Abstract
Ectopic gene expression through introduction of cDNA and gene silencing by RNA interference each facilitate the elucidation of molecular pathways in both normal and pathologic states. As transfection efficiency in some primary and established cells is low, lentivirus based expression systems with high infection rates can improve experimental design. For example, glioblastoma cells and particularly the cancer stem cell (CSC) fraction can be difficult to transfect but are amenable to viral infection. Greater utilization of lentivirus for expression of cDNA and shRNA in CSCs may be limited due to technical challenges, including elimination of pro-differentiating fetal bovine serum (FBS). We therefore generated a subline of 293Ts that can proliferate and efficiently produce virus in CSC media, designated CSC293Ts. We provide detailed protocols for the generation of CSC293Ts and for the production of lentivirus for CSC infection using glioblastoma as a model. Our data demonstrate that serum free media from CSC293Ts consistently produces greater than 80% infection rates without virus concentration. We believe that the detailed protocols provided here can be adapted for multiple cell types for broad utility.
Keywords: lentivirus, cancer stem cell infection, cellular transduction
INTRODUCTION
Cancer stem cells (CSCs) contribute to tumor maintenance and recurrence via therapeutic resistance, angiogenesis, and invasion [1-6]. The molecular mechanisms mediating these protumorigenic effects are of significant interest as identification of novel pathways can provide novel opportunities for targeted therapy development [5-6]. To elucidate CSC specific signals and the biologies they control, pathways can be modulated via gene overexpression and knockdown. While CSC transfection may be difficult, infection can provide an efficient mechanism through which to express specific genes or short hairpin sequences in CSCs. One method through which CSCs can be transduced is through the use of lentivirus, a member of a family of enveloped viruses which can infect mammalian cells [7-9].
As lentivirus is capable of infecting and stably incorporating into the human genome, biological safety is increased through the use of multi-vector systems which typically are based on human immunodeficiency virus type I (HIV) and include packaging, envelope, and transfer vectors [7-9]. Packaging vectors contain Gag and Pol domains which provide structural proteins including matrix, capsid, and nucleocapsid proteins as well as reverse transcriptase and integrase for conversion of viral RNA to DNA with genomic integration. Envelope vectors contain vesicular stomatitis virus G glycoprotein (VSVG), which is used to pseudotype lentiviral vectors to provide broad tropism for transduction. Transfer vectors contain DNA or shRNAs of interest located between long terminal repeats (LTRs) that facilitate viral integration into the genome. To improve biosafety, transfer vectors often have deletions in the 3’ LTR to yield self-inactivating virus. The packaging, envelope, and transfer vectors are transfected into simian virus 40 T antigen expressing human embryonic kidney 293 (239T or HEK 293T) cells to produce virus in cell culture media that is then concentrated or used directly for experiments [7-9].
The use of lentivirus for transduction of CSCs has several advantages. Lentivirus integration can occur in both proliferating and non-dividing cells, suggesting that if a quiescent CSC population does exist within tumors both this tumor subset as well as any proliferative progenitor populations would be transduced. Infection will result in the integration of a gene or shRNA, providing stable expression that can be important for further in vivo studies. Infection rates can be modulated by alteration of amounts of virus applied and number of cells infected to achieve the desired multiplicity of infection [7-9]. Another advantage for lentivirus usage in experimental design is that cDNAs and shRNAs for many genes as well as some reporter systems are commercially available from companies such as Addgene, GE Healthcare, GeneCopoeia, Sigma, and System Biosciences allowing for project acceleration.
While there are benefits of lentivirus use for CSC infection, there are concerns and limitations. Precautions against viral integration into the handler’s DNA requires BSL2 conditions with appropriate Institutional Biosafety Committee approvals. In addition, standard 293T culture requires fetal bovine serum (FBS), a pro-differentiating agent that will reduce CSC maintenance. To avoid this issue, many laboratories are concentrating virus with commercially available polyethylene glycol based products or using less than optimal amounts of virus containing media to ensure FBS concentrations remain low. Even with these approaches, successful lentivirus-based transduction of CSCs has been elusive for some groups. We therefore developed an efficient method of transduction of CSCs in the absence of FBS without the need for viral concentration and now provide detailed protocols with evidence of glioblastoma CSCs infection.
