Abstract
Background:
We aimed to understand the dependence of MEK and m-TOR inhibition in EGFRWT/ALKnon-rearranged NSCLC cell lines.
Methods:
In a panel of KRASM and KRASWT NSCLC cell lines, we determined growth inhibition (GI) following maximal reduction in p-ERK and p-S6RP caused by trametinib (MEK inhibitor) and AZD2014 (m-TOR inhibitor), respectively.
Results:
GI caused by maximal m-TOR inhibition was significantly greater than GI caused by maximal MEK inhibition in the cell line panel (52% vs 18%, P<10−4). There was no significant difference in GI caused by maximal m-TOR compared with maximal m-TOR+MEK inhibition. However, GI caused by the combination was significantly greater in the KRASM cell lines (79% vs 61%, P=0.017).
Conclusions:
m-TOR inhibition was more critical to GI than MEK inhibition in EGFRWT/ALKnon-rearranged NSCLC cells. The combination of MEK and m-TOR inhibition was most effective in KRASM cells.
Keywords: drug combinations, NSCLC, KRAS mutant, MEK inhibitor, trametinib, m-TOR inhibitor, AZD2014
Lung cancer is the leading cause of death in the world. The advent of personalised medicine has seen the introduction of a number of targeted treatments for the adenocarcinoma subset of NSCLC (NSCLC-adeno). Although EGFR (Maemondo et al, 2010) and ALK (Solomon et al, 2014) inhibitors are established in EGFR mutant and ALK rearranged lung cancer, there are currently no molecularly targeted agents licensed for use in the remainder of adenocarcinomas (NSCLC-adeno-EGFRWT/ALKnon-rearranged). There have been recent advances in the treatment of NSCLC-adeno with the introduction of immune checkpoint inhibitors. However, response rates and median overall survival are low with 19% and 12.2 months, respectively (Borghaei et al, 2015). Thus, finding new treatments for NSCLC-adeno is an area of unmet need.
We have focused our efforts on the subgroup of NSCLC-adeno-EGFRWT/ALKnon-rearranged. There are multiple activating mutations in this subset (KRAS, BRAF, MET, HER-2 and STK11), and gene fusions of which KRAS mutations form the largest group (Cancer Genome Atlas Research Network, 2014). We tested inhibitors of MEK and m-TOR that target nodes that are downstream of many of these activating events. We studied on a cell line panel the dependence on MEK or m-TOR signalling. We used trametinib, a FDA- and EMA-approved drug (Infante et al, 2012), and AZD2014, a drug under clinical investigation (Basu et al, 2015), MEK and m-TOR inhibitors, which are known to cause robust pharmacodynamics suppression of signals.
The specific aims of the study were to investigate growth inhibition (GI) following maximal inhibition of MEK or m-TOR in NSCLC-adeno-EGFRWT/ALKnon-rearranged cells and to identify any differences between KRASM and KRASWT cell lines. We then studied the effect of additive GI by inhibiting signalling through both MEK and m-TOR nodes compared with GI caused by inhibiting MEK or m-TOR node.
Materials and methods
Cell lines and drugs
A panel of six NSCLC cell lines, including three KRASM (A549, Calu6 and H23) and three KRASWT (H522, H1838 and H1651) cell lines, were purchased from ATCC-LGC Standards (Teddington, UK). Trametinib and AZD2014 were sourced from Selleckchem (Munich, Germany).
Quantification of inhibition of signalling
Maximal reduction of p-ERK1/2 (Thr202/Tyr204; Thr185/Tyr187) and p-S6RP (Ser235/236) was determined by exposing the cell lines to increasing concentrations of Trametinib and AZD2014 over 24 h and quantifying p-ERK1/2 and p-S6RP by ELISA (MesoScale Discovery, Rockville, MD, USA; kit K151DWD; K150DFD) following manufacturer's instructions. Experiments were conducted in triplicates.
Growth inhibition
Cell lines were exposed to concentrations of Trametinib and AZD2014 shown to maximally inhibit MEK and m-TOR signalling, respectively, for 72 h. The effects of inhibiting these nodes alone or in combination on cell growth were studied using a WST-1 assay (Roche Diagnostics, Burgess Hill, UK). Experiments were carried out in triplicates.
