Abstract
L-3,4-dihydroxyphenylalanine (L-DOPA) is the most common treatment for patients with Parkinson's disease (PD). However, long term use of L-DOPA for PD therapy lead to abnormal involuntary movements (AIMs) known as dyskinesia. Fatty acid amide hydrolase (FAAH) is enriched protein in basal ganglia, and inhibition of the protein reduces dyskinetic behavior of mice. Palmitoyl serotonin (PA-5HT) is a hybrid molecule patterned after arachidonoyl serotonin, antagonist of FAAH. However, the effect of PA-5HT on L-DOPA-induced dyskinesia (LID) in PD have not yet been elucidated. To investigate whether PA-5HT relieve LID in PD and decrease hyperactivation of dopamine D1 receptors, we used the 6-hydroxydopomine (6-OHDA)-lesioned mouse model of PD and treated the L-DOPA (20 mg/kg) for 10 days with PA-5HT (0.3 mg/kg/day). The number of wall contacts with the forelimb in the cylinder test was significantly decreased by 6-OHDA lesion in mice and the pharmacotherapeutic effect of L-DOPA was also revealed in PA-5HT-treated mice. Moreover, in AIMs test, PA-5HT-treated mice showed significant reduction of locomotive, axial, limb, and orofacial AIMs score compared to the vehicle-treated mice. LID-induced hyper-phosphorylation of ERK1/2 and overexpression of FosB/ΔFosB was markedly decreased in 6-OHDA-lesioned striatum of PA-5HT-treated mice, indicating that PA-5HT decreased the dopamine D1 receptor-hyperactivation induced by chronic treatment of L-DOPA in dopamine-denervated striatum. These results suggest that PA-5HT effectively attenuates the development of LID and enhance of ERK1/2 phosphorylation and FosB/ΔFosB expression in the hemi-parkinsonian mouse model. PA-5HT may have beneficial effect on the LID in PD.
Keywords: palmitoyl serotonin, L-DOPA-induced dyskinesia, Parkinson's disease, pERK1/2, FosB/ΔFosB
INTRODUCTION
Parkinson's disease (PD) is the second most common neurodegenerative disorder and is associated with progressive degeneration of dopaminergic neurons in the substantia nigra pars compacta (SNc) [1]. L-3, 4-dihydroxyphenylalanine (L-DOPA) is widely used as a standard therapy for patients with PD, but prolonged use of L-DOPA causes involuntary movements known as dyskinesia in patients with PD [2,3]. Currently, LID in PD is mediated by changes in the basal ganglia activity and alteration of the serotonin (5-HT) terminal [4,5,6]. Serotonergic neurons synthesise dopamine (DA) through decarboxylation of L-DOPA to dopamine by amino-acid decarboxylase, and store and release dopamine into their synaptic cleft [7,8]. L-DOPA-induced DA release is sensitive to serotonin pharmacological manipulation and the lesion of 5-HT neurons [9,10].
Conjugates of fatty acid with amino acids or monoamine neurotransmitters act as a signalling molecule in various biological processes [11,12,13]. N-acyl serotonin, with fatty acid chains, is endogenously formed and is present in the gastrointestinal tract of pigs and mice [13]. N-acyl serotonin has the ability to inhibit fatty acid amide hydrolase (FAAH) in vitro [13] and prevent glutamate-induced oxidative cytotoxicity in HT-22 cells [14]. N-palmitoyl serotonin (PA-5-HT), the N-acyl serotonin with palmitoyl chain, showed a remarkable protective effect against glutamate-induced cytotoxicity and oxidative stress, and markedly suppressed the glutamate-induced activation of ERK in the late phase [14]. The modulation of fatty acid amide (FAA) levels via inhibition of FAAH is of potential therapeutic interest for patients with PD [15]. Inhibition of FAAH and the transient receptor potential vanilloid 1 (TRPV1) receptor reduces L-DOPA-induced dyskinesia and hyperactivity in an animal model of PD [16,17]. PA-5-HT is a hybrid molecule patterned after arachidonoyl serotonin, a dual antagonist of FAAH and TRPV1 receptor [18,19]. However, the effect of PA-5-HT on LID in PD has not yet been investigated.
Previous studies reported that alternate of D1 dopamine receptor (D1R) associated protein in L-DOPA-induced dyskinesia (LID; [20,21]). Chronic treatment of L-DOPA triggers a D1R pathway that includes cAMP-dependent kinase [22,23,24]. The enhanced D1R signalling is observed in the phosphorylation of extracellular signal-regulated protein kinases (ERKs)1/2, mitogen- and stress-activated kinase 1 (MSK1), and histone H3 [25,26,27]. Therefore, chronic treatment of L-DOPA induces transcriptional alteration in the striatum such as FosB/ΔFosB expression [28,29,30]. The increase of FosB/ΔFosB expression by prolonged treatment of L-DOPA triggers the development of L-DOPA-induced abnormal behavioural responsiveness and dyskinesia [31].
