Abstract
Mononuclear Ru(III) complexes of the type [Ru(LL)Cl2(H2O)] (LL = monobasic tridentate Schiff base anion: (1Z)-N′-(2-{(E)-[1-(2,4-dihydroxyphenyl)ethylidene]amino}ethyl)-N-phenylethanimidamide [DAE], 4-[(1E)-N-{2-[(Z)-(4-hydroxy-3-methoxybenzylidene)amino]ethyl}ethanimidoyl]benzene-1,3-diol [HME], 4-[(1E)-N-{2-[(Z)-(3,4-dimethoxybenzylidene)amino]ethyl}ethanimidoyl]benzene-1,3-diol [MBE], and N-(2-{(E)-[1-(2,4-dihydroxyphenyl)ethylidene]amino}ethyl)benzenecarboximidoyl chloride [DEE]) were synthesized and characterized using the microanalytical, conductivity measurements, electronic spectra, and FTIR spectroscopy. IR spectral studies confirmed that the ligands act as tridentate chelate coordinating the metal ion through the azomethine nitrogen and phenolic oxygen atom. An octahedral geometry has been proposed for all Ru(III)-Schiff base complexes. In vitro anticancer studies of the synthesized complexes against renal cancer cells (TK-10), melanoma cancer cells (UACC-62), and breast cancer cells (MCF-7) was investigated using the Sulforhodamine B assay. [Ru(DAE)Cl2(H2O)] showed the highest activity with IC50 valves of 3.57 ± 1.09, 6.44 ± 0.38, and 9.06 ± 1.18 μM against MCF-7, UACC-62, and TK-10, respectively, order of activity being TK-10 < UACC-62 < MCF-7. The antioxidant activity by DPPH and ABTS inhibition assay was also examined. Scavenging ability of the complexes on DPPH radical can be ranked in the following order: [Ru(DEE)Cl2(H2O)] > [Ru(HME)Cl2(H2O)] > [Ru(DAE)Cl2(H2O)] > [Ru(MBE)Cl2(H2O)].
1. Introduction
Coordination chemistry of transition metal Schiff base complexes possessing N, O, and S-donor atoms has received consideration over the past few decades, due to the imperative roles these compounds have played in a variety of biochemical procedures like haloperoxidation [1], insulin mimicking [2, 3], fixation of nitrogen [4], inhibition of cancer growth, and prophylaxis against carcinogenesis [5, 6]. A huge variety of carbonyl compounds (>C=O) and amines (R-NH2) have been exploited in the preparation of Schiff bases [7, 8]. The reactivity of aldehyde compounds is generally faster than those of the ketones in condensation reaction, thereby resulting in the formation of Schiff bases with a centre that are less steric than the ketone's, relatively unstable and freely polymerizable [9]. This important attribute of Schiff base ligands offers prospects for prompting substrate chirality and metal centred electronic factor tuning and improving the solubility and steadiness of either homogeneous or heterogeneous catalysts [10–12].
Schiff bases have shown an interesting application as an active corrosion inhibitor that is established on their capability to spontaneously form a monolayer upon the surface to be glazed [13], as it is a type of interaction existing between an inhibitor and a metal surface known as chemisorption [14]. It is interesting to note that several commercial inhibitors contain amines and aldehydes, but seemingly because of the presence of >C=N bond, this makes Schiff bases function more resourcefully in many ways [15]. Stabilization of metal ions in various oxidation states and monitoring their reactivity for catalytic applications have been linked to Schiff bases [16]. The nitrogen-oxygen Schiff bases geometry largely relies on the diamine structural unit, nature of the ancillary ligand, and the central metal ion [17]. Schiff base-transition metal complexes have been known to be one of the most modifiable and comprehensively studied systems [18] with applications in clinical and analytical fields [19, 20]. Antioxidants derived from metal Schiff base ligand combinations have received current attention for their capability to safeguard living systems and cells from impairment caused by oxidative stress or free radicals [21].
DNA binding, cleavage potentials, scavenging potentials, and anticancer investigations of Schiff base-ruthenium(III) complexes have been accounted for [22]. Synthesis, spectral, redox, catalytic, and biological action investigation of mononuclear Ru(III)-Schiff base structures are reported [23]. 2,2′-Bipyridine and tetradentate Schiff base ancillary ligands of mixed-ligand Ru(II) complexes have been reported for their electrochemical and Na+ binding properties [24]. Catalytic and growth inhibitory activities of Ru(III) mixed ligand complexes of 2-hydroxy-1-naphthylideneimines have been reported [25].
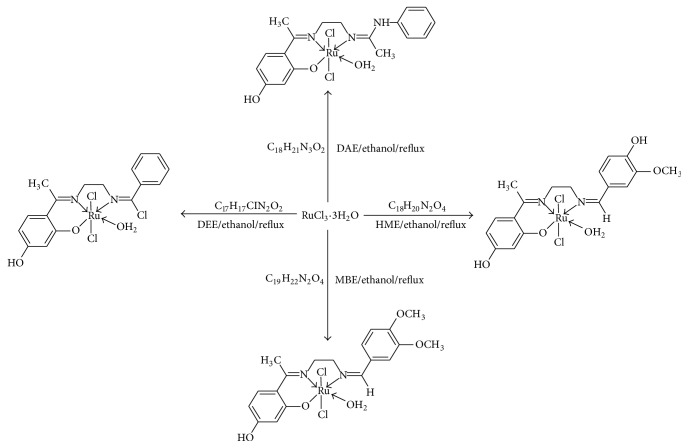
In this study, we report the synthesis, characterization, free radical scavenging, and anticancer studies of four mononuclear ruthenium(III) complexes of Schiff bases derived from 2′,4′-dihydroxyacetophenone and ethylenediamine as the bridging ligand with RCHO moiety alongside their radicals scavenging action on 1,1-diphenyl-2-picrylhydrazyl (DPPH) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) and antiproliferative potentials. The Schiff base ligands containing N2O type tridentate partitions were utilized for the synthesis of the mononuclear ruthenium(III)-Schiff base complexes (Scheme 1).
Scheme 1.
Structure of mononuclear ruthenium(III)-Schiff base complexes.
2. Experimental
2.1. Chemicals and Instrumentations
All reagents used were of analytical grade and used as purchased commercially. Ethylenediamine, N,N′-dimethylformamide (DMF) and ascorbic acid (Vit. C) were received from Merck, 2′,4′-dihydroxyacetophenone and RuCl3·3H2O were obtained from Aldrich. 1,1-Diphenyl-2-picrylhydrazyl (DPPH), 2,2′-azinobis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS), butylated hydroxytoluene (BHT), and rutin hydrate were received from Sigma Chemical Co. (St. Louis, MO, USA). Elemental analysis was carried out using Perkin-Elmer elemental analyzer. IR spectra were recorded on an FT-IR spectrometer: Perkin-Elmer System (Spectrum 2000) via KBr disk method was used for the IR spectra analysis. Freshly prepared DMF solutions of about 10−3 M containing Ru(III) complexes gave the molar conductance at room temperature with Crison EC-Meter Basic 30+ conductivity cell. Electronic absorption spectra ranging from 200 to 900 nm were recorded on a Perkin-Elmer Lambda-25 spectrophotometer. Stuart melting point (SMP 11) was used for the melting points. Four N2O type tridentate ligands, (1Z)-N′-(2-{(E)-[1-(2,4-dihydroxyphenyl)ethylidene]amino}ethyl)-N-phenylethanimidamide [DAE], 4-[(1E)-N-{2-[(Z)-(4-hydroxy-3-methoxybenzylidene)amino]ethyl}ethanimidoyl]benzene-1,3-diol [HME], 4-[(1E)-N-{2-[(Z)-(3,4-dimethoxybenzylidene)amino]ethyl}ethanimidoyl]benzene-1,3-diol [MBE], and N-(2-{(E)-[1-(2,4-dihydroxyphenyl)ethylidene]amino}ethyl)benzenecarboximidoyl chloride [DEE], were synthesized and reported previously [26].
