Abstract
The aim of the study was to investigate the resistant mechanisms and homology of imipenem-resistant Acinetobacter baumannii (A. baumannii). A total of 46 non-duplicate imipenem-resistant A. baumannii clinical isolates were collected from three tertiary hospitals between July, 2011 and June, 2012. The minimal inhibitory concentrations (MICs) of antimicrobial agents were determined using the agar dilution method. Phenylalanine-arginine β-naphthylamide was used to detect the presence of the efflux pump-mediated resistant mechanism. Polymerase chain reaction was employed to amplify genes associated with drug resistance, including β-lactamase genes, efflux pump genes and outer membrane protein gene CarO. A few amplicons were randomly selected and sequenced. Multilocus sequence analysis (MLST) was employed in typing A. baumanni. A. baumannii was resistant to imipenem, simultaneously showing resistance to several other antimicrobials. In addition, 13 A. baumannii were found to mediate drug resistance through operation of the efflux pump. Of the various drug resistance genes tested, blaOXA-51 was present in 46 isolates, blaOXA-23 gene was present in 44 isolates and blaNDM gene was found in only one strain. Other drug resistant-associated genes, including blaKPC, blaIMP, blaOXA-24, blaOXA-58, blaSHV, blaGIM and blaVIM were not detected. Mutation of adeS and outer membrane protein gene CarO were found in a few of the imipenem-resistant isolates. The MLST analysis revealed that all 46 clinical isolates were clustered into 11 genotypes and the most frequent genotype was ST208. In conclusion, β-lactamase genes, genes involved in efflux pump and mutation of outer membrane protein encoding gene may be important in mediating imipenem resistance in A. baumannii. Of the 11 different genotypes, ST11 was shared by the majority of A. baumannii, which may be due to horizontal transfer of patients from hospitals.
Keywords: Acinetobacter baumannii, imipenem, efflux pump, minimal inhibitory concentration, multilocus sequence analysis, molecular epidemiology
Introduction
Acinetobacter baumannii (A. baumannii) has emerged as a major pathogen of nosocomial infections and is associated with high rates of morbidity and mortality in recent years (1,2). A nationwide surveillance program, including hospitals from 14 geographically different regions in China revealed that the ratio of A. baumannii is on the increase annually (3).
Carbapenem has good antibacterial activity against A. baumannii and was the first choice in treatment of infection caused by A. baumannii in the past years (4). However, the emergence of resistance to carbapenem was reported in 1991 (5), followed by similar reports from different parts of the world (6,7). In China, 57 and 61% of Acinetobacter spp. (A. baumannii accounted for 89.6%) showed resistance to imipenem and meropenem, respectively (3). International studies in China as well as in other parts of the world focused only on evaluating the resistance of A. baumannii to various antimicrobials (8–10). However, to the best of our knowledge, few studies have investigated the molecular mechanism underlying drug resistance. Additionally, no data are available on the epidemiological characteristics of imipenem-resistant A. baumannii in Shanghai.
Thus, A. baumannii clinical isolates were collected from three tertiary hospitals in Shanghai and their drug resistance pattern to a spectrum of antimicrobials, molecular mechanisms (including carbapenemase, efflux pumps and membrane proteins) behind their resistance and multilocus sequence analysis (MLST) were analyzed to assess their molecular epidemiology.
Materials and methods
Bacterial strains
During the period July, 2011 to June, 2012, 46 non-duplicate imipenem-resistant A. baumannii strains were collected from three tertiary hospitals located in Shanghai, China.
Reconfirmation of strains
The collected strains were subjected to gram staining, biochemical tests, and recA gene and 16S-23S rRNA gene intergenic spacer region to reconfirm them as A. baumannii (7).
