Keywords: nerve regeneration, brain injury, CD163, soluble CD163, intracerebral hemorrhage, hemoglobin scavenging, haptoglobin, hemoglobin, hematoma, absorption, adult, brain, neural regeneration
Abstract
Clinical outcomes are positively associated with hematoma absorption. The monocyte-macrophage scavenger receptor, CD163, plays an important role in the metabolism of hemoglobin, and a soluble form of CD163 is present in plasma and other tissue fluids; therefore, we speculated that serum CD163 affects hematoma absorption after intracerebral hemorrhage. Patients with intracerebral hemorrhage were divided into high- and low-level groups according to the average CD163 level (1,977.79 ± 832.91 ng/mL). Compared with the high-level group, the low-level group had a significantly slower hematoma absorption rate, and significantly increased National Institutes of Health Stroke Scale scores and modified Rankin Scale scores. These results suggest that CD163 promotes hematoma absorption and the recovery of neurological function in patients with intracerebral hemorrhage.
Introduction
Intracerebral hemorrhage (ICH) is a common type of stroke, from a non-intracranial vascular traumatic spontaneous bleed. The bleeding site can be in the brain parenchyma and the hematoma may rupture into the ventricle or subarachnoid space. ICH accounts for 10–15% of all strokes, and is one of the major causes of human death (Lei et al., 2013; Paciaroni and Agnelli, 2014; Specogna et al., 2014).
Hematoma is an initial factor for mechanical compression of brain tissue and secondary neurological damage; therefore, controlling hematoma is essential for the treatment of ICH. In clinical practice, outcome is positively associated with the rate of hematoma absorption (van Asch et al., 2010). Internal bleeding can result in the release of hemoglobin (Hb), which causes tissue damage (Aronowski and Zhao, 2011). The primary mechanism protecting against the deleterious effects of ‘free’ Hb is governed by the monocyte-macrophage scavenger receptor, CD163 (Kristiansen et al., 2001; Schaer et al., 2013). CD163 is an important marker in atherosclerosis, sepsis, hepatitis, and type 2 diabetes (Moreno et al., 2009; Møller et al., 2011; Ye et al., 2013; Kjærgaard et al., 2014). It remains unclear whether CD163 can be used as an evaluation index of ICH.
CD163, also known as M130, belongs to the scavenger receptor cysteine-rich superfamily, and was discovered in 1987 by Zwadlo et al. CD163 is mainly localized on the surface of monocytes/macrophages; however, its function is unclear (Högger et al., 1998). CD163 can be shed from the cell surface to produce a soluble form of CD163 in plasma and other tissue fluids (Sulahian et al., 2001; Møller et al., 2002b). CD163 might mediate endocytosis of Hb when bound with the Hb-haptoglobin (Hp) complex (Droste et al., 1999; Kristiansen et al., 2001). Under pathological conditions, CD163-mediated Hb clearance increases significantly. Liu et al. (2015) found that CD163 expression in brain tissue around a hematoma gradually increased after ICH. Based on the key role of CD163 in the Hb metabolic pathway, we speculated that CD163 functions in the absorption of hematoma after ICH and affects the prognosis of ICH. However, the relationship between the peripheral serum concentrations of CD163 and ICH is poorly understood.
In the present study, by measuring changes in blood levels of CD163 and making neurological assessments, we aimed to identify whether CD163 promotes absorption of hematoma following ICH. We also evaluated whether CD163 can serve as both a biomarker and a potential target for new ICH therapy.
Subjects and Methods
Patients
The study was performed with approval from the Ethics Committee of the First Hospital of Jilin University of China. Written informed consent was obtained from all patients or guardians of patients participating in the study.