2.2. Preparation of the Tridentate Schiff Bases (DAE, HME, MBE, and DEE)
Ethylenediamine (0.015 mol) dissolved in 20 mL of alcohol was slowly added to 2′,4′-dihydroxyacetophenone (0.015 mol) dissolved in same alcohol (30 mL) and allowed to stir for 60 minutes at room temperature and then followed by drop-wise addition of appropriate aldehyde (RCHO, 15 mmol) dissolved in 30 mL alcohol for 20 minutes time interval at room temperature and further stirred for 120 minutes. The mixture was left standing with continuous stirring for approximately 36 hours at room temperature, after which the desired tridentate compounds were filtered and washed with ethanol to give crystalline solid. The crude product was recrystallized from warm ethanol. The products were dried in the vacuum at 50°C overnight to give analytically pure products in good yields (64.2% to 73.8%).
2.3. Synthesis of Ru(III)-Tridentate Schiff Base Complexes
Ru(III) complexes were prepared by adding (0.5 mmol) ethanol solution of ruthenium(III) chloride to a warm ethanolic solution (0.5 mmol) of [DAE]/[HME]/[MBE]/[DEE], respectively. The colour of the solutions changed immediately, magnetically stirred and kept under reflux for 6 hours. The precipitated solids were filtered by suction from the reaction medium, washed with ethanol and then with diethyl ether, and dried over anhydrous calcium chloride. The yields were about 55.7–61.9%. The synthesis of the complexes is explained in Scheme 1.
2.3.1. Synthesis of [OHC6H3OH:C(CH3):N(C2H4)N:C(CH3):NHC6H5RuCl2(H2O)]
[Ru(DAE)Cl 2 (H 2 O)]·H 2 O. Dark-green solid; Yield: 156.6 mg (60.4%); F. Wt: 518.38 g; Anal. Calcd. for C18H24N3O4RuCl2 (%): C 41.71, H 4.67, N 8.11; Found (%): C 41.43, H 4.54, N 8.29; IR (KBr) ν max/cm−1: 3436 (O-H), 1621 (C=N), 1242, 1170 (C-O), 520 (Ru-N), 438 (Ru-O); UV-Vis (DMF): λ max/nm (cm−1): 281 (35 587), 310 (32 258), 391 (25 576), 452 (22 124), 525 (19 048), 613 (16 313); Decomp. Temp, °C, 238-239°C; Λμ: 31.8 μScm−1.
2.3.2. Synthesis of [OHC6H3OH:C(CH3):N(C2H4)N:CH:C6H3OHOCH3RuCl2(H2O)]
[Ru(HME)Cl 2 (H 2 O)]·H 2 O. Darkish-green Solid; Yield: 165.7 mg (61.9%); F. Wt: 535.37 g; Anal. Calcd. for C18H23N2O6RuCl2 (%): C 40.38, H 4.33, N 5.23; Found (%): C 40.58, H 4.21, N 5.44; IR (KBr) ν max/cm−1: 3422 (O-H), 1637 (C=N), 1245, 1173 (C-O), 485 (Ru-N), 437 (Ru-O); UV-Vis (DMF): λ max/nm (cm−1): 277 (36 101), 309 (32 363), 381 (26 247), 393 (25 446), 513 (19 493), 623 (16 051); Decomp. Temp, °C, 218-219°C; Λμ: 30.5 μScm−1.
2.3.3. Synthesis of [OHC6H3OH:C(CH3):N(C2H4)N:CH:C6H5(OCH3)2RuCl2(H2O)]
[Ru(MBE)Cl 2 (H 2 O)]·H 2 O. Darkish-green Solid; Yield: 160.4 mg (58.4%); F. Wt: 549.39 g; Anal. Calcd. for C19H25N2O6RuCl2 (%): C 41.54, H 4.59, N 5.10; Found (%): C 41.29, H 4.32, N 4.98; IR (KBr) ν max/cm−1: 3435 (O-H), 1639 (C=N), 1244, 1171 (C-O), 548 (Ru-N), 475 (Ru-O); UV-Vis (DMF): λ max/nm (cm−1): 277 (36 101), 311 (32 155), 380 (26 316), 393 (25 446), 510 (19 608), 623 (16 051); Decomp. Temp, °C, 226-227°C; Λμ: 30.1 μScm−1.
2.3.4. Synthesis of [OHC6H3OH:C(CH3):N(C2H4)N:C(Cl):C6H5RuCl2(H2O)]
[Ru(DEE)Cl 2 (H 2 O)]·H 2 O. Dark-green Solid; Yield: 145.9 mg (55.7%); F. Wt: 523.79 g; Anal. Calcd. for C17H20N2O4RuCl3 (%): C 38.98, H 3.85, N 5.35; Found (%): C 39.11, H 3.67, N 5.11; IR (KBr) ν max/cm−1: 3416 (O-H), 1617 (C=N), 1243, 1169 (C-O), 475 (Ru-N), 436 (Ru-O); UV-Vis (DMF): λ max/nm (cm−1): 275 (31 364), 306 (32 680), 385 (25 974), 521 (19 231), 632 (15 823); Decomp. Temp, °C, 228-229°C; Λμ: 38.8 μScm−1.
2.4. In Vitro Antiproliferative Activity
The potentials of the Ru(III)-tridentate Schiff base complexes to interfere with the growth of TK-10 renal cell line, UACC-62 melanoma cell line, and MCF-7 breast cell lines were determined by SRB assay as previously described [22]. 3–19 passages of MCF-7, TK-10, and UACC-62 cell lines with plating densities of 7–10 000 cells per well were precultured into 96-well microtitre plates for 24 h at 37°C with 95% air, 5% CO2, and 100% relative humidity in RPMI medium, supplemented with 5% fetal bovine serum (FBS), 50 μg mL−1 (gentamicin), and 2 mM L-glutamine [27]. The compounds were dissolved in DMSO and treated with the cells after 24 h and diluted in RPMI medium giving rise to 5 concentrations comprising 0.01, 0.1, 0, 10, and 100 μM.
Wells containing culture medium were used as control while the wells containing complete culture medium with no cells were used as the blanks. Parthenolide was used as the standard drug in this study. The plates were then incubated for 48 h after the addition of the compounds. Viable cells were fixed to the bottom of each well with cold 50% trichloroacetic acid, washed, dried, and dyed by SRB. The unbounded dye was separated, while the protein-bound dye was extracted with 10 mM Tris base and multiwell spectrophotometer at the wavelength 540 nm was used for its optical density determination. IC50 values were determined by plotting the percentage viability against concentration of compounds on a logarithmic graph to obtain 50% of cell growth inhibition relative to the control.
