Abstract
OBJECTIVES
We have carried out a systematic review of longitudinal studies examining the association between oral health and cognitive decline.
DESIGN
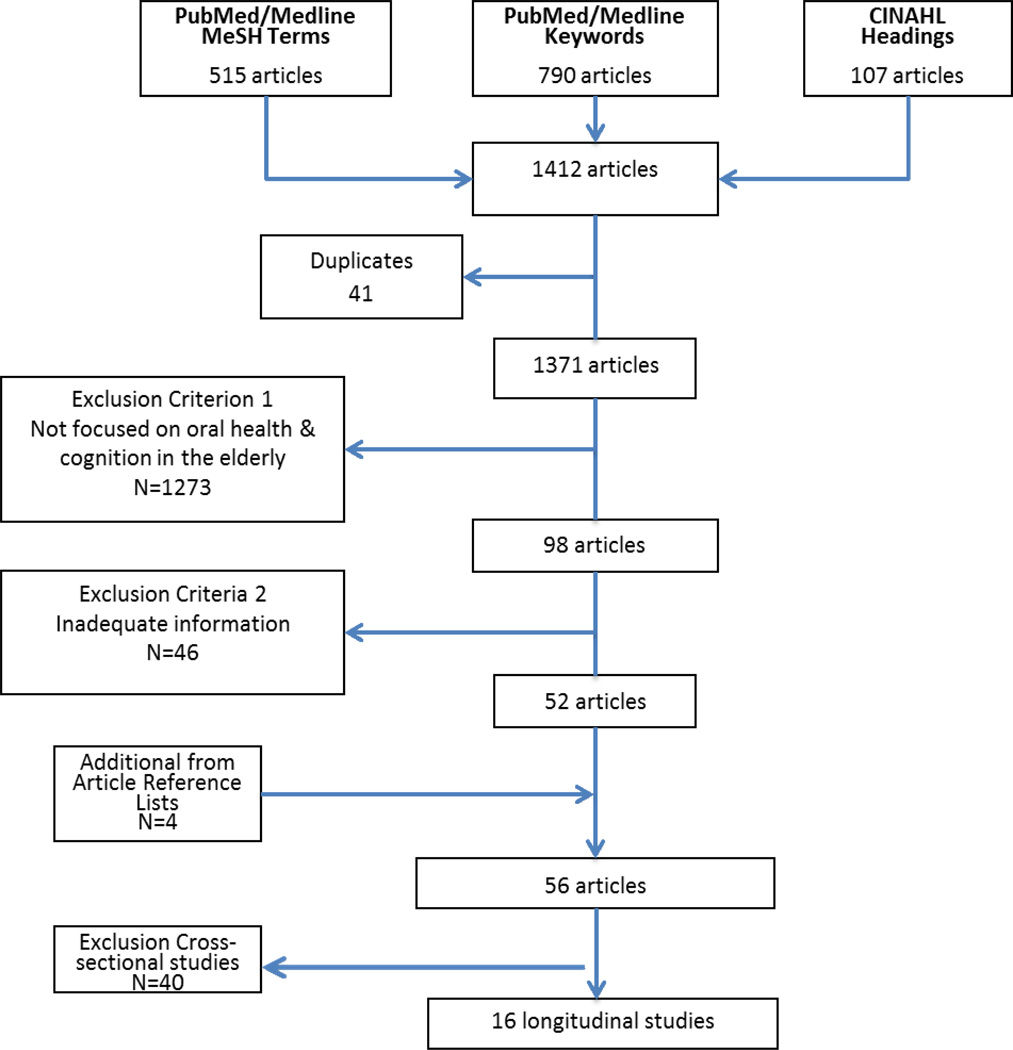
Studies, published 01/1993-03/2013, were identified by search of English language publications in PubMed/Medline using relevant MeSH terms and title/abstract keywords, and from CINAHL using relevant subject headings. After applying eligibility criteria, and adding four studies identified from article references, 56 of the 1412 articles identified remained: 40 were cross-sectional, and 16 longitudinal; 11 of the latter examined the impact of oral health on change in cognitive health or dementia incidence, five examined the reverse.
SETTING
Sources of information included administrative data, subject evaluations in parent studies, medical and dental records, self-reports, and in-person evaluations.
MEASUREMENTS
Most studies used subjects whose oral or cognitive status was known, adding the missing piece using standard measures. The oral health information most frequently studied included number of teeth, periodontal and caries problems, and denture use. Cognition was most frequently evaluated using the MMSE, or by determination of dementia.
RESULTS
Some studies found that oral health measures such as number of teeth and periodontal disease were associated with increased risk of cognitive decline or incident dementia, while others did not find the association. Similarly, cognitive decline was not consistently associated with greater loss of teeth or number of caries. Methodological limitations likely play a major role in explaining the inconsistent findings.
CONCLUSION
It is unclear how or whether oral health and cognitive status are related. Additional research is needed in which there is greater agreement on how oral health and cognitive states are assessed, in order to better examine the linkages between these two health outcomes.
Keywords: Oral health, dementia, cognitive decline
INTRODUCTION
In the past two decades, an increasing number of studies has examined the relationship between cognitive impairment and oral health in older adults, since question has arisen as to whether, in the elderly, there is an association between oral health status and cognitive status, possibly through a common inflammatory pathway.1,2 This issue is of considerable relevance given the rapid increase in the proportion and number of older persons in the population, an increasing number of whom are retaining their natural teeth.3 Further, approximately 36% of those over age 70 are cognitively impaired,4 and the prevalence of cognitive impairment and dementia increases with age.5 Up to 5.1 million Americans currently have a dementing disorder,6 with an expected doubling by 2050.7
Concomitantly, evidence from clinical samples suggests that the elderly have an increased incidence of oral disease, the frequency of oral health problems increasing significantly in cognitively impaired elderly, particularly those with dementia. Any intervention that might help delay the onset or progression of dementia, including improvement of oral hygiene and dental services, could have a significant impact on personal and family well-being, and health care costs.
In order to examine the association between oral health and cognition in the elderly, and provide direction for future studies, we conducted a systematic review of the existing literature. Our focus is on longitudinal studies, since they may be informative with respect to causation.
METHODS
Search Strategy
The time frame considered was from the first relevant publication found in 1993,8 to 03/01/2013. We searched for human study publications in English in the PubMed/Medline database, using the MeSH terms (Memory Disorders or Cognition or Cognition Disorders or Dementia) AND (Oral Health or Mouth Diseases or Tooth Diseases), which identified 515 articles; a title/abstract keyword search using (mouth or tooth or oral AND cogni* or Alzheimer or dement*), which identified 790 articles; and the CINAHL database using the subject headings (dementia+ or Alzheimer’s diseases or cognition+ or cognition disorders+ or memory disorders+) AND (mouth diseases or tooth loss or dental caries), identifying 107 articles. After removing 41 duplicates from the 1412 articles identified, 1371 unique articles remained (Figure 1).
Figure 1.
Identification of relevant articles
We applied two exclusionary criteria to identify non-qualifying articles. The first criterion excluded 1,273 articles that, by title/abstract, did not examine the association between cognition and oral health diseases and conditions in adults; this left 98 articles. The second criterion excluded articles that, based on review of the abstract by two readers, had inadequate information on oral health or cognitive status. Any disagreement was resolved by a third reviewer. In consequence, an additional 46 articles were excluded and 52 retained. To these were added four articles culled from article reference lists, resulting in a total of 56 articles, based on 55 studies. Of these, 40 articles represented cross-sectional studies, and 16 represented longitudinal studies.