A total of 54 patients (18 females and 36 males) with acute ICH that were hospitalized in the First Hospital of Jilin University of China from August 2013 to December 2014 were enrolled. Immediate treatment with osmotic dehydration and rehabilitation were given to all the patients. Inclusion criteria: (1) Age of the patient was in range 18 to 80 years; (2) ICH occurred within 24 hours after onset; (3) patients without prior history of ICH; hemorrhage was located in the basal ganglia diagnostically and confirmed by head computed tomography (CT); (4) the hematoma did not rupture into the ventricle.
Exclusion criteria: (1) treated with surgery; (2) in a coma or died within 14 days; (3) ICH caused by non-hypertension, e.g. as a result of a brain tumor, trauma, substance abuse, abnormal blood clotting, anticoagulation therapy or vascular malformation; (4) three months before recruitment, patient suffered from an infectious disease, an autoimmune disease or used hormone therapy; (5) acute myocardial infarction, liver damage, kidney damage or bacterial sepsis/infection; (6) diabetes; (7) consent not given.
Detection of hematoma volume and hematoma absorption rate
A cranial CT (64-slice spiral CT, Siemens, Germany) scan was performed on the day of ICH onset and 14 days after onset. We used hematoma volume = π/6 × length (cm) × width (cm) × height (cm) to measure hematoma volume and [hematoma volume (on the 14th day) – hematoma volume (on the day of admission)]/hematoma volume (on the day of admission) × 100% to calculate hematoma absorption rate.
On the day of onset and 7, 14 and 30 days after onset, the National Institutes of Health Stroke Scale (NIHSS) was used to evaluate severity of ICH (scores range from 0 to 25, higher scores indicate more severe stroke). We used modified Rankin Scale (mRS) scores (scores range from 0 to 6, higher scores indicate poorer recovery) to measure the recovery of neurological function of patients 90 days after the onset of the ICH (Lyden et al., 2007). The scale consists of six grades, arranged from 0–5, where 0 corresponds to no symptoms and 5 corresponds to severe disability (Sulter et al., 1999).
Determination of soluble CD163
Forearm venous blood was sampled post fast to measure plasma levels of soluble CD163 within 24 hours after ICH onset. Blood samples were centrifuged at 1,000 × g for 15 minutes and the supernatant stored at −20°C. Soluble CD163 was quantified using a highly sensitive enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Minneapolis, MN, USA) (Møller et al., 2002a). All steps were completed according to the instructions of the ELISA kit. Absorbance was measured at 450 nm. Absorbance values of standard concentrations were used to draw a standard curve and sample CD163 concentrations were determined from their absorbance values against the standard curve. All the steps were repeated twice and mean levels used.
Statistical analysis
Values for the concentration of soluble CD163 are expressed as the mean ± SD. All data were analyzed with SPSS 17.0 software (SPSS, Chicago, IL, USA). The normality test was used to determine if data were normally distributed. Normally distributed data were analyzed with the paired t-test. Correlation between soluble CD163 levels and hematoma absorption rate was assessed using Pearson correlation analysis. P < 0.05 was considered statistically significant.
Results
Quantitative analysis of participants
All patients (18 females and 36 males; aged 28–79 years) were included in the final analysis. There was no significant difference in sex ratio, average age or past medical history (stroke, hypertension, diabetes, dyslipidemia, drinking, and smoking) between the low-level group (L group) and high-level group (H group).
Blood level of soluble CD163
According to the average CD163 level among patients (1,977.79 ± 832.91 ng/mL), all patients were divided into two groups: L group (below the average soluble CD163 level, n = 28) and H group (above the average soluble CD163 level, n = 26). A significant difference in the concentration of soluble CD163 was observed between the L group (1,347.90 ± 963.13 ng/mL) and the H group (2,656.13 ± 1,002.04 ng/mL) (P < 0.01).