Statistics
Data are reported as means±s.d. Statistical significance was evaluated by non-parametric Mann–Whitney's tests. Results were subjected to statistical analysis by using the GraphPad Prism software package, v6.01 (GraphPad Software, La Jolla, CA, USA).
Results
Determination of Trametinib and AZD2014 concentrations inducing maximal inhibition of signal transduction
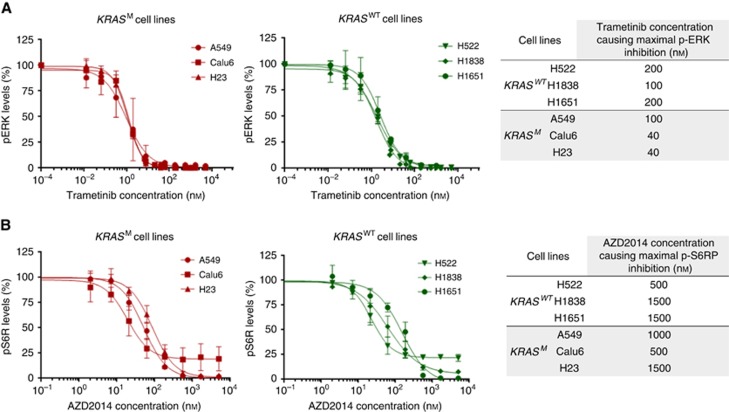
NSCLC-adeno-EGFRWT/ALKnon-rearranged cell lines were exposed to increasing concentrations of Trametinib and AZD2014 over 24 h and the concentrations of Trametinib and AZD2014 to cause maximal reduction in levels of p-ERK and p-S6RP were determined (Figure 1).
Figure 1.
Determination of degrees of signalling inhibition caused by MEK and m-TOR inhibitors in KRASM and KRASWT cell lines. (A) Trametinib concentrations needed to maximally reduce p-ERK levels (table). (B) AZD2014 concentrations needed to maximally reduce p-S6RP levels (table). The error bars represent s.d.
Determination of dependence of MEK and m-TOR signalling in KRASM and KRASWT cell lines
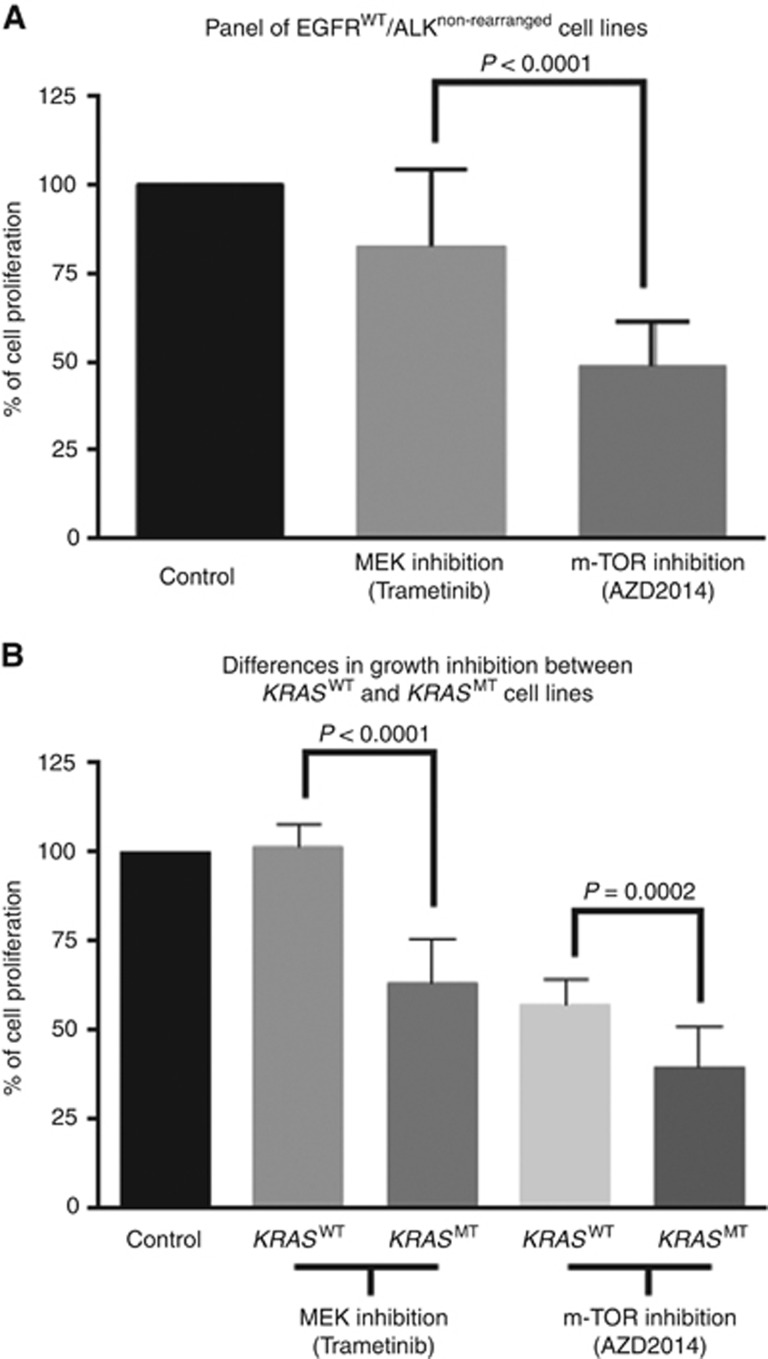
Using the concentrations previously determined for each cell line, the mean GI caused by maximal MEK and m-TOR inhibition was 18%(SD22) vs 52%(SD13), P<0.0001, respectively, in all cell lines (Figure 2A). These data suggest