Antimicrobial susceptibility and efflux phenotype tests
The collected A. baumannii isolates were subjected to an antimicrobial susceptibility test against imipenem, meropenem, amikacin, piperacillin, ceftazidime, cefotaxime, mino-cycline, ciprofloxacin, ampicillin/sulbactam, sulbactam, cefoperazone/sulbactam, piperacillin/tazobactam, colistin, tigecycline and trimethoprim/sulfamethoxazole using agar dilution method. Escherichia coli strain ATCC25922 and Pseudomonas aeruginosa (P. aeruginosa) strain ATCC27853 were used as reference strains.
Strains in which efflux pump operation was detected by agar dilution method where imipenem- and meropenem-resistant isolates were cultured in Mueller-Hinton agar contained the efflux pump inhibitor phenylalanine-arginine β-naphthylamide (PAβN) at a final concentration of 20 mg/l (11,12). A ≥4-fold reduction of imipenem or meropenem minimal inhibitory concentrations (MICs) in the presence of PAβN possessed an operating drug efflux pump.
Analysis of genes responsible for drug resistance, drug efflux and outer membrane protein
Polymerase chain reaction (PCR) was performed for the genes, blaKPC, blaIMP, blaNDM, blaOXA-51, blaOXA-23, blaOXA-24, blaOXA-58, blaSHV, blaGIM and blaVIM, CarO, adeA, adeB, adeC, adeS and adeR. Thus, obtained amplicons were subjected to sequencing analysis.
A fresh and pure bacterial colony was suspended in distilled water and boiled at 100°C for 15 min. After centrifu-gation at 8,000 × g for 15 min, 1 µl of the supernatant was used for PCR analysis with the primers (Table I). PCR was performed in a total volume of 50 µl containing 0.25 µl Taq DNA polymerase (Takara Bio, Inc., Tokyo, Japan), 5 µl 10X PCR buffer (Mg2+ Plus), 4 µl dNTP mixture (2.5 mM each), 2.5 µl DNA template, 1 µl of each primer (20 µM), and 36.25 µl ddH2O. The PCR thermal cycle consisted of initial denaturation at 94°C for 5 min, followed by 30 cycles of 94°C for 30 sec, annealing 55°C for 1 min and 72°C for 1 min and a final extension at 72°C for 7 min. The PCR products were electrophoresed in 1% agarose gel and visualized under ultraviolet light, and subsequently sequenced (Sangon Biotech Co., Ltd., Shanghai, China).
Table I.
Gene-specific primers used in this study.
| Genes | Primer sequences |
|---|---|
| recA | F: CCTGAATCTTCTGGTAAAAC R: GTTTCTGGGCTGCCAAACATTAC |
| ITS | F: CATTATCACGGTAATTAGTG R: AGAGCACTGTGCACTTAAG |
| blaKPC | F: TTACTGCCCGTTGACGCCCAATCC R: TCGCTAAACTCGAACAGG |
| blaIMP | F: AACCAGTTTTGCCTTACCAT R: CTACCGCAGCAGAGTCTTTG |
| blaNDM | F: CCGCCCAGATCCTCAACT R: ATCAGGCAGCCACCAAAA |
| blaOXA-51 | F: TAATGCTTTGATCGGCCTTG R: TGGATTGCACTTCATCTTGG |
| blaOXA-23 | F: GATCGGATTGGAGAACCAGA R: ATTTCTGACCCATTTCCAT |
| blaOXA-24 | F: GGTTAGTTGGCCCCCTTAAA R: AGTTGAGCGAAAAGGGGATT |
| blaOXA-58 | F: AAGTATTGGGGCTTGTGCTG R: CCCCTCTGCGCTCTACATAC |
| blaSHV | F: GGTTATGCGTTATATTCGCC R: TTAGCGTTGCCAGTGCTC |
| blaGIM | F: AGAACCTTGACCGAACGCAG R: ACTCATGACTCCTCACGAGG |
| blaVIM | F: TCCACGCACTTTCATGACGA R: AGACGTGCGTGACAACTCAT |
| adeA | F: GAAATCCGTCCGCAAGTC R: ACACGCACATACATACCC |