Absorption of hematoma
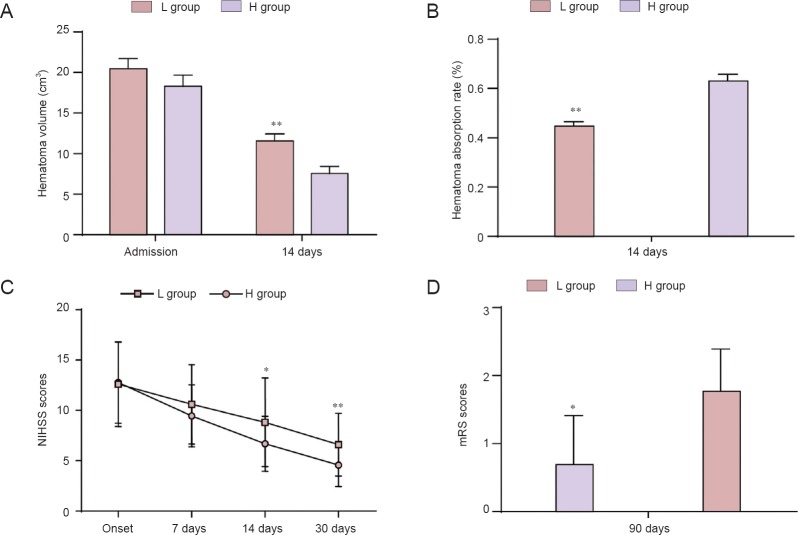
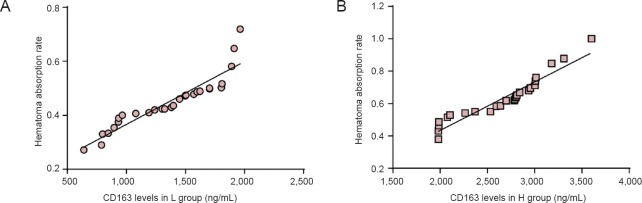
On the day of ICH onset, no significant difference was observed in the hematoma volume between the L and H groups (P > 0.05), but on day 14, hematoma volume in the L group (11.68 ± 4.63 mL) was significantly larger compared with that in the H group (P < 0.01) (Figures 1, 2A). According to changes in hematoma volume, we calculated the hematoma absorption rate and found that the hematoma absorption rate in the L group was significantly lower compared with that in the H group (P < 0.01) (Figure 2B). Furthermore, correlations between soluble CD163 levels and hematoma absorption rate were observed in the H group (r2 = 0.91, P < 0.01) and the L group (r2 = 0.86, P < 0.01) (Figure 3).
Figure 1.
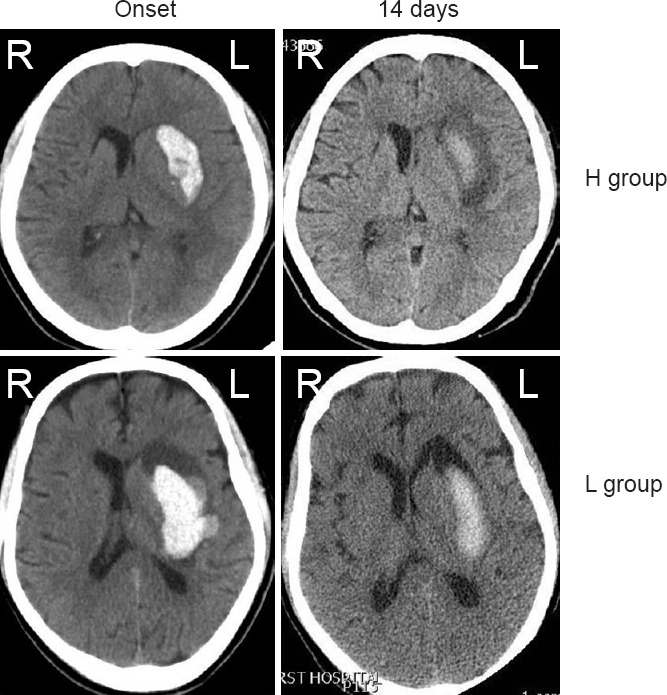
Typical cranial computed tomography images of intracerebral hemorrhage patients showing the hemorrhage absorption process.
