Significance
Eukaryotic transcripts contain spliceosomal introns that need to be removed by pre-mRNA splicing. Although several models have been proposed to identify the mechanism of intron gain over the evolution of eukaryotes, they remain models due to a lack of experimental validation. We developed a reporter system to detect selected intron gain and loss events and captured two intron gain events in which the intron derived from the reporter was transposed into the chromosomal loci of RPL8B and ADH2. This is, to our knowledge, the first demonstration of intron gain via intron transposition in any organism, and we suggest that these events are likely to have occurred by a reversal of the pre-mRNA splicing reaction followed by homologous recombination.
Keywords: spliceosome, intron, evolution
Abstract
The presence of intervening sequences, termed introns, is a defining characteristic of eukaryotic nuclear genomes. Once transcribed into pre-mRNA, these introns must be removed within the spliceosome before export of the processed mRNA to the cytoplasm, where it is translated into protein. Although intron loss has been demonstrated experimentally, several mysteries remain regarding the origin and propagation of introns. Indeed, documented evidence of gain of an intron has only been suggested by phylogenetic analyses. We report the use of a strategy that detects selected intron gain and loss events. We have experimentally verified, to our knowledge, the first demonstrations of intron transposition in any organism. From our screen, we detected two separate intron gain events characterized by the perfect transposition of a reporter intron into the yeast genes RPL8B and ADH2, respectively. We show that the newly acquired introns are able to be removed from their respective pre-mRNAs by the spliceosome. Additionally, the novel allele, RPL8Bint, is functional when overexpressed within the genome in a strain lacking the Rpl8 paralogue RPL8A, demonstrating that the gene targeted for intronogenesis is functional.
One of the defining features of all eukaryotic organisms is the presence of intervening sequences termed introns in at least some nuclear genes (1, 2). The removal of introns from eukaryotic pre-mRNA within the spliceosome is mechanistically related to self-cleaving group II introns from prokaryotes and eukaryotic organelles (3–5) (Fig. S1). Although much work has been done in examining the mechanism and machinery of spliceosomal intron removal (1, 2), several evolutionary mysteries remain regarding introns: How did spliceosomal introns invade and persist in eukaryotic genomes? How are they removed from the genomes of organisms undergoing intron loss? Have introns been added over evolutionary time, and if so, how does that occur?
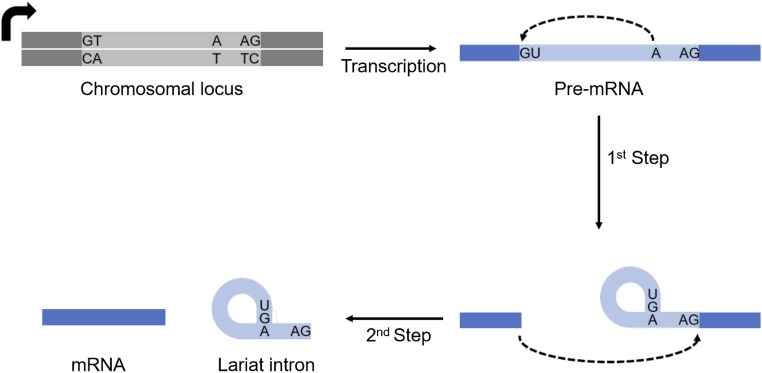
Fig. S1.
Pre-mRNA splicing mechanism. Pre-mRNA splicing occurs by two ordered transesterification steps. In the first step, the 2′-OH of the branch point adenosine performs a nucleophilic attack at the 5′ splice site, yielding a lariat intermediate RNA and a 5′ exon. The second step takes place when the 3′-OH of the 5′ exon attacks the 3′ splice site, ligating the exons and releasing the lariat intron.
Several models exist for how introns might be lost (6–8), and experimental intron loss has been demonstrated in at least one organism (9, 10) (Fig. S2). Indeed, at this time in evolution, budding yeast is a species in which widespread intron loss has been proposed to have occurred, likely through RNA-mediated homologous recombination of cDNA (9, 11). This model is likely to be correct, as it reflects the genomic reality that budding yeast introns generally exist close to the 5′ end of intron-containing genes, as would be expected in a reaction mediated by reverse transcriptase, which begins copying the mRNA from the 3′ end (7, 12–14).
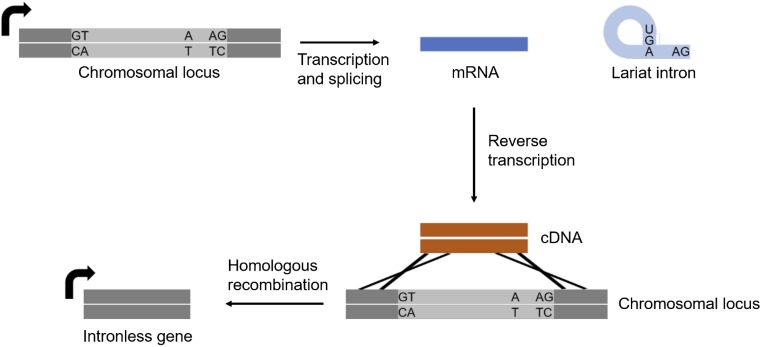
Fig. S2.
Proposed mechanism of intron loss. After an intron-containing gene is transcribed and spliced, the resulting mRNA is reverse transcribed to the cDNA by cellular reverse transcriptase. The cDNA is then incorporated into the genome by homologous recombination, leading to the generation of a new, intronless allele.
Models for how introns are gained are numerous, however no model has yet been experimentally validated (15, 16). These models include intron transposition, intron gain during double-stranded DNA break repair, transfer of an intron from a paralogous gene, and an appealing model involving reverse-splicing. This model invokes the incorporation of an intron retained in the residual spliceosome into an intron-naïve mRNA that has encountered and stably interacted with this species (17) (Fig. S3). Although all of these models could potentially lead to intronogenesis, none have yet been demonstrated in vivo.
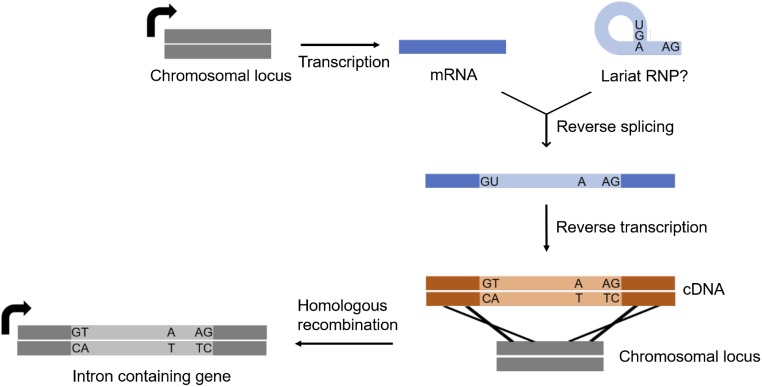
Fig. S3.
Proposed mechanism of intron gain. The model involving reverse splicing employs a spliced-out lariat RNA as the source of a new intron gain. This lariat RNA remains engaged in the residual spliceosome after mRNA release. Reverse splicing and subsequent reverse transcription generate the cDNA copy containing the newly inserted intron. Homologous recombination between the genome and cDNA copy yields a new intron gain allele.
Most of the published work on the phenomena of intron gain and loss has used phylogenetic comparisons of intron presence and position across intron-containing genes, made possible by the existence of extensive genome sequence databases (16). Definitive conclusions of intron gain or loss are difficult to make by these analyses, however it is clear that introns massively infiltrated the genome of the last eukaryotic common ancestor and that introns have continued to be gained and lost over evolutionary time (11, 18).
