a forward step would potentially exacerbate instability through synchronization of instructed proactive stepping and forward perturbation‐induced stepping. When older subjects initiated instructed rapid stepping in response to a light cue (voluntary–certain), step initiation tended to be slower than for younger adults (Fig. 4). However, older subjects were as fast as the young to initiate an induced step in response to a postural perturbation (induced–certain). When a voluntary step was initiated under the threat of a forward perturbation (voluntary–uncertain), step initiation was delayed, particularly in older subjects, who remained slower than the young. On the other hand, when subjects performed RT stepping and were perturbed forward before the reaction cue was activated (induced–uncertain), younger and older individuals demonstrated a context‐dependent capacity to delay substantially (by 200–300 ms) the triggering of induced protective stepping. This indicated that the initiation timing for triggering an induced step does not reflect entirely an immediate necessity or a last resort response to balance instability. While stepping occurred 50 ms earlier for the old during the induced–certain condition, their initiation timing during induced–uncertain trials occurred 130 ms earlier than for the young (Fig. 4). Thus, the trend for older individuals to trigger stepping earlier was exacerbated by the contextual uncertainty about balance stability. At least for these experimental conditions, perturbation‐induced stepping was apparently not triggered directly by specific sensory input reflecting the state of balance stability but appeared to involve a pre‐selection process that was initiated before it may have actually been needed. With balance uncertainty, subjects might have prolonged their initiation timing to a threshold level that more directly approached their mechanical limits of stability to recover balance and step initiation timing might have been estimated via on‐line sensory information.
Figure 4. Effects of age and contextual uncertainty on the timing of step initiation .
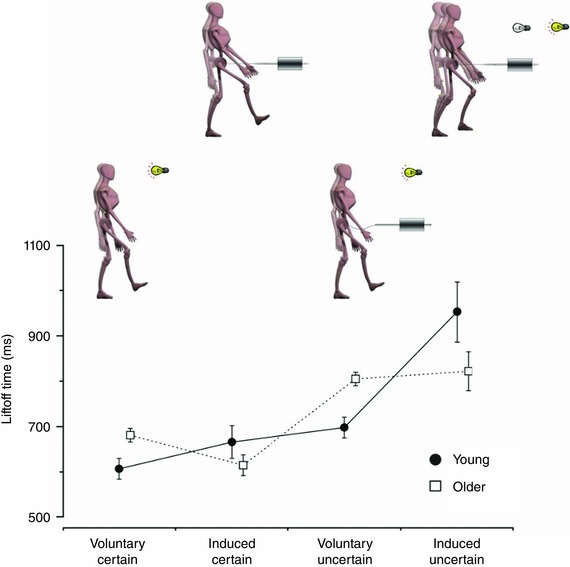
The loading–unloading forces registered by a force platform beneath the stepping limb provided timing information about the instant of first step lift‐off under four different experimental conditions: voluntary–certain (instructed reaction time (RT) stepping without perturbations), induced–certain (perturbation‐induced stepping without instruction), voluntary–uncertain (instructed RT stepping with threat of random perturbations), induced–uncertain (instructed RT stepping with actual random perturbations). In the induced–uncertain trials, the perturbation was presented at different times prior to the imperative RT light cue. When older subjects initiate a voluntary step in response to a light cue (voluntary–certain), step initiation is slower than for younger adults. Paradoxically, older subjects are as fast as the young to initiate a step in response to the postural perturbation (induced–certain). When a voluntary step was initiated under the threat of a perturbation (induced–uncertain), step initiation timing was delayed, particularly in the old, who remained slower than the young. When subjects performed RT stepping but were perturbed prior to the imperative go‐cue (induced–uncertain), both groups delayed the onset of stepping. This indicated that the initiation timing for triggering induced stepping did not reflect entirely an immediate necessity or a last resort response to balance instability (values represent group means and SDs for 32 older subjects and 14 young; adapted from Rogers et al. 2003 a).
The earlier triggering of induced stepping with ageing and falls might reflect a decision to step rather than a necessity. Psychophysical studies have shown that the threshold for detecting an event is lower than the threshold for detecting the specific properties of movement (Hall & McCloskey, 1983; Refshauge et al. 1995). Sensory information about the occurrence of an event does not have information about movement size and direction. When a mechanical perturbation occurs (Fig. 5), those with greater fall risk might step in response to event detection rather than to specific movement detection information. This would explain the earlier timing of induced stepping with older age and fall risk and the ineffectiveness of the first step observed for many of these individuals (Mille et al. 2013).