| adeB | F: AAAGACTTCAAAGAGCGG R: TCACGCATTGCTTCACCC |
| adeC | F: ATTTCAGGTCGTAGCATT R: CTTGATAAGTAGAGTAGGGATT |
| adeS | F: ACTGTTATCTTCTGTGGCTGTA R: GTGGACGTTAGGTCAAGTTCTG |
| adeR | F: AAACGGTTGGGAAGTATTA R: ATGGCTATCTACGGTTCG |
| CarO | F: AAGGAGAAAACGATGA R: TTATTACGTGGTTATGG |
| gltA | F: AATTTACAGTGGCACATTAGGTCCC R: GCAGAGATACCAGCAGAGATACACG |
| gyrB | F: TGAAGGCGGCTTATCTGAGT R: GCTGGGTCTTTTTCCTGACA |
| gdhB | F: ACCACATGCTTTGTTATG R: GTTGGCGTATGTTGTGC |
| recA | F: CCTGAATCTTCYGGTAAAAC R: GTTTCTGGGCTGCCAAACATTAC |
| cpn60 | F: GGTGCTCAACTTGTTCGTGA R: CACCGAAACCAGGAGCTTTA |
| Gpi | F: GAAATTTCCGGAGCTCACAA R: TCAGGAGCAATACCCCACTC |
| rpoD | F: ACCCGTGAAGGTGAAATCAG R: TTCAGCTGGAGCTTTAGCAAT |
MLST
Seven housekeeping genes including homologous recombination factor (recA), citrate synthase (gltA), DNA gyrase subunit (gyrB), glucose-6-phosphate isomerase isomerase (gpi), glucose dehydrogenase B (gdhB), 60-kDa chaperonin (cpn60), and RNA polymerase 70 factor (rpoD) were amplified in PCR using relevant primers (Table I) and appropriate thermal conditions. The amplicons were sequenced and the sequences were submitted to the MLST database (http://pubmlst.org.net) to compare them with sequences submitted from other parts of the world. Each strain was then characterized by a pattern of numbers defining its allelic profile.
Results
Antimicrobial susceptibility
A. baumannii resistant to imipenem simultaneously showed resistance to several other common antimicrobials. The resistance rate was >80% for all the antimicrobials except minocycline and colistin. Antibiotic susceptibility of the 46 clinical isolates is shown in Table II. Thirteen imipenem-resistant A. baumannii isolates were positive for efflux pump.
Table II.
The drug-resistant rates of imipenem-resistant Acinetobacter baumannii.
| Drug | Resistance rate No. of resistant strains (%) |
|---|---|
| Meropenem | 41 (89) |
| Amikacin | 38 (83) |
| Piperacillin | 46 (100) |
| Ceftazidime | 46 (100) |
| Minocycline | 34 (74) |
| Ciprofloxacin | 45 (98) |
| Ampicillin/sulbactam | 43 (93) |
| Piperacillin/tazobactam | 46 (100) |
| Colistin | 1 (2) |
| Trimethoprim/sulfamethoxazole | 43 (93) |
| Cefotaxime | 45 (98) |
Detection of genes involved in drug resistance, drug efflux and outer membrane protein
Of the various drug resistance genes tested, blaOXA-51 was present in 46 isolates, blaOXA-23 gene was present in 44 isolates and blaNDM gene was found in only one strain. Other drug-resistant genes including blaKPC, blaIMP, blaOXA-24, blaOXA-58, blaSHV, blaGIM and blaVIM were not detected in the isolates.
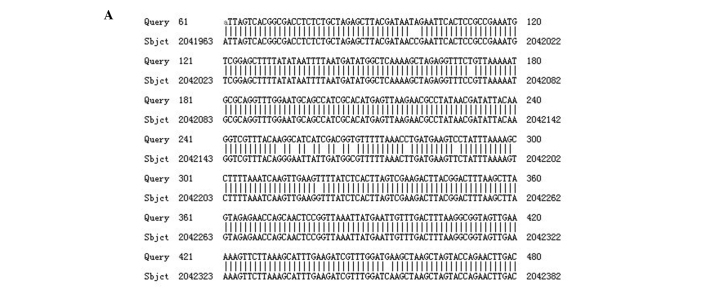
Of the five genes associated with the drug efflux pump tested, all five were found to be present in the isolates. Several mutations were found in the sequences of adeS gene in isolates with efflux phenotype. Differences were observed at three places when nucleotide sequences were translated into an amino acid sequence. This amino acid sequence was then compared to the amino acid sequence of the reference strain ATCC17978 (Fig. 1).