Here we report the use of a reporter system designed to detect events of intron gain and intron loss in the budding yeast Saccharomyces cerevisiae. With this reporter, we have captured, to our knowledge, the first verified examples of intron gain via intron transposition in any eukaryote.
Results
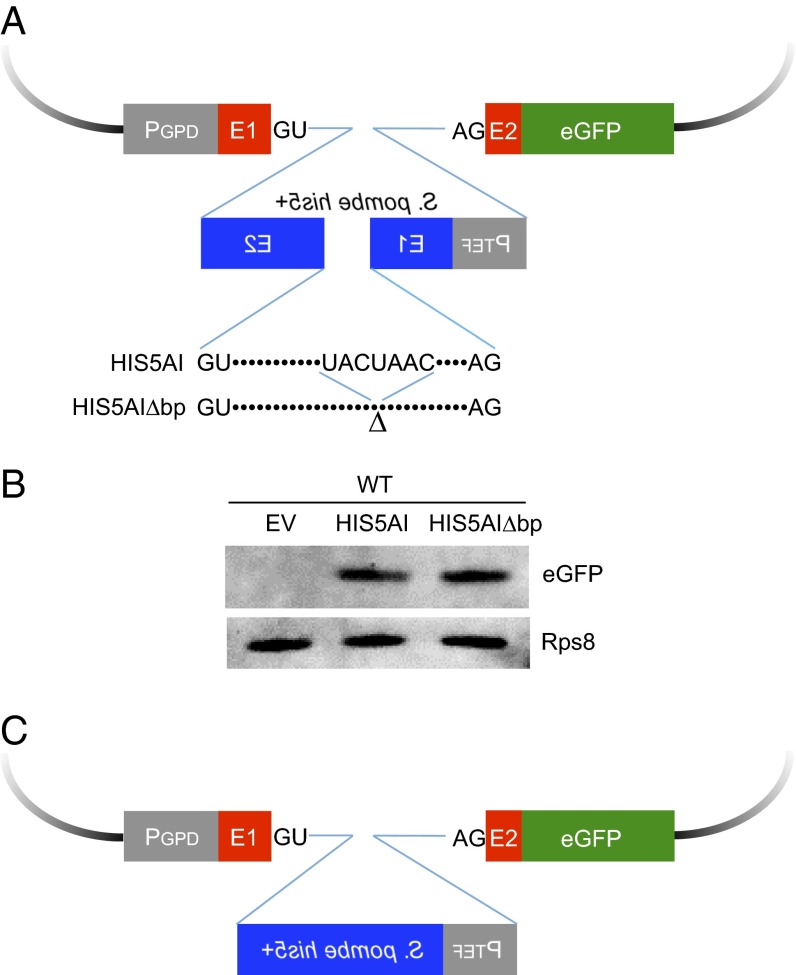
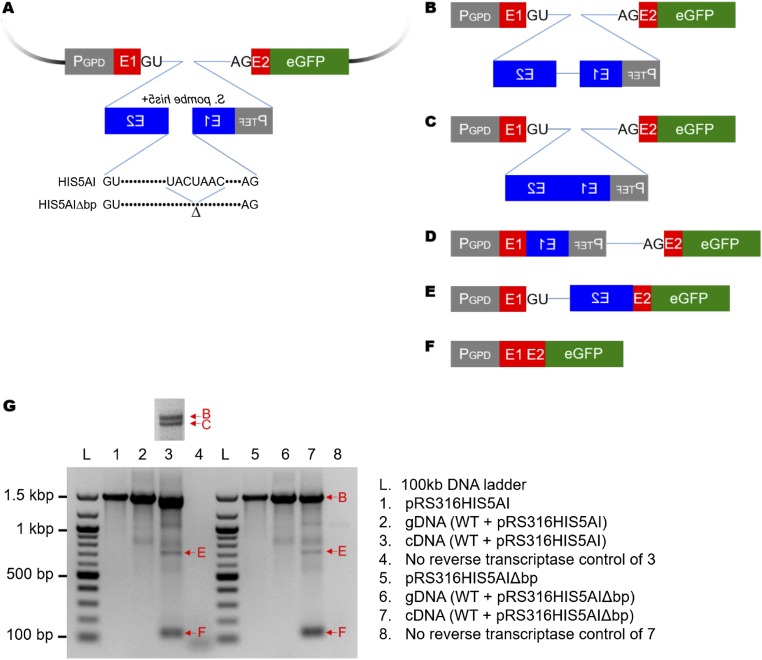
The design of the reporter is outlined in Fig. 1A. In this construct, which has been modified from one used previously in our other work (19), the intron and short stretches of exons 1 and 2 of the RPL28 gene (ribosomal protein L28) have been fused in-frame to EGFP (enhanced green fluorescent protein). Within the intron, we have used the heterologous Schizosaccharomyces pombe TEF1 promoter to drive transcription of the S. pombe his5+ gene, which confers histidine prototrophy in S. cerevisiae his3 mutants. The transcriptional direction of his5+ is in the opposite orientation to that of the EGFP construct. Within the his5+ gene, we have inserted an artificial intron (modeled on those described in refs. 9 and 20) containing pre-mRNA splicing signals that are only capable of being spliced from the EGFP transcript but not from the his5+ transcript. Verification of the proper splicing of this reporter by virtue of EGFP protein production is shown in Fig. 1B. The array of recursive splicing products was also tested by RT-PCR and sequence analysis of the PCR products (Fig. S4). We note that similar to our previous work with a related construct (19), this intron is poorly spliced, however there is reasonably good EGFP splicing and EGFP protein production.
Fig. 1.
Design and splicing of the intron gain/loss reporter. (A) Model of the intron gain/loss reporter. Short stretches of exon 1 and exon 2 of the RPL28 gene with portions of the intron were fused to EGFP. The S. pombe his5+ gene was inserted into the RPL28 intron (EGFP gene). Backwards text represents the opposite transcriptional orientation of the S. pombe his5+ gene. The his5+ gene was interrupted by an artificial intron (AI) containing splice sites that are only spliced from the EGFP transcript, due to their orientation. The AI lacking the branch point sequence (AIΔbp) cannot be spliced from the EGFP transcript. After splicing of the artificial intron followed by RNA-mediated recombination, transcription of S. pombe his5+ gene driven by TEF1 promoter confers histidine prototrophy in S. cerevisiae his3 mutants. (B) Demonstration of EGFP production. Splicing and expression of EGFP from the reporter was demonstrated by Western blot analysis using an antibody to GFP. Western blot analysis of Rps8 was used as a loading control. (C) Plasmid-borne intron loss events. The plasmids rescued from most his+ cells have lost the artificial intron within the S. pombe his5+ gene.
Fig. S4.
Detection of recursive intron splicing. (A) The intron reporter constructs of both HIS5AI and HIS5AI∆bp. (B–F) Predicted and detected recursive intron splicing products. (B) The reporter pre-mRNA transcribed without being processed by splicing. (C) Only the his5+ intron is spliced. (D) Splicing occurs at the 5′ splice site of the EGFP intron and the 3′ splice site of the his5+ intron. (E) Splicing occurs at the 5′ splice site of the his5+ intron and the 3′ splice site of the EGFP intron. (F) The entire EGFP intron is spliced. Note that the HIS5AIΔbp construct is not able to produce C or D. (G) Detection of the transcripts from the reporter construct. Total RNA extracted from the wild-type strain harboring HIS5AI (lane 3) or HIS5AIΔbp (lane 7) was subjected to reverse transcription. The resulting cDNA was amplified by PCR with the primers complementary to the exon 1 of RPL28 and EGFP to capture the possible spectrum of transcripts. The reporter plasmids (lanes 1 and 5) and the corresponding genomic DNA (lanes 2 and 6) were also subjected to PCR as controls for HIS5AI and HIS5AI∆bp, respectively. Reverse transcription of total RNA in the absence of reverse transcriptase followed by PCR was also performed as a negative control (lanes 4 and 8). Due to inefficient splicing, the unspliced transcript was amplified in all conditions, except the “no reverse transcriptase” control (species B, 1,473 bp). The top band in cDNA from the wild-type strain containing the HIS5AI construct was separated by further electrophoresis, revealing two distinct bands corresponding to B and C (1,370 bp; see Inset above lane 3). Transcripts E (717 bp) and F (134 bp) were detected in both strains harboring HIS5AI and HIS5AIΔbp, respectively, whereas transcript D (787 bp) was not detected. All PCR products except transcript C were verified by DNA sequence analysis.