Figure 5. Conceptual model of induced‐step triggering with ageing and fall risk .
An external postural perturbation modifies the CoM–BoS relationship. Instead of using on‐line processing of the perturbation characteristics to estimate the state of evolving instability (based on an internal representation of the body) and trigger an induced step with the correct direction, timing and amplitude to compensate for the perturbation (see Fig. 1), particularly older adults at greater risk of falling might react to event information (red arrow) that would trigger a preset stepping response that is not adaptively tuned to the perturbation characteristics.
Conclusions
The features of the age‐related differences in the triggering of voluntary and induced stepping and their relationship with the risk of falls are compatible with the conceptual theoretical model that we propose. The characteristic delay in initiating rapid voluntary stepping with ageing, especially when spatial or temporal uncertainties about the nature of the task are not known in advance (e.g. CRT) or when cognitive demands are increased (e.g. dual task performance), is somewhat counterintuitively accompanied by an equivalent or earlier triggering of externally induced stepping. While the triggering delay in voluntary RT stepping has generally been attributed primarily to changes in neuronal conduction and central processing, the specific neural substrates and processes that underlie this slowing remain to be elucidated. For example, although white matter hyperintensities linked with sensorimotor processing and cognitive executive functions may play a role in delaying voluntary stepping (Zheng et al. 2012), alterations in motor preparation or response generation integrating posture and locomotion that involve cortical and brainstem networks may also undergo changes with age that prolong step triggering (Drew et al. 2004; Jacobs & Horak, 2007; MacKinnon et al. 2007; Rogers et al. 2011). Furthermore, older individuals with motor slowing and altered coordination show a reduced capability to modulate GABA‐mediated cortical inhibitory processes that appears to be related to age‐related loss of white and grey matter and increased brain activation in cortical and subcortical areas (Levin et al. 2014).
In future work, it will be important to determine whether or not age‐related changes in the ability to modulate motor cortical or brainstem excitability and inhibitory processes during response preparation and generation of stepping, or in suppressing stepping actions, are linked with altered protective stepping and falls. These processes have been previously shown to be affected by older age for isolated single joint and limb movements (Fujiyama et al. 2012; Levin et al. 2014), and to a lesser extent during stepping (Jacobs and Horak, 2007; MacKinnon et al. 2007; Rogers et al. 2011). Determining the associations between ageing impairments in structural and/or functional integrity of brain networks with changes in CNS modulatory mechanisms and motor deficits are additional gaps and emerging areas that could be fruitful to investigate. Given the pervasive sarcopenic changes in skeletal muscle accompanying older age with diminished motor performance (Addison et al. 2014; Cruz‐Jentoft et al. 2014; Inacio et al. 2014), better understanding of the relative contributions of neural and muscular deficits and their interaction to altered neuromotor control of stepping and other balance stabilizing actions is another promising line of study in this area.
For induced stepping, the fact that steps are triggered more often and earlier within the CoM–BoS position–velocity state space, and may be as fast or faster than for younger adults, is consistent with the postulation that older people at greater risk of falling might step in response to an event‐detection strategy rather than to specific movement detection information. Although the causal factors underlying this possibility are currently unknown, it is conceivable that heightened arousal or anxiety related to one's vulnerability to falls may be contributing to a lower threshold for triggering stepping (Pai et al. 1998; Carpenter et al. 2004). A further and intriguing possibility is whether or not external perturbations of posture induce startle reactions that may be interspersed with postural responses and differentially integrated and modulated with age and fall risk. Studies have shown a superimposition of startle reflex activity on the postural response during first trial balance and freefall perturbations, and that postural responses can be accelerated by a startling acoustic stimulus even when the perturbation direction is not known in advance (Oude Nijhuis et al. 2010; Campbell et al. 2013; Sanders et al. 2015). It is possible that the presence of startle reaction coincident with the balance response may interfere with the parametric control of protective stepping and its effectiveness in stabilizing balance. Therefore, a further direction for future studies would be to determine whether or not age‐related changes in startle contributions involving brainstem and cortical pathways influence the triggering of induced stepping responses in relation to fall risk.
