Abstract
Functional neural competence and integrity require interactive exchanges among sensory and motor neurons, interneurons and glial cells. Recent studies have attributed some of the tasks needed for these exchanges to extracellular vesicles (such as exosomes and microvesicles), which are most prominently involved in shuttling reciprocal signals between myelinating glia and neurons, thus promoting neuronal survival, the immune response mediated by microglia, and synapse assembly and plasticity. Such vesicles have also been identified as important factors in the spread of neurodegenerative disorders and brain cancer. These extracellular vesicle functions add a previously unrecognized level of complexity to transcellular interactions within the nervous system.
Considerable effort has been devoted to understanding the mechanisms by which neurons and glia communicate with each other, as well as with innervated tissue, such as muscles. Such studies have revealed, in great molecular detail, processes such as transcellular signalling through chemical and electrical synapses, the plasticity-inducing influence of specific patterns of electrical activity, and mechanisms of anterograde signalling and retrograde signalling through neurotrophins and growth factors, and their effects on synaptic strength. In recent years, signal transmission through extracellular vesicles (EVs) has emerged as a new aspect of transcellular signalling, and our growing appreciation of this process is likely to have a considerable influence on our understanding of how glia, neurons and innervated targets communicate.
EVs carry cargo that was previously not thought to readily cross the plasma membrane barrier; such cargo includes membrane proteins, cytosolic proteins and genetic material such as mRNAs and non-coding RNAs, such as microRNAs (miRNAs). Thus, the observation that EVs can function in the intercellular transport of these molecules within the nervous system opens an entirely new perspective on cellular communication in the brain. Here, we summarize the evidence for the existence and relevance of EVs in neuron–neuron, neuron–glial cell and neuron–muscle communication, as well as major emerging themes in the involvement of EVs in neurological disease.
Biology of EVs
The study of EVs is still in its infancy and much investigation needs to be conducted to understand EV biogenesis, composition and function, and we refer readers to an excellent and extensive review that discusses these aspects1. Nevertheless, as a foundation to our Review, in which we describe our current knowledge of the roles of EVs in the nervous system, we provide a brief definition of the types and known features of EVs below.
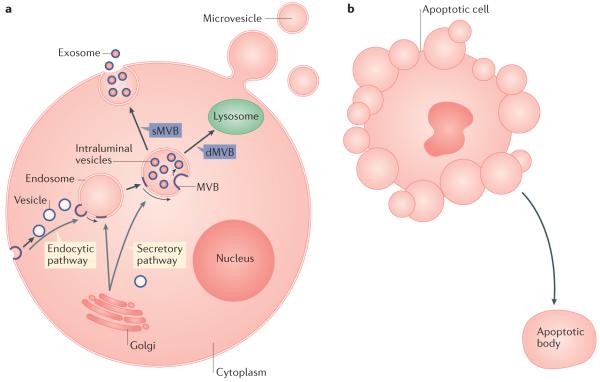
EVs are vesicles that are liberated from cells into the extracellular space and can be subdivided into microvesicles (100 nm to 1 μm in diameter) and exosomes (30–100 nm in diameter), depending on their subcellular origin: the plasma membrane or multivesicular bodies (MVBs)1 (FIG. 1a). Apoptotic cells also shed vesicles (0.8–5.0 μm in diameter) into the extracellular space; these vesicles are called apoptotic bodies and share some characteristics with microvesicles2. However, apoptotic bodies probably do not have a role in transcellular communication in the nervous system, as, following their release, they are rapidly engulfed by phagocytic cells (FIG. 1b).
Figure 1. Extracellular vesicle formation and release.
a ∣ Multivesicular bodies (MVBs), the cellular source of exosomes, can form via the endocytic or the secretory pathway. In this process, vesicles that originate by endocytosis at the plasma membrane or that are generated by the Golgi complex fuse with the limiting membrane of an endosomal compartment and bud inwardly into the lumen of the endosome (the movement of membrane constituents from the plasma membrane or Golgi complex to the exosome membranes is depicted by a blue line boundary). Although degradative MVBs (dMVBs) subsequently fuse with the lysosome, leading to MVB content degradation, secretory MVBs (sMVBs) fuse with the plasma membrane, releasing exosomes into the extracellular space. In contrast to exosomes, microvesicles form by the outward budding of the plasma membrane, which releases microvesicles after the bud pinches off from the cell surface. b ∣ A cell undergoing apoptosis sheds apoptotic bodies, which bud off from the plasma membrane.
The mechanisms of EV formation and release into the extracellular space rely on the endosomal sorting complex required for transport (ESCRT)3 (BOX 1) or lipid-dependent processes4 (BOX 2), among other factors.
Box 1. The endosomal sorting complex required for transport.
The endosomal sorting complex required for transport (ESCRT) is involved in transporting cargo to the lysosome for degradation. In this process, a multivesicular body (MVB) is formed by the inward budding of an endosomal limiting membrane, giving rise to intraluminal vesicles (ILVs; FIG. 1a). The ESCRT is crucial for selecting and recruiting cargoes for ILVs and for forming and pinching off the inwardly budding vesicles that become ILVs. However, a plethora of functions seem to require ESCRT function, particularly ESCRT-III function, from the exit of a virus from the cell to nuclear envelope sealing during cytokinesis.
The ESCRT comprises more than 30 proteins, which are organized into five discrete complexes with distinct functions. In Metazoa, ESCRT-0 is composed of hepatocyte growth factor-regulated tyrosine kinase substrate (HRS) and signal transducing adaptor molecule (STAM). ESCRT-0 binds and clusters ubiquitylated proteins that are destined for degradation. Moreover, it binds to phosphatidylinositol 3-phosphate at the endosomal surface, thus recruiting ubiquitin-tagged proteins to the endosome. ESCRT-0 also recruits ESCRT-I to the endosome.
ESCRT-I is a heterotetramer composed of tumour susceptibility gene 101 (TSG101), vacuolar protein sorting-associated protein 28 (VPS28), VPS37 and ubiquitin-associated protein 1 (UBAP1). It clusters ubiquitylated proteins, establishing a link between ESCRT-0 and ESCRT-II, recognizes and remodels membranes during scission, and recruits ESCRT-III.
ESCRT-II is a heterotetrameric complex consisting of SNF8 (also known as EAP30), VPS25 (also known as EAP20) and VPS36 (also known as EAP45). After binding ESCRT-0 and ESCRT-I, ubiquitylated proteins bind to ESCRT-II, which establishes a bridge with ESCRT-III.
ESCRT-III is composed of charged multivesicular body proteins (CHMP2–CHMP7) and IST1. Unlike ESCRT-0, ESCRT-I and ESCRT-II, which are stable complexes, ESCRT-III is a transient complex. It assembles into a long filamentous polymer that coils around the neck of the inwardly budding vesicles, just before membrane scission, and pinches off the neck. It also forms a plug that prevents the escape of vesicle cargo before scission. VPS4 is an AAA ATPase that disassembles polymerized ESCRT-III. Before VPS4 performs its function, ALG 2 interacting protein X (ALIX; also known as PDCD6IP) recruits deubiquitinases to the ESCRT-III complex to remove ubiquitin from cargo proteins.
Box 2. Lipid-dependent mechanisms of extracellular vesicle formation.
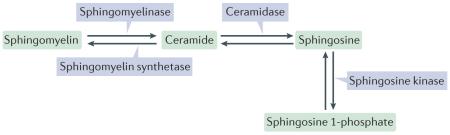
Evidence for an endosomal sorting complex required for transport (ESCRT)-independent model of multivesicular body (MVB) formation has been presented, and this mechanism might be used for the formation of secretory MVBs (sMVBs). However, this model still requires thorough experimental investigation. The lipid composition of exosomes and microvesicles has been related to their ability to form or to pinch off from the plasma membrane. Sphingomyelin and its metabolic products, particularly ceramide and sphingosine 1-phosphate (see the figure), have a major role in exosome biogenesis and microvesicle shedding, two topologically similar membrane-budding events (inward budding occurs away from the cytosol). Pharmacological inhibition of neutral sphingomyelinase, which catalyses the synthesis of ceramide from sphingomyelin, inhibits exosome release from oligodendrocytes. By contrast, the synthesis of ceramide by acidic sphingomyelinase promotes microvesicle shedding by microglial cells.
Ceramide promotes membrane bending owing to its cone-like structure, and its downstream product, sphingosine 1-phosphate, regulates the constitutive production of exosomes by sMVBs and microvesicle shedding from microglial cells. Several sphingosine 1-phosphate target receptors have been identified and subgrouped into distinct G protein-coupled receptor families. Ceramide can also give rise to sphingomyelin, which is catalysed by sphingomyelin synthetase (see the figure). Blocking sphingomyelin synthetase leads to a rise in ceramide levels and consequently enhanced exosome secretion. Thus, sphingosine metabolism appears to be a common theme in extracellular vesicle production. Phospholipase D1 and phospholipase D2, which metabolize phosphatidylcholine into choline and phosphatidic acid, are also implicated in both microvesicle and exosome budding by eliciting changes in membrane curvature through interactions of their charged head groups with the membrane. Whether the ESCRT complex could link membrane budding to lipid composition is unknown, but it has been hypothesized that membrane asymmetries in phosphatidylcholine and phosphatidylinositol could lead to the binding of ESCRT-III to the membrane, thus facilitating vesicle budding.
Microvesicles
Microvesicles are formed through outward budding and vesicle shedding at the plasma membrane (FIG. 1a). They typically form at plasma membrane microdomains called lipid rafts, which are ordered membrane entities that are enriched in cholesterol and glycosphingolipids5. Microglia-derived microvesicles are enriched in phosphatidylserine and phophatidylinositol in the external membrane leaflet, a feature that is shared with apoptotic bodies but that distinguishes microvesicles from exosomes.
Exosomes
Exosomes, also referred to as intraluminal vesicles (ILVs) when they are inside cells, are thought to be derived from the endomembrane system and contain ceramide, a lipid produced from sphingosine6 (FIGS 1,2). This composition favours lipid raft organization at the exosomal membrane, similar to that in microvesicles. Exosomes are formed by the inward budding of the limiting membrane of an endomembrane compartment, which then gives rise to an MVB containing exosomes or ILVs in its lumen6. MVBs can be subdivided into two types, which we refer to as secretory MVBs (sMVBs) and degradative MVBs (dMVBs) in this Review.
