Abstract
A series of new 5-imino-4-thioxo-2-imidazolidinone derivatives 3 with various halogenated and alkylated aromatic substituents at N1 and N3 was synthesized. Imidazolidineiminothione derivatives 3 were prepared from the reaction of N-arylcyanothioformamide derivatives with aryl isocyanates. These compounds were used as key synthons for the preparation of wide variety of new substituted imidazole compounds. Imine hydrolysis of 3 with ethanolic HCl produced the corresponding 4-thioxo-2,5-imidazolidindiones 4. Condensation of 3 with benzophenonhydrazone furnished the corresponding 4-azine derivatives 5. Monohydrazono and dihydrazono derivatives 6 and 8 were obtained upon treatment of imidazolidinone derivatives 3 with hydrazine hydrate. Finally, imidazolidinones 3 were reacted with o-phenylenediamines or pyrazol-5(4H)-ones and afforded the corresponding imidazoquinoxaline and imidazolidin-4-ylidenepyrazolone-5(4H)-one derivatives 11 and 12, respectively. Evaluation of the antibacterial and antifungal activities for the synthesized compounds was carried out to probe their activities. Most of the tested compounds showed significant activities. The best antimicrobial activity was observed for 1-(3-ethoxyphenyl)-6-methyl-1-phenyl-1H-imidazo[4,5-b]quinoxalin-2(3H)-ones (11c) followed by 5-imino-3-(3-methoxy- phenyl)-1-phenyl-4-thioxoimidazolidin-2-one (3f).
Keywords: N-arylcyanothioformanilides, imidazolidineiminothiones, imidazolidinone, imidazoquinoxaline, antibacterial and antifungal activities
Introduction
Infectious diseases caused by bacteria and fungi remain a major worldwide health problem due to rapid development of resistance to the existing antimicrobial drugs. The increasing use and misuse of the existing antimicrobial drugs have resulted in the development of resistant pathogens. The medical community faces a serious problem when treating infections caused by pathogenic microbes and needs an effective therapy and search for novel antimicrobial agents [1-6]. In addition, systemic and dermal fungal infections have significantly increased, specifically in individuals with suppressed immune systems such as those receiving cancer chemotherapy and AIDS patients. Although there are different antifungal drugs used in the treatment of fungal infections, some of them have undesirable side effects because of the biochemical similarity between human cell and fungi forms. The search for new and effective antimicrobial agents, resistant to the mechanisms of defense of these bacteria, is of paramount importance [1, 2]. Imidazoles and their fused derivatives are keys in many bioactive compounds of both natural and synthetic origins [7] such as histidine, purines, biotin and hydantoin. Nitroimidazoles are a well-known family of antibacterial and antiprotozoal drugs [8, 9], including antitrypanosomal drugs or compounds with known anti-trypanosomal activity [10, 11]. Metronidazole, the first drug to be introduced for this purpose and probably also the best known drug in this class, has been in use for more than 50 years [12]. 4-Oxoimidazolidine-2-thiones and their 2-
alkylthio-3,5-dihydro-4H-imidazol-4-one derivatives are important compounds in the fields of drugs, pharmaceutical intermediates. As an example isatinylidene derivatives exhibit immunosuppressive activity [13]. 2-Thiohydantoin derivatives have not only been used in medicinal chemistry, but have also been developed as fungicides e.g. fenamidone [14, 15].
In light of these facts, and in continuation of our ongoing study on antimicrobial agents [16-19], the present study was designed to synthesize new imidazolinone derivatives and evaluate their antimicrobial activity. Various functional groups were introduced into the target compounds in order to investigate their preliminary structure activity relationships.
Results and Discussion
Chemistry
The starting compounds, N-(4-fluoro, 3-chloro, 4-methyl)-cyanothioformanilides 2a-c were prepared according to the previously reported procedure [20]. The new compounds, N-(3-methoxy, 3-ethoxy)-cyanothioformanilides 2d,e were prepared from the reaction of N-aryl isothiocyanates with potassium cyanide. The elemental analysis and spectroscopic data of 2d,e are consistent with the assigned structure. IR measurement of 2e for example displayed absorption bands at: 3264 and 2227 cm-1 for NH and C≡N groups. 1H NMR spectrum revealed triplet and quartet signals at: δ = 1.23 and 4.14 ppm corresponding to CH3 and CH2, respectively and the presence of a D2O exchangeable broad singlet at: δ = 13.36 ppm attributable to the NH proton. In addition, 13C NMR spectrum of 2e showed two signals at 14.1 (CH3), 63.3 (CH2), also it showed a farthermost downfield signal for the thione group (C=S) at: 161.1 due to the tautomeric thione and thiole mixture and an upfield signal resulting from the nitrile group (107.9). The behavior of the cyanothioformanilides towards isocyanate derivatives was investigated. Thus, reaction of cyanothioformanilides with aryl isocyanates in ether and in the presence of triethylamine caused cyclization to furnish the corresponding 5-imino-4-thioxo-2-imidazoli-dinones 3a-k. The structure of 3 was assigned on the basis of analytical and spectral data. IR spectrum of 3f, as representative example, displayed absorption bands at: 3261 cm-1 due to NH and at 1777 cm-1 due to C=O functional group. 1H NMR spectrum exhibited two singlets at: δ = 3.80 and 9.66 ppm indicative of methoxy protons and NH proton, respectively. 13C NMR spectrum of 3f displayed four signals at: 183.3, 160.1, 154.5 and 65.3 for C=S, C=O, C=N and OCH3, respectively, whereas the aromatic signals ranged from 114.1 to 154.1 corresponding for 12C. Mass spectrum showed a molecular ion peak at m/z = 311, corresponding to a molecular formula C16H13N3O2S. Hydrolysis of 3h,i with dilute HCl in boiling ethanol afforded the corresponding dione derivatives 4a,b (Scheme 1). IR spectrum has no absorption band characteristic to an NH group.
Scheme 1.
Synthesis of N-arylcyanothioformanilide, imidazolidin-2-one and imidazolidineiminothione derivatives.
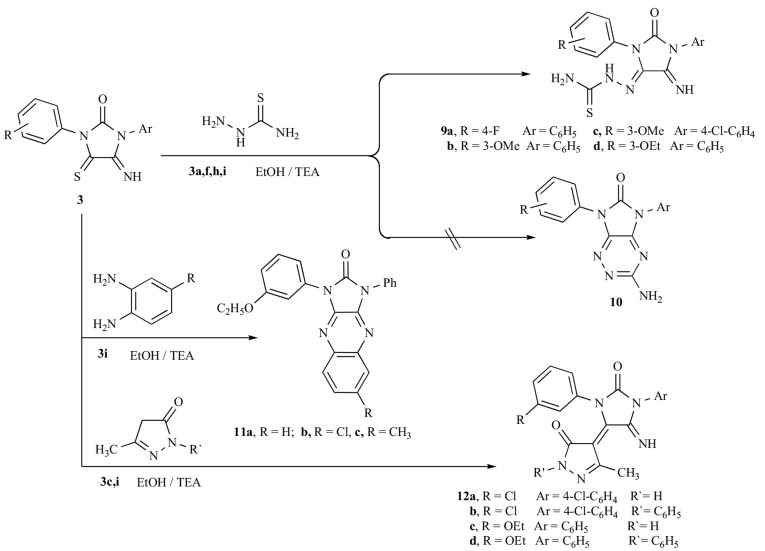
The imidazolidine derivatives contain adjacent imino and thione functional groups in the 5-and 4-positions appear promising for further chemical transformations. Therefore, it was interesting to study the reaction of imidazolidineiminothiones 3 with some amino compounds as nitrogen nucleophiles. Condensation of 3a,d,i with benzophenonehydrazone in boiling ethanol using triethylamine as a basic catalyst furnished the corresponding 4-azine derivatives 5a-c. The analytical and spectral data are in agreement with the proposed structure. Thus IR spectrum of 5a showed the presence of three signals at 3265, 1758 and 1624 cm-1 due to the presence of three groups NH, C=O and C=N, respectively. 1H NMR spectrum showed singlet signal at: 10.11 ppm corresponding NH proton. While, 13C NMR showed the complete disappearance of the signal in the range around 180 ppm due to C=S and the presence of four signals at 160.4, 160.7, 162.4 and 167.5 corresponding 3C=N and C=O groups. In addition, the reactivity of iminothione derivatives 3 towards binucliophiles was also investigated. Thus equimolecular amounts of 3h,i and hydrazine hydrate furnished the monohydrazono derivatives for which structure 6 or 7 seemed possible. The positive element test for sulfur and spectral data favored the 5-hydrazono derivatives 6a,b. Elemental analysis, IR and NMR are in agreement with the proposed structure. The formation of 6 may be rationalized via the nucleophilic addition of nitrogen atom of hydrazine to the thio group followed by nucleophilic addition of the other nitrogen to the imino group followed by elimination of ammonia as shown in Scheme 2 Dihydrazono derivatives 8a,b were achieved as a sole product by treating the iminothione 3h,i with two moles of hydrazine hydrate in ethanol (Scheme 2). The product of this reaction was identified on the basis of both elemental analyses and spectral data. 1H NMR spectrum of 8b displayed triplet and quartet signals at: δ =1.32, 4.01 due to the ethoxy group and broad singlet signals at: δ = 5.65 assignable the 2NH2. 13C NMR showed complete disappearance of the signal in the range around 180 ppm due to the functional group change from C=S to C=N and the presence of signals at: 152.6, 159.0 corresponding C=N and C=O groups.
