Abstract
Background
Cholinesterase inhibitor-based reversal agents, given in the absence of neuromuscular block, evoke a partial upper airway obstruction by decreasing skeletal upper airway muscle function. Sugammadex reverses neuromuscular block by encapsulating rocuronium. However, its effects on upper airway integrity and breathing are unknown.
Methods
Fifty-one adult male rats were anaesthetized with isoflurane, tracheostomized, and a femoral artery and vein were cannulated. First, we compared the efficacy of sugammadex 15 mg kg−1 and neostigmine 0.06 mg kg−1 to reverse respiratory effects of rocuronium-induced partial paralysis [train-of-four ratio (T4/T1)=0.5]. Subsequently, we compared the safety of sugammadex and neostigmine given after recovery of the T4/T1 to 1, by measuring phasic genioglossus activity and breathing.
Results
During partial paralysis (T4/T1=0.5), time to recovery of minute volume to baseline values was 10.9 (2), 75.8 (18), and 153 (54) s with sugammadex, neostigmine, and placebo, respectively (sugammadex was significantly faster than neostigmine and placebo, P<0.05). Recovery of T4/T1 was also faster for sugammadex than neostigmine and placebo. Neostigmine administration after complete recovery of T4/T1 decreased upper airway dilator muscle activity to 64 (30)% of baseline and decreased tidal volume (P<0.05 for both variables), whereas sugammadex had no effect on either variable.
Conclusions
In contrast to neostigmine, which significantly impairs upper airway dilator muscle activity when given after recovery from neuromuscular block, a reversal dose of sugammadex given under the same conditions does not affect genioglossus muscle activity and normal breathing. Human studies will be required to evaluate the clinical relevance of our findings.
Keywords: airway, muscles; airway, obstruction; airway, patency; airway, pharynx; airway, reflexes; airway, tongue; neuromuscular block, antagonism: neuromuscular block, neurotransmission effects; neuromuscular block, pharmacology; neuromuscular block, recovery; ventilation, airway muscles; ventilation, pattern
Residual neuromuscular block occurs frequently after surgery and may put a patient at risk of developing postoperative respiratory complications.1–3 Reversal of neuromuscular block is therefore recommended to ensure recovery in all patients regardless of whether they are monitored objectively or show signs of weakness.4–6 The standard practice of reversal with cholinesterase inhibitors (ChEI) is relatively safe in patients who have residual block, but if these agents are given after complete spontaneous recovery, they can actually produce muscle weakness7,8 and possibly upper airway collapse.9 Until recently, however, adverse effects of ChEI-based reversal on muscle function were not well substantiated or universally accepted.7
We have recently demonstrated in the rat that neostigmine can evoke partial neuromuscular transmission failure and impair genioglossus function.9 The mechanisms proposed for this effect include desensitization of acetylcholine receptors,10 depolarization block,11 or open channel block.12 This may be a clinically relevant effect, since quantitative neuromuscular transmission monitoring is not always utilized in clinical practice,13,14 and patients fully recovered from neuromuscular block may thus occasionally receive ChEIs.
Sugammadex (Org 25969) reverses neuromuscular block by encapsulating rocuronium.15–17 On the basis of this mechanism of action, we hypothesized that sugammadex would not affect upper airway dilator muscles, even when administered in the absence of neuromuscular block. The effects of sugammadex alone on breathing and upper airway dilator muscle function have not been studied. This preclinical study compares the effects of sugammadex with those of neostigmine:
on breathing when reversal agents are administered during partial paralysis (T4/T1=0.5; efficacy study);
on upper airway dilator muscle function when reversal agents are administered after recovery of the T4/T1 to 1 from rocuronium-induced neuromuscular block (safety study).
Methods
The Institutional Animal Care and Use Committees at Harvard and Beth Israel Deaconess Medical Centre approved all procedures involving animals. Fifty-one spontaneously breathing adult male Sprague–Dawley rats (300–400 g; Harlan Sprague Dawley, Indianapolis, IN, USA) were used.
Experimental preparation
After induction of anaesthesia with 2–3.5 vol% isoflurane, the trachea and a femoral artery and vein were cannulated proximally. Body temperature was measured with a rectal probe and maintained at a mean (sem) of 37 (1)°C by a thermostatically controlled heating blanket (CWE Inc., Ardmore, PA, USA). In protocol 2 (described below), we also inserted electromyographic (EMG) recording electrodes into the diaphragm and the genioglossus muscles (one on each side of the midline by open surgery). After surgery, we allowed the anaesthesia to stabilize at 1.5 vol% for 30 min before we initiated the measurements.