2.5. Antioxidant Assay
2.5.1. Scavenging Activity of 1,1-Diphenyl-2-picrylhydrazyl (DPPH) Radical
The antioxidant activity of the prepared Ru(III) complexes was studied using spectrophotometer by 1,1-diphenyl-2-picrylhydrazyl (DPPH) method. This compound is known as a stable readily accessible free radical, with solubility in methanol giving a purple solution, and when reacted with antioxidant species changes to an equivalent light yellow colour. The radical scavenging potentials of the complexes with DPPH radical were evaluated as described [22]. 1 mL solution of the compounds in DMF with concentrations ranging from 100 to 500 μg/mL was mixed thoroughly with equivalent amount of 0.4 mM DPPH in methanol; the mixtures were then allowed to react in the dark for half an hour. Measurement of the mixture absorbance was achieved spectrophotometrically at 517 nm. Vitamin C and rutin were used as the standard drugs. All test analysis was carried out in triplicate. The ability of the ruthenium compounds to scavenge DPPH radical was calculated via the following equation:
| (1) |
2.5.2. ABTS: 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) Radical Scavenging Assay
ABTS scavenging ability of the Ru(III)-tridentate Schiff base complexes adopted a described method [28]. 7 mM ABTS solution and 2.4 mM potassium persulfate solution in equal amounts (1 : 1) were used for working solution preparation and allowed to react in the dark for 12 h at room temperature. An absorbance of 0.706 ± 0.001 units at 734 nm required for the analysis was obtained by diluting 1 mL ABTS+ solution. Test samples (1 mL) were mixed with 1 mL of the ABTS+ solution, and absorbance was read spectrophotometrically at 734 nm. The test samples' ABTS scavenging capacity alongside standard drugs was evaluated. Triplicate analysis was carried out. The percentage inhibition of ABTS radical scavenging activity was obtained following a previous report [28].
3. Results and Discussion
3.1. Synthesis and Characterization
The obtained compounds were of coloured powders, stable in atmosphere with a general formula: [Ru(LL)Cl2(H2O)] (LL = monobasic tridentate Schiff base anion: DAE, HME, MBE, and DEE). They were prepared by treating [RuCl3·3H2O] with the corresponding Schiff base in an equal mole ratio in alcohol as depicted in the Scheme 1. All the complexes are dark-green and sparingly soluble in general organic solvents but soluble in polar aprotic solvent such as DMF and DMSO; the melting point analysis showed that the Ru(III) complexes were decomposing before melting. The physicoanalytical data collected for the compounds are in agreement with the structural formulae proposed, thus confirming the suggested mononuclear composition for the Ru(III) complexes (Scheme 1).
3.2. Molar Conductivity Measurements
The molar conductance of the synthesized Ru(III) complexes was measured in DMF at 10−3 M solution. The values were found to be in the range of 30.1–38.8 μScm−1 suggesting the nonelectrolytic nature of the complexes in solution [22, 29].
3.3. Infrared Spectra
Valuable evidence concerning the environment of the functional group attached to the ruthenium atom has been obtained from the FTIR spectra. The IR spectra of the ligands, when compared with those of the newly synthesized complexes, confirm the coordination of N2O type tridentate ligands to the ruthenium ion. The classification was achieved by comparing the spectra of the ligands with those originating from the coordination between ruthenium(III) metal ion and the active sites. The Schiff bases showed the broad bands in the 3462–3477 cm−1 range attributable to the ν(OH) cm−1 vibration. Ligand infrared spectra showed that a band at 1605–1619 cm−1 is attributed to ν(C=N) stretching of the azomethine group based on earlier reports [30]. This ν(C=N) shift to 1617–1639 cm−1 in all the complexes by about 5–23 cm−1 signifies the participation of azomethine nitrogen in the coordination sphere with the ruthenium(III) ion for all the complexes [21, 31]. A medium band that corresponds to phenolic oxygen atom ν(C-O) is observed at 1167 and 1245 cm−1 for the free ligands.
The higher shifting of ν(C-O) stretching vibrations as observed in the ruthenium(III) complexes spectra suggests that the phenolic OH group of Schiff base, DAE, HME, MBE, and DEE, is involved in coordination with ruthenium ion after deprotonation [32, 33]. Seemingly, the DAE, HME, MBE, and DEE ligands act as a tridentate chelating compound, coordinating to the metal ion via the two nitrogen atoms of the azomethine group as well as O atom of phenolic group [21, 25]. This is further supported by the displacement of ν(O-H) in the range 3462–3477 cm−1 in all the complexes. The presence of coordinated water gave a broad band that appeared in the regions 3416–3436 and 813–851 cm−1; this can be due to ν(O-H) stretching and ν(O-H) rocking vibrations, respectively, which further confirms the presence of nonligand assignable to the rocking mode of water [28, 34]. New weak nonligand bands that are not found in the DAE, HME, MBE, and DEE ligands appeared in the ranges 475–548 cm−1 and 436–475 cm−1 in the complexes spectra attributed to ν(Ru-N) and ν(Ru-O) vibrations, respectively [35, 36]. A band ranging from 311–346 cm−1 appeared in the spectra of the Ru(III)-Schiff base complexes indicating the presence of two chloride ions in trans position around ruthenium centre [37–40].
3.4. Electronic Absorption Spectra Studies
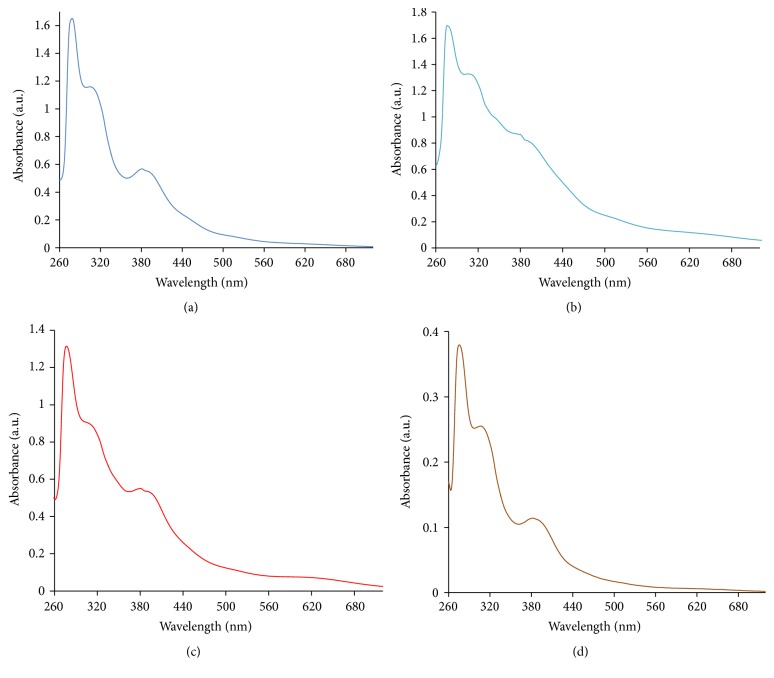
The UV-Vis spectra of the Ru(III)-Schiff base complexes in DMF solutions were recorded at room temperature ranging from 200 to 900 nm. The nature of DAE, HME, MBE, and DEE ligands field around the ruthenium ion was obtained from the electronic spectra. The free ligands showed absorption bands within the range of 277–393 nm attributable to π ∗ ← π and π ∗ ← n transitions relating the benzene ring (Figure 1). The shifting of these bands in the complexes spectra followed the participation of the imine group nitrogen and phenolic group oxygen in bonding [22, 25]. Ground state of ruthenium(III) is 2T2g, where initial excited doublet levels in order of increasing energy are 2A2g and 2T1g, arising from t2g 4eg 1 configuration [41].
Figure 1.
Electronic absorption spectra of the Ru(III) complexes: (a) [Ru(DAE)Cl2(H2O)]; (b) [Ru(HME)Cl2(H2O)]; (c) [Ru(MBE)Cl2(H2O)]; (d) [Ru(DEE)Cl2(H2O)].
Ru3+ ion, with a d5 electronic configuration, possesses high oxidizing properties and large crystal field parameter. Also, charge transfer bands of the type Lπy → T2g were noticeable within low energy region, obscuring weaker bands that is due to d-d transitions [22, 25]. The extinction coefficient bands around 613–632 nm regions are found to be low when compared to the charge transfer bands. These bands have been assigned to 2T2g → 2A2g transition and are in agreement with the assignment made for similar octahedral ruthenium(III) complexes [42, 43]. Absorption bands within the 452–525 nm regions were assigned to the charge transfer transitions [22, 44]. Overall, the absorption spectra of the Ru(III)-Schiff base complexes are typical of octahedral environment about the ruthenium(III) ions [22].