Scheme 2.
Reactions of imidazolidineiminothiones with hydrazone derivatives and hydrazine hydrate.
Furthermore, upon reaction of the iminothiones 3a,f,h,i with thiosemicarbazide, the nucleophilic addition occurred at the thio group and the corresponding 4-thiosemicarbazone derivatives 9a-d were obtained in good yield. Compounds 9 were not converted into the triazine derivatives 10 under these conditions. The chemical structure of 9a-d was elucidated on the basis of elemental analyses and spectral data. 1HNMR spectrum of 9d as a representative example revealed triplet and quartet signals at: δ = 1.30 and 4.03 ppm, respectively, characteristic for ethoxy protons, in addition to three broad singlet signals at: 8.46, 9.47, 12.79 due to NH and NH2 protons. Now, we have extended our synthetic program to the synthesis of otherwise inaccessible heterocyclic ring system utilizing imidazolidineiminothiones 3 as the key starting material. Thus condensation of the imidazolidineiminothione 3i with o-phenylenediamine derivatives as 1,4-binucleophile in ethanol under reflux afforded yellow products which were identified as imidazo[4,5-b]quinoxalines 11a-c. The spectral data of the isolated product were in complete agreement with structure 11. IR spectrum revealed lack of an absorption band corresponding to a C=NH functional group. Mass spectrum of compound 11c showed molecular ion peak at m/z 396 (base peak) corresponding to molecular formula C24H20N6O2. Finally, it was interesting to study the behavior of imidazolidi-
neiminothiones toward carbon nucleophiles. Thus, condensation of the imidazolidineiminothiones 3c,i with 3-methyl-1H-pyrazol-5(4H)-one derivatives in boiling ethanol using triethylamine as a basic catalyst afforded a product which was identified as imidazolidin-4-ylidenpyrazolones 12a-d (Scheme 3 ). IR spectrum of 12d as a representative example revealed absorption bands at: 3280, 1717 and 1603 characteristic to NH, C=O and C=N functional groups, respectively. The 1H NMR spectrum exhibited triplet, quartet and singlet at: 1.14, 4.19 and 2.41 ppm due to ethoxy and methyl protons, singlet signal at 12.83 specific for NH. Moreover, its 13C NMR spectrum revealed signals at: 14.8, 15.1, 63.3 assigned ethoxy and methyl carbons signals 105.0, 144.7 corresponding for C=C and signals at 159.3, 159.4, 162.3 and 166.6 assigned to 2 imino and 2 carbonyl carbons.
Scheme 3.
Reactions of imidazolidineiminothione derivatives with different reagents.
Antibacterial and Antifungal Activities
The synthesized compounds were tested in vitro for antibacterial and antifungal activities against the following strains: three Gram-positive bacteria, Staphylococcus aureus RCMB 010027, Staphylococcus epidermidis RCMB 010024 and Bacillis subtilis RCMB 010063; three Gram-negative bacteria, Neisseria gonorrhoeae RCMB 010079, Esherichia coli RCMB 010052 and Klebsiella pneumoniae RCMB 010093, and three Fungi, Aspergillus fumigatus RCMB 02564, Aspergillus clavatus RCMB 02593 and Geotricum Candidum RCMB 05096, and the results were summarized in Table 1. The synthesized compounds were tested for antimicrobial activity by the agar diffusion method [21] using a 1 cm microplate well diameter and a 100 μL of each concentration. The antifungal agents were evaluated against clinical isolates of standard strains of fungi by the broth dilution methods Broth dilution method according to NCCLs [22, 23]. Tested compound solution prepared by dissolving 5 mg of the chemical compound in 1 mL of dimethyl sulfoxide (DMSO). The inoculated plates were then incubated for 24 h at 37 °C. Ampicillin, Gentamycin and Amphotericin B (1 mg/mL) were used as standard references for Gram positive bacteria, Gram negative bacteria and antifungal activity, respectively. After incubation time, antimicrobial activity was evaluated by measuring the inhibition zone diameters against the test organisms and compared with standard zone size ranges that determine susceptibility, intermediate susceptibility, or resistance to the screened compounds. Visual bacterial growth is observed only in areas in which the drug concentrations are below those required for growth inhibition. The experiment was carried out in triplicate and the average zone of inhibition was calculated.
The mean values of the inhibition zone diameter obtained for these compounds suggest that all of the imidazole derivatives evaluated possess significant antibacterial activity against most of the test organisms used in these assays. A moderate difference in antibacterial activity is noted between the tested compounds, this indicate that the main effect related to the presence of the imidazolidinone moiety. Using the general structure provided in (Fig. 1), certain aspects of the structure activity relationships for these compounds can be more clearly highlighted.
Fig. (1).

General formula of the synthesized compounds.
A series of imidazolidineiminothione derivatives 3 which contain 4-fluorophenyl, 3-methoxyphenyl, 3-ethoxyphenyl moieties (Ar) at N1 and phenyl, 2-fluorophenyl, 4-chlorophenyl, 3,5-di(trifluoromethyl)-phenyl moieties (Ar') at N3, X = O, Y = S and Z = NH was synthesized and their biological activates were measured and studied. Regarding the effect of Ar and Ar' groups. The type of the substitutions on the benzene ring of aryl moiety is important. It was noticed that the presence of 3-methoxyphenyl and phenyl in 3f and 3-ethoxyphenyl and 4-chlorophenyl in 3j showed the maximum activities against most of the test organisms which showed near the reference drug. On the other hand, compounds 3b,g,h,i showed moderate to good activity against most of the tested organisms. Compound 3k with 4-fluorophenyl and 3,5-di(trifluoromethyl)-phenyl moieties has moderate activity the Gram +ve bacteria and no activity toward the Gram -ve bacteria. Regarding the effect of changing the substituents on N1 and N3, the results of screening demonstrated the presence of good correlation between the antifungal and antibacterial activities. Changing the substituent on C4 from imino to oxo (3h,i → 4a,b) was carried out to show the deference between imino and oxo on the effect of the antimicrobial activity. Compound 4a showed higher activity more than its imino analogues 3h while compound 4b has less activity than its imino analogues compound 3i. Y and Z substituents in imidazolidineiminothione derivatives 3 were changed to monohydrazono, dihydrazono and thiosemicarbazono moieties to do the same study. Compounds 6, 8, 9 with mono hydrazono, dihydrazono and thiosemicarbazono moieties have weak activity against Gram +ve bacteria and no activity against Gram -ve activity. Changing the substituent at C4 from imino to hydrazono (3h,i → 6a,b) showed lower activity than their imino analogues 3h,i. Also, changing the substituents on C4 and C5 from thioxo and imino to hydrazono (3h,i → 8a,b) showed lower activity than its analogues 3h,i. Moreover, changing the substituent on C4 from thioxo to thiosemicarbazono (3a,f,h → 9a,b,c) showed lower activity than their analogues. The authors also studied the effect of changing of the substituent at C4 and C5 to cyclic substituents at imidazolidine ring on the biological activities. Compounds 11a-c with quinoxaline moiety showed strong activity against all of the tested organisms, where compound 11c was equipotent to the reference drug. Finally, the vary substitution on C5 from thioxo to substituted pyrazolone moiety (3i → 12c,d) had slight effect on antibacterial activity. Compounds 12c,d with moiety showed moderate activity with all the tested Gram positive and negative bacteria. The comparison between the antimicrobial activity of our potent synthesized compounds and standard reference drug against the used Gram positive, Gram negative bacteria and fungi is represented graphically in Figure 2.
Fig. (2).
The comparison between the antimicrobial activity of our potent synthesized compounds and standard drug against the used Gram positive, Gram negative bacteria and fungi.
MIC of the Most Active Compounds
Minimum inhibitory concentration (MIC) of the most active synthesized compounds 3f, 3k, 4a, 11c, 12a, and 12b was evaluated in vitro using the Broth dilution method according to NCCLs [22, 23]. The results of minimum inhibitory concentration are depicted in Table 2. Regarding the effect of each substituent at C2, C4, C5 and those at N1 and N3 against bacterial and fungal strains, results of antimicrobial activity in this study revealed that the presence of quinoxaline moiety in compound 11c resulted in highest antimicrobial activity among all the investigated compounds. The latter compound showed better results when compared with standard drugs as revealed from its MIC values (0.12 - 0.98 μg/mL). Compound 11c was equipotent to Ampicillin in inhibiting the growth of B. subtilis (MIC 0.15 μg/mL). This compound was equipotent to Gentamycin in inhibiting the growth of K. pneumonia (MIC 0.12 μg/mL). Also, this compound was equipotent to Amphotericin B in inhibiting the growth of A. clavatus (MIC 0.49 μg/mL) and G. candidum (MIC 0.12 μg/mL). In certain cases, such values are low enough to render such agents as potential candidates for further studies.