Data Extraction
Two reviewers independently extracted information from each longitudinal article on: first author and date of publication, study name, location; study date(s), sample demographics; cognitive and oral health measures; covariates; and key findings. Disagreements on data extraction were resolved by consensus with the assistance of a third person.
RESULTS
The characteristics of the 16 longitudinal studies are summarized in Table 1. The first section summarizes the 11 studies of oral health predicting cognitive decline, the second section summarizes the five studies of cognitive status/dementia predicting oral health.
Table 1.
Characteristics of longitudinal studies of oral health and cognitive statusa
| Author/Location/ Study Title |
Sample/ Study Design | Cognitive Measures |
Oral Health Measures |
Primary Covariates |
Key findings and analytic proceduresb |
|---|---|---|---|---|---|
| ORAL DISEASE PREDICTS COGNITIVE IMPAIRMENT | |||||
| Shimazaki et al., 200111 Kitakyushu City, Japan 29 institutions for elderly |
Baseline: 10/1988-02/1989 N=1929 (87% of residents) Followup:10/1994-03/1995 N=719 (856 died, 167 status unknown, 187 hospitalized) M=169; F=550 Age range: 59–107; mean age 79.7±7.5y |
From medical records Mental health status: Good/ Fair/ Poor re: symptoms of dementia or cognitive disability Followup: Deterioration = symptoms of dementia, or cognitive disability status at lower level |
Baseline: 2 dentists trained in use of epidemiological indices for oral health No. teeth (excluding retained roots) categorized, denture use for eating |
From medical records Age, sex, mobility, type of institution, chronic diseases |
Logistic regression adjusted for age, physical health status, type of institution, cerebrovascular disorder: No. teeth, denture use, not associated with decline in mental health status Reference = 20+ teeth (OR, 95% CI) 1–19 teeth+dentures 1.9 (0.8–4.6) 1–19 teeth-dentures 2.3 (0.9–5.8) 0 teeth+dentures 1.0 (0.7–4.0) 0 teeth-dentures 2.4 (0.9–6.5) |
| Yoneyama et al., 200212 Japan 11 nursing homes |
Baseline: 09/1996, followed at 6, 12, 18, 24 months N=366, randomized: 184 oral care (M=36, F=148; mean age 82.0±7.8y) 182 no oral care (M=37, F=145; mean age 82.1±7.5y) Oral intervention – brush teeth for 5” after each meal, or clean oral cavity. Clean dentures daily. Plaque and calculus control as necessary. |
MMSE score at baseline, 24 months |
Debris index for plaque score |
Survivors only. 24 month change in MMSE score: Oral care: −1.5 +/− 4.9 No oral care: −3.0 +/− 5.9 P <.05 |
|
| Gatz M et al., 200615 Sweden, Swedish Twin Registry HARMONY study |
Questionnaires mailed 1961, 1963, 1967 (1970 if no response in 1967) 1998 survivors age 65+ of same sex twins born before 1926, and contacted for HARMONY study N=310 dementia (M=85, F=225), average onset age: 78.95±7.00y N=3,063 nondemented (M=1,200, F=1,863), mean age 79.23 ± 4.73y |
Dementia determined from Swedish Inpatient Discharge Registry and Swedish Cause of Death Registry (sensitivity 63%, specificity 98% for dementia) |
Self-report of own teeth at age 35 (all or most/ half/ few or none) |
Age, sex, education CVD Lifestyle before age 40: Mental activity (reading, cultural activities, studies) Physical exercise Adult height (>1sd shorter vs. rest) Parents’ social class (upper, middle, lower) |
Analyses adjusted for all covariates. Loss of all teeth Case control analysis predicting (1) all dementias, (2) Alzheimer’s disease: (1) OR 1.49 (1.14–1.95) (2) OR 1.68 (1.21–2.32) Monozygotic co-twin control analyses: (1) OR 3.60 (1.34–9.70) OR 5.50 (1.22–24.80) |
| Stein et al., 20079 Milwaukee, WI Nun Study25 (Snowdon, 1997) |
Baseline: 1991–1992 N=144 nuns with cognitive assessment and known APOE ε4 status Age range 75–98y N=32 with dementia N=112 without dementia. N=101 with adequate followup data Neuropathology data on 118/129 decedents Followup: annual, 12 years |
CERAD neuro- psychology battery, administered annually for 12 years Dementia criteria: NINCDS-ADRDA Braak staging for AD neuropathology |
40 years of dental records No. teeth excluding non- third molars present at first cognitive evaluation. Presence of periodontal damage |
Age, education APOE ε4 status Medical history in dental records (N=133) Medical conditions associated with inflammatory process |
Incident dementia over 12 years: Adjusted for age, education, APOE ε4: 0–9 teeth vs 10–28 teeth: HR 2.20 (1.1–4.5) Further adjusted for periodontal damage, no association HR 2.4 (0.86–6.6) For 118 deceased, adjusted for age, education, APOE ε4: No association between no. of teeth and severity of AD neuropathology (Braak stage): 1.1 (0.49–2.7); or brain infarcts: 1.14 (0.49–2.66) |
| Kaye et al., 201016 US Dept. of Veterans Affairs Dental Longitudinal Study Boston, Mass metropolitan area |
Baseline: 1968 Original N = 1,231 Race: 97% White, 3% Black Current study: N=597 men with necessary dental and cognitive data Age 24–84 Convenience sample, community dwellers Followup: up to 32 years (until 2002) |
Given at each dental exam. (~every 3 years) MMSE (low score = <25, or <90% of age/education specific median) Spatial copying task (SCT) |
Dental exam ~every 3 years by calibrated periodontist, reliability of measurements determined Number of teeth, maximum probing pocket depth for each tooth, alveolar bone loss Caries or restoration (5 tooth surfaces examined) |
Age, education smoking aspirin use, NSAIDs BMI, CHD, stroke, hypertension, CVD, cancer, diabetes alcohol, coffee, tea, folate, B6, B12 |
Adjusted analyses Additional tooth loss/decade associated with low: MMSE: HR 1.09 (1.01–1.18) SCT: HR 1.12 (1.05–1.18) Increased alveolar bone loss associated with low: MMSE: HR 1.03 (1.00–1.07) SCT: HR 1.03 (1.01–1.06) Increased pocket depth associated with low: MMSE: HR1.04 (1.01–1.09) SCT: HR 1.04 (1.01–1.06) New caries, restoration: associated with low: MMSE: HR 1.02 (0.97–1.08) SCT: HR 1.05 (1.01–1.08) |
| Stein et al., 201010 Milwaukee, WI |
Same sample as Stein et al., 2007 |
Delayed word recall (from CERAD Neuropsychology battery) |
Same as Stein et al., 2007. |
Education (≤high school vs >high school) APOE ε4 |
Persons with APOE ε4 allele, ≤9 teeth, or both, had poorer delayed recall scores at baseline and declined faster than those with either one or neither of these risk factors. |
| Arrivé, et al., 201217 PAQUIDENT (PAQUID Dental study), substudy of Personnes Agées QUID (PAQUID) study (Dartigues et al., 1992) Gironde, southwest France |
PAQUIDENT baseline: 1989–1990 N=405 Age 66–80 Median age=70 M=184, F=221 No dementia Median followup=10y PAQUID baseline: 1988 Followup: 1,3,5,8,10,13,15y (through 2003) Community dwellers |
MMSE, Benton Visual Retention Test, Wechsler Paired Associates, verbal fluency, visuospatial attention, digit symbol Dementia criteria: DSM-III-R NINCDS-ADRDA (for AD) Hachinski score (vascular dementia) Dementia status ascertained after in- person assessment by neurologist (demented vs normal). |
