Abstract
Background/Aims
Human mesenchymal stromal/stem cells (MSCs), derived from many different tissues, are characterized by a fibroblast-like morphology, expression of certain cell surface markers, and their ability to differentiate into adipocytes, chondrocytes, and osteoblasts. A number of studies have shown that MSCs share many characteristics with fibroblasts, however, there is no well-defined set of phenotypic characteristics that could distinguish between these two types of cells.
Methods
We used four well-established human fibroblast strains from three different tissue sources and several human MSC strains from two different tissue sources to compare phenotypic and immunological characteristic of these cells.
Results
Fibroblast strains show a morphology similar to MSCs, express the same cell surface markers as MSCs, and could also differentiate into adipocytes, chondrocytes, and osteoblasts. Also, similar to MSCs, these fibroblasts were capable of suppressing T cell proliferation and modulating the immunophenotype of macrophages. We also show that MSCs deposit extracellular matrices of collagen type-I and fibronectin, and express FSP1 in patterns similar to fibroblasts.
Conclusions
Based on currently accepted definitions for cultured human MSCs and fibroblasts, we could not find any immunophenotypic property that could make a characteristic distinction between MSCs and fibroblasts.
Keywords: mesenchymal stromal/stem cells, fibroblasts, stroma, immunomodulatory, macrophages
INTRODUCTION
The cells that we currently know as mesenchymal stromal/stem cells (MSCs) were originally discovered by A.J. Friedenstein [1] and characterized by their ability to adhere to culture plates, generate fibroblast-like colonies, and be repeatedly passaged upon proliferation [2–4]. Furthermore, Friedenstein was the first to show that these stromal cells were responsible for creating the hematopoietic tissue microenvironment, as the transplantation of these cells under the kidney capsule of guinea pigs resulted in not only bone formation, but also transferred the hematopoietic supportive properties of bone marrow stroma [4]. These cells were originally denoted as colony forming unit fibroblasts (CFU-F) and could be derived from non-bone marrow organs as well with the ability to be differentiated into bone and cartilage tissues [2,5,6]. While “mesenchymal stem cell” terminology was later adapted to indicate the multipotential differentiation capabilities of these cells [7,8], they are also commonly referred to as “mesenchymal stromal cells” [9].
In 2006, the International Society for Cellular Therapy (ISCT) developed a, now widely accepted, consensus definition for MSCs that includes their ability to adhere to plastic under standard culture conditions; ability to differentiate in vitro to osteoblasts, adipocytes, or chondroblasts; expression of CD73, CD90, and CD105; and lack of expression of CD34, CD45, CD14 or CD11b, CD19 or CD79α, and HLA-DR [10]. Similar to MSCs, fibroblasts are present in almost every human tissue, contribute to wound healing, and represent a heterogeneous population of cells [11,12]. However, in contrast to recently defined and standardized characteristics of MSCs, there are still no widely accepted criteria for defining fibroblasts [13], even though the 3T3 cell line established in 1962 has been utilized as the standard fibroblast cell line in innumerable experimental studies [14].
Over the years, many studies have compared MSCs and fibroblasts, and most of these show that fibroblasts express many of the the same markers as MSCs; can be induced to differentiate into adipocytes, chondrocytes, or osteoblasts; and even have immunomodulatory properties similar to those of MSCs [15–22]. However, there are also publications reporting differences between MSCs and fibroblasts. For example, some studies have shown that fibroblasts are incapable of differentiating into adipocytes, chondrocytes, and osteoblasts [8,19,23]. Furthermore, another study showed that fibroblasts from foreskin and adult dermis differed from bone marrow MSCs in their expression of CD10 (positive in fibroblasts but not MSCs), CD26 (positive in fibroblasts but not MSCs), CD106 (positive in MSCs but not fibroblasts), and collagen VII (12-fold higher expression in fibroblasts) [23]. In two studies that compared global gene expression of MSCs and fibroblasts, one found that MSCs express higher levels of genes related to embryogenesis and neural development [24] and the second study found that MSCs express more MHC-DR-α, MHC-DR-β, and CD73 than fibroblasts, but fibroblasts express more adrenomedullin, apolipoprotein D, collagen type XV α1, and matrix metalloproteinase-1 than MSCs [25]. In the study herein, we selected four fibroblast strains that were derived a decade or more ago from different human tissues and have been used by multiple laboratories as paradigms of fibroblast cell lines. We compared the phenotypical characteristics and differentiation capabilities of these fibroblasts to MSCs from human bone marrow or adipose tissue derived in our laboratory for the purpose of being able to distinguish the two populations. Elucidation of the relationship between MSCs and fibroblasts could help to reconcile many of the immunomodulatory and anti-inflammatory properties of MSCs with the corresponding biological effects of fibroblasts in tissue repair following inflammatory injuries.
MATERIALS AND METHODS
Isolation and Culture of Mesenchymal Stem Cells
Human bone marrow and adipose MSCs were derived from healthy donors using protocols approved by the UW-Madison Institutional Review Board (IRB), as previously described [15,26]. Briefly, bone marrow mononuclear cells from discarded collection filters after bone marrow donation were separated by density gradient separation using Ficoll-Hypaque (GE Lifesciences, Piscataway, NJ), followed by treatment with ACK lysis buffer to reduce contaminating red blood cells. Cells were collected from adipose tissue that was resected or from aspirates from abdominoplasty flaps, after mechanical mincing followed by enzymatic digestion with type I collagenase (Sigma-Aldrich, St. Louis, MO). Bone marrow mononuclear cells or digested adipose tissues were grown in αMEM media supplemented with 10% FBS, 1% non-essential amino acids (NEAA), and 2μM L-alanyl-L-glutamine (Hyclone, Logan, UT). Non-adherent cells were discarded within 24 hours. Cells were dissociated and passaged 1:3 upon reaching 80–90% confluency, using TrypLE (Invitrogen, Carlsbad, CA) per manufacturer’s directions. Passages 4–6 were used for experiments.
Culture of Fibroblasts
Neonatal foreskin dermal fibroblasts (PCS-201-010; denoted as “Foreskin FB 1” in the text) [27,28] and embryonic lung fibroblasts (IMR-90; denoted as “Lung FB” in the text) [29] were obtained from American Type Culture Collection (ATCC) and cultured according to protocols recommended by ATCC. A second strain of neonatal foreskin dermal fibroblasts (AH1F; denoted as “Foreskin FB 2” in the text) [30] was derived originally by Dr. Lynn Allen-Hoffmann at the University of Wisconsin [31]. Mammary fibroblasts (RMF/EG; denoted as Breast FB in the text) kindly provided by Dr. Andreas Freidl of the University of Wisconsin, were originally derived in the Dr. Robert Weinberg laboratory from tissue obtained at the time of reduction mammoplasty [32]. All four fibroblast strains were cultured in DMEM media supplemented with 10% FBS. Passages 4–6 (from receiving from other laboratories) were used for experiments.