Finally, from a rehabilitation standpoint, the capacity for both volitionally triggered and perturbation‐induced protective stepping to be at least acutely improved with step training interventions among older individuals suggests that there is a residual adaptive capacity that can be exploited to advance therapeutic approaches to enhance balance function and reduce the risk of falls (Rogers et al. 2003 b; Mille et al. 2009; Barrett et al. 2012; Yungher et al. 2012). Therefore, it appears that targeting improvements in age‐associated paradoxical triggering of voluntary and induced protective stepping could be a useful goal of rehabilitation interventions. In this regard, however, an important gap in current understanding is in determining the underlying CNS and neuromotor mechanisms that drive activity and exercise‐based adaptations in motor performance to improve protective balance and other motor actions. Greater insights into these areas should be valuable for optimizing the design of effective balance rehabilitation and fall prevention interventions by providing directives for tailored programmes that target the specific problems for different health conditions and individuals.
Additional information
Competing interests
None declared.
Author contributions
Both authors have approved the final version of the manuscript and agree to be accountable for all aspects of the work. All persons designated as authors qualify for authorship, and all those who qualify for authorship are listed.
Biographies
Mark W. Rogers received his undergraduate degree in Physical Therapy from the University of Connecticut, Storrs and graduate degrees from the University of Massachusetts, Amherst and the University of Iowa, Iowa City followed by postdoctoral studies at McGill University, Montreal, Canada. His research interests are focused on human neuromotor control of balance and locomotion in health and disease including the interaction of neural, musculoskeletal and behavioural systems that underlie function and its restoration through rehabilitation.

Marie‐Laure Mille has a PhD in Motor Control from the Aix‐Marseille University. She started focusing on protective stepping and ageing during her postdoctoral fellowship at Northwestern University. She has broad interests in human sensorimotor control processes in health and disease with a focus on the coordination of posture and locomotion. She has a tenured position at the University of Toulon and is furthering her research at the Movement Sciences Institut in Marseille.
This review was presented at the symposium “From TOP to TOE: What falls reveal about physiological ageing and degeneration”, which took place at Ageing and Degeneration: A Physiological Perspective in Edinburgh, UK, 10–11 April 2015.
References
- Addison O, Young P, Inacio M, Bair W‐N, Prettyman MG, Beamer BA, Ryan AS & Rogers MW (2014). Hip but not thigh intramuscular adipose tissue is associated with poor balance and increased temporal gait variability in older adults. Curr Aging Sci 7, 137–143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrett RS, Cronin NJ, Lichtwark GA, Mills PM & Carty CP (2012). Adaptive recovery responses to repeated forward loss of balance in older adults. J Biomech 45, 183–187. [DOI] [PubMed] [Google Scholar]
- Breniere Y, Do MC & Bouisset S (1987). Are dynamic phenomena prior to stepping essential to walking? J Mot Behav 19, 62–76. [DOI] [PubMed] [Google Scholar]
- Campbell AD, Squair JW, Chua R, Inglis JT & Carpenter MG (2013). First trial and StartReact effects induced by balance perturbations to upright stance. J Neurophysiol 110, 2236–2245. [DOI] [PubMed] [Google Scholar]
- Carlsoo S (1966). The initiation of walking. Acta Anat (Basel) 65, 1–9. [DOI] [PubMed] [Google Scholar]
- Carpenter MG, Frank JS, Adkin AL, Paton A & Allum JHJ (2004). Influence of postural anxiety on postural reactions to multi‐directional surface rotations. J Neurophysiol 92, 3255–3265. [DOI] [PubMed] [Google Scholar]