Table 2. Minimum inhibitory concentration (µg/mL) of the more potent synthesized compounds against the pathological organisms.
| Compd. No. | Gram +ve | Gram -ve | Fungi | ||||||
|---|---|---|---|---|---|---|---|---|---|
| S. aureus | S.epidermidis | B. subtilis | N.gonorrhoeae | E. coli | K.pneumoniae | A. fumigatus | A. clavatus | G. candidum | |
| 3f | 1.95 | 0.98 | 0.49 | 31.25 | 7.81 | 7.81 | 0.98 | 7.81 | 0.49 |
| 3k | 3.9 | 1.95 | 0.98 | 125 | 15.63 | 3.9 | 7.81 | 7.81 | 1.95 |
| 4a | 1.95 | 15.63 | 1.95 | 125 | 31.25 | 15.63 | 31.25 | 3.9 | 1.95 |
| 11c | 0.49 | 0.49 | 0.12 | 0.98 | 0.49 | 0.12 | 0.98 | 0.49 | 0.12 |
| 12a | 15.63 | 3.9 | 1.95 | 500 | 125 | 125 | 62.5 | 15.6 | 1.95 |
| 12b | 3.9 | 0.98 | 0.98 | 500 | 31.25 | 31.25 | 15.63 | 7.8 | 1.95 |
| St. (A) | 0.03 | 0.12 | 0.15 | ----- | ----- | ----- | ----- | ----- | ----- |
| St. (B) | ----- | ----- | ----- | 0.49 | 0.24 | 0.12 | ----- | ----- | ----- |
| St. (C) | ----- | ----- | ----- | ----- | ----- | ----- | 0.12 | 0.49 | 0.12 |
St. (A): Ampicillin, St. (B): gentamycin and St. (C): Amphotericin b
Conclusion
The aim of the present investigation is to synthesize different series of imidazolidin-2-one bearing various substituents at N1 and others at N3 beside various substituents at C1, C3 and C4. The authors measured the antibacterial and antifungal activities of these derivatives. They studied the effect of each substituent on these activities and make a comparative study between them to deduce a structure activity relationship. Most compounds displayed antibacterial and antifungal activities. A moderate difference in antimicrobial activity is noted between the tested compounds, this indicates the main effect related to the presence of the imidazolidinone moiety. Our results clearly revealed that the best antimicrobial activity was observed for 1-(3-ethoxyphenyl)-6-methyl-1-phenyl-1H-imidazo[4,5-b]quinoxalin-2(3H)-ones (11c) followed by 5-imino-3-(3-methoxy- phenyl)-1-phenyl-4-thioxoimidazolidin-2-one (3f).
Experimental Section
All melting points are recorded on digital Gallen Kamp MFB-595 instrument and are uncorrected. The IR spectra (KBr) (cm-1) were measured on a Shimadzu 440 spectrophotometer. NMR spectra (δ, ppm) were obtained in deuterated dimethyl sulfoxide on a Varian Gemini 500 (500 MHz) spectrometer, using TMS as an internal standard; chemical shifts are reported as δ ppm units. Mass spectra (m/z, %) were obtained on GC MS-QP 100 Exmass spectrometer at 70 eV. Elemental analyses were carried out at Microanalytical Unit, Cairo University, Cairo, Egypt.
General Procedure for the Synthesis of Arylcarbamothioyl Cyanide Derivatives 2d,e
To a solution of isothiocyanate derivative (3-methoxyphenyl isothiocyanate or 3-ethoxyphenyl isothiocyanate) (0.01 mol) in ethanol (50 ml), a solution of KCN (0.01 mol) in 5 ml water was added slowly while stirring. After complete addition, stirring of the reaction mixture was continued for additional 1 h. The reaction mixture was poured into acidified crushed ice. The resulting precipitate was filtrated off, dried and crystallized from ethanol to give 2d, e.
(3-Methoxyphenyl)carbamothioyl cyanide (2d): as yellow crystals; yield 65%; mp 75-77 °C; IR: ν/cm-1 = 3251 (NH), 3041 (CH-Ar), 2996 (CH-aliph.), 2221 (C≡N); 1H NMR: δ/ppm = 3.83 (s, 3H, CH3), 6.96 -7.68 (m, 4H, Ar-H), 9.55 (br, 1H, NH; cancelled with D2O); 13C NMR: 55.6 (OCH3), 113.7, 114.4, 124.2, 129.9, 130.2, 137.4, 159.0, 161.0; Anal. Calcd for C9H8N2OS (192.24): C, 56.23; H, 4.19; N, 14.57; Found: C, 56.44; H, 4.35; N, 14.81%.
(3-Ethoxyphenyl)carbamothioyl cyanide (2e): as yellow crystals; yield 73%; mp 83-85 °C; IR: ν/cm-1 = 3264 (NH), 3066 (CH-Ar), 2980 (CH-aliph.), 2227 (C≡N); 1H NMR: δ/ppm = 1.23 (t, 3H, J = 6.9 Hz, CH3), 4.14 (q, 2H, J = 7.0 Hz, CH2), 6.83-7.68 (m, 4H, Ar-H), 13.36 (br, 1H, NH; cancelled with D2O); 13CNMR: 14.1 (CH3), 63.3 (CH2), 107.9, 113.7, 114.5, 130.3, 138.7, 140.3, 158.8, 161.1; Anal. Calcd. for C10H10N2OS (206.26): C, 58.23; H, 4.89; N, 13.58; Found: C, 58.42; H, 4.76; N, 13.84%.
General Procedure for the Synthesis of 5-imino-4-thioxoimidazolidin-2-one Derivatives 3
To a solution of 2 (0.0l mo1) in ether (20 ml), isocyanates (0.0l mo1) and triethylamine (0.5 ml) were added; the reaction mixture was stirred at room temperature for 1 hr. The obtained products were crystallized from the chloroform / n-hexane to afford 3.
5-Imino-4-thioxoimidazolidin-2-ones (3a,c-e) were prepared according to the reported method [ 20 ]. 1-(3,5-Bis(trifluoromethyl)phenyl)-3-(4-fluorophenyl)-5-imino-4-thioxoimidazolidin-2-one (3b): as orange crystals; yield 83%; mp 105-107 °C; IR: ν/cm-1 = 3215 (NH), 1774 (C=O), 1664 (C=N); 1H NMR: δ/ppm = 7.12-8.35 (m, 7H, Ar-H), 10.44 (br, 1H, NH; cancelled with D2O); 13C NMR: 116.8, 117.0, 120.1, 122.2, 124.4, 126.6, 127.9, 129.7, 130.5, 131.3, 131.5, 134.9, 153.5, 153.8, (12C-Ar + 2CF3), 161.6 (C=N), 163.6 (C=O), 182.9 (C=S); Anal. Calcd. for C17H8F7N3OS (435.32): C, 46.90; H, 1.85; N, 9.65; Found: C, 46.55;H, 1.87; N, 9.76%.
4-Imino-1-(3-methoxyphenyl)-3-phenyl-5-thioxoimida-zolidin-2-one (3f): as orange crystals; yield 78%; mp 118-120 °C; IR: ν/cm-1 = 3261 (NH), 2988, 2933 (CH-aliph), 1777 (C=O), 1600 (C=N); 1H NMR: δ/ppm = 3.80 (s, 3H, OCH3), 7.10-7.50 (m, 9H, Ar-H), 9.66 (br, 1H, NH; cancelled with D2O); 13C NMR: 65.3 (OCH3), 115.3, 118.5, 120.3, 122.3, 128.5, 129.3, 130.3, 133.1, 134.9, 140.1, 153.0, 154.1 (12C-Ar), 154.5 (C=N), 160.1 (C=O), 183.3 (C=S); MS, m/z (%): 311 (M+, 1.30), 261 (13.67), 205 (2.4), 180 (13.25), 166 (14.11), 165 (81.01), 119 (20.49), 104 (13.67), 91 (46.48), 77 (100); Anal. Calcd. for C16H13N3O2S (311.36): C, 61.72; H, 4.21; N, 13.50; Found: C, 61.53; H, 4.36; N, 13.62%.
1-(2-Fluorophenyl)-5-imino-3-(3-methoxyphenyl)-4-thioxoimidazolidin-2-one (3g): as orange crystals, yield 62%; mp 143-145 °C; IR: ν/cm-1 = 3264 (NH), 3064 (CH-Ar), 2986, 2936 (CH-aliph), 1778 (C=O), 1602 (C=N); 1H NMR: δ/ppm = 3.80 (s, 3H, OCH3), 7.10-7.69 (m, 8H, Ar-H), 9.82 (br, 1H, NH; cancelled with D2O), 13CNMR 65.3 (OCH3), 115.7, 116.9, 117.1, 120.2, 125.1, 125.5, 130.3, 131.8, 134.3, 153.5, 153.5, 153.5 (12C-Ar), 154.0 (C=N), 160.1 (C=O), 183.1 (C=S); Anal. Calcd. for C16H12FN3O2S (329.35): C, 58.35; H, 3.67; N, 12.76; Found: C, 58.53; H, 3.84; N, 12.54%.