In-home oral exam by 10 trained, calibrated dentists. No. decayed, missing, filled teeth (DFMT) (for 28 teeth) No. missing teeth (denture substitution counts as own teeth) Plaque/calculus Periodontal exam for Community Periodontal Index (CPI) |
Sex, education (lower vs higher, years not specified), living condition, Tobacco, alcohol use Health status: CES-D depression, BMI, vascular risk factors |
Adjusted for sex, BMI, vascular risk factors Predicting incident dementia Higher education ≥11 missing teeth vs <11: HR 1.07 (0.57–2.02) Bleed/calculus: HR 0.71 (0.31–1.63) Pockets: HR 0.42 (0.15–1.15) No eligible sextant: HR 1.51 (0.63–3.57) Lower education ≥11 missing teeth vs <11: HR 0.30 (0.11–0.79) Bleed/calculus: HR 1.24 (0.39–3.88) HR Pockets: 0.97 (0.29–3.19) No eligible sextant: HR 0.02 (0.28–3.66) |
| Paganini-Hill et al., 201219 Leisure World Laguna Hills, CA, USA |
Study dates: 1992–2010 N=5,468 Median age: 81y, range 52– 105 M=1,733; F=3,735 No. with incident dementia: 1,145 (followed up to 18 years) Retirement community |
Dementia determined by: In-person evaluation for age 90+ (n=519 with dementia, identified by different study). Self-report (n=90), hospital records (n= 46), death certificates (n=490) Date of diagnosis = date dementia first mentioned |
Self-report: # natural teeth: <16/ 16–25/ 26+ Adequate natural dentition (10 in upper jaw, 6 in lower jaw) Inadequate natural dentition: wears dentures/ does not Oral health habits |
1992 -- Self-reported education, head injury, first degree relative with Alzheimer’s disease, senility or dementia 1983/1985 – smoke, alcohol, caffeine BMI, hypertension, angina pectoris, heart attack, rheumatoid arthritis, cancer, HBP, stroke, diabetes Active/sedentary |
Fully adjusted, predicting incident dementia. Separate analyses for men and women No. natural teeth – NS (men and women) Inadequate natural dentition, no dentures, men: HR 1.91 (1.13–3.21), women: HR 1.22 (0.86–1.73) Inadequate dentition, alone or with dentures: – NS (men + women) Oral health habits (40 comparisons) No daily tooth brushing: HR 1.65 (1.05–2.62), women only. No dental visits last 12 months: HR 1.89 (1.21–2.95), men only |
| Yamamoto et al., 201223 Aichi Gerontological Evaluation Study (AGES) Six municipalities in Aichi province, Japan |
Baseline: 2003 N=4,425 (from mail survey of random sample of 9,783 community residents) Age: 65+ Sex: M=2,158; F=2,267 Race/ethnicity: Asian Follow up: 4-year cohort study Community residents |
Dementia onset: from administrative data -- Certified to receive insurance benefits, based on Ministry questionnaire on functional, cognitive, behavioral, communication problems in dementia. Graded I-IV, M (I=can still live independently, IV=consistent care needed, M=severe psychiatric symptoms, specialized medical service needed) |
Self-report ≥20 teeth/≤19 teeth Few teeth + denture use Few teeth no dentures Data missing Mastication: Chew anything/ cannot chew at all Have regular dentist (yes/no) Personal dental care good (yes/no) |
Age, gender, adjusted household income BMI, current illnesses (up to 18 conditions) Smoking history, alcohol use Exercise Forgetfulness |
Adjusted for all characteristics excluding “forgetfulness”. Predicting incident dementia. Only significant findings reported. Oral health status. Reference: ≥20 teeth Few teeth no dentures: HR 1.85 (1.04–3.29) Mastication Can’t chew very well: HR 1.47 (0.95–2.25) No regular dentist HR 1.44 (1.04–2.01) Poor personal dental care HR 2.01 (1.11–3.63) |
| Batty et al., 201314 ADVANCE trial Impact of reducing blood pressure, intensive glucose control, on Type II diabetes. 215 centers, 20 countries |
Baseline: 2001–2003 N=11,140 M=6,407; F=4,733 Age: 55–88 (all with type II diabetes and history of major macro- or microvascular disease, or at least 1 other cardiovascular risk factor) White (67%) (other ethnicities not reported) Followup: for 5 years |
MMSE (3 assessments over 5 years) If MMSE <24, or MD or nurse suspected dementia, referred to specialist for dementia evaluation, DSM IV criteria Cognitive decline = drop ≥ 3 MMSE points by 3rd assessment |
Baseline: Self-reported no. of natural teeth connected to gum or jawbone. No. days bleeding gums in past 12 mos. |
Age, sex, EQ-5D, socioeconomic, CVD risk factors: -behavioral -physiological -psychological |
Fully adjusted Incident dementia (N=109) No. of teeth Reference ≥22 teeth 0 teeth HR 1.48 (1.24–1.78) 1–21 teeth HR 1.24 (1.05–1.46) Bleeding gums ≥12 days HR 1.19 (0.51–2.75) <12 days HR 0.42 (0.10–1.71) Cognitive decline (N=1,806) No. of teeth 0 teeth HR 1.39 (1.21–1.59) 1–21 teeth HR 1.23 (1.10–1.38) Bleeding gums ≥12 days HR 0.94 (0.77–1.15) <12 days HR 0.92 (0.75–1.13) |
| Stewart et al., 201320 Memphis and Pittsburgh, US Health Aging & Body Composition (Health ABC) study |
Baseline: 1997/1998 N=947 (with cognitive data, y5) Age 70–79 M=~50%; F=~50% Race/ethnicity: Black 34%, White 66% Healthy Medicare community residents in local area, unimpaired basic ADL or mobility. Included all Blacks, and random sample of Whites Sample based on year 2 oral health participants Followup: year 5 |
Cognitive assessment: 1997/1998 (y1), 1999/2000 (y3), 2000/2001 (y5) Cognitive impairment: 3MS score <80 (bottom 10%) Cognitive decline: Decline ≥5 points (~20%) Year 3 -- Digit Symbol Substitution test (DSST), clock drawing Cognitive impairment = bottom 10% |
Dental examination 1998/99 (year 2) No. of teeth No. occluding tooth pairs Probing depth Attachment loss (no. & proportion of sites) Mean gingival index Mean plaque score No. sites bleeding on probing All measures categorized into quartiles |
Age, sex, education, race Self-reported cardiovascular disease and risk factors CES-D score C-reactive protein, IL- 6 at y1; weight loss y1-y3 Anticholinergic medication APOE ε4 |
Change in year1-year3 3MS 3MS cognitive impairment and decline; stratified by APOE ε4 genotype to ascertain effect modification. No significant association of any dental measure with cognitivedecline on 3MS Closest: no. of teeth, OR 0.88 (0.77– 1.00), gingival index OR 1.17 (0.99–1.38) DSST decline: no association Dichotomized gingival index (Q4/rest): associated with decline in 3MS, fully adjusted: OR 2.28 (1.60– 3.25) No association with DSST |
| COGNITIVE IMPAIRMENT PREDICTS ORAL DISEASE | |||||
| Study | Sample | Cognitive Assessment |
Oral Health Measures |
Primary covariates | Key findings |
| Jones et al., 19938 Dept. of Veterans Affairs (VA) Longitudinal Study of Dementia. VAMC, Bedford, MA Dept. of Veterans Affairs Dental Longitudinal Study – VA-DLS (subset of VA Normative Aging Study). Boston VA |
Baseline: 1986–1989 Dementia (AD) N=23 male veterans (from Dept. of Veterans Affairs Longitudinal Study of Dementia) Age: 67.4±7.5 Controls (matched on age, number of teeth, education) N=46 male veterans (VA-DLS) Age: 65±12 Followup: 23, 50 months Retrospective data |
AD cases drawn from VA Longitudinal Study of Dementia, with previously determined diagnosis. MMSE |