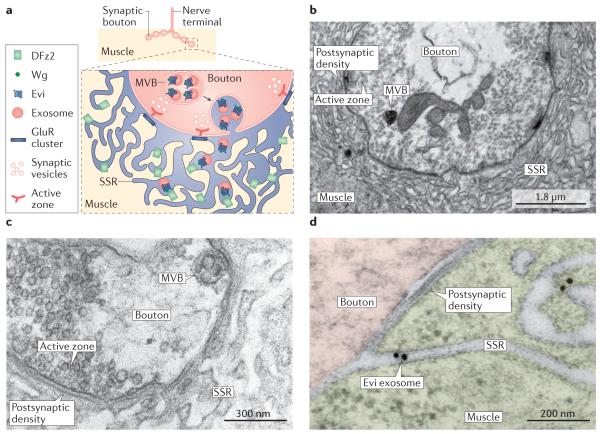
Figure 2. Release of Evi-containing exosomes at the Drosophila melanogaster neuromuscular junction.
a ∣ At the Drosophila melanogaster neuromuscular junction, exosomes are released by motor neuron terminals (synaptic boutons) and are received by muscles. In the synaptic boutons (see inset box), multivesicular bodies (MVBs) fuse with the plasma membrane at extrasynaptic sites, away from active zones, and release exosomes containing Evi and Wingless (Wg). Exosomes travel through extracellular canals formed by the subsynaptic reticulum (SSR), a folded structure that is formed at the junctional region of the muscle cell, and eventually interact with DFrizzled-2 (DFz2) receptors, localized deep in the SSR. b ∣ Cross-section of a synaptic bouton showing an MVB at the ultrastructural level. An active zone and its apposed postsynaptic density, as well as the folded muscle SSR, are indicated. c ∣ Electron micrograph of a synaptic bouton showing an MVB docked at the presynaptic bouton membrane, away from sites of neurotransmitter release. d ∣ High-magnification view of a presynaptic bouton region and the postsynaptic SSR in an animal that expressed Evi-GFP (green fluorescent protein) exclusively in neurons, immunolabelled with gold granules. Note the presence of a gold-labelled Evi exosome that is outside the bouton and within an SSR canal. A nearby postsynaptic density is also shown. Part b is modified with permission from REF. 49, Elsevier. Parts c and d are modified with permission from REF. 48, © 2016 The American Society for Biochemistry and Molecular Biology.
In sMVBs, exosomes are thought to be derived from endocytic vesicles that fuse with the limiting membrane of the endomembrane compartment and then undergo inward budding from this compartment. There is also evidence to indicate that proteins produced in the Golgi are routed to MVBs7–9, although this evidence remains limited. The limiting membrane of sMVBs can fuse with the plasma membrane, releasing the exosomal content into the extracellular space. In dMVBs, exosomes are thought to be formed by the fusion of endosomal vesicles with an endosomal compartment that subsequently fuses with a lysosome, leading to degradation.
To date, no protein markers have been identified that can discriminate between sMVBs and dMVBs, or indeed between exosomes and microvesicles10. Tetraspanins (CD63 and CD81) or the ESCRT proteins (BOX 1) ALG2-interacting protein X (ALIX; also known as PDCD6IP) and tumour susceptibility gene 101 protein (TSG101) are frequently used to identify exosomes; however, microvesicles also contain tetraspanins11. Moreover, there are examples of ESCRT-independent MVB formation4, and ESCRT-III takes part both in exosome and microvesicle formation3.
Cargoes
Cytosolic and membrane proteins, as well as RNA and lipids, can traverse across cells in EVs. However, it has been difficult to make generalizations about EV types based on their reported cargoes because of the heterogeneity of the methods used to purify EVs in proteomics studies1,12. Such heterogeneity in purification methods is likely to influence the type (or types) of EV populations selected for analysis. It has also been reported that EVs might contain mitochondrial DNA13, ribosomal RNA14 and genomic DNA15. However, it remains unclear whether these findings might have reflected contamination by dying cells, particularly as large vesicle size ranges were selected for analysis in these studies and therefore might have included apoptotic bodies (for example, see REFS 2,13). How an EV cargo becomes selected and whether the cargo has a function in the recipient cell is of great interest, but our knowledge of these matters is currently limited. There are now at least two public repositories where published information about EV cargoes is curated: Vesiclepedia (previously Exocarta) and EVpedia (see Further information).
A cautionary note
Most studies documenting EV function in the mammalian nervous system were performed in cell cultures, raising the significant challenge of corroborating the findings in vivo. Compounding this challenge are the experimental problems associated with the use of heterogeneous EV populations as a result of isolating these EVs from mixtures of different cell types. That is, each cell type probably releases a different EV population and target cells potentially respond to a restricted set of EVs carrying specific cargo1. In addition, published studies have used dissimilar criteria for designating EVs as microvesicles or exosomes; such criteria include the presence of specific markers and the size and shape of EVs, which are not necessarily specific, as alluded to above (see also REFS 10,16). Thus, it is not always possible to determine whether outcomes from different studies are comparable. Finally, much of the data that provide insight into the function of EVs in the intact brain originated from experiments involving the injection of concentrated EV preparations that were purified from the medium of cultured cells, and it is unclear whether the concentrations of EVs used in these experiments are in the same range as EV concentrations to which cells are exposed in intact organisms.
Although all of the above issues need to be considered when interpreting the findings of studies conducted so far on the neural function of EVs in mammals17, EVs are present in cerebrospinal fluid and blood in healthy mammalian organisms18–20, suggesting that they are part of normal physiological processes. Furthermore, convincing evidence for a functional role of EVs in the nervous system in vivo has been gained in invertebrate model systems, which exhibit developmental and physiological mechanisms that are highly conserved in nervous systems across various phyla.
EVs in the mammalian brain
EV function in neurons
Cultured neurons from embryonic and mature mammalian neural tissue release exosome-like EVs after stimulation with depolarization or alternative means to induce excitation, such as the application of Ca2+ ionophores, GABA receptor blockers, AMPA or NMDA21,22. These EVs, collected from culture media, are of typical exosomal size and shape, and contain EV markers such as ALIX and flotillin 2, as well as AMPA-type glutamate receptor subunits and the neuronal cell adhesion molecule L1. Corroboration of their MVB derivation has been obtained by pulse-chase experiments using a carboxy-terminal fragment of tetanus toxin heavy chain, which binds to the neuronal surface and is then internalized and accumulates in MVB ILVs. In such experiments, neuron stimulation resulted in the release of exosome-like EVs that contained tetanus toxin21. Although neuroblastoma cell-derived exosomes are reported to be predominantly taken up by glia, exosomes derived from stimulated dissociated cortical neurons were taken up by other neurons23.
It has been conjectured that the release of neurotransmitter receptors via exosomes may reflect a lysosomeindependent mechanism for disposing of such receptors for protein quality control or to regulate excitability22. A similar hypothesis has been proposed for other exosome cargoes, such as miRNAs24. Differentiated neuroblast cultures release a large number of miRNAs, which are initially localized to neurites upon cell depolarization and can then be recovered from the medium in RNA nuclease-resistant exosomes24. Exosomal release of a miRNA species is often associated with a decrease in the intracellular level of that miRNA, raising the possibility that exosome release is a means to adjust silencing levels; for example, during synaptic plasticity24.
Exosome release also serves to influence the physiological state of the recipient cell. Exosomes that are isolated from the medium of mouse primary cortical neuron cultures contain miR-124a, and these exosomes are internalized by astrocytes. This process results in the upregulation of excitatory amino acid transporter 2 (EAAT2; also known as GLT1), an important mediator of glutamate uptake in the brain25. These studies provide examples of transcellular communication through exosomes.
Oligodendrocyte communication via EVs
Oligodendrocytes, which are the glial cells that are responsible for myelinating axons in the brain, contain MVBs at periaxonal sites and release exosomes containing proteolipid protein (PLP), myelin proteins and proteins associated with protection against oxidative stress26–28. These MVBs appear to fuse with the plasma membrane and release exosomes upon glutamate release from neurons27. This process was elegantly demonstrated using a cell contact-free transwell device (Boyden chamber), in which mouse primary cortical neurons and oligodendrocytes could be cultured in different chambers separated by a filter membrane with 1-μm pores, thus preventing cell contact but allowing the exchange of metabolites and particles by diffusion. Potassium-induced depolarization of the neurons or stimulation of glutamatergic activity by treating the neurons with the GABAA receptor antagonist bicuculline enhanced the release of exosomes from the oligodendrocytes. Exosome release from oligodendrocytes was dependent on Ca2+ influx into the oligodendrocytes through NMDA-type and, to a lesser extent, AMPA-type glutamate receptors27. Accordingly, oligodendrocytes derived from an NMDA receptor NR1 subunit conditional knockout mouse were unable to increase exosome release upon neuronal depolarization27. Studies using the oligodendrocyte precursor cell line Oli-neu29 or mouse primary oligodendrocyte cultures27 have also shown that the release of exosomes by these cells is inhibited by reducing RAB35 activity29, potentially also implicating this small G protein in the regulation of exosome release by oligodendrocytes.