In this study, to assess the anti-dyskinetic effect of PA-5HT in PD, we used the 6-hydroxydopamine (6-OHDA)-induced hemiparkinsonian mouse model, and evaluated LID using the abnormal involuntary movements (AIMs) scoring test. We administered the L-DOPA at 10 mg/kg/day dosage for 10 days in vehicle- and PA-5HT (0.3 mg/kg/day)-treated mice. The effects of the PA-5HT on hyper-phosphorylation of ERK1/2 and overexpression of FosB/ΔFosB was investigated in the striatum of an animal model of PD showing LID.
MATERIALS AND METHODS
Animals
Eight-week-old male C57BL/6J mice (25~28 g) were provided by the Korea Research Institute of Bioscience and Biotechnology. The animals were housed in a temperature (21~22℃) and humidity (50~60%) controlled environment and specific-pathogen-free condition with a 12-h light/dark cycle (light: 7 AM to 7 PM, dark: 7 PM to 7 AM). The animals were allowed free access to autoclaved food and water (Purina Inc., Hwasung-si, Korea). The cages were filled with bedding made of chopped wood particles (J. Rettenmaier & Söhne GmbH + Co. KG, Rosenberg, Germany). All animal experiments were approved by the Institutional Animal Use and Care Committee of the Korea Research Institute of Bioscience and Biotechnology and were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health.
Drugs
6-OHDA was purchased from Sigma-Aldrich Co. LLC (St. Louis, MO, USA) and diluted with 0.02% ascorbic acid in saline. Desipramine (25 mg/kg), L-DOPA (20 mg/kg), and the peripheral DOPA decarboxylase inhibitor benserazide hydrochloride (12 mg/kg) were purchased from Sigma-Aldrich Co. LLC and dissolved in saline immediately before use. PA-5HT (0.3 mg/kg) was purchased from Cayman Chemical (MI, USA), evaporated with ethanol and immediately diluted in saline. D-amphetamine (D-AMPH, 5 mg/kg) was purchased from USP (Rockville, MD, USA) and then dissolved in saline.
6-OHDA lesions
Mice were treated with desipramine (25 mg/kg, intraperitoneal (i.p.)) 30 min before the surgery to prevent noradrenergic neuron damage. Mice were anaesthetised with a mixture of ketamine hydrochloride and xylazine hydrochloride, as described previously [32], and mounted on a stereotactic frame (Stoelting Europe, Dublin, Ireland) that was equipped with a mouse adaptor. Mice received unilateral injections of 6-OHDA in 3-µL volumes (5 µg/µL, at the injection speed of 1 µL/min) into the left side of the SNc at the following coordinates according to the mouse brain atlas of Paxinos and Franklin (2001): anteroposterior (AP), -3.0 mm; lateral (L), -1.3 mm; and dorsoventral (DV), -4.7 mm [33]. Mice were left on a warming plate until they woke up from the anaesthesia and then returned to their home cages until use. To avoid dehydration, lesioned mice received sterile saline (10 mL/kg, i.p.) for 3 days. In addition, during the first week post-surgery, food pellets soaked in water were placed in a shallow vessel on the floor of the cages in the evening.
Cylinder test
Cylinder test was described in a previous study [33]. Cylinder test assessed the behavioural effects of the 6-OHDA-induced lesions and improvements of acute treatments with L-DOPA on sensorimotor function. Before the 6-OHDA lesion, 2 weeks later the 6-OHDA injection, and 30 min after the first injection of L-DOPA, each mouse was placed in an acrylic cylinder (diameter, 15 cm; height, 27 cm), and we counted the number of contacts on the wall with the right or left forepaw for 5 min. The use of the impaired (right) forelimb was expressed as a percentage of the total number of supporting wall contacts.
D-AMPH-induced rotation test
For mice administered with PA-5-HT, D-AMPH-induced rotation was measured 2 weeks after the 6-OHDA injections. We screened for mice showing high levels of asymmetric rotations in response to administration of D-AMPH. Turning behaviours that were induced after D-AMPH (5 mg/kg, i.p.) administration were recorded for 60 min in an observation cylinder (diameter, 20 cm; height 13 cm). The number of ipsilateral rotations was analysed by a SMART video-tracking program (Panlab S.I., Barcelona, Spain). The selected mice were separated into two groups for an injection of either vehicle (n=9) or PA-5-HT (n=8, 0.3 mg/kg/day, peroral).