Flow Cytometry
Fibroblasts and MSCs were harvested using TrypLE and resuspended at a density of 1 x 106 cells per 100 μL PBS, then incubated with 10 μL surface antibodies (1:10) for 30 minutes at 4° C in the dark. The following antibodies were used: CD14 FITC, CD19 PE, CD29 PE, CD31 PE, CD34 APC, CD44 PE, CD45 PE, CD73 PE, CD90 PE, CD105 PE, CD206 PE, HLA-ABC FITC, HLA-DR FITC (BD Biosciences, San Jose, CA). As a control, cells were stained with the appropriate isotype antibodies. Samples were analyzed using an Accuri C6 Flow Cytometer. FlowJo software (Tree Star) was used to analyze the data.
Differentiation Assays
Fibroblasts and MSCs were plated in 24-well plates and grown to 80–90% confluency. Adipogenic or osteogenic differentiation media (Miltenyi Biotech, Auburn, CA) was added and changed every 3–4 days for a total of 21 days. Adipocyte lipid droplets were detected by oil red O staining (Sigma-Aldrich). Osteoblast calcification was detected by alizarin red S staining (Sigma-Aldrich). For chondrogenic differentiation, 2.5x105 cells were put in a deep well 96-well plate and centrifuged to make a pellet. Media was changed every 3–4 days for a total of 24 days. Pellets were embedded in 2% agar, fixed in formalin, and sectioned. Chondrocytes were detected by staining with Alcian blue.
IFNγ Stimulation
Fibroblasts and MSCs were harvested using TrypLE digestion, and 1x105 cells were plated in 6-well plates in appropriate culture media and stimulated with 100 IU/mL IFNγ. Unstimulated cells served as controls. Cells were harvested after 4 days and analyzed by flow cytometry as described above after staining with HLA-ABC FITC, HLA-DR PE, CD80 FITC, and CD86 PE.
T Cell Suppression Assay
Our reference standard BM MSCs [33] and four fibroblast strains were plated in titrated numbers. Peripheral blood samples from healthy donors were separated by density centrifugation, and peripheral blood mononuclear cells (PBMCs) were collected and labeled with carboxyfluorescein succinimidyl ester (CFSE, Sigma-Aldrich) according to the manufacturer’s directions. CFSE-labeled PBMCs were then cultured alone (1-0 PBMC:MSC/FB ratio) for use as a positive control, or co-cultured with titrated numbers of fibroblasts or MSCs ranging from 1-1 down to a 1-0.05 PBMC:MSC/FB ratio. Soluble anti-huCD3 and anti-huCD28 monoclonal antibodies (RnD Systems, Minneapolis, MN) were used to stimulate T cell populations. After four days, cultures were harvested and stained with anti-huCD4 APC (RnD Systems, Inc., Minneapolis, MN). Data acquisition was performed with an Accuri C6 Flow Cytometer (Accuri Cytometer, Ann Arbor, MI). Percent CD4+ T cell stimulation (% CD4+/CFSE-low cells) was gauged using the 1-0.05 non-stimulated negative control gate.
Culture of Macrophages
Macrophages were isolated and cultured as previously described [26,34]. CD14+ cells were plated in IMDM media supplemented with 10% human serum AB, 1% NEAA, 2μM L-alanyl-L-glutamine, 1% sodium pyruvate, and 5μM human recombinant insulin and cultured for 7 days to allow for differentiation into macrophages. Following this time, MSCs or fibroblasts were plated with these macrophages in a 1:10 ratio (105 MSCs:106 macrophages in 6-well plate).
Quantitative Polymerase Chain Reaction (Q-PCR)
RNA was isolated from cells using the RNeasy Micro Kit (Qiagen, Valencia, CA), and converted to cDNA using the Quantitect Reverse Transcription Kit (Qiagen). Q-PCR was performed using Power SYBR green master mix (Applied Biosystems, Foster City, CA) on StepOne Plus instrument (Applied Biosystems). Quantification of IL-10 and IL-12 expressed as mRNA level was normalized to the mRNA of three housekeeping genes (RRN18S, GAPDH and ACTB). Primers were obtained from Qiagen.
Collagen and Fibronectin Immunocytochemistry
Fibroblasts and MSCs were plated on glass coverslips in 24-well plates. For wells to be analyzed for collagen I, cells were treated with 0.2mM ascorbate (Sigma-Aldrich). Monolayers were fixed with 3.7% paraformaldehyde in PBS for 10 minutes, blocked with 1% bovine serum albumin in PBS for 1 hour, incubated with rabbit anti-collagen I or anti-fibronectin [35] for 1 h, and then incubated with anti-rabbit secondary antibody conjugated with Alexa 546 (Jackson ImmunoResearch Laboratories, West Grove, PA). Nonspecific rabbit anti-serum was used as a negative control. Coverslips were counterstained for DNA with 4′,6-diamidino-2-phenylindole (DAPI) and mounted with ProLong Gold anti-fade reagent (Thermo Fisher, Waltham, MA). Coverslips were then visualized by fluorescence microscopy with a Nikon Eclipse Ti inverted microscope and CoolSNAP HQ2 charge-coupled device camera (Photometrics) using the appropriate filters for Alexa 546.
Western Blotting
Equal amounts of protein from cells were electrophoresed on a 10% SDS–PAGE gel. The proteins were transferred to PVDF membranes, which were probed with the following antibodies: anti-FSP1 (Millipore, Billerica, MA) and anti-alpha tubulin (Abcam, Cambridge, MA). HRP-conjugated goat anti-rabbit secondary antibody was used (Cell Signaling) along with Pierce ECL Western Blotting Substrate (Thermo Fisher, Waltham, MA). Protein bands were quantified using ImageJ (NIH, Bethesda, MD).
RESULTS
Fibroblasts and MSCs Share Similar Morphology and Cell Surface Markers
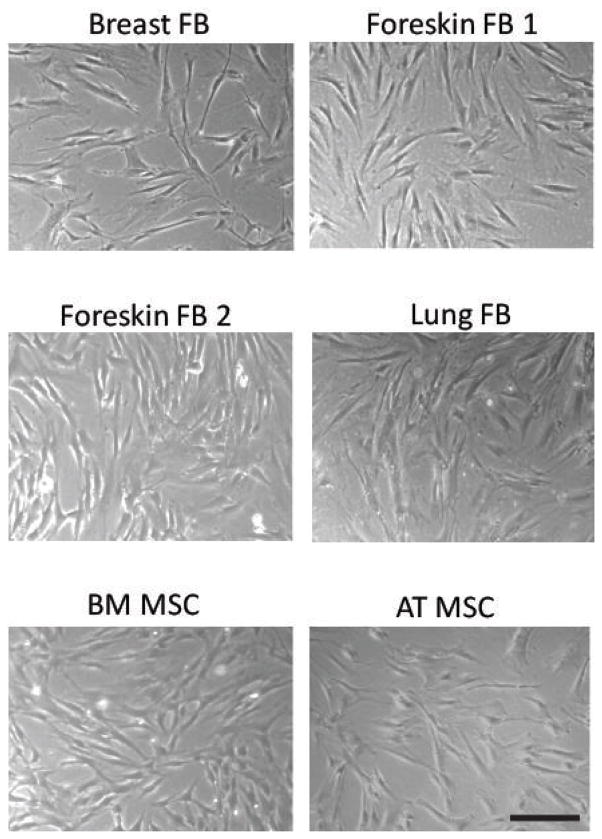
Four fibroblast cell strains from three different human tissue sources (breast, foreskin, and lung) and MSCs from two different human tissue sources (bone marrow and adipose tissue) were grown under appropriate culture conditions. MSCs and fibroblasts adhered to plastic under standard cell culture conditions and did not show any discernible differences in morphology by phase contrast microscopy (Figure 1). We then analyzed fibroblasts for MSC cell surface markers based on criteria from the International Society for Cellular Therapy (ISCT) using flow cytometry [10]. All the fibroblasts tested were positive for CD73, CD90, and CD105; negative for CD14, CD34, CD45, CD19; and HLA-DR, similar to MSCs (Figure 2; Supplemental Figure 1). We also analyzed additional markers that are characteristic of MSCs but are not part of the ISCT consensus criteria; we found that all the fibroblasts were positive for CD29 and CD44, and negative for CD31, similar to MSCs (Figure 2; Supplemental Figure 1).