Oligodendrocyte exosomes can be taken up by neurons and microglia27. The retrieval of oligodendrocyte-derived exosomes by neurons was validated in the transwell co-culture system described above. Oligodendrocytes were labelled with a lipophilic dye that is released in association with exosomes, and the uptake of fluorescent particles by the cultured cortical neurons was observed. The neurons were also able to take up the labelled exosomes, which, as expected, contained PLP, from a purified exosomal fraction derived from the oligodendrocyte culture medium. To determine whether a functional exosomal cargo was retrieved by neurons, oligodendrocyte cultures were transduced with a construct containing CRE recombinase under the control of an oligodendrocyte-specific promoter. These oligodendrocytes were then co-cultured in the transwell system with mouse primary cortical neurons that had been previously transfected with a CRE activity reporter. The CRE reporter was activated in neurons, and this activation was prevented by inhibition of exosome secretion, through the application of a sphingomyelinase inhibitor or Rab35 knockdown27. The results of these experiments suggest that CRE is transferred from oligodendrocytes to neurons, probably through exosomes, and that this exosomal cargo is active in the receiving neuron. Moreover, when an exosome preparation isolated from the culture medium of CRE recombinase-transduced cultured mouse oligodendrocytes was injected into the cerebellum and hippocampus of mice carrying a CRE reporter, the exosomes activated the reporter in neurons in these brain regions27. This result provides solid evidence that exosomes are capable of transferring enzymatic activities to recipient cells. Exosome release by oligodendrocytes, and their internalization by neurons in the transwell set up described above, were associated with enhanced neuronal viability under conditions of cell stress (oxygen and glucose deprivation)28. Moreover, the application of oligodendrocyte exosomes to neurons induced changes in neuronal physiology, such as an increase in firing rate28.
Oligodendrocytes also secrete exosomes that inhibit oligodendrocyte differentiation and myelin formation30. This autoinhibition is partially blocked by incubating glial cells with conditioned neuronal medium, suggesting that this autoinhibitory mechanism serves to control premature myelin biogenesis in the absence of neurons30.
Oli-neu cells release exosomes containing PLP that can be recovered from the culture medium. When this exosome fraction, labelled with a lipophilic dye, was applied to primary cultures of mouse cortical neurons, microglia, astrocytes or oligodendrocytes, these exosomes were taken up by microglia but not by the other cell types31. The internalized exosomes colocalized with a lysosomal or late endosomal marker. This colocalization was also observed in resident microglial cells when the exosome fraction was injected into the spinal cord of transgenic mice expressing enhanced green fluorescent protein (GFP) in microglia31. The levels of PLP in lysates derived from the primary microglial cultures decreased over time, suggesting that microglia degrade exosome-derived PLP. Inhibition of factors and processes involved in micropinocytosis inhibited exosome uptake; this result suggests that the uptake of exosomes by microglia occurs through this mode of endocytosis31. The uptake of oligodendrocyte-derived exosomes by primary cultures of microglia does not activate the immune functions of microglia, as there was no increase in the secretion of several cytokines and chemokines by the microglia in response to inflammatory stimuli after the internalization of exosomes31. Thus, this microglial cell population seems to take part in the clearance of oligodendrocyte exosomal cargo.
Schwann cell EVs during development and regeneration
Schwann cells myelinate axons in the peripheral nervous system, and these cells de-differentiate and support nerve regeneration in response to nerve injury. Cultured de-differentiated Schwann cells release exosome-like EVs that contain the neurotrophin receptor p75NTR (REF. 32). These exosomes are internalized by axons, as shown in cell culture experiments in which exosomal preparations were applied to dorsal root ganglion neurons. Treatment of dorsal root ganglion explants with Schwann cell exosomes, but not with fibroblast-derived exosomes, augmented the axon regeneration rate, providing evidence that exosomes have cell-specific actions. Schwann cell-derived exosomes also effectively decreased RHOA activity in growth cones, causing axon elongation inhibition and promoting growth cone collapse32. Injection of Schwann cell-derived exosomes also resulted in their internalization by axons in vivo and in enhanced axon regeneration after sciatic nerve crush32. In addition, brain-derived neurotrophic factor-mediated stimulation of p75NTR in cultured sympathetic neurons or nerve growth factor-mediated stimulation of p75NTR in PC12 cells promoted the internalization and accumulation of this receptor in RAB11-positive endosomes and MVBs33. Upon neuronal depolarization, these MVBs fuse with the plasma membrane, releasing p75NTR-containing exosomes.
Microglia-derived EVs in inflammation and synaptic talk
Microglia, which are macrophages engaged in defence against infection and in tissue repair in the brain, shed microvesicle-like EVs upon ATP activation of P2X7 receptors at their surface34. These microvesicles contain the cytokine interleukin-1β (IL-1β) proprotein, its protein processing enzyme caspase 1 and P2X7 receptors. When microvesicles come into contact with high levels of extracellular ATP, which is released from astrocytes or injured tissue, microvesicle P2X7 receptors are activated, causing the caspase 1-mediated cleavage of IL-1β proprotein and the release of mature IL-1β from these vesicles34. Thus, the release of immature IL-1β in P2X7-containing microvesicles serves as a mechanism to survey the brain and cerebrospinal fluid for tissue damage. Upon encountering sites of injury, often propagated by astrocytes, these microvesicles convert immature IL-1β into a biologically active molecule that is a key initiator of the acute inflammatory response.
There is also evidence to suggest that microgliaderived microvesicles can regulate neuronal excitability. The application of microvesicles derived from the N9 microglia cell line or rat primary microglial cells to rat hippocampal neuron cultures, or the injection of such microvesicles directly into the visual cortex of rats, resulted in an increase in the probability of synaptic vesicle release by presynaptic terminals35. This enhancement was accompanied by a rapid stimulation of neuronal ceramide and sphingosine production and was blocked by pharmacological or genetic inhibition of sphingosine synthesis in neurons35,36 (BOX 2). These findings are consistent with previous evidence showing that sphingosine metabolism regulates synaptic vesicle exocytosis by facilitating SNARE (soluble-N-ethylmaleimide sensitive factor attachment protein receptor) complex assemby37. However, the active component in microvesicles that causes increased synaptic vesicle release in neurons is not yet known.
Microglia-derived microvesicles have also been reported to enclose endocanabinoids38, known lipid regulators of synapse development and homeostasis. These microvesicles carry the endocannabinoid N-arachidonoylethanolamine on their surface, which is able to stimulate cannabinoid receptor type 1 (CB1) in target GABA neurons, suppressing spontaneous inhibitory transmission. This effect appears to be the result of stimulating CB1, as application of a selective CB1 antagonist prevents the suppression of spontaneous inhibitory transmission38. However, this action of microvesicles seems to be independent from the ability of such EVs to increase excitatory transmission35,38, as blocking CB1 did not prevent the increase in glutamatergic transmission elicited by microvesicles. Thus, microglia-derived microvesicles appear to regulate the excitation–inhibition balance.
Microglia also release exosomes that resemble those released by B lymphocytes and dendritic cells under basal conditions39, consistent with their role in the immune system. Microglia-derived exosomes contain MHC (major histocompatibility complex) class II receptors, chaperones, tetraspanins and membrane receptors, in addition to CD13 (REF. 39), an aminopeptidase that is responsible for the degradation of encephalin, the opioid receptor-binding neuropeptide. In addition, microglia-derived exosomes are released in response to WNT3A, which is internalized and in turn stimulates the release of WNT3A-containing exosomes by microglial cells40 (WNTs are signalling molecules implicated both in development and in several neurodegenerative conditions, including Alzheimer disease (AD)41). The exosomes also contain monocarboxylate transporter 1 (MCT1), which transports lactate. Indeed, by loading lactate into exosomes, these EVs function as an auxiliary source of energy for neurons during synaptic activity39.
Exosome release by microglia can also be influenced by neurotransmitters; microglia contain serotonin (5-HT) receptors, which can be blocked by 5-HT2 and 5-HT4 receptor antagonists42. Stimulation of 5-HT receptors results in the release of exosomes containing insulin-degrading enzyme, an enzyme that can degrade amyloid-β, a neurotoxic peptide linked to AD (see below). This result might explain the observation that high levels of 5-HT are associated with a reduction in amyloid-β levels in mouse models of this disease43.
Astrocyte EVs
Astrocytes are involved in controlling ionic balance, constitute the brain–blood barrier (BBB), provide trophic support for neurons and participate in repair after injury. Similar to microglia, astrocytes release IL-1β in exosomes and microvesicles in response to ATP binding to P2X7 receptors44. This process depends on the rapid activation of acid sphingomyelinase (a-SMase) (BOX 2) and results in the movement of this enzyme to the outer leaflet of the plasma membrane. Indeed, ATP-induced microvesicle shedding and IL-1β release are markedly reduced by inhibition of a-SMase, and completely blocked in glial cultures derived from a-SMase knockout mice44.
Astrocyte-enriched cultures also release synapsin 1 into the medium45. Although synapsin 1 has been classically associated with synaptic vesicles, it is an oligomannose-binding protein that interacts with neural cell adhesion molecule 1 (NCAM1) in an oligomannose-dependent manner, thus promoting neurite outgrowth45.
Taken together, the findings described above indicate that both neurons and glia release EVs, and that these EVs are engaged in the modulation of neuronal electrical activity, repair and protection from cellular stress.
EVs in the invertebrate nervous system
Cells in the nervous systems of Caenorhabditis elegans and Drosophila melanogaster also release EVs. The ease of genetic manipulation, the availability of tools that enable effective regulation of gene and transgene expression and the relative simplicity of the nervous systems in these organisms have facilitated the study of EV function in cellular communication in vivo.
EVs at the fly larval neuromuscular junction
The D. melanogaster larval neuromuscular junction (NMJ) has served as a powerful model to dissect genetic mechanisms of synaptic assembly, development and plasticity. In this system, the role of EVs in a physiological process has been demonstrated through a combination of studies in the whole organism and cell culture studies that were subsequently validated in vivo46–48. An attribute of the larval body wall muscle preparation is the dynamic nature of the NMJ, which continuously expands in line with muscle cell growth. Thus, this preparation is particularly useful for studying the genetic mechanisms of synaptic growth.
Studies of the release and transport of Wingless (Wg), the fly orthologue of mouse WNT1, which is secreted by neurons and glia49,50, and mediates coordinated pre- and postsynaptic expansion during growth, have suggested possible functions for exosomes in vivo at the larval NMJ. The Wg receptor DFrizzled-2 (DFz2) is localized both in motor neuron terminals and in postsynaptic muscle sites that are apposed to these terminals49. In particular, DFz2 is concentrated at extrasynaptic regions in the convoluted folding of the muscle junctional region, the subsynaptic reticulum (SSR) (FIG. 2a). Owing to hydrophobic post-translational modifications, Wg does not readily diffuse in the extracellular space. Instead, Wg is released in association with exosomes through its binding to the multipass transmembrane protein Evi (also known as Wntless)47, which is present in MVBs48,51. Through this shuttling mechanism, Wg can reach DFz2 receptors localized at distant regions within the SSR. Consistent with this idea, WNT proteins have also been found to be released in association with exosomes by human cells and during D. melanogaster development52.