Abnormal involuntary movements (AIMs) test
Four weeks after the 6-OHDA injections, lesioned mice were treated with L-DOPA (20 mg/kg, i.p.) and benserazide hydrochloride (12 mg/kg, i.p.) for 10 days. On days 5 and 10 of the L-DOPA injections, AIMs were assessed by observers who were blind to the treatment groups. Each mouse was individually placed in a separate glass cylinder, and dyskinetic behaviours were assessed for 1 min (monitoring period) in every 20-min block for a period of 120 min. The AIMs score corresponds to the sum of the individual scores for each subtype. AIMs subtypes: axial (contralateral dystonic posture of the neck and upper body toward the side contralateral to the lesion), limb (jerky and fluttering movement of the limb contralateral to the side of the lesion), orolingual (vacuous jaw movements and tongue protrusions), and locomotive (tight contralateral turns). Each subtypes was scored from 0 to 4: 0, absent; 1, occasional; 2, frequent; 3, continuous; 4, continuous and not interruptible by other stimulations. A composite score was obtained by the addition of the scores for axial, limb, and orofacial AIMs (ALO score) according to the report that composite AIMs scores more closely reflect human dyskinetic behaviour compared to the locomotive AIMs score (LOC score) [27,33,34].
Immunohistochemistry
Immunohistochemistry was conducted as described previously [35]. Briefly, 30 min after the last L-DOPA injection, the brains were removed, post-fixed overnight, and then cut into 40-µm coronal sections with a vibratome (Vibratome VT1000A, Leica Microsystems GmbH, Wetzlar, Germany). Free-floating sections were incubated in phosphate buffer saline (PBS) containing 3% hydrogen peroxide (H2O2) (v/v), rinsed 3 times in PBS, and blocked with 5% horse serum (HS) for 1 h at room temperature. Sections were incubated overnight at 4℃ with the primary antibodies. The primary antibodies were rabbit polyclonal antibodies for TH (Pel-Freez, RRID:AB_461064). After washing, the sections were incubated with biotinylated secondary anti-rabbit IgG (Vector Laboratories, Inc., Burlingame, CA, USA), which was followed by the avidin-biotinylated peroxidase complex (ABC kit, Vector Laboratories, Inc., PK6100) and 3,3'-diaminobenzidine (DAB; Sigma-Aldrich Co. LLC, D5637). Sections containing the SNc at AP -3.0 to -3.6 mm from the bregma were selected, and immunoreactive cells from the lesioned and unlesioned SNc were counted under a microscope. Qualitative evaluations of the immunoreactive cells were performed in a blind manner in terms of genotype and treatment by following the procedure introduced by Park et al. (2014) [33].
Immunoblotting
Western blot analysis was described in a previous study [36]. Thirty minutes after the last L-DOPA injection, mice were sacrificed, and the striatum was quickly removed and homogenised in a RIPA buffer (50 mM Tris-HCl, pH 8.0, 150 mM NaCl, 1% Nonidet P-40, 0.1% SDS, and 0.1% sodium deoxycholate) containing a cocktail of protease inhibitors (Roche). Protein samples were separated by 10% SDS-PAGE, and then transferred onto a polyvinylidene fluoride (PVDF) membrane (Bio-Rad, Hercules, CA). Blots were incubated with primary antibodies (Phospho-ERK1/2 (Thr202/Tyr204; Cell Signaling Technology, RRID:AB_331646), ERK1/2 (Cell Signaling Technology, RRID:AB_10695746), FosB/ΔFosB (Cell Signaling Technology, RRID:AB_2106903) or actin (Millipore) followed by secondary antibodies, and specific signals were visualised using an enhanced Ez West Lumi plus (WSE-7120L, ATTO Corporation, Tokyo, Japan). Western blot images were quantified using Quantity One 1-D analysis software, version 4.6.1 (Bio-Rad).
Statistical analysis
GraphPad PRISM software (GraphPad Software, Inc., La Jolla, CA, USA) was used to perform statistical analyses. Two-sample comparisons were conducted with Student's t tests, whereas multiple comparisons were made with a two-way analysis of variance (ANOVA), followed by a Bonferroni post hoc test or one-way ANOVA, followed by a Tukey-Kramer's post hoc test. All the results are presented as means±standard error of the mean. Any difference with a p value less than 0.05 were considered statistically significant.