Figure 1.
(A) Comparison of nucleotide sequence of adeS gene between resistant strain and reference strain ATCC17978. (B) Comparison of amino acid sequence of adeS gene between resistant strain and reference strain ATCC17978.
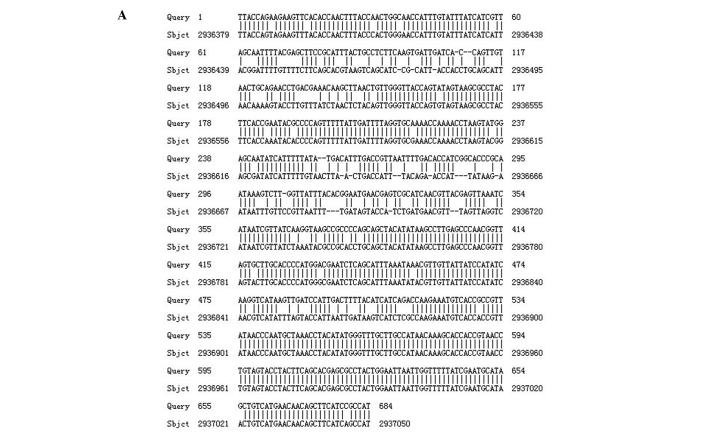
Similarly, the nucleotide sequence of the outer membrane protein encoding gene CarO, when compared with the nucleotide sequence of reference strain ATCC17978, harbored mutations that were reflected in the amino acid sequence (Fig. 2).
Figure 2.
(A) Comparison of nucleotide sequence of CarO gene between resistant and reference strain ATCC17978. (B) Comparison of amino acid sequence of CarO gene between resistant and reference strain ATCC17978.
Genotyping of isolates by MLST
The MLST analysis revealed that the isolates were clustered in 11 different genotypes or STs. The ST208 genotype was shared by the majority of isolates (58.7%, 27/46), followed by ST191 (10.9%, 5/46) and ST451 (6.5%, 3/46). We also detected some other STs shared by certain isolates such as ST75 (2.1%, 1/46), ST90 (4.2%, 2/46), ST92 (2.1%, 1/46), ST108 (2.1%, 31/46), ST109 (2.1%, 1/46), ST172 (2.1%, 1/46), ST368 (4.2%, 2/46) and ST69 (4.2%, 2/46). These STs were grouped into the three clonal complexes, CC92, CC109 and CC28.
Discussion
A. baumannii develops resistance to imipenem through a variety of mechanisms. Carbapenemase is an important factor responsible for imipenem resistance. In the present study, common carbapenemases were detected in the isolates, including blaOXA-51, blaOXA-23, blaOXA-24, blaOXA-58, blaKPC, blaIMP, blaSHV, blaGIM, blaNDM and blaVIM. OXA-type enzymes are naturally present in Acinetobacter spp. and are usually expressed in small amounts (13). The expression of such genes is markedly higher under the effect of a strong promoter (insertion sequence ISAba1 is the most shared) and induce drug resistance only when combined with a reduction in outer membrane permeability and/or activation of the efflux pump (14). In the present study, blaOXA-51 and blaOXA-23 genes were prevalent among the isolates, results that are consistent with other reports (15–17). Carbapenemases that are different from OXA, such as KPC, IMP, SHV, GIM, NDM and VIM have strong carbapenem-hydrolysing activity (14). However, such types of carbapenemases were rarely detected in A. baumannii. The blaNDM gene was identified in only one strain while the remaining resistant genes were not detected. NDM was first identified in Escherichia coli and Klebsiella pneumoniae in 2008 in India (18). This finding was followed by reports on NDM-producing P. aeruginosa, Enterobacter cloacae, Citrobacter freundii and Enterococcus faecium (19–23). In China, NDM-producing A. baumannii was first reported in 2011. Of the anti microbials tested one NDM-positive isolate in the present study was identified that was multidrug-resistant, and only susceptible to amikacin, colistin and minocycline.
