Abstract
Background and Aims
The pathophysiology of some gastrointestinal neuromuscular diseases (GINMD) remains largely unknown. This is in part due to the inability to obtain ample deep gastric wall biopsies that include the intermuscular layer of the muscularis propria (MP) to evaluate the enteric nervous system, interstitial cells of Cajal (ICC) and related cells. We report on a novel technique for gastric endoscopic muscle biopsy (gEMB).
Methods
Patients with idiopathic gastroparesis were prospectively enrolled in a feasibility study using a novel “no hole” gEMB. Main outcome measures were technical success, adverse events and histological confirmation of the intermuscular layer, including myenteric neurons and ICC. The gEMB was a double resection clip–assist technique. A site was identified on the anterior wall of the gastric body as recommended by the International Working Group on histological techniques. Endoscopic mucosal resection was performed to unroof and expose the underlying MP. The exposed MP was then retracted into the cap of an over-the-scope clip. The clip was deployed and the pseudopolyp of MP created was resected. This resulted in a “nohole” gEMB.
Results
Three patients with idiopathic gastroparesis underwent gEMB. Patients had severe delayed gastric emptying with a mean of 49 ± 16.8% of retained gastric contents at 4 hours. They had no history of gastric or small-bowel surgery, and did not use steroids or other immunosuppressive drugs. The gEMB procedure was successfully performed with no procedural adverse events. Postprocedure abdominal pain was controlled with nonsteroidal anti-inflammatory agents and opioid analgesics. Mean length of resected MP was 10.3 ± 1.5 mm. Mean procedure time was 25.7 ± 6 minutes. Tissue samples on hematoxylin and eosin (H&E) stain confirmed presence of both inner circular and outer longitudinal muscle as well as the intermuscular layer. H&E stain showed reduced myenteric ganglia in 1 patient. In 2 patients, specialized immunohistochemistry was performed, which showed marked decrease in myenteric neurons as delineated by an antibody to Protein Gene Product (PGP) 9.5 and severe decrease in ICC across the muscle layers. At 1 month follow-up, upper endoscopy showed a well-healed scar in 2 patients and minimal ulceration with retained clip in 1 patient. CT abdomen confirmed the integrity of the gastric wall in all patients. Due to lack of an immune infiltrate in the resected samples, patients were not considered suitable for immunosuppressive or steroid therapy.
Conclusions
gEMB is feasible and easy to perform, with acquisition of tissue close to surgical samples to identify myenteric ganglia, ICC and multiple cell types. The ability to perform gEMB represents a paradigm shift in endoscopic tissue diagnosis of gastric neuromuscular pathologies.
The pathophysiology of many gastrointestinal neuromuscular diseases (GINMD) including idiopathic gastroparesis and functional dyspepsia, is not well understood1,2. There is growing evidence to support an underlying heterogeneous neuromuscular pathology in patients with gastroparesis and emerging evidence for cellular changes in functional diseases such as functional dypspesia3,4, 5. Endoscopic mucosal-based biopsies do not allow for evaluation of the myenteric plexus or interstitial cells of Cajal (ICC) networks that lie within the muscularis propria (MP) or the intermuscular layer of the MP. Currently, we rely on surgical approaches such as laparoscopic wedge biopsy to obtain sufficient tissue samples of the gastric wall.
A readily available, effective, and safe endoscopic technique that enables ample deep gastric wall biopsies to include the intermuscular layer of the MP for evaluation of the enteric nervous system, immune cells and ICC may provide invaluable insights into the pathogenesis of these disorders. The aims of this study were to (1) determine the efficacy of an innovative gastric endoscopic muscle biopsy (gEMB) technique; (2) identify the muscle layers included in the resected specimen and the presence of myenteric ganglia and ICC; (3) determine the procedural and long-term safety of the technique and (4) identify neuromuscular pathologic changes in patients with idiopathic gastroparesis.
METHODS
Patients
Patients were prospectively enrolled in this feasibility study approved by the Institutional Review Board. Patients diagnosed with symptomatic refractory idiopathic gastroparesis were recruited. Only patients with documentation within the last 2 years of delayed gastric emptying with >30% retained gastric contents at 4 hours based on 296 kcal solid-liquid, fat-containing standard meal gastric emptying test were included. Patients were excluded if there was a history of oropharyngeal, esophageal, gastric, or small-bowel surgery, esophageal stricture, abdominal radiation therapy, percutaneous endoscopic gastrostomy or jejunostomy, coagulopathy, and use of steroids or immunosuppressive drugs.
Patients were admitted after the procedure for 24-hour observation. Patients were maintained on clear liquids on day 1 of the procedure, and the diet was advanced as tolerated thereafter. Omeprazole 40 mg twice daily orally for 4 weeks and perioperative antibiotics were administered. Patients were contacted by phone 48 hours and 1 week after the procedure. At 1 month follow-up, a repeat upper endoscopy to review the mucosal aspect of the resection site and a CT abdomen to assess the integrity of the resection site were performed.