MVBs are localized at presynaptic boutons of the NMJ48 (FIG. 2b) and they are also observed to dock at the bouton surface membrane at extrasynaptic bouton areas (FIG. 2c). Evidence for transfer of Evi–Wg from synaptic boutons to muscles in vivo was obtained by selectively expressing Evi–GFP in motor neurons, which led to the appearance of Evi in the SSR in muscle tissue47,48 (FIG. 2d). Similarly, downregulating Evi in neurons led to a decrease in endogenous Evi levels in muscles, suggesting that a postsynaptic Evi protein pool is derived from Evi release by presynaptic boutons. The decrease in Evi levels suppressed Wg release and resulted in stunted NMJs, which exhibited fewer numbers of synaptic boutons than NMJs from wild-type controls and contained undifferentiated boutons47.
Similar to the larval NMJ, cultured insect Schneider 2 (S2) cells can secrete endogenous or epitope-tagged Evi-containing EVs that are internalized by naive S2 cells. Proteomics analysis of these EVs suggest that they harbour endosomal markers that are typically associated with exosomes48.To understand the molecular mechanisms by which Evi exosomes are released into the extracellular space, the following strategy was utilized48. First, the above S2 cell culture assay was used as a primary screen to search for genes that, when downregulated, prevented the release of Evi-containing exosomes. Second, the identified genes from the primary screen were then downregulated in motor neurons in vivo to validate the model that the genes required for exosome release in S2 cells are also required for motor neuron exosome release in vivo at the NMJ.
Downregulating Rab11 or syntaxin 1A (Syx1A) inhibited exosome release, which was validated in vivo by RNAi-mediated silencing of Rab11 or Syx1A, or expression of a dominant-negative variant of Rab11 in neurons. Further evidence for the requirement of Syx1A in Evi-containing exosome release was obtained by expressing a hyperactive Syx1A mutant variant in the motor neurons of larvae, which enhanced Evi release48. Rab11 is present in recycling endosomes, but its localization has been linked to MVBs and exosome release in haematopoietic cells53,54. Syx1A is also part of the SNARE fusion machinery that is required for exocytosis of synaptic vesicles, indicating that MVB fusion with the plasma membrane might share some of the components required for neurotransmitter release. Rab11 might serve to tether MVBs at extrasynaptic regions, whereas Rab3a might direct synaptic vesicles to active zones, the sites of neurotransmitter release.
Another significant aspect of Evi–Wg neuronal release via exosomes is their activity dependency. Evi-containing exosome secretion is inhibited by lowering extracellular Ca2+ levels and it is enhanced by a spaced stimulation paradigm (repeated bouts of excitation separated by a period of rest)55. Thus, electrical activity is one of the regulatory mechanisms controlling the release of neuronal EVs that could link an EV mechanism of transcellular communication in the nervous system to neuronal function.
An important retrograde signal at the D. melanogaster embryonic NMJ is the release of the vesicle protein synaptotagmin 4 (Syt4) from muscles to stimulate synaptic plasticity in presynaptic terminals56. Highfrequency stimulation of motor neurons results in a marked increase in the frequency of spontaneous synaptic release events in a postsynaptic Ca2+-, endocytosis- and Syt4-dependent manner56. Similarly, at the larval NMJ, the rapid formation of new presynaptic boutons can be induced by spaced patterns of activity in a manner that depends on postsynaptic activity and postsynaptic Syt4 (REF. 46). Notably, postsynaptic Syt4 is derived from its exosomal release by motor neurons and its internalization by muscle46. These studies suggest that, at the NMJ, presynaptic boutons are capable of releasing exosomes that are taken up by muscle cells, and that these exosomes have a physiological function in synaptic plasticity in the recipient muscle cell in vivo.
Although WNTs have emerged as prominent factors in the regulation of synaptic development and plasticity in the vertebrate brain57, functioning both as anterograde and retrograde regulators, their release via exosomes in the mammalian brain has been less explored. Nevertheless, as discussed above, WNT3A can be internalized and secreted via exosomes by mammalian microglia40, and EV-mediated release of WNTs has been reported both during vertebrate development and cancer progression58. Peripheral glia at the D. melanogaster NMJ, similar to motor neurons, release Wg, and glia-derived Wg has a physiological role, which is partially distinct from the function of motor neuronderived Wg50. Although the release of Wg by motor neurons or glia regulates synaptic growth and differentiation of the NMJ, glia-derived Wg also regulates the localization of glutamate receptors at postsynaptic sites of the NMJ. There are additional common properties between fly and mammalian exosomes, such as their protein content (determined by proteomics studies of exosomes released by cultured S2 cells)48, the requirement of Rab11 for exosome release48 and the activity-dependent release of exosomes by neurons55. Thus, it is highly likely that communication of WNTs and other signals via EVs will be conserved in nervous systems throughout phyla, as is the case in most fundamental biological processes.
EVs in C. elegans sensory neurons
C. elegans type B male-specific ciliated sensory neurons expose their cilia to the environment through a cuticular pore59. These cilia shed microvesicle-like EVs containing the polycystin receptors, location of vulva defective 1 (LOV-1) and pyruvate dehydrogenase kinase 2 (PDK-2). Microvesicle release depends on intraflagellar transport protein 88 (IFT-88) and a kinesin-3, KLP-66 (REF. 59). When microvesicles isolated from the rearing medium of C. elegans were added to the rearing medium of naive animals, increased locomotor reversal frequency and tail chasing behaviour, a strategy used during mating, were observed59. This result suggests that microvesicles are involved in inter-organism communication59. A forward genetic screen to identify genes required for the ciliary localization of PKD-2 (REF. 60) led to the identification of CIL-7, a myristolated protein that regulates microvesicle biogenesis. Mutations in cil-7 were associated with mating defects and a block on PKD-2 release60.
Collectively, the above studies in invertebrate systems provide compelling evidence that EVs function in communication in vivo, providing proof of principle that these vesicles have physiological roles.
EVs in disease
In recent years, EVs have emerged as important participants in the spreading of misfolded proteins in neurodegenerative diseases, in tumorigenic activity in brain cancer and in neuroinflammation. Below, we provide a brief summary of some of the emerging themes connecting EVs to neurological disorders61,62, the mechanisms by which EVs participate in neuroprotection and the exciting new approaches for the potential use of EVs to deliver therapeutic agents.
EVs as prion carriers
The idea that neurodegenerationcausing agents may have a common mode of spreading in the brain, even before cell death, has become attractive. Supporting evidence has been provided in several disorders, although such evidence is still being debated in other diseases63. A common trait in many of the mutated genes that cause neurodegenerative diseases is that they lead to the misfolding and aggregation of their encoded proteins63. This misfolding and aggregation spreads from one or more focal points in the brain to contiguous neuroanatomical regions. The spreading of the affected protein is thought to be similar to the spread of prions, which are misfolded proteins that can induce the misfolded state in pre-existing normally folded proteins, and that can spread from cell to cell64. Polymerization of the misfolded protein results in the formation of stable protein aggregates. These aggregates are usually fibrillar structures (referred to as amyloid) and contain the misfolded protein in a β-sheet conformation; amyloid may subsequently cause tissue damage and cell death. Prion-like properties, such as those observed for prion protein (PrP) in Creutzfeldt–Jakob disease65, have been suggested for other proteins involved neurodegenerative disorders. Such proteins include amyloid-β and tau in AD66,67; α-synuclein in Parkinson disease68; and TAR DNA-binding protein 43 (TDP43), copper zinc superoxide dismutase 1 (SOD1) and other proteins in amyotrophic lateral sclerosis and frontotemporal lobal degeneration69. A role for EVs in this spreading capacity is beginning to emerge70, as these prion-like proteins are known to be packaged and released in exosome-like EVs. However, the interpretation of the available data is still challenging owing to substantial experimental issues, as only a minute quantity of prion-like protein is sufficient to seed protein unfolding and whether EV preparations were devoid of contaminating prion-like protein aggregates that were not contained within EVs is unclear (for example, see REF. 71). In addition, it is currently uncertain which protein species (folded, unfolded or aggregated) are packaged into EVs. Below, we provide some examples of the potential role of EVs in the propagation of unfolding.
In vitro experiments have provided evidence that mutant SOD1 induces misfolding of human wild-type SOD1, and that misfolded human wild-type SOD1 propagates misfolding across cultured HEK, neuroblastoma, mesenchymal and human neuron-like cells, even in the absence of mutant SOD1 (REFS 69,71). This propagation has been proposed to be mediated both via exosome-dependent and exosome-independent (aggregates released by dying cells and taken up via micropinocytosis) mechanisms. The evidence for EV-dependent spreading of mutant SOD1 or misfolded human wild-type SOD1 has been obtained in NSC-34 motor neuron-like cultures, utilizing reagents such as antibodies to specifically detect the misfolded but not the properly folded protein. These cells were able to release either mutant SOD1 or misfolded human wild-type SOD1 in exosome-like EVs, and these EVs were able to spread misfolding of human wild-type SOD1 through multiple cell passages71. Nevertheless, misfolded human wild-type SOD1 aggregates not contained within EVs, albeit at minimal levels, were also found in the EV fraction that was added to the cultures71. Therefore, it is plausible that the spread of misfolding might be caused via non-EV-dependent mechanisms.
α-synuclein has also been proposed to be secreted in exosomes in a Ca2+-dependent manner by SH-SY5Y neuron-like cells, spreading toxicity to the culture72. However, in these experiments, there are also questions about the specific fraction (or fractions) of α-synuclein (that is, α-synuclein within EVs, or as a soluble contaminant of the EV fraction, or as a contaminant in the form of small or large aggregates within the EV fraction) that conveys toxicity to the cells73.