This reporter is conceptually related to ones used previously to detect de novo Ty1 retrotransposition events (20) and RNA-mediated intron loss (9); however, the selection signal for the other reporters was contained on the resulting mRNA, whereas this reporter selection signal is contained within the spliced intron. Intron mobilization leading to histidine prototrophy can be a result of a few key events. The most common event in our screen is plasmid-borne intron loss, related to the RNA-mediated recombination events shown previously (9, 20) (Fig. 1C). In this intron loss, the intron within the his5+ gene has been spliced out, and a species of the EGFP intron RNA has been reverse-transcribed and recombined back into the reporter plasmid. From these data, it is not possible to determine if this is a singly spliced, intron-containing EGFP pre-mRNA or the liberated EGFP intron that gave rise to these events, however sequencing of the his5+ gene in the plasmid revealed that the his5+ intron has been precisely removed. As a control, a screen with a reporter construct lacking the branchpoint sequence within the his5+ intron produced no histidine prototrophs (Fig. 1A), indicating that removal of the artificial intron from the EGFP intron by splicing followed by RNA transposition of the EGFP intron is required for histidine prototroph formation.
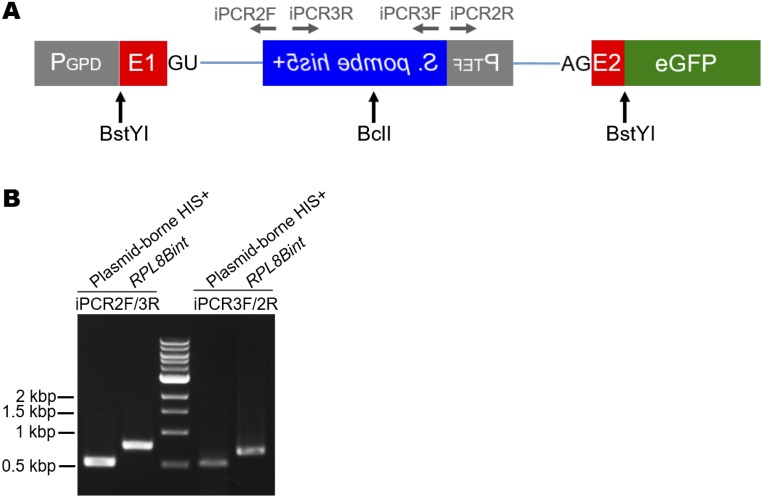
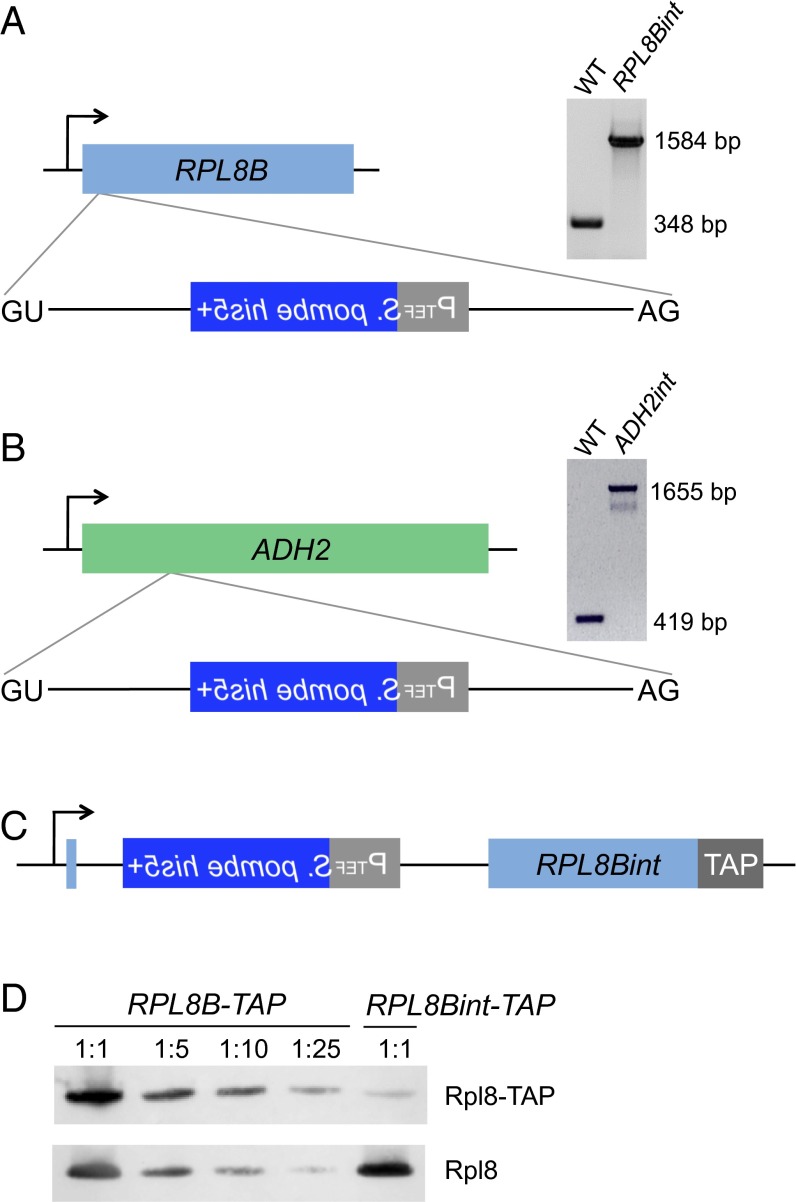
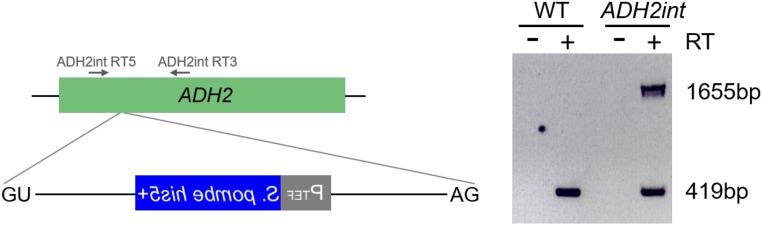
Our screen was conducted on nearly 2 × 1011 cells from which we detected ∼10,000 plasmid-borne intron loss events, resulting in an RNA-mediated recombination rate similar to that previously reported (9). In addition to the plasmid-related events, we detected two instances of intron gain that resulted from a perfect chromosomal addition of the EGFP intron into an mRNA-encoding gene. Direct genomic sequencing (21) and inverse PCR from genomic DNA (Fig. S5) in the first captured intron gain strain revealed that an intronogenesis event occurred in the RPL8B gene (ribosomal protein L8B) (Fig. 2A). PCR amplification and sequencing of the entire RPL8B locus confirmed that the transposed intron was inserted 18 nucleotides downstream from the start codon of the gene and that after intron removal from the RPL8B pre-mRNA, the resulting mRNA is competent for translation into a proper Rpl8 protein. In the second intron gain strain, the EGFP intron was transposed into the ADH2 gene (alcohol dehydrogenase), 204 nucleotides downstream from the start codon (Fig. 2B). PCR amplification of genomic DNA and cDNA in this strain demonstrated that the newly formed intron is able to be spliced from the ADH2 pre-mRNA (Fig. 2B and Fig. S6). The new alleles of RPL8B and ADH2 were termed RPL8Bint and ADH2int, respectively. Additionally, we observed two events that were not plasmid-borne but that resulted in histidine prototrophy, likely by virtue of nonhomologous recombination of plasmid sequences containing the his5+ gene from which the intron was previously lost. These two events were not related to the intronogenesis process, as they also included EGFP exon sequences, indicating that the mechanism did not involve the lariat intron (Discussion).