that in this panel, the cells were more dependent on growth on m-TOR signalling compared with MEK. This result is confirmed in KRASM and KRASWT cell lines with GI of 0%(SD6) vs 36%(SD12), P<0.0001 and 43%(SD7) vs 61%(SD11), P=0.0002, respectively (Figure 2B).
Figure 2.
Growth inhibition upon maximal inhibition of MEK and m-TOR signalling.(A) Growth inhibition in the cell lines panel. (B) Growth inhibition in KRASM and KRASWT cell lines. The error bars represent s.d.
Combination of MEK and m-TOR signalling
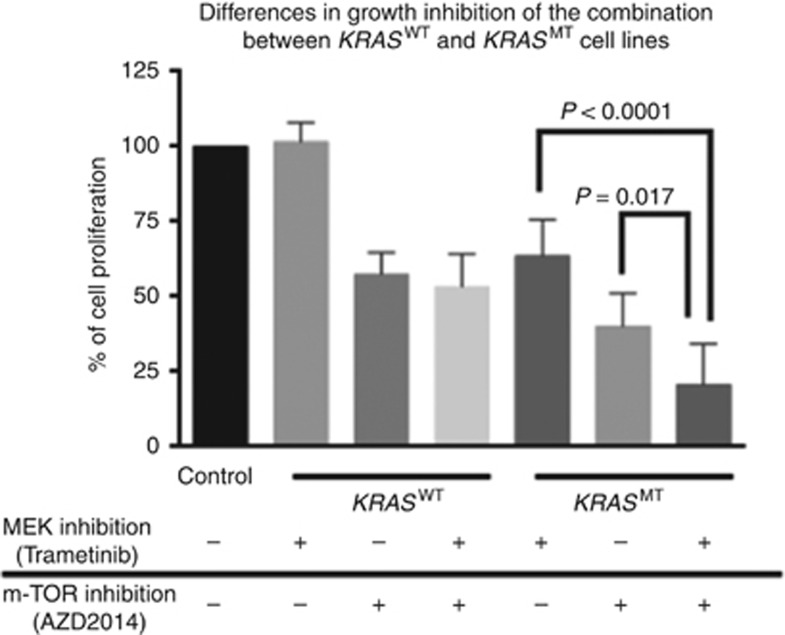
As the GI caused by MEK and m-TOR inhibitions were modest, combinations of these were studied, using the same concentrations. Across the cell line panel, the difference between mean GI caused by maximal m-TOR inhibition (52%, SD13) when compared with GI caused by maximal m-TOR+MEK inhibition (64%, SD21) was not significant (P=0.089). However, there was a significant difference of GI caused by inhibition of MEK signalling alone, compared with the combination of maximal MEK+m-TOR signalling inhibition, 18%(SD22) vs 64%(SD21), P<0.0001 (data not shown). Importantly, in KRASM cell lines, inhibition of m-TOR+MEK signalling caused significantly greater GI than inhibition of m-TOR or MEK signalling alone, 79%(SD14) vs 61%(SD11), P=0.017 and 79%(SD14) vs 36(SD12), P<0.0001 (Figure 3).