Figure 1. Fibroblasts and MSCs display similar morphologies.
Representative micrographs at 50x magnification of the different fibroblast strains compared to BM and Ad MSCs. Scale bar = 200μm.
Figure 2. Fibroblasts express cell surface markers that define MSCs.
The graph shows the percent of cells expressing the indicated cell surface markers. Bars represent means ±SE for three independent experiments. Representative flow cytometry histograms are shown in Supplemental Figure 1.
Fibroblasts Differentiate into Adipocytes, Chondrocytes, and Osteoblasts
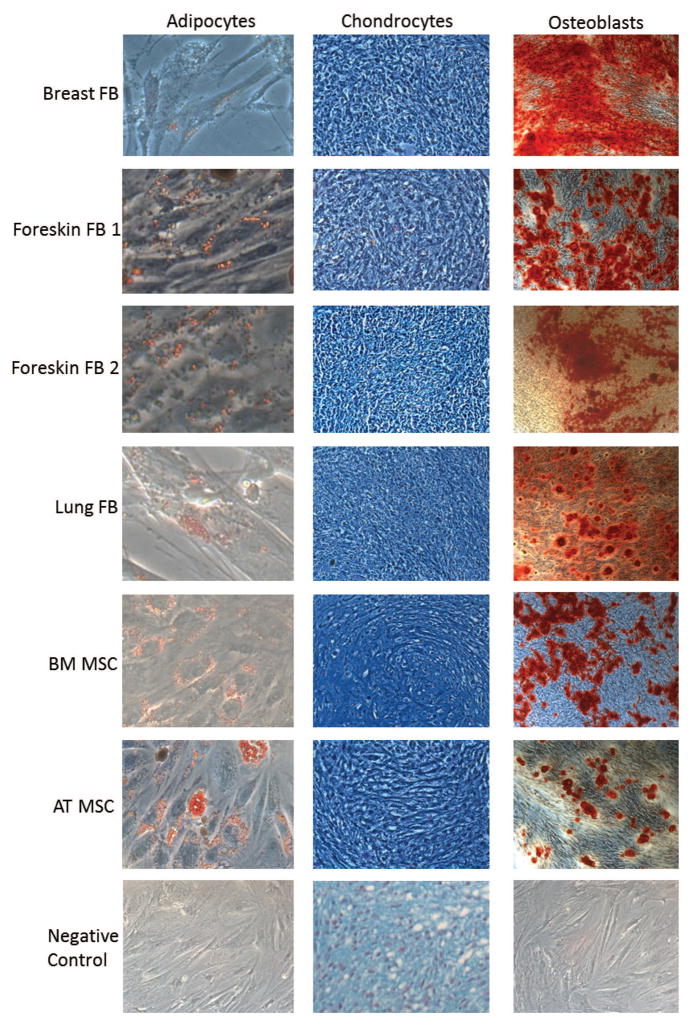
The third criterion that defines a MSC is the ability to differentiate in vitro into adipocytes, chondrocytes, and osteoblasts [10]. Therefore, we investigated whether or not fibroblasts could differentiate into these three cell types. Fibroblasts and MSCs were grown in adipogenic, chondrogenic, and osteogenic conditions, as described in Materials and Methods. Adipocyte lipid droplets were stained with oil red O, osteoblast calcium deposits were stained with Alizarin red S, and paraffin-embedded chondrocyte pellets were stained with Alcian blue (Figure 3). Similar to MSCs, all four fibroblast cell strains were able to differentiate into adipocytes, chondrocytes, and osteoblasts (Figure 3).
Figure 3. Fibroblasts can differentiate into adipocytes, chondrocytes, and osteoblasts.
Fibroblasts were grown in adipogenic, chondrogenic, and osteogenic medias for 21, 24, and 21 days and stained with oil red O, Alcian blue, and Alizarin red S, respectively. BM and Ad MSCs were differentiated the same way and used as positive controls. The micrographs are representative images at 200x magnification for adipocytes, 100x magnification for chondrocytes, and 100x magnification for osteoblasts. Negative controls are BM MSCs cultured for the same lengths of time with normal MSC growth media, fixed and stained the same way.
Fibroblasts Express HLA-DR After IFNγ Stimulation
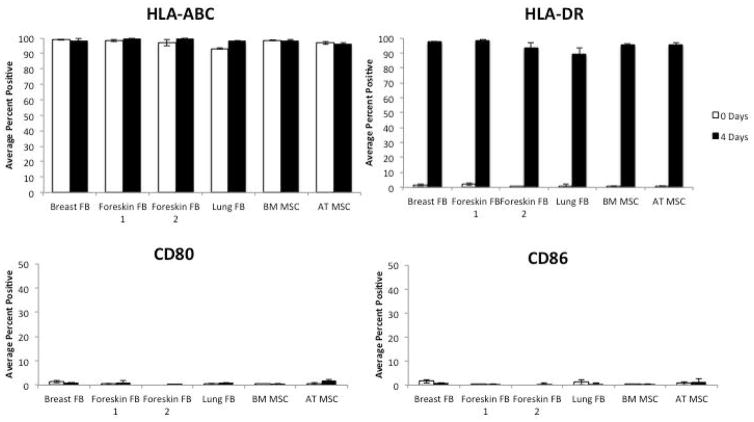
Unstimulated fibroblasts and MSCs were positive for HLA-ABC and negative for HLA-DR and the costimulatory molecules CD80 and CD86 (Figure 4). MSCs are known to express the major histocompatibility complex class II, HLA-DR, following stimulation with IFNγ [10]. Fibroblasts stimulated with IFNγ expressed HLA-DR, similar to MSCs stimulated with IFNγ (Figure 4).
