Abstract
The aim of this analysis was to examine the association between tea intake during pregnancy and maternal and infant metal exposures. Data from the Maternal-Infant Research on Environmental Chemicals (MIREC) Study, a pan-Canadian pregnancy cohort, were used. All participants with a gestational age of ⩾20 weeks (n=1954) with available biomarkers were included. Geometric means (GMs) for lead, arsenic, mercury, cadmium and manganese in maternal (first and third trimesters) and cord blood, as well as speciated arsenic in maternal urine in the first trimester, were calculated for participants who drank regular, green or herbal tea and for those who did not. Differences between groups were examined using chi-square tests. Adjusted least squares geometric means (LSGMs) were estimated by tea intake, controlling for factors such as country of birth, coffee intake and maternal smoking. Concentrations of all metals were above the limits of detection in most participants in the first trimester: lead (GM): 0.62 μg/dl), mercury (GM: 2.99 nmol/l); cadmium (GM 1.93 nmol/l), arsenic (GM 9.75 nmol/l) and manganese (GM 160.1 nmol/l). Adjusted LSGMs for lead in the first trimester were higher for tea drinkers than for those who were non-tea drinkers (LSGM 0.65 μg/dl, 95%CI: 0.62, 0.69 and 0.61 μg/dl, 95%CI: 0.59, 0.62), and there was evidence of a dose–response relationship for green and herbal tea. Those who consumed herbal tea in the third trimester had significantly higher third trimester maternal and cord blood lead concentrations than non-herbal tea drinkers. This study provides evidence of an association between blood lead concentrations and green or herbal tea consumption. However, the GM blood lead concentrations of the highest tea consumers were still less than 1 μg/dl and within the normal range of blood lead concentrations in the Canadian population.
Keywords: lead, metals, MIREC, tea
INTRODUCTION
Elevated metal concentrations in a prenatal population could indicate an increased risk for adverse health outcomes. Prenatal lead, mercury, cadmium and manganese concentrations have been associated with adverse fetal growth and poor neurological outcomes in the offspring.1, 2, 3, 4, 5, 6, 7 Although several sources of metal exposure have been identified,8 there is some concern that tea consumption may contribute to exposure.9 Tea leaves that are cultivated in certain countries, including China10 and Iran,11 have detectable concentrations of metals, including lead, mercury, cadmium and arsenic. Tea leaves may be contaminated with metals when grown in regions with polluted soil, heavy industry or where certain pesticides and fertilizers are used.12 Women may choose to reduce their consumption of tea during pregnancy, which generally stems from prenatal guidance to consume less than 300 mg of caffeine per day,13 though some women will continue to drink tea during pregnancy.14, 15 The potential link between consumption of tea during pregnancy and increased blood concentrations of these metals is not well understood. Thus, the aim of this study is to examine the association between tea intake during pregnancy and metal concentrations in the blood or urine.
METHODS
Data from the Canadian Maternal-Infant Research on Environmental Chemicals (MIREC) Study cohort were used for these analyses. The MIREC Study methods are outlined briefly here and in detail elsewhere.16, 17 Two thousand women were recruited in the first trimester of pregnancy (<14 weeks gestation) from obstetric and prenatal clinics in 10 cities across Canada, from 2008 to 2011. Eligibility criteria included the ability to consent and to communicate in English or French, 18 years of age or older, planning to deliver at a local hospital, and participation in the cord blood collection component of the MIREC Study. Women with a medical history of major chronic disease, threatened abortion or illicit drug use were excluded. The study was reviewed and approved by the Health Canada Research Ethics Board and the ethics committees at the participating hospitals and research centers across Canada. Potential participants were provided with information on the objectives and design of the study and asked to sign the consent forms.
Questionnaires were administered during the first and third trimester clinic visits to collect sociodemographic (i.e., maternal age, highest level of maternal education, annual household income, country of birth [Canada or other] and marital status) and behavioural (e.g., beverage consumption — including alcohol and coffee intake — and smoking) information. Pre-pregnancy body mass index (BMI) was calculated by dividing self-reported weight (kg) by measured height squared (m2).
Tea Intake
Intake of regular (black), green and herbal tea was evaluated in the first and third trimesters. Women reported their frequency of consumption for each type of tea in terms of number of 6 ounce cups per day, week or month. A dichotomous variable (tea drinkers or non-tea drinkers) was created for each type of tea, as well as all types of tea combined. Further, a variable was derived to describe frequency as number of cups per week (none to <1; 1 to <7; ⩾7) for each type of tea, as well as all types combined. Since the majority of participants in the “none to <1 cup/week” category reported consuming tea “never” or “less than 2 times/month” and because a sensitivity analysis examining women with no tea consumption at all as a category did not change the results of the analyses, we collapsed these two categories into the “none to <1 cup/week” category.
Metals Analysis
Metal concentrations of lead, cadmium, arsenic, total mercury and manganese in maternal whole blood were measured in the first and third trimester visits. Cord blood concentrations of these metals were also measured. Urine analysis for speciated arsenic was conducted in the first trimester.
Maternal and cord blood were analysed by sample dilution followed by inductively coupled plasma mass spectrometry (ICP-MS) analysis [ICP-MS DRC-II; Elan Perkin Elmer]. Urinary analysis for speciated arsenic was carried out by HPLC coupled to a Varian 820 MS ICP-MS. All chemical analyses were carried out by the Toxicology Laboratory, located in the Institut national de santé publique du Québec, which is accredited by the Standards Council of Canada under ISO 17025 and CAN-P-43. The accuracy and precision of the analyses are evaluated on a regular basis through the laboratory's participation in external quality assessment programmes.