Fig. S5.
Analysis of the intron gain in the RPL8B gene by inverse PCR. (A) 5-FOA–resistant histidine prototrophs were subjected to the restriction digestion and inverse PCR to analyze the insertion sites. The cartoon displays the location of the BstYI and BclI cleavage sites within the reporter construct after splicing of the artificial intron. Two pairs of primers, iPCR2F/iPCR3R and iPCR3F/iPCR2R, were designed to amplify the sequences containing the partial Schizosaccharomyces pombe his5+ gene. (B) For the plasmid-borne intron loss event, 529-bp and 535-bp PCR products are expected to be amplified with the two sets of primers, respectively, whereas the intron insertion into the chromosome gives larger PCR products. Inverse PCR analysis of RPL8Bint produced 780-bp and 680-bp products.
Fig. 2.
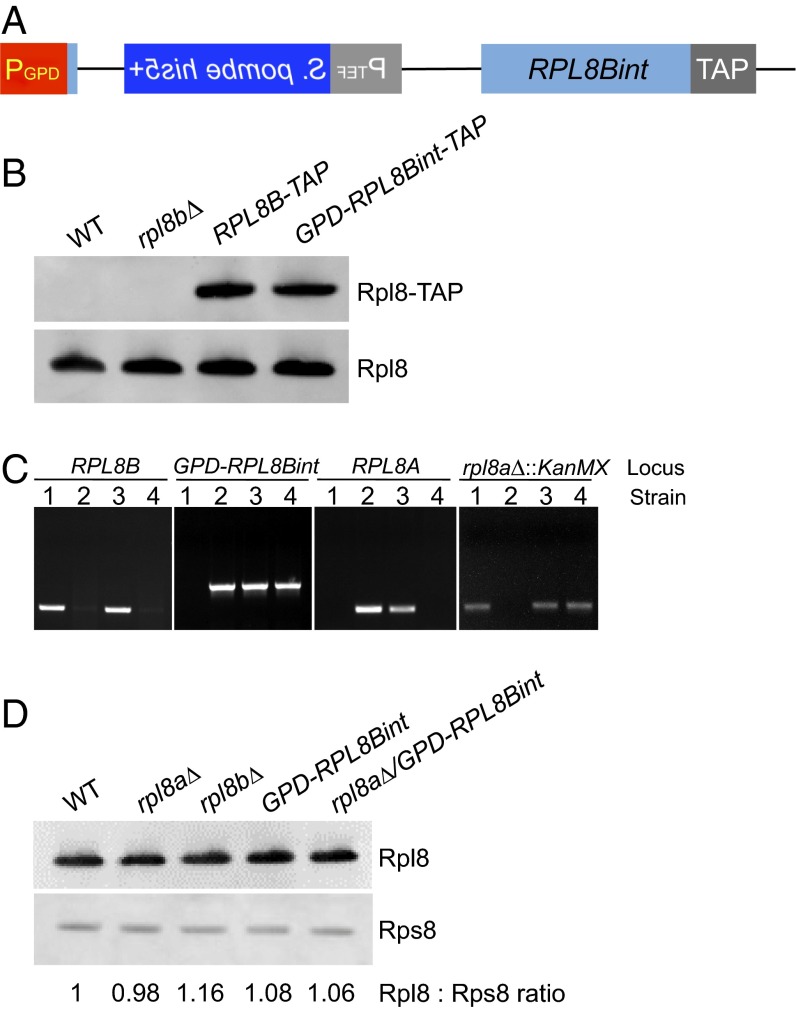
Transposition of the EGFP intron into the chromosomal locus, leading to a gain of a spliceable intron. (A) The RPL8Bint locus. (Left) The entirety of the EGFP intron (lacking the spliced-out artificial intron) was transposed 18 nucleotides downstream from the start codon of RPL8B. It contains all of the pre-mRNA splicing signals required to splice the RPL8Bint transcript. (Right) PCR products of the RPL8B locus from genomic DNA of wild-type and RPL8Bint strains. The size difference in the PCR products of RPL8Bint indicates the acquisition of the reporter intron (1,236 bp) in RPL8Bint. (B) The ADH2int locus. (Left) The EGFP intron was inserted 204 nucleotides downstream from the start codon of ADH2 in the same manner observed in RPL8Bint. (Right) PCR products of the genomic ADH2 locus in wild-type and ADH2int strains reflect the reporter intron transposition in ADH2int. (C) TAP tagging of RPL8Bint. The TAP tag (22) was inserted into the C terminus of the RPL8Bint gene. (D) Quantitation of Rpl8 expressed from RPL8Bint-TAP. To quantitatively compare Rpl8-TAP protein levels in the indicated strains, RPL8B-TAP was serially diluted as shown in the figure. Rpl8 and Rpl8-TAP were detected by an antibody to Rpl8 and quantitated by chemifluorescence.
Fig. S6.
RT-PCR analysis of ADH2int allele. Total RNA extracted from the wild-type strain and ADH2int allele was subjected to reverse transcription and cDNA amplification with the primers ADH2int RT5 and ADH2int RT3, designed to amplify the DNA fragment containing the transposed intron. Two PCR products were detected from ADH2int allele, spliced (419 bp) and unspliced (1,655 bp), indicating that the newly transposed intron is able to be spliced. Reverse transcription of total RNA in the absence of reverse transcriptase followed by PCR was performed as a negative control (RT –).
The RPL8Bint pre-mRNA is spliced, as shown by Western blotting of a strain in which RPL8Bint has been tandem affinity purification (TAP) tagged (22) (Fig. 2C). Quantitative comparisons of the levels of the RPL8B-TAP (23) to that of RPL8Bint-TAP show that Rpl8 protein produced from the RPL8Bint-TAP locus is ∼2% of that from the RPL8B-TAP locus (Fig. 2D). This is most likely due to the inefficient splicing of the very large intron containing the his5+ gene (19) (Fig. S4). Budding yeast introns are typically much shorter than in other species, and as the size of the intron increases, splicing efficiency dramatically decreases (24). Indeed, at 1,236 nucleotides, this intron is 235 nucleotides longer than the largest natural intron in S. cerevisiae (25). Thus, it is not surprising that knocking out the Rpl8 paralog RPL8A in RPL8Bint proved to be impossible either by direct removal or by crossing and dissection of tetrads.