Figure 4. Fibroblasts express HLA-DR when stimulated with IFNγ.
Fibroblasts were plated in a 6-well plate and stimulated with 100 IU/mL IFNγ for 4 days and subsequently probed for HLA-ABC, HLA-DR, CD80, and CD86. BM and Ad MSCs were used as positive controls. Bars represent means ±SE from 3 independent experiments.
Fibroblasts and MSCs Share Immunomodulatory Properties
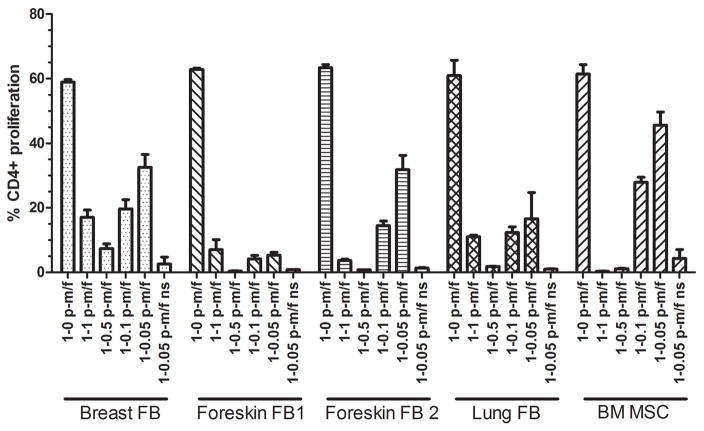
MSCs are well known to suppress T cell proliferation [36]. We sought to determine whether fibroblast strains from different tissues could also inhibit CD4+ T cell proliferation. We used a BM MSC strain that we have established at our laboratory as our reference standard for MSC immunosuppression [33]. For this assay, PBMCs were stained with CFSE and stimulated with anti-CD3 and anti-CD28 antibodies. Fibroblasts or MSCs were co-cultured with CFSE-labeled PBMCs at ratios ranging from a 1-1 PBMC:MSC/FB ratio down to a 1-0.05 PBMC:MSC/FB ratio. 1-0 PBMC:MSC/FB ratios (PBMCs cultured without MSCs or fibroblasts) were used as positive controls. Non-stimulated 1-0.05 ratios were used as negative controls such that gates could be set to measure extent of proliferation for the other (stimulated) cultures. After a four-day incubation period, flow cytometry was used to analyze the percent of CFSE low (proliferative) CD4+ T cells. Fibroblasts were able to suppress CD4+ T cell proliferation levels similar to that of our reference BM MSCs at all ratios tested (Figure 5). Non-stimulated co-cultures (without anti-CD3 and anti-CD28 antibodies; 1-0.05 p/mf ns in Figure 5) demonstrated that fibroblasts, like MSCs, were not immunostimulatory in and of themselves.
Figure 5. Fibroblasts suppress CD4+ T cell proliferation.
PBMCs from a healthy donor were stained with CFSE. Labled PBMCs were plated with four fibroblast strains (FB) or our standard BM MSCs (positive control) at different PBMC:MSC/FB ratios. After a 4-day co-culture, cells were collected, stained with anti-CD4, and analyzed by flow cytometry. A non-stimulated (ns) co-culture at 1-0.05 PBMC:MSC/FB ratio served as a negative control as well as to measure the level of stimulation induced by the strain itself. The value of p-m/f indicates the ratio of PBMCs to MSCs/fibroblasts. Bars represent means ±SE.
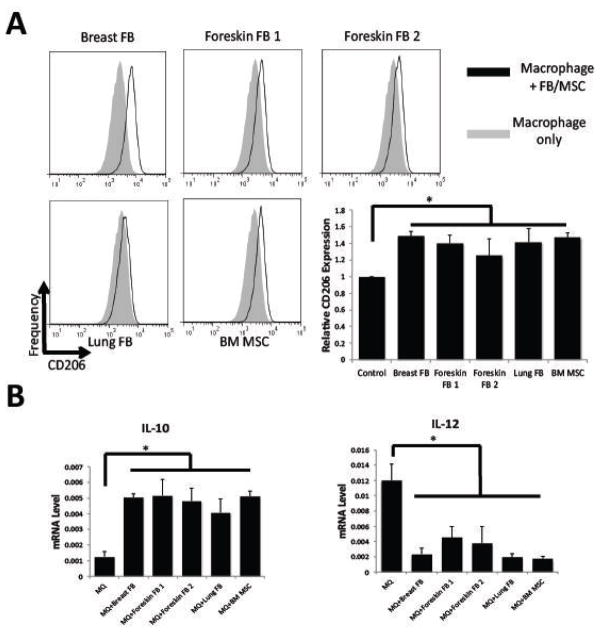
We have previously shown that MSCs are able to modulate the immunophenotype of macrophages towards a unique type of alternatively activated macrophages, which are characterized by increased expression of CD206, high expression of IL-10, and low expression of IL-12 [26] We co-cultured macrophages with fibroblasts and MSCs for 3 days and analyzed expression of CD206 by flow cytometry. We found that all fibroblast strains, similar to MSCs, were able to increase CD206 expression in macrophages (Figure 6A). Furthermore, fibroblasts cultured with macrophages in trans-well experiments induced macrophages to express more IL-10 and less IL-12, as seen in MSC-educated macrophages (Figure 6B).
Figure 6. Immunophenotype of Fibroblast-educated macrophages.
CD14+ cells were isolated from peripheral blood of 3 different donors and plated in 6-well plates (1x106/well) for 7 days to allow for differentiation into macrophages. Then fibroblasts or MSCs (2x105/well) were added and co-cultured for 3 days. Macrophages were harvested by scraping the plates. Cells were stained with anti-CD14 and anti-CD206, and CD206 expression was analyzed in CD14-positive cells. Representative histograms are shown. The bar graph represents the average CD206 mean channel fluorescence in units relative to the control (no MSCs or fibroblasts added to CD14+ cells) ±SE from 3 independent experiments. (B) Q-PCR was used to assess gene expression of IL-10 and IL-12 normalized to 3 housekeeping genes (18S rRNA, GAPDH, and beta-actin). Relative mRNA levels (2^-ΔCt) are plotted. Samples were analyzed in triplicate, and bars represent means ±SE. *P<0.05 compared to control.