Initial caries examination: after teeth scaled and polished 50 month followup: caries examination (teeth not scaled or polished) NIDR criteria Katz criteria for root caries evaluation Annualized net caries increment (% per 100 available surfaces) |
Demographic (from medical records) |
Annualized followup: Veterans with AD had increased coronal caries increment 2.29±4.29 vs 0.88±1.14; root caries 2.38±5.57 vs. 0.31±0.69 per 100 available surfaces compared with control group. All findings NS. |
| Ship & Puckett, 199413 National Institute on Aging Normative Aging Program Also, Clinical Center, National Institutes of Health, Bethesda, MD |
Baseline: AD: N = 21 M=13; F=8 Age: 64±9 Controls (age, gender matched) N=21; M=13; F=8 (from National Institute on Aging Normative Aging Program Age: 65±12 Community residents, unmedicated Persons with other medical, neurological, psychiatric conditions excluded. Followup interval: AD 23±11 months Controls: 36±1.5 months |
NINCDS-ADRDA criteria for AD (vigorous screening) MMSE Change in MMSE score |
Unstimulated & stimulated major salivary gland flow rates Extensive assessment of gingival, periodontal (NIDR criteria), dental and oral mucosal tissues. Standard criteria and scales used in assessment. Test-retest reliability of assessment determined. Annualized change |
No association between change in salivary flow rate and change in MMSE Gingival health: Poorer MMSE scores associated with increased gingival bleeding and plaque. Periodontal health: no difference between groups Number of teeth, restored surfaces no difference Loss of tooth attachment: findings unclear Intraoral mucosal tissue: no difference between groups, but lower MMSE scores related to diminished mucosal health |
|
| Chalmers et al., 200221 “Oral health of community dwelling older adults with dementia” South Australian Dental Longitudinal Study (SADLS) Adelaide, Australia |
Baseline: 1998/99 116 dementia 116 no dementia Gender, age matched (M=66, F=50), age (≤79 = 91, ≥80 = 25) Followup: 1999/2000 103 dementia 113 no dementia. Exclusionary factors: Edentulous, too ill, refused Dementia cases -- in-home assessment only No dementia – in-home or clinic assessment Community residents |
Physician confirmed diagnosis Dementia: 76% AD, 24% other MMSE ≥26 no dementia 21–25 mild 11–20 moderate ≤10 severe dementia GDS |
Two calibrated dentists NIDR protocol Tooth status (present, absent, replaced, root status) Caries (initial, incident): coronal, root surface; sound, decayed, treated; gingival recession Assessed as crude caries increment (CCI), net caries increment (NCI), adjusted caries increment (ADJCI) Reliability of assessing caries incidence determined |
ADL (Katz) IADL (Lawton Brody) No. medical conditions Anticholinergics: (severity: none, low, high) Dental care issues Zarit Burden Scale Medical status, medication status, cognitive status |
Caries increments (dementia vs nondementia): Coronal: CCI=3.7 vs 1.5 NCI=3.5 vs 1.4 ADJCI=3.6 vs 1.4, all P<0.01. Incidence: 71.8% vs 48.7%, P<0.01. Root surfaces: CCI=1.9 vs 0.9 NCI=1.7 vs 0.8 ADJCI=1.8 vs 0.9, all P<0.01. Incidence: 62.1% vs 44.2%, P<0.01. Greater root caries incidence in high maintenance patients. Dementia group – coronal caries increment: 3.5 (for 1 increment in nondemented) (p=0.001) GDS moderate-severe dementia -- additional 1.78 coronal increments (p=0.01); 0.95 root caries increment (p = 0.001) |
| Ellefsen et al., 200922 Copenhagen, Denmark Two hospital memory clinics |
Baseline: 2002–2004 N=106 (AD=61, other dementia (OD)=26, no dementia=19) M=37, F=69 Followup: ~1 year later N = 77 (AD=49, other dementia=15, no dementia=13) M=28, F=49 Mean age: 81.9 (no information on age range) |
MMSE Dementia criteria: ICD- 10 AD vs. other dementia vs. no dementia |
Oral evaluation in home NIDR criteria for coronal and root decayed surfaces and filled surfaces, gingival recession. Calculated Adjusted Caries and filling Increments (ADJCIs) Number of teeth present, dental prostheses |
Demographic, functional, social variables from structured questionnaire |
No statistically significant differences for decayed and filled coronal and root surfaces, although rates higher for AD, OD, than for nondemented. ADJCIs higher for age 80+, but not always significant. No difference for MMSE <24 vs ≥24. Predicting coronal caries AD: OR 0.64 (0.09–4.50) OD: OR 4.13 (0.44–39.05) Predicting root caries AD: OR 0.08 (0.01–0.79) OD: OR 1.01 (0.11–9.56) |
| Chen et al., 201024 St. Paul, Minnesota USA Geriatric dental clinic affiliated with U Minnesota School of Dentistry, for age 55+ , persons with disabilities, community and nursing home residents. |
Study years: 10/1999- 12/2006, followup 1–90 months, mean 38.8 months N = 491 Dementia N=119 M=30, F=89 Mean age: 81.5±9.2 Range: 49–102 No dementia (propensity score matched) N=372 M=110, F=262 Mean age: 73.8±10.7 Range: 44–103 New patients, dentate after initial treatment, returned for routine care at least once. |
Medical history from dental records; diagnosis of AD, other dementia, CBS, or ICD- 9CM-R codes 290.x, 294.1, 331.2 Medical, cognitive, functional assessment from dental records |
From dental records Followup treatment focused on continued treatment as needed |
Charlson comorbidity index Anticholinergic drug scale (ADS), to measure medication- related anticholinergic burden |
Fully adjusted models No. teeth lost: HR 0.92 (0.59–1.63) P=0.99 Rate of tooth loss: P=0.52, slightly higher in dementia patients, but difference NS |
If there was no indication of race, or of number of men or women, the information was not mentioned in the paper.
Psychological measures for which results are not reported have been excluded to reduce space.
All abbreviations in the table have been spelled out below, together with references where appropriate.
Values in parentheses in this column indicate 95% confidence interval
3MS = Modified Mini-Mental State Examination: Teng EL, Chui HC. The Modified Mini-Mental State (3MS) Examination. J Clin Psychiatry 1987;48:314–318.
AD = Alzheimer’s disease
ADJCI = Adjusted Caries Increment, determined as CCI (crude caries increment [number of surfaces] (1-examiner reversals/examiner reversals + x [decayed/recurrent/filled/filled unsatisfactory/root sound])
ADL = activities of daily living: Katz S, Ford AB, Moskowitz RW et al. Studies of illness in the aged. The index of ADL: a standardized measure of biological and psychosocial functioning. J Am Med Assoc 1963;185:914–919.
ADS = Anticholinergic drug scale: Carnahan RM, Lund BC, Perry PJ et al. The Anticholinergic Drug Scale as a measure of drug-related anticholinergic burden: association with serum anticholinergic activity. J Clin Pharmacol 2006;46:1481–1486.
Anticholinergic drug scale: Carnahan RM, Lund BC, Perry PJ, Pollock BG, Culp KR. The anticholinergic drug scale as a measure of drug-related anticholinergic burden: associations with serum anticholinergic activity. J Clin Pharmacol 2006;46:1481–1486.