1-(4-Chlorophenyl)-5-imino-3-(3-methoxyphenyl)-4-thioxoimidazolidin-2-one (3h): as yellow crystals; yield 68%; mp 127-129 °C, IR: ν/cm-1 = 3231 (NH), 2934 (CH-aliph.), 1772 (C=O), 1665 (C=N); 1H NMR: δ/ppm = 3.79 (s, 3H, OCH3), 7.11-7.66 (m, 8H, Ar-H), 9.74 (s, 1H, NH; cancelled with D2O), 13C NMR: 55.9 (OCH3), 114.0, 115.5, 120.2, 122.3, 129.1, 129.3, 129.4, 130.5, 131.9, 133.0, 134.8, 153.9 (12C-Ar), 154.3 (C=N), 160.1 (C=O), 183.2 (C=S); MS, m/z (%): 345 (M+, 100), 347 (M+2; 39.0), 314 (2.0), 192 (22.1), 191 (20.4), 165 (44.1), 161 (10.8), 135 (10.2), 125 (4.3), (3.8), 77 (15.9); Anal. Calcd. for C16H12ClN3O2S (345.80): C, 55.57; H, 3.50; N, 12.15; Found: C, 55.76; H, 3.37; N, 12.23%.
1-(3-Ethoxyphenyl)-4-imino-3-phenyl-5-thioxoimidazolidin-2-one (3i): as orange crystals; yield 82%; mp 100-102 °C; IR: ν/cm-1 = 3217 (NH), 2974, 2879 (CH-aliph), 1769 (C=O), 1599 (C=N); 1H NMR: δ/ppm = 1.34 (t, 3H, J = 6.9 Hz, CH3), 4.05 (q, 2H, J = 6.9 Hz, CH2), 7.08-7.59 (m, 9H, Ar-H), 9.67 (br, 1H, NH; cancelled with D2O); 13C NMR: 15.0 (CH3), 63.9 (CH2), 114.4, 116.0, 120.1, 127.6, 128.6, 129.3, 130.4, 130.5, 131.9, 133.0, 134.9, 154.1 (12C-Ar), 154.6 (C=N), 159.3 (C=O), 183.3 (C=S); Anal. Calcd. for C17H15N3O2S (325.38): C, 62.75; H, 4.65; N, 12.91; Found: C, 62.84; H, 4.42; N, 12.75%.
1-(3-Ethoxyphenyl)-3-(2-fluorophenyl)-4-imino-5-thioxoimidazolidin-2-one (3j): as orange crystals; yield 71%, mp 105-107 °C; IR: ν/cm-1 = 3262 (NH), 2969, 2886 (CH-aliph.), 1775 (C=O), 1597 (C=N); 1H NMR: δ/ppm = 1.34 (t, 3H, J = 6.8 Hz, CH3), 4.04 (q, 2H, J = 6.9 Hz, CH2), 7.08-7.68 (m, 8H, Ar-H), 9.81 (br, 1H, NH; cancelled with D2O); 13C NMR: 14.3 (CH3), 63.9 (CH2), 114.3, 116.1, 116.9, 117.1, 120.3, 125.5, 130.5, 133.5, 130.9, 131.9, 134.7, 153.5 (12C-Ar), 157.0 (C=N), 159.4 (C=O), 183.1 (C=S); MS, m/z (%): 343 (M+, 100), 344 (M+1; 20.6), 324 (12.9), 205 (7.6), 179 (16.0), 161 (8.2), 151 (28.8), 93 (6.4), 65 (9.3); Anal. Calcd. for C17H14FN3O2S (343.38): C, 59.46; H, 4.11; N, 12.24; Found: C, 59.65; H, 4.32; N, 12.42%.
1-(4-Chlorophenyl)-3-(3-ethoxyphenyl)-5-imino-4-thioxoi-midazolidin-2-one (3k): as golden crystals; yield 78%; mp 142-144 °C; IR: ν/cm-1 = 3242 (NH), 2980, 2921 (CH-aliph), 1770 (C=O), 1604 (C=N); 1H NMR: δ/ppm = 1.36 (t, 3H, J = 7.0 Hz, CH3), 4.09 (q, 2H, J = 6.9 Hz, CH2), 7.07-7.63 (m, 8H, Ar-H), 9.72 (s, 1H, NH; cancelled with D2O); 13C NMR: 14.9 (CH3), 63.9 (CH2), 114.4, 116.0, 120.1, 120.3, 125.9, 129.0, 129.4, 130.5, 133.0, 134.1, 139.1, 152.4 (12C-Ar), 153.3 (C=N), 154.3 (C=O), 183.3 (C=S); Anal. Calcd. for C17H14ClN3O2S (359.83): C, 56.74; H, 3.92; N, 11.68; Found: C, 56.91; H, 3.75; N, 11.76%.
Synthesis of Imidazolidin-2,5-diones 4a,b
The imidazolidineiminothione derivative 3h or 3i (5 mmol) was dissolved in boiling ethanol (20 mL) and treated with dil. HCl (1:1 molar ratio). The obtained products were filtered off, washed with cold water, air-dried, and recrystallized from chloroform/n-hexane to give the corresponding diones 4a,b, respectively.
3-(4-Chlorophenyl)-1-(3-methoxyphenyl)-5-thioxoimida-zolidine-2,4-dione (4a): as orange crystals; yield 62%; mp 100-102 °C; IR: ν/cm-1 = 2918, 2826 (CH-aliph), 1749, 1797 (C=O); 1H NMR: δ/ppm = 3.82 (s, 3H, OCH3), 7.06-7.68 (m, 8H, Ar-H); Anal. Calcd. for C16H11ClN2O3S (346.79): C, 55.41; H, 3.20; N, 8.08; Found: C, 55.64; H, 3.42; N, 8.26%.
1-(3-Ethoxyphenyl)-3-phenyl-5-thioxoimidazolidine-2,4-dione (4b): as orange crystals; mp 157-159 °C; yield 76%; IR: ν/cm-1 = 2982, 2895 (CH-aliph), 1745, 1802 (C=O); 1H NMR: δ/ppm = 1.34 (t, 3H, J = 6.8 Hz, CH3), 4.11 (q, 2H, J = 6.9 Hz, CH2), 7.03-7.65 (m, 8H, Ar-H); Anal. Calcd. for C17H13ClN2O3S (360.81): C, 56.59; H, 3.63; N, 7.76; Found: C, 56.75; H, 3.84; N, 7.45%.
Synthesis of Azine Derivatives 5a-c
To a solution of 3a, 3d, 3i (0.0l mo1) in ethanol (20 ml), benzophenonehydrazone (0.01mol) was added and the reaction mixture was heated under reflux for 3hr. The obtained product was filtered, washed with ethanol and crystallized from ethanol/dioxane to afford 5a-c.
4-((Diphenylmethylene)hydrazono)-3-(4-fluorophenyl)-5-imino-1-phenylimidazolidin-2-one (5a): as yellow crystals; yield 62%, mp 170-172 °C, IR: ν/cm-1 = 3265 (NH), 1758 (C=O), 1624 (C=N); 1H NMR: δ/ppm = 7.34-7.62 (m, 19H, Ar-H), 10.11 (br, 1H, NH; cancelled with D2O); 13C NMR: 115.6, 126.5, 127.3, 128.1, 128.3, 128.4, 128.5, 128.8, 129.2 (3C), 129.4 (2C), 130.0, 130.1, 130.2, 133.7, 134.4, 134.8, 137.4, 142.4, 150.7, 152.1, 152.8 (24 C-Ar), 160.4, 160.7, 162.4 (3C=N), 167.5 (C=O); Anal. Calcd. For C28H20FN5O (461.49): C, 72.87; H, 4.37; N, 15.18; Found: C, 72.77; H, 4.27; N, 15.29%.
4-((Diphenylmethylene)hydrazono)-5-imino-1-phenyl-3-p-tolylimidazolidin-2-one 5b: as yellow crystals; mp 162-164 °C; yield 72%; IR: ν/cm-1 = 3249 (NH), 1757 (C=O), 1612 (C=N); 1H NMR: δ/ppm = 2.27 (s, 3H, CH3), 7.13-7.61 (m, 19H, Ar-H), 10.04 (br, 1H, NH; cancelled with D2O); 13C NMR: 21.1 (CH3), 115.4, 126.5, 127.3, 128.1, 128.3, 128.4, 128.5, 128.8, 129.1 (2C), 129.2, 129.3, 129.9, 130.0, 130.1, 131.3, 132.8, 134.3, 134.4, 137.7, 137.5, 142.2, 145.3, 148.1 (24 C-Ar), 150.8, 153.7, 153.0 (3C=N), 167.3 C=O); Anal. Calcd. for C29H23N5O (457.53): C, 76.13; H, 5.07; N, 15.31; Found: C, 76.28; H, 5.19; N, 15.43%.