Description of Technique
gEMB involves a double resection clip-assist technique. A site was identified along the anterior wall of the gastric body, midway between the greater and lesser curves, as recommended by the International Working Group on histological techniques which is a consensus document by expert neurogastroenterologists6. From an endoscopic perspective the anterior wall is ideal both from safety (avoiding the gastroepiploic and gastric vessels) and ease of procedure performance. Endoscopic mucosal resection (EMR) using the band ligation approach (Duette; Cook Endoscopy, Bloomington, Ind) was initially performed to unroof and expose the underlying MP. An over-the-scope clip (Padlock Clip – 11 mm; Aponos, Kingston, NH) was fitted on a diagnostic upper endoscope (GIF-180; Olympus America, Center Valley, Pa) and advanced to the exposed MP. A tri-pronged tissue retraction device (OTSC Anchor, Ovesco Endoscopy, Tübingen, Germany) was passed through the working channel of the endoscope and deployed through the MP, which was then retracted and suctioned into the cap of the clip. Once an adequate amount of tissue was entrapped within the cap, the clip was deployed. The pseudopolyp of MP created was resected using hot snare (Acusnare; Cook Endoscopy, Bloomington, Ind). Resection was done approximately 5 mm above the deployed clip to ensure ample tissue remained for a secure closure. This approach resulted