On the onset day, there was no significant difference in the hematoma volume between groups L and H. However, on day 14, hematoma volume was larger in the L group compared with that in the H group. L group: Low-level group, below the average level of soluble CD163; H group: high-level group, above the average level of soluble CD163. L: Left; R: right.
Figure 2.
Comparison of hematoma absorption, NIHSS scores and mRS scores between L and H groups.
(A) On day 14, hematoma volume was significantly higher in the L group than in the H group (P < 0.01). (B) Hematoma absorption rate was significantly lower in the L group than in the H group. (C) The NIHSS scores were significantly higher in the L group than in the H group at 14 and 30 days. (D) The mRS scores were also significantly higher in the L group than in the H group at 90 days. *P < 0.05, **P < 0.01, vs. H group. Data are expressed as the mean ± SD (L group: n = 28, H group: n = 26; paired t-test). L group: Low-level group, below the average level of soluble CD163 (1,977.79 ± 832.91 ng/mL); H group: high-level group, above the average level of soluble CD163 (1,977.79 ± 832.91 ng/mL). NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale.
Figure 3.
Correlations between soluble CD163 levels and hematoma absorption rate in the H and L groups.
There is a positive linear correlation between CD163 levels and absorption rate of hematoma in the L group (A) and H group (B) (H group: n = 26, r2 = 0.91, P < 0.01; L group: n = 28, r2 = 0.86, P < 0.01; Pearson linear correlation analysis). L group: Low-level group, below the average level of soluble CD163; H group: high-level group, above the average level of soluble CD163.
Improvement of neurological deficit
NIHSS scores were higher in the L group compared with those in the H group at 7, 14 and 30 days and were significantly different at 14 and 30 days (P < 0.05; Figure 2C). The mRS scores were also significantly higher in the L group compared with those in the H group at 90 days (P < 0.05; Figure 2D).
Discussion
Many studies have shown that primary and secondary damage resulting from brain injury, including hemorrhagic volume and hematoma expansion, cerebral edema, inflammation, and cellular apoptosis, ultimately lead to blood-brain barrier disruption and massive brain cell death (Kaindl et al., 2012; Vitner et al., 2012; Roth et al., 2014). The pathophysiology of cerebral hemorrhage is very complex (Zou et al., 2015). Within the first few hours after ICH, primary brain injury occurs as the bleed in the brain tissue clots forming a hematoma causing a physical mass effect on the surrounding tissue (Xi et al., 2006; Keep et al., 2012). During coagulation, blood clots contract and plasma permeates into the brain tissue around the hematoma. This increases the internal hydrostatic pressure of peri-hematomal tissue and brain edema occurs, leading to secondary ischemia. In addition, blood components also contribute to ICH-induced secondary injury (Xi et al., 2006; Aronowski and Zhao, 2011; Babu et al., 2012); for example, the lysis of erythrocytes after ICH leads to the release of Hb, causing tissue damage (Aronowski and Zhao, 2011). It has been shown that the monocyte-macrophage scavenger receptor, CD163, is involved in protecting against the deleterious effects of ‘free’ Hb (Kristiansen et al., 2001; Schaer et al., 2013). CD163 is expressed only on cells of the monocyte/macrophage lineage and its expression increases as monocytes differentiate into macrophages (Graversen and Moestrup, 2015). Under physiological conditions, microglial cells do not express CD163, until activation by Hb stimulation (Xue and Del Bigio, 2000). Hp is the primary Hb-binding protein in plasma. Hb-bound Hp targets a specific clearance pathway through CD163. Hb is then converted by the enzyme heme oxygenase-1 into neurotoxic components, such as heme and iron which are major contributors to secondary brain injury (Kenneth, 2003; Wu et al., 2003; Keep et al., 2012). Under physiological conditions, approximately 10% of erythrocytes are degraded through this pathway, corresponding to approximately 0.5–1.0 gram of Hb being cleared by CD163 every day (Garby and Noyes, 1959a, b; Thomsen et al., 2013). However, under pathological conditions, CD163-mediated Hb clearance is significantly increased. Liu et al. (2015) found that CD163 expression in brain tissue around a hematoma gradually increased after ICH. Augmenting the capacity of the intrathecal CD163-Hp–Hb scavenging system prevents delayed cerebral ischemia (Galea et al., 2012).