Statistical Analysis
Analyte results below the limits of detection (LOD), defined as results indistinguishable from zero, were counted as half the detection limit. Descriptive statistics were used to characterize the population by intake of regular, green or herbal tea. The proportions of women consuming regular, green or herbal tea during the first and third trimesters of pregnancy were described by geometric mean (GM) of each metal in the maternal whole blood collected at these time points. The concentration of metals in cord blood in relation to consumption of each type of tea in the third trimester was also examined. Differences between categories were examined using chi-square tests. Where significant differences in GM metal concentrations were observed between tea drinkers and non-tea drinkers, adjusted least squares geometric means (LSGMs), and their confidence intervals, were calculated by frequency of tea intake for each type of tea consumed. The LSGM was estimated controlling for maternal age, household income, highest level of maternal education, country of birth, coffee intake, maternal smoking, exposure to second hand smoke and maternal pre-pregnancy BMI. Where concentrations of metals were not normally distributed, log transformations were used in the regression models. Further, cumulative percentile curves were generated for metals with significant adjusted LSGM for each type of tea consumed.
LSGMs were calculated to adjust for confounders. To limit the bias caused by a high percentage of samples with no detectable metal concentrations, only those samples with >50% detectable concentrations were included in the analyses. All participants with an offspring gestational age of ⩾20 weeks were included in the analyses (n=1954). The sample size varied by availability of biomarkers data. The analyses were conducted using the SAS version 9.3 software.
RESULTS
Population Characteristics
MIREC participant characteristics, by prevalence of reported tea drinking (regular, green or herbal) in the first and third trimesters, are described in Table 1. The majority of the women were Canadian born (81%), 30 years of age or older (70%), non-smokers (92%), first pregnancy (71%) and had an annual household income of >$50,000 (78%). Less than 1% of tea-drinking women reported consuming all three types of tea in either the first or third trimester, and 15% and 10% reported drinking two types of tea in the first or third trimester, respectively (data not shown). The prevalence of reporting green tea intake was lower in the third trimester, with no difference observed between the first and third trimesters for reported intake of regular or herbal tea.
Table 1. Characteristics of study participants (n=1954), by tea intake in the first and third trimesters, MIREC, Canada, 2009–2012.
|
Prevalence tea drinkers, n (%) |
|||||||
|---|---|---|---|---|---|---|---|
|
Regular |
Green |
Herbal |
|||||
| N (%) | First trimester | Third trimester | First trimester | Third trimester | First trimester | Third trimester | |
| Overall | 1954 | 290 (14.8) | 280 (14.8) | 118 (6.0) | 83 (4.4)a | 69 (3.5) | 56 (2.9) |
| Maternal age (years) | |||||||
| ⩽29 | 592 (30.3) | 94 (15.9) | 70 (11.8) | 35 (5.9) | 22 (3.7) | 12 (2.0)b | 14 (2.4) |
| 30–34 | 699 (35.8) | 94 (13.5) | 109 (15.6) | 37 (5.3) | 37 (5.3) | 19 (2.7) | 22 (3.2) |
| ⩾35 | 663 (33.9) | 102 (15.4) | 108 (16.3) | 46 (6.9) | 30 (4.5) | 38 (5.7)c | 20 (3.0) |
| Maternal country of birth | |||||||
| Canada | 1590 (81.4) | 231 (14.5) | 230 (14.5) | 79 (5.0)b | 58 (3.7)b | 53 (3.3) | 45 (2.8) |
| Other | 364 (18.6) | 59 (16.2) | 57 (15.7) | 39 (10.7)c | 31 (8.5)c | 16 (4.4) | 11 (3.0) |
| First pregnancy | |||||||
| No | 561 (28.7) | 75 (13.4) | 84 (15.0) | 34 (6.1) | 23 (4.1) | 22 (3.9) | 14 (2.5) |
| Yes | 1393 (71.3) | 215 (15.4) | 203 (14.6) | 84 (6.0) | 66 (4.7) | 47 (3.4) | 42 (3.0) |
| Marital status | |||||||
| Single, divorced or widowed | 91 (4.7) | 17 (18.7) | 10 (11.0) | 6 (6.6) | 2 (2.2) | 3 (3.3) | 1 (1.1) |
| Married or common law | 1863 (95.3) | 273 (14.7) | 277 (14.9) | 112 (6.0) | 87 (4.7) | 66 (3.5) | 55 (3.0) |
| Maternal education | |||||||
| College educated or less | 735 (37.6) | 111 (15.1) | 100 (13.6) | 41 (5.6) | 30 (4.1) | 21 (2.9) | 12 (1.6)b |
| Completed university | 716 (36.6) | 98 (13.7) | 101 (14.1) | 39 (5.5) | 29 (4.1) | 26 (3.6) | 17 (2.4) |
| Graduate university | 503 (25.7) | 81 (16.1) | 86 (17.1) | 38 (7.8) | 30 (6.0) | 22 (4.4) | 27 (5.4)c |
| Household income ($) | |||||||
| ⩽50,000 | 338 (18.1) | 56 (16.6) | 42 (12.4) | 20 (5.9) | 22 (6.5) | 10 (3.0) | 9 (2.7) |
| 50,001–100,000 | 778 (41.8) | 117 (15.0) | 117 (15.0) | 52 (6.7) | 33 (4.2) | 26 (3.3) | 21 (2.7) |
| >100,000 | 747 (40.1) | 104 (13.9) | 119 (15.9) | 43 (5.8) | 34 (4.6) | 31 (4.2) | 25 (3.4) |
| Maternal pre-pregnancy body mass index | |||||||
| <25 | 1150 (63.5) | 183 (15.9) | 171 (14.9) | 75 (6.5) | 70 (6.1)b | 46 (4.0) | 40 (3.5) |
| 25–29.9 | 396 (21.9) | 50 (12.6) | 62 (15.7) | 26 (6.6) | 8 (2.0)c | 12 (3.0) | 7 (1.8) |
| ⩾30 | 265 (14.6) | 42 (15.9) | 41 (15.5) | 11 (4.2) | 7 (2.6)c | 5 (1.9) | 5 (1.9) |
| Smoking during pregnancy | |||||||
| Daily or occasionally | 80 (4.2) | 20 (17.7) | 14 (12.4) | 4 (3.5) | 6 (7.5) | 3 (2.7) | 2 (2.5) |
| Never | 1818 (95.8) | 270 (14.7) | 274 (14.8) | 114 (6.2) | 83 (4.4) | 66 (3.6) | 54 (2.9) |
| Exposure to second hand smoke | |||||||
| Yes | 884 (45.2) | 136 (15.4) | 143 (16.2) | 74 (6.9) | 34 (3.9) | 33 (3.7) | 20 (3.4) |
| No | 1070 (54.8) | 154 (14.4) | 144 (13.5) | 44 (5.0) | 55 (5.1) | 36 (3.4) | 36 (2.3) |
| Alcohol during pregnancy | |||||||
| Never or <1/week | 1890 (96.7) | 280 (14.8) | 279 (14.8) | 112 (5.9) | 86 (4.6) | 65 (3.4) | 54 (2.9) |
| ⩾1/week | 64 (3.3) | 10 (15.6) | 8 (12.5) | 6 (9.4) | 3 (4.7) | 4 (6.3) | 2 (3.1) |
Abbreviations: BMI, body mass index; MIREC, Maternal-Infant Research on Environmental Chemicals. Missing data not shown, 7.3% of sample for maternal pre-pregnancy BMI.