Drug efflux systems including AdeABC, AdeIJK, AdeDE and AdeXYZ (RND family) have been found in A. baumannii. Of these, the AdeABC efflux system is common in A. baumannii (24). This efflux pump, together with other resistant mechanisms, can lead to high-level imipenem resistance. Although mediated by the substrate, its expression may increase when a single point mutation occurs in the adeR or adeS gene (25). PAβN was proven to be an effective inhibitor of drug efflux. In the present study, adeA, adeB and adeC were present in all of the isolates because when PAβN was added the MICs inherent to imipenem in 13 isolates were decreased. The adeS gene differed from the adeS of standard strain and this is the possible reason for increased drug efflux associated with drug resistance.
Few studies concerning the impact of changes on membrane proteins in A. baumannii are available. In 2002, a laboratory in Argentina advocated for the first time that inducible resistance by imipenem can trigger loss of a 29-kDa membrane protein. In 2005, the same laboratory furthering their study, demonstrated that the outer membrane protein is encoded by the CarO gene and when there is an insertion mutation or any other mutation in the CarO gene makes it off and thus the strain become resistant to certain drugs (26). In the present study, the sequence of CarO gene had nucleotide insertions, deletions and point mutations in comparison with the standard strains and there were also differences in their nucleotide and amino acid sequences.
In summary, for a global epidemiologic analysis, a comparison of the results between different laboratories is required. MLST is a powerful tool used to transfer typing data and compare results via relevant databases. The MLST analysis revealed that the major epidemic clone of A. baumannii in Shanghai was ST208 (CC92 clone complex), which differed from the results obtained in other regions in China (27).
Acknowledgments
The project was supported by a grant from the Natural Science Foundation of Shanghai Science and Technology Committee (no. 12ZR1426200), the Medical Guide Program of Shanghai Science and Technology Committee (no. 14411962900), Key project of Shanghai Municipal Health and Family Planning Commission (no. 201540367) and Central Universities Basic Research Program (no. 1511219024).
References
- 1.Perez F, Hujer AM, Hujer KM, Decker BK, Rather PN, Bonomo RA. Global challenge of multidrug-resistant Acinetobacter baumannii. Antimicrob Agents Chemother. 2007;51:3471–3484. doi: 10.1128/AAC.01464-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Giamarellou H, Antoniadou A, Kanellakopoulou K. Acinetobacter baumannii: A universal threat to public health? Int J Antimicrob Agents. 2008;32:106–119. doi: 10.1016/j.ijantimicag.2008.02.013. [DOI] [PubMed] [Google Scholar]
- 3.Wang F, Zhu D, Hu F, Jiang X, Hu Z, Li Q, Sun Z, Chen Z, Xu Y, Zhang X, et al. CHINET 2012 surveillance of bacterial resistance in China. China J Infect Chemother. 2013;13:321–330. Chinese. [Google Scholar]
- 4.Kim YJ, Kim SI, Hong KW, Kim YR, Park YJ, Kang MW. Risk factors for mortality in patients with carbapenem-resistant Acinetobacter baumannii bacteremia: Impact of appropriate antimicrobial therapy. J Korean Med Sci. 2012;27:471–475. doi: 10.3346/jkms.2012.27.5.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Go ES, Urban C, Burns J, Kreiswirth B, Eisner W, Mariano N, Mosinka-Snipas K, Rahal JJ. Clinical and molecular epidemiology of acinetobacter infections sensitive only to polymyxin B and sulbactam. Lancet. 1994;344:1329–1332. doi: 10.1016/S0140-6736(94)90694-7. [DOI] [PubMed] [Google Scholar]