Soluble CD163 is present in plasma (Møller et al., 2002b) and, related to macrophage activity, its concentration increases during disease (Møller, 2012). The physiological role of soluble CD163 is not yet clear. Soluble CD163 can combine with Hp-Hb complexes, but to a far lesser degree than CD163 on the cell membrane (Møller et al., 2010). In a large cohort study, soluble CD163 was shown to positively correlate with insulin resistance, which is the fundamental problem in type-2 diabetes (Moestrup, 1994; Zanni et al., 2012). Plasma levels of soluble CD163 were markedly increased and correlated with disease severity and prognosis in acute-on-chronic hepatitis B liver failure patients (Ye et al., 2013). The levels of soluble CD163 were also increased in acute inflammations, such as bacterial sepsis/infection (Møller et al., 2006). Glucocorticoids have been reported to regulate the expression of CD163. The upregulation of CD163 by glucocorticoid has been verified by human volunteers injecting prednisone (Zwadlo-Klarwasser et al., 1990). Based on this, we excluded patients with diabetes, liver damage and other conditions that might affect shedding of CD163. Currently, there are not many studies on the correlation between CD163 and ICH, especially in vivo studies. We believe that CD163 plays an important role in the absorption of hematoma after ICH and also affects the prognosis of ICH. Our aim is to identify a biological marker to predict the prognosis of ICH.
In our study, the absorption of hematoma in patients with higher concentrations of soluble CD163 was increased and the neurological deficits more improved compared with patients with lower CD163 levels. The level of soluble CD163 in the serum of patients with ICH was positively correlated with hematoma absorption rate. This indicated that CD163 accelerates absorption and promotes removal of hematoma. Rapid neurological recovery has been observed in patients with fast absorption of hematoma. We believe that CD163 can improve the prognosis of patients with cerebral hemorrhage. Our study provides evidence that CD163 is a potential target for the treatment of ICH. Further studies are necessary to examine how CD163 functions in the absorption of hematoma.
There were some limitations of our study. First, the measurements of CD163 serum levels by ELISA were not stable due to interference by many factors. Although we minimized their impact, there were still many non-specific factors. In addition, more research is needed to confirm the effect of factors on the relationship between CD163 and ICH. Second, the number of patients enrolled was small, resulting in potential bias. Given the limitations of our study, further studies are necessary to verify our results.
In spite of these limitations, our study revealed that CD163 functions in promoting hematoma absorption and improving the prognosis of patients with ICH and may provide possible therapeutic options for future treatment of ICH.
Footnotes
Conflicts of interest: None declared.
Plagiarism check: This paper was screened twice using Cross-Check to verify originality before publication.
Peer review: This paper was double-blinded and stringently reviewed by international expert reviewers.