APOE = Apolipoprotein E
BentonVisual Retention Test: Benton AL. Manuel pour l’application du test de retention visuelle. Applications cliniques et expérimentales. 2e edition franaise. Paris, Centre de Psycholgie Appliquée, 1965.
BMI = body mass index (weight[kg]/height[m2])
Braak staging: Braak H, Braak E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol (Berl) 1991;82:239–59.
CBS = chronic brain syndrome
CCI = crude caries increment
CERAD: Morris JC, Heyman A, Mohs RC et al. The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part 1. Clinical and Neuropsychological Assessment of Alzheimer’s Disease. Neurology 1989;39:1159–1165.
CES-D = Center for Epidemiologic Studies-Depression Scale: Radloff LS. The CES-D scale: a self-report depression scale for research in the general population. J Appl Psychol Meas 1977;1:385–401.
Charlson comorbidity index: Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chron Dis 1987;40:373–383.
CHD = coronary heart disease
CPI = Community Periodontal Index: World Health Organization, Community Periodontal Index (CPI) in Oral Health Surveys – Basic methods, World Health Organization, Editor. 1997: Geneva. Pp.36–38.
CVD = cerebrovascular disease
Debris Index: Green JC, Vermillion JR. The oral hygiene index: a method for classifying oral hygiene status. J Am Dent Assoc 1957;61:171–179.
DSST = Digital Symbol Substitution Test
Digit symbol test: Wechsler D. WAIS-R Manual. New York, Psychological Corporation, 1981.
EQ-5D = Quality of life: The EuroQol Group. EuroQol-a new facility for the measurement of healthrelated quality of life. Health Policy 1990;16:199–208. See http://www.euroqol.org/ for most recent information. Accessed October 21, 2014
GDS = Global Deterioration Scale: Reisberg B, Ferris SH, DeLeon MJ, et al. The Global Deterioration Scale (GDS): an instrument for the assessment of primary degenerative dementia. Am J Psychiatry 1982;139:1136–1139.
Gingival Index: Loe H. The Gingival Index, the Plaque Index and the Retention Index systems. J Periodontol 1967;38:610–616.
HBP = High blood pressure
HR = Hazard ratio, analysis in which time to an event is taken into account
IADL = instrumental activities of daily living
IL-6 = interleukin-6, an inflammatory marker
Katz criteria for root caries: Katz RV. Assessing root caries in populations: the evolution of the root caries index. J Pub Hlth Dent 1980; 40:7–16.
L-B = Lawton MP, Brody E. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontotogist 1969;9:179–186.
MMSE: Folstein MF, Folstein SE, McHugh PR. ‘Mini-Mental State’. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975;12:189–198.
NCI = net caries increment
NIDR = National Institute of Dental Research
NINCDS-ADRDA = National Institute of Neurological, Communicative Disorders, and Stroke-Alzheimer’s Disease and Related Disorders Association
NSAID = Non-steroidal anti-inflammatory drug
OR = Odds ratio, analysis concerned with whether an event has taken place
VA = Veterans Administration
VA-DLS = Veterans Administration Dental Longitudinal Study
VAMC = Veterans Administration Medical Center
Verbal Fluency: Isaacs B, Kennie AT. The Set Test as an aid to detecting dementia in old people. J Psychiatry 1973;123:467–470.
Visuo-spatial attention: Zazzo R. Test des deux barrages. Actualités pédagogique et psychologiques, vol 7. Neuchâtel, Delachaux et Nestlé, 1974.
Wechsler Paired Associates: Wechsler D. A standardized scale for clinical use. J Psychol 1945;19:87–95.
Zarit Burden Scale: Zarit SH, Reever KE, Bach-Peterson J. Relatives of the impaired elderly: correlates of feelings of burden. Gerontologist 1980;20:649–655.
Several developed countries are represented: U.S. (n=8), Japan (n=3), Australia (n=1), France (n=1), Denmark (n=1), Sweden (n=1), and a European consortium (n=1). Two studies used the same sample, but different outcomes and statistical analyses.9,10 All but four studies9–12 were conducted among community residents, but some community-based studies may have included nursing home residents. Sample sizes ranged from small (N=42)13 to large (N=11,140, from 215 centers in 20 European countries).14
Subject selection and source of data reflected several creative, overlapping approaches. These included use solely of administrative data, 15 use of data already gathered in longitudinal studies (often designed for other purposes), 9,10,16–20 selecting subjects for whom cognitive status or oral health status had been established and adding an oral or cognitive evaluation, 8,11,13,21,22 and linking subject data to administrative records.23
Study design varied: intervention studies of good oral care;12,24 samples matched on demographics, by randomization,8,12,13,21 or through use of propensity scores;24 and natural history studies.9–11,14–17,19,20,22,23
Measures of Oral Health Status (information is summarized in on-line Table S1.)
Of the 16 studies, nine used oral health evaluation, and seven used dental records. In studies gathering oral health data, examiners were trained, calibrated if more than one participated, and reliability determined for assessing a variety of oral health measures. The amount of oral health information gathered in person or from records, and used in analysis, varied from extensive and highly detailed, particularly in studies of oral health predicting cognitive impairment (number of decayed, missing, filled teeth; plaque; periodontal disease), 8,13,20,22 to minimal (self-reported approximate number of teeth currently or when younger). 14,15,19,23 Subject-provided information included pain or discomfort, 17 bleeding gums, 14 and denture use.11,19,23 Information from dental records was used in three studies, 9,10,24 two of which relied on up to 40 years of records.9, 10, 25
Tooth loss was the most common measure of oral health, recorded in 14 of the 16 studies, with a significant relationship to cognition reported in 11.9–11,13,15–17,19,20,22,23 When used in analysis, the number of teeth present was typically categorized, with categorization varying from study to study, and sometimes linked to denture use. 9,11,15,17
Eleven studies used clinical evaluation to assess oral health status.8–13,16,17,20,21,24 In some studies, standard oral health indices were used: Debris Index for plaque score, 12 Community Periodontal Index, 17 Gingival Index, 20 Plaque Index, 20 Katz criteria for roots, and adjusted caries increment, 21 and National Institute for Dental Research (NIDR) criteria.8,13,21,22
In examining periodontal disease, attention was paid to 1) extent of gingival bleeding on probing (ratio of examined sites), 2) proportion of examined sites with loss of attachment >3 mm, and 3) mean pocket depth. Periodontal diseases were assessed in five studies, four on the impact of oral health on cognitive impairment (three studies used clinical assessment, 16,17,20 one used self-report14) and in one study of the effect of cognitive impairment on oral health.13 In addition, two studies assessed periodontal damage based on decades of dental records.9,10
Measures of Cognitive Assessment (information is summarized in on-line Table S2.)
Assessment of the presence of dementia included self-reported diagnosis, use of standard clinical diagnostic criteria, neuropathological diagnostic criteria, and information from medical, dental, and administrative records. Cognitive status was based on a variety of neuropsychology measures, ranging from use of a substantial battery to reliance on a single brief measure.