in a “no-hole” gEMB. Effective closure of the biopsy site was confirmed during endoscopy by visible closure without disruption, sustained distension of the gastric lumen with insufflation of CO2, and absence of radiopaque contrast leakage into the peritoneal cavity after intragastric injection determined fluoroscopically. The resected specimens of MP, as well as mucosa and submucosa from the EMR, were retrieved, pinned, measured, and submitted for histology.
RESULTS
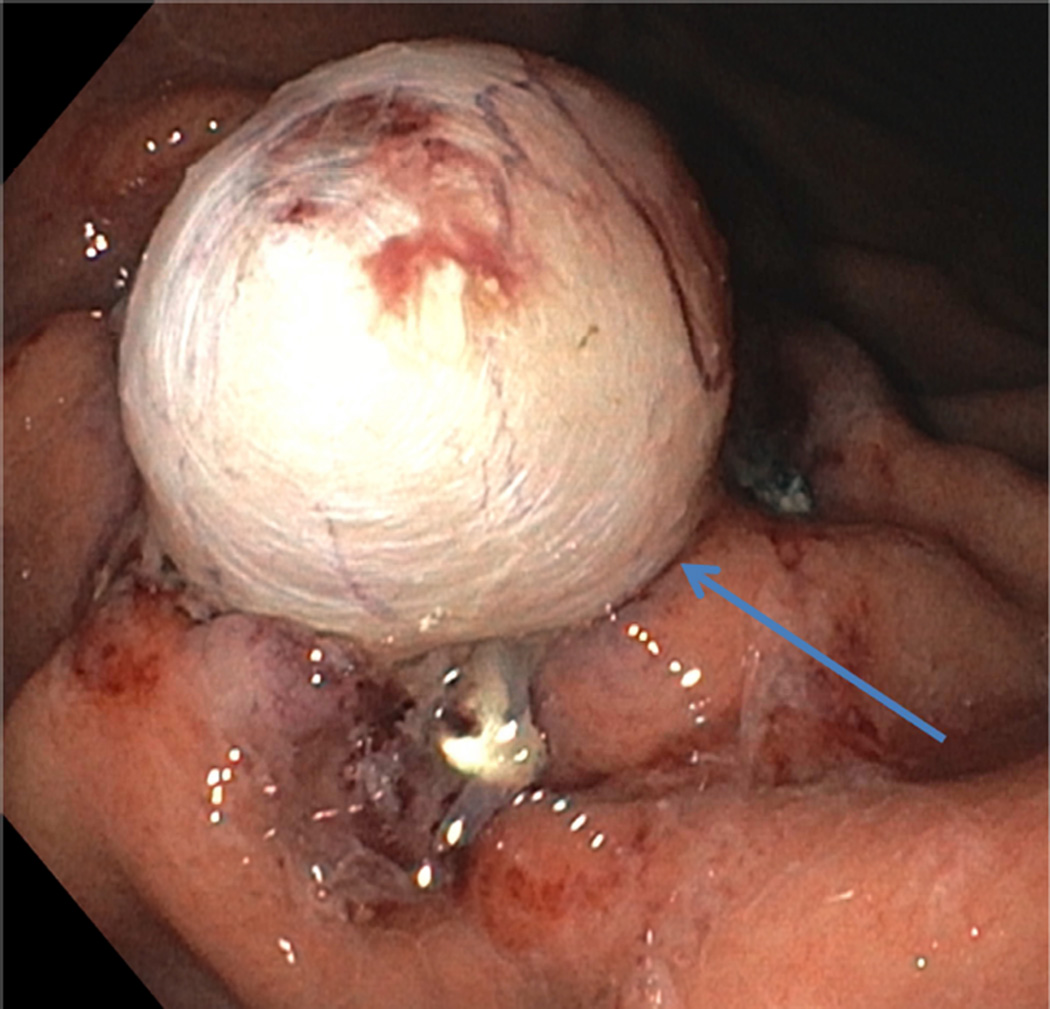
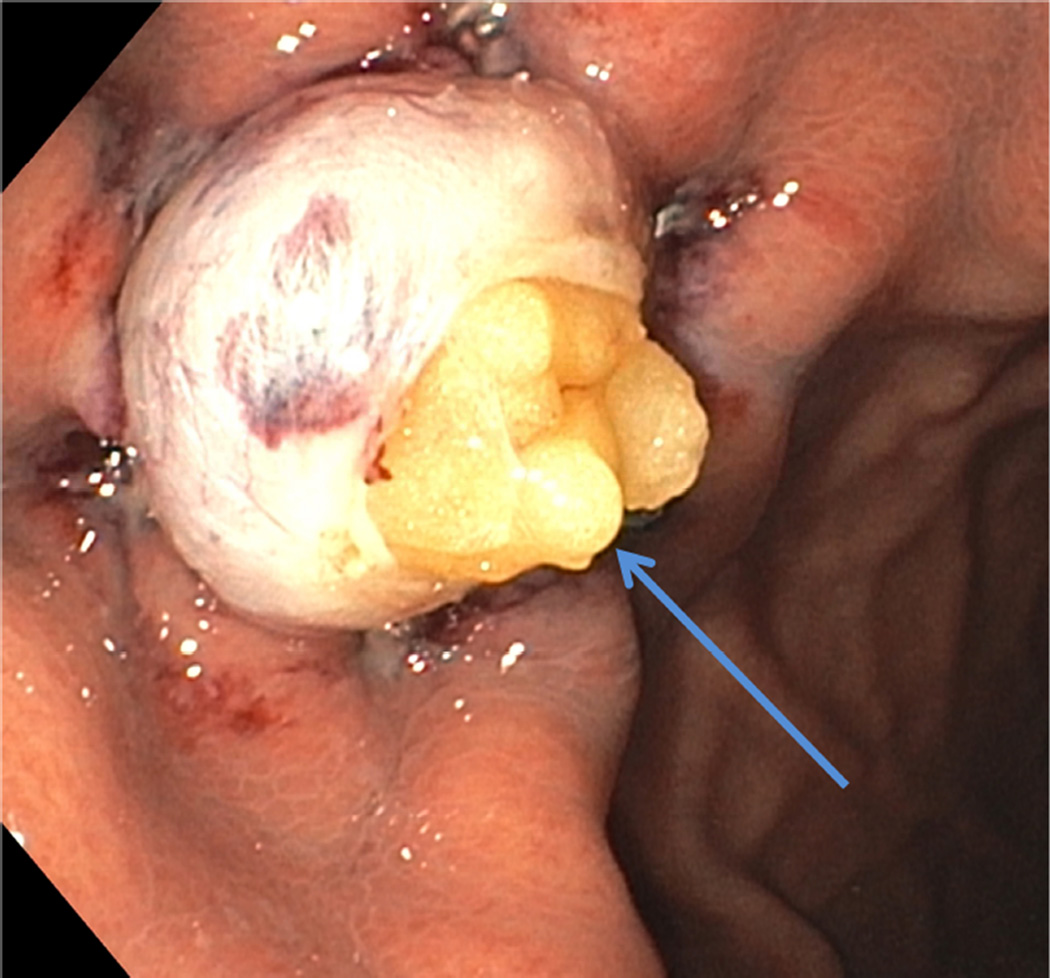
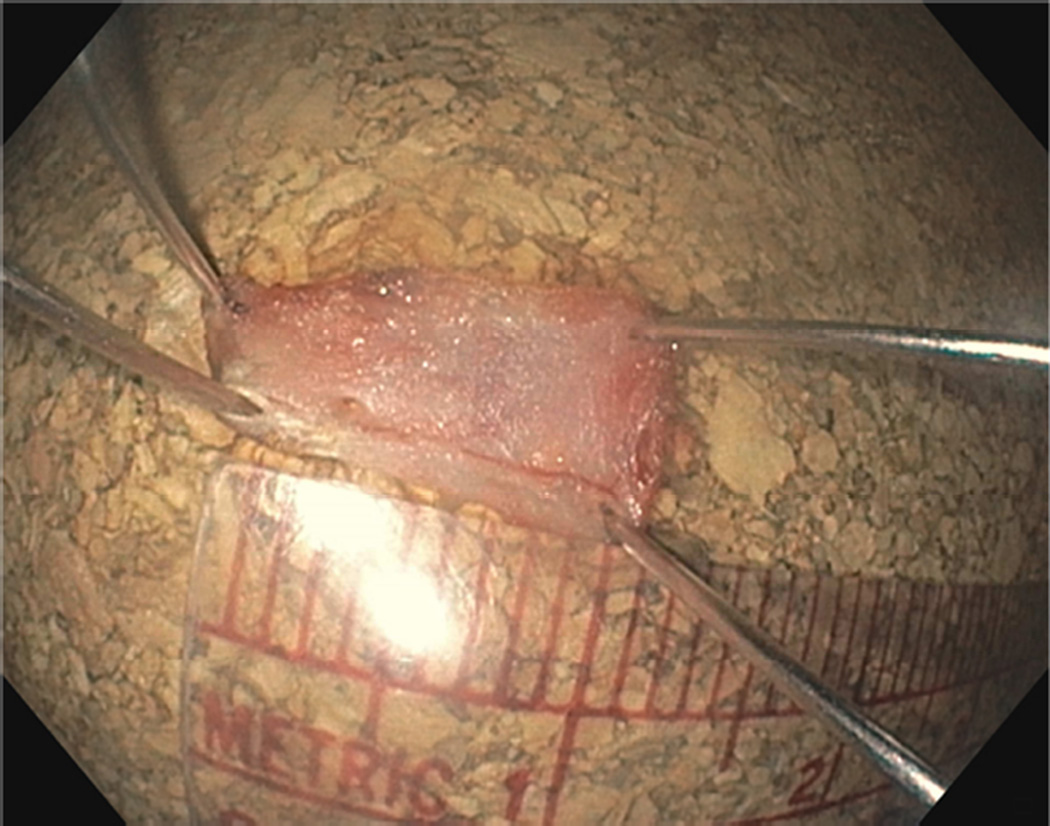
We report on 3 female patients with a mean age of 33 ± 8.5 years with symptomatic refractory idiopathic gastroparesis who underwent gEMB. Predominant symptoms were nausea, vomiting, abdominal pain, and weight loss that were ongoing for several years. Severe delayed gastric emptying with a mean of 49 ± 16.8% of retained gastric contents at 4 hours based on 296 kcal solid-liquid, fat-containing standard meal gastric emptying test was documented in study patients. A double resection clip-assist gEMB was performed successfully in all patients (Figures 1A and 1B). There were no intraprocedural adverse events. The mean size of resected MP was 10.3 ± 1.5 mm (Figure 2). The mean size of resected mucosa and submucosa was 22 ± 6.1 mm. Mean procedure time from endoscope intubation to retrieval of tissue specimens was 25.7 ± 6 minutes.
FIGURE 1.
A Pseudopolyp of muscularis propria (arrow) created after clip deployment
B After resection of muscularis propria with exposed omentum (arrow)
FIGURE 2.
Resected muscularis propria specimen
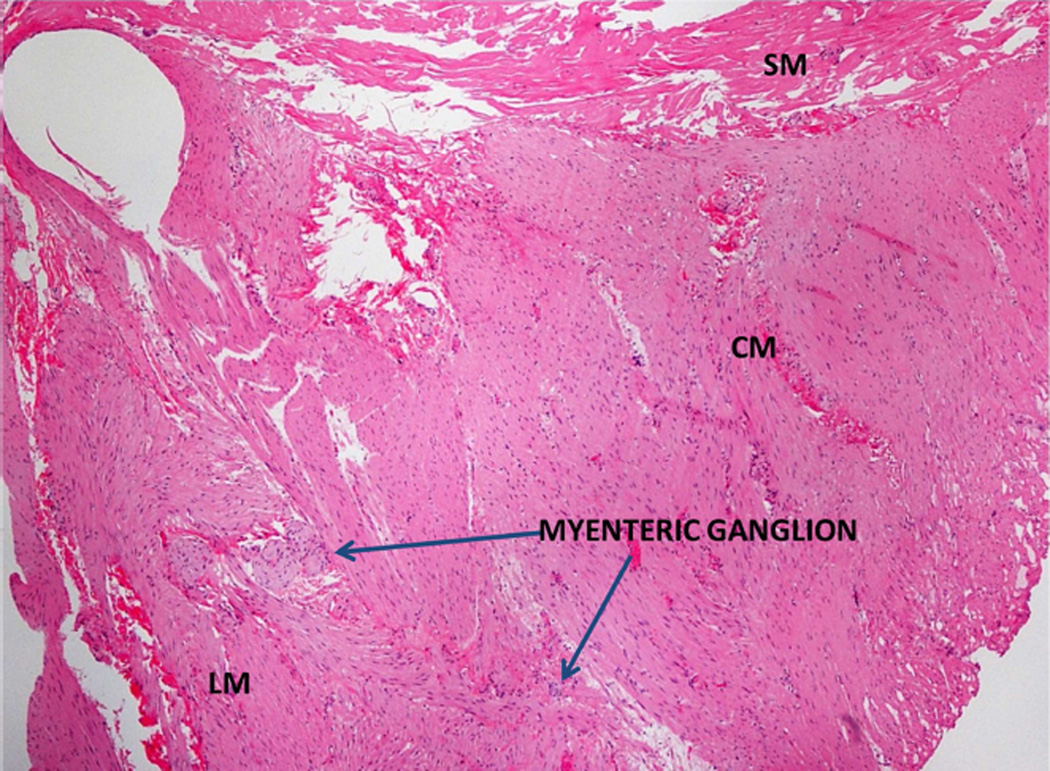
Tissue samples on hematoxylin and eosin (H&E) stain confirmed the presence of MP with inner circular and outer longitudinal muscle, as well as intermuscular layer in all patients (Figure 3). Abnormalities were noted in 2 out of 3 (66%) patients. H&E stain showed reduced myenteric ganglia in 1 patient. In 2 of the 3 patients, additional specialized immunohistochemistry was carried out, which showed a marked decrease in myenteric neurons in both patients as delineated by an antibody to Protein Gene Product (PGP) 9.5 (Figure 4A) and a severe decrease in Kit (CD 117) positive ICC across the muscle layers (Figure 4B). Immunohistochemical stain for CD3 showed no increase in T-cells, either in the myenteric plexus or smooth muscle. Smooth muscle actin was normal. Trichrome stain showed no muscle fibrosis. Mucosa and submucosa tissue samples showed submucosal fibrosis of unclear clinical significance in 1 patient.
FIGURE 3.
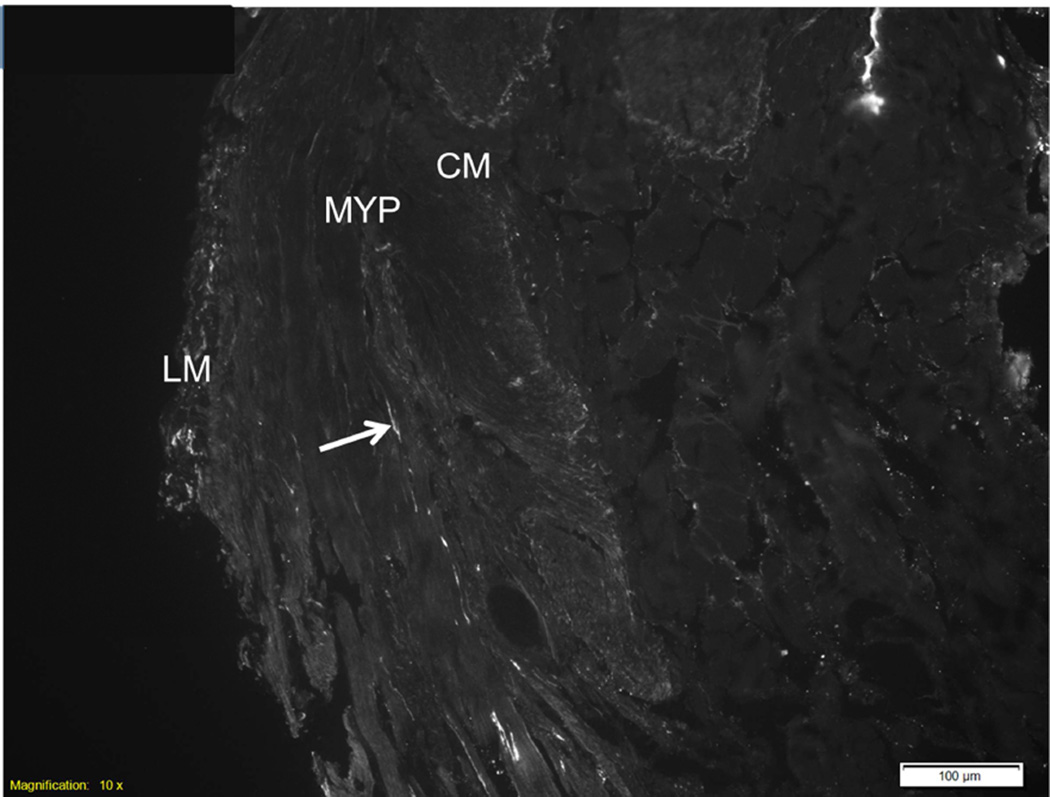
Hematoxylin and eosin (H&E) staining of the muscularis propria and myenteric ganglia. Tissue shows the presence of the circular muscle (CM), myenteric ganglia, longitudinal muscle (LM) and submucosa (SM). Scale bar = 40 µm
FIGURE 4.
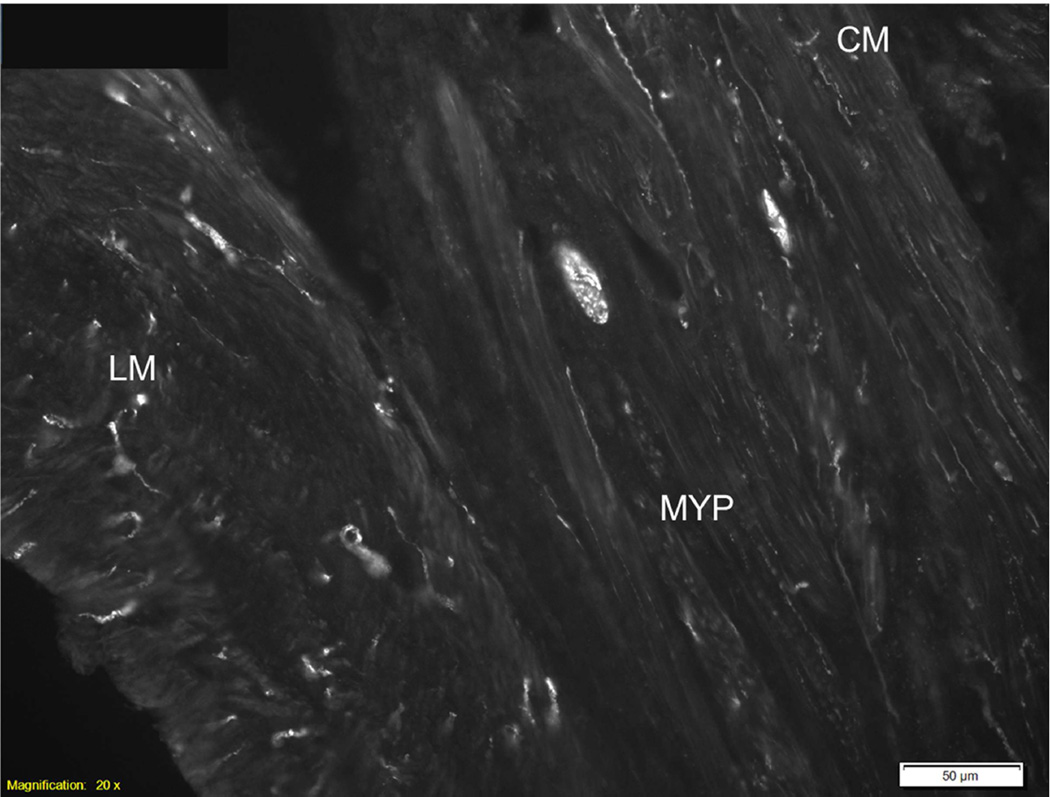
A Myenteric region showing marked decreased in Protein Gene Product 9.5 (PGP 9.5) positive neurons. CM = circular muscle; LM = longitudinal muscle; MYP = myenteric plexus. Scale bar = 50 µm.
B Intermuscular layer of the muscularis propria showing marked decrease in Kit (CD 117) positive interstitial cells of Cajal (ICC). CM = circular muscle; LM = longitudinal muscle; MYP = myenteric plexus. Scale bar = 100 µm.
In all 3 patients, upper abdominal pain with pleuritic component was experienced post-procedure which was controlled with nonsteroidal anti-inflammatory agents and opioid analgesics. No fever or leukocytosis was noted. Given this represented our initial experience, CT abdomen and chest CT angiography 48 hours after the procedure was performed in 1 patient with left upper quadrant abdominal pain. The studies were negative for intra-abdominal pathology and pulmonary embolism. Repeat upper endoscopy in this same patient showed ulceration and retained clip at the biopsy site. Patients were contacted at 48 hours and 1 week after the procedure. A visual analog scale was used for pain assessment, and patients reported moderate abdominal pain at 48 hours and mild pain at 1 week.
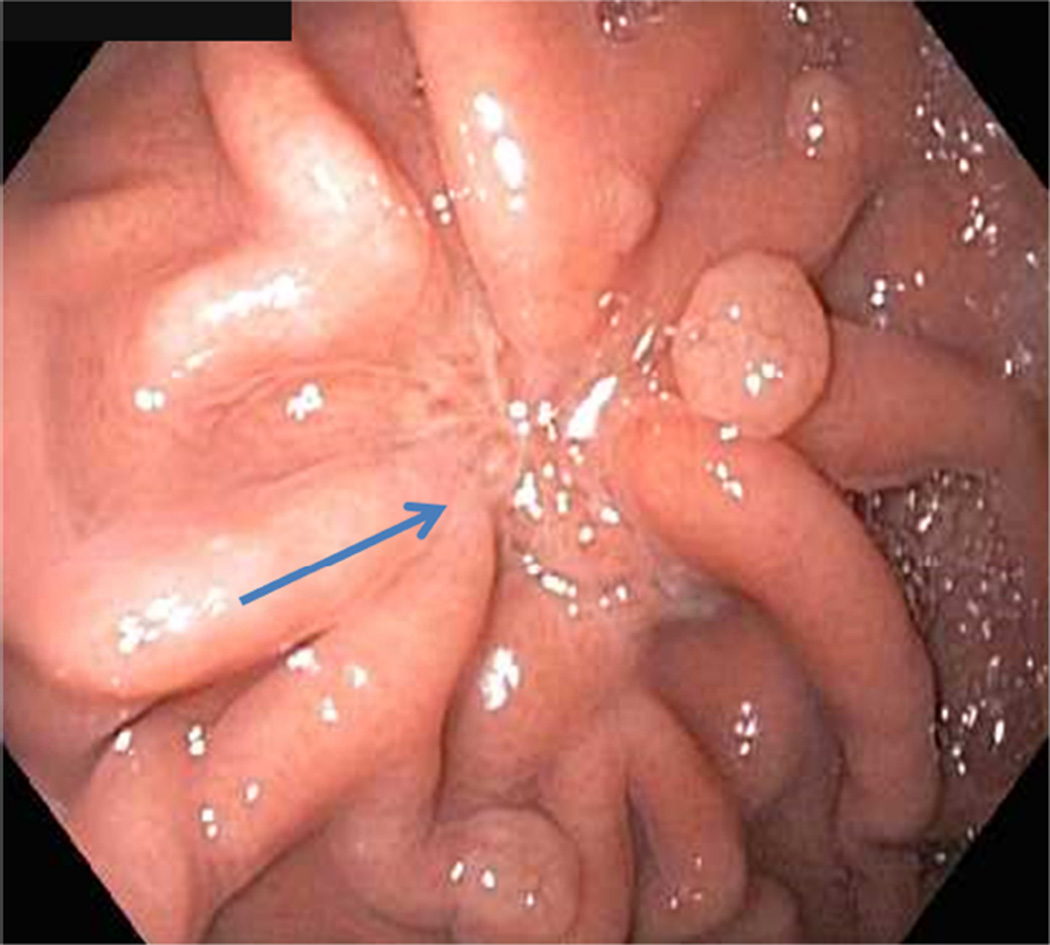
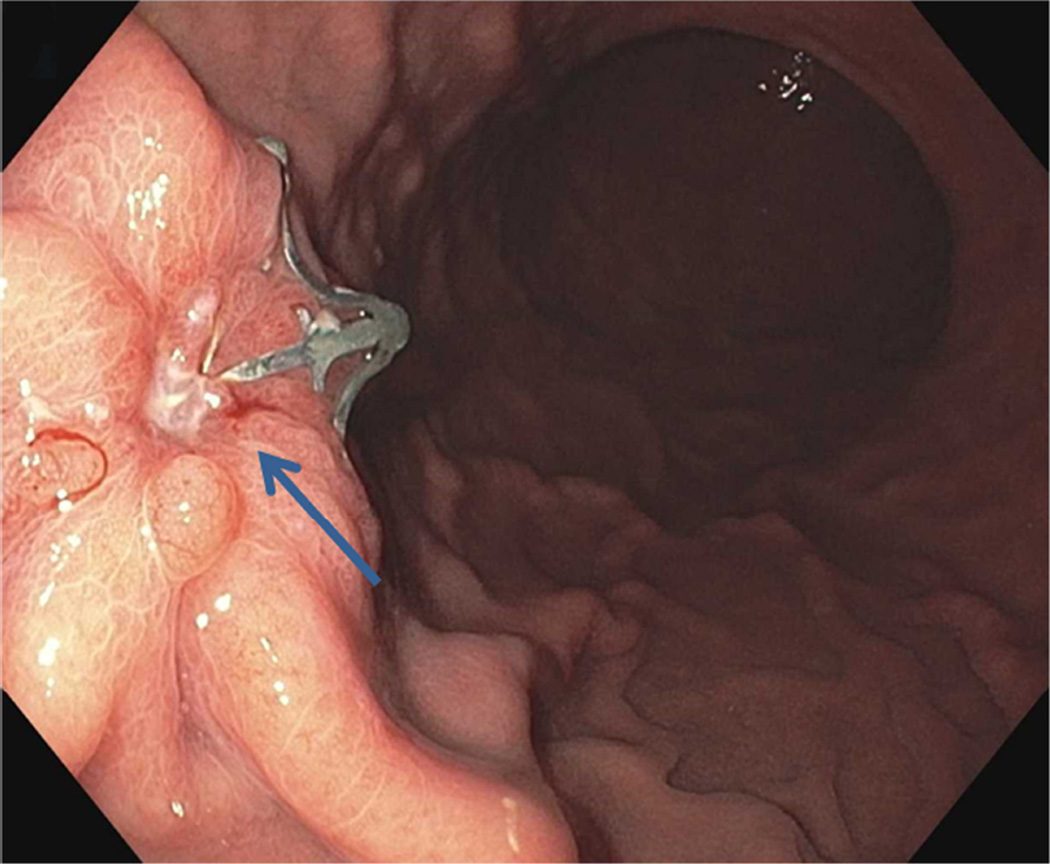
Repeat upper endoscopy and CT abdomen were performed at 1 month per study protocol. All patients reported baseline symptoms at 1 month. Endoscopy showed a well-healed scar in 2 patients and minimal residual ulceration with a retained clip in 1 patient (Figures 5A and 5B). A CT abdomen examination showed negative results in all patients.
FIGURE 5.
A Endoscopy at 1 month showing well-healed scar (arrow)
B Endoscopy at 1 month showing ulceration and retained clip (arrow)
DISCUSSION
The etiology of several GINMD, including functional GI disorders, remains largely unknown7,8,9,10. Surgically obtained gastric muscle biopsy specimens from patients with gastroparesis have shown a decrease in ICC in the majority of patients, changes in macrophages, and a variable decrease in nerve fibers11. We have previously reported on different experimental endoscopic techniques for deep gastric wall biopsies to include the entire MP12,13. Our group introduced the “no hole” concept (close, then cut) to eliminate the risk of peritonitis from leaked gastric contents14. However, the techniques were limited by lack of adequate sample size and/or safety issues. Percutaneous endoscopically assisted transenteric full-thickness biopsy using a 14-gauge needle for assessing histopathological abnormalities in GINMD showed abnormalities in 44% of patients15. Recently an EUS-guided fine-needle aspiration of the gastric antrum MP using a 19 gauge core-needle was shown to be safe with adequate tissue for histologic assessment of myenteric plexus in 54.5% and ICC in 81% of patients16,17. The limitations of these 2 techniques are the small sample volume of MP and the fragmented samples that can impair proper tissue orientation and impact diagnostic yield. The use of confocal laser endomicroscopy for optical histologic imaging is promising, but depth of imaging may not be sufficient in the stomach and the applicability of dye-based methods in humans is unclear18,19,20,21.
The selection of a star-shaped, over-the-scope clip was intentional because when deployed, the flat clip configuration facilitates easy snare placement without entrapment of the clip prongs (Figure 6). Other preferences for the selection of this closure device included a simple pushbutton deployment, a clip diameter of 11 mm allowing for atraumatic peroral intubation, and a cap depth of 10 mm allowing for substantial tissue capture. The trigger wire is located alongside the endoscope shaft, thus freeing the working channel for use of additional accessories and suction of luminal contents. The double resection technique is fundamental for deep gastric wall biopsies due to the relative thickness of the gastric wall. Exposing the underlying MP after EMR ensures the entire muscle layer is captured in the subsequent resection. All devices used are commercially available in the United States.
FIGURE 6.
Star-shaped over-the-scope clip
Large tissue samples of the entire MP, closer to the criterion standard of surgically acquired specimens (≥10 mm), with myenteric ganglia and ICC were obtained in all patients. Harberson et al reported on laparoscopic full-thickness gastric antral biopsy specimens obtained from the anterior wall of the stomach in patients with gastroparesis2. The range in size of tissue samples were from 0.3 × 0.3 × 0.3 cm to 1.5 × 1.0 × 1.0 cm. The postprocedure abdominal pain may be explained by thermal effect on the serosa causing inflammation, edema and pain akin to postpolypectomy syndrome. Abnormalities with reduction of myenteric plexus and loss of ICC were noted, which are considered hallmark features of gastroparesis22. Due to the lack of an immune infiltrate in the resected specimens, patients were not considered suitable candidates for immunosuppressive or steroid therapy.
Based on our initial experience, the minimally invasive gEMB approach appears to be safe, effective, and easy to perform. This innovative gEMB method using a double resection clip-assist technique represents a paradigm shift in endoscopic tissue diagnosis. Our ability to acquire large tissue samples allows for both quantitative and qualitative analysis of multiple cell types. Future research may contribute to our knowledge of the pathophysiology of GINMD and other inflammatory and neoplastic conditions, leading to potential targeted and curative therapies and likely challenging current clinical practice.
Supplementary Material
Gastric endoscopic muscle biopsy (gEMB) technique
Acknowledgments
Grant support: Weissman Fund, NIH grants DK68055 and DK57061 and Center for Individualized Medicine, Mayo Clinic, Rochester, MN
ACRONYMS
- GINMD
Gastrointestinal neuromuscular disease
- MP
Muscularis propria
- ICC
Interstitial cells of Cajal
- gEMB
Gastric endoscopic muscle biopsy
- H&E
Hematoxylin and Eosin
- PGP
Protein Gene Product
- CM
Circular muscle
- LM
Longitudinal muscle
- SM
Submucosa
- MYP
Myenteric plexus
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Author contribution
ER, CJG, GF, LMWKS and TCS: conception and design, analysis and interpretation of the data, drafting of the article, critical revision of the article and final approval for submission.
LAS, PK, JB, JLD, MAK and CEB: critical revision of the article and final approval for submission
REFERENCES
- 1.Farrugia G. Interstitial cells of Cajal in health and disease. Neurogastroenterol Motil. 2008;20(Suppl 1):54–63. doi: 10.1111/j.1365-2982.2008.01109.x. [DOI] [PubMed] [Google Scholar]
- 2.Harberson J, Thomas RM, Harbison SP, Parkman HP. Gastric neuromuscular pathology in gastroparesis: analysis of full-thickness antral biopsies. Dig Dis Sci. 2010;55(2):359–370. doi: 10.1007/s10620-009-1071-2. [DOI] [PubMed] [Google Scholar]
- 3.Faussone-Pellegrini MS, Grover M, Pasricha P, et al. Ultrastructural differences between diabetic and idiopathic gastroparesis. J Cell Mol Med. 2012;16:1573–1581. doi: 10.1111/j.1582-4934.2011.01451.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Angeli TR, Cheng LK, Du P, et al. Loss of interstitial cells of Cajal and patterns of gastric dysrhythmia in patients with chronic unexplained nausea and vomiting. Gastroenterology. 2015;149(1):56–66. doi: 10.1053/j.gastro.2015.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cirillo C, Bessissow T, Desmet AS, Vanheel H, Tack J, Vanden Berghe P. Evidence for neuronal and structural changes in submucous ganglia of patients with functional dyspepsia. Am J Gastroenterol. 2015;110(8):1205–1215. doi: 10.1038/ajg.2015.158. [DOI] [PubMed] [Google Scholar]
- 6.Knowles CH, De Giorgio R, Kapur RP, et al. Gastrointestinal neuromuscular pathology: guidelines for histological techniques and reporting on behalf of the Gastro 2009 International Working Group. Acta Neuropathol. 2009;118(2):271–301. doi: 10.1007/s00401-009-0527-y. [DOI] [PubMed] [Google Scholar]
- 7.Kashyap P, Farrugia G. Enteric autoantibodies and gut motility disorders. Gastroenterol Clin North Am. 2008;37:397–410. doi: 10.1016/j.gtc.2008.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kashyap P, Farrugia G. Diabetic gastroparesis: what we have learned and had to unlearn in the past 5 years. Gut. 2010;59(12):1716–1726. doi: 10.1136/gut.2009.199703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pasricha PJ, Yates KP, Nguyen L, et al. Outcomes and factors associated with reduced symptoms in patients with gastroparesis. Gastroenterology. 2015;149(7):1762–1774. doi: 10.1053/j.gastro.2015.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Neshatian L, Gibbons SJ, Farrugia G. Macrophages in diabetic gastroparesis--the missing link? Neurogastroenterol Motil. 2015;27(1):7–18. doi: 10.1111/nmo.12418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Grover M, Farrugia G, Lurken MS, et al. NIDDK Gastroparesis Clinical Research Consortium. Cellular changes in diabetic and idiopathic gastroparesis. Gastroenterology. 2011;140(5):1575–1585. doi: 10.1053/j.gastro.2011.01.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rajan E, Gostout CJ, Lurken MS, et al. Evaluation of endoscopic approaches for deep gastricmuscle-wall biopsies: what works? Gastrointest Endosc. 2008;67(2):297–303. doi: 10.1016/j.gie.2007.06.024. [DOI] [PubMed] [Google Scholar]
- 13.Rajan E, Gostout CJ, Aimore Bonin E, et al. Endoscopic full-thickness biopsy of the gastric wall with defect closure by using an endoscopic suturing device: survival porcine study. Gastrointest Endosc. 2012;76(5):1014–1019. doi: 10.1016/j.gie.2012.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rajan E, Gostout CJ, Lurken MS, et al. Endoscopic "no hole" full-thickness biopsy of the stomach to detect myenteric ganglia. Gastrointest Endosc. 2008;68(2):301–307. doi: 10.1016/j.gie.2007.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Andrews CN, Mintchev P, Neshev E, et al. Percutaneous endoscopically assisted transenteric full-thickness gastric biopsy: initial experience in humans. Gastrointest Endosc. 2011;73(5):949–954. doi: 10.1016/j.gie.2010.12.037. [DOI] [PubMed] [Google Scholar]
- 16.Othman MO, Davis B, Saroseik I, Torabi A, McCallum RW. EUS-guided FNA biopsy of the muscularis propria of the antrum in patients with gastroparesis is feasible and safe. Gastrointest Endosc. 2016;83:327–333. doi: 10.1016/j.gie.2015.06.056. [DOI] [PubMed] [Google Scholar]
- 17.Grover M, Farrugia G, Pasricha PJ. Endoscopy of the "brain": the next frontier in gastroenterology. Gastrointest Endosc. 2016;83(2):334–336. doi: 10.1016/j.gie.2015.10.011. [DOI] [PubMed] [Google Scholar]
- 18.Sumiyama K, Kiesslich R, Ohya TR, et al. In vivo imaging of enteric neuronal networks in humans using confocal laser endomicroscopy. Gastroenterology. 2012;143(5):1152–1153. doi: 10.1053/j.gastro.2012.09.001. [DOI] [PubMed] [Google Scholar]
- 19.Ohya TR, Sumiyama K, Takahashi-Fujigasaki J, et al. In vivo histologic imaging of the muscularis propria and myenteric neurons with probe-based confocal laser endomicroscopy in porcine models (with videos) Gastrointest Endosc. 2012;75(2):405–410. doi: 10.1016/j.gie.2011.09.045. [DOI] [PubMed] [Google Scholar]
- 20.Pasricha PJ. Endoscopy 20 years into the future. Clin Gastroenterol Hepatol. 2013;11(2):119–122. doi: 10.1016/j.cgh.2012.11.017. [DOI] [PubMed] [Google Scholar]
- 21.Boesmans W, Hao MM, Vanden Berghe P. Optical Tools to Investigate Cellular Activity in the Intestinal Wall. J Neurogastroenterol Motil. 2015;21(3):337–351. doi: 10.5056/jnm15096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Grover M, Bernard CE, Pasricha PJ, et al. Clinical-histological associations in gastroparesis: results from the Gastroparesis Clinical Research Consortium. Neurogastroenterol Motil. 2012;24(6):531–539. e249. doi: 10.1111/j.1365-2982.2012.01894.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Gastric endoscopic muscle biopsy (gEMB) technique