Copyedited by Allen J, Haase R, Wang J, Qiu Y, Li CH, Song LP, Zhao M
References
- 1.Aronowski J, Zhao X. Molecular pathophysiology of cerebral hemorrhage secondary brain injury. Stroke. 2011;42:1781–1786. doi: 10.1161/STROKEAHA.110.596718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Babu R, Bagley JH, Di C, Friedman AH, Adamson C. Thrombin and hemin as central factors in the mechanisms of intracerebral hemorrhage-induced secondary brain injury and as potential targets for intervention. Neurosurg Focus. 2012;32:E8. doi: 10.3171/2012.1.FOCUS11366. [DOI] [PubMed] [Google Scholar]
- 3.Droste A, Sorg C, Högger P. Shedding of CD163 a novel regulatory mechanism for a member of the scavenger receptor cysteine-rich family. Biochem Biophys Res Commun. 1999;256:110–113. doi: 10.1006/bbrc.1999.0294. [DOI] [PubMed] [Google Scholar]
- 4.Galea J, Cruickshank G, Teeling JL, Boche D, Garland P, Perry VH, Galea I. The intrathecal CD163-haptoglobin–hemoglobin scavenging system in subarachnoid hemorrhage. J Neurochem. 2012;121:785–792. doi: 10.1111/j.1471-4159.2012.07716.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Garby L, Noyes WD. Studies on hemoglobin metabolism. I. The kinetic properties of the plasma hemoglobin pool in normal man. J Clin Invest. 1959a;38:1479. doi: 10.1172/JCI103925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Garby L, Noyes WD. Studies on hemoglobin metabolism. II. Pathways of hemoglobin iron metabolism in normal man. J Clin Invest. 1959b;38:1484. doi: 10.1172/JCI103926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Graversen JH, Moestrup SK. Drug trafficking into macrophages via the endocytotic receptor CD163. Membranes. 2015;5:228–252. doi: 10.3390/membranes5020228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Högger P, Dreier J, Droste A, Buck F, Sorg C. Identification of the integral membrane protein RM3/1 on human monocytes as a glucocorticoid-inducible member of the scavenger receptor cysteine-rich family (CD163) J Immunol. 1998;161:1883–1890. [PubMed] [Google Scholar]
- 9.Kaindl AM, Degos V, Peineau S, Gouadon E, Chhor V, Loron G, Le Charpentier T, Josserand J, Ali C, Vivien D. Activation of microglial N-methyl-D-aspartate receptors triggers inflammation and neuronal cell death in the developing and mature brain. Ann Neurol. 2012;72:536–549. doi: 10.1002/ana.23626. [DOI] [PubMed] [Google Scholar]
- 10.Keep RF, Hua Y, Xi G. Intracerebral haemorrhage: mechanisms of injury and therapeutic targets. Lancet Neurol. 2012;11:720–731. doi: 10.1016/S1474-4422(12)70104-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kenneth R. Heme and iron metabolism: role in cerebral hemorrhage review article. J Cereb Blood Flow Metab. 2003;23:629–652. doi: 10.1097/01.WCB.0000073905.87928.6D. [DOI] [PubMed] [Google Scholar]
- 12.Kjærgaard AG, Rødgaard-Hansen S, Dige A, Krog J, Møller HJ, Tønnesen E. Monocyte expression and soluble levels of the haemoglobin receptor (CD163/shedding CD163) and the mannose receptor (MR/sMR) in septic and critically ill non-septic ICU patients. PLoS One. 9:e92331. doi: 10.1371/journal.pone.0092331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kristiansen M, Graversen JH, Jacobsen C, Sonne O, Hoffman HJ, Law SA, Moestrup SK. Identification of the haemoglobin scavenger receptor. Nature. 2001;409:198–201. doi: 10.1038/35051594. [DOI] [PubMed] [Google Scholar]