The standard diagnostic criteria for dementia included DSM-III-R, DSM-IV, ICD-9CM-R, ICD-10, reflecting the date of evaluation.13–15,17,22 At times these criteria were supplemented by NINCDS-ADRDA (Alzheimer’s disease [AD]), 13,15,17 NINDS-AIREN (vascular dementia), 15 Hachinski scale (vascular dementia),17 Lund & Manchester (frontal lobe dementia),15 Geriatric Mental State examination,17 and the Global Deterioration Scale,26 which assesses both presence and severity of dementia.21,22 Neuropathological criteria27 were also used, but available for only one dataset.9
Non-AD dementias15,17,22 were rarely analyzed separately because of limited numbers. The validity of administrative records, which were frequently used, was evaluated for only one study.15 Both cognitive batteries and individual measures were used to assess cognitive change and ascertain the presence of dementia. The most popular single measure was the Mini-Mental State Exam (MMSE), 28 used in 10 studies – in eight as a single measure, four of which examined the effect of oral condition on cognitive state, 12,14,16,19 and four the reverse, 8,13,21,22 and in two studies as part of a larger neuropsychology battery.9,17 All neuropsychology measures had established validity and reliability. Criteria used to assess cognitive change included self-perceived deterioration, change in score on neuropsychology measures, a specific decline (which differed across studies), crossing a cut-point (with cut-points differing across studies), or reaching an administrative criterion indicating need for assistance.
Covariates Used in Analysis (summarized in online Table S3.)
The covariates used varied widely across studies. They included demographic characteristics (sex, race, education, income, -- none uniformly reported); and specific health conditions and associated risk factors or the number of health conditions present (12 studies), and the Charlson Index.31 Also considered were biomarker and genetic information (interleukin (IL)-6, C-reactive protein, APOE ε4, 1st degree kin with dementia); 9,10,19,20 prescription and over-the-counter medications, particularly anticholinergics;16,20,21,24 scales of depression and caregiver burden;14,17,21 and measures of life style and health behaviors (included by 10 studies). No study included all these areas.
Findings
The impact of oral health on cognitive status (11 articles)
Two studies indicated that having fewer teeth at baseline or decline in number of teeth, was not associated significantly with cognitive decline.11,20 This was contradicted by other studies which found an association with cognitive decline, 10,16 and with dementia.14,15,23 Findings are further complicated: one reported an association only in women; 19 another reported that with higher education the number of missing teeth was not associated with dementia, but among those with lower education, individuals with a greater number of missing teeth were less likely to have dementia;17 and a third reported that having fewer teeth increased the hazard of dementia, but not when periodontal damage was present.9 Development of caries or the presence of new restoration was associated with lower performance on the MMSE and a spatial copying task.16 Mastication difficulty was not associated with incident dementia.23
Five studies examined the effect of periodontal disease on incident dementia, cognitive decline, or low level of cognitive functioning.9,14,16,17,20 Two of them found some indication of a relationship: alveolar bone loss and pocket depth was associated with poor cognitive functioning,16 the other found a significant decline on the Modified Mini-Mental State (3MS) Examination (which assesses several areas of cognition), but only when the continuous form of the gingival index was categorized, and the bottom quartile compared with the rest; no significant decline was found on the Digit Symbol Substitution Test.20
Three studies asked about or provided personal dental care. An intervention in nursing homes to make sure that teeth, dentures, and the oral cavity were cleaned, and plaque and calculus controlled, found a significantly smaller decline in MMSE score in the group receiving this oral care, however, variation was large and the numbers present at study end were unclear.12 In one study, poor oral hygiene and no regular visits to the dentist predicted incident dementia.23 Similar findings were reported in another study but were sex-specific -- no daily tooth brushing (women only), and no dental visits in the previous 12 months (men only) predicted incident dementia.19
The impact of cognitive status on oral health (5 articles)
Sample sizes tended to be small when examining the impact of cognitive status on longitudinal oral status, while the number of oral outcomes of interest was large. One study, which provided routine dental care and treatment, focused specifically on tooth loss; neither number of teeth lost, nor rate of loss differed significantly between the dementia and no dementia groups.24 Other studies also found no statistically significant differences in number of teeth or restored surfaces.13,22
Considerable attention was paid to increase in the number of caries, which, variously, was higher in patients with dementia,21 higher but not significant because of small sample size,8 or not significantly different.22 Poorer MMSE scores were associated with increased gingival bleeding and plaque and diminished mucosal health, but with small sample size these findings were only suggestive.12,14,16
Assessment of strength of the eligible studies
We used a set of criteria to assess each of the studies, including sample selection, adequate sample size, description of the sample, validated assessment of the exposure (e.g., cognitive status or oral health measure) and outcome (e.g., cognitive status or oral health measure), length of follow-up period, attrition, controls for confounding, and appropriate analytic approach.40 The evidence suggests that there are methodological deficiencies in this area, most notably due to small sample size or lack of representativeness of the population, inadequate assessment of cognitive function and lack of clinical evaluation of oral health. Thus, there was insufficient evidence to date for a causal association between cognitive function and oral health.
DISCUSSION
Our systematic review of the first two decades of longitudinal studies designed to examine the association between oral health and cognitive status identified 16 studies, 11 examined the effect of oral status on change in cognitive status, and five the reverse. The uneven distribution likely reflects the costs and difficulties of carrying out oral health assessments. It is easier and cheaper to add cognitive assessments to a study for which oral health has already been measured, than to do the reverse.
It is currently unclear how or whether oral health conditions and cognitive status are related. Findings based on number or change in number of teeth or caries, are conflicting, or, because of inadequate sample size, do not reach statistical significance. Limited studies (two out of five) found periodontal conditions, such as gingival health or pocket depth, was associated with poorer cognitive status or cognitive decline, with findings possibly sensitive to the neuropsychology assessments used. There are indications, albeit weak, that better oral hygiene and regular dental visits may reduce the rate of cognitive decline, and the hazard of incident dementia.12,19,23 Certainly, with good dental care, the dental status of patients with dementia remained comparable to that of patients without a dementing disorder.24 These findings may reflect that incipient cognitive decline or dementia leads to decline in hygiene, but that oral health can be maintained with self or assisted oral hygiene, and dental appointments.
While the argument of a common underlying cause associated with inflammation may be enticing,1,2 it was examined directly in only one study, and there found to be only marginally relevant.20 Current studies are not adequate to indicate whether poor oral health and cognitive decline have a common underlying cause in inflammation, but with the increase in major surveys with biomarker data, test of this hypothesis is becoming more feasible.
Because of the costs of detailed dental examination, there may be a temptation to carry out small sample studies. As seen in the current review, this results in a lack of power. There is also a temptation to match cases and controls, but matching must be considered carefully. Doing so eliminates the possibility of examining the “matching” conditions (e.g., demographic characteristics), which may be associated with both oral health and cognitive status. With an older population, the duration of a study becomes an issue because of incidence of additional chronic conditions which may affect oral health and cognition, attrition due to sickness, and death.
To better compare findings across studies, agreement is needed regarding the oral health issues to be explored, oral health and neuropsychological measures to use, and covariates for which to adjust. In particular, to reduce the costs of dental studies, additional information is needed on the extent to which self-report accurately determines oral health condition, oral hygiene behavior, and dental visits. While it appears that self-assessment of obvious conditions (e.g., number of teeth), and those that are salient (e.g., root canal, dentures) can be reported reliably (with accuracy varying by demographic status), report is less reliable for conditions requiring oral health expertise (e.g., caries, periodontal disease).32,33 However, since no studies have been conducted to examine the reliability of self-reported data among individuals with cognitive impairment, we are less certain how reliable the self-reported oral health data is in such individuals.
There is a considerable literature on assessment of cognitive state, with agreement on preferred measures to use in Memory Disorders clinics to assess dementing disorders,34 and procedures to follow in epidemiological studies, such as diagnostic adjudication. With increasing information on dementia, diagnostic criteria are changing, and diagnosis at an earlier stage of disease is becoming possible. Consequently, the characteristics of diagnosed subjects may change, with findings on an oral health/dementia association changing also.