4-((Diphenylmethylene)hydrazono)-3-(3-ethoxyphenyl)-5-imino-1-phenylimidazolidin-2-one (5c): as yellow crystals; yield 67%; mp 165-167 °C; IR: ν/cm-1 = 3240 (NH), 1751 (C=O), 1609 (C=N); 1H NMR: δ/ppm = 1.23 (t, 3H, J = 6.8 Hz, CH3), 3.86 (q, 2H, J = 6.8 Hz, CH2), 6.86-7.61 (m, 19H, Ar-H), 10.07 (br, 1H, NH; cancelled with D2O); 13C NMR: 14.9 (CH3), 63.6 (CH2), 114.5, 120.0, 125.9, 127.6, 127.7, 128.1, 128.2, 128.3, 128.5, 128.7, 128.9, 129.0, 129.1, 129.2, 129.4, 129.8, 130.0, 130.7, 131.3, 132.8, 133.5, 134.2, 137.6, 142.3 (24 C-Ar), 150.7, 152.9, 158.8 (3C=N), 167.4 (C=O); MS, m/z (%): 487 (M+, 3.0), 488 (M+1, 1.6), 459 (41.2), 458 (100), 430 (14.5), 410 (30.9), 368 (15.9), 323 (51.6), 291 (18.6), 135 (11.0), 119 (14.8), 77 (44.3); Anal. Calcd. for C30H25N5O2 (487.55): C, 73.90; H, 5.17; N, 14.36; Found: C, 73.76; H, 5.29; N, 14.44%.
Synthesis of Hydrazone Derivatives 6a,b
A solution of 3h, 3i (0.01 mol) in ethanol (30 ml) was treated with hydrazine hydrate (0.012 mol), and the reaction mixture was stirred at room temperature for 15 min. The product was collected and recrystallized from ethanol/dioxane to give 6a,b.
1-(4-Chlorophenyl)-5-hydrazono-3-(3-methoxyphenyl)-4-thioxoimidazolidin-2-one (6a): as yellow crystals; yield 61%; mp 213-215 °C; IR: ν/cm-1 = 3372, 3119 (NH2), 2965, 2886 (CH-aliph), 1725 (C=O), 1598 (C=N); 1H NMR: δ/ppm = 1.22 (s, 3H, CH3), 5.78 (br, 2H, NH2), 7.02-7.87 (m, 8H, Ar-H); MS, m/z (%): 360 (M+, 100), 361 (M+1, 22.7), 362 (M+2, 36.4), 334 (17.1), 332 (40.8), 166 (11.0), 164 (9.4), 148 (9.1), 134 (60.2), 111 (11.7), 107 (18.4), 77 (14.3); Anal. Calcd. for C16H13ClN4O2S (360.82): C, 53.26; H, 3.63; N, 15.53; Found: C, 53.42; H, 3.76; N, 15.48%.
1-(3-Ethoxyphenyl)-4-hydrazono-3-phenyl-5-thioxoimidazolidin-2-one (6b): as yellow crystals; yield 65%; mp 158-160 °C; IR: ν/cm-1 = 3327, 3169 (NH2), 3109 (CH-Ar), 2984, 2884 (CH-aliph.), 1722 (C=O), 1598 (C=N); 1H NMR: δ/ppm = 1.42 (t, 3H, J = 7.1 Hz, CH3), 4.05 (q, 2H, J = 7.0 Hz, CH2), 5.56 (br, 2H, NH2), 7.01-7.62 (m, 9H, Ar-H); 13C NMR: 14.7 (CH3), 63.6 (CH2), 113.7, 114.0, 116.1, 120.7, 126.1, 127.8, 128.9, 130.1, 132.1, 133.1, 135.2, 150.1, 152.6, 159.0, 183.6 (C=S); MS, m/z (%): 340 (M+, 100), 341 (M+1, 25.1), 313 (17.2), 312 (82.1), 295 (19.7), 164 (11.4), 149 (14.2), 148 (36.7), 136 (20.4), 135 (17.3), 121 (20.1), 104 (16.8), 93 (11.6), 77 (38.8); Anal. Calcd. for C17H16N4O2S (340.40): C, 59.98; H, 4.74; N, 16.46; Found: C, 59.75; H, 4.63; N, 16.73%.
Synthesis of Dihydrazone Derivatives 8a,b
A mixture of 3h, 3i (0.01 mol) and hydrazine hydrate (0.03 mol) in ethanol (30 ml) was stirred for 30 min. at room temperature. The product was recrystallized from ethanol/dioxane to give 8a, b.
1-(4-Chlorophenyl)-4,5-dihydrazono-3-(3-methoxyphenyl)imidazolidin-2-one (8a): as yellow crystals; yield 63%; mp 193-195 °C; IR: ν/cm-1 = 3373, 3193 (NH2), 2978 (CH-aliph), 1724 (C=O), 1603 (C=N); 1H NMR: δ/ppm = 1.21 (s, 3H, CH3), 5.65 (br, 4H, 2NH2), 6.91-7.58 (m, 8H, Ar-H); MS, m/z (%): 358 (M+, 1.76), 343 (32.7), 177 (12.8), 173 (15.6), 161 (16.7), 151 (100), 136 (18.7), 135 (37.4), 122 (33.6), 107 (41.8), 80 (13.3); Anal. Calcd. for C16H15ClN6O2 (358.78): C, 53.56; H, 4.21; N, 23.42; Found: C, 53.78; H, 4.45; N, 23.56%.
1-(3-Ethoxyphenyl)-4,5-dihydrazono-3-phenylimidazolidin-2-one (8b): as yellow crystals; yield 77%; mp 150-152 °C, IR: ν/cm-1 = 3372, 3210 (NH2), 3067 (CH-Ar), 2925, 2833 (CH-aliph), 1724 (C=O), 1600 (C=N); 1H NMR: δ/ppm = 1.32 (t, 3H, J = 7.0 Hz, CH3), 4.01 (q, 2H, J = 6.9 Hz, CH2), 5.65 (br, 4H, 2NH2), 6.91-7.58 (m, 9H, Ar-H); 13C NMR: 15.0 (CH3), 63.6 (CH2), 113.8, 114.5, 116.0, 120.2, 127.4, 128.4, 129.0, 129.7, 131.2, 133.0, 134.7, 150.1 (12C-Ar), 152.6 (2C=N), 159.0 (C=O); MS, m/z (%): 338 (M+, 100), 339 (M+1; 22.5), 307 (34.0), 175 (11.9), 148 (53.3), 135 (13.4), 119 (14.2), 77 (32.4); Anal. Calcd. for C17H18N6O2 (338.36): C, 60.34; H, 5.36; N, 24.84; Found: C, 59.95; H, 5.53; N, 24.73%.
Synthesis of Thiosemicarbazone Derivatives 9a-d
To a solution of 3a, 3f, 3h, 3i (0.0l mo1) in ethanol (20 ml), thiosemicarbazide (0.01mol) was added and the reaction mixture was heated under reflux for 3hr. The obtained product was filtered, washed with ethanol and crystallized from ethanol/dioxane to afford 9a-d.
2-(3-(4-Fluorophenyl)-5-imino-2-oxo-1-phenylimidazolidin-4- ylidene)hydrazinecarbothioamide (9a): as orange crystals; yield 62%; mp 215-217 °C; IR: ν/cm-1 = 3426, 3260, 3148 (NH2, NH), 1758 (C=O), 1611 (C=N); 1H NMR: δ/ppm = 6.87-7.74 (m, 9H, Ar-H), 8.43, 9.72, 11.63 (3br, 4H, NH2+2NH); Anal. Calcd. For C16H13FN6OS (356.38): C, 53.92; H, 3.68; N, 23.58; Found: C, 53.69; H, 3.88; N, 23.76%.
2-(5-Imino-3-(3-methoxyphenyl)-2-oxo-1-phenylimidazolidin-4-ylidene)hydrazinecarbothioamide (9b): as yellow crystals; yield 61%; mp 218-220 °C; IR: ν/cm-1 = 3442, 3251, 3203 (NH2, NH), 2964, 2930 (CH-aliph), 1765 (C=O), 1645 (C=N); 1H NMR: δ/ppm = 1.23 (s, 3H, CH3), 6.91-7.63 (m, 9H, Ar-H), 8.52, 9.33, 11.87 (3br, 4H, NH2+2NH); Anal. Calcd. for C17H16N6O2S (368.41): C, 55.42; H, 4.38; N, 22.81; Found: C, 55.67; H, 4.69; N, 22.75%.
2-(1-(4-Chlorophenyl)-5-imino-3-(3-methoxyphenyl)-2-oxoimidazolidin-4-ylidene)hydrazinecarbothioamide (9c): as yellow crystals; mp 222-224 °C; yield 59%; IR: ν/cm-1 = 3421, 3238, 3139 (NH2, NH), 2934 (CH-aliph), 1757 (C=O), 1640 (C=N); 1H NMR: δ/ppm = 1.21 (s, 3H, CH3), 6.95-7.87 (m, 8H, Ar-H), 8.53, 9.34, 11.92 (3br, 4H, NH2+2NH); MS, m/z (%): 402 (M+, 9.2), 346 (21.3), 255 (37.0), 253 (100), 252 (50.6), 249 (32.0), 180 (11.9), 166 (13.7), 155 (31.6), 153 (99.0), 119 (13.5), 77 (49.4); Anal. Calcd. For C17H15ClN6O2S (402.86): C, 50.68; H, 3.75; N, 20.86; Found: C, 50.39; H, 3.86; N, 20.79%.