Significantly different from tea intake at the first trimester visit, McNemar test (P<0.05).
Reference.
Significantly different from reference (P<0.05).
At the first trimester visit, a significantly higher proportion of green tea drinkers were not Canadian born, and this was also observed at the third trimester visit. Women ⩾35 years of age had a higher prevalence of herbal tea intake compared with those <29 years of age at the first trimester. At the third trimester visit, prevalence of herbal tea intake was higher for those with graduate level university education than for those who had a college education or lower. Those with a pre-pregnancy BMI of <25 had a higher prevalence of green tea intake than for those who were overweight or obese (BMI ⩾25).
Metals in Maternal and Cord Blood and Speciated Arsenic in Maternal Urine
Most maternal blood metal concentrations were above the LOD in the first trimester: lead (GM): 0.62 μg/dl (0.03 μmol/l), 100% detected), mercury (GM 2.99 nmol/l, 90% detected), cadmium (GM 1.93 nmol/l, 97% detected), arsenic (GM 9.75 nmol/l, 92% detected) and manganese (GM 160.1 nmol/l, 100% detected) (Table 2). With the exception of dimethylarsinic acid (GM 0.03 μmol/l; 86% detected), most of the speciated arsenic concentrations in maternal urine were not detected (trivalent arsenic 84% below LOD; pentavalent arsenic 98% monomethylarinic acid 92% and arsenobetaine and arsenocholine 58%). Manganese concentrations were higher in the third trimester (GM 222.6 nmol/l, 100% detected) compared with the first trimester (160.1 nmol/l, 100% detected), but all other metals tended to be lower in the third trimester. Cord blood concentrations were somewhat higher than maternal blood concentrations for lead, mercury and manganese, but lower for cadmium and arsenic (Table 2).
Table 2. Detection and geometric mean concentrations of metals in maternal whole blood, in the first and third trimesters, cord blood and maternal urine in the first trimester, by tea intake in the first or third trimester, in MIREC participants, Canada, 2009–2012.
|
Regular tea |
Green tea |
Herbal tea |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Yes | No | Yes | No | Yes | No | ||||
| N | %<LODa | Geometric mean | Geometric mean | Geometric mean | Geometric mean | ||||
| First trimester | |||||||||
| Lead (μg/dl) | 1910 | 0 | 0.62 | 0.65b | 0.61 | 0.75b | 0.61 | 0.77b | 0.61 |
| Mercury (nmol/l) | 1910 | 10 | 2.99 | 3.01 | 2.99 | 4.07b | 2.93 | 3.90 | 2.96 |
| Cadmium (nmol/l) | 1910 | 3 | 1.93 | 2.12b | 1.90 | 1.95 | 1.93 | 2.23 | 1.92 |
| Arsenic (nmol/l) | 1910 | 8 | 9.75 | 9.71 | 9.75 | 11.2b | 9.66 | 11.7 | 9.69 |
| Manganese (nmol/l) | 1910 | 0 | 160.1 | 162.9 | 159.7 | 152.6 | 160.6 | 159.8 | 160.2 |
| Speciated arsenic in urine (μmol/l, adjusted for specific gravity) | |||||||||
| Trivalent arsenic | 1895 | 84 | <0.005 | — | — | — | — | — | — |
| Pentavalent arsenic | 1904 | 98 | <0.005 | — | — | — | — | — | — |
| Monomethylarsinic acid | 1904 | 92 | <0.005 | — | — | — | — | — | — |
| Dimethylarsinic acid | 1904 | 14 | 0.03 | 0.03 | 0.03 | 0.05b | 0.03 | 0.04 | 0.03 |
| Arsenobetaine, arsenocholine | 1904 | 51 | <0.005 | — | — | — | — | — | — |
| Third trimester | |||||||||
| Lead (μg/dl) | 1673 | 2 | 0.57 | 0.57 | 0.57 | 0.66b | 0.57 | 0.70b | 0.57 |
| Mercury (nmol/l) | 1673 | 11 | 2.41 | 2.46 | 2.40 | 2.68 | 2.39 | 2.93 | 2.39 |
| Cadmium (nmol/l) | 1673 | 4 | 1.75 | 1.78 | 1.75 | 2.12 | 1.73 | 1.88 | 1.75 |
| Arsenic (nmol/l) | 1673 | 13 | 8.57 | 9.47 | 8.40 | 9.21 | 8.54 | 10.3 | 8.52 |
| Manganese (nmol/l) | 1673 | 0.1 | 222.6 | 225.3 | 222.1 | 232.5 | 222.1 | 216.0 | 222.8 |
| Cord blood | |||||||||
| Lead (μg/dl) | 1398 | 4 | 0.74 | 0.77 | 0.73 | 0.86b | 0.73 | 1.00b | 0.73 |
| Mercury (nmol/l) | 1398 | 18 | 3.06 | 3.10 | 3.06 | 4.18 | 3.02 | 4.88b | 3.02 |
| Cadmium (nmol/l) | 1399 | 11 | 0.58 | 0.57 | 0.58 | 0.60 | 0.58 | 0.54 | 0.58 |
| Arsenic (nmol/l) | 1397 | 2 | 8.03 | 9.00b | 7.86 | 9.44 | 7.96 | 10.1 | 7.97 |
| Manganese (nmol/l) | 1398 | 0 | 571.2 | 573.3 | 570.9 | 604.1 | 569.7 | 566.8 | 571.4 |
Abbreviations: LOD, Limits of detection; MIREC, Maternal-Infant Research on Environmental Chemicals.