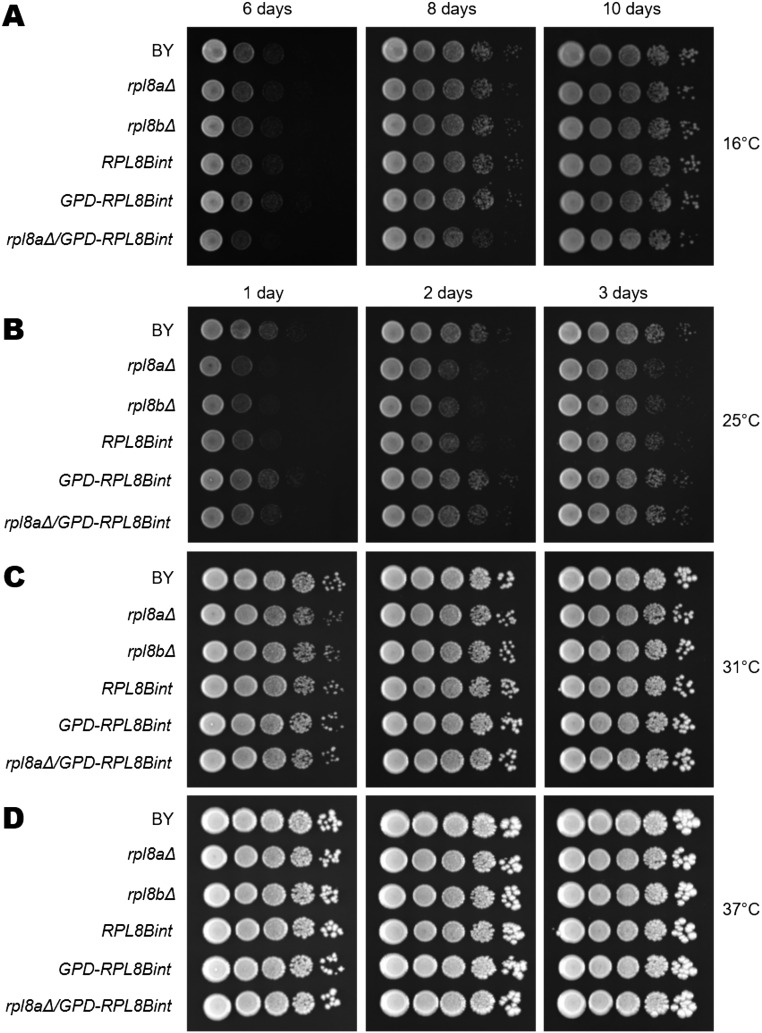
Because the levels of Rpl8 produced from RPL8Bint were so low and because we wished to show that the new intron added to RPL8B could suffice for production of Rpl8, we modified both RPL8Bint and RPL8Bint-TAP to be driven from a strong heterologous promoter derived from the TDH3 gene (26) (Fig. 3A). This modification restored the levels of Rpl8 to wild-type levels as determined by Western blotting in the GPD-RPL8Bint-TAP strain (Fig. 3B). Removal of RPL8A from the GPD-RPL8Bint strain was achieved by crossing GPD-RPL8Bint with the rpl8A∆ strain followed by sporulation (Fig. 3C). Indeed, total levels of Rpl8 are very similar in wild-type, GPD-RPL8Bint, and rpl8a∆/GPD-RPL8Bint strains (Fig. 3D), indicating that the Rpl8 expressed from the GPD-RPL8Bint locus is sufficient in these cells. Not only is the expression level comparable to the wild type, Rpl8 in the rpl8aΔ/GPD-RPL8Bint strain is also fully functional, as it exhibits no detectable growth defects (Fig. S7).
Fig. 3.
The Rpl8Bint locus is sufficient for Rpl8 production when overexpressed. (A) GPD-RPL8Bint construction. The original promoter of the RPL8Bint and RPL8Bint-TAP was replaced with the GPD promoter to increase the expression level. (B) Quantitation of Rpl8 expressed from RPL8B-TAP to GPD-RPL8Bint-TAP. To determine the effect of the GPD promoter, the amount of Rpl8 expressed from RPL8B-TAP and GPD-RPL8Bint-TAP strains was determined by Western blotting. Rpl8 and Rpl8-TAP were detected by an antibody to Rpl8. (C) PCR confirmation of rpl8aΔ/GPD-RPL8Bint and related strains. Shown is the genomic DNA amplification of a portion of RPL8B, GPD-RPL8Bint, RPL8A, and rpl8aΔ::KanMX loci from each strain, labeled 1–4 (1, rpl8aΔ; 2, GPD-RPL8Bint; 3, diploid formed from rpl8aΔ and GPD-RPL8Bint; 4, haploid rpl8aΔ/GPD-RPL8Bint). (D) Quantitative Western blot. Total levels of Rpl8 were determined in wild-type, rpl8aΔ, rpl8bΔ, GPD-RPL8Bint, and rpl8a∆/GPD-RPL8Bint strains by Western blotting using chemifluorescence.
Fig. S7.
Overexpressed RPL8Bint allele is functionally comparable to the wild type. Relative growth rate of RPL8Bint was determined by serial dilution analysis. RPL8Bint shows a slight growth defect as seen in rpl8aΔ and rpl8bΔ strains. When RPL8Bint is overexpressed, no significant growth defect is detected even in the absence of RPL8A. For serial dilutions, each strain was grown in YPD liquid culture, adjusted to OD600 of 1.0, and serially diluted by 10-fold with the same media. Growth on YPD plates was monitored at 16 °C (A) for 10 d and at 25 °C (B), 31 °C (C), and 37 °C (D) up to 3 d.
Discussion
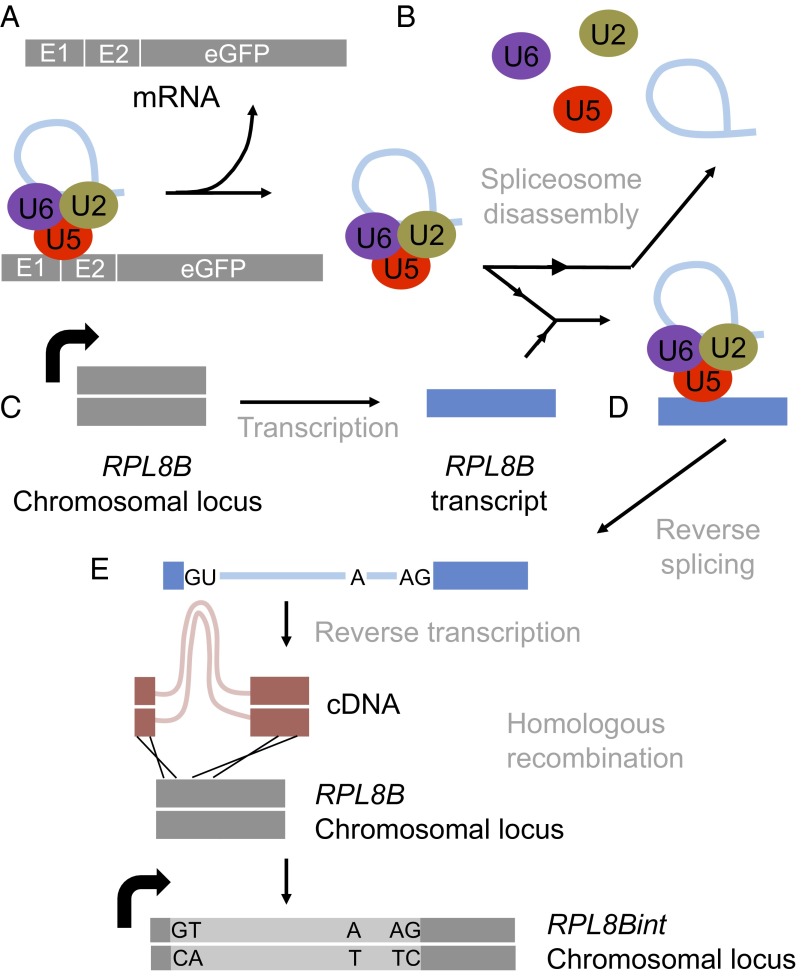
Here we report, to our knowledge, the first experimental demonstration of spliceosomal intron transposition in any eukaryotic organism. We detected the insertion of an EGFP reporter intron into the genomic RPL8B and ADH2 using a reporter construct. In our model of RPL8Bint formation, the EGFP intron RNA lacking the his5+ artificial intron was retained in the residual spliceosome after Prp22-mediated mRNA release (Fig. 4). This complex then encountered an RPL8B mRNA and inserted the reporter intron by reverse splicing (15, 17). RPL8Bint pre-mRNA was then reverse-transcribed by a cellular reverse transcriptase (likely derived from the Ty1 retroelement) (9, 20), and the resulting RPL8Bint cDNA was incorporated into the RPL8B genomic locus by homologous recombination. We suggest that the formation of ADH2int is likely to have occurred by the same model.