Standardization of assessment of cognitive status should be considered. The most frequently used measure has been the MMSE,28 indicating preference for a brief, easy to administer assessment. The MMSE has several drawbacks: multiple versions with unknown equivalence; performance, as with many brief cognitive screens, that is age-, race-, and education-biased; a ceiling effect, and payment required for use. Improved screens, such as the Montreal Cognitive Assessment35 have become available since data were gathered for many of the current studies, and these screens should be considered. Agreement on use of a brief screen would facilitate comparison of findings, and aggregation of data.
Covariates varied from study to study, with a large variety reported: demographic, health conditions (in particular cardiovascular conditions and risk factors), medications (anticholinergics), health behaviors, and an assortment of other characteristics (e.g., scales of depression, caregiver burden). Certain relevant information was rarely included: e.g., nutrition, which affects oral health and general health, and environmental characteristics (dental insurance, access to dentists, cultural expectations regarding dentition). Although many studies have found self-report of medical conditions to be valid,36 caution is needed regarding medical and dental records and administrative data. Diagnosis of dementia based on administrative records which determine level of care to be provided may yield over-diagnosis, while dementing disorders may be under-reported in administrative medical records and death certificates.37,38 Agreement is needed on which covariates to include in analysis, and the manner in which they should be handled, in particular, regarding categorization of age, and education.
Current studies that capitalize on already available information on oral health, cognition, or biomarkers, illustrate how sample size and possibly representativeness can be increased, and costs reduced. In addition to secondary analysis of data,14,16,20 some studies have used administrative records with known sensitivity and specificity for diagnosis of dementia,15 while others have linked the main study data to dental records.9,10 Although there is no report as to whether those dental records were evaluated, studies that have examined pre-existing radiographs have found them to be usable for assessment of periodontal disease.39
This is the first systematic review to examine longitudinal studies focused on oral health and cognition. As such, it shows substantial diversity across studies with regard to data and sample sources, sample size, and variety of oral health data and cognitive data obtained. In summary, the strength of the evidence is weak, and findings were often inconsistent. For example, some studies found a significant association, albeit relatively weak, between periodontal disease and cognitive decline; but other studies found no association. Similarly the association between dental caries and cognition was inconsistent across studies. Findings are even more complex for the association between tooth loss and cognitive decline, ranging from a positive association, to no association, to a negative association (only in the group with a lower level of education) across different studies. However, these studies lay the groundwork for future longitudinal work.
While research in the field appears to be moving in the right direction, we would like to make some specific suggestions, operationalization of which may permit more accurate determination of the grounds for an association, if one is present, between oral health and cognitive status. In the current studies, cognitive function has been evaluated using a great variety of cognitive measures. Information could be better evaluated if there was agreement on a uniform set of cognitive assessments. When change in cognition is assessed, it may be preferable to examine decline in cognitive function (which is more straightforward to assess), instead of development of dementing disorders, with attendant complexities in diagnosis. Regarding the assessment of oral health status, several studies used standardized oral health examination protocols, such as the U.S. National Institute of Dental Research protocol,21,22, a procedure which should be emphasized. Future research should pay more attention to calibration of dental assessment, both between and within dental evaluators. The oral health outcomes used should reflect oral health status which can change during the study period. With respect to other aspects of study design, use of standard covariates, samples sized adequately for the number and type of outcomes to be measured and better representative of the study population, a study duration appropriate to the planned outcome, and sophisticated statistical approaches already in use should be encouraged.
Supplementary Material
Acknowledgments
Funding sources: This study was supported by NIH/NIDCR grant number 1R01DE019110 (BW), NIH/NINR grant number 1P30NR014139 (BW), and NIH/NIA grant number 1P30AG028716 (GGF). This study was presented at the annual scientific meeting of the Gerontological Society of America, 2013. We would like to thank Qin Lu for his assistance with the literature review.
Sponsor’s role: No sponsor was involved in the design, methods, subject recruitment, data collections, analysis or preparation of this paper.
Footnotes
Conflict of Interest: The editor in chief has reviewed the conflict of interest checklist provided by the authors and has determined that the authors have no financial or any other kind of personal conflicts with this paper.
Author contributions:
- Substantial contributions to conception/design, acquisition of data, analysis and interpretation of data: Bei Wu, Gerda G. Fillenbaum, Brenda L. Plassman, Liang Guo
- Drafting the article or revising it critically for important intellectual content: Bei Wu, Gerda G. Fillenbaum, Brenda L. Plassman, Liang Guo
- Final approval of the version to be published: Bei Wu, Gerda G. Fillenbaum, Brenda L. Plassman, Liang Guo
REFERENCES
- 1.Kamer AR, Craig RG, Dasanayake AP, et al. Inflammation and Alzheimer’s disease: Possible role of periodontal diseases. Alzheimers Dement. 2008;4:242–250. doi: 10.1016/j.jalz.2007.08.004. [DOI] [PubMed] [Google Scholar]
- 2.Noble JM, Scarmeas N, Papapanou PN. Poor oral health as a chronic, potentially modifiable dementia risk factor: Review of the literature. Curr Neurol Neurosci Rep. 2013;13:1–8. doi: 10.1007/s11910-013-0384-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wu B, Liang J, Landerman L, et al. Trends of edentulism among middle-aged and older Asian Americans. Am J Public Health. 2013;103:e76–e82. doi: 10.2105/AJPH.2012.301190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Plassman BL, Langa KM, Fisher GG, et al. Prevalence of cognitive impairment without dementia in the United States. Ann Intern Med. 2008;148:427–434. doi: 10.7326/0003-4819-148-6-200803180-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Plassman BL, Langa KM, Fisher GG, et al. Prevalence of dementia in the United States: The aging, demographics, and memory study. Neuroepidemiology. 2007;29:125–132. doi: 10.1159/000109998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Daviglus M, Bell C, Berrettini W, et al. NIH State-of-the-Science Conference Statement: Preventing Alzheimer’s disease and cognitive decline. NIH Consensus and State-of-the-Science Statements. 2010;27:1–30. [PubMed] [Google Scholar]
- 7.Hebert LE, Weuve J, Scherr PA, et al. Alzheimer disease in the United States (2010–2050) estimated using the 2010 census. Neurology. 2013;80:1778–1783. doi: 10.1212/WNL.0b013e31828726f5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jones JA, Lavallee N, Alman J, et al. Caries incidence in patients with dementia. Gerodontology. 1993;10:76–82. doi: 10.1111/j.1741-2358.1993.tb00086.x. [DOI] [PubMed] [Google Scholar]