2-(3-(3-Ethoxyphenyl)-5-imino-2-oxo-1-phenylimidazolidin-4-ylidene)hydrazinecarbothioamide (9d): as yellow crystals yield 75%, mp 188-190 °C, IR: ν/cm-1 = 3382, 3254, 3162 (NH2, NH), 2978 (CH-aliph), 1775 (C=O), 1607 (C=N); 1H NMR: δ/ppm = 1.30 (t, 3H, J = 7.0 Hz, CH3), 4.03 (q, 2H, J = 7.0 Hz, CH2), 6.91-7.63 (m, 9H, Ar-H), 8.46, 9.47, 12.79 (3s, 4H, NH2+2NH); MS, m/z (%): 382 (M+, 16.5), 383 (M+1, 8.1), 365 (38.9), 348 (15.8), 323 (1.5), 263 (17.3), 219 (66.4), 218 (50.2), 180 (28.7), 163 (27.9), 135 (47.5), 119 (62.0), 93 (100), 77 (45.0); Anal. Calcd. for C18H18N6O2S (382.44): C, 56.53; H, 4.74; N, 21.97; Found: C, 56.34; H, 4.84; N, 21.76%.
Synthesis of imidazo[4,5-b]quinoxalin-2(3H)-one derivatives 11a-c
A solution of (3i, 0.0l mo1) in ethanol (20 ml) was treated with o-phenylenediamine derivatives (0.0l mo1) and triethylamine (0.5 ml). The reaction mixture was heated under reflux for 3hr and the solid that obtained after filtration was crystallized from ethanol/dioxane to give 11a-c.
1-(3-Ethoxyphenyl)-3-phenyl-1H-imidazo[4,5-b]quinoxalin-2(3H)-one (11a): as white crystals; yield 62%; mp 160-162 °C; IR: ν/cm-1 = 3055 (CH-Ar), 2975 (CH-aliph.), 1744 (C=O), 1597 (C=N); 1H NMR: δ/ppm = 1.37 (t, 3H, J = 7.0 Hz, CH3), 4.10 (q, 2H, J = 7.0 Hz, CH2), 7.06-7.98 (m, 13H, Ar-H); 13C NMR: 15.0 (CH3), 63.8 (CH2), 113.6, 114.6, 119.1, 119.2, 127.2, 127.5, 128.6, 129.3, 129.6, 130.3 (2C), 132.9, 133.9, 136.9, 137.0, 137.4, 138.8, 140.6 (18C-Ar), 152.6 (2C=N), 159.2 (C=O); Anal. Calcd. for C23H18N4O2 (382.41): C, 72.24; H, 4.74; N, 14.65; Found: C, 72.36; H, 4.44; N, 14.76%.
6-Chloro-1-(3-ethoxyphenyl)-3-phenyl-1H-imidazo[4,5-b]quinoxalin-2(3H)-one (11b): as gray crystals; yield 63%; mp 130-132 °C; IR: ν/cm-1 = 3064 (CH-Ar), 2960 (CH-aliph.), 1748 (C=O), 1600 (C=N); 1H NMR: δ/ppm = 1.37 (t, 3H, J = 6.8 Hz, CH3), 4.09 (q, 2H, J = 7.0 Hz, CH2), 7.09-7.98 (m, 12H, Ar-H); 13C NMR: 16.0 (CH3), 53.9 (CH2), 113.6, 115.4, 118.5, 119.3, 126.2, 126.8, 127.3 (3C), 127.8, 128.9, 129.5, 130.9, 131.5, 132.3, 132.5, 138.5, 152.6, 159.3 (C=N), 159.4 (C=N), 160.0 (C=O); MS, m/z (%): 416 (M+, 100), 417 (M+1, 54.2), 418 (M+2, 30.3), 402 (11.2), 387 (71.4), 320 (46.1), 253 (20.3), 163 (10.0), 176 (14.5), 163 (17.1), 135 (12.5), 91 (19.5), 77 (58.5); Anal. Calcd. for C23H17ClN4O2 (416.86): C, 66.27; H, 4.11; N, 13.44; Found: C, 66.13; H, 4.24; N, 13.21%.
1-(3-Ethoxyphenyl)-6-methyl-3-phenyl-1H-imidazo[4,5-b]quinoxalin-2(3H)-one (11c): as brown crystals; yield 67%; mp 118-120 °C; IR: ν/cm-1 = 3054 (CH-Ar.), 2975, 2922 (CH-aliph.), 1744 (C=O), 1597 (C=N); 1H NMR: δ/ppm = 1.37 (t, 3H, CH3), 2.48 (s, 3H, CH3), 4.09 (q, 2H, CH2), 7.09-7.92 (m, 12H, Ar-H); 13C NMR: 15.0 (CH3), 21.4 (CH3), 63.8 (CH2), 113.6, 114.7, 119.3, 119.4, 127.3, 127.6, 127.8, 127.9, 128.7, 129.6, 130.4, 132.8, 133.8, 138.9, 139.0, 140.6, 140.7, 150.5 (18C-Ar), 152.6 (2C=N), 159.3 (C=O); MS, m/z (%): 396 (M+, 100), 397 (M+1, 31.4), 381 (27.6), 368 (50.7), 320 (46.1), 77 (8.5); Anal. Calcd. for C24H20N4O2 (396.44): C, 72.71; H, 5.08; N, 14.13; Found: C, 72.86; H, 5.21; N, 14.24.
Synthesis of Imidazolidin-4-Ylidene Derivatives 12a-d
To a solution of iminothione 3c,i (0.0l mo1) in ethanol (20 ml) pyrazolone derivatives (0.01 mol) and triethylamine (0.5 ml) was added. The reaction mixture was heated under reflux for 10 hr. left to cool, the solid that obtained after filtration was crystallized from ethanol/dioxane to give 12a-d.
4-(3-(3-Chlorophenyl)-1-(4-chlorophenyl)-5-imino-2-oxoimidazolidin-4-ylidene)-3-methyl-1H-pyrazol-5(4H)-one (12a): as red crystals; yield 72%; mp 118-120 °C; IR: ν/cm-1 = 3335 (NH), 3047 (CH-Ar), 2980, 2927 (CH-aliph.), 1726 (C=O), 1594 (C=N); 1H NMR: δ/ppm: 2.31 (s, 3H, CH3), 7.08-8.41 (m, 8H, Ar-H), 8.08 (br, 1H, NH; cancelled with D2O); 12.72 (br, 1H, NH; cancelled with D2O); Anal. Calcd. for C19H13Cl2N5O2 (414.24): C, 55.09; H, 3.16; N, 16.91; Found: C, 55.29; H, 3.34; N, 16.71%.
4-(3-(3-Chlorophenyl)-1-(4-chlorophenyl)-5-imino-2-oxoimidazolidin-4-ylidene)-3-methyl-1-phenyl-1H-pyrazol-5(4H)-one (12b): as violet crystals; yield 60%; mp 93-95 °C, IR: ν/cm-1 = 3345 (NH), 3063 (CH-Ar), 2922 (CH-aliph.), 1718 (C=O), 1621 (C=N); 1H NMR: δ/ppm: 2.41 (s, 3H, CH3), 7.11-8.79 (m, 13H, Ar-H), 12.64 (br, 1H, NH; cancelled with D2O); MS, m/z (%): 490 (M+, 1.0), 478 (2.4), 417 (5.1), 405 (3.6), 379 (4.4), 358 (3.1), 355 (5.3), 333 (30.9), 315 (4.3), 304 (6.3), 278 (41.6), 255 (21.2), 208 (38.9), 179 (49.0), 159 (16.73), 122 (60.4), 106 (100), 68 (8.47); Anal. Calcd. for C25H17Cl2N5O2 (490.34): C, 61.24; H, 3.49; N, 14.28; Found: C, 61.39; H, 3.54; N, 14.59%.
4-(3-(3-Ethoxyphenyl)-5-imino-2-oxo-1-phenylimidazolidin-4-ylidene)-3-methyl-1Hpyrazol-5(4H)-one (12c): as red crystals; yield 65%; mp 175-177 °C; IR: ν/cm-1 = 3291 (NH), 3050 (CH-Ar), 2978 (CH-aliph.), 1713 (C=O), 1610 (C=N); 1H NMR: δ/ppm: 1.12 (t, 3H, J = 6.9 Hz, CH3), 2.32 (s, 3H, CH3), 4.18 (q, 2H, J = 6.8 Hz, CH2), 7.11-8.79 (m, 9H, Ar-H), 8.11 (br, 1H, NH; cancelled with D2O); 12.71 (br, 1H, NH; cancelled with D2O); MS, m/z (%): 389 (M+, 3.1), 390 (M+1, 3.1), 309 (12.2), 304 (100), 295 (12.6), 287 (13.6), 275 (13.9), 244 (24.9), 200 (23.2), 163 (13.6), 135 (30.3), 124 (44.1), 119 (18.0), 90 (2.3), 77 (26.3); Anal. Calcd. for C21H19N5O3 (389.41): C, 64.77; H, 4.92; N, 17.98; Found: C, 64.86; H, 4.72; N, 17.76%.