MATERIALS AND METHODS
Cell Culture of 293Ts
LentiX 293T (Parental 293T) cells were obtained from Clontech (632180). Cells were thawed in Gibco DMEM, high glucose media (Life Technologies, 11965-092) with 10% Fetal Bovine Serum (Corning, 35-016-CV) and 1% Penicillin/Streptomycin (Corning, 30-002-CI). When cells grew to a density of 60-70%, they were split 1:4 using Gibco StemPro Accutase Cell Dissociation Reagent (Life Technologies, A11105-01) on a canted neck T-75 flask (Corning, 430641). For the generation of CSC293Ts, an aliquot of the parental 293T cells was first cultured in 50% DMEM high glucose media with 10% FBS and 50% CSC media [DMEM/F-12 50/50 media (Corning, 16-405-CV), Gibco B-27 supplement w/o vitamin A (Life Technologies, 12587-010), 1% Penicillin/Streptomycin, 1% Sodium Pyruvate (Corning, 25-000-CI), 10μg Recombinant Human EGF (R&D Systems, 236-EG-01M), and 10 μg Recombinant Human FGF basic 145aa (R&D Systems, 4114-TC-01M)]. After the first passage, 293T cells were continuously cultured without FBS as follows. 4 mL of Gibco Geltrex (Life Technologies, A1413302) at a concentration of 0.1mg/ml in CSC media was used to coat a T-75 flask. The flask was incubated at 37°C for a minimum of 20 minutes prior to plating cells. 293Ts were allowed to adhere overnight. The cells continuously grew and were split at each passage once they reached 60-70% confluence.
Transfection of 293Ts
293T cells were plated in triplicate in the absence of penicillin/streptomycin at a density of 250,000 per 6 well (Corning, 3516) 16-24 hours prior to transfection. Parental 293Ts were plated in 1 mL DMEM high glucose with 10% FBS whereas CSC293Ts were plated in 1 mL CSC media. Each well received 0.5 μg pGreenFire1-CMV (pGF1-CMV, transfer vector, System Biosciences), 0.25 μg ps-PAX2 (packaging vector, Addgene #12260), and 0.25 μg pCL-VSVG (envelope vector, Addgene #1733). Plasmids were diluted in 50 μL of DMEM without any supplements, vortexed, and then centrifuged for 5 seconds. 3 μL of FuGene HD (Promega, E2311) per well was added directly to the media in the DNA mixture. The tube was again vortexed and centrifuged for 5 seconds. The DNA and transfection reagent mixture was then incubated for 10 minutes at room temperature, and the entire solution in the tube subsequently added to a 6 well. Plates were then placed in a tissue culture incubator designated for lentivirus use.
Harvest of Virus Containing Media from 293Ts with Biosafety Precautions
After the initial transfection, cells were assumed to be producing lentivirus and handled accordingly. Biosafety precautions included the following: wearing a disposable, waterproof gown (18-567, Fisher Scientific) with double gloves, exposing all pipettes contacting virus to 0.825% sodium hypochlorite, disposing of pipette tips and other small consumables in a biohazard bag closed inside the hood, and decontaminating all surfaces with 0.825% sodium hypochlorite followed by 70% ethanol at the completion of experiments. 24 hours post transfection, the media containing transfection reagent was removed and 1.5 mL of media containing penicillin/streptomycin added. As during the transfection, parental 293Ts were cultured in DMEM high glucose with 10% FBS and CSC293Ts were cultured in CSC media on geltrex. After an additional 24 hours (48 hrs post transfection), virus containing media was removed from the parental 293Ts and CSC293Ts using a disposable pipette. Media was placed in a disposable syringe and filtered through a low protein binding 0.45 μm filter (SLHV033RS, Millipore) to remove any residual 293Ts.
Infection of Glioblastoma Stem Cells
Glioblastoma stem cells (GSC83, graciously provided by Dr. Ichiro Nakano of Ohio State University) were plated in triplicate at a density of 150,000 cells per 6-well in 1 mL CSC media with penicillin/streptomycin 16-24 hours prior to infection. Each well received 500 μL of filtered, parental 293T or CSC293T generated virus. At 24-72 hours post infection, cells were observed using an EVOS (Life Technologies, 12563471) fluorescent microscope to visualize the green fluorescent protein (GFP) expressed by pGF1-CMV. Representative images were taken to demonstrate successful infection shown via GFP positive cells. 72 hours after infection, cells were harvested for flow cytometry.
Flow Cytometry
293T cells were analyzed by flow cytometry for transfection efficiency whereas GSC83s were analyzed for infection efficiency. For 293Ts and GSC83s, viral supernatant was removed, cells harvested, cells passaged through a 30 μm filter, and SYTOX blue (Life Tech, S34857) added as a viability marker prior to analysis by flow cytometry. For each experiment, negative controls (not transfected or uninfected cells) without SYTOX Blue were used to establish the gates for GFP and SYTOX high and low populations.