Studies of the involvement of EVs in AD are particularly intriguing, although their involvement in disease remains unresolved. Amyloid-β is produced during cleavage of the β-amyloid precursor protein (APP) by the consecutive actions of the β- and γ-secretases. β-secretasemediated cleavage of APP occurs in early endosomes and amyloid-β is directed to MVBs. Fusion of MVBs with the plasma membrane liberates amyloid-β-containing exosomes; the majority of amyloid-β is found in the medium in a soluble, non-EV-enclosed fraction74, and soluble rather than fibrillar amyloid-β is the most toxic to neurons and glia75. In addition to amyloid-β, several C-terminal APP fragments and full-length APP, but not amino-terminal APP fragments, have been found in exosomes from cultured neuroblastoma N2a cells76.
Because amyloid plaques contain exosomal markers, such as ALIX, it has been suggested that exosomes might participate in the pathogenesis of AD74. However, this concept and the potential role of exosomes in spreading and inducing amyloid-β aggregation in a prion-like manner have not been convincingly established77. For example, it has been reported that exosomes derived from a neuroblastoma cell line or from cultured mouse primary cortical neurons induce the aggregation of soluble amyloid-β and bind to these amyloid-β aggregates78. When the exosomes that were bound to fibrillar amyloid-β were then added to primary microglia cultures, they were taken up by microglia and degraded. This process resulted in a decrease in toxic soluble amyloid-β in the medium, suggesting that exosomes might represent a protective pathway for the removal of excess soluble amyloid-β78 (see REF. 77 for a review). Other studies have shown that reactive microglia release microvesicles that bind and promote amyloid-β pathogenesis by increasing its solubility75, raising the possibility that exosomes and microvesicles might have antagonistic functions. Nevertheless, another study examining the mechanisms of tau propagation in the brain provided evidence that spreading of tau is accomplished through the release of exosomes by microglia, and that blocking exosome synthesis prevents such spreading79. Thus, the interplay of EV types, their cellular derivation and their role in the context of AD pathology remain unclear.
EVs in brain tumours
Cancer cells show enhanced EV release, and released EVs often contain an altered cargo composition compared with their normal counterparts80. This modified cargo can include increased amounts of cargo normally present in EVs derived from wild-type cells and transforming proteins and RNA, including tumour suppressors, epidermal growth factor variant III (EGFRvIII), histones and miRNAs, normally not present in EVs derived from wild-type cells. The uptake of EVs that have been released by cancer cells can result in the transfer of oncogenic activity61. A prominent example is the case of glioblastomas, which are the most common and aggressive forms of primary brain cancers. Glioblastomas are composed of a heterogeneous population of cells, including a hierarchy of stem cells that may contribute to the initiation and propagation of tumours through clonal expansions. It has been postulated that the diversification of cell types and their phenotypes (for example, proliferation rate, survival rate and motility rate) might be guided by the uptake of EVs containing diverse cargoes61. For instance, EGFRvIII is an oncogenic receptor tyrosine kinase that usually accumulates in plasma membrane lipid rafts. Cell culture studies have shown that EGFRvIII is released into the culture medium in flotillin 1-containing microvesicles80. Uptake of these EVs by naive cells leads to the incorporation of the oncogenic receptor into the plasma membrane and activation of transforming signalling pathways, such as the ERK1–ERK2 and AKT pathways80,81. Furthermore, when EGFRvIII-containing glioma cells are injected into immunodeficient mice, these cells form subcutaneous tumours, which release EGFRvIII-containing EVs into the bloodstream and neighbouring cells. Thus, EVs are likely to contribute to the propagation of oncogenes.
Notably, glioblastoma cells also release EVs containing the tumour suppressor miR-1 (REF. 82). Among other targets, miR-1 silences annexin A2 mRNA, whose protein product is a major pro-oncogenic protein released in EVs, thus ameliorating the pro-oncogenic effect of glioblastoma EVs82. In addition to glioblastomas, brain metastasis is also a major mortality factor in breast cancer83. Central to this brain metastasis is the breach of the BBB. This breach is promoted by the delivery of miR-181c via cancer cell-derived EVs, as shown with EVs derived from D3H2LN cells, which are human mammary tumour cells with a high tumorigenic and metastatic capacity. miR-181c promotes the destruction of the BBB by downregulating its target mRNA, PDPK1 (3-phosphoinositide-dependent protein kinase 1), whose protein product is required for actin organization and thus for proper tight junction function in establishing the BBB83.
EVs as carriers of neuroinflammatory signals
EVs derived from both microglia and astrocytes have an active role in neuroinflammation, in part by spreading proinflammatory signals; for example, during multiple sclerosis (MS), a demyelinating disease84,85. Such microvesicles contain pro-inflammatory cytokine IL-1β, interferon-γ, tumour necrosis factor (TNF), caspase 1 and the P2X7 receptor, among others. They also contain metalloproteinases, which, together with TNF and IL-1β, can disrupt the BBB by promoting the degradation of the extracellular matrix and tight and adherens junctions, enhance nitric oxide expression, and enable brain infiltration by leukocytes and T cells86,87. Astrocytes that form part of the BBB become highly activated in response to neuronal injury. Akin to microglia, they release EVs containing IL-1β, which exacerbates tissue deterioration, thus promoting DNA damage and apoptosis88.
Oligodendrocytes activate microglia in patients with MS, and microglia propagate the inflammatory response89. Consistent with the role of microglia-derived microvesicles in propagating inflammatory responses, patients with acute MS have increased numbers of microvesicles in blood and cerebrospinal fluid. In addition, the injection of microglia-derived microvesicles into the brains of mice with subclinical experimental autoimmune encephalomyelitis (a model of MS) led to the recruitment of inflammatory cells to the injection site85,90. Microglia-derived microvesicles also affect sphingosine metabolism (BOX 2) in responsive cells. Indeed, a-SMase knockout mice, in which the production of microvesicles is impaired, are significantly protected from experimental autoimmune encephalomyelitis, and cultured astrocytes or microglia from mice with pharmacological or genetic ablation of a-SMase have a diminished release of IL-1β upon P2X7 receptor stimulation44,85.
Toll-like receptors (TLRs), a family of receptors that initiate inflammatory responses, are also present in the nervous system, including in neurons, oligodendrocytes, astrocytes and microglia91. In the CNS, TLRs are activated by signals that are released from injured or stressed cells, such as pro-inflammatory factors and heat shock proteins (HSPs), leading to nuclear factor-κB activation and changes in gene expression that lead to tissue damage. Notably, TLRs have also been associated with the pathology of amyotrophic lateral sclerosis, AD and Parkinson disease92. It has been suggested that the release of EVs bearing altered miRNA content in these diseases, particularly those belonging to a class of mediators of inflammation, results in the activation of TLRs, setting in motion an inflammatory cascade91.
EV function in neuroprotection
Although EVs might contribute significantly to the spreading of certain neurological conditions, they also have neuroprotective actions. They serve as delivery vehicles for factors involved in repair after stress, disease and injury, and they can become a sink for the removal of unwanted or toxic material. In the context of prion-like proteins, protein aggregates that are resistant to the cell’s degradation machinery are released to the medium in EVs. Although this process may serve to spread disease (possibly similar to an infective mechanism), it might also function in mitigating the toxic effects of protein aggregates within the cell by disposing of this material93. A case in point, as discussed earlier, regards the ability of exosome-like EVs to trap94 and to promote amyloid-β removal by microglia78. In this regard, although microglia are involved in propagating inflammation, they are also engaged in neuroprotection95.
There is also evidence to suggest that EVs participate in protection from oxidative stress26. Oligodendrocyte-derived EVs contain many enzymes involved in protecting against oxidative stress, such as SOD1, catalase and peroxiredoxin26,28. Consequently, neurons taking up these exosome-like EVs have increased oxidative stress tolerance and exhibit improved survival upon nutrient deprivation27,28. In in vitro models of cerebral ischaemia, in which neurons are deprived of glucose and oxygen, oligodendrocyte-derived EVs ameliorate neuronal apoptosis and necrosis, and neurons exhibit increased metabolic activity28. In addition to providing antioxidative enzymes, the increased metabolic activity of neurons is partly the consequence of differential gene expression and activation of survival-promoting ERK1–ERK2 and AKT transduction cascades, among others, after exosome uptake27,28.
EVs also contain HSPs, such as HSP70. Although HSPs were originally thought to operate exclusively within the cells that synthesize them, it is now evident that HSPs are also released in EVs and taken up by cells. It has been suggested that released HSPs, in addition to their role as chaperones, serve as warning signals to prevent the propagation of cellular stress96. The transfer of HSP70 from glia to axons was first discovered in the giant axon of squid in response to thermal shock97. Subsequently, the release of HSPs has been documented for a variety of cells and tissues. For example, astrocytes subjected to hyperthermia or exposed to prion-like proteins increase the release of HSP70 via EVs98.
Exosome-like EVs also appear to bear age-related properties. Young mice carry EVs enriched in miR-219 (REF. 99), an miRNA that promotes the differentiation of myelinating oligodendrocytes100,101. Treatment of demyelinated rat hippocampal slices with EVs derived from the serum of young animals improved remyelination and enhanced myelination in aged rats99.
It has also been suggested that synapsin 1, which as described in previous sections is an oligomannose-containing protein released in EVs by astrocyte-enriched cultures, might have neuroprotective functions under conditions of oxidative stress, ischaemia and high depolarization levels45. Collectively, these examples demonstrate the vast potential of the content of EVs in the functional recovery after spinal injuries, stroke or MS.