MSCs Exhibit Immunohistochemical Properties Characteristic of Fibroblasts
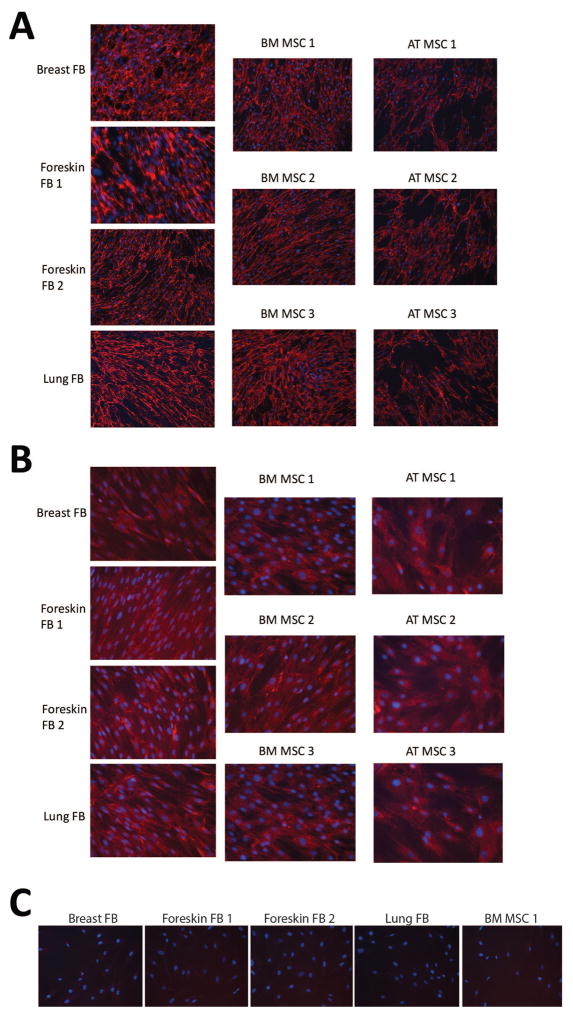
Fibroblasts are typically characterized by their ability to produce fibronectin and collagen matrices, so we investigated whether MSCs produce similar extracellular matrices. Bone marrow-derived MSCs and adipose-tissue derived MSCs, each from 3 different donors, were utilized for these experiments and compared to the 4 different fibroblast strains. Fibroblasts and MSCs were plated on coverslips in 24-well cluster dishes for 3 days and examined by fluorescence microscopy. The fibronectin and type I collagen matrices produced by fibroblasts and MSCs both varied slightly depending on the tissue of origin, but there were no obvious differences that could be used to distinguish fibroblasts from MSCs (Figure 7).
Figure 7. MSCs express fibronectin and collagen matrices that are indistinguishable from that of fibroblasts.
MSCs and fibroblasts were grown on coverslips in 24-well plates to near confluency. For collagen formation, 0.2mM ascorbic acid was added to the media. Samples were probed for fibronectin (A), collagen I (B), or non-specific rabbit serum (C) and analyzed with fluorescent microscopy using the 10x (A) or 20x (B, C) objectives. DNA was counterstained with DAPI (blue). Images are representative micrographs.
Some reports suggest that the fibroblast specific protein 1 (FSP1), a cytoplasmic filament-associated, calcium-binding protein, is a specific marker for fibroblasts [37–39]. Therefore, we analyzed FSP1 expression in fibroblasts and MSCs by western blot, and there were no obvious differences between expression by fibroblasts and MSCs (Supplemental Figure 2). The differences in FSP1 expression among the fibroblast strains were comparable to the differences in FSP1 expression among the MSC strains.
DISCUSSION
Herein, we provide evidence that fibroblasts and MSCs could not be distinguished phenotypically based on their morphology, cell surface markers, differentiation potential, and immunomodulatory properties. We demonstrate that four well-established fibroblast strains express all the cell surface markers of the ISCT consensus criteria that are used to define MSCs [10], and are also capable of differentiating into adipocytes, chondrocytes, and osteoblasts. Additionally, we show that the extracellular matrix secreted by MSCs is similar to that secreted by fibroblasts [40,41]. We also show that the four fibroblast lines we tested can suppress T cell proliferation in vitro, and change the immunophenotype of macrophages similar to MSC-mediated alternative activation of macrophages [26]. The latter observation is intriguing as it could provide an explanation for how anti-inflammatory properties of MSCs are reminiscent of fibroblast proliferation during a wound healing process in vivo, as fibroblasts are presumed to orchestrate the resolution of the inflammatory process via converting inflammatory macrophages to anti-inflammatory macrophages [41,42].
We previously investigated characteristics of vocal fold fibroblasts and found that they have the same cell surface markers, differentiation potential, and immunologic properties as bone marrow- and adipose tissue-derived MSCs [15]. Our results are supported by numerous previous studies. It has been shown that fibroblasts from human bronchus, dermis, and foreskin are similar to bone marrow-derived MSCs in regard to their cell surface markers [16,17,43–46]. Many reports have also demonstrated the ability of fibroblasts from different tissues, including dermis, lung, and orbit, to differentiate into adipocytes, chondrocytes, and osteoblasts [16–19,43,45,47–49]. Furthermore, dermal and foreskin fibroblasts have been shown to be suppress T cell proliferation [20,23,50,51]. Interestingly, as long as thirty years ago fibroblasts were found to suppress lymphocytes via a PGE2-dependent mechanism [52], a mechanism that is now known to partially confer the immunomodulatory properties of MSCs [53]. In addition, large-scale gene expression profiles demonstrate that fibroblasts and MSCs from a number of different sources show very similar patterns [19].
Prior studies have also presented evidence that MSCs exhibit properties typically thought to characterize fibroblasts. One study showed that bone marrow MSCs express the same types of collagens as skin fibroblasts [54]. Another study demonstrated that adipose tissue-derived MSCs make collagen type I, fibronectin, vimentin, and nestin matrices similar to those of dermal fibroblasts [17], and another report demonstrated these same findings in bone marrow MSCs compared to adult bronchial and fetal lung fibroblasts [16].
While some studies have shown certain similarities between MSCs and fibroblasts, the literature is beset by contrary reports. For example, Pittenger et al. and Cappellesso-Fleury et al. demonstrated that fibroblasts are incapable of differentiating into adipocytes, chondrocytes, and osteoblasts [8], while other studies show that fibroblasts do possess these differentiation capabilities [15,16]. Some studies suggest that such differentiation capability may depend on the tissue of origin; for example, Covas et al. showed that skin fibroblasts but not foreskin fibroblasts are capable of differentiating into adipocytes, chondrocytes, and osteoblasts [19]. Several large-scale gene expression studies comparing MSCs and fibroblasts have noted some differences in a variety of genes too [24,25]. However, it is also well known that even fibroblasts display distinct and characteristic transcriptional gene expression patterns depending on the anatomic site or origin [55,56]. Furthermore, the magnitude of differences reported in the literature between MSCs and fibroblasts are comparable to differences reported in the literature among MSCs derived from different tissues. Furthermore, in studies where fibroblasts and MSCs have been reported to be different, the differences are no greater than the differences between MSCs from different tissues and no greater than the differences between fibroblasts from different tissues [18]. Given that differences in culture conditions or derivation methodologies could affect the phenotype of these cells [57,58], it is not surprising that these differences are found in gene expression studies comparing MSCs and fibroblasts.