RESULTS
Generation of CSC293Ts
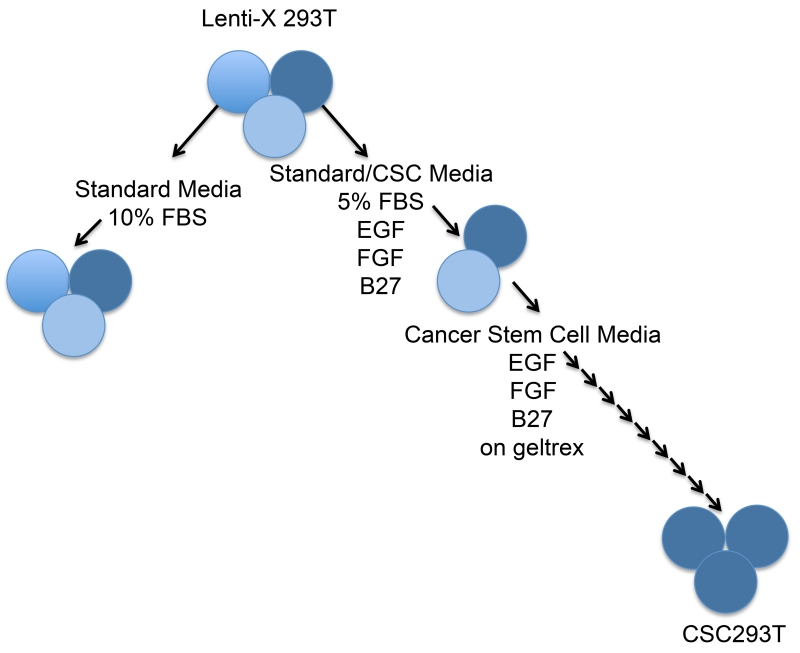
Fetal bovine serum (FBS) is a standard component of 293T cell culture media in which lentivirus is produced, but FBS promotes the differentiation of cancer stem cells (CSCs). We therefore developed a strategy to generate a subline of 293Ts that could grow and efficiently produce virus in CSC media. To accomplish this goal, we utilized Lenti-X 293Ts as a parental population because the cells are reported by Clontech to produce six fold higher virus particles than 293FT cells typically used for lentivirus production. We initially cultured the parental Lenti-X 293Ts in a 1:1 mixture of CSC media and the recommended, standard cell culture media containing FBS. After this first passage in a 5% FBS media, Lenti-X 293Ts were cultured in CSC media on geltrex and never allowed to reach greater than 70% confluence. Although early passage of the Lenti-X 293Ts in CSC media resulted in cells which produced minimal virus (data not shown), the subline resulting after nine passages in CSC media efficiently generated virus and was designated CSC293T (Figure 1).
Figure 1. Strategy for Generation of 293T Cells Producing Lentivirus in Cancer Stem Cell Media.
Diagram representing the methods used to isolate a subline of LentiX-293Ts capable of growing and producing virus in cancer stem cell media. LentiX-293Ts were initially cultured in a 1:1 mixture of the 293T standard media (DMEM +10% FBS) and cancer stem cell media (DMEM/F12+EGF+FGF+B27). Once LentiX-293Ts grew to confluence, they were passaged into cancer stem cell media on geltrex coated dishes. Cells were then continually passaged in cancer stem cell media on geltrex to obtain a LentiX-293T subline that proliferated in the absence of serum, designated CSC293T. We anticipate that a similar strategy can be used to develop 293T sublines for producing lentivirus in many other medias required for specific cell types.