4-(3-(3-Ethoxyphenyl)-5-imino-2-oxo-1-phenylimidazolidin-4-ylidene)-3-methyl-1-phenyl-1H-pyrazol-5(4H)-one (12d): as brown crystals; yield 68%; mp 70-72 °C; IR: ν/cm-1 = 3280 (NH), 3055 (CH-Ar), 2978, 2914 (CH-aliph.), 1717 (C=O), 1603 (C=N); 1H NMR: δ/ppm: 1.14 (t, 3H, J = 6.8 Hz, CH3), 2.41 (s, 3H, CH3), 4.19 (q, 2H, J = 6.9 Hz, CH2), 7.13- 8.01 (m, 14H, Ar-H), 12.83 (br, 1H, NH; cancelled with D2O); 13CNMR 14.8 (CH3), 15.1 (CH3), 63.3 (CH2), 105.0, 110.7, 112.8, 115.4, 119.2, 120.7, 122.7, 123.7, 124.2, 127.3, 127.9, 128.8, 129.4, 130.3, 137.3, 139.8, 140.5, 144.7, 147.8, 153.9, 159.34 (C=N), 159.4 (C=N), 162.3 (C=O), 166.6 (C=O); Anal. Calcd. for C27H23N5O3 (465.50): C, 69.66; H, 4.98; N, 15.04; Found: C, 69.49; H, 4.76; N, 15.21%.
Antimicrobial Activity
Chemical compounds were individually tested against a panel of gram positive and gram negative bacterial pathogens and fungi. Antimicrobial tests were carried out by the agar well diffusion method using 100 μL of suspension containing 1 x108 CFU/mL of pathological tested bacteria and 1 x106 CFU/ml of fungi spread on nutrient agar and Sabouraud dextrose agar media, respectively. After the media had cooled and solidified, wells (10 mm in diameter) were made in the solidified agar and loaded with 100 μL of tested compound solution prepared by dissolving 5 mg of the chemical compound in one mL of dimethyl sulfoxide (DMSO). The inoculated plates were then incubated for 24 h at 37 °C. Negative controls were prepared using DMSO employed for dissolving the tested compound. Ampicillin, Gentamycin and Amphotericin B (1 mg/mL) were used as standard for antibacterial and antifungal activity, respectively. After incubation time, antimicrobial activity was evaluated by measuring the zone of inhibition against the test organisms and compared with that of the standard. The observed zone of inhibition is presented in Table 1. Antimicrobial activities were expressed as inhibition diameter zones in millimeters (mm). The experiment was carried out in triplicate and the average zone of inhibition was calculated.
Table 1. Antimicrobial activity of the synthesized compounds against the pathological organisms expressed as inhibition diameter zones in millimeters (mm) based on well diffusion assay.
|
Compd.
No. |
Gram Positive Bacteria | Gram Negative Bacteria | Fungi | ||||||
|---|---|---|---|---|---|---|---|---|---|
| S. aureus | S. epidermidis | B. subtilits | N. gonorrhoeae | E. coli | K. pneumoniae | A. fumigatus | A. clavatus | G. candidum | |
| 3b | 0 | 14.6 ± 0.43 | 16.2 ± 0.53 | 10.2 ± 0.72 | 13.7 ± 0.63 | 15.6 ± 0.25 | 12.6 ± 0.58 | 13.6 ± 0.25 | 15.2 ± 0.38 |
| 3f | 20.3 ± 0.63 | 20.7 ± 0.22 | 22.4 ± .036 | 16.2 ± .058 | 17.6 ± 0.58 | 18.3 ± 1.2 | 20.5 ± 0.22 | 18.3 ± 0.26 | 22.1 ± 0.15 |
| 3g | 14.3 ± 0.58 | 15.9 ± 0.44 | 17.2 ± 0.58 | 16.3 ± 0.63 | 18.9 ± 0.63 | 20.3 ± 0.63 | 16.3 ± 0.44 | 18.4 ± 0.58 | 19.1 ± 0.37 |
| 3h | 13.6 ± 0.58 | 15.2 ± 0.44 | 16.1 ± 0.67 | 10.2 ± 0.46 | 13.0 ± 0.46 | 14.5 ± 0.46 | 10.6 ± 0.25 | 11.7 ± 0.34 | 16.5 ± 0.58 |
| 3i | 18.9 ± 0.14 | 16.2 ± 0.15 | 19.8 ± 0.42 | 12.3 ± 0.53 | 15.3 ± 0.53 | 16.2 ± 0.53 | 15.7 ± 0.33 | 15.9 ± 0.25 | 16.8 ± 0.34 |
| 3j | 18.9 ± 0.58 | 20.3 ± 0.43 | 21.4 ± 0.53 | 14.3 ± 0.25 | 16.9 ± 0.25 | 19.2 ± 0.25 | 17.6 ± 0.58 | 18.2 ± 0.25 | 20.3 ± 0.38 |
| 3k | 15.8 ± 0.44 | 17.8 ± 0.17 | 19.8 ± 0.22 | 0 | 0 | 0 | 18.7 ± 0.11 | 19.3 ± 0.23 | 20.3 ± 0.27 |
| 4a | 20.3 ± 0.58 | 17.3 ± 0.63 | 20.2 ± 0.44 | 13.6 ± 0.37 | 15.9 ± 0.37 | 16.8 ± 0.37 | 16.3 ± 0.44 | 18.6 ± 0.58 | 19.8 ± 0.25 |
| 4b | 13.6 ± 0.25 | 20.6 ± 0.63 | 22.4 ± 0.44 | 14.3 ± 0.63 | 16.9 ± 0.58 | 17.8 ± 1.2 | 17.8 ± 0.63 | 18.9 ± 0.44 | 20.3 ± 0.25 |
| 6a | 12.8 ± 0.58 | 14.3 ± 0.63 | 15.2 ± 0.72 | 0 | 0 | 0 | 13.3 ± 0.44 | 16.2 ± 0.58 | 17.2 ± 0.37 |
| 6b | 17.3 ± 0.58 | 18.4 ± 0.17 | 20.3 ± 0.22 | 0 | 0 | 0 | 16.3 ± 0.39 | 18.2 ± 0.16 | 20.4 ± 0.58 |
| 8a | 10.3 ± 0.58 | 11.6 ± 0.58 | 13.3 ± 0.72 | 0 | 0 | 0 | 12.3 ± 0.25 | 13.2 ± 0.25 | 15.2 ± 0.58 |
| 8b | 9.6 ± 1.2 | 10.6 ± 0.58 | 12.7 ± 0.72 | 0 | 0 | 0 | 8.6 ± 0.36 | 9.3 ± 0.44 | 11.4 ± 0.58 |
| 9a | 11.6 ± 0.58 | 12.4 ± 0.63 | 14.3 ± 0.63 | 0 | 0 | 0 | 16.3 ± 0.25 | 15.2 ± 0.58 | 17.3 ± 0.17 |
| 9b | 16.3 ± 0.37 | 17.2 ± 0.20 | 19.4 ± 0.29 | 0 | 11.2 ± 0.33 | 15.9 ± 0.77 | 15.6 ± 0.44 | 16.2 ± 0.58 | 17.9 ± 0.37 |
| 9c | 12.3 ± 0.58 | 14.2 ± 0.17 | 16.4 ± 0.22 | 0 | 0 | 0 | 10.3 ± 0.25 | 11.6 ± 0.34 | 13.4 ± 0.58 |
| 11a | 16.6 ± 0.20 | 19.8 ± 0.12 | 21.4 ± 0.17 | 11.4 ± 0.42 | 18.3 ± 0.58 | 16.3 ± 0.81 | 20.6 ± 0.63 | 21.1 ± 0.27 | 21.9 ± 0.35 |
| 11b | 15.6 ± 0.44 | 18.6 ± 0.44 | 21.1 ± 0.58 | 14.3 ± 0.20 | 16.3 ± 0.33 | 17.2 ± 0.34 | 18.3 ± 0.34 | 21.1 ± 0.25 | 21.3 ± 0.38 |
| 11c | 21.6 ± 0.25 | 22.3 ± 0.25 | 25.8 ± 0.25 | 20.6 ± 0.25 | 21.6 ± 0.19 | 23.2 ± 0.42 | 20.9 ± 0.25 | 22.3 ± 0.25 | 24.8 ± 0.58 |
| 12a | 17.2 ± 0.24 | 19.2 ± 0.15 | 19.8 ± 0.42 | 10.4 ± 0.25 | 13.4 ± 0.63 | 14.5 ± 0.51 | 15.7 ± 0.33 | 17.2 ± 0.25 | 19.8 ± 0.34 |
| 12b | 19.2 ± 0.25 | 20.6 ± 0.25 | 20.9 ± 0.63 | 11.6 ± 0.43 | 15.7 ± 0.56 | 15.9 ± 0.77 | 16.8 ± 025 | 18.3 ± 0.44 | 20.4 ± 0.63 |
| St. (A) | 28.9 ± 0.14 | 25.4 ± 0.18 | 29.8 ± 0.35 | ------- | ------- | ------- | ------- | ------- | ------- |
| St. (B) | ------- | ------- | ------- | 22.3 ± 0.58 | 23.4 ± 0.3 | 26.3 ± 0.15 | ------- | ------- | ------- |
| St. (C) | ------- | ------- | ------- | ------- | ------- | ------- | 23.7 ± 0.10 | 21.9 ± 0.12 | 25.4 ± 0.16 |
St. (A): Ampicillin, St. (B): gentamycin and St. (C): Amphotericin b
Minimal Inhibitory Concentration (MIC) Measurement
Screening was evaluated in vitro using the Broth dilution method according to NCCLs [22, 23]. All the bacteria were incubated and activated at 30 °C for 24 h inoculation into nutrient broth and the fungi were incubated in malt extract broth for 48 h. The compounds were dissolved in DMSO and then diluted using cautiously adjusted Mueller-Hinton broth. Two-fold serial concentrations of the compounds were employed to determine the (MIC). In each case triplicate tests were performed and the average was taken as the final reading. The tubes were then inoculated with the test organisms, grown in their suitable broth at 37 °C for 24 hours for tested microorganisms (1×108 CFU/mL for bacteria and 1 x106 CFU/mL of fungi), each 5 ml received 0.1 mL of the above inoculum and incubated at 37 °C for 24 hours. The lowest concentration showing no growth was taken as the minimum inhibitory concentration (MIC).