3.5. Antiproliferative Activity
Investigation into the structure-activity relationship of the isolated Ru(III)-N2O Schiff base complexes with respect to different functional groups on the ligands used for ruthenium ion complex formation has been conducted via antiproliferative studies. Three of the Ru(III)-Schiff base compounds alongside parthenolide were subjected to cell lines tests at different sample concentrations ranging from 0.01 to 100 μM towards renal cancer cell (TK-10), melanoma cancer cell (UACC-62), and breast cancer cell (MCF-7). The cancer cell lines were incubated for 48 h, followed by the addition of the compounds of various concentrations via Sulforhodamine B (SRB) assay [22].
The ruthenium(III) compounds and standard drug (parthenolide) IC50 values are presented in Table 1 and revealed that the test samples showed significant inhibition against the tested cell lines. Figures 2 –4 represent the cell viability percentages of ruthenium(III)-Schiff base complexes and parthenolide drug against TK-10, UACC-62, and MCF-7 cell lines, at different concentrations of ruthenium(III) compounds or parthenolide. A high level of antiproliferative potentials against the studied cell lines was exhibited by parthenolide in accordance with earlier reports [45]. The obtained results revealed that treatment of cell lines with different concentrations of Ru(III)-Schiff base complexes efficiently affected cell viability towards MCF-7 cells, as displayed in Figures 2 –4 and Table 1. The Ru(III) compounds exhibited low to strong in vitro antiproliferative activities against the selected cell lines as compared to the standard drug (parthenolide). [Ru(DAE)Cl2(H2O)], [Ru(HME)Cl2(H2O)], and [Ru(DEE)Cl2(H2O)] induced more efficient cell death with IC50 values of 3.57 ± 1.09, 4.88 ± 1.28, and 3.43 ± 1.48 μM, respectively, towards human breast cancer cell (MCF-7) cells than other investigated cell lines, compared with IC50 values of 0.44 ± 2.02 μM MCF-7, for the standard cytotoxin drug parthenolide.
Table 1.
In vitro antiproliferative studies of Ru(III)-Schiff base complexes against TK-10, UACC-62, and MCF-7 cell lines.
| Compounds | Molecular formula | Anticancer activity IC50 (µM) 48 h | ||
|---|---|---|---|---|
| TK-10 | UACC-62 | MCF-7 | ||
| [Ru(DAE)Cl2(H2O)] | C18H24N3O4RuCl2 | 9.06 ± 1.18 | 6.44 ± 0.38 | 3.57 ± 1.09 |
| [Ru(HME)Cl2(H2O)] | C18H23N2O6RuCl2 | 41.09 ± 4.44 | 6.31 ± 1.47 | 4.88 ± 1.28 |
| [Ru(DEE)Cl2(H2O)] | C17H20N2O4RuCl3 | 13.10 ± 2.81 | 5.14 ± 1.09 | 3.43 ± 1.48 |
| Parthenolide∗ | C15H20O3 | 0.50 ± 1.43 | 0.89 ± 2.18 | 0.44 ± 2.02 |
∗Standard cytotoxin drug: cell lines were treated with different concentrations of the compounds to achieve 50% inhibition of the culture growth when cultured for 48 h. Value represents mean ± SD of three independent experimentations.
Figure 2.
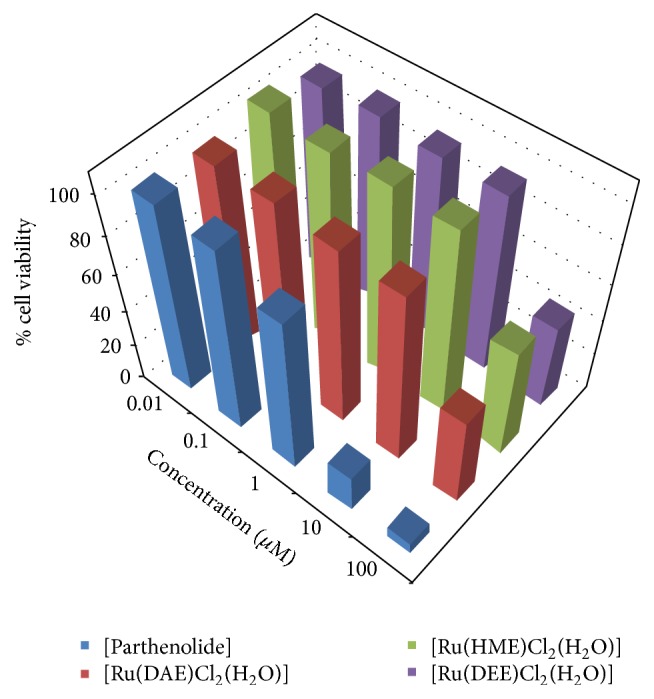
In vitro antiproliferative activity of Ru(III) complexes and parthenolide against human breast cancer cell line (MCF-7).
Figure 3.
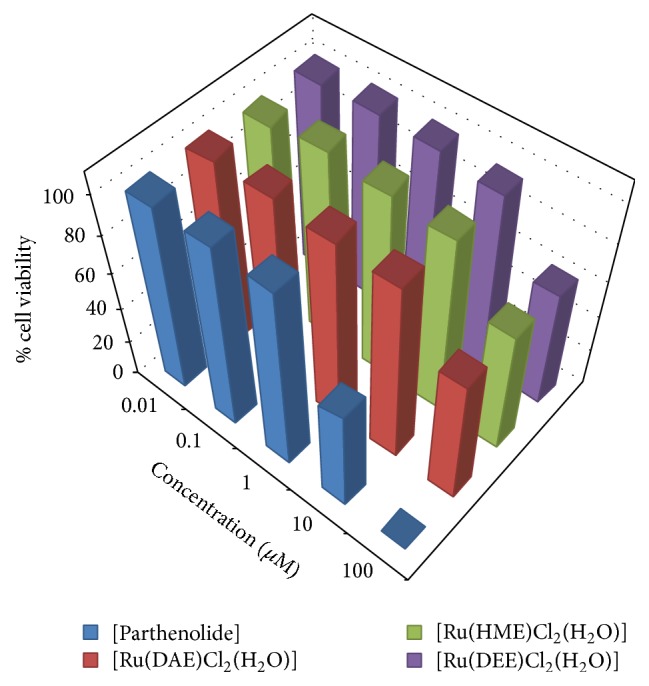
In vitro antiproliferative activity of Ru(III) complexes and parthenolide against human melanoma cancer cell (UACC-62).
Figure 4.
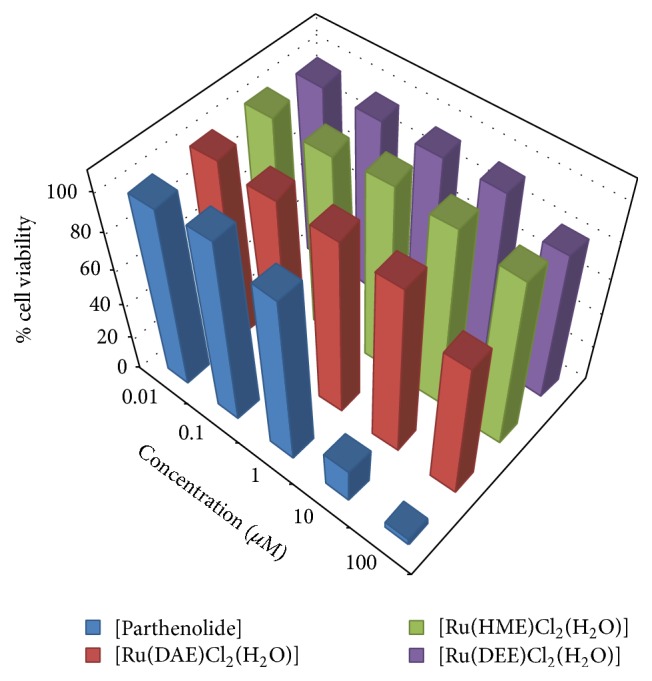
In vitro antiproliferative activity of Ru(III) complexes and parthenolide against human renal cancer cell (TK-10).