Thus, neither the MSC criteria established by the ISCT, including cell surface markers and differentiation capabilities, nor immunohistochemical properties that are characteristic of fibroblasts are capable of clearly distinguishing MSCs from fibroblasts. We found that fibroblasts meet all the criteria set forth by the ISCT for defining an MSC, and conversely, our assays show that MSCs display fibroblast immunohistochemical characteristics. Importantly, recent in vivo data is emerging in the literature that point to MSC-like cells as a major cellular origin of fibrosis by demonstrating that in mice perivascular Gli1(+) cells express a typical MSC marker pattern in many organs, are plastic adherent, and can differentiate into adipocytes, chondrocytes and osteoblasts in vitro [59]. Importantly, after organ injury resident Gli1(+) cells expand and become myofibroblasts and contribute to organ fibrosis.
As we and others have proposed, fibrosis is an evolutionarily conserved defense mechanism against many different pathologies through which fibrosis is the consequence of (myo)fibroblast cell efforts to suppress inflammation [60,61]. This could provide an explanation for why MSC/fibroblasts have such a wide range of immunomodulatory and anti-inflammatory properties, both in vitro and in vivo. We propose MSCs and fibroblasts could not be distinguished based on the commonly used phenotypic characterization methodologies and could likely represent the same cell type.
CONCLUSIONS
Fibroblasts and MSCs cannot be distinguished based on morphology, cell surface markers, differentiation potential, or immunomodulatory properties. Notably, fibroblasts share immunomodulatory properties of MSCs, such as suppression of T cell proliferation and modulation of macrophage immunophenotype. Our results suggest that MSCs and fibroblasts could likely represent the same cell type.
Supplementary Material
Acknowledgments
We would like to thank Fran Fogerty for her assistance with the analysis of collagen and fibronectin expression and the University of Wisconsin Carbone Cancer Center’s Experimental Pathology Lab, supported by University of Wisconsin Carbone Cancer Center Cancer Center Support Grant P30 CA014520, for processing and staining the chondrogenic pellets.
Abbreviations
- AT MSC
adipose tissue-derived mesenchymal stem cell
- BM MSC
bone marrow-derived mesenchymal stem cell
- CFSE
carboxyfluorescein succinimidyl ester
- FB
fibroblast
- FSP1
fibroblast specific protein 1
- IDO
indoleamine 2,3-dioxygenase (IDO)
- IFNγ
interferon gamma
- ISCT
International Society for Cellular Therapy
- MSC
mesenchymal stromal/stem cell
Footnotes
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Author Contributions:
R.A.D.: conception and design, collection and assembly of data, manuscript writing;
D.D.B.: collection and assembly of data, manuscript writing;
S.N.: collection and assembly of data;
A.D.G.: data interpretation, manuscript writing;
J.K.: collection and assembly of data;
D.F.M.: conception and design, provision of study material, manuscript writing;
P.H.: conception and design, provision of study material, data interpretation, manuscript writing
Contributor Information
Ryan A Denu, Email: rdenu@wisc.edu.
Steven Nemcek, Email: stevennemcek@gmail.com.
Debra D Bloom, Email: ddbloom@medicine.wisc.edu.
A Daisy Goodrich, Email: daisy@unr.edu.
Jaehyup Kim, Email: kim.jaehyup@gmail.com.
Deane F Mosher, Email: dfm1@medicine.wisc.edu.
Peiman Hematti, Email: pxh@medicine.wisc.edu.
References
- 1.Horwitz EM, Le Blanc K, Dominici M, Mueller I, Slaper-Cortenbach I, Marini FC, Deans RJ, Krause DS, Keating A. Clarification of the nomenclature for msc: The international society for cellular therapy position statement. Cytotherapy. 2005;7:393–395. doi: 10.1080/14653240500319234. [DOI] [PubMed] [Google Scholar]
- 2.Friedenstein AJ, Chailakhjan RK, Lalykina KS. The development of fibroblast colonies in monolayer cultures of guinea-pig bone marrow and spleen cells. Cell and tissue kinetics. 1970;3:393–403. doi: 10.1111/j.1365-2184.1970.tb00347.x. [DOI] [PubMed] [Google Scholar]
- 3.Friedenstein AJ. Marrow stromal fibroblasts. Calcif Tissue Int. 1995;56(Suppl 1):S17. [Google Scholar]
- 4.Friedenstein AJ, Chailakhyan RK, Latsinik NV, Panasyuk AF, Keiliss-Borok IV. Stromal cells responsible for transferring the microenvironment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo Transplantation. 1974;17:331–340. doi: 10.1097/00007890-197404000-00001. [DOI] [PubMed] [Google Scholar]
- 5.Friedenstein AJ. Precursor cells of mechanocytes. International review of cytology. 1976;47:327–359. doi: 10.1016/s0074-7696(08)60092-3. [DOI] [PubMed] [Google Scholar]
- 6.Friedenstein AJ, Lalykina KS. Thymus cells are inducible to osteogenesis. Eur J Immunol. 1972;2:602–603. doi: 10.1002/eji.1830020624. [DOI] [PubMed] [Google Scholar]
- 7.Caplan AI. Mesenchymal stem cells. J Orthop Res. 1991;9:641–650. doi: 10.1002/jor.1100090504. [DOI] [PubMed] [Google Scholar]
- 8.Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 9.Viswanathan S, Keating A, Deans R, Hematti P, Prockop D, Stroncek DF, Stacey G, Weiss DJ, Mason C, Rao MS. Soliciting strategies for developing cell-based reference materials to advance mesenchymal stromal cell research and clinical translation. Stem Cells Dev. 2014;23:1157–1167. doi: 10.1089/scd.2013.0591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy. 2006;8:315–317. doi: 10.1080/14653240600855905. [DOI] [PubMed] [Google Scholar]
- 11.Gurd FB. A histological study of the skin lesions of pellagra. J Exp Med. 1911;13:98–114. doi: 10.1084/jem.13.1.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rinkevich Y, Walmsley GG, Hu MS, Maan ZN, Newman AM, Drukker M, Januszyk M, Krampitz GW, Gurtner GC, Lorenz HP, Weissman IL, Longaker MT. Skin fibrosis. Identification and isolation of a dermal lineage with intrinsic fibrogenic potential. Science. 2015;348:aaa2151. doi: 10.1126/science.aaa2151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Darby IA, Hewitson TD. Fibroblast differentiation in wound healing and fibrosis. Int Rev Cytol. 2007;257:143–179. doi: 10.1016/S0074-7696(07)57004-X. [DOI] [PubMed] [Google Scholar]