The femoral nerve was stimulated supramaximally with needle electrodes, and the evoked response of the quadriceps femoris muscle was measured with the TOF-Watch SX Monitor (Organon Ireland Ltd, a part of Schering-Plough Corporation, Dublin, Ireland), as described previously.10 The transducer was fixed to the skin ventromedially at the proximal end of the thigh, next to the tibial tuberosity (insertion point of the patellar ligament). After determination of the supramaximal stimulation current, the femoral nerve was continuously stimulated at 1 Hz until twitch height reached a stable plateau. Thereafter, we changed the stimulation pattern to train-of-four (TOF) stimulation (2 Hz) applied for a minimum of 5 min, and calibrated the TOF-Watch SX® monitor (calibration mode 1). TOF stimulation was then continued for at least 2 min before drugs were injected.
Measurement of genioglossus EMG
EMG signals were led to differential amplifiers in a Grass Polygraph, filtered (100 Hz low pass, 1 kHz high pass), and digitized by a desktop computer equipped with Digidata A/D hardware and Axotape software (Molecular Devices, Union City, CA, USA). Voltage signals were analysed with Clampfit, Igor Pro (WaveMetrics, Inc., Lake Oswego, OR, USA), Microsoft Excel, and Sigmastat software. Phasic genioglossus activity was measured during the entire EMG burst that occurred in phase with each single breath. To measure the peak amplitude of genioglossus bursts (expressed in microvolt), we measured the peak moving time average (time base: 100 ms). Values of 10 consecutive breaths were averaged and used for statistical comparison.18
Drugs
Isoflurane (Flurane, Baxter Healthcare Corporation, Deerfield, IL, USA), rocuronium (Zemuron, Organon, NJ, USA), neostigmine (Scicor Pharmaceuticals, Irvine, CA, USA), and glycopyrrolate (American Regent, Inc., Shirley, NY, USA) were obtained from clinical supplies. Sugammadex was kindly supplied by Organon (Organon Ireland Ltd, a part of Schering-Plough Corporation).
Pilot study
To obtain an estimate of the efficacy of rocuronium at the rat quadriceps femoris muscle and its reversibility by sugammadex, we generated a cumulative dose–response curve by administering rocuronium 0.4 mg kg−1 every 30 s until the twitch response was abolished. We subsequently injected sugammadex (0, 5, 10, or 15 mg kg−1) 30 s after onset of complete neuromuscular block (n=15). On the basis of this pilot study, sugammadex 15 mg kg−1 was chosen as an appropriate reversal dose for this regimen of neuromuscular block, and this dose was used in protocols 1 and 2 (described below) for comparison with neostigmine 0.06 mg kg−1.9
Protocol 1 (efficacy): effects of sugammadex and neostigmine given at T4/T1=0.5 on T4/T1 ratio and breathing
All rats were anaesthetized with isoflurane 2–3.5 vol% during induction and surgery (inspiratory gas: 40% O2 and 60% N2), and 1.5 vol% during measurement. Rats were then paralysed with rocuronium 3.5 mg kg−1 (two times the ED90), and mechanically ventilated (SAR-830 Ventilator, CWE Inc.) until recovery of the T4/T1 ratio to 0.5. We then administered either neostigmine 0.06 mg kg−1 plus glycopyrrolate 0.012 mg kg−1 (n=7), sugammadex 15 mg kg−1 (n=7), or saline placebo (n=4). T4/T1 ratio, arterial pressure, respiratory flow, diaphragm and genioglossus EMG (GG-EMG), and end-tidal CO2 concentration were measured continuously until recovery of the T4/T1 to 1 for at least 15 min.
Protocol 2 (safety): effects of sugammadex and neostigmine administration at T4/T1=1 on GG-EMG and breathing
Anaesthesia and rocuronium doses were the same as in protocol 1. Within 30 s of spontaneous recovery of the T4/T1 ratio to 1, we administered neostigmine 0.06 mg kg−1 and glycopyrrolate 0.012 mg kg−1 (n=9), or sugammadex 15 mg kg−1 (n=9). Respiratory and upper airway muscle function was recorded until 15 min after reversal agent injection.