CSC293T Transfection in the Absence of FBS
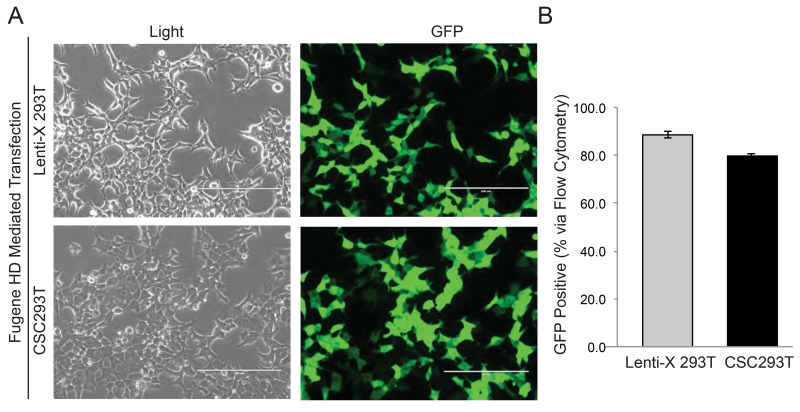
High rates of transfection of viral transfer, packaging, and envelope plasmids is critical to facilitate virus production. Beginning with transfection, use of a transfer vector containing a fluorescent protein to monitor and troubleshoot the steps of viral production is an important control (Figure 2). Using this strategy, 293Ts were transfected with the green fluorescent protein (GFP) expressing pGF1-CMV vector to determine if CSC293Ts had rates of transfection comparable to parental 293Ts. Observation of cells using a fluorescent microscope demonstrated strong GFP expression in the majority of cells within twenty-four hours of transfection using Fugene HD (Figure 3A). Rates of transfection did vary with cellular confluence and transfection reagents (data not shown), and optimal conditions visually observed were further repeated and quantified. Although Lenti-X 293Ts did have slightly higher rates of transfection than CSC293Ts, the percentage of GFP positive cells seventy-two hours after transfection determined via flow cytometry was always greater than 75% transfection efficiency in both parental Lenti-X 293Ts and CSC293Ts (Figure 3B). These data demonstrated that CSC293Ts could be cultured and successfully transfected under CSC permissive conditions in which serum is absent.
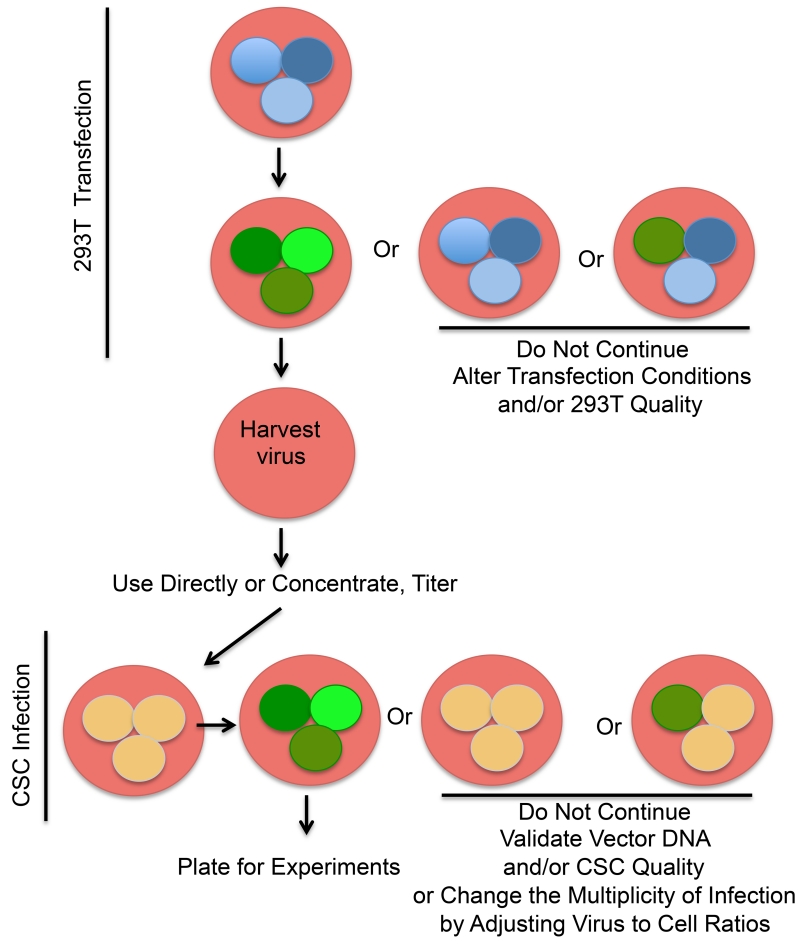
Figure 2. Fluorescent Proteins for Monitoring Lentivirus Production and CSC Infection.
Diagram representing the use of a fluorescent protein as a reporter for transfection efficiency in 293Ts and infection efficiency in CSCs. Inclusion of a transfer vector expressing fluorescent protein as a positive control allows monitoring of virus production at each step and helps to save time and money by facilitating troubleshooting. If expression is not observed within 48 hours of 293T transfection, transfection reagents or 293T cell quality (including viability or confluence) are likely causes. If fluorescent protein expression is observed post transfection, but not within 72 hours of CSC infection, packaging or envelope vector DNA may be degraded. Alternatively, the viability of CSCs may be low. If none of these issues is causative, the multiplicity of infection for the CSCs of interest may need to be increased by lowering the cell number or increasing the amount of virus applied.