RESULTS
6-OHDA lesion-induced motor deficit was rescued by acute L-DOPA treatment in vehicle-treated and PA-5HT-treated groups
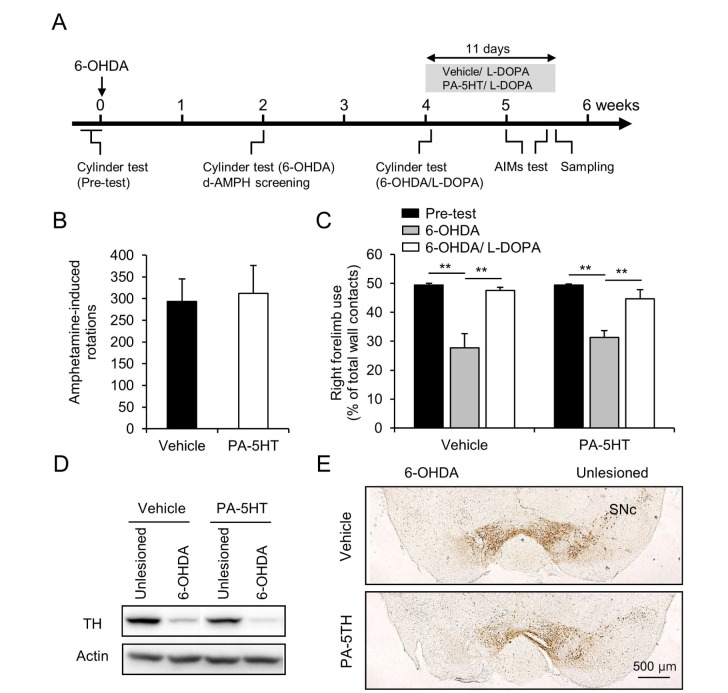
To investigate the effect of PA-5-HT on LID in PD, 6-OHDA-induced hemiparkinsonian mouse model was used [33]. Two weeks after the 6-OHDA lesion, we screened for mice showing high level of asymmetric rotations in responses to administration of D-amphetamine (D-AMPH) (Fig. 1B). The selected mice were then separated into two groups: vehicle-treated and PA-5HT-treated groups (D-AMPH-induced rotations: vehicle, 293.42±52.15 and PA-5HT, 312.2±63.72 and percentage of forelimb use: vehicle, 27.69±4.89% and PA-5HT, 31.30±2.33%). Two weeks after the behavioural screening, L-DOPA was administered in both groups. 6-OHDA-lesion induced reduced number of wall contacts with the forelimb of mice in the cylinder test (Fig. 1C, p<0.01). In the first administration of L-DOPA, we observed the therapeutic effects in both groups in the cylinder test (Fig. 1C, p<0.01). The efficacy of the 6-OHDA lesions was verified using western blotting and immunohistochemistry to detect the decrease in tyrosine hydroxylase (TH) level in the striatum (Fig. 1D) and SNc (Fig. 1E). In immunoblotting, TH depletion levels in 6-OHDA lesioned side were 70.35±9.19 % and 65.79±7.31% in vehicle and PA-5HT. And in immunohistochemistry, TH deletion levels were 78.99±4.98 and 69.96±7.71 in vehicle and PA-5-HT. We did not observe any correlation between the reduction of TH and the severity of AIMs (TH fiber in the striatum, r=0.156, p=0.549; TH-positive cells in SNc, r=0.293, p=0.2526). 6-OHDA lesion in the SNc in mice effectively induced dopaminergic neuronal death in the SNc and fibre loss in the striatum.
Fig. 1. Effects of 6-OHDA lesions on vehicle- and PA-5HT-treated mice. (A) Schematic of the 6-OHDA-lesion, drug injection and behavioral tests. (B) D-AMPH-induced rotations in vehicle- and PA-5HT- treated mice for 60 min (n=8-9). (C) Right forelimb use by vehicle- and PA-5HT-treated mice in the cylinder test before (pre-test) and after 6-OHDA injection (6-OHDA), and 30 min after the first treatment of L-DOPA (6-OHDA/L-DOPA) (n=8-9). Western blots (D) and immunostaining (E) of TH in the striatum and SNc of vehicle- and PA-5HT- treated mice. Scale bar 500 µm. **p<0.01, significant differences between indicated groups. One-way ANOVA followed by a Tukey-Kramer post hoc test. Data are presented as the mean±SEM. D-AMPH: D-amphetamine; PA-5HT: Palmitoyl serotonin; 6-OHDA: 6-hydroxydopamine; L-DOPA: L-3,4-dihydroxyphenylalanine; TH: tyrosine hydroxylase; SNc: substantia nigra pars compacta.
PA-5HT reduced the L-DOPA induced dyskinesia in hemiparkinsonian mice