Arterial blood gas samples were obtained (OPTI CCA-TS, Osmetech, Roswell, GA, USA) before injection of rocuronium, during mechanical ventilation, and after injection of sugammadex.
Statistical analysis
The primary outcome was safety of reversal of neuromuscular block after recovery of the T4/T1 ratio to 1. We tested the primary hypothesis that phasic GG-EMG is significantly lower after injection of neostigmine 0.06 mg kg−1 compared with sugammadex 15 mg kg−1, when administered in this circumstance. The percentage decrease in genioglossus activity was compared using the t-test for independent samples. The sample size estimation was based on the assumption9 of a 20% difference in minimum phasic GG-EMG between neostigmine and sugammadex groups, and a standard deviation of 12%. A sample size of nine rats was calculated to provide a power of 90% (alpha error P=0.05, two-sided). All other comparisons were performed with an exploratory intent. Statistical significance was evaluated with an independent sample and matched samples t-tests, as appropriate (P<0.05). Data are expressed as mean (sem).
Results
Fifty-one rats were included in this study and experiments were successfully completed in all animals.
Pilot study
On the basis of the cumulative rocuronium dose–response curve, we calculated by logistic regression analysis that rocuronium 1.75 mg kg−1 has a 90% probability to evoke complete neuromuscular block at the quadriceps femoris muscle (T4/T1=0). Sugammadex 0, 5, 10, and 15 mg kg−1 fully reversed complete rocuronium-induced [3.5 mg kg−1 (two times the ED90)] neuromuscular block within 16 (1.5), 10 (3.4), 6 (0.6), and 2.5 (1) min, respectively. On the basis of these recovery times, we chose sugammadex 15 mg kg−1 as an appropriate reversal dose that should be used for comparison with neostigmine.
Protocol 1 (efficacy): effects of sugammadex and neostigmine given at T4/T1=0.5 on recovery of T4/T1 and breathing
Before rocuronium injection, minute volume amounted to 25.6 (1.9) ml. At T4/T1=0.5, minute volume was significantly decreased from pre-rocuronium baseline and amounted to 78 (6)% of baseline. Accordingly, end-tidal CO2 was increased at T4/T1=0.5 amounting to 142 (15)% of baseline.
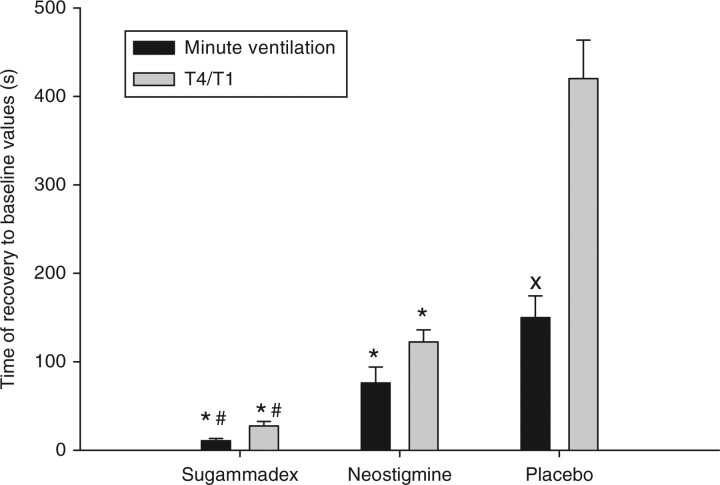
Time to recovery of minute volume to baseline values was significantly shorter with sugammadex 15 mg kg−1 compared with neostigmine 0.06 mg kg−1 and placebo amounting to 10.9 (2), 75.8 (18), and 153 (54) s, respectively (Fig. 1). The recovery of minute ventilation occurred significantly earlier than recovery of the T4/T1 to baseline, and this was particularly true for the placebo group (Fig. 1).
Fig 1.
Effects of reversal of partial paralysis (T4/T1=0.5) on breathing and T4/T1 ratio (efficacy data). Recovery time of minute volume to baseline values observed before administration of neuromuscular blocking agents was significantly shorter with sugammadex and neostigmine compared with placebo. The accelerating effect was significantly stronger with sugammadex compared with neostigmine. The effect of reversal agents on breathing paralleled those on T4/T1 recovery. In the placebo group, the T4/T1 recovery time was significantly longer than the time to recovery of the minute volume. *P<0.05 vs placebo; #P<0.05 vs neostigmine; XP<0.05 vs T4/T1 (placebo group).