Figure 3. High Rates of Transfection in CSC293Ts.
(A) Representative images under light and fluorescent microscopy of Lenti-X 293Ts and CSC293Ts 24 hours post transfection with pGF1-CMV vector expressing GFP are shown with the white bar representing 200 μm. (B) Quantification of the percentage of GFP positive 293Ts 72 hours post transfection is shown.
CSC293Ts Produce Virus That Efficiently Infects Glioma Stem Cells
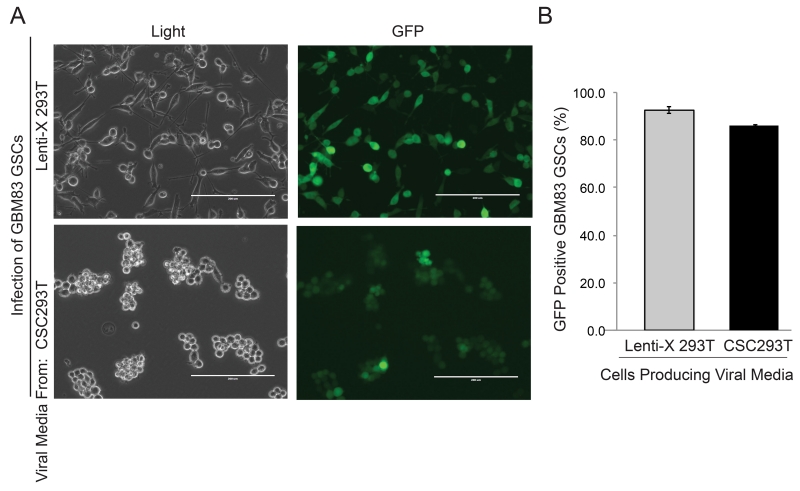
To determine if CSC293Ts could produce virus which could have similar rates of infection to the parental cells, media collected from Lenti-X 293Ts or CSC293Ts was added to glioblastoma stem cells (GSCs). Visualization of fluorescence demonstrated strong GFP expression in the majority of GSCs infected with media from either Lenti-X 293Ts or CSC293Ts within 72 hours (Figure 4A). However, observation via light microscopy demonstrated a strong change in cell morphology in GSCs infected with media from Lenti-X 293Ts that contained FBS (Figure 4A). Infection with FBS containing viral media promoted cell attachment and an astrocyte like morphology consistent with differentiation. In striking contrast, GSCs infected with viral media from CSC293Ts continued to grow non-adherently (Figure 4A). Quantification of the percentage of GFP positive GSCs via flow cytometry demonstrated that both Lenti-X 293Ts and CSC293Ts generated virus capable of infecting 85% or more of the live GSCs (Figure 4B). Using the number of GFP positive cells per mL of viral supernatant to measure the functional titer [9], we estimate number of transduction units per mL without viral concentration to be approximately 1.5×105. Thus, CSC293Ts can produce virus capable of infecting GSCs at high efficiency.
Figure 4. CSC293T Produce Virus Capable of Efficiently Infecting Cancer Stem Cells without Concentration.
(A) Representative images under light and fluorescent microscopy of GBM83 CSCs 72 hours post infection are shown with the white bar representing 200 μm. CSCs were infected with GFP expressing virus containing media produced by Lenti-X 293Ts or CSC293Ts. (B) Quantification of the percentage of GFP positive GSCs 72 hours post infection is shown.
DISCUSSION
The ability to perturb CSC signaling pathways and elucidate consequences on biological functions is an important requirement for tumor studies. There are many mechanisms through which this goal may be achieved, including use of: transient transfection of mammalian expression plasmids; clonal selection for stable transfection; genome editing with transcription activator-like effector nucleases (TALEN) or clustered regularly interspaced short palindromic repeats (CRISPR); infection with adeno- or lentivirus, and small molecule inhibitors. Each method has advantages and disadvantages, but, once a laboratory has established the technique, lentivirus provides a relatively low-cost method of achieving high rates of stable cell transduction. For CSC transduction, establishment of the technique can be hindered by either lack of successful lentivirus production or cell maintenance post-infection. We therefore sought to provide detailed methodologies for lentivirus production and use, and derived a subline of Lenti-X 293Ts that could specifically be used for generation of lentivirus for CSC infection.