The order of activity of the complexes against human melanoma cancer cell (UACC-62) is as follows: [Ru(DEE)Cl2(H2O)] > [Ru(HME)Cl2(H2O)] > [Ru(DAE)Cl2(H2O)]. With respect to previous report by Shier [46], compounds exhibiting IC50 activity ranging from 10 to 25 μM are referred to as weak anticancer drugs, while those with IC50 action between 5 and 10 μM are moderate, and the compounds possessing activity less than (<) 5.00 μM are considered as strong agents. Thus, the Ru(III) complexes exhibited a weak to strong activity against the investigated cancer cell lines with the following order of activity: MCF-7 > UACC-62 > TK-10. However, [Ru(DAE)Cl2(H2O)] showed the highest antiproliferative activity with IC50 valves of 3.57 ± 1.09, 6.44 ± 0.38, and 9.06 ± 1.18 μM for MCF-7, UACC-62, and TK-10, respectively. The biochemical activity could be due to the methoxy, alkyl, chloride group substituents and bridge spacer: ethylenediamine, which could have played a vital role in antiproliferative potentials of the Ru(III)-N2O Schiff base complexes. In vitro anticancer activity of the synthesized Ru(III) complexes in this study was compared with Ru complexes reported by other authors and found that [Ru(DAE)Cl2(H2O)], [Ru(HME)Cl2(H2O)], and [Ru(DEE)Cl2(H2O)] complexes exhibited higher antitumor activities. [RuCl(CO)(PPh3)L] reported by Raja et al. [47] against human cervical carcinoma cell line, (HeLa) after exposure for 48 h, gave an IC50 value in the range of 31.6 μM and [RuCl2(AsPh3)L] with an IC50 value of 37.8 μM [48]. Raju et al. [43] reported ruthenium(III) Schiff base complexes of the type [RuX2(PPh3)2(L)] (where X = Cl or Br; L = monobasic bidentate ligand) complex to have IC50 value in the range of 45.2 μM.
3.6. Antioxidant Capacity
Different antioxidant techniques and modifications have been put forward to evaluate antioxidants reactivity and functionality in foods and biological systems as a means of checkmating variety of pathological activities such as cellular injury and aging process; these damaging occurrences are caused by free radicals. Hence, two free radicals were used for in vitro antioxidants activities of the test samples in this study, namely, 1,1-diphenyl-2-picrylhydrazyl (DPPH) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS).
3.6.1. DPPH Radical Scavenging Assay
The activity of antioxidants on DPPH radical is believed to be centred on their ability to donate hydrogen [22]. DPPH has been a stable free radical, with the ability to accept hydrogen radical or an electron and then become a stable molecule [49].
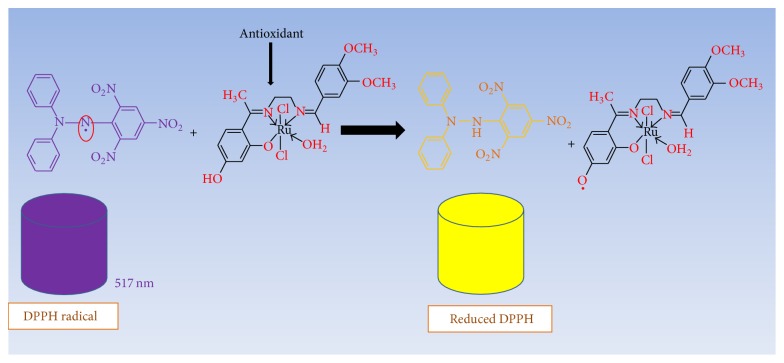
The mode of rummaging the DPPH radical has extensively been used to appraise antioxidant activities of test samples in a moderately short period of time compared to other procedures [49]. The reduction in the DPPH radical capability is calculated by the decrease in its absorbance at 517 nm prompted by antioxidants [50]. The reduction of DPPH radical intensity in this study is due to the interaction of Ru(III) complexes with radical and as such scavenging the radicals by hydrogen donation (Scheme 2). The DPPH activities by the Ru(III)-N2O Schiff base complexes exhibit strong electron donating power when compared to the standards: ascorbic acid and rutin as displayed in Figure 5. The calculated IC50 and its corresponding R 2 (correlation coefficient) values of Ru(III) compounds are listed in Table 2. Compounds [Ru(DAE)Cl2(H2O)], [Ru(HME)Cl2(H2O)], [Ru(MBE)Cl2(H2O)], and [Ru(DEE)Cl2(H2O)] with an IC50 value of 1.60 ± 0.68, 1.54 ± 0.44, 1.63 ± 1.05, and 1.51 ± 0.50 μM, respectively, exhibited higher activity against DPPH than the commercially available Vit. C and rutin (standard); however, [Ru(DEE)Cl2(H2O)] showed the highest activity of all investigated ruthenium(III) samples with an IC50 value of 1.51 ± 0.50 μM.
Scheme 2.
Conversion of DPPH∗ (purple) to its corresponding hydrazine form (yellow) by the addition of Ru(III) compounds to DPPH∗ due to proton transfer.
Figure 5.
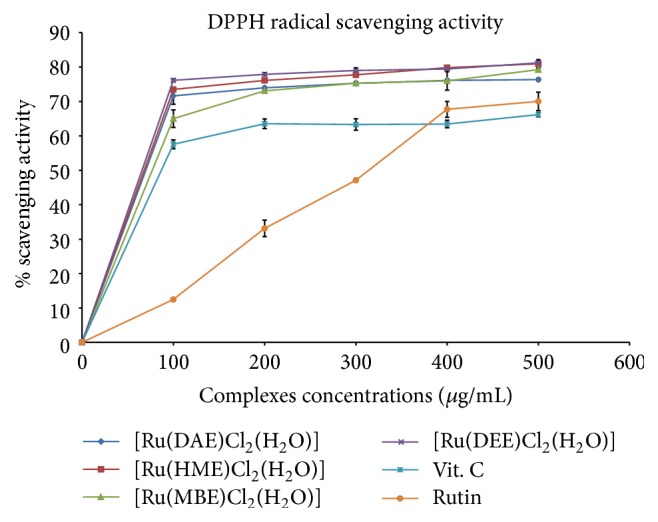
DPPH scavenging potential of Ru(III)-Schiff base complexes.
Table 2.
Radical scavenging abilities (IC50 ± SD, µM) of Ru(III)-Schiff base complexes and standard drugs.
| Compounds | DPPH radical scavenging activity | ABTS radical scavenging activity | ||
|---|---|---|---|---|
| IC50 (µM) | R 2 | IC50 (µM) | R 2 | |
| Ru(DAE)Cl2(H2O) | 1.60 ± 0.68 | 0.965 | 3.30 ± 0.89 | 0.959 |
| Ru(HME)Cl2(H2O) | 1.54 ± 0.44 | 0.974 | 4.27 ± 1.17 | 0.808 |
| Ru(MBE)Cl2(H2O) | 1.63 ± 1.05 | 0.991 | 3.30 ± 1.48 | 0.877 |
| Ru(DEE)Cl2(H2O) | 1.51 ± 0.50 | 0.963 | 3.24 ± 0.93 | 0.855 |
| Rutin∗ | 2.52 ± 1.60 | 0.798 | 2.83 ± 1.84 | 0.983 |
| Vit. C∗ | 1.92 ± 1.07 | 0.978 | — | — |
| BHT∗ | — | — | 1.64 ± 1.54 | 0.919 |
n = 3, X ± SEM; IC50: growth inhibitory concentration; when the inhibition of the tested compounds was 50%, the tested compound concentration was IC50. R 2: correlation coefficient. ∗Standards.