Limits of detection: arsenic <3 nmol/l; cadmium <0.4 nmol/l; mercury <0.6 nmol/l; lead <0.1036 μg/dl (<0.005 μmol/l); manganese <10 nmol/l; for all speciated arsenic <0.01 μmol/l.
Significant difference (P<0.05).
Concentrations of Metals by Reported Tea Intake
Those reporting drinking regular tea at the first trimester had significantly higher GM blood lead concentrations (0.65 μg/dl) than those who did not drink regular tea (0.61 μg/dl) (Table 2). Higher blood lead was also observed for who reported green tea drinking (0.75 μg/dl) and herbal tea drinking (0.77 μg/dl) compared with those who did not drink these types of tea (0.61 μg/dl for both types of tea). The difference in blood lead concentrations between tea drinkers and non-drinkers persisted for green (0.66 and 0.57 μg/dl, respectively) and herbal tea (0.70 and 0.57 μg/dl, respectively) in the third trimester. Cord blood lead concentrations were also significantly different for third trimester green tea (0.86 μg/dl) and herbal tea (1.00 μg/dl) drinkers in comparison with non-tea drinkers (0.73 μg/dl for both types of tea).
A significant difference for mean cadmium concentrations between regular tea drinkers (2.12 nmol/l) and non-tea drinkers (1.90 nmol/l) was observed in the first trimester, but this did not persist into the third trimester nor was it observed in cord blood.
A significant difference between green tea drinkers and non-tea drinkers for GM blood mercury (4.07 and 2.93 nmol/l, respectively) and arsenic concentrations was observed (11.2 and 9.7 nmol/l, respectively), but this association was not observed in the third trimester or in cord blood. Arsenic concentrations were higher in cord blood for regular tea drinkers (9.0 nmol/l) compared with non-drinkers (7.9 nmol/l). Significant differences between green tea drinkers and non-tea drinkers were also observed in the first trimester for dimethylarsinic acid (0.05 and 0.03 nmol/l, respectively).
No significant associations with tea consumption were observed with blood manganese concentrations.
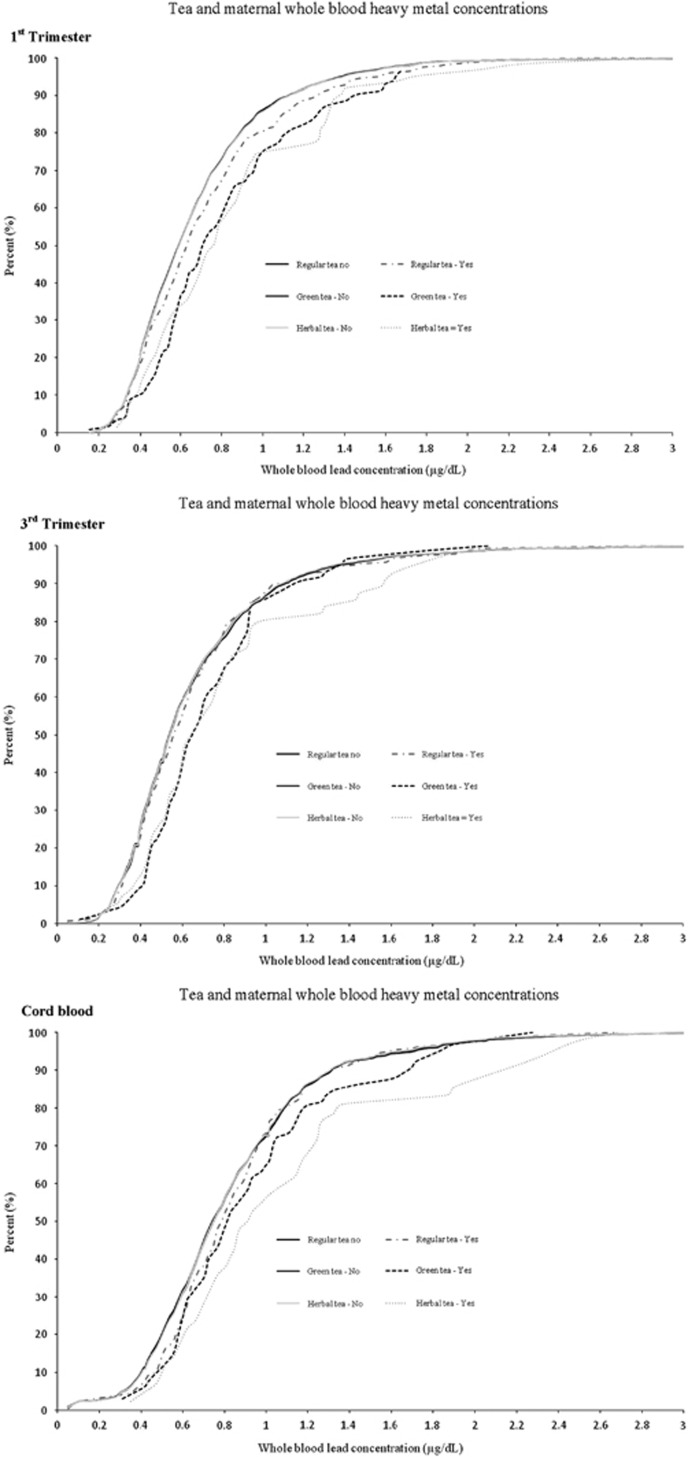
First trimester blood lead concentrations at the 95th percentile for regular, green and herbal tea drinkers were higher for tea drinkers (1.57, 1.65 and 1.76 μg/dl, respectively) than for those who were non-tea drinkers for each type (1.38 μg/dl) (Figure 1). However, 95th percentile blood lead concentrations were not consistently higher across tea drinker versus non-drinkers in the third trimester and cord blood (Figure 1).