EVs as shuttles for therapeutic intervention
Several features place EVs, and in particular exosomes, as potentially powerful shuttles for the delivery of therapeutic agents to the brain102. EVs also constitute important and non-invasive biomarkers for the diagnosis and the progression of disease, as they are found in all bodily fluids1. Exosomes are thought to cross the BBB103. They are stable, have low immunogenicity, can be loaded with therapeutic silencing RNAs, miRNAs and drugs12,104, and might have endogenous mechanisms that target them to specific cell types. Exosomes from adult stem cells are particularly promising in providing a strategy for delivering therapeutic agents into the brain via EVs. For example, mesenchymal stromal cells (MSCs), self-renewing multipotent progenitors that can be isolated from a variety of tissues for cell culture, produce substantial amounts of exosomelike EVs with different characteristics, particularly when stimulated with different factors105,106. Endogenous MSCs derived from rat brain regions subjected to middle cerebral artery occlusion represent a case in point. These MSCs secrete exosomes containing miR-133b, and these exosomes can increase plasticity and neurite remodelling in the ischaemic boundary107. Other examples are EVs derived from human adipose tissue MSCs, which carry the amyloid-β-degrading enzyme neprilysin and are capable of decreasing both secreted and intracellular amyloid-β in cell culture, providing a potential avenue to treat AD108. In addition, exosomes from interferon-γ-stimulated dendritic cells can increase myelination and tolerance to oxidative stress109. These exosomes contain miR-219, which promotes oligodendrocyte differentiation and myelination100,101. Macrophage exosomes loaded with the antioxidant catalase have also been used to treat mouse models of Parkinson disease via intranasal application, and this treatment provided considerable neuroprotection110. In addition, self-derived dendritic cell exosomes (exosomes derived from dendritic cells of one individual that are modified and used to treat that individual) may be an approach to treat AD. When such exosomes were loaded with exogenous smallinterfering RNAs directed against BACE1 (which encodes β-secretase 1 and is a therapeutic target for AD) and were injected into wild-type mice, an effective downregulation of BACE1 was observed (REF. 104).
However, despite the many promising features of EVs, there are still many hurdles to overcome before their use as therapeutic interventions can be realized. First, we are still in the very early stages of understanding EV biology. Deciphering the mechanisms that trigger EV release, promote EV uptake, provide cell target specificity and determine cargo selectivity is critical to the development of EVs as therapeutic agents in a clinical setting. Second, given that evidence suggests that exosomes also have the potential to spread nefarious factors, such as prion-like proteins110 and pathological inflammation84,85, as discussed above, as well as viruses111, many safety concerns for their use in the clinic remain. Although the idea of generating artificial EVs is attractive, a more thorough understanding of EV biology will be paramount to their construction and use as a Trojan horse to combat disease.
Conclusions and perspectives
Taken together, the studies briefly discussed in this Review make a promising case that EVs are used by animals with nervous systems as intercellular communication devices, both in health and during disease. Although much needs to be done to truly understand the biological context and the physiological range in which EVs are utilized, the available examples of EV function in vivo in invertebrate systems makes it likely that this will be an expanding field. In the same way that pioneering studies in invertebrate genetic model organisms have paved the way for our understanding of conserved mechanisms (such as pattern formation, cell death and RNA-mediated gene silencing), the contribution of invertebrate model organisms to fully grasping the significance of EVs in the nervous system is likely to be forthcoming.
In addition to providing novel mechanisms of intercellular communication in the healthy organism, the study of EVs during disease is also providing important clues to measure the progression of neurodegenerative diseases and cancer. This knowledge is cultivating a fertile ground for the design of clinical interventions to alleviate disease.
However, many outstanding questions relating to EV biology remain. For example, it is unclear whether sMVBs are produced in the neurons or glia, how EV cargo is selected and how many EV populations are derived from the same cell. Moreover, it is unclear what the molecular mechanisms that define MVB fusion sites in the plasma membrane are, and what the constituents of regulated or constitutive MVB plasma membrane fusion are. It will also be crucial to know whether there are signatures that target EVs to disparate targets, how recipient cells discriminate among EVs, what the mechanisms of exosome and microvesicle uptake by recipient cells are, and, indeed, how EV cargoes are unloaded and utilized by such cells. Other questions to be answered include how EVs cross the BBB in healthy organisms and how EVs escape the immune response. Finally, whether EVs function in a synapse-specific manner during plasticity and what plasticity signals they carry remain to be answered.
Acknowledgements
V.B. is supported by a US National Institutes of Health grant (R37 MH070000).
Footnotes
Competing interests statement
The authors declare no competing interests
References
- 1.Colombo M, Raposo G, Thery C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu. Rev. Cell Dev. Biol. 2014;30:255–289. doi: 10.1146/annurev-cellbio-101512-122326. An important review of the current understanding of EV biogenesis and secretion, including protocols for EV isolation and current challenges for the field.
- 2.Crescitelli R, et al. Distinct RNA profiles in subpopulations of extracellular vesicles: apoptotic bodies, microvesicles and exosomes. J. Extracell. Vesicles. 2013 doi: 10.3402/jev.v2i0.20677. http://dx.doi.org/10.3402/jev.v2i0.20677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hurley JH. ESCRTs are everywhere. EMBO J. 2015;34:2398–2407. doi: 10.15252/embj.201592484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Babst M. MVB vesicle formation: ESCRT-dependent, ESCRT-independent and everything in between. Curr. Opin. Cell Biol. 2011;23:452–457. doi: 10.1016/j.ceb.2011.04.008. An excellent discussion on the role of the ESCRT versus lipids in forming MVB ILVs.
- 5.Lorent JH, Levental I. Structural determinants of protein partitioning into ordered membrane domains and lipid rafts. Chem. Phys. Lipids. 2015;192:23–32. doi: 10.1016/j.chemphyslip.2015.07.022. [DOI] [PubMed] [Google Scholar]
- 6.Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J. Cell Biol. 2013;200:373–383. doi: 10.1083/jcb.201211138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Friend DS. Cytochemical staining of multivesicular body and Golgi vesicles. J. Cell Biol. 1969;41:269–279. doi: 10.1083/jcb.41.1.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nilsson P, et al. Autophagy-related protein 7 deficiency in amyloid β (Aβ) precursor protein transgenic mice decreases Aβ in the multivesicular bodies and induces Aβ accumulation in the Golgi. Am. J. Pathol. 2015;185:305–313. doi: 10.1016/j.ajpath.2014.10.011. [DOI] [PubMed] [Google Scholar]
- 9.Kolesnikova L, Berghofer B, Bamberg S, Becker S. Multivesicular bodies as a platform for formation of the Marburg virus envelope. J. Virol. 2004;78:12277–12287. doi: 10.1128/JVI.78.22.12277-12287.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gould SJ, Raposo G. As we wait: coping with an imperfect nomenclature for extracellular vesicles. J. Extracell. Vesicles. 2013 doi: 10.3402/jev.v2i0.20389. http://dx.doi.org/10.3402/jev.v2i0.20389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Andreu Z, Yanez-Mo M. Tetraspanins in extracellular vesicle formation and function. Front. Immunol. 2014;5:442. doi: 10.3389/fimmu.2014.00442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Batrakova EV, Kim MS. Using exosomes, naturally-equipped nanocarriers, for drug delivery. J. Control. Release. 2015;219:396–405. doi: 10.1016/j.jconrel.2015.07.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Guescini M, Genedani S, Stocchi V, Agnati LF. Astrocytes and glioblastoma cells release exosomes carrying mtDNA. J. Neural Transm. 2010;117:1–4. doi: 10.1007/s00702-009-0288-8. [DOI] [PubMed] [Google Scholar]
- 14.Miranda KC, et al. Nucleic acids within urinary exosomes/microvesicles are potential biomarkers for renal disease. Kidney Int. 2010;78:191–199. doi: 10.1038/ki.2010.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cai J, et al. Extracellular vesicle-mediated transfer of donor genomic DNA to recipient cells is a novel mechanism for genetic influence between cells. J. Mol. Cell. Biol. 2013;5:227–238. doi: 10.1093/jmcb/mjt011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lotvall J, et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J. Extracell. Vesicles. 2014;3:26913. doi: 10.3402/jev.v3.26913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gangoda L, Boukouris S, Liem M, Kalra H, Mathivanan S. Extracellular vesicles including exosomes are mediators of signal transduction: are they protective or pathogenic? Proteomics. 2015;15:260–271. doi: 10.1002/pmic.201400234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tietje A, Maron KN, Wei Y, Feliciano DM. Cerebrospinal fluid extracellular vesicles undergo age dependent declines and contain known and novel noncoding RNAs. PLoS ONE. 2014;9:e113116. doi: 10.1371/journal.pone.0113116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chiasserini D, et al. Proteomic analysis of cerebrospinal fluid extracellular vesicles: a comprehensive dataset. J. Proteom. 2014;106:191–204. doi: 10.1016/j.jprot.2014.04.028. [DOI] [PubMed] [Google Scholar]
- 20.Marzesco AM, et al. Release of extracellular membrane particles carrying the stem cell marker prominin-1 (CD133) from neural progenitors and other epithelial cells. J. Cell Sci. 2005;118:2849–2858. doi: 10.1242/jcs.02439. [DOI] [PubMed] [Google Scholar]
- 21.Lachenal G, et al. Release of exosomes from differentiated neurons and its regulation by synaptic glutamatergic activity. Mol. Cell. Neurosci. 2011;46:409–418. doi: 10.1016/j.mcn.2010.11.004. [DOI] [PubMed] [Google Scholar]
- 22.Faure J, et al. Exosomes are released by cultured cortical neurones. Mol. Cell. Neurosci. 2006;31:642–648. doi: 10.1016/j.mcn.2005.12.003. A seminal study showing that primary cultured neurons secrete EVs in response to depolarization.
- 23.Chivet M, et al. Exosomes secreted by cortical neurons upon glutamatergic synapse activation specifically interact with neurons. J. Extracell. Vesicles. 2014;3:24722. doi: 10.3402/jev.v3.24722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Goldie BJ, et al. Activity-associated miRNA are packaged in Map1b-enriched exosomes released from depolarized neurons. Nucleic Acids Res. 2014;42:9195–9208. doi: 10.1093/nar/gku594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Morel L, et al. Neuronal exosomal miRNA-dependent translational regulation of astroglial glutamate transporter GLT1. J. Biol. Chem. 2013;288:7105–7116. doi: 10.1074/jbc.M112.410944. A fine study documenting a potentially physiological action of an miRNA when transferred from neurons to astrocytes.