Scavenging ability of the test samples on the DPPH radical can be ranked in the following order: [Ru(DEE)Cl2(H2O)] > [Ru(HME)Cl2(H2O)] > [Ru(DAE)Cl2(H2O)] > [Ru(MBE)Cl2(H2O)] > [Vit. C] > [rutin]. The scavenging effect of the DAE, HME, MBE, and DEE ligands is lower as compared to their corresponding Ru(III) complexes, owing to the coordination of the organic molecules to the Ru3+ ion. It is further supported by the observed discolouration from purple DPPH radical solution to yellow solution showing scavenging of the DPPH radicals by hydrogen donation (Scheme 2). Hence, these complexes could be effective therapeutic agent's preparation for the treatment of chronic conditions such as cardiovascular, neurodegenerative, and arteriosclerosis diseases [21].
3.6.2. 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) Radical Scavenging Activity
To further confirm the synthesized Ru(III)-N2O Schiff base complexes antiradical potential, we examined the ABTS assay in this study. A well-known protonated radical like 2,2′-azinobis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) possesses characteristic absorbance maxima at 734 nm and decreases with the scavenging of the proton radicals [51]. The assay measures radical scavenging by electron donation. The outcome of Ru(III)-N2O Schiff base complexes alongside the standard drugs on ABTS radical is presented in Table 2. At 734 nm, the absorbance of active ABTS∗ solution noticeably declined upon the addition of different concentrations of ruthenium(III) samples; the same trend was also observed for the standard drugs: butylated hydroxytoluene (BHT) and rutin hydrate with the percentage inhibition displayed in Figure 6.
Figure 6.
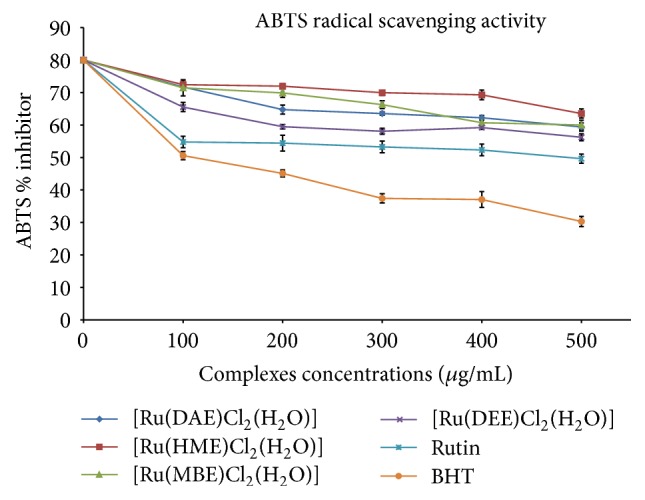
ABTS rummaging activity of Ru(III)-Schiff base complexes.
The efficacy of the tested samples in quenching ATBS∗ radicals in the system was observed at 100 μg/mL, the lowest concentration, and Ru(III) complexes exhibited higher ABTS % inhibition than the standards. [Ru(DEE)Cl2(H2O)] complex exhibited the highest ABTS scavenging activity amongst the studied ruthenium(III) complexes with an IC50 value of 3.24 ± 0.93 μM and 0.855 R 2 (correlation coefficient) as listed in Table 2 while complexes of [Ru(DAE)Cl2(H2O)], [Ru(HME)Cl2(H2O)], and [Ru(MBE)Cl2(H2O)] had an IC50 value of 3.30 ± 0.89, 4.27 ± 1.17, and 3.30 ± 1.48 μM, respectively.
The ABTS scavenging activity pattern of the complexes is ranked in the following order: [Ru(HME)Cl2(H2O)] < [Ru(MBE)Cl2(H2O)] = [Ru(DAE)Cl2(H2O)] < [Ru(DEE)Cl2(H2O)]. With this result, the antiradical studies showed that the synthesised Ru(III)-N2O Schiff base complexes may be useful in developing therapeutic agent for averting cell oxidative damage and as radicals chain terminator. This is because various free radicals generated in the system often lead to cancer, cellular injury, aging process, and cardiovascular diseases [21].
4. Conclusion
In this study, we present the synthesis of Ru(III) Schiff base complexes formulated as [Ru(LL)Cl2(H2O)] (LL = DAE, HME, MBE, and DEE). The complexes were characterized using the microanalytical, conductance, electronic, and vibrational spectral analysis. FTIR spectral data showed that the ligand acts as tridentate chelating ligand, coordinating through azomethine nitrogen and phenol oxygen atom. The microanalyses were in conformity with the proposed structures. Conductance measurements showed the complexes to be nonelectrolytes in DMF. Octahedral structures were assigned to these complexes based on the elemental and spectral information. In vitro antiproliferative studies of the Ru(III) complexes gave a weak to strong inhibition against the studied cancer cell lines, with the following activity order: MCF-7 > UACC-62 > TK-10. Significantly, further investigation on the compounds free radical scavenging properties revealed that Ru(III)-Schiff base complexes possessed considerable antioxidant activities. The outcome from DPPH and ABTS inhibition studies revealed that the compounds are proficient in donating electron or hydrogen atom and subsequently terminate the chain reactions in a dose-dependent pattern. Scavenging ability of the test samples on the DPPH radicals can be ranked in the following order: [Ru(DEE)Cl2(H2O)] > [Ru(HME)Cl2(H2O)] > [Ru(DAE)Cl2(H2O)] > [Ru(MBE)Cl2(H2O)]. Thus, Ru(III)-N2O Schiff base complexes showed stronger inhibition of DPPH at various concentrations.
Acknowledgments
The authors acknowledge Govan Mbeki Research and Development Centre (GMRDC), University of Fort Hare, for financial support and IPE acknowledges National Research Foundation and Sasol Inzalo Foundation for the award of Ph.D. scholarship.
Competing Interests
No conflict of interests regarding the publication of this paper is declared by the authors.