- 14.Todaro GJ, Green H. Quantitative studies of the growth of mouse embryo cells in culture and their development into established lines. J Cell Biol. 1963;17:299–313. doi: 10.1083/jcb.17.2.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hanson SE, Kim J, Johnson BH, Bradley B, Breunig MJ, Hematti P, Thibeault SL. Characterization of mesenchymal stem cells from human vocal fold fibroblasts. Laryngoscope. 2010;120:546–551. doi: 10.1002/lary.20797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sabatini F, Petecchia L, Tavian M, Jodon de Villeroche V, Rossi GA, Brouty-Boye D. Human bronchial fibroblasts exhibit a mesenchymal stem cell phenotype and multilineage differentiating potentialities. Laboratory investigation; a journal of technical methods and pathology. 2005;85:962–971. doi: 10.1038/labinvest.3700300. [DOI] [PubMed] [Google Scholar]
- 17.Lorenz K, Sicker M, Schmelzer E, Rupf T, Salvetter J, Schulz-Siegmund M, Bader A. Multilineage differentiation potential of human dermal skin-derived fibroblasts. Exp Dermatol. 2008;17:925–932. doi: 10.1111/j.1600-0625.2008.00724.x. [DOI] [PubMed] [Google Scholar]
- 18.Jääger K, Neuman T. Human dermal fibroblasts exhibit delayed adipogenic differentiation compared with mesenchymal stem cells. Stem Cells Dev. 2011;20:1327–1336. doi: 10.1089/scd.2010.0258. [DOI] [PubMed] [Google Scholar]
- 19.Covas DT, Panepucci RA, Fontes AM, Silva WA, Orellana MD, Freitas MC, Neder L, Santos AR, Peres LC, Jamur MC, Zago MA. Multipotent mesenchymal stromal cells obtained from diverse human tissues share functional properties and gene-expression profile with cd146+ perivascular cells and fibroblasts. Exp Hematol. 2008;36:642–654. doi: 10.1016/j.exphem.2007.12.015. [DOI] [PubMed] [Google Scholar]
- 20.Haniffa MA, Wang XN, Holtick U, Rae M, Isaacs JD, Dickinson AM, Hilkens CM, Collin MP. Adult human fibroblasts are potent immunoregulatory cells and functionally equivalent to mesenchymal stem cells. J Immunol. 2007;179:1595–1604. doi: 10.4049/jimmunol.179.3.1595. [DOI] [PubMed] [Google Scholar]
- 21.Alt E, Yan Y, Gehmert S, Song YH, Altman A, Vykoukal D, Bai X. Fibroblasts share mesenchymal phenotypes with stem cells, but lack their differentiation and colony-forming potential. Biol Cell. 2011;103:197–208. doi: 10.1042/BC20100117. [DOI] [PubMed] [Google Scholar]
- 22.Chang Y, Li H, Guo Z. Mesenchymal stem cell-like properties in fibroblasts. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology. 2014;34:703–714. doi: 10.1159/000363035. [DOI] [PubMed] [Google Scholar]
- 23.Cappellesso-Fleury S, Puissant-Lubrano B, Apoil PA, Titeux M, Winterton P, Casteilla L, Bourin P, Blancher A. Human fibroblasts share immunosuppressive properties with bone marrow mesenchymal stem cells. J Clin Immunol. 2010;30:607–619. doi: 10.1007/s10875-010-9415-4. [DOI] [PubMed] [Google Scholar]
- 24.Brendel C, Kuklick L, Hartmann O, Kim TD, Boudriot U, Schwell D, Neubauer A. Distinct gene expression profile of human mesenchymal stem cells in comparison to skin fibroblasts employing cdna microarray analysis of 9600 genes. Gene Expr. 2005;12:245–257. doi: 10.3727/000000005783992043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ishii M, Koike C, Igarashi A, Yamanaka K, Pan H, Higashi Y, Kawaguchi H, Sugiyama M, Kamata N, Iwata T, Matsubara T, Nakamura K, Kurihara H, Tsuji K, Kato Y. Molecular markers distinguish bone marrow mesenchymal stem cells from fibroblasts. Biochem Biophys Res Commun. 2005;332:297–303. doi: 10.1016/j.bbrc.2005.04.118. [DOI] [PubMed] [Google Scholar]
- 26.Kim J, Hematti P. Mesenchymal stem cell-educated macrophages: A novel type of alternatively activated macrophages. Experimental hematology. 2009;37:1445–1453. doi: 10.1016/j.exphem.2009.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Toledo-Piza AR, Nakano E, Rici RE, Maria DA. Proliferation of fibroblasts and endothelial cells is enhanced by treatment with phyllocaulis boraceiensis mucus. Cell Prolif. 2013;46:97–108. doi: 10.1111/cpr.12003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Du ZQ, Yang CX, Rothschild MF, Ross JW. Novel microrna families expanded in the human genome. BMC Genomics. 2013;14:98. doi: 10.1186/1471-2164-14-98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nichols WW, Murphy DG, Cristofalo VJ, Toji LH, Greene AE, Dwight SA. Characterization of a new human diploid cell strain, imr-90. Science. 1977;196:60–63. doi: 10.1126/science.841339. [DOI] [PubMed] [Google Scholar]
- 30.Tomasini-Johansson BR, Johnson IA, Hoffmann FM, Mosher DF. Quantitative microtiter fibronectin fibrillogenesis assay: Use in high throughput screening for identification of inhibitor compounds. Matrix Biol. 2012;31:360–367. doi: 10.1016/j.matbio.2012.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Allen-Hoffmann BL, Schlosser SJ, Brondyk WH, Fahl WE. Fibronectin levels are enhanced in human fibroblasts overexpressing the c-sis protooncogene. J Biol Chem. 1990;265:5219–5225. [PubMed] [Google Scholar]
- 32.Kuperwasser C, Chavarria T, Wu M, Magrane G, Gray JW, Carey L, Richardson A, Weinberg RA. Reconstruction of functionally normal and malignant human breast tissues in mice. Proc Natl Acad Sci U S A. 2004;101:4966–4971. doi: 10.1073/pnas.0401064101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bloom DD, Centanni JM, Bhatia N, Emler CA, Drier D, Leverson GE, McKenna DH, Gee AP, Lindblad R, Hei DJ, Hematti P. A reproducible immunopotency assay to measure mesenchymal stromal cell-mediated t-cell suppression. Cytotherapy. 2015;17:140–151. doi: 10.1016/j.jcyt.2014.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kim J, Denu RA, Dollar BA, Escalante LE, Kuether JP, Callander NS, Asimakopoulos F, Hematti P. Macrophages and mesenchymal stromal cells support survival and proliferation of multiple myeloma cells. Br J Haematol. 2012;158:336–346. doi: 10.1111/j.1365-2141.2012.09154.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Furcht LT, Smith D, Wendelschafer-Crabb G, Mosher DF, Foidart JM. Fibronectin presence in native collagen fibrils of human fibroblasts: Immunoperoxidase and immunoferritin localization. J Histochem Cytochem. 1980;28:1319–1333. doi: 10.1177/28.12.7014712. [DOI] [PubMed] [Google Scholar]