- 26.Kramer-Albers EM, et al. Oligodendrocytes secrete exosomes containing major myelin and stress-protective proteins: trophic support for axons? Proteom. Clin. Appl. 2007;1:1446–1461. doi: 10.1002/prca.200700522. [DOI] [PubMed] [Google Scholar]
- 27.Fruhbeis C, et al. Neurotransmitter-triggered transfer of exosomes mediates oligodendrocyte–neuron communication. PLoS Biol. 2013;11:e1001604. doi: 10.1371/journal.pbio.1001604. An elegant study documenting a potential role of EVs in glia–neuron communication in the mammalian brain.
- 28.Frohlich D, et al. Multifaceted effects of oligodendroglial exosomes on neurons: impact on neuronal firing rate, signal transduction and gene regulation. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014;369:20130510. doi: 10.1098/rstb.2013.0510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hsu C, et al. Regulation of exosome secretion by Rab35 and its GTPase-activating proteins TBC1D10A-C. J. Cell Biol. 2010;189:223–232. doi: 10.1083/jcb.200911018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bakhti M, Winter C, Simons M. Inhibition of myelin membrane sheath formation by oligodendrocyte-derived exosome-like vesicles. J. Biol. Chem. 2011;286:787–796. doi: 10.1074/jbc.M110.190009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fitzner D, et al. Selective transfer of exosomes from oligodendrocytes to microglia by macropinocytosis. J. Cell Sci. 2011;124:447–458. doi: 10.1242/jcs.074088. [DOI] [PubMed] [Google Scholar]
- 32.Lopez-Verrilli MA, Picou F, Court FA. Schwann cell-derived exosomes enhance axonal regeneration in the peripheral nervous system. Glia. 2013;61:1795–1806. doi: 10.1002/glia.22558. An important study documenting the role of EVs liberated from Schwann cells during axonal regeneration.
- 33.Escudero CA, et al. The p75 neurotrophin receptor evades the endolysosomal route in neuronal cells, favouring multivesicular bodies specialised for exosomal release. J. Cell Sci. 2014;127:1966–1979. doi: 10.1242/jcs.141754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bianco F, et al. Astrocyte-derived ATP induces vesicle shedding and IL-1β release from microglia. J. Immunol. 2005;174:7268–7277. doi: 10.4049/jimmunol.174.11.7268. [DOI] [PubMed] [Google Scholar]
- 35.Antonucci F, et al. Microvesicles released from microglia stimulate synaptic activity via enhanced sphingolipid metabolism. EMBO J. 2012;31:1231–1240. doi: 10.1038/emboj.2011.489. An intriguing study showing that microgliaderived microvesicles might influence synaptic activity in mammals.
- 36.Turola E, Furlan R, Bianco F, Matteoli M, Verderio C. Microglial microvesicle secretion and intercellular signaling. Front. Physiol. 2012;3:149. doi: 10.3389/fphys.2012.00149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Darios F, et al. Sphingosine facilitates SNARE complex assembly and activates synaptic vesicle exocytosis. Neuron. 2009;62:683–694. doi: 10.1016/j.neuron.2009.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gabrielli M, et al. Active endocannabinoids are secreted on extracellular membrane vesicles. EMBO Rep. 2015;16:213–220. doi: 10.15252/embr.201439668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Potolicchio I, et al. Proteomic analysis of microgliaderived exosomes: metabolic role of the aminopeptidase CD13 in neuropeptide catabolism. J. Immunol. 2005;175:2237–2243. doi: 10.4049/jimmunol.175.4.2237. [DOI] [PubMed] [Google Scholar]
- 40.Hooper C, et al. Wnt3a induces exosome secretion from primary cultured rat microglia. BMC Neurosci. 2012;13:144. doi: 10.1186/1471-2202-13-144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Freese JL, Pino D, Pleasure SJ. Wnt signaling in development and disease. Neurobiol. Dis. 2010;38:148–153. doi: 10.1016/j.nbd.2009.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Glebov K, et al. Serotonin stimulates secretion of exosomes from microglia cells. Glia. 2015;63:626–634. doi: 10.1002/glia.22772. [DOI] [PubMed] [Google Scholar]
- 43.Cirrito JR, et al. Serotonin signaling is associated with lower amyloid-β levels and plaques in transgenic mice and humans. Proc. Natl Acad. Sci. USA. 2011;108:14968–14973. doi: 10.1073/pnas.1107411108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bianco F, et al. Acid sphingomyelinase activity triggers microparticle release from glial cells. EMBO J. 2009;28:1043–1054. doi: 10.1038/emboj.2009.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wang S, et al. Synapsin I is an oligomannose-carrying glycoprotein, acts as an oligomannose-binding lectin, and promotes neurite outgrowth and neuronal survival when released via glia-derived exosomes. J. Neurosci. 2011;31:7275–7290. doi: 10.1523/JNEUROSCI.6476-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Korkut C, et al. Regulation of postsynaptic retrograde signaling by presynaptic exosome release. Neuron. 2013;77:1039–1046. doi: 10.1016/j.neuron.2013.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Korkut C, et al. Trans-synaptic transmission of vesicular Wnt signals through Evi/Wntless. Cell. 2009;139:393–404. doi: 10.1016/j.cell.2009.07.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Koles K, et al. Mechanism of evenness interrupted (evi)-exosome release at synaptic boutons. J. Biol. Chem. 2012;287:16820–16834. doi: 10.1074/jbc.M112.342667. Together with reference 47, this study provides in vivo evidence for the role of EVs in trans-synaptic communication of WNT signals.
- 49.Packard M, et al. The Drosophila wnt, wingless, provides an essential signal for pre- and postsynaptic differentiation. Cell. 2002;111:319–330. doi: 10.1016/s0092-8674(02)01047-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kerr KS, et al. Glial wingless/Wnt regulates glutamate receptor clustering and synaptic physiology at the Drosophila neuromuscular junction. J. Neurosci. 2014;34:2910–2920. doi: 10.1523/JNEUROSCI.3714-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Franch-Marro X, et al. Wingless secretion requires endosome-to-Golgi retrieval of Wntless/Evi/Sprinter by the retromer complex. Nat. Cell Biol. 2008;10:170–177. doi: 10.1038/ncb1678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gross JC, Chaudhary V, Bartscherer K, Boutros M. Active Wnt proteins are secreted on exosomes. Nat. Cell Biol. 2012;14:1036–1045. doi: 10.1038/ncb2574. [DOI] [PubMed] [Google Scholar]
- 53.Savina A, Vidal M, Colombo MI. The exosome pathway in K562 cells is regulated by Rab11. J. Cell Sci. 2002;115:2505–2515. doi: 10.1242/jcs.115.12.2505. [DOI] [PubMed] [Google Scholar]
- 54.Savina A, Fader CM, Damiani MT, Colombo MI. Rab11 promotes docking and fusion of multivesicular bodies in a calcium-dependent manner. Traffic. 2005;6:131–143. doi: 10.1111/j.1600-0854.2004.00257.x. [DOI] [PubMed] [Google Scholar]
- 55.Ataman B, et al. Rapid activity-dependent modifications in synaptic structure and function require bidirectional wnt signaling. Neuron. 2008;57:705–718. doi: 10.1016/j.neuron.2008.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yoshihara M, Adolfsen B, Galle KT, Littleton JT. Retrograde signaling by Syt 4 induces presynaptic release and synapse-specific growth. Science. 2005;310:858–863. doi: 10.1126/science.1117541. [DOI] [PubMed] [Google Scholar]
- 57.Budnik V, Salinas PC. Wnt signaling during synaptic development and plasticity. Curr. Opin. Neurobiol. 2011;21:151–159. doi: 10.1016/j.conb.2010.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhang L, Wrana JL. The emerging role of exosomes in Wnt secretion and transport. Curr. Opin. Genet. Dev. 2014;27:14–19. doi: 10.1016/j.gde.2014.03.006. [DOI] [PubMed] [Google Scholar]
- 59.Wang J, et al. C. elegans ciliated sensory neurons release extracellular vesicles that function in animal communication. Curr. Biol. 2014;24:519–525. doi: 10.1016/j.cub.2014.01.002. A pioneering study documenting the role of EVs in inter-organismal communication.
- 60.Maguire JE, et al. Myristoylated CIL-7 regulates ciliary extracellular vesicle biogenesis. Mol. Biol. Cell. 2015;26:2823–2832. doi: 10.1091/mbc.E15-01-0009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Nakano I, Garnier D, Minata M, Rak J. Extracellular vesicles in the biology of brain tumour stem cells — implications for inter-cellular communication, therapy and biomarker development. Semin. Cell Dev. Biol. 2015;40:17–26. doi: 10.1016/j.semcdb.2015.02.011. An excellent discussion on the role of EVs in brain tumours.
- 62.Pegtel DM, Peferoen L, Amor S. Extracellular vesicles as modulators of cell-to-cell communication in the healthy and diseased brain. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014;369:20130516. doi: 10.1098/rstb.2013.0516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Grad LI, Fernando SM, Cashman NR. From molecule to molecule and cell to cell: prion-like mechanisms in amyotrophic lateral sclerosis. Neurobiol. Dis. 2015;77:257–265. doi: 10.1016/j.nbd.2015.02.009. [DOI] [PubMed] [Google Scholar]
- 64.Aguzzi A, Lakkaraju AK. Cell biology of prions and prionoids: a status report. Trends Cell Biol. 2015;26:40–51. doi: 10.1016/j.tcb.2015.08.007. [DOI] [PubMed] [Google Scholar]
- 65.Guest WC, Plotkin SS, Cashman NR. Toward a mechanism of prion misfolding and structural models of PrP(Sc): current knowledge and future directions. J. Toxicol. Environ. Health A. 2011;74:154–160. doi: 10.1080/15287394.2011.529065. [DOI] [PubMed] [Google Scholar]
- 66.Eisele YS, et al. Peripherally applied Aβ-containing inoculates induce cerebral β-amyloidosis. Science. 2010;330:980–982. doi: 10.1126/science.1194516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Clavaguera F, et al. Transmission and spreading of tauopathy in transgenic mouse brain. Nat. Cell Biol. 2009;11:909–913. doi: 10.1038/ncb1901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Luk KC, et al. Pathological α-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice. Science. 2012;338:949–953. doi: 10.1126/science.1227157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Grad LI, et al. Intermolecular transmission of superoxide dismutase 1 misfolding in living cells. Proc. Natl Acad. Sci. USA. 2011;108:16398–16403. doi: 10.1073/pnas.1102645108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Coleman BM, Hill AF. Extracellular vesicles — their role in the packaging and spread of misfolded proteins associated with neurodegenerative diseases. Semin. Cell Dev. Biol. 2015;40:89–96. doi: 10.1016/j.semcdb.2015.02.007. An excellent discussion about the role of EVs in spreading misfolded proteins associated with neurodegenerative disorders.