- 14.Lei B, Dawson HN, Roulhac-Wilson B, Wang H, Laskowitz DT, James ML. Tumor necrosis factor alpha antagonism improves neurological recovery in murine intracerebral hemorrhage. J Neuroinflammation. 2013;10:103. doi: 10.1186/1742-2094-10-103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu B, Hu B, Shao S, Wu W, Fan L, Bai G, Shang P, Wang X. CD163/hemoglobin oxygenase-1 pathway regulates inflammation in hematoma surrounding tissues after intracerebral hemorrhage. J Stroke Cerebrovasc Dis. 2015;24:2800–2809. doi: 10.1016/j.jstrokecerebrovasdis.2015.08.013. [DOI] [PubMed] [Google Scholar]
- 16.Lyden PD, Shuaib A, Lees KR, Davalos A, Davis SM, Diener HC, Grotta JC, Ashwood TJ, Hardemark HG, Svensson HH. CHANT Trial Investigators (2007) Safety and tolerability of NXY-059 for acute intracerebral hemorrhage. Stroke. 38:2262–2269. doi: 10.1161/STROKEAHA.106.472746. [DOI] [PubMed] [Google Scholar]
- 17.Møller H, Hald K, Moestrup SK. Characterization of an enzyme-linked immunosorbent assay for soluble CD163. Scand J Clin Lab Invest. 2002a;62:293–299. doi: 10.1080/003655102760145852. [DOI] [PubMed] [Google Scholar]
- 18.Møller HJ. Soluble CD163. Scand J Clin Lab Invest. 2012;72:1–13. doi: 10.3109/00365513.2011.626868. [DOI] [PubMed] [Google Scholar]
- 19.Møller HJ, Peterslund NA, Graversen JH, Moestrup SK. Identification of the hemoglobin scavenger receptor/CD163 as a natural soluble protein in plasma. Blood. 2002b;99:378–380. doi: 10.1182/blood.v99.1.378. [DOI] [PubMed] [Google Scholar]
- 20.Møller HJ, Nielsen MJ, Maniecki MB, Madsen M, Moestrup SK. Soluble macrophage-derived CD163: a homogenous ectodomain protein with a dissociable haptoglobin–hemoglobin binding. Immunobiology. 2010;215:406–412. doi: 10.1016/j.imbio.2009.05.003. [DOI] [PubMed] [Google Scholar]
- 21.Møller HJ, Frikke-Schmidt R, Moestrup SK, Nordestgaard BG, Tybjærg-Hansen A. Serum soluble CD163 predicts risk of type 2 diabetes in the general population. Clin Chem. 2011;57:291–297. doi: 10.1373/clinchem.2010.154724. [DOI] [PubMed] [Google Scholar]
- 22.Møller HJ, Moestrup SK, Weis N, Wejse C, Nielsen H, Pedersen SS, Attermann J, Nexø E, Kronborg G. Macrophage serum markers in pneumococcal bacteremia: Prediction of survival by soluble CD163. Crit Care Med. 2006;34:2561–2566. doi: 10.1097/01.CCM.0000239120.32490.AB. [DOI] [PubMed] [Google Scholar]
- 23.Moestrup SK. The α2-macroglobulin receptor and epithelial glycoprotein-330: two giant receptors mediating endocytosis of multiple ligands. Biochim Biophys Acta. 1994;1197:197–213. doi: 10.1016/0304-4157(94)90005-1. [DOI] [PubMed] [Google Scholar]
- 24.Moreno JA, Muñoz-García B, Martín-Ventura JL, Madrigal-Matute J, Orbe J, Páramo JA, Ortega L, Egido J, Blanco-Colio LM. The CD163-expressing macrophages recognize and internalize TWEAK: potential consequences in atherosclerosis. Atherosclerosis. 2009;207:103–110. doi: 10.1016/j.atherosclerosis.2009.04.033. [DOI] [PubMed] [Google Scholar]
- 25.Paciaroni M, Agnelli G. Should oral anticoagulants be restarted after warfarin-associated cerebral haemorrhage in patients with atrial fibrillation. Thromb Haemost. 2014;111:14–18. doi: 10.1160/TH13-08-0667. [DOI] [PubMed] [Google Scholar]