Fig. 4.
Proposed model for the mechanism of RPL8B intron gain. (A) The last step of the pre-mRNA splicing reaction. The spliced mRNA (Top) is disengaged from the residual spliceosome composed of the lariat intron; the U2, U5, and U6 snRNPs; and other splicing proteins. (B) Disassembly of the residual spliceosome under normal conditions. The U2, U5, and U6 snRNPs and the lariat intron are dissociated from each other at the end of the splicing reaction. (C) Transcription of the RPL8B mRNA, the proposed substrate for the intron transposition. (D) Model of the transposition of the reporter intron contained within the intact residual spliceosome into the RPL8B transcript. The RPL8B transcript was captured by an intact, lariat intron-containing spliceosome before proceeding through both catalytic steps of pre-mRNA splicing, in reverse (17). (E) Proposed mechanism of transposed intron mobilization into the RPL8B locus. The transposed reporter intron within the RPL8B mRNA underwent a conversion to double-stranded cDNA by a reverse transcriptase, followed by homologous recombination into the genome at the RPL8B locus.
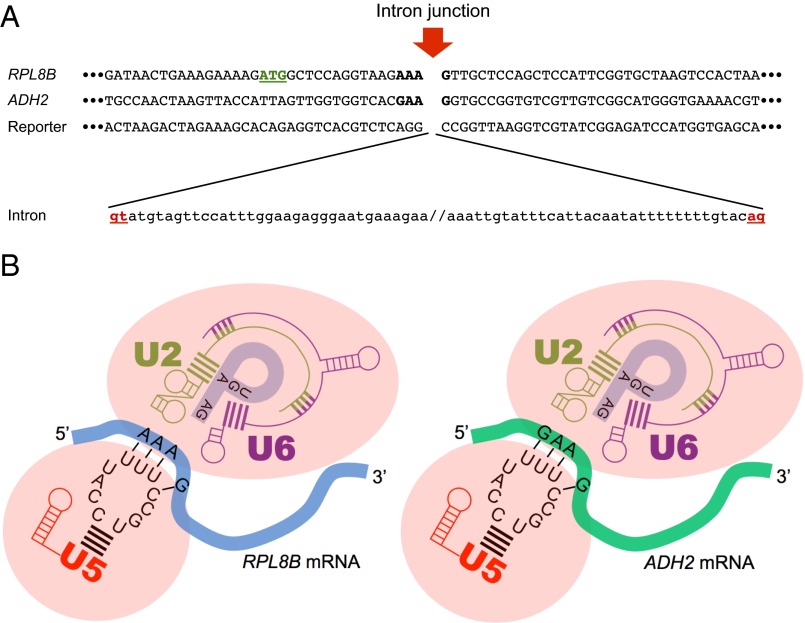
It may not be surprising that in ∼2 × 1011 cells we captured only two intron gain events while observing over 10,000 plasmid-borne intron loss events, considering that our model of intron gain involves an additional reverse splicing step that is expected to be rare in vivo (15). Previous work showed the capability of the spliceosome to achieve the reversal of the two catalytic steps of splicing in vitro (17), however those experiments were performed at nonstandard salt and pH conditions to promote the reverse reactions. We would like to point out that the sequences of our reporter construct and the location of insertion in RPL8B and ADH2 do not have even short regions of homology, which could explain this intron gain as having taken place via a microhomology-based recombination event (27) (Fig. 5A). Interestingly, the four nucleotides in the RPL8B transcript at the site of intron insertion (the –3, –2, –1, and +1 positions relative to the intron) possess base-pairing potential with the invariant U5 loop at the proper positions (28, 29) (Fig. 5). Comparable base-pairing potential exists between the four nucleotides in the ADH2 transcript at the intron junction (–3, –2, –1, and +1) and the U5 loop (Fig. 5). This provides further evidence that reverse splicing may have occurred and suggests that in this mode of intron gain, new mRNAs may be selected for intron addition by the spliceosome at sites at which splicing may be more efficient in the forward reaction (Fig. 4).
Fig. 5.
The sequence information at the intron insertion sites supports an intron gain model involving reverse splicing. (A) Microhomology-mediated end-joning rules preclude recombination as a means of this intron acquisition. The coding strand sequences of RPL8B, ADH2, and the reporter construct at both ends of the inserted intron were juxtaposed to examine the possibility of very short sequence homology. Neither RPL8B nor ADH2 show potential microhomology to the reporter construct at the intron junctions. Note the start codon of RPL8B highlighted in green. (B) Closer view of the RPL8B (Left) and ADH2 (Right) transcript engaging with the spliceosome showing the proposed base-pairing interactions between the new splice site and loop 1 of the U5 snRNA. The nucleotides of RPL8B and ADH2 participating in the potential base-pairing are marked in bold in A.
Although how the spliceosomal introns emerged and why only eukaryotes possess this class of introns remain unclear, it is widely accepted that the spliceosomal introns originated from the group II self-splicing introns and appeared at the time of eukaryogenesis (11, 18). It is likely that the proto-spliceosomal introns resembled the group II introns and for some period retained their mobility and self-splicing ability (5). This may have enabled and promoted considerable proliferation of these introns at the time of eukaryogenesis (11, 18). The group II intron-derived features are likely to have degenerated over time, leading to a necessity for the development of the pre-mRNA splicing machinery composed of trans-acting snRNAs and trans-acting protein factors (11, 18). Within this context, reverse splicing-catalyzed intron transposition is an appealing model for intron gain.
Demonstration of intron gain events in vivo has been a major obstacle to test existing models of intron gain. The in vivo intron gain presented in this work strengthens the intron gain model involving reverse splicing by providing, to our knowledge, the first evidence that it can occur on a laboratory time scale. At this point in the arc of evolution, for an intron gain event to become fixed in a population, either the intron insertion needs to result in a selective advantage to that organism or individuals possessing a new intron insertion event need to find themselves in ecological niches in which they can further speciate. Indeed in recent reports, some intron gain events have been suggested to have occurred by means other than reverse splicing and are highly mosaic depending on the individuals that have been analyzed (30–33), indicating that it may be that there are multiple mechanisms of intron gain that may change over time and genetic circumstance.
Materials and Methods
Experimental Design of a Reporter System.
The intron reporter was generated by modification of the splicing reporter used in our previous work (19). The two-color fluorescence reporter containing the RPL28 intron fused to EGFP and mCherry in the intron (pRS316 backbone) was restriction enzyme-digested with SphI and AflIII to remove the mCherry ORF. A synthetic construct (Genscript) containing the S. pombe tef1+ promoter driving the S. pombe his5+ gene and terminating with the S. pombe tef1+ termination sequence was cloned into those same restriction sites.
Yeast Strains.