Figure 3.
Growth inhibition caused by MEK and m-TOR inhibitors alone and in combination in KRASM and KRASWT cell lines.The error bars represent s.d.
Discussion
We have used an approach of titrating inhibition of signalling output to decide upon concentrations of drugs to be used in GI experiments as we believe that this approach better reflects the dependence of cancer cell growth to different signalling networks and reduces the reporting predominantly off-target effects of drugs (Stewart et al, 2015).
In the NSCLC cell lines studied, we have shown for the first time that maximal inhibition of m-TOR caused significantly more GI compared with maximal MEK inhibition. This is interesting as most efforts of clinically developing signal transduction inhibitors in NSCLC-adeno-EGFRWT/ALKnon-rearranged cancers is focussed on using MEK inhibitors. In clinical practice MEK inhibitors response, when used alone, is confined to KRASM tumours, where response rates typically range between 0 and 15% (Zimmer et al, 2014; Blumenschein et al, 2015). Our data showed that upon maximal MEK inhibition, KRASM cell lines showed significantly greater GI compared with KRASWT cell lines. However, the GI caused by MEK inhibition in KRASM cell lines was modest (36%). This observation is in line with previously published literature (Pratilas et al, 2008). In the panel studied, our data suggest that inhibition of MEK signalling has very little effect on growth in KRASWT cell lines within the group of EGFRWT/ALKnon-rearranged cell lines.
There have been few single agent studies of m-TOR inhibitors in lung cancer and with very low response rates of 0–5% (Soria et al, 2009; Reungwetwattana et al, 2012). This discrepancy between the clinical outcomes following m-TOR inhibitor treatment and our data showing that NSCLC cells are more sensitive to m-TOR inhibitors compared with MEK inhibitors, may be explained by signalling though feedback loops and parallel signal transduction pathways (Al-Lazikani et al, 2012). Main m-TOR inhibitors evaluated in clinic so far are mTORC1 inhibitors, known to induce feedback loops leading to mTORC2–IRS-1-mediated hyperactivation of PI3K–AKT (Jokinen and Koivunen, 2015), while AZD2014 is a potent inhibitor of mTORC1 and mTORC2 preventing this feedback.
Preclinical studies have suggested combinations of m-TOR and MEK inhibitors in KRASM lung cancers (Engelman et al, 2008) but also in N- and HRAS mutant tumours (Kiessling et al, 2015). However, these attempted combinations have proven challenging to deliver due to diarrhoea, skin rash and fatigue (Tolcher et al, 2015a). Intermittent dosing could be used to reduce the degree of toxicity (Yap et al, 2013). We have shown for the first time that inhibition of m-TOR contributes to the majority of the GI in this combination. As our results suggest, in the event of toxicity, clinical trial designs using intermittent dosing should prioritise reducing the dose/frequency of the MEK signalling rather than m-TOR signalling. MEK inhibitors have been used in combination with PI3K and AKT inhibitors which are proximal nodes in the canonical PI3K–AKT–m-TOR pathway and such treatment regimens have shown early promise in the treatment of KRASM NSCLC (Shimizu et al, 2012; Tolcher et al, 2015b). The policy of prioritising inhibition of m-TOR over MEK signalling within the combination may be broadly applicable in across MEK combinations with AKT and PI3K inhibitors but this needs to be validated in those settings.
The combinations of MEK inhibitors and m-TOR inhibitors or more broadly PI3K pathway inhibitors should be evaluated in KRASM NSCLC. Toxicity limits such combinations and intermittent schedules are the only way to deliver drugs at doses that are pharmacodynamically active. In case of toxicity, our data suggest dose interruptions/reductions should be considered in inhibitors of MEK signalling rather than inhibitors of m-TOR signalling. This approach will help refine doses/schedules of MEK and PI3K pathway inhibitors to provide new treatment paradigms for KRASM NSCLC.