- 26.Roth TL, Nayak D, Atanasijevic T, Koretsky AP, Latour LL, McGavern DB. Transcranial amelioration of inflammation and cell death after brain injury. Nature. 2014;505:223–228. doi: 10.1038/nature12808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schaer DJ, Buehler PW, Alayash AI, Belcher JD, Vercellotti GM. Hemolysis and free hemoglobin revisited: exploring hemoglobin and hemin scavengers as a novel class of therapeutic proteins. Blood. 2013;121:1276–1284. doi: 10.1182/blood-2012-11-451229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Specogna AV, Patten SB, Turin TC, Hill MD. Cost of Spontaneous intracerebral hemorrhage in canada during 1 decade. Stroke. 2014;45:284–286. doi: 10.1161/STROKEAHA.113.003276. [DOI] [PubMed] [Google Scholar]
- 29.Sulahian TH, Hintz KA, Wardwell K, Guyre PM. Development of an ELISA to measure soluble CD163 in biological fluids. J Immunol methods. 2001;252:25–31. doi: 10.1016/s0022-1759(01)00328-3. [DOI] [PubMed] [Google Scholar]
- 30.Sulter G, Steen C, De Keyser J. Use of the Barthel index and modified Rankin scale in acute stroke trials. Stroke. 1999;30:1538–1541. doi: 10.1161/01.str.30.8.1538. [DOI] [PubMed] [Google Scholar]
- 31.Thomsen JH, Etzerodt A, Svendsen P, Moestrup SK. The haptoglobin-CD163-heme oxygenase-1 pathway for hemoglobin scavenging. Oxid Med Cell Longev. 2013:523652. doi: 10.1155/2013/523652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.van Asch CJ, Luitse MJ, Rinkel GJ, van der Tweel I, Algra A, Klijn CJ. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurol. 2010;9:167–176. doi: 10.1016/S1474-4422(09)70340-0. [DOI] [PubMed] [Google Scholar]
- 33.Vitner EB, Farfel-Becker T, Eilam R, Biton I, Futerman AH. Contribution of brain inflammation to neuronal cell death in neuronopathic forms of Gaucher’s disease. Brain. 2012;135:1724–1735. doi: 10.1093/brain/aws095. [DOI] [PubMed] [Google Scholar]
- 34.Wu J, Hua Y, Keep RF, Nakamura T, Hoff JT, Xi G. Iron and iron-handling proteins in the brain after intracerebral hemorrhage. Stroke. 2003;34:2964–2969. doi: 10.1161/01.STR.0000103140.52838.45. [DOI] [PubMed] [Google Scholar]
- 35.Xi G, Keep RF, Hoff JT. Mechanisms of brain injury after intracerebral haemorrhage. Lancet Neurol. 2006;5:53–63. doi: 10.1016/S1474-4422(05)70283-0. [DOI] [PubMed] [Google Scholar]
- 36.Xue M, Del Bigio MR. Intracortical hemorrhage injury in rats relationship between blood fractions and brain cell death. Stroke. 2000;31:1721–1727. doi: 10.1161/01.str.31.7.1721. [DOI] [PubMed] [Google Scholar]
- 37.Ye H, Wang LY, Zhao J, Wang K. Increased CD163 expression is associated with acute-on-chronic hepatitis B liver failure. World J Gastroenterol. 2013;19:2818. doi: 10.3748/wjg.v19.i18.2818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zanni MV, Burdo TH, Makimura H, Williams KC, Grinspoon SK. Relationship between monocyte/macrophage activation marker soluble cd163 and insulin resistance in obese and normal-weight subjects. Clin Endocrinol. 2012;77:385–390. doi: 10.1111/j.1365-2265.2011.04284.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zou W, Chen QX, Sun XW, Chi QB, Kuang HY, Yu XP, Dai XH. Acupuncture inhibits Notch1 and Hes1 protein expression in the basal ganglia of rats with cerebral hemorrhage. Neural Regen Res. 2015;10:457–462. doi: 10.4103/1673-5374.153696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zwadlo-Klarwasser G, Bent S, Haubeck HD, Sorg C, Schmutzler W. Glucocorticoid-induced appearance of the macrophage subtype RM 3/1 in peripheral blood of man. Int Arch Allergy Appl Immunol. 1990;91:175–180. doi: 10.1159/000235111. [DOI] [PubMed] [Google Scholar]