- Carty CP, Mills P & Barrett R (2011). Recovery from forward loss of balance in young and older adults using the stepping strategy. Gait Posture 33, 261–267. [DOI] [PubMed] [Google Scholar]
- Cohen RG, Nutt JG & Horak FB (2011). Errors in postural preparation lead to increased choice reaction times for step initiation in older adults. J Gerontol A Biol Sci Med Sci 66, 705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cruz‐Jentoft AJ, Landi F, Schneider SM, Zúñiga C, Arai H, Boirie Y, Chen L‐K, Fielding RA, Martin FC, Michel J‐P, Sieber C, Stout JR, Studenski SA, Vellas B, Woo J, Zamboni M & Cederholm T (2014). Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing 43, 748–759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidson PR & Wolpert DM (2005). Widespread access to predictive models in the motor system: a short review. J Neural Eng 2, S313–S319. [DOI] [PubMed] [Google Scholar]
- Drew T, Prentice S & Schepens B (2004). Cortical and brainstem control of locomotion. Prog Brain Res 143, 251–261. [DOI] [PubMed] [Google Scholar]
- Fujimoto M, Bair W‐N & Rogers MW (2015). Center of pressure control for balance maintenance during lateral waist‐pull perturbations in older adults. J Biomech 48, 963–968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujiyama H, Hinder MR, Schmidt MW, Tandonnet C, Garry M & Summers JJ (2012). Age‐related differences in corticomotor excitability and inhibitory processes during a visuomotor RT task. J Cogn Neurosci 24, 1253–1263. [DOI] [PubMed] [Google Scholar]
- Hall LA & McCloskey DI (1983). Detections of movements imposed on finger, elbow and shoulder joints. J Physiol 335, 519–533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hof AL, Gazendam MGJ & Sinke WE (2005). The condition for dynamic stability. J Biomech 38, 1–8. [DOI] [PubMed] [Google Scholar]
- Inacio M, Ryan AS, Bair W‐N, Prettyman M, Beamer BA & Rogers MW (2014). Gluteal muscle composition differentiates fallers from non‐fallers in community dwelling older adults. BMC Geriatr 14, 37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs JV & Horak FB (2007). Cortical control of postural responses. J Neural Transm 114, 1339–1348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin O, Fujiyama H, Boisgontier MP, Swinnen SP & Summers JJ (2014). Aging and motor inhibition: A converging perspective provided by brain stimulation and imaging approaches. Neurosci Biobehav Rev 43, 100–117. [DOI] [PubMed] [Google Scholar]
- Lord SR & Fitzpatrick RC (2001). Choice stepping reaction time: a composite measure of falls risk in older people. J Gerontol A Biol Sci Med Sci 56, M627–M632. [DOI] [PubMed] [Google Scholar]
- Lord SR, Menz HB, Sherrington C & Close JC (2007). Falls in Older People: Risk Factors and Strategies for Prevention, 2nd edn Cambridge University Press, Cambridge, UK. [Google Scholar]
- Luchies CW, Schiffman J, Richards LG, Thompson MR, Bazuin D & DeYoung AJ (2002). Effects of age, step direction, and reaction condition on the ability to step quickly. J Gerontol A Biol Sci Med Sci 57, M246–M249. [DOI] [PubMed] [Google Scholar]
- Luchies CW, Wallace D, Pazdur R, Young S & DeYoung AJ (1999). Effects of age on balance assessment using voluntary and involuntary step tasks. J Gerontol A Biol Sci Med Sci 54, M140–M144. [DOI] [PubMed] [Google Scholar]
- MacKinnon CD, Bissig D, Chiusano J, Miller E, Rudnick L, Jager C, Zhang Y, Mille M‐L & Rogers MW (2007). Preparation of anticipatory postural adjustments prior to stepping. J Neurophysiol 97, 4368–4379. [DOI] [PubMed] [Google Scholar]
- McIlroy WE & Maki BE (1993). Task constraints on foot movement and the incidence of compensatory stepping following perturbation of upright stance. Brain Res 616, 30–38. [DOI] [PubMed] [Google Scholar]
- McIlroy WE & Maki BE (1996). Age‐related changes in compensatory stepping in response to unpredictable perturbations. J Gerontol A Biol Sci Med Sci 51, M289–M296. [DOI] [PubMed] [Google Scholar]
- McIlroy WE & Maki BE (1999). The control of lateral stability during rapid stepping reactions evoked by antero‐posterior perturbation: does anticipatory control play a role? Gait Posture 9, 190–198. [DOI] [PubMed] [Google Scholar]