Protocol 2 (safety): respiratory effects of sugammadex and neostigmine given after recovery of T4/T1 to 1
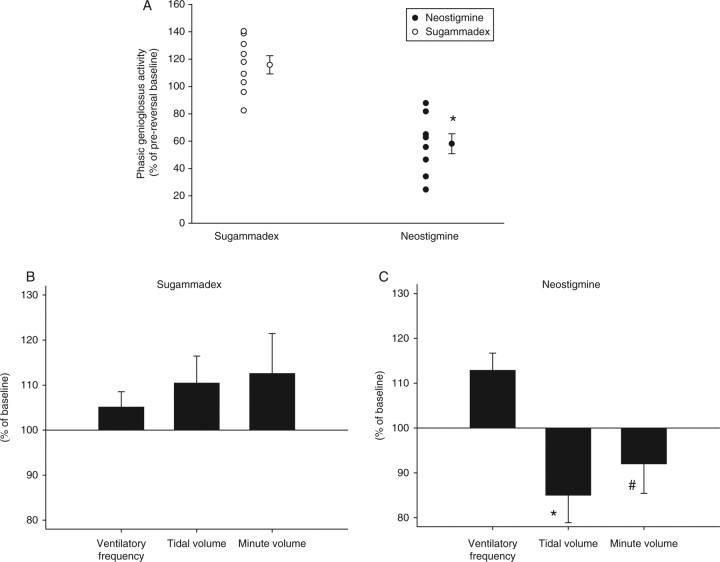
Neostigmine plus glycopyrrolate significantly decreased phasic genioglossus activity, but sugammadex did not (Fig. 2). In the neostigmine group, baseline genioglossus activity was achieved after 13 (2) min. Differences in phasic genioglossus activity observed between the two drugs were paralleled by significantly lower values of tidal volume (P<0.05) for neostigmine. There was also a trend towards lower minute volume with neostigmine (P=0.08, Fig. 2). At 2.5 min after injection of reversal agents, heart rate was significantly (P<0.001) higher in the neostigmine+glycopyrrolate group compared with sugammadex amounting to 106 (2.2) vs 99 (2) beats min−1, respectively. Mean arterial pressure did not differ between the groups, amounting to 105 (5.5) mm Hg after neostigmine vs 105 (4.2) mm Hg after sugammadex.
Fig 2.
Effects of sugammadex and neostigmine reversal after recovery of T4/T1 to 1 (safety data). Drugs were given after spontaneous recovery of the T4/T1 to 1. Values are given as per cent of values observed immediately after injection of reversal agents. (a) Primary outcome. Effects on upper airway dilator muscle activity. Neostigmine significantly decreases EMG activity of the genioglossus muscle, whereas sugammadex does not have significant effects. Individual values, means and sem. (b and c) Effects on ventilation. Tidal volume was significantly lower, and minute volume tended to be lower after neostigmine reversal compared with sugammadex. *P<0.05 vs sugammadex; #P<0.1 vs sugammadex.
Discussion
These data show that a reversal dose of sugammadex, given after complete recovery from rocuronium-induced neuromuscular block, does not affect upper airway dilator muscle activity, whereas neostigmine reduces genioglossus muscle activity by approximately one-third. Sugammadex reverses the respiratory effects of partial paralysis more rapidly than neostigmine.
Neuromuscular transmission in rats is relatively resistant to non-depolarizing neuromuscular blocking agents.19 Accordingly, compared with humans, rats required higher doses of rocuronium to achieve complete neuromuscular block, and consequently, higher sugammadex doses were needed for reversal. Rocuronium and sugammadex doses used in our study have been selected based on our pilot experiments. After complete rocuronium-induced neuromuscular block (T4/T1=0), sugammadex produced complete recovery in approximately 2.5 min, a time that agrees with recovery data in humans.20 Therefore, this dose was chosen as the appropriate reversal dose in rats. Our data show that sugammadex 15 mg kg−1 reverses a partial paralysis (T4/T1=0.5) in <60 s (Fig. 1).
In agreement with our previous report,9 we observed that the ChEI neostigmine causes a decrease in upper airway dilator muscle activity. The degree of impairment of GG-EMG observed after injection of neostigmine 0.06 mg kg−1 (Fig. 2) was lower than that we found previously.9 Residual concentrations of rocuronium may have been sufficient for partial antagonism of neostigmine-induced block.