SS4056 (parent BY4739) (MATα his3::KanMX, leu2Δ0, lys2Δ0, ura3Δ0), BY4733 (MATa his3Δ200, leu2Δ0, met15Δ0, trp1Δ63, ura3Δ0), and SS4019 (parent BY4741) (MATa his3Δ1, leu2Δ0, met15Δ0, ura3Δ0, trp1Δ) were used for intron transposition assays. SS5230 (parent SS4056) (MATα his3::KanMX, leu2Δ0, lys2Δ0, ura3Δ0, RPL8B::his5+::int) and SS5252 (MATα his3::KanMX, leu2Δ0, lys2Δ0, ura3Δ0, ADH2::his5+::int) were obtained by intron gain. SS5231 (MATα his3::KanMX, leu2Δ0, lys2Δ0, ura3Δ0, RPL8B::his5+::int::TAP::HYG), SS5232 (MATα his3::KanMX, leu2Δ0, lys2Δ0, ura3Δ0, URA3::GPD-RPL8B::his5+::int), SS5233 (MATα his3::KanMX, leu2Δ0, lys2Δ0, ura3Δ0, URA3::GPD-RPL8B::his5+::int::TAP::HYG), and SS5235 (MATα his3::KanMX, leu2Δ0, lys2Δ0, ura3Δ0, rpl8aΔ, URA3::GPD-RPL8B::his5+::int) were generated to further analyze the RPL8Bint strain. RPL8B-TAP–tagged strain (background BY4741) (MATa his3Δ1, leu2Δ0, met15Δ0, ura3Δ0, RPL8B-TAP::HIS3MX6) (Yeast-TAP–tagged ORF library, GE Dharmacon), rpl8aΔ strain (background BY4741) (MATa his3Δ1, leu2Δ0, met15Δ0, ura3Δ0, rpl8a::KanMX) (GE Dharmacon), and rpl8bΔ strain (background BY4741) (MATa his3Δ1, leu2Δ0, met15Δ0, ura3Δ0, rpl8b::KanMX) (GE Dharmacon) were used to quantitatively analyze Rpl8 expressed in the RPL8Bint strains.
Intron Transposition Assay.
Yeast cells transformed with pRS416 (empty vector), pRS316HIS5AI, or pRS316HIS5AIΔbp were plated on SD-ura and grown for 72 h at 31 °C. Individual uracil prototrophs were then grown in 10 mL SD-ura liquid medium overnight at 31 °C and cells harvested by centrifugation. Pelleted cells were resuspended in water and plated on 150-mm SD-his plates and grown at 31 °C for 4–6 d. All his+ colonies were patched onto YPD plates, grown overnight at 31 °C, and then streaked on SD-his 5-FOA. After incubation at 31 °C for 6 d, his+ cells were scored for their resistance to 5-FOA.
Inverse PCR.
Genomic DNA was isolated from his+/5-FOAr cells and cut sequentially with BstYI and BclI restriction endonucleases. This DNA was phenol-extracted, ethanol-precipitated, and then resuspended in water to be ligated using T4 DNA ligase (New England Biolabs) under conditions favoring self-ligation (9). After overnight ligation at 16 °C, the DNA was extracted, precipitated, and resuspended in 50 µL of water for use in an inverse PCR. DNA was amplified by PCR with primers hybridizing within the reporter intron region using Taq DNA polymerase with ThermoPol buffer (New England Biolabs): SpHIS5iPCR2F, 5′-CCAAGCACGAAGGGAGTGTTGTAAAGTAAC-3′; SpHIS5iPCR3R, 5′-ACATGCAAAGTAATTCCAGC-3′; SpHIS5iPCR3F, 5′-CAAGTAATCCAAGTAGACAC-3′; and SpHIS5iPCR2R, 5′- GTTTATGTTCGGATGTGATGTGAG-3′
RPL8Bint and ADH2int Identification and Confirmation by PCR.
The intron insertion was first identified by direct genomic DNA sequencing (21) and inverse PCR and confirmed by PCR with the following primer sets, respectively: RPL8Bint, 2014.7, 5′-GTATTGTGATGCGCACGTTGAAATTCAC-3′; RPL8Bint, 2014.5, 5′-CAGCGGTGTTTCTGTCCAAAGTG-3′; ADH2int, ADH2int RT5, 5′-CAACGGCAAGTTGGAGCATAAG-3′; ADH2int RT3, 5′-CACACAAGATTGGCGCGACTTCAG-3′.
TAP Tagging of RPL8Bint.
The TAP tag was introduced at the C terminus of the genomic RPL8Bint locus by homologous recombination with the hygromycin B-resistant selectable marker adjacent to the TAP cassette (22). Oligonucleotide primers contain a 40-bp-long region identical to the yeast genome for homologous recombination. Primers A and B hybridize at the 5′ end of the CBP (calmodulin binding peptide) coding sequence and at the downstream of the coding sequence of hygromycin B phosphotransferase, respectively: RPL8BtapA-HYG, 5′-AGCTAAGATGGACAAGAGAGCTAAGACTTCCGACTCCGCTATGGAAAAGAGAAGATGGAA-3′; RPL8BtapBpRS41H, 5′-TACAAAATATAATTATATTACGATGTTCGAAATTCTATATACTGAGAGTGCAGCGACATG-3′.
RPL8Bint Promoter Switch.
The GPD (TDH3) promoter was introduced to replace the promoter of RPL8Bint at the genomic locus by homologous recombination. Primers contain a 55 nucleotide-long region identical to regions upstream and downstream of the RPL8Bint start codon for homologous recombination. Oligonucleotides KIA and KIB were designed to amplify a DNA fragment containing the GPD promoter with the URA3 marker for selection purposes: RPL8B-GPD-URA-KIA, 5′-ATAGAACGCATTGAAACTTTTCCCATCTCAAAATCCAGGGACAATAGTATGGGATGCGCGTTTCGGTGATGACGGTG-3′; RPL8Bint-GPD-URA-KIB, 5′-GGTTCTTTCATTCCCTCTTCCAAATGGAACTACATACTTTCTTACCTGGAGCCATTTTGTTTGTTTATGTGTGTTTATTCG-3′.
Yeast Sporulation and Tetrad Dissection.
GPD-RPL8Bint (SS5232) and rpl8aΔ strains were mixed on an YPD plate within a drop of water, incubated overnight at 31 °C, and then diploids were selected by streaking on SD-his-lys media. The diploid cells were inoculated into 2 mL of SD-his, incubated for 30 h at 31 °C, harvested, and washed with water. The cells were resuspended in 2 mL of SPM (100 mM Potassium acetate and 400 µM Raffinose) and incubated on a roller drum at room temperature for 5 d to sporulate. We treated 100 µL of the sporulated culture with 15 µg of Zymolyase T100 in 20 mM β-mercaptoethanol at 30 °C for 20 min and dissected it on a tetrad dissection microscope.
Protein Isolation and Quantitation.
Yeast cells were grown in YPD at 31 °C, and 1 OD unit of the cells was harvested by centrifugation, resuspended in 1 mL of YPD, chilled on ice for 10 min, and then mixed with 150 µL of ice-cold 2 N NaOH and 8% (vol/vol) β-mercaptoethanol. After an additional 10-min incubation on ice, the cells were mixed with 150 µL of ice-cold 50% (vol/vol) TCA, chilled on ice for another 10 min, and harvested by centrifuging at 4 °C at (16,000 × g) for 2 min. The pellets were washed with 1 mL of ice-cold acetone and resuspended in 100 µL of 1× LDS sample buffer containing 100 mM DTT. Extracted protein was subjected to Western blot analysis using a chemifluorescence detection kit. Quantitation of proteins in Western blots was performed using the Quantity One analysis software (Bio-Rad) and ImageJ [National Institutes of Health (https://imagej.nih.gov/ij)].
Acknowledgments
We thank Arlen Johnson and the Johnson laboratory for contributions of antibodies and technical help, and Albert Mackrell and the students in the Vertebrate Interactome Stream of the University of Texas Freshman Research Initiative for help with the screen. This work was funded by support from the Department of Molecular Biosciences, University of Texas at Austin.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1605113113/-/DCSupplemental.