Figure 1.
Cumulative distribution of maternal whole blood lead concentrations in first and third trimesters, and cord blood, by type of tea consumed in MIREC participants, Canada, 2009–2012. MIREC, Maternal-Infant Research on Environmental Chemicals.
Adjusted LSGMs for mercury, cadmium and arsenic in maternal — at the first or third trimester — and in cord blood were not significantly different for those who drank any type of tea versus non-tea drinkers (data not shown). Differences in maternal urinary dimethylarsinic acid for green tea drinkers, in comparison with non-green tea drinkers, were small but significant [green tea LSGM 0.03 nmol/l, 95%CI: 0.032, 0.035; non-green tea drinkers LSGM 0.04 nmol/l, 95% CI: 0.038, 0.049]. A comparison of first trimester blood lead concentrations and type of tea consumed in the first trimester demonstrated a significant difference between green tea, herbal tea or intake of any type of tea, versus non-drinkers (Table 3). An examination of the third trimester and cord blood results showed a significant difference for lead concentrations for herbal tea drinkers versus non-herbal tea drinkers. Increasing frequency of tea intake — for any type of tea — showed some evidence of a dose–response relationship between number of cups of tea per week and the adjusted LSGM for lead in the first trimester (none to <1 cup/week LSGM 0.61, 95% CI: 0.59, 0.62; 1 to <7 cups/week (LSGM 0.66, 95% CI: 0.61, 0.68); ⩾7 cups/week 0.67, 95% CI: 0.62, 0.74) (Table 4).
Table 3. Adjusted least squares geometric means of blood lead concentrations, by tea drinkers in the first or third trimester, in MIREC participants, Canada, 2009–2012.
|
Lead (μg/dl) |
|||
|---|---|---|---|
| LSGM | 95% CI | P-value | |
| First trimester blood concentration | |||
| Regular tea | |||
| None to <1 cup/week | 0.61 | 0.60, 0.63 | |
| Yes | 0.64 | 0.60, 0.67 | 0.24 |
| Green tea | |||
| None to <1 cup/week | 0.61 | 0.60, 0.62 | |
| Yes | 0.71 | 0.65, 0.78 | 0.0007 |
| Herbal tea | |||
| None to <1 cup/week | 0.62 | 0.60, 0.63 | |
| Yes | 0.71 | 0.63, 0.80 | 0.0195 |
| All tea | |||
| None to <1 cup/week | 0.61 | 0.59, 0.62 | |
| Yes | 0.65 | 0.62, 0.69 | 0.0044 |
| Third trimester blood concentration | |||
| Regular tea | |||
| None to <1 cup/week | 0.57 | 0.55, 0.58 | |
| Yes | 0.56 | 0.53, 0.59 | 0.6727 |
| Green tea | |||
| None to <1 cup/week | 0.57 | 0.55, 0.58 | |
| Yes | 0.61 | 0.55, 0.68 | 0.1958 |
| Herbal tea | |||
| None to <1 cup/week | 0.56 | 0.55, 0.58 | |
| Yes | 0.68 | 0.59, 0.78 | 0.0075 |
| All tea | |||
| None to <1 cup/week | 0.56 | 0.55, 0.58 | |
| Yes | 0.58 | 0.55, 0.62 | 0.2270 |
| Cord blood concentrationa | |||
| Regular tea | |||
| None to <1 cup/week | 0.74 | 0.72, 0.76 | |
| Yes | 0.76 | 0.71, 0.82 | 0.4468 |
| Green tea | |||
| None to <1 cup/week | 0.74 | 0.72, 0.76 | |
| Yes | 0.81 | 0.71, 0.93 | 0.2206 |
| Herbal tea | |||
| None to <1 cup/week | 0.74 | 0.72, 0.76 | |
| Yes | 0.92 | 0.77, 1.09 | 0.0144 |
| All tea | |||
| None to <1 cup/week | 0.74 | 0.72, 0.77 | |
| Yes | 0.75 | 0.70, 0.80 | 0.7923 |
Abbreviations: CI, confidence interval; LSGM, least squares geometric mean; MIREC, maternal-infant research on environmental chemicals. Each model controlled for maternal age, household income, highest level of maternal education, country of birth, maternal coffee intake, maternal smoking, exposure to second hand smoke and maternal pre-pregnancy body mass index.
Tea drinking in the third trimester.