- Maki BE & McIlroy WE (1997). The role of limb movements in maintaining upright stance: the “change‐in‐support” strategy. Phys Ther 77, 488–507. [DOI] [PubMed] [Google Scholar]
- Massion J, Alexandrov A & Frolov A (2004). Why and how are posture and movement coordinated? Prog Brain Res 143, 13–27. [DOI] [PubMed] [Google Scholar]
- Medell JL & Alexander NB (2000). A clinical measure of maximal and rapid stepping in older women. J Gerontol A Biol Sci Med Sci 55, M429–M433. [DOI] [PubMed] [Google Scholar]
- Melzer I, Kurz I, Shahar D, Levi M & Oddsson L (2007). Application of the voluntary step execution test to identify elderly fallers. Age Ageing 36, 532–537. [DOI] [PubMed] [Google Scholar]
- Melzer I, Kurz I, Shahar D & Oddsson LIE (2010). Do voluntary step reactions in dual task conditions have an added value over single task for fall prediction? A prospective study. Aging Clin Exp Res 22, 360–366. [DOI] [PubMed] [Google Scholar]
- Melzer I & Oddsson LI (2004). The effect of a cognitive task on voluntary step execution in healthy elderly and young individuals. J Am Geriatr Soc 52, 1255–1262. [DOI] [PubMed] [Google Scholar]
- Mille M‐L, Creath RA, Prettyman MG, Johnson Hilliard M, Martinez KM, Mackinnon CD & Rogers MW (2012). Posture and locomotion coupling: a target for rehabilitation interventions in persons with Parkinson's disease. Park Dis 2012, 754186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mille M‐L, Hilliard MJ, Martinez KM, Simuni T, Zhang Y & Rogers MW (2009). Short‐term effects of posture‐assisted step training on rapid step initiation in Parkinson's disease. J Neurol Phys Ther 33, 88–95. [DOI] [PubMed] [Google Scholar]
- Mille M‐L, Johnson Hilliard M, Martinez KM, Simuni T & Rogers MW (2007). Acute effects of a lateral postural assist on voluntary step initiation in patients with Parkinson's disease. Mov Disord 22, 20–27. [DOI] [PubMed] [Google Scholar]
- Mille M‐L, Johnson‐Hilliard M, Martinez KM, Zhang Y, Edwards BJ & Rogers MW (2013). One step, two steps, three steps more … directional vulnerability to falls in community‐dwelling older people. J Gerontol A Biol Sci Med Sci 68, 1540–1548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mille M‐L, Rogers MW, Martinez K, Hedman LD, Johnson ME, Lord SR & Fitzpatrick RC (2003). Thresholds for inducing protective stepping responses to external perturbations of human standing. J Neurophysiol 90, 666–674. [DOI] [PubMed] [Google Scholar]
- Mille M‐L, Simoneau M & Rogers MW (2014). Postural dependence of human locomotion during gait initiation. J Neurophysiol 112, 3095–3103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada S, Hirakawa K, Takada Y & Kinoshita H (2001). Age‐related differences in postural control in humans in response to a sudden deceleration generated by postural disturbance. Eur J Appl Physiol 85, 10–18. [DOI] [PubMed] [Google Scholar]
- Oude Nijhuis LB, Allum JHJ, Valls‐Solé J, Overeem S & Bloem BR (2010). First trial postural reactions to unexpected balance disturbances: a comparison with the acoustic startle reaction. J Neurophysiol 104, 2704–2712. [DOI] [PubMed] [Google Scholar]
- Pai YC, Maki BE, Iqbal K, McIlroy WE & Perry SD (2000). Thresholds for step initiation induced by support‐surface translation: a dynamic center‐of‐mass model provides much better prediction than a static model. J Biomech 33, 387–392. [DOI] [PubMed] [Google Scholar]
- Pai YC & Patton J (1997). Center of mass velocity‐position predictions for balance control. J Biomech 30, 347–354. [DOI] [PubMed] [Google Scholar]
- Pai YC, Rogers MW, Patton J, Cain TD & Hanke TA (1998). Static versus dynamic predictions of protective stepping following waist‐pull perturbations in young and older adults. J Biomech 31, 1111–1118. [DOI] [PubMed] [Google Scholar]
- Patton JL, Hilliard MJ, Martinez K, Mille M‐L & Rogers MW (2006). A simple model of stability limits applied to sidestepping in young, elderly and elderly fallers. Conf Proc IEEE Eng Med Biol Soc 1, 3305–3308. [DOI] [PubMed] [Google Scholar]