ACKNOWLEDGEMENTS
The authors would like to extend their sincere appreciation to the Deanship of Scientific Research at King Saud University for its funding of this research through the Research Group Project no. RGP.VPP-302.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
REFERENCES
- 1.Abbas S.Y., El-Sharief A.M., Basyouni W.M., Fakhr I.M., El-Gammal E.W. Thiourea derivatives incorporating a hippuric acid moiety: Synthesis and evaluation of antibacterial and antifungal activities. Eur. J. Med. Chem. 2013;64:111–120. doi: 10.1016/j.ejmech.2013.04.002. [DOI] [PubMed] [Google Scholar]
- 2.El-Sharief A.M., Abbas S.Y., El-Bayouki K.A., El-Gammal E.W. Synthesis of thiosemicarbazones derived from N-(4-hippuric acid)thiosemicarbazide and different carbonyl compounds as antimicrobial agents. Eur. J. Med. Chem. 2013;67:263–268. doi: 10.1016/j.ejmech.2013.06.031. [DOI] [PubMed] [Google Scholar]
- 3.Aneja D.K., Lohan P., Arora S., Sharma C., Aneja K.R., Prakash O. Synthesis of new pyrazolyl-2, 4-thiazolidinediones as antibacterial and antifungal agents. Bioorg. Med. Chem. Lett. 2011:1–15. doi: 10.1186/2191-2858-1-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Berber I., Cokmus C., Atalan E. Characterization of Staphylococcus species by SDS–page of whole-cell and extracellular proteins. Microbiology. 2003;72:42–47. [PubMed] [Google Scholar]
- 5.Bildirici I., Sener A., Tozlu I. Further derivatives of 4-benzoyl-1, 5-diphenyl-1H pyrazole-3-carboxylic acid and their antibacterial activities. Med. Chem. Res. 2007;16:418–426. [Google Scholar]
- 6.Appelbaum P.C. The emergence of vancomycin-intermediate and vancomycin-resistant Staphylococcus aureus. Clin. Microbiol. Infect. 2006;12:16. doi: 10.1111/j.1469-0691.2006.01344.x. [DOI] [PubMed] [Google Scholar]
- 7.Graziak M.P., Ding H. Halogenated (acylamino)imidazoles and benzimidazoles for directed halogen-metal exchange-based functionalization. Acta Chim. Slov. 2000;47:1. [Google Scholar]
- 8.Trunz B.B., Jedrysiak R., Tweats D., Brun R., Kaiser M., Suwinski J., Torreele E. 1-Aryl-4-nitro-1H-imidazoles, a new promising series for the treatment of human African trypanosomiasis. Eur. J. Med. Chem. 2011;46:1524–1535. doi: 10.1016/j.ejmech.2011.01.071. [DOI] [PubMed] [Google Scholar]
- 9.Raether W., Hänel H. Nitroheterocyclic drugs with broad spectrum activity. Parasitol. Res. 2003;90:519–539. doi: 10.1007/s00436-002-0754-9. [DOI] [PubMed] [Google Scholar]
- 10.Winkelmann E., Raether W., Gebert U., Sinharay A. Chemotherapeutically active nitro compounds. 4. 5-Nitroimidazoles (Part I-IV). Arzneimittelforschung. 1977:27–28. [PubMed] [Google Scholar]
- 11.Marie-Daragon A., Rouillard M.C., Bouteille B., Bisser S., de Albuquerque C., Chauvière G., Périé J., Dumas M. An efficacy trial on Trypanosoma brucei brucei of molecules permeating the blood-brain barrier and of megazol. Bull. Soc. Pathol. Exot. 1994;87:347–352. [PubMed] [Google Scholar]
- 12.Freeman C.D., Klutman N.E., Lamp K.C. Metronidazole. A therapeutic review and update. Drugs. 1997;54:679–708. doi: 10.2165/00003495-199754050-00003. [DOI] [PubMed] [Google Scholar]
- 13.Chazeau V., Cussac M., Boucherle A. Synthesis and immunomodulating activity of 1-amino-2- thiohydantoin derivatives. Eur. J. Med. Chem. 1992;27:839–843. [Google Scholar]
- 14.Bascou J.P., Lacroix G., Gadras A., Perez J. Dérivés optiquement actifs de 2-imidazoline-5-ones et 2-imidazoline-5-thiones fongicides. EP Patent 0 629616 A2, 1994 [Google Scholar]
- 15.Ding M.W., Fu B.Q., Yuan J-Z. Facile synthesis of 2-alkylthio-3-amino-4 H-imidazol-4-ones and 2H-imidazo[2,1-b]-1,3,4-thiadiazin-6(7H)-ones via N-vinylic iminophosphorane. Heteroatom Chem. 2005;16:76–80. [Google Scholar]
- 16.Aly M.M., Mohamed Y.A., El-Bayouki Kh.A., Basyouni W.M., Abbas S.Y. Synthesis of some new 4 (3H)-quinazolinone-2-carboxaldehyde thiosemicarbazones and their metal complexes and a study on their anticonvulsant, analgesic, cytotoxic and antimicrobial activities–Part-1. Eur. J. Med. Chem. 2010;45:3365–3373. doi: 10.1016/j.ejmech.2010.04.020. [DOI] [PubMed] [Google Scholar]
- 17.Helal M.H., Abbas S.Y., Salem M.A., Farag A.A., Ammar Y.A. Synthesis and characterization of new types of 2-(6-methoxy-2-naphthyl) propionamide derivatives as potential antibacterial and antifungal agents. Med. Chem. Res. 2013;22:5598–5609. [Google Scholar]
- 18.Abbas S.Y., Farag A.A., Ammar Y.A., Atrees A.A., Mohamed A.F., El-Henawy A.A. Synthesis, characterization, and antiviral activity of novel fluorinated isatin derivatives. Monatsh. Chem. 2013;144:1725–1733. doi: 10.1007/s00706-013-1034-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bondock S., Naser T., Ammar Y.A. Synthesis of some new 2-(3-pyridyl)-4,5-disubstituted thiazoles as potent antimicrobial agents. Eur. J. Med. Chem. 2013;62:270–279. doi: 10.1016/j.ejmech.2012.12.050. [DOI] [PubMed] [Google Scholar]
- 20.El-Sharief A.M., Moussa Z. Synthesis, characterization and derivatization of some novel types of mono- and bis-imidazolidineiminothiones and imidazolidineiminodithiones with antitumor, antiviral, antibacterial and antifungal activities – part I. Eur. J. Med. Chem. 2009;44:4315. doi: 10.1016/j.ejmech.2009.07.019. [DOI] [PubMed] [Google Scholar]
- 21.Cooper R.E. Analytical Microbiology. In: Kavangeh F.W., editor. 1-2. New York and London: Academic press; 1972. [Google Scholar]
- 22.Clinical and Laboratory Standards Institute (CLSI, formerly National Committee for Clinical and Laboratory Standards NCCLS); Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. NCCLS M7 A4, 4th. Pennsylvania: Approved Standard, NCCLS:; 1997. [Google Scholar]
- 23.Clinical and Laboratory Standards Institute (CLSI, formerly National Committee for Clinical and Laboratory Standards NCCLS); Method M27-A2. 2th. Pennsylvania: NCCLS; 2002. [Google Scholar]