Neostigmine impaired upper airway dilator muscle function without affecting minute volume. This finding suggests that these muscles are more susceptible to neuromuscular blocking agents2,21 and ChEI than the respiratory pump muscles.2,21 In the dog, the upper airway dilating geniohyoid muscle is more sensitive to vecuronium than the diaphragm.21 Moreover, the EMG of the genioglossus, the muscle that has been assessed in this study, is markedly impaired by neostigmine at a level that does not induce diaphragmatic dysfunction or a decrease in lung volume.9 The high susceptibility of the upper airway dilator muscles to non-depolarizing neuromuscular blocking agents2,21,22 and neostigmine9 may be due to a number of factors, including nerve discharge rate, chemosensitivity, blood flow, fibre size, and acetylcholine receptor density. Although our study does not address these mechanisms, we speculate that the high firing frequency of the genioglossus motor units (15–25 Hz during quiet breathing vs a diaphragmatic firing rate of only at 8–13 Hz)23 may account for the high sensitivity.
In the placebo group, the T4/T1 recovery time was significantly longer than the time to recovery of minute volume, indicating a ‘respiratory sparing effect’,24 reflecting the relative ease with which weakened respiratory muscles are able to drive the normal respiratory system in the supine subject.
Reversal of residual neuromuscular block is an important goal in terms of patients’ postoperative safety, since it is associated with a decreased risk of 24 h postoperative morbidity and mortality.6 Omitting antagonism introduces a significant risk of residual paralysis even with short-acting neuromuscular blocking agents.25 Optimally, administration of reversal agents should be guided by evaluation of the T4/T1 ratio.26,27
In this study, we were interested in addressing the clinically interesting question as to whether adverse effects of neostigmine on neuromuscular transmission may outweigh its beneficial effects when administered during minimal neuromuscular block (TOF=0.5). Our data show that both neostigmine and sugammadex improve neuromuscular transmission and consequently respiratory function during minimal neuromuscular block. Therefore, reversal under these conditions should improve safety based on our observations in the rat.
In contrast, neostigmine reversal following complete recovery impairs upper airway dilator function and upper airway size,9 which may put the upper airway at risk of collapse. This may be clinically important, because quantitative neuromuscular transmission monitoring is not uniformly applied in clinical practice.13,14 Thus, ChEIs are routinely administered by clinicians at the end of surgical cases to reverse suspected effects of neuromuscular blocking agents regardless of whether such residual effects have been demonstrated.
Sugammadex administered in adequate dosage rapidly decreases the concentration of free or unbound rocuronium to values below the threshold necessary to achieve significant receptor occupancy. Although the efficacy of sugammadex is high,28–30 we also know that rare side-effects from any new drug that becomes clinically available are not usually detected until several thousand patient exposures have occurred.17 Therefore, the clinician, and also the regulatory authorities, will have to perform a risk–benefit analysis in due time. For that purpose, effects and side-effects will be evaluated in direct comparison with comparators that have already been approved for the same indication. Our data may provide some useful information on this issue. Sugammadex provides a better risk–benefit ratio with respect to respiratory function than the appropriate comparator, a cholinesterase inhibitor.
Species-dependent effects of neuromuscular blocking agents have been reported and rats are believed to be more resistant to depolarizing neuromuscular blocking drugs than humans.19 In theory, neostigmine and sugammadex effects on GG-EMG may be different in humans and rats, so dose–response studies in humans are required.
In summary, our data show that sugammadex given after recovery from rocuronium-induced neuromuscular block does not affect upper airway dilator muscle activity, but neostigmine significantly impairs genioglossus muscle activity. Furthermore, sugammadex reverses the respiratory effects from partial paralysis more rapidly than neostigmine.
Funding
This work was supported by a grant from Organon International Inc., a part of Schering-Plough Corporation. Organon did not participate in generation of the protocol, data analysis and interpretation, or writing.