- 6.Pournaras S, Markogiannakis A, Ikonomidis A, Kondyli L, Bethimouti K, Maniatis AN, Legakis NJ, Tsakris A. Outbreak of multiple clones of imipenem-resistant Acinetobacter baumannii isolates expressing OXA-58 carbapenemase in an intensive care unit. J Antimicrob Chemother. 2006;57:557–561. doi: 10.1093/jac/dkl004. [DOI] [PubMed] [Google Scholar]
- 7.Kuo LC, Teng LJ, Yu CJ, Ho SW, Hsueh PR. Dissemination of a clone of unusual phenotype of pandrug-resistant Acinetobacter baumannii at a university hospital in Taiwan. J Clin Microbiol. 2004;42:1759–1763. doi: 10.1128/JCM.42.4.1759-1763.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Xu J, Sun Z, Li Y, Zhou Q. Surveillance and correlation of antibiotic consumption and resistance of Acinetobacter baumannii complex in a tertiary care hospital in northeast China, 2003–2011. Int J Environ Res Public Health. 2013;10:1462–1473. doi: 10.3390/ijerph10041462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kwon NY, Kim JD, Pai HJ. The resistance mechanisms of b-lactam antimicrobials in clinical isolates of Acinetobacter baumannii. Korean J Intern Med. 2002;17:94–99. doi: 10.3904/kjim.2002.17.2.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.García-Quintanilla M, Pulido MR, Moreno-Martínez P, Martín-Peña R, López-Rojas R, Pachón J, McConnell MJ. Activity of host antimicrobials against multidrug-resistant Acinetobacter baumannii acquiring colistin resistance through loss of lipopolysaccharide. Antimicrob Agents Chemother. 2014;58:2972–2975. doi: 10.1128/AAC.02642-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen TL, Siu LK, Wu RC, Shaio MF, Huang LY, Fung CP, Lee CM, Cho WL. Comparison of one-tube multiplex PCR, automated ribotyping and intergenic spacer (ITS) sequencing for rapid identification of Acinetobacter baumannii. Clin Microbiol Infect. 2007;13:801–806. doi: 10.1111/j.1469-0691.2007.01744.x. [DOI] [PubMed] [Google Scholar]
- 12.Clinical Laboratory Standards Institute (CLSI) Performance standards for antimicrobial susceptibility testing; Twenty-second informational supplement. CLSI; Wayne, PA: 2012. CLSI document M100-S22. [Google Scholar]
- 13.Zhang JP, Zhu W, Tian SF, Chu YZ, Chen BY. Molecular characteristics and resistant mechanisms of imipenem-resistant Acinetobacter baumannii isolates in Shenyang. China J Microbiol. 2010;48:689–694. doi: 10.1007/s12275-010-0137-3. [DOI] [PubMed] [Google Scholar]
- 14.Merkier AK, Centrón D. bla(OXA-51)-type beta-lactamase genes are ubiquitous and vary within a strain in Acinetobacter baumannii. Int J Antimicrob Agents. 2006;28:110–113. doi: 10.1016/j.ijantimicag.2006.03.023. [DOI] [PubMed] [Google Scholar]
- 15.He C, Xie Y, Zhang L, Kang M, Tao C, Chen Z, Lu X, Guo L, Xiao Y, Duo L, et al. Increasing imipenem resistance and dissemination of the ISAba1-associated blaOXA-23 gene among Acinetobacter baumannii isolates in an intensive care unit. J Med Microbiol. 2011;60:337–341. doi: 10.1099/jmm.0.022681-0. [DOI] [PubMed] [Google Scholar]