References
- 1.Butler A., Walker J. V. Marine haloperoxidases. Chemical Reviews. 1993;93(5):1937–1944. doi: 10.1021/cr00021a014. [DOI] [Google Scholar]
- 2.Shechter Y., Goldwaser I., Mironchik M., Fridkin M., Gefel D. Historic perspective and recent developments on the insulin-like actions of vanadium; toward developing vanadium-based drugs for diabetes. Coordination Chemistry Reviews. 2003;237(1-2):3–11. doi: 10.1016/S0010-8545(02)00302-8. [DOI] [Google Scholar]
- 3.Bastos A. M. B., da Silva J. G., Maia P. I. S., et al. Oxovanadium(IV) and (V) complexes of acetylpyridine-derived semicarbazones exhibit insulin-like activity. Polyhedron. 2008;27(6):1787–1794. doi: 10.1016/j.poly.2008.02.014. [DOI] [Google Scholar]
- 4.Eady R. R. Current status of structure function relationships of vanadium nitrogenase. Coordination Chemistry Reviews. 2003;237(1-2):23–30. doi: 10.1016/S0010-8545(02)00248-5. [DOI] [Google Scholar]
- 5.Thompson K. H., McNeill J. H., Orvig C. Vanadium compounds as insulin mimics. Chemical Reviews. 1999;99(9):2561–2572. doi: 10.1021/cr980427c. [DOI] [PubMed] [Google Scholar]
- 6.Grivani G., Bruno G., Rudbari H. A., Khalaji A. D., Pourteimouri P. Synthesis, characterization and crystal structure determination of a new oxovanadium(IV) Schiff base complex: the catalytic activity in the epoxidation of cyclooctene. Inorganic Chemistry Communications. 2012;18:15–20. doi: 10.1016/j.inoche.2011.12.044. [DOI] [Google Scholar]
- 7.Pyrz J. W., Roe A. L., Stern L. J., Que L., Jr. Model studies of iron-tyrosinate proteins. Journal of the American Chemical Society. 1985;107(3):614–620. doi: 10.1021/ja00289a013. [DOI] [Google Scholar]
- 8.Tümer M., Erdogan B., Köksal H., Serin S., Nutku M. Y. Preparation, spectroscopic characterisation and thermal analyses studies of the Cu(II), Pd(II) and VO(IV) complexes of some Schiff base ligands. Synthesis and Reactivity in Inorganic and Metal-Organic Chemistry. 1998;28(4):529–542. doi: 10.1080/00945719809351663. [DOI] [Google Scholar]
- 9.Hine J., Yeh C. Y. Equilibrium in formation and conformational isomerization of imines derived from isobutyraldehyde and saturated aliphatic primary amines. Journal of the American Chemical Society. 1967;89(11):2669–2676. doi: 10.1021/ja00987a030. [DOI] [Google Scholar]
- 10.Opstal T., Verpoort F. Synthesis of highly active ruthenium indenylidene complexes for atom-transfer radical polymerization and ring-opening-metathesis polymerization. Angewandte Chemie—International Edition. 2003;42(25):2876–2879. doi: 10.1002/anie.200250840. [DOI] [PubMed] [Google Scholar]
- 11.De Clercq B., Lefebvre F., Verpoort F. Immobilization of multifunctional Schiff base containing ruthenium complexes on MCM-41. Applied Catalysis A: General. 2003;247(2):345–364. doi: 10.1016/S0926-860X(03)00126-1. [DOI] [Google Scholar]
- 12.Gölcü A., Tümer M., Demirelli H., Wheatley R. A. Cd(II) and Cu(II) complexes of polydentate Schiff base ligands: synthesis, characterization, properties and biological activity. Inorganica Chimica Acta. 2005;358(6):1785–1797. doi: 10.1016/j.ica.2004.11.026. [DOI] [Google Scholar]
- 13.Mishra N., Poonia K., Kumar D. An overview of biological aspects of Schiff base metal complexes. International Journal of Advancements in Research & Technology. 2013;2(8):52–66. [Google Scholar]
- 14.Allen L.-A. H., Schlesinger L. S., Kang B. Virulent strains of Helicobacter pylori demonstrate delayed phagocytosis and stimulate homotypic phagosome fusion in macrophages. The Journal of Experimental Medicine. 2000;191(1):115–127. doi: 10.1084/jem.191.1.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Calderon L. A., Teles R. C. L., Leite J. R. S. A., Bloch C., Jr., Astolfi-Filho S., Freitas S. M. Serine protease inhibitors from Amazon Leguminosae seeds: purification and preliminary characterization of two chymotrypsin inhibitors from Inga umbratica . Protein and Peptide Letters. 2001;8(6):485–493. doi: 10.2174/0929866013409175. [DOI] [Google Scholar]
- 16.Cozzi P. G. Metal-Salen Schiff base complexes in catalysis: practical aspects. Chemical Society Reviews. 2004;33(7):410–421. doi: 10.1039/b307853c. [DOI] [PubMed] [Google Scholar]
- 17.Katsuki T. Unique asymmetric catalysis of cis-β metal complexes of salen and its related Schiff-base ligands. Chemical Society Reviews. 2004;33(7):437–444. doi: 10.1039/b304133f. [DOI] [PubMed] [Google Scholar]
- 18.Fakhr I. M. I., Hamdy N. A., Radwan M. A., Ahmed Y. M. Synthesis of new bioactive benzothiophene derivatives. Egyptian Journal of Chemistry. 2004;47:201–215. [Google Scholar]
- 19.Ammar R. A. A., Alaghaz A.-N. M. A. Synthesis, spectroscopic characterization and potentiometric studies of a tetradentate [N2O2] schiff base, N,N′-bis(2-hydroxybenzylidene)-1,1-diaminoethane and its Co(II),Ni(II),Cu(II) and Zn(II) complexes. International Journal of Electrochemical Science. 2013;8(6):8686–8699. [Google Scholar]
- 20.Gaballa A. S., Asker M. S., Barakat A. S., Teleb S. M. Synthesis, characterization and biological activity of some platinum(II) complexes with Schiff bases derived from salicylaldehyde, 2-furaldehyde and phenylenediamine. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 2007;67(1):114–121. doi: 10.1016/j.saa.2006.06.031. [DOI] [PubMed] [Google Scholar]
- 21.Ejidike I. P., Ajibade P. A. Transition metal complexes of symmetrical and asymmetrical Schiff bases as antibacterial, antifungal, antioxidant, and anticancer agents: progress and prospects. Reviews in Inorganic Chemistry. 2015;35(4):191–224. doi: 10.1515/revic-2015-0007. [DOI] [Google Scholar]
- 22.Ejidike I. P., Ajibade P. A. Synthesis, characterization, and in vitro antioxidant and anticancer studies of ruthenium(III) complexes of symmetric and asymmetric tetradentate Schiff bases. Journal of Coordination Chemistry. 2015;68(14):2552–2564. doi: 10.1080/00958972.2015.1043127. [DOI] [Google Scholar]
- 23.Priya N. P., Arunachalam S., Manimaran A., Muthupriya D., Jayabalakrishnan C. Mononuclear Ru(III) Schiff base complexes: synthesis, spectral, redox, catalytic and biological activity studies. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 2009;72(3):670–676. doi: 10.1016/j.saa.2008.10.028. [DOI] [PubMed] [Google Scholar]
- 24.Mishra L., Prajapati R., Pandey K. K. Mixed-ligand Ru(II) complexes with 2,2′-bipyridine and tetradentate Schiff bases auxiliary ligands: Synthesis, physico-chemical study, DFT analysis, electrochemical and Na+ binding properties. Spectrochimica Acta—Part A: Molecular and Biomolecular Spectroscopy. 2008;70(1):79–85. doi: 10.1016/j.saa.2007.07.017. [DOI] [PubMed] [Google Scholar]
- 25.Venkatachalam G., Ramesh R. Catalytic and biological activities of Ru(III) mixed ligand complexes containing N,O donor of 2-hydroxy-1-naphthylideneimines. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 2005;61(9):2081–2087. doi: 10.1016/j.saa.2004.08.008. [DOI] [PubMed] [Google Scholar]
- 26.Ejidike I. P., Ajibade P. A. Synthesis and in vitro anticancer, antibacterial, and antioxidant studies of unsymmetrical Schiff base derivatives of 4-[(1E)-N-(2-aminoethyl)ethanimidoyl]benzene-1,3-diol. Research on Chemical Intermediates. 2016;42(8):6543–6555. doi: 10.1007/s11164-016-2479-x. [DOI] [Google Scholar]