References
- 1.Sundman E, Witt H, Olsson R, Ekberg O, Kuylenstierna R, Eriksson LI. The incidence and mechanisms of pharyngeal and upper esophageal dysfunction in partially paralyzed humans: pharyngeal videoradiography and simultaneous manometry after atracurium. Anesthesiology. 2000;92:977–84. doi: 10.1097/00000542-200004000-00014. [DOI] [PubMed] [Google Scholar]
- 2.Eikermann M, Vogt FM, Herbstreit F, et al. The predisposition to inspiratory upper airway collapse during partial neuromuscular blockade. Am J Respir Crit Care Med. 2007;175:9–15. doi: 10.1164/rccm.200512-1862OC. [DOI] [PubMed] [Google Scholar]
- 3.Berg H. Is residual neuromuscular block following pancuronium a risk factor for postoperative pulmonary complications? Acta Anaesthesiol Scand Suppl. 1997;110:156–8. doi: 10.1111/j.1399-6576.1997.tb05541.x. [DOI] [PubMed] [Google Scholar]
- 4.Viby-Mogensen J. Postoperative residual curarization and evidence-based anaesthesia. Br J Anaesth. 2000;84:301–3. doi: 10.1093/oxfordjournals.bja.a013428. [DOI] [PubMed] [Google Scholar]
- 5.Eriksson LI. Evidence-based practice and neuromuscular monitoring: it’s time for routine quantitative assessment. Anesthesiology. 2003;98:1037–9. doi: 10.1097/00000542-200305000-00002. [DOI] [PubMed] [Google Scholar]
- 6.Arbous MS, Meursing AE, van Kleef JW, et al. Impact of anesthesia management characteristics on severe morbidity and mortality. Anesthesiology. 2005;102:257–68. doi: 10.1097/00000542-200502000-00005. quiz 491–2. [DOI] [PubMed] [Google Scholar]
- 7.Caldwell JE. Reversal of residual neuromuscular block with neostigmine at one to four hours after a single intubating dose of vecuronium. Anesth Analg. 1995;80:1168–74. doi: 10.1097/00000539-199506000-00018. [DOI] [PubMed] [Google Scholar]
- 8.Churchill-Davidson HC, Christie TH. The diagnosis of neuromuscular block in man. Br J Anaesth. 1959;31:290–301. doi: 10.1093/bja/31.7.290. [DOI] [PubMed] [Google Scholar]
- 9.Eikermann M, Fassbender P, Malhotra A, et al. Unwarranted administration of acetylcholinesterase inhibitors can impair genioglossus and diaphragm muscle function. Anesthesiology. 2007;107:621–9. doi: 10.1097/01.anes.0000281928.88997.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yost CS, Maestrone E. Clinical concentrations of edrophonium enhance desensitization of the nicotinic acetylcholine receptor. Anesth Analg. 1994;78:520–6. doi: 10.1213/00000539-199403000-00018. [DOI] [PubMed] [Google Scholar]
- 11.Payne JP, Hughes R, Al Azawi S. Neuromuscular blockade by neostigmine in anaesthetized man. Br J Anaesth. 1980;52:69–76. doi: 10.1093/bja/52.1.69. [DOI] [PubMed] [Google Scholar]
- 12.Legendre P, Ali DW, Drapeau P. Recovery from open channel block by acetylcholine during neuromuscular transmission in zebrafish. J Neurosci. 2000;20:140–8. doi: 10.1523/JNEUROSCI.20-01-00140.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cammu G, De Witte J, De Veylder J, et al. Postoperative residual paralysis in outpatients versus inpatients. Anesth Analg. 2006;102:426–9. doi: 10.1213/01.ane.0000195543.61123.1f. [DOI] [PubMed] [Google Scholar]
- 14.Foster JG, Kish SK, Keenan CH. National practice with assessment and monitoring of neuromuscular blockade. Crit Care Nurs Q. 2002;25:27–40. doi: 10.1097/00002727-200208000-00004. [DOI] [PubMed] [Google Scholar]
- 15.de Boer HD, van Egmond J, van de Pol F, Bom A, Booij LH. Sugammadex, a new reversal agent for neuromuscular block induced by rocuronium in the anaesthetized Rhesus monkey. Br J Anaesth. 2006;96:473–9. doi: 10.1093/bja/ael013. [DOI] [PubMed] [Google Scholar]