Acknowledgments
This work was supported by Cancer Research UK (grant number C309/A11566), the Experimental Cancer Medicine Centre award (grant number C12540/A15573), and the National Institute for Health Research Biomedical Research Centre award. SB has received the support of Fondation de France – Unicancer (Aide à la mobilité internationale de médecins ou pharmaciens). Dr UB has received research funding from AstraZeneca.
The authors declare no conflict of interest.
References
- Al-Lazikani B, Banerji U, Workman P (2012) Combinatorial drug therapy for cancer in the post-genomic era. Nat Biotechnol 30(7): 679–692. [DOI] [PubMed] [Google Scholar]
- Basu B, Dean E, Puglisi M, Greystoke A, Ong M, Burke W, Cavallin M, Bigley G, Womack C, Harrington EA, Green S, Oelmann E, de Bono JS, Ranson M, Banerji U (2015) First-in-Human Pharmacokinetic and Pharmacodynamic Study of the dual m-TORC 1/2 inhibitor AZD2014. Clin Cancer Res 21(15): 3412–3419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blumenschein GR Jr, Smit EF, Planchard D, Kim DW, Cadranel J, De Pas T, Dunphy F, Udud K, Ahn MJ, Hanna NH, Kim JH, Mazieres J, Kim SW, Baas P, Rappold E, Redhu S, Puski A, Wu FS, Jänne PA (2015) A randomized phase II study of the MEK1/MEK2 inhibitor trametinib (GSK1120212) compared with docetaxel in KRAS-mutant advanced non-small-cell lung cancer (NSCLC)dagger. Ann Oncol 26(5): 894–901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E, Barlesi F, Kohlhäufl M, Arrieta O, Burgio MA, Fayette J, Lena H, Poddubskaya E, Gerber DE, Gettinger SN, Rudin CM, Rizvi N, Crinò L, Blumenschein GR Jr, Antonia SJ, Dorange C, Harbison CT, Graf Finckenstein F, Brahmer JR (2015) Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 373(17): 1627–1639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cancer Genome Atlas Research Network (2014) Comprehensive molecular profiling of lung adenocarcinoma. Nature 511(7511): 543–550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelman JA, Chen L, Tan X, Crosby K, Guimaraes AR, Upadhyay R, Maira M, McNamara K, Perera SA, Song Y, Chirieac LR, Kaur R, Lightbown A, Simendinger J, Li T, Padera RF, García-Echeverría C, Weissleder R, Mahmood U, Cantley LC, Wong KK (2008) Effective use of PI3K and MEK inhibitors to treat mutant Kras G12D and PIK3CA H1047R murine lung cancers. Nat Med 14(12): 1351–1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Infante JR, Fecher LA, Falchook GS, Nallapareddy S, Gordon MS, Becerra C, DeMarini DJ, Cox DS, Xu Y, Morris SR, Peddareddigari VG, Le NT, Hart L, Bendell JC, Eckhardt G, Kurzrock R, Flaherty K, Burris HA 3rd, Messersmith WA (2012) Safety, pharmacokinetic, pharmacodynamic, and efficacy data for the oral MEK inhibitor trametinib: a phase 1 dose-escalation trial. Lancet Oncol 13(8): 773–781. [DOI] [PubMed] [Google Scholar]
- Jokinen E, Koivunen JP (2015) MEK and PI3K inhibition in solid tumors: rationale and evidence to date. Ther Adv Med Oncol 7(3): 170–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiessling MK, Curioni-Fontecedro A, Samaras P, Atrott K, Cosin-Roger J, Lang S, Scharl M, Rogler G (2015) Mutant HRAS as novel target for MEK and mTOR inhibitors. Oncotarget 6(39): 42183–42196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I, Fujita Y, Okinaga S, Hirano H, Yoshimori K, Harada T, Ogura T, Ando M, Miyazawa H, Tanaka T, Saijo Y, Hagiwara K, Morita S, Nukiwa T North-East Japan Study Group (2010) Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med 362(25): 2380–2388. [DOI] [PubMed] [Google Scholar]