We derived and continue to passage CSC293Ts on geltrex as an extracellular matrix. While geltrex promotes adherence in the absence of serum and reduces the potential for 293T contamination of viral supernatants, cost is increased. When we tested the ability of CSC293Ts to grow in the absence of geltrex, we determined that the cells thrived as spheres. These non-adherent CSC293Ts could also be transfected at high rates and produce virus efficiently (data not shown). As cells can be plated at higher density when grown as spheres, this cell culture method may provide benefit if large and/or more concentrated amounts of virus are needed. However, it would be important to transfect single cells or doublets (prior to sphere formation) to ensure high transfection rates. In addition, non-adherent CSC293Ts should first be centrifuged and then the viral supernatant passaged through a filter prior to use. Therefore, our CSC293T cell culture protocols can be further modified as needed.
We produced the infection rates provided here using small amounts of DNA (total 1 μg/6 well) with a ratio of 2:1:1 for the transfer vector to the packaging and envelope vectors during CSC293T transfection. The low DNA amounts reduce costs and worked well for multiple transfer vectors in our laboratory, but may be optimized for different transfer vectors and packaging systems. For example, we found that a transfer vector with a large insert (>5 kb) optimally generated virus when a 1:2:2 ratio of transfer vector to the packaging and envelop vectors was used instead of our previously established standard. This change in DNA ratios only slightly affected transfection rates, but had strong effects on transduction efficiency. These observations reiterate the importance of having a well established and highly successful system that can be slightly altered in specific instances to improve efficacy. We believe that our protocols and improved cell line provide such a system for lentivirus production and CSC transduction.
ACKNOWLEDGMENTS
These studies were supported by National Institutes of Health grant R01 CA1515122 (ABH), the UAB Brain Tumor SPORE Career Development Award, and startup funds from the University of Alabama at Birmingham Department of Cell, Developmental and Integrative Biology, the Comprehensive Cancer Center, the Civitan International Research Center for Glial Biology in Medicine, the Center for Free Radical Biology, and the Neuro-Oncology Brain SPORE. We appreciate the technical assistance of Marion Spell at the UAB Flow Cytometry Facility. We thank Nathaniel Boyd and Anh Tran for assistance with manuscript editing.
Footnotes
CONFLICT OF INTEREST STATEMENT
We have no conflicts of interest.
REFERENCES
- [1].Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105–111. doi: 10.1038/35102167. [DOI] [PubMed] [Google Scholar]
- [2].Venere M, Fine HA, Dirks PB, Rich JN. Cancer stem cells in gliomas: identifying and understanding the apex cell in cancer’s hierarchy. Glia. 2011;59:1148–1154. doi: 10.1002/glia.21185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Magee JA, Piskounova E, Morrison SJ. Cancer stem cells: impact, heterogeneity, and uncertainty. Cancer Cell. 2012;21:283–296. doi: 10.1016/j.ccr.2012.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Vlashi E, Pajonk F. Cancer stem cells, cancer cell plasticity and radiation therapy. Semin Cancer Biol. 2014 doi: 10.1016/j.semcancer.2014.07.001. doi: 10.1016/j.semcancer.2014.07.001 [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Pattabiraman DR, Weinberg RA. Tackling the cancer stem cells - what challenges do they pose? Nat Rev Drug Discov. 2014;13:497–512. doi: 10.1038/nrd4253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Maccalli C, De Maria R. Cancer stem cells: perspectives for therapeutic targeting. Cancer Immunol Immunother. 2014 doi: 10.1007/s00262-014-1592-1. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Delenda C. Lentiviral vectors: optimization of packaging, transduction and gene expression. J Gene Med. 2004;6(Suppl 1):S125–38. doi: 10.1002/jgm.501. [DOI] [PubMed] [Google Scholar]
- [8].Mendenhall A, Lesnik J, Mukherjee C, Antes T, Sengupta R. Packaging HIV- or FIV-based Lentivector Expression Constructs & Transduction of VSV-G Pseudotyped Viral Particles. J Vis Exp. 2012:e3171. doi: 10.3791/3171. doi:10.3791/3171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Sastry L, 1, Johnson T, Hobson MJ, Smucker B, Cornetta K. Titering lentiviral vectors: comparison of DNA, RNA and marker expression methods. Gene Ther. 2002;9:1155–1162. doi: 10.1038/sj.gt.3301731. [DOI] [PubMed] [Google Scholar]