References
- 1.Matera AG, Wang Z. A day in the life of the spliceosome. Nat Rev Mol Cell Biol. 2014;15(2):108–121. doi: 10.1038/nrm3742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Will CL, Lührmann R. Spliceosome structure and function. Cold Spring Harb Perspect Biol. 2011;3(7):a003707. doi: 10.1101/cshperspect.a003707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cech TR. The generality of self-splicing RNA: Relationship to nuclear mRNA splicing. Cell. 1986;44(2):207–210. doi: 10.1016/0092-8674(86)90751-8. [DOI] [PubMed] [Google Scholar]
- 4.Jacquier A. Self-splicing group II and nuclear pre-mRNA introns: How similar are they? Trends Biochem Sci. 1990;15(9):351–354. doi: 10.1016/0968-0004(90)90075-m. [DOI] [PubMed] [Google Scholar]
- 5.Lambowitz AM, Zimmerly S. Group II introns: Mobile ribozymes that invade DNA. Cold Spring Harb Perspect Biol. 2011;3(8):a003616. doi: 10.1101/cshperspect.a003616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fink GR. Pseudogenes in yeast? Cell. 1987;49(1):5–6. doi: 10.1016/0092-8674(87)90746-x. [DOI] [PubMed] [Google Scholar]
- 7.Mourier T, Jeffares DC. Eukaryotic intron loss. Science. 2003;300(5624):1393. doi: 10.1126/science.1080559. [DOI] [PubMed] [Google Scholar]
- 8.Boeke JD, Garfinkel DJ, Styles CA, Fink GR. Ty elements transpose through an RNA intermediate. Cell. 1985;40(3):491–500. doi: 10.1016/0092-8674(85)90197-7. [DOI] [PubMed] [Google Scholar]
- 9.Derr LK, Strathern JN, Garfinkel DJ. RNA-mediated recombination in S. cerevisiae. Cell. 1991;67(2):355–364. doi: 10.1016/0092-8674(91)90187-4. [DOI] [PubMed] [Google Scholar]
- 10.Derr LK. The involvement of cellular recombination and repair genes in RNA-mediated recombination in Saccharomyces cerevisiae. Genetics. 1998;148(3):937–945. doi: 10.1093/genetics/148.3.937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Irimia M, Roy SW. Origin of spliceosomal introns and alternative splicing. Cold Spring Harb Perspect Biol. 2014;6(6):a016071. doi: 10.1101/cshperspect.a016071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sakurai A, et al. On biased distribution of introns in various eukaryotes. Gene. 2002;300(1-2):89–95. doi: 10.1016/s0378-1119(02)01035-1. [DOI] [PubMed] [Google Scholar]
- 13.Roy SW, Gilbert W. The pattern of intron loss. Proc Natl Acad Sci USA. 2005;102(3):713–718. doi: 10.1073/pnas.0408274102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sverdlov AV, Babenko VN, Rogozin IB, Koonin EV. Preferential loss and gain of introns in 3′ portions of genes suggests a reverse-transcription mechanism of intron insertion. Gene. 2004;338(1):85–91. doi: 10.1016/j.gene.2004.05.027. [DOI] [PubMed] [Google Scholar]
- 15.Roy SW, Irimia M. Mystery of intron gain: New data and new models. Trends Genet. 2009;25(2):67–73. doi: 10.1016/j.tig.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 16.Yenerall P, Zhou L. Identifying the mechanisms of intron gain: Progress and trends. Biol Direct. 2012;7:29. doi: 10.1186/1745-6150-7-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tseng CK, Cheng SC. Both catalytic steps of nuclear pre-mRNA splicing are reversible. Science. 2008;320(5884):1782–1784. doi: 10.1126/science.1158993. [DOI] [PubMed] [Google Scholar]
- 18.Rogozin IB, Carmel L, Csuros M, Koonin EV. Origin and evolution of spliceosomal introns. Biol Direct. 2012;7:11. doi: 10.1186/1745-6150-7-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sorenson MR, Stevens SW. Rapid identification of mRNA processing defects with a novel single-cell yeast reporter. RNA. 2014;20(5):732–745. doi: 10.1261/rna.042663.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Curcio MJ, Garfinkel DJ. Single-step selection for Ty1 element retrotransposition. Proc Natl Acad Sci USA. 1991;88(3):936–940. doi: 10.1073/pnas.88.3.936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Horecka J, Jigami Y. Identifying tagged transposon insertion sites in yeast by direct genomic sequencing. Yeast. 2000;16(10):967–970. doi: 10.1002/1097-0061(200007)16:10<967::AID-YEA597>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 22.Puig O, et al. The tandem affinity purification (TAP) method: A general procedure of protein complex purification. Methods. 2001;24(3):218–229. doi: 10.1006/meth.2001.1183. [DOI] [PubMed] [Google Scholar]
- 23.Ghaemmaghami S, et al. Global analysis of protein expression in yeast. Nature. 2003;425(6959):737–741. doi: 10.1038/nature02046. [DOI] [PubMed] [Google Scholar]
- 24.Klinz FJ, Gallwitz D. Size and position of intervening sequences are critical for the splicing efficiency of pre-mRNA in the yeast Saccharomyces cerevisiae. Nucleic Acids Res. 1985;13(11):3791–3804. doi: 10.1093/nar/13.11.3791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Spingola M, Grate L, Haussler D, Ares M., Jr Genome-wide bioinformatic and molecular analysis of introns in Saccharomyces cerevisiae. RNA. 1999;5(2):221–234. doi: 10.1017/s1355838299981682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bitter GA, Egan KM. Expression of heterologous genes in Saccharomyces cerevisiae from vectors utilizing the glyceraldehyde-3-phosphate dehydrogenase gene promoter. Gene. 1984;32(3):263–274. doi: 10.1016/0378-1119(84)90002-7. [DOI] [PubMed] [Google Scholar]
- 27.Truong LN, et al. Microhomology-mediated end joining and homologous recombination share the initial end resection step to repair DNA double-strand breaks in mammalian cells. Proc Natl Acad Sci USA. 2013;110(19):7720–7725. doi: 10.1073/pnas.1213431110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Newman AJ, Norman C. U5 snRNA interacts with exon sequences at 5′ and 3′ splice sites. Cell. 1992;68(4):743–754. doi: 10.1016/0092-8674(92)90149-7. [DOI] [PubMed] [Google Scholar]
- 29.Newman AJ, Teigelkamp S, Beggs JD. snRNA interactions at 5′ and 3′ splice sites monitored by photoactivated crosslinking in yeast spliceosomes. RNA. 1995;1(9):968–980. [PMC free article] [PubMed] [Google Scholar]
- 30.van der Burgt A, Severing E, de Wit PJ, Collemare J. Birth of new spliceosomal introns in fungi by multiplication of introner-like elements. Curr Biol. 2012;22(13):1260–1265. doi: 10.1016/j.cub.2012.05.011. [DOI] [PubMed] [Google Scholar]
- 31.Worden AZ, et al. Green evolution and dynamic adaptations revealed by genomes of the marine picoeukaryotes Micromonas. Science. 2009;324(5924):268–272. doi: 10.1126/science.1167222. [DOI] [PubMed] [Google Scholar]
- 32.Li W, Tucker AE, Sung W, Thomas WK, Lynch M. Extensive, recent intron gains in Daphnia populations. Science. 2009;326(5957):1260–1262. doi: 10.1126/science.1179302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Simmons MP, et al. Intron invasions trace algal speciation and reveal nearly identical arctic and antarctic Micromonas populations. Mol Biol Evol. 2015;32(9):2219–2235. doi: 10.1093/molbev/msv122. [DOI] [PMC free article] [PubMed] [Google Scholar]