- Pratilas CA, Hanrahan AJ, Halilovic E, Persaud Y, Soh J, Chitale D, Shigematsu H, Yamamoto H, Sawai A, Janakiraman M, Taylor BS, Pao W, Toyooka S, Ladanyi M, Gazdar A, Rosen N, Solit DB (2008) Genetic predictors of MEK dependence in non-small cell lung cancer. Cancer Res 68(22): 9375–9383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reungwetwattana T, Molina JR, Mandrekar SJ, Allen-Ziegler K, Rowland KM, Reuter NF, Luyun RF, Dy GK, Marks RS, Schild SE, Jett JR, Adjei AA (2012) Brief report: a phase II "window-of-opportunity" frontline study of the MTOR inhibitor, temsirolimus given as a single agent in patients with advanced NSCLC, an NCCTG study. J Thorac Oncol 7(5): 919–922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu T, Tolcher AW, Papadopoulos KP, Beeram M, Rasco DW, Smith LS, Gunn S, Smetzer L, Mays TA, Kaiser B, Wick MJ, Alvarez C, Cavazos A, Mangold GL, Patnaik A (2012) The clinical effect of the dual-targeting strategy involving PI3K/AKT/mTOR and RAS/MEK/ERK pathways in patients with advanced cancer. Clin Cancer Res 18(8): 2316–2325. [DOI] [PubMed] [Google Scholar]
- Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, Felip E, Cappuzzo F, Paolini J, Usari T, Iyer S, Reisman A, Wilner KD, Tursi J, Blackhall F PROFILE 1014 Investigators (2014) First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med 371(23): 2167–2177. [DOI] [PubMed] [Google Scholar]
- Soria JC, Shepherd FA, Douillard JY, Wolf J, Giaccone G, Crino L, Cappuzzo F, Sharma S, Gross SH, Dimitrijevic S, Di Scala L, Gardner H, Nogova L, Papadimitrakopoulou V (2009) Efficacy of everolimus (RAD001) in patients with advanced NSCLC previously treated with chemotherapy alone or with chemotherapy and EGFR inhibitors. Ann Oncol 20(10): 1674–1681. [DOI] [PubMed] [Google Scholar]
- Stewart A, Thavasu P, de Bono JS, Banerji U (2015) Titration of signalling output: insights into clinical combinations of MEK and AKT inhibitors. Ann Oncol 26(7): 1504–1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tolcher AW, Bendell JC, Papadopoulos KP, Burris HA 3rd, Patnaik A, Jones SF, Rasco D, Cox DS, Durante M, Bellew KM, Park J, Le NT, Infante JR (2015. a) A phase IB trial of the oral MEK inhibitor trametinib (GSK1120212) in combination with everolimus in patients with advanced solid tumors. Ann Oncol 26(1): 58–64. [DOI] [PubMed] [Google Scholar]
- Tolcher AW, Khan K, Ong M, Banerji U, Papadimitrakopoulou V, Gandara DR, Patnaik A, Baird RD, Olmos D, Garrett CR, Skolnik JM, Rubin EH, Smith PD, Huang P, Learoyd M, Shannon KA, Morosky A, Tetteh E, Jou YM, Papadopoulos KP, Moreno V, Kaiser B, Yap TA, Yan L, de Bono JS (2015. b) Antitumor activity in RAS-driven tumors by blocking AKT and MEK. Clin Cancer Res 21(4): 739–748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yap TA, Omlin A, de Bono JS (2013) Development of therapeutic combinations targeting major cancer signaling pathways. J Clin Oncol 31(12): 1592–1605. [DOI] [PubMed] [Google Scholar]
- Zimmer L, Barlesi F, Martinez-Garcia M, Dieras V, Schellens JH, Spano JP, Middleton MR, Calvo E, Paz-Ares L, Larkin J, Pacey S, Venturi M, Kraeber-Bodéré F, Tessier JJ, Eberhardt WE, Paques M, Guarin E, Meresse V, Soria JC (2014) Phase I expansion and pharmacodynamic study of the oral MEK inhibitor RO4987655 (CH4987655) in selected patients with advanced cancer with RAS-RAF mutations. Clin Cancer Res 20(16): 4251–4261. [DOI] [PubMed] [Google Scholar]