Table 4. Adjusted least squares geometric means of blood lead concentrations, by frequency of tea intake in the first or third trimester, in MIREC participants, Canada, 2009–2012.
|
Lead (μg/dl) |
||||
|---|---|---|---|---|
| N (%) | LSGM | 95% CI | P-value | |
| First trimester blood concentration | ||||
| Regular tea (cupsa/week) | ||||
| None to <1 | 1626 (85.1) | 0.61 | 0.60, 0.63 | |
| 1 to <7 | 197 (10.3) | 0.62 | 0.58, 0.66 | 0.8768 |
| ⩾7 | 87 (4.6) | 0.68 | 0.61, 0.75 | 0.0587 |
| Green tea (cupsa/week) | ||||
| None to <1 | 1795 (94.0) | 0.61 | 0.60, 0.62 | |
| 1 to <7 | 101 (5.3) | 0.71 | 0.64, 0.77 | 0.0028 |
| ⩾7 | 14 (0.7) | 0.78 | 0.60, 0.98 | 0.0634 |
| Herbal tea (cupsa/week) | ||||
| None to <1 | 1847 (96.7) | 0.62 | 0.60, 0.63 | |
| 1 to <7 | 58 (3.0) | 0.72 | 0.64, 0.81 | 0.0132 |
| ⩾7 | 5 (0.3) | 0.52 | 0.33, 0.81 | 0.4450 |
| All tea (cupsa/week) | ||||
| None to <1 | 1511 (79.1) | 0.61 | 0.59, 0.62 | |
| 1 to <7 | 294 (15.4) | 0.65 | 0.61, 0.68 | 0.0323 |
| ⩾7 | 105 (5.5) | 0.67 | 0.62, 0.74 | 0.0287 |
| Third trimester blood concentration | ||||
| Regular tea (cupsa/week) | ||||
| None to <1 | 1398 (83.6) | 0.57 | 0.55, 0.58 | |
| 1 to <7 | 206 (12.3) | 0.56 | 0.52, 0.60 | 0.73 |
| ⩾7 | 69 (4.1) | 0.56 | 0.50, 0.63 | 0.79 |
| Green tea (cupsa/week) | ||||
| None to <1 | 1588 (94.9) | 0.57 | 0.55, 0.58 | |
| 1 to <7 | 74 (4.4) | 0.61 | 0.55, 0.69 | 0.17 |
| ⩾7 | 11 (0.7) | 0.57 | 0.43, 0.77 | 0.92 |
| Herbal tea (cupsa/week) | ||||
| None to <1 | 1617 (96.7) | 0.56 | 0.55, 0.58 | |
| 1 to <7 | 48 (2.9) | 0.71 | 0.61, 0.82 | 0.002 |
| ⩾7 | 8 (0.5) | 0.51 | 0.36, 0.74 | 0.5987 |
| All tea (cupsa/week) | ||||
| None to <1 | 1306 (78.1) | 0.56 | 0.55, 0.58 | |
| 1 to <7 | 281 (16.8) | 0.58 | 0.55, 0.62 | 0.35 |
| ⩾7 | 86 (5.1) | 0.56 | 0.50, 0.62 | 0.91 |
| Cord blood concentration | ||||
| Regular tea (cupsa/week) | ||||
| None to <1 | 1182 (84.6) | 0.74 | 0.72, 0.76 | |
| 1 to <7 | 162 (11.6) | 0.75 | 0.69, 0.81 | 0.88 |
| ⩾7 | 54 (3.9) | 0.82 | 0.71, 0.96 | 0.17 |
| Green tea (cupsa/week) | ||||
| None to <1 | 1333 (95.4) | 0.74 | 0.72, 0.76 | |
| 1 to <7 | 57 (4.1) | 0.82 | 0.70, 0.94 | 0.23 |
| ⩾7 | 8 (0.6) | 0.79 | 0.54, 0.87 | 0.74 |
| Herbal tea (cupsa/week) | ||||
| None to <1 | 1356 (97.0) | 0.74 | 0.72, 0.76 | |
| 1 to <7 | 38 (2.7) | 0.92 | 0.77, 1.10 | 0.0171 |
| ⩾7 | 4 (0.3) | 0.89 | 0.48, 1.63 | 0.5655 |
| All tea (cupsa/week) | ||||
| None to <1 | 1112 (79.5) | 0.73 | 0.71, 0.76 | |
| 1 to <7 | 220 (15.7) | 0.77 | 0.72, 0.83 | 0.24 |
| ⩾7 | 66 (4.7) | 0.82 | 0.72, 0.94 | 0.11 |
Abbreviations: CI, confidence interval; LSGM, least squares geometric mean; MIREC, maternal-infant research on environmental chemicals. Each model controlled for age, household income, highest level of maternal education, country of birth, maternal smoking, maternal coffee intake, exposure to second hand smoke and maternal pre-pregnancy body mass index
Defined in questionnaire as 6 ounces.
DISCUSSION
This study provides new insight into possible associations between tea intake during pregnancy and blood metal concentrations. Adjusted mean blood concentrations of mercury, cadmium, arsenic and manganese — with the exception of dimethylarsinic acid — were similar for any type of tea drinkers and non-tea drinkers. However, adjusted mean blood lead concentrations were higher among those who reported consuming tea, and more specifically green or herbal tea, in the first trimester, even after adjusting for several factors, including smoking and coffee intake.
The positive association between tea intake and blood lead concentrations observed in MIREC participants is inconsistent with those from the Avon Longitudinal Study of Parents and Children (ALSPAC), a British study of over 14,000 parents and their offspring (1991–1992), which did not find that tea intake was independently associated with first trimester blood lead concentrations (GM 3.43 μg/dl).18 However, in the ALSPAC, caffeinated coffee was a stronger predictor of blood lead levels than decaffeinated coffee when the model was adjusted for tea consumption. There are several possible explanations for this disparity, including the following: the tea sold in the United Kingdom during that time period may have differed in lead content from that in Canada (e.g., increase in industrialization in tea growing regions), variance in the frequency of tea intake in the two populations, or dissimilar predictors because of the higher blood lead concentrations in the ALSPAC cohort (GM 3.67 μg/dl) as compared with MIREC women (GM 0.62 μg/dl).