- 9.Stein PS, Desrosiers M, Donegan SJ, et al. Tooth loss, dementia and neuropathology in the Nun study. J Am Dent Assoc. 2007;138:1314–1322. doi: 10.14219/jada.archive.2007.0046. [DOI] [PubMed] [Google Scholar]
- 10.Stein P, Kryscio R, Desrosiers M, et al. Tooth loss, apolipoprotein E, and decline in delayed word recall. J Dent Res. 2010;89:473–477. doi: 10.1177/0022034509357881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shimazaki Y, Soh I, Saito T, et al. Influence of dentition status on physical disability, mental impairment, and mortality in institutionalized elderly people. J Dent Res. 2001;80:340–345. doi: 10.1177/00220345010800010801. [DOI] [PubMed] [Google Scholar]
- 12.Yoneyama T, Yoshida M, Ohrui T, et al. Oral care reduces pneumonia in older patients in nursing homes. J Am Geriatr Soc. 2002;50:430–433. doi: 10.1046/j.1532-5415.2002.50106.x. [DOI] [PubMed] [Google Scholar]
- 13.Ship JA, Puckett SA. Longitudinal study on oral health in subjects with Alzheimer’s disease. J Am Geriatr Soc. 1994;42:57–63. doi: 10.1111/j.1532-5415.1994.tb06074.x. [DOI] [PubMed] [Google Scholar]
- 14.Batty GD, Li Q, Huxley R, et al. Oral disease in relation to future risk of dementia and cognitive decline: Prospective cohort study based on the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified-Release Controlled Evaluation (ADVANCE) trial. Eur Psychiatry. 2013;28:49–52. doi: 10.1016/j.eurpsy.2011.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gatz M, Mortimer JA, Fratiglioni L, et al. Potentially modifiable risk factors for dementia in identical twins. Alzheimers Dement. 2006;2:110–117. doi: 10.1016/j.jalz.2006.01.002. [DOI] [PubMed] [Google Scholar]
- 16.Kaye EK, Valencia A, Baba N, et al. Tooth loss and periodontal disease predict poor cognitive function in older men. J Am Geriatr Soc. 2010;58:713–718. doi: 10.1111/j.1532-5415.2010.02788.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Arrivé E, Letenneur L, Matharan F, et al. Oral health condition of French elderly and risk of dementia: A longitudinal cohort study. Community Dent Oral Epidemiol. 2012;40:230–238. doi: 10.1111/j.1600-0528.2011.00650.x. [DOI] [PubMed] [Google Scholar]
- 18.Batty GD, Li Q, Czernichow S, et al. Erectile dysfunction and later cardiovascular disease in men with type 2 diabetes prospective cohort study based on the ADVANCE (Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified-Release Controlled Evaluation) Trial. J Am Coll Cardiol. 2010;56:1908–1913. doi: 10.1016/j.jacc.2010.04.067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Paganini-Hill A, White SC, Atchison KA. Dentition, dental health habits, and dementia: The Leisure World Cohort Study. J Am Geriatr Soc. 2012;60:1556–1563. doi: 10.1111/j.1532-5415.2012.04064.x. [DOI] [PubMed] [Google Scholar]
- 20.Stewart R, Weyant RJ, Garcia ME, et al. Adverse oral health and cognitive decline: The health, aging and body composition study. J Am Geriatr Soc. 2013;61:177–184. doi: 10.1111/jgs.12094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chalmers JM, Carter KD, Spencer AJ. Caries incidence and increments in community-living older adults with and without dementia. Gerodontology. 2002;19:80–94. doi: 10.1111/j.1741-2358.2002.00080.x. [DOI] [PubMed] [Google Scholar]
- 22.Ellefsen B, Holm-Pedersen P, Morse DE, et al. Assessing caries increments in elderly patients with and without dementia: A one-year follow-up study. J Am Dent Assoc. 2009;140:1392–1400. doi: 10.14219/jada.archive.2009.0076. [DOI] [PubMed] [Google Scholar]
- 23.Yamamoto T, Kondo K, Hirai H, et al. Association between self-reported dental health status and onset of dementia: A 4-year prospective cohort study of older Japanese adults from the Aichi Gerontological Evaluation Study (AGES) Project. Psychosom Med. 2012;74:241–248. doi: 10.1097/PSY.0b013e318246dffb. [DOI] [PubMed] [Google Scholar]
- 24.Chen X, Shuman SK, Hodges JS, et al. Patterns of tooth loss in older adults with and without dementia: A retrospective study based on a Minnesota cohort. J Am Geriatr Soc. 2010;58:2300–2307. doi: 10.1111/j.1532-5415.2010.03192.x. [DOI] [PubMed] [Google Scholar]
- 25.Snowdon DA, Greiner LH, Mortimer JA, et al. Brain infarction and the clinical expression of Alzheimer disease: The Nun Study. JAMA. 1997;277:813–817. [PubMed] [Google Scholar]
- 26.Reisberg B, Ferris SH, de Leon MJ, et al. The Global Deterioration Scale for assessment of primary degenerative dementia. Am J Psychiatry. 1982;139:1136–1139. doi: 10.1176/ajp.139.9.1136. [DOI] [PubMed] [Google Scholar]
- 27.Braak H, Braak E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991;82:239–259. doi: 10.1007/BF00308809. [DOI] [PubMed] [Google Scholar]
- 28.Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–198. doi: 10.1016/0022-3956(75)90026-6. [DOI] [PubMed] [Google Scholar]
- 29.Dartigues JF, Gagnon M, Barberger-Gateau P, et al. The Paquid epidemiological program on brain ageing. Neuroepidemiology. 1992;11(Suppl. 1):14–18. doi: 10.1159/000110955. [DOI] [PubMed] [Google Scholar]
- 30.Morris J, Heyman A, Mohs R, et al. The Consortium to Establish a Registry for Alzheimer’s. Disease (CERAD). Part I. Clinical and neuropsychological assesment of Alzheimer’s disease. Neurology. 1989;39:1159–1165. doi: 10.1212/wnl.39.9.1159. [DOI] [PubMed] [Google Scholar]
- 31.Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J Chronic Dis. 1987;40:373–383. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- 32.Liu H, Maida CA, Spolsky VW, et al. Calibration of self-reported oral health to clinically determined standards. Community Dent Oral Epidemiol. 2010;38:527–539. doi: 10.1111/j.1600-0528.2010.00562.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pitiphat W, Garcia RI, Douglass CW, et al. Validation of self-reported oral health measures. J Public Health Dent. 2002;62:122–128. doi: 10.1111/j.1752-7325.2002.tb03432.x. [DOI] [PubMed] [Google Scholar]
- 34.Weintraub S, Salmon D, Mercaldo N, et al. The Alzheimer’s Disease Centers’ uniform data set (UDS): The neuropsychological test battery. Alzheimer Dis Assoc Disord. 2009;23:91–101. doi: 10.1097/WAD.0b013e318191c7dd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695–699. doi: 10.1111/j.1532-5415.2005.53221.x. [DOI] [PubMed] [Google Scholar]
- 36.Baumeister H, Kriston L, Bengel J, et al. High agreement of self-report and physician-diagnosed somatic conditions yields limited bias in examining mental-physical comorbidity. J Clin Epidemiol. 2010;63:558–565. doi: 10.1016/j.jclinepi.2009.08.009. [DOI] [PubMed] [Google Scholar]
- 37.Taylor DH, Jr, Fillenbaum GG, Ezell ME. The accuracy of medicare claims data in identifying Alzheimer’s disease. J Clin Epidemiol. 2002;55:929–937. doi: 10.1016/s0895-4356(02)00452-3. [DOI] [PubMed] [Google Scholar]
- 38.Romero JP, Benito-León J, Louis ED, et al. Under reporting of dementia deaths on death certificates: A systematic review of population-based cohort studies. J Alzheimers Dis. 2014;41:213–221. doi: 10.3233/JAD-132765. [DOI] [PubMed] [Google Scholar]
- 39.Pitiphat W, Crohin C, Williams P, et al. Use of preexisting radiographs for assessing periodontal disease in epidemiologic studies. J Public Health Dent. 2004;64:223–230. doi: 10.1111/j.1752-7325.2004.tb02757.x. [DOI] [PubMed] [Google Scholar]
- 40.Williams JW, Plassman BL, Burke J, et al. Preventing Alzheimer’s disease and cognitive decline. Evidence Report/Technology Assessment No. 193 (Prepared by the Duke Evidence-based Practice Center under Contract No. HHSA 290-2007-10066-I.) AHRQ Publication No. 10-E005. Rockville, MD: Agency for Healthcare Research and Quality; 2010. Apr, [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.