- 71.Grad LI, et al. Intercellular propagated misfolding of wild-type Cu/Zn superoxide dismutase occurs via exosome-dependent and -independent mechanisms. Proc. Natl Acad. Sci. USA. 2014;111:3620–3625. doi: 10.1073/pnas.1312245111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Emmanouilidou E, et al. Cell-produced α-synuclein is secreted in a calcium-dependent manner by exosomes and impacts neuronal survival. J. Neurosci. 2010;30:6838–6851. doi: 10.1523/JNEUROSCI.5699-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Melachroinou K, et al. Deregulation of calcium homeostasis mediates secreted α-synuclein-induced neurotoxicity. Neurobiol. Aging. 2013;34:2853–2865. doi: 10.1016/j.neurobiolaging.2013.06.006. [DOI] [PubMed] [Google Scholar]
- 74.Rajendran L, et al. Alzheimer’s disease β-amyloid peptides are released in association with exosomes. Proc. Natl Acad. Sci. USA. 2006;103:11172–11177. doi: 10.1073/pnas.0603838103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Joshi P, et al. Microglia convert aggregated amyloid-β into neurotoxic forms through the shedding of microvesicles. Cell Death Differ. 2014;21:582–593. doi: 10.1038/cdd.2013.180. An important review highlighting the complexity of protein forms involved in neurodegeneration and the role of EVs.
- 76.Sharples RA, et al. Inhibition of γ-secretase causes increased secretion of amyloid precursor protein C-terminal fragments in association with exosomes. FASEB J. 2008;22:1469–1478. doi: 10.1096/fj.07-9357com. [DOI] [PubMed] [Google Scholar]
- 77.Joshi P, Benussi L, Furlan R, Ghidoni R, Verderio C. Extracellular vesicles in Alzheimer’s disease: friends or foes? Focus on aβ-vesicle interaction. Int. J. Mol. Sci. 2015;16:4800–4813. doi: 10.3390/ijms16034800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yuyama K, Sun H, Mitsutake S, Igarashi Y. Sphingolipid-modulated exosome secretion promotes clearance of amyloid-β by microglia. J. Biol. Chem. 2012;287:10977–10989. doi: 10.1074/jbc.M111.324616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Asai H, et al. Depletion of microglia and inhibition of exosome synthesis halt tau propagation. Nat. Neurosci. 2015;18:1584–1593. doi: 10.1038/nn.4132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Al-Nedawi K, et al. Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells. Nat. Cell Biol. 2008;10:619–624. doi: 10.1038/ncb1725. [DOI] [PubMed] [Google Scholar]
- 81.Al-Nedawi K, Meehan B, Kerbel RS, Allison AC, Rak J. Endothelial expression of autocrine VEGF upon the uptake of tumor-derived microvesicles containing oncogenic EGFR. Proc. Natl Acad. Sci. USA. 2009;106:3794–3799. doi: 10.1073/pnas.0804543106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Bronisz A, et al. Extracellular vesicles modulate the glioblastoma microenvironment via a tumor suppression signaling network directed by miR-1. Cancer Res. 2014;74:738–750. doi: 10.1158/0008-5472.CAN-13-2650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Tominaga N, et al. Brain metastatic cancer cells release microRNA-181c-containing extracellular vesicles capable of destructing blood–brain barrier. Nat. Commun. 2015;6:6716. doi: 10.1038/ncomms7716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Carandini T, et al. Microvesicles: what is the role in multiple sclerosis? Front. Neurol. 2015;6:111. doi: 10.3389/fneur.2015.00111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Verderio C, et al. Myeloid microvesicles are a marker and therapeutic target for neuroinflammation. Ann. Neurol. 2012;72:610–624. doi: 10.1002/ana.23627. [DOI] [PubMed] [Google Scholar]
- 86.Minagar A, Alexander JS. Blood–brain barrier disruption in multiple sclerosis. Mult. Scler. 2003;9:540–549. doi: 10.1191/1352458503ms965oa. [DOI] [PubMed] [Google Scholar]
- 87.Saenz-Cuesta M, Osorio-Querejeta I, Otaegui D. Extracellular vesicles in multiple sclerosis: what are they telling us? Front. Cell. Neurosci. 2014;8:100. doi: 10.3389/fncel.2014.00100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Prada I, Furlan R, Matteoli M, Verderio C. Classical and unconventional pathways of vesicular release in microglia. Glia. 2013;61:1003–1017. doi: 10.1002/glia.22497. [DOI] [PubMed] [Google Scholar]
- 89.Peferoen L, Kipp M, van der Valk P, van Noort JM, Amor S. Oligodendrocyte–microglia cross-talk in the central nervous system. Immunology. 2014;141:302–313. doi: 10.1111/imm.12163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Saenz-Cuesta M, et al. Circulating microparticles reflect treatment effects and clinical status in multiple sclerosis. Biomark. Med. 2014;8:653–661. doi: 10.2217/bmm.14.9. [DOI] [PubMed] [Google Scholar]
- 91.Paschon V, et al. Interplay between exosomes, microRNAs and Toll-like receptors in brain disorders. Mol. Neurobiol. 2015;11:11. doi: 10.1007/s12035-015-9142-1. [DOI] [PubMed] [Google Scholar]
- 92.Gupta A, Pulliam L. Exosomes as mediators of neuroinflammation. J. Neuroinflammation. 2014;11:68. doi: 10.1186/1742-2094-11-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Frühbeis C, Fröhlich D, Krämer-Albers EM. Emerging roles of exosomes in neuron–glia communication. Front. Physiol. 2012;3:119. doi: 10.3389/fphys.2012.00119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.An K, et al. Exosomes neutralize synaptic-plasticity-disrupting activity of Aβ assemblies in vivo. Mol. Brain. 2013;6:47. doi: 10.1186/1756-6606-6-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Polazzi E, Monti B. Microglia and neuroprotection: from in vitro studies to therapeutic applications. Prog. Neurobiol. 2010;92:293–315. doi: 10.1016/j.pneurobio.2010.06.009. [DOI] [PubMed] [Google Scholar]
- 96.De Maio A, Vazquez D. Extracellular heat shock proteins: a new location, a new function. Shock. 2013;40:239–246. doi: 10.1097/SHK.0b013e3182a185ab. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Tytell M, Greenberg SG, Lasek RJ. Heat shock-like protein is transferred from glia to axon. Brain Res. 1986;363:161–164. doi: 10.1016/0006-8993(86)90671-2. [DOI] [PubMed] [Google Scholar]
- 98.Taylor AR, Robinson MB, Gifondorwa DJ, Tytell M, Milligan CE. Regulation of heat shock protein 70 release in astrocytes: role of signaling kinases. Dev. Neurobiol. 2007;67:1815–1829. doi: 10.1002/dneu.20559. [DOI] [PubMed] [Google Scholar]
- 99.Pusic AD, Kraig RP. Youth and environmental enrichment generate serum exosomes containing miR-219 that promote CNS myelination. Glia. 2014;62:284–299. doi: 10.1002/glia.22606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Dugas JC, et al. Dicer1 and miR-219 are required for normal oligodendrocyte differentiation and myelination. Neuron. 2010;65:597–611. doi: 10.1016/j.neuron.2010.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zhao X, et al. MicroRNA-mediated control of oligodendrocyte differentiation. Neuron. 2010;65:612–626. doi: 10.1016/j.neuron.2010.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Aryani A, Denecke B. Exosomes as a nanodelivery system: a key to the future of neuromedicine? Mol. Neurobiol. 2014 doi: 10.1007/s12035-014-9054-5. http://dx.doi.org/10.1007/s12035-014-9054-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Zhuang X, et al. Treatment of brain inflammatory diseases by delivering exosome encapsulated anti-inflammatory drugs from the nasal region to the brain. Mol. Ther. 2011;19:1769–1779. doi: 10.1038/mt.2011.164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Alvarez-Erviti L, et al. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 2011;29:341–345. doi: 10.1038/nbt.1807. [DOI] [PubMed] [Google Scholar]
- 105.Yeo RW, et al. Mesenchymal stem cell: an efficient mass producer of exosomes for drug delivery. Adv. Drug Deliv. Rev. 2013;65:336–341. doi: 10.1016/j.addr.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 106.Park JS, Suryaprakash S, Lao YH, Leong KW. Engineering mesenchymal stem cells for regenerative medicine and drug delivery. Methods. 2015;84:3–16. doi: 10.1016/j.ymeth.2015.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Xin H, et al. Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J. Cereb. Blood Flow Metab. 2013;33:1711–1715. doi: 10.1038/jcbfm.2013.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Katsuda T, et al. Human adipose tissue-derived mesenchymal stem cells secrete functional neprilysin-bound exosomes. Sci. Rep. 2013;3:1197. doi: 10.1038/srep01197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Pusic AD, Pusic KM, Clayton BL, Kraig RP. IFNγ-stimulated dendritic cell exosomes as a potential therapeutic for remyelination. J. Neuroimmunol. 2014;266:12–23. doi: 10.1016/j.jneuroim.2013.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Haney MJ, et al. Exosomes as drug delivery vehicles for Parkinson’s disease therapy. J. Control. Release. 2015;207:18–30. doi: 10.1016/j.jconrel.2015.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Schwab A, et al. Extracellular vesicles from infected cells: potential for direct pathogenesis. Front. Microbiol. 2015;6:1132. doi: 10.3389/fmicb.2015.01132. [DOI] [PMC free article] [PubMed] [Google Scholar]