- 16.de Boer HD, van Egmond J, van de Pol F, Bom A, Driessen JJ, Booij LH. Time course of action of sugammadex (Org 25969) on rocuronium-induced block in the Rhesus monkey, using a simple model of equilibration of complex formation. Br J Anaesth. 2006;97:681–6. doi: 10.1093/bja/ael240. [DOI] [PubMed] [Google Scholar]
- 17.Hunter JM, Flockton EA. The doughnut and the hole: a new pharmacological concept for anaesthetists. Br J Anaesth. 2006;97:123–6. doi: 10.1093/bja/ael158. [DOI] [PubMed] [Google Scholar]
- 18.Chamberlin NL, Eikermann M, Fassbender P, White DP, Malhotra A. Genioglossus premotoneurons and the negative pressure reflex in rats. J Physiol. 2007;579:515–26. doi: 10.1113/jphysiol.2006.121889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zaimis EJ. Motor end-plate differences as a determining factor in the mode of action of neuromuscular blocking substances. J Physiol. 1953;122:238–51. doi: 10.1113/jphysiol.1953.sp004995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.de Boer HD, Driessen JJ, Marcus MA, Kerkkamp H, Heeringa M, Klimek M. Reversal of rocuronium-induced (1.2 mg/kg) profound neuromuscular block by sugammadex: a multicenter, dose-finding and safety study. Anesthesiology. 2007;107:239–44. doi: 10.1097/01.anes.0000270722.95764.37. [DOI] [PubMed] [Google Scholar]
- 21.Isono S, Kochi T, Ide T, Sugimori K, Mizuguchi T, Nishino T. Differential effects of vecuronium on diaphragm and geniohyoid muscle in anaesthetized dogs. Br J Anaesth. 1992;68:239–43. doi: 10.1093/bja/68.3.239. [DOI] [PubMed] [Google Scholar]
- 22.D’Honneur G, Gall O, Gerard A, Rimaniol JM, Lambert Y, Duvaldestin P. Priming doses of atracurium and vecuronium depress swallowing in humans. Anesthesiology. 1992;77:1070–3. doi: 10.1097/00000542-199212000-00004. [DOI] [PubMed] [Google Scholar]
- 23.Saboisky JP, Gorman RB, De Troyer A, Gandevia SC, Butler JE. Differential activation among five human inspiratory motoneuron pools during tidal breathing. J Appl Physiol. 2007;102:772–80. doi: 10.1152/japplphysiol.00683.2006. [DOI] [PubMed] [Google Scholar]
- 24.Gal TJ, Goldberg SK. Relationship between respiratory muscle strength and vital capacity during partial curarization in awake subjects. Anesthesiology. 1981;54:141–7. doi: 10.1097/00000542-198102000-00008. [DOI] [PubMed] [Google Scholar]
- 25.Tramer MR, Fuchs-Buder T. Omitting antagonism of neuromuscular block: effect on postoperative nausea and vomiting and risk of residual paralysis. A systematic review. Br J Anaesth. 1999;82:379–86. doi: 10.1093/bja/82.3.379. [DOI] [PubMed] [Google Scholar]
- 26.Kopman AF, Kopman DJ, Ng J, Zank LM. Antagonism of profound cisatracurium and rocuronium block: the role of objective assessment of neuromuscular function. J Clin Anesth. 2005;17:30–5. doi: 10.1016/j.jclinane.2004.03.009. [DOI] [PubMed] [Google Scholar]
- 27.Kopman AF. Tactile evaluation of train-of-four count as an indicator of reliability of antagonism of vecuronium- or atracurium-induced neuromuscular blockade. Anesthesiology. 1991;75:588–93. doi: 10.1097/00000542-199110000-00008. [DOI] [PubMed] [Google Scholar]
- 28.Kopman AF. Sugammadex: a revolutionary approach to neuromuscular antagonism. Anesthesiology. 2006;104:631–3. doi: 10.1097/00000542-200604000-00003. [DOI] [PubMed] [Google Scholar]
- 29.Naguib M. Sugammadex: another milestone in clinical neuromuscular pharmacology. Anesth Analg. 2007;104:575–81. doi: 10.1213/01.ane.0000244594.63318.fc. [DOI] [PubMed] [Google Scholar]
- 30.Sparr HJ, Vermeyen KM, Beaufort AM, et al. Early reversal of profound rocuronium-induced neuromuscular blockade by sugammadex in a randomized multicenter study: efficacy, safety, and pharmacokinetics. Anesthesiology. 2007;106:935–43. doi: 10.1097/01.anes.0000265152.78943.74. [DOI] [PubMed] [Google Scholar]