A blood lead concentration considered as safe during pregnancy has not been established, though the American Congress of Obstetricians and Gynecologists recommends blood lead testing when any risk factor is identified, and follow-up testing if results exceed 5 μg/dl.19 Infants and children are undergoing rapid neurodevelopment, and absorb lead more easily and excrete lead less efficiently than adults; thus, sensitive adverse health end points in this sub-group have been associated with blood lead levels as low as >1.3 μg/dl.20 The GM blood lead concentrations of the MIREC population (tea and non-tea drinkers) were below these thresholds and were lower than for females 20–39 years of age in the 2007–2009 Canadian Health Measures Survey (CHMS) (MIREC: 0.62 μg/dl vs CHMS: 0.89 μg/dl).21 Though lead mobilization from bone, increased gut absorption and lead retention in soft tissue may result in higher concentrations as pregnancy progresses, the hemodilution that occurs in pregnancy may be partly responsible for the lower lead concentrations in in the MIREC cohort in comparison with non-pregnant women in the CHMS study.22
Tea is produced from the Camellia sinensis, plant of the Theaceae family. Different processing methods are used to make different types of tea, including green tea (non-oxidized, non-fermented) and black tea (fully-oxidized, fermented). Herbal tea is commonly made from dried herbs, flowers or roots, and may not contain leaves from the Camellia sinensis. The soil where plants are grown may be exposed to metals through the application of fertilizers and pesticides, which are then available for uptake by the plant.23 Detectable levels of certain metals, including lead, arsenic and cadmium, have been observed in dried tea. For example, the Canadian Food Inspection Agency (CFIA) tested 100 samples of dried tea (including black, green and herbal) and found that metals, including manganese, lead, cadmium and arsenic, were detected in 100% of samples, at varying levels.23 Further, green tea had higher mean lead amounts (1.31 p.p.m.) than herbal tea (1.14 p.p.m.) and black tea (0.64 p.p.m.).23 These dried tea samples do not indicate the amount of lead in prepared tea, as steeping time impacts the transfer of contaminants. For example, in a Canadian study, convenience samples of 30 steeped black, green, white and oolong tea were examined9 to determine the presence of metals; the study revealed that lead and arsenic were detected in all dried tea leaves and steeped tea, with Chinese oolong teas having the highest levels, followed by green tea and regular black tea. Though the study accounted for the amount of lead in the water used for preparation, the mean amount found in steeped tea — standard oolong tea with a steeping time of 3-4 min (3.05 μg/l) and 15–17 min (3.45 μg/l) — was below 10 μg/l, the current maximum acceptable concentration of lead in Canadian drinking water.24 Although mercury was detected in the dry tea leaves, none of the brewed teas had detectable levels, suggesting that mercury is bound in the leaf and does not leach into the brewed tea. Cadmium was measured in all dry tea leaves with some transfer to brewed tea; and the highest levels of manganese were found in black tea. The world's top tea producing countries are China (1.94 million tonnes) and India (1.21 million tonnes), followed by Kenya (430,000 tonnes) and Sri Lanka (340,000 tonnes).25 In highly industrialized countries, tea leaves may be exposed to higher lead in the soil. For example, in a systematic review of Chinese surveillance data gathered from 2006 to 2012, it was determined that dried tea had the highest lead concentration in the food samples studied, with an estimated weighted mean concentration of 1.937 mg/kg.10
The MIREC cohort has multiple strengths, including the large, national-level prenatal population with biomonitoring data on metal exposures at multiple time points in pregnant women. Further, chemical analyses for MIREC and the CHMS were conducted at the same laboratory, which minimizes potential inter-laboratory variation. As MIREC is not population based, participants differed from the Canadian population giving birth in 2009; for example, our population tended to be older, more educated and less likely to be a current smoker. Thus, our results may not be generalizable to the Canadian population.16 Though measured blood metal concentrations were available in early and late pregnancy, concentrations across time points were not compared as we could not control for hemodilution during pregnancy. Our sample included mostly low-to-moderate tea drinkers, which indicates the need for future studies to examine the association between blood lead concentrations in heavy tea drinkers. Further, other than general tea categories (e.g., regular and green) we did not collect specific data on the source of the tea consumed, such as country of origin. Studies indicate that the transfer of lead from dried to liquid tea increases with length of steeping time — though the amount transferred may be modest — and we did not have this information for the MIREC participants. A study examining the metal content of tea by type, country of origin and steeping time in relation to metal biomarkers may provide further insight needed to formulate appropriate guidance for pregnant women. This study suggests that green and herbal tea drinkers may have higher blood lead concentrations than non-drinkers. However, other environmental exposures that may contribute to this increase in blood lead levels and be correlated with tea intake should be investigated in future studies.
This study provides novel insight into tea intake as a potential route of exposure for metals in Canada. Lead was the only metal found to be related to intake of green and herbal tea, after adjusting for potential confounders. However, the GM blood lead concentrations of the highest tea consumers were still less than 1 μg/dl, thus within the normal range of blood lead concentrations and lower than those in a representative sample of similarly aged non-pregnant Canadian women. Thus, the potential for higher blood lead levels through low-to-moderate tea consumption does not appear to increase blood lead levels to those associated with health concerns. As there are many potential sources of lead exposure in the environment, such as contaminated drinking water and house dust which were not considered in this study, our findings should be interpreted cautiously. Further investigation of this association is warranted.
Acknowledgments
The MIREC Study was funded by Health Canada's Chemicals Management Plan, the Canadian Institute of Health Research (CIHR grant # MOP-81285) and the Ontario Ministry of the Environment. CK Colapinto was supported by a CIHR-Quebec Training Network in Perinatal Research (QTNPR) Post-doctoral fellowship.