- Refshauge KM, Chan R, Taylor JL & McCloskey DI (1995). Detection of movements imposed on human hip, knee, ankle and toe joints. J Physiol 488, 231–241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers MW, Hain TC, Hanke TA & Janssen I (1996). Stimulus parameters and inertial load: effects on the incidence of protective stepping responses in healthy human subjects. Arch Phys Med Rehabil 77, 363–368. [DOI] [PubMed] [Google Scholar]
- Rogers MW, Hedman LD, Johnson ME, Cain TD & Hanke TA (2001). Lateral stability during forward‐induced stepping for dynamic balance recovery in young and older adults. J Gerontol A Biol Sci Med Sci 56, M589–M594. [DOI] [PubMed] [Google Scholar]
- Rogers MW, Hedman LD, Johnson ME, Martinez KM & Mille M‐L (2003. a). Triggering of protective stepping for the control of human balance: age and contextual dependence. Brain Res Cogn Brain Res 16, 192–198. [DOI] [PubMed] [Google Scholar]
- Rogers MW, Johnson ME, Martinez KM, Mille M‐L & Hedman LD (2003. b). Step training improves the speed of voluntary step initiation in aging. J Gerontol A Biol Sci Med Sci 58, 46–51. [DOI] [PubMed] [Google Scholar]
- Rogers MW, Kennedy R, Palmer S, Pawar M, Reising M, Martinez KM, Simuni T, Zhang Y & MacKinnon CD (2011). Postural preparation prior to stepping in patients with Parkinson's disease. J Neurophysiol 106, 915–924. [DOI] [PubMed] [Google Scholar]
- Rogers MW & Mille M‐L (2003). Lateral stability and falls in older people. Exerc Sport Sci Rev 31, 182–187. [DOI] [PubMed] [Google Scholar]
- St George RJ, Fitzpatrick RC, Rogers MW & Lord SR (2007). Choice stepping response and transfer times: effects of age, fall risk, and secondary tasks. J Gerontol Biol Sci Med Sci 62, 537–542. [DOI] [PubMed] [Google Scholar]
- Sanders OP, Savin DN, Creath RA & Rogers MW (2015). Protective balance and startle responses to sudden freefall in standing humans. Neurosci Lett 586, 8–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shumway‐Cook A & Woollacott MH (2001). Motor Control: Theory and Practical Applications. Lippincott Williams & Wilkins, Baltimore. [Google Scholar]
- Sparto PJ, Aizenstein HJ, Vanswearingen JM, Rosano C, Perera S, Studenski SA, Furman JM & Redfern MS (2008). Delays in auditory‐cued step initiation are related to increased volume of white matter hyperintensities in older adults. Exp Brain Res 188, 633–640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sparto PJ, Fuhrman SI, Redfern MS, Jennings JR, Perera S, Nebes RD & Furman JM (2013). Postural adjustment errors reveal deficits in inhibition during lateral step initiation in older adults. J Neurophysiol 109, 415–428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tinetti ME, Speechley M & Ginter SF (1988). Risk factors for falls among elderly persons living in the community. N Engl J Med 319, 1701–1707. [DOI] [PubMed] [Google Scholar]
- Van den Bogert AJ, Pavol MJ & Grabiner MD (2002). Response time is more important than walking speed for the ability of older adults to avoid a fall after a trip. J Biomech 35, 199–205. [DOI] [PubMed] [Google Scholar]
- Woollacott MH (2000). Systems contributing to balance disorders in older adults. J Gerontol A Biol Sci Med Sci 55, M424–M428. [DOI] [PubMed] [Google Scholar]
- Yungher DA, Morgia J, Bair W‐N, Inacio M, Beamer BA, Prettyman MG & Rogers MW (2012). Short‐term changes in protective stepping for lateral balance recovery in older adults. Clin Biomech 27, 151–157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng JJJ, Delbaere K, Close JCT, Sachdev PS & Lord SR (2011). Impact of white matter lesions on physical functioning and fall risk in older people: a systematic review. Stroke 42, 2086–2090. [DOI] [PubMed] [Google Scholar]
- Zheng JJJ, Lord SR, Close JCT, Sachdev PS, Wen W, Brodaty H & Delbaere K (2012). Brain white matter hyperintensities, executive dysfunction, instability, and falls in older people: a prospective cohort study. J Gerontol A Biol Sci Med Sci 67, 1085–1091. [DOI] [PubMed] [Google Scholar]