- 36.Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P, Grisanti S, Gianni AM. Human bone marrow stromal cells suppress t-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood. 2002;99:3838–3843. doi: 10.1182/blood.v99.10.3838. [DOI] [PubMed] [Google Scholar]
- 37.Strutz F, Okada H, Lo CW, Danoff T, Carone RL, Tomaszewski JE, Neilson EG. Identification and characterization of a fibroblast marker: Fsp1. J Cell Biol. 1995;130:393–405. doi: 10.1083/jcb.130.2.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mazzucchelli L. Protein s100a4: Too long overlooked by pathologists? Am J Pathol. 2002;160:7–13. doi: 10.1016/S0002-9440(10)64342-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Barraclough R. Calcium-binding protein s100a4 in health and disease. Biochim Biophys Acta. 1998;1448:190–199. doi: 10.1016/s0167-4889(98)00143-8. [DOI] [PubMed] [Google Scholar]
- 40.Connor NS, Aubin JE, Sodek J. Independent expression of type i collagen and fibronectin by normal fibroblast-like cells. J Cell Sci. 1983;63:233–244. doi: 10.1242/jcs.63.1.233. [DOI] [PubMed] [Google Scholar]
- 41.Ross R. The fibroblast and wound repair. Biol Rev Camb Philos Soc. 1968;43:51–96. doi: 10.1111/j.1469-185x.1968.tb01109.x. [DOI] [PubMed] [Google Scholar]
- 42.Mosser DM, Edwards JP. Exploring the full spectrum of macrophage activation. Nature reviews. 2008;8:958–969. doi: 10.1038/nri2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wagner W, Wein F, Seckinger A, Frankhauser M, Wirkner U, Krause U, Blake J, Schwager C, Eckstein V, Ansorge W, Ho AD. Comparative characteristics of mesenchymal stem cells from human bone marrow, adipose tissue, and umbilical cord blood. Experimental hematology. 2005;33:1402–1416. doi: 10.1016/j.exphem.2005.07.003. [DOI] [PubMed] [Google Scholar]
- 44.Wirths S, Malenke E, Kluba T, Rieger S, Müller MR, Schleicher S, Hann von Weyhern C, Nagl F, Fend F, Vogel W, Mayer F, Kanz L, Bühring HJ, Kopp HG. Shared cell surface marker expression in mesenchymal stem cells and adult sarcomas. Stem Cells Transl Med. 2013;2:53–60. doi: 10.5966/sctm.2012-0055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Brohem CA, de Carvalho CM, Radoski CL, Santi FC, Baptista MC, Swinka BB, de Urban AC, de Araujo LR, Graf RM, Feferman IH, Lorencini M. Comparison between fibroblasts and mesenchymal stem cells derived from dermal and adipose tissue. Int J Cosmet Sci. 2013 doi: 10.1111/ics.12064. [DOI] [PubMed] [Google Scholar]
- 46.Bae S, Ahn JH, Park CW, Son HK, Kim KS, Lim NK, Jeon CJ, Kim H. Gene and microrna expression signatures of human mesenchymal stromal cells in comparison to fibroblasts. Cell Tissue Res. 2009;335:565–573. doi: 10.1007/s00441-008-0729-y. [DOI] [PubMed] [Google Scholar]
- 47.Lorenz K, Sicker M, Schmelzer E, Rupf T, Salvetter J, Schulz-Siegmund M, Bader A. Multilineage differentiation potential of human dermal skin-derived fibroblasts. Exp Dermatol. 2008;17:925–932. doi: 10.1111/j.1600-0625.2008.00724.x. [DOI] [PubMed] [Google Scholar]
- 48.Friedenstein AJ, Chailakhyan RK, Gerasimov UV. Bone marrow osteogenic stem cells: In vitro cultivation and transplantation in diffusion chambers. Cell Tissue Kinet. 1987;20:263–272. doi: 10.1111/j.1365-2184.1987.tb01309.x. [DOI] [PubMed] [Google Scholar]
- 49.Sorisky A, Pardasani D, Gagnon A, Smith TJ. Evidence of adipocyte differentiation in human orbital fibroblasts in primary culture. J Clin Endocrinol Metab. 1996;81:3428–3431. doi: 10.1210/jcem.81.9.8784110. [DOI] [PubMed] [Google Scholar]
- 50.Price S. Experimental arthritis: Dermal fibroblasts have msc-like immunosuppressive effects in vivo. Nat Rev Rheumatol. 2011;7:437. doi: 10.1038/nrrheum.2011.89. [DOI] [PubMed] [Google Scholar]
- 51.Jones S, Horwood N, Cope A, Dazzi F. The antiproliferative effect of mesenchymal stem cells is a fundamental property shared by all stromal cells. J Immunol. 2007;179:2824–2831. doi: 10.4049/jimmunol.179.5.2824. [DOI] [PubMed] [Google Scholar]
- 52.Korn JH. Modulation of lymphocyte mitogen responses by cocultured fibroblasts. Cell Immunol. 1981;63:374–384. doi: 10.1016/0008-8749(81)90016-2. [DOI] [PubMed] [Google Scholar]
- 53.Prockop DJ, Oh JY. Mesenchymal stem/stromal cells (mscs): Role as guardians of inflammation. Molecular therapy: the journal of the American Society of Gene Therapy. 2012;20:14–20. doi: 10.1038/mt.2011.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Castro-Malaspina H, Gay RE, Resnick G, Kapoor N, Meyers P, Chiarieri D, McKenzie S, Broxmeyer HE, Moore MA. Characterization of human bone marrow fibroblast colony-forming cells (cfu-f) and their progeny. Blood. 1980;56:289–301. [PubMed] [Google Scholar]
- 55.Chang HY, Chi JT, Dudoit S, Bondre C, van de Rijn M, Botstein D, Brown PO. Diversity, topographic differentiation, and positional memory in human fibroblasts. Proc Natl Acad Sci U S A. 2002;99:12877–12882. doi: 10.1073/pnas.162488599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Rinn JL, Bondre C, Gladstone HB, Brown PO, Chang HY. Anatomic demarcation by positional variation in fibroblast gene expression programs. PLoS Genet. 2006;2:e119. doi: 10.1371/journal.pgen.0020119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Samuelsson H, Ringden O, Lonnies H, Le Blanc K. Optimizing in vitro conditions for immunomodulation and expansion of mesenchymal stromal cells. Cytotherapy. 2009;11:129–136. doi: 10.1080/14653240802684194. [DOI] [PubMed] [Google Scholar]
- 58.Schellenberg A, Stiehl T, Horn P, Joussen S, Pallua N, Ho AD, Wagner W. Population dynamics of mesenchymal stromal cells during culture expansion. Cytotherapy. 2011 doi: 10.3109/14653249.2011.640669. [DOI] [PubMed] [Google Scholar]
- 59.Kramann R, Schneider RK, DiRocco DP, Machado F, Fleig S, Bondzie PA, Henderson JM, Ebert BL, Humphreys BD. Perivascular gli1(+) progenitors are key contributors to injury-induced organ fibrosis. Cell Stem Cell. 2015;16:51–66. doi: 10.1016/j.stem.2014.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hematti P. Mesenchymal stromal cells and fibroblasts: A case of mistaken identity? Cytotherapy. 2012;14:516–521. doi: 10.3109/14653249.2012.677822. [DOI] [PubMed] [Google Scholar]
- 61.Thannickal VJ, Zhou Y, Gaggar A, Duncan SR. Fibrosis: Ultimate and proximate causes. J Clin Invest. 2014;124:4673–4677. doi: 10.1172/JCI74368. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.