The authors declare no conflict of interest.
References
- Liu J, Chen Y, Gao D, Jing J, Hu Q. Prenatal and postnatal lead exposure and cognitive development of infants followed over the first three years of life: A prospective birth study in the Pearl River Delta Region, China. Neurotoxicology 2014; 44: 326–334. [DOI] [PubMed] [Google Scholar]
- Chung SE, Cheong HK, Ha EH, Kim BN, Ha M, Kim Y et al. Maternal blood manganese and early neurodevelopment: The Mothers and Children's Environmental Health (MOCEH) study. Environ Health Perspect 2015; 123: 717–722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kippler M, Tofail F, Gardner R, Rahman A, Hamadani JD, Bottai M et al. Maternal cadmium exposure during pregnancy and size at birth: a prospective cohort study. Environ Health Perspect 2012; 120: 284–289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu J, Ying T, Shen Z, Wang H. Effect of low-level prenatal mercury exposure on neonate neurobehavioral development in China. Pediatr Neurol 2014; 51: 93–99. [DOI] [PubMed] [Google Scholar]
- Xie X, Ding G, Cui C, Chen L, Gao Y, Zhou Y et al. The effects of low-level prenatal lead exposure on birth outcomes. Environ Pollut 2013; 175: 30–34. [DOI] [PubMed] [Google Scholar]
- Zhu M, Fitzgerald EF, Gelberg KH, Lin S, Druschel CM. Maternal low-level lead exposure and fetal growth. Environ Health Perspect 2010; 118: 1471–1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews KW, Savitz DA, Hertz-Picciotto I. Prenatal lead exposure in relation to gestational age and birth weight: A review of epidemiologic studies. Am J Ind Med 1994; 26: 13–32. [DOI] [PubMed] [Google Scholar]
- Tchounwou PB, Yedjou CG, Patlolla AK, Sutton DJ. Heavy metal toxicity and the environment. EXS 2012; 101: 133–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwalfenberg G, Genuis SJ, Rodushkin I. The benefits and risks of consuming brewed tea: beware of toxic element contamination. J Toxicol 2013; 2013: 370460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin Y, Liu P, Wu Y, Min J, Wang C, Sun J et al. A systematic review on food lead concentration and dietary lead exposure in China. Chin Med J (Engl) 2014; 127: 2844–2849. [PubMed] [Google Scholar]
- Falahi E, Hedaiati R. Heavy metal content of black teas consumed in Iran. Food Addit Contam Part B Surveill 2013; 6: 123–126. [DOI] [PubMed] [Google Scholar]
- Karak T, Bhagat RM. Trace elements in tea leaves, made tea and tea infusion: a review. Food Res Int 2010; 43: 2234–2252. [Google Scholar]
- Canadian Prenatal Nutrition Guidelines: Caffeine. Available from http://www.phac-aspc.gc.ca/hp-gs/know-savoir/caffeine-eng.php. Updated 2014.
- Chen L, Bell EM, Browne ML, Druschel CM, Romitti PA. National Birth Defects Prevention Study. Exploring maternal patterns of dietary caffeine consumption before conception and during pregnancy. Matern Child Health J 2014; 18: 2446–2455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei SQ, Xu H, Xiong X, Luo ZC, Audibert F, Fraser WD. Tea consumption during pregnancy and the risk of pre-eclampsia. Int J Gynaecol Obstet 2009; 105: 123–126. [DOI] [PubMed] [Google Scholar]
- Arbuckle TE, Fraser WD, Fisher M, Davis K, Liang CL, Lupien N et al. Cohort profile: The Maternal-Infant Research on Environmental Chemicals research platform. Paediatr Perinat Epidemiol 2013; 27: 415–425. [DOI] [PubMed] [Google Scholar]
- Ashley-Martin J, Dodds L, Arbuckle TE, Levy AR, Platt RW, Marshall JS. Predictors of interleukin-33 and thymic stromal lymphopoietin levels in cord blood. Pediatr Allergy Immunol 2015; 26: 161–167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor CM, Golding J, Hibbeln J, Emond AM. Environmental factors predicting blood lead levels in pregnant women in the UK: the ALSPAC study. PLoS One 2013; 8: e72371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Committee on Obstetric Practice. Committee opinion no. 533: Lead screening during pregnancy and lactation. Obstet Gynecol 2012; 120: 416–420. [DOI] [PubMed] [Google Scholar]
- Health Canada. Final human health state of the science report on lead 2013. Available from: http://www.hc-sc.gc.ca/ewh-semt/pubs/contaminants/dhhssrl-rpecscepsh/index-eng.php (Accessed 15 September 2015).
- Bushnik T, Haines D, Levallois P, Levesque J, Van Oostdam J, Viau C. Lead and bisphenol A concentrations in the Canadian population. Health Rep 2010; 21: 7–18. [PubMed] [Google Scholar]
- Manton WI, Angle CR, Stanek KL, Kuntzelman D, Reese YR, Kuehnemann TJ. Release of lead from bone in pregnancy and lactation. Environ Res 2003; 92: 139–151. [DOI] [PubMed] [Google Scholar]
- Canadian Food Inspection Agency. Pesticide residues and metals in dried tea. Available from http://www.inspection.gc.ca/food/chemical-residues-microbiology/chemical-residues/dried-tea/eng/1348263667494/1348263826430. Updated 2014. Accessed 2 May 2015.
- Health Canada. Guidelines for Canadian drinking water quality. Available from http://www.hc-sc.gc.ca/ewh-semt/pubs/water-eau/sum_guide-res_recom/index-eng.php#t2. Updated 2014. Accessed 10 June 2015.
- FAOSTAT: Production quantities of tea by country, 2013. Available from http://faostat3.fao.org/browse/Q/QC/E. Updated 2015.