- 16.Martínez P, Mattar S. Imipenem-resistant Acinetobacter baumannii carrying the ISAba1-bla OXA-23,51 and ISAba1-bla ADC-7 genes in Monteria, Colombia. Braz J Microbiol. 2012;43:1274–1280. doi: 10.1590/S1517-83822012000400006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Corvec S, Poirel L, Naas T, Drugeon H, Nordmann P. Genetics and expression of the carbapenem-hydrolyzing oxacil-linase gene blaOXA-23 in Acinetobacter baumannii. Antimicrob Agents Chemother. 2007;51:1530–1533. doi: 10.1128/AAC.01132-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yong D, Toleman MA, Giske CG, Cho HS, Sundman K, Lee K, Walsh TR. Characterization of a new metallo-beta-lactamase gene, bla(NDM-1), and a novel erythromycin esterase gene carried on a unique genetic structure in Klebsiella pneumoniae sequence type 14 from India. Antimicrob Agents Chemother. 2009;53:5046–5054. doi: 10.1128/AAC.00774-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cabanes F, Lemant J, Picot S, Simac C, Cousty J, Jalin L, Naze F, Boisson V, Cresta MP, André H, et al. Emergence of Klebsiella pneumoniae and Salmonella metallo-beta-lactamase (NDM-1) producers on reunion island. J Clin Microbiol. 2012;50:3812. doi: 10.1128/JCM.01029-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Savard P, Gopinath R, Zhu W, Kitchel B, Rasheed JK, Tekle T, Roberts A, Ross T, Razeq J, Landrum BM, et al. First NDM-positive Salmonella sp. strain identified in the United States. Antimicrob Agents Chemother. 2011;55:5957–5958. doi: 10.1128/AAC.05719-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jovcic B, Lepsanovic Z, Suljagic V, Rackov G, Begovic J, Topisirovic L, Kojic M. Emergence of NDM-1 metallo-β-lactamase in Pseudomonas aeruginosa clinical isolates from Serbia. Antimicrob Agents Chemother. 2011;55:3929–3931. doi: 10.1128/AAC.00226-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Poirel L, Dortet L, Bernabeu S, Nordmann P. Genetic features of blaNDM-1-positive Enterobacteriaceae. Antimicrob Agents Chemother. 2011;55:5403–5407. doi: 10.1128/AAC.00585-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ho PL, Lo WU, Yeung MK, Lin CH, Chow KH, Ang I, Tong AH, Bao JY, Lok S, Lo JY. Complete sequencing of pNDM-HK encoding NDM-1 carbapenemase from a multidrug-resistant Escherichia coli strain isolated in Hong Kong. PLoS One. 2011;6:e17989. doi: 10.1371/journal.pone.0017989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chu YW, Chau SL, Houang ET. Presence of active efflux systems AdeABC, AdeDE and AdeXYZ in dierent AbeM (MATE) Ade ABC Acinetobacter genomic DNA groups. J Med Microbiol. 2006;55:477–478. doi: 10.1099/jmm.0.46433-0. [DOI] [PubMed] [Google Scholar]
- 25.Marchand I, Damier-Piolle L, Courvalin P, Lambert T. Expression of the RND-type efflux pump AdeABC in Acineto-bacter baumannii is regulated by the AdeRS two-compo nent system. Antimicrob Agents Chemother. 2004;48:3298–3304. doi: 10.1128/AAC.48.9.3298-3304.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mussi MA, Limansky AS, Viale AM. Acquisition of resistance to carbapenems in multidrug-resistant clinical strains of Acinetobacter baumannii: Natural insertional inactivation of a gene encoding a member of a novel family of β-barrel outer membrane proteins. Antimicrob Agents Chemother. 2005;49:1432–1440. doi: 10.1128/AAC.49.4.1432-1440.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhou Z, Du X, Wang L, Yang Q, Fu Y, Yu Y. Clinical carbapenem-resistant Acinetobacter baylyi strain coharboring blaSIM-1 and blaOXA-23 from China. Antimicrob Agents Chemother. 2011;55:5347–5349. doi: 10.1128/AAC.00425-11. [DOI] [PMC free article] [PubMed] [Google Scholar]