- 27.Ansari K. I., Hussain I., Das H. K., Mandal S. S. Overexpression of human histone methylase MLL1 upon exposure to a food contaminant mycotoxin, deoxynivalenol. The FEBS Journal. 2009;276(12):3299–3307. doi: 10.1111/j.1742-4658.2009.07055.x. [DOI] [PubMed] [Google Scholar]
- 28.Ejidike I. P., Ajibade P. A. Synthesis, characterization and biological studies of metal(II) complexes of (3E)-3-[(2-{(E)-[1-(2,4-Dihydroxyphenyl) ethylidene]amino}ethyl)imino]-1-phenylbutan-1-one schiff base. Molecules. 2015;20(6):9788–9802. doi: 10.3390/molecules20069788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bolos C. A., Chaviara A. T., Mourelatos D., et al. Synthesis, characterization, toxicity, cytogenetic and in vivo antitumor studies of 1,1-dithiolate Cu(II) complexes with di-, tri-, tetra- amines and 1,3-thiazoles. Structure-activity correlation. Bioorganic & Medicinal Chemistry. 2009;17(8):3142–3151. doi: 10.1016/j.bmc.2009.02.059. [DOI] [PubMed] [Google Scholar]
- 30.Das P. K., Panda N., Behera N. K. Synthesis, characterization and antimicrobial activities of Schiff base complexes derived from isoniazid and diacetylmonoxime. International Journal of Innovative Science, Engineering & Technology. 2016;3(1):42–54. [Google Scholar]
- 31.Mitu L., Ilis M., Raman N., Imran M., Ravichandran S. Transition metal complexes of isonicotinoyl–hydrazone-4-diphenylaminobenzaldehyde: synthesis, characterization and antimicrobial studies. E-Journal of Chemistry. 2012;9(1):365–372. doi: 10.1155/2012/298175. [DOI] [Google Scholar]
- 32.Ali S. A., Soliman A. A., Aboaly M. M., Ramadan R. M. Chromium, molybdenum and ruthenium complexes of 2-hydroxyacetophenone schiff bases. Journal of Coordination Chemistry. 2002;55(10):1161–1170. doi: 10.1080/0095897021000023509. [DOI] [Google Scholar]
- 33.Kumar K. N., Ramesh R., Liu Y. Synthesis, structure and catalytic activity of cycloruthenated carbonyl complexes containing arylazo phenolate ligands. Journal of Molecular Catalysis A: Chemical. 2007;265(1-2):218–226. doi: 10.1016/j.molcata.2006.10.015. [DOI] [Google Scholar]
- 34.Ejidike I. P., Ajibade P. A. Synthesis, characterization, antioxidant, and antibacterial studies of some metal(II) complexes of tetradentate schiff base ligand: (4E)-4-[(2-{(E)-[1-(2,4-dihydroxyphenyl)ethylidene]amino}ethyl)imino]pentan-2-one. Bioinorganic Chemistry and Applications. 2015;2015:9. doi: 10.1155/2015/890734.890734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Casas J. S., Castiñeiras A., Condori F., et al. Diorganotin(IV)-promoted deamination of amino acids by pyridoxal: SnR2 2+ complexes of pyridoxal 5′-phosphate and of the Schiff base pyridoxal-pyridoxamine (PLPM), and antibacterial activities of PLPM and [SnR2(PLPM-2H)] (R=Me, Et, Bu, Ph) Polyhedron. 2003;22(1):53–65. doi: 10.1016/s0277-5387(02)01331-1. [DOI] [Google Scholar]
- 36.Inba P. J. K., Annaraj B., Thalamuthu S., Neelakantan M. A. Cu(II), Ni(II), and Zn(II) complexes of salan-type ligand containing ester groups: synthesis, characterization, electrochemical properties, and in vitro biological activities. Bioinorganic Chemistry and Applications. 2013;2013:11. doi: 10.1155/2013/439848.439848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shanker K., Rohini R., Ravinder V., Reddy P. M., Ho Y.-P. Ru(II) complexes of N4 and N2O2 macrocyclic Schiff base ligands: their antibacterial and antifungal studies. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 2009;73(1):205–211. doi: 10.1016/j.saa.2009.01.021. [DOI] [PubMed] [Google Scholar]
- 38.Alias M., Kassum H., Shakir C. Synthesis, physical characterization and biological evaluation of Schiff base M(II) complexes. Journal of the Association of Arab Universities for Basic and Applied Sciences. 2014;15(1):28–34. doi: 10.1016/j.jaubas.2013.03.001. [DOI] [Google Scholar]
- 39.Shivakumar K., Shashidhar, Reddy P. V., Halli M. B. Synthesis, spectral characterization and biological activity of benzofuran Schiff bases with Co(II), Ni(II), Cu(II), Zn(II), Cd(II) and Hg(II) complexes. Journal of Coordination Chemistry. 2008;61(14):2274–2287. doi: 10.1080/00958970801905239. [DOI] [Google Scholar]
- 40.Thangadurai T. D., Ihm S.-K. Novel bidentate ruthenium(III) Schiff base complexes: synthetic, spectral, electrochemical, catalytic and antimicrobial studies. Transition Metal Chemistry. 2004;29(2):189–195. doi: 10.1023/b:tmch.0000019419.40754.63. [DOI] [Google Scholar]
- 41.Ballhausen C. J. Introduction to Ligand Field Theory. New York, NY, USA: McGarw Hill; 1962. [Google Scholar]
- 42.Lever A. B. P. Inorganic Electronic Spectroscopy. 2nd. New York, NY, USA: Elsevier; 1984. [Google Scholar]
- 43.Raju V. V., Balasubramanian K. P., Jayabalakrishnan C., Chinnusamy V. Synthesis, characterization, antimicrobial activities and DNA-Binding studies of some Ru(III) complexes of Schiff bases. International Journal of Applied Biology and Pharmaceutical Technology. 2012;3(2):76–87. [Google Scholar]
- 44.Balasubramanian K. P., Parameswari K., Chinnusamy V., Prabhakaran R., Natarajan K. Synthesis, characterization, electro chemistry, catalytic and biological activities of ruthenium(III) complexes with bidentate N, O/S donor ligands. Spectrochimica Acta—Part A: Molecular and Biomolecular Spectroscopy. 2006;65(3-4):678–683. doi: 10.1016/j.saa.2005.12.029. [DOI] [PubMed] [Google Scholar]
- 45.Ghantous A., Saikali M., Rau T., Gali-Muhtasib H., Schneider-Stock R., Darwiche N. Inhibition of tumor promotion by parthenolide: epigenetic modulation of p21. Cancer Prevention Research. 2012;5(11):1298–1309. doi: 10.1158/1940-6207.capr-12-0230. [DOI] [PubMed] [Google Scholar]
- 46.Shier W. T. Mammalian Cell Culture on $5 a Day: A Lab Manual of Low Cost Methods. Los Banos, Calif, USA: University of the Philippines; 1991. [Google Scholar]
- 47.Raja G., Butcher R. J., Jayabalakrishnan C. Studies on synthesis, characterization, DNA interaction and cytotoxicity of ruthenium(II) Schiff base complexes. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 2012;94:210–215. doi: 10.1016/j.saa.2012.03.035. [DOI] [PubMed] [Google Scholar]
- 48.Raja G., Butcher R. J., Jayabalakrishnan C. Synthesis, characterization, DNA binding and cleavage properties and anticancer studies of ruthenium(III) Schiff base complexes. Transition Metal Chemistry. 2012;37(2):169–174. doi: 10.1007/s11243-011-9571-2. [DOI] [Google Scholar]
- 49.Gülçin I., Küfrevioglu Ö. I., Oktay M., Büyükokuroglu M. E. Antioxidant, antimicrobial, antiulcer and analgesic activities of nettle (Urtica dioica L.) Journal of Ethnopharmacology. 2004;90(2-3):205–215. doi: 10.1016/j.jep.2003.09.028. [DOI] [PubMed] [Google Scholar]
- 50.Viuda-Martos M., Navajas Y. R., Zapata E. S., Fernández-López J., Pérez-Álvarez J. A. Antioxidant activity of essential oils of five spice plants widely used in a Mediterranean diet. Flavour and Fragrance Journal. 2010;25(1):13–19. doi: 10.1002/ffj.1951. [DOI] [Google Scholar]
- 51.Mathew S., Abraham T. E. In vitro antioxidant activity and scavenging effects of Cinnamomum verum leaf extract assayed by different methodologies. Food and Chemical Toxicology. 2006;44(2):198–206. doi: 10.1016/j.fct.2005.06.013. [DOI] [PubMed] [Google Scholar]