Neuropathologies include the deterioration and damage of the nervous system, especially neurons present in the brain, spinal cord and peripheral nervous system. Damage or alternations in neurons makes their structure and functionality abnormal. Every year over 90,000 people get affected by neurodegenerative diseases in the USA. Among all the neurological pathologies, the spinal cord injuries alone influence 10,000 people in the USA every year. Millions of cases of neurodegenerative pathologies are registered worldwide. Without proper treatment and cure to these cases, the toll for patients suffering from neurode-generative disease is expected to reach at least 12 million in the USA 30 years from now (John, 2015).
Another aspect of neural pathologies includes peripheral nerve injury (PNI). Since mid 1980s, Food and Drug Administration have approved various natural and synthetic biomaterials based devices for repairing nerve defects arising from PNI. Worldwide, more than 8.5 million restricted activity days and approximately 5 million bed/disability days each year are results of PNIs (Belkas et al., 2004). The most unfortunate part of this is that all types of PNIs are not manageable via surgical intervention. The surgical procedures mainly neurorrhaphy (direct suture repair without employing any grafted materials) can only be employed for shorter (< 5 mm) nerve gaps. Trials on repairing larger nerve defects by the similar neurorhaphy exert excessive tension on the suture line and offer poor surgical outcomes (Kehoe et al., 2012). The present scenario necessitates an urgent need to discover new therapies and cure for neuropathological conditions.
It is well established that physical guidance of axons plays a vital role in nerve repair. Earlier in the 19th century, various kinds of materials were employed to provide physical guidance for the regeneration of injured nerves, namely, autologous nerve grafts, bone, metal tubes, and fat sheaths. During 1960s, Millesi et al. developed microsurgical techniques for aligning nerve fascicles precisely in the direct resection of nerve ends, with better functional recovery (Schmidt and Leach, 2003). Further, they also established the fact that nerve grafts placed between the damaged nerves makes the damaged nerve tension free and enhanced functional recovery. With the advancement in the medical innovations, various physical guidance conduits have been proposed with different biochemical stimuli for nerve regeneration in the course of time (John, 2015).
Polymers are mostly being used for nerve conduits due to their following characteristics: (i) soft and flexible nature similar to that of neural tissues; (ii) structural and functional similarity to the biomolecules existing in the extracellular matrix; (iii) biocompatibility and (iv) easy functionalization with essential proteins and growth factors for better performance. But, these polymeric scaffolds lack in mechanical strength and electrical conductivity, primary requisite for neural growth. Thus, the search was always on to have a suitable scaffold material with mechanical strength and electrical conductivity, along with other prerequisites for replacing neural tissue. Recently, neuroscientists are getting attracted towards conductive hybrid materials modulating the single neurons or neuronal networks being grown integrated to such surfaces. Indeed, the main driving force for such research approaches lies in the limited regenerative power of the central nervous system. Interdisciplinary approach in modern neuroscience research allows fabricating hybrid circuits via nanomaterial assemblies, with well defined physical-chemical properties, and cultured neurons. With the help of such substrates, we can also govern physical features to be sensed by cells, which activate molecular level events of neural regeneration and repair. In this connection, carbon nanotubes (CNT), due to their large surface area, high electrical conductivity and mechanical properties, can serve as suitable material for neural electrical interfacing applications. The properties including roughness, charge, polarity, and chemistry of CNT scaffolds, can alter the affinity of neurons linked to CNT-containing surface. The neurite regrowth is favorable in spinal explants with higher growth cone activity in smoother and electrically conductive the surface. The direct interactions between neurites and CNTs, act as an exoskeleton, with more membrane/material tight junctions formation. Greater surface area of CNT significantly resuits in stronger charge injection capacity and lesser interfacial impedance as they help in electrochemical coupling via electron transfer between CNTs and neuritis (Fabbro et al., 2012).
With the progress in neural tissue regeneration techniques, it was explored that oriented topographical cues on polymer scaffolds stimulate the endogenous repair mechanisms, predominantly by increasing the Schwann cell migration and axonal outgrowth across the nerve gap even without the exogenous growth promoting proteins (Kim et al., 2008). A study by Fan et al. (2012) used aligned CNT yarn patterned substrates as the topographic scaffold to provide guidance to the neurite outgrowth. Parallel-aligned and cross-linked CNT yarn patterned substrates have shown better axonal path finding in comparison to the plain polystyrene substrates (Fan et al., 2012). The fascinating outcomes of this study led the direction of our research. But the above mentioned breakthrough researches have some drawbacks, as noted below, which are needed to be taken care for fabricating a successful neural scaffold. The CNTs, being patterned on some substrates, are directly exposed on the surface of the scaffolds. The direct exposure of the CNTs presents a high probability of detachment from the substrate during implantation inside body. Even if the implantation surgery avoids all the risks of detachment, post-implantation risks are also associated with these patterned substrates. Biological interactions with tissue interfaces offer the chance of the CNTs getting detached or leached out into the tissue fluid and ultimately to the blood stream, thus failing to retain the expected functionalities of the scaffold for neural regeneration.
Apart from the problem mentioned above about leaching of CNTs from bare surface, the other serious problem with the above mentioned scaffolds were the substrate on which CNTs were patterned. The scaffolds, with patterned CNTs, are fabricated on various types of hard and non-polymeric substrates, e.g., glass, aluminium foils and silicon, using photolithography, chemical vapor deposition (CVD) and other micro-patterning techniques. These hard, non-polymeric, and non-biodegradable substrates would not be a good candidate for neural tissue engineering, due to their in vivo incompatibility with surrounding tissues. The over rigidity of these scaffolds causes nerve compression, imposing more deterioration to the damaged part. This is also the main drawback of some of the commercially available nerve guidance conduits.
Both of these was overcome by our group, in a recent study, through reinforcing the CNT patterns inside biocompatible soft polymer matrix (Gupta et al., 2015). The alignment of CNTs was maintained within the matrix, without any chance of CNT leaching and detachment from surface. The orientation of the CNTs can be clearly observed in Figure 1B and 1C. Figure 1B shows the CNTs oriented randomly in radial direction, while CNTs are well aligned in a particular direction in aligned scaffold (Figure 1C). The alignment of CNTs inside chitosan matrix also provided the anisotropic electrical conductivity, leading to the preferential alignment of neurons in the alignment direction.
Figure 1.
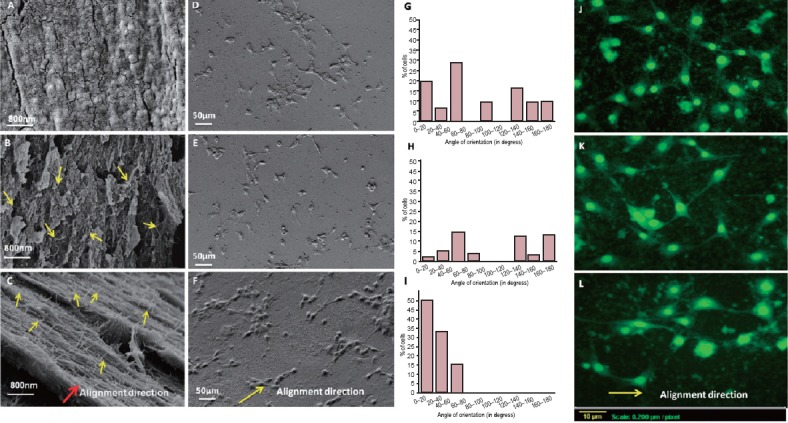
Effect of MWCNT alignment direction on orientation of HT-22 neurons.
Fracture surfaces of the chitosan, random and aligned scaffolds are shown in FESEM images A, B and C respectively. The images D, E and F show orientation of the grown HT-22 rat hippocampal neurons on chitosan, chitosan-random MWCNT scaffold and chitosan-aligned MWCNT scaffold respectively. Bar charts show percentage of hippocampal neurons oriented in various angles on chitosan (G), random (H) and aligned (I) scaffolds corresponding to figures D, E and F respectively. The florescent images depicting the alignment of the neurites are shown in the figures J, K and L for chitosan, random and aligned scaffolds respectively. Alignment direction of MWCNT in chitosan matrix has been shown with yellow arrows while red arrow in image C shows the MWCNT alignment direction in chitosan matrix (Gupta et al., 2015). MWCNTs: Multi walled carbon nanotubes.
However, the beneficial effects of alignment of CNTs inside polymer matrix for neural tissue engineering have already been described, but it is rather difficult to get alignment of CNTs inside soft, biodegradable polymer substrates. The polymers generally cannot withstand the high temperature and strong chemical agents, required for most of the CNT alignment techniques. Therefore, the selection of the alignment method also became critical for our study. Finally the a.c. electric field application method was selected due to its simple and cost-effective nature to align the multi walled carbon nanotubes (MWCNTs) inside chitosan polymer matrix. This technique allows the easy and robust way to align the CNTs inside polymer-based scaffold, and not loosely attached to the surface. After characterizing the scaffold it was also confirmed that the scaffold offers all the expected cues/functionalities, particularly electrical conductivity. The alignment of the neurites and the cell bodies of HT-22 hippocampal neurons on aligned CNT-chitosan scaffold was studied and compared with the neurons grown on randomly oriented MWCNTs reinforced chitosan scaffold as well as chitosan scaffold alone.
It was observed that the neurites do not prefer any direction for growth and show random orientation on both chitosan and chitosan-random MWCNT scaffolds (Figure 1D and 1E). However, the HT-22 rat hippocampal neurons on the chitosan-aligned MWCNT scaffolds (Figure 1F) behave distinctly different from other two scaffolds. The neurons, grown on the chitosan-aligned MWCNT scaffolds, show a favored direction of growth, which happens to be the alignment direction of nanotubes in the scaffolds.
This directional neurite growth suggests the prospect of this scaffold for treating pathological conditions ranging from spinal cord injury to peripheral nerve repair, using transplanted cells. It was really a challenging task in itself, and there were no previous reports available for developing such kind of scaffolds. However, taking all the requisites into consideration, the study was designed to fabricate a modified polymer CNT scaffold that can simultaneously offer biological, mechanical, electrical, and topographical cues needed for neural tissue engineering.
References
- 1.Belkas JS, Shoichet MS, Midha R. Peripheral nerve regeneration through guidance tubes. Neurol Res. 2004;26:151–160. doi: 10.1179/016164104225013798. [DOI] [PubMed] [Google Scholar]
- 2.Fabbro A, Villari A, Laishram J, Scaini D, Toma FM, Turco A, Prato M, Ballerini L. Spinal cord explants use carbon nanotube interfaces to enhance neurite outgrowth and to fortify synaptic inputs. ACS Nano. 2012;6:2041–2055. doi: 10.1021/nn203519r. [DOI] [PubMed] [Google Scholar]
- 3.Fan L, Feng C, Zhao W, Qian L, Wang Y, Li Y. Directional neurite outgrowth on superaligned carbon nanotube yarn patterned substrate. Nano Lett. 2012;12:3668–3673. doi: 10.1021/nl301428w. [DOI] [PubMed] [Google Scholar]
- 4.Gupta P, Sharan S, Roy P, Lahiri D. Aligned carbon nanotube reinforced polymeric scaffolds with electrical cues for neural tissue regeneration. Carbon. 2015;95:715–724. [Google Scholar]
- 5.John AA. Carbon nanotubes and graphene as emerging candidates in neuroregeneration and neurodrug delivery. Int J Nanomedicine. 2015;10:4267–4277. doi: 10.2147/IJN.S83777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kehoe S, Zhang XF, Boyd D. FDA approved guidance conduits and wraps for peripheral nerve injury: a review of materials and efficacy. Injury. 2012;43:553–572. doi: 10.1016/j.injury.2010.12.030. [DOI] [PubMed] [Google Scholar]
- 7.Kim Y, Haftel VK, Kumar S, Bellamkonda RV. The role of aligned polymer fiber-based constructs in the bridging of long peripheral nerve gaps. Biomaterials. 2008;29:3117–3127. doi: 10.1016/j.biomaterials.2008.03.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schmidt CE, Leach JB. Neural tissue engineering: strategies for repair and regeneration. Annu Rev Biomed Eng. 2003;5:293–347. doi: 10.1146/annurev.bioeng.5.011303.120731. [DOI] [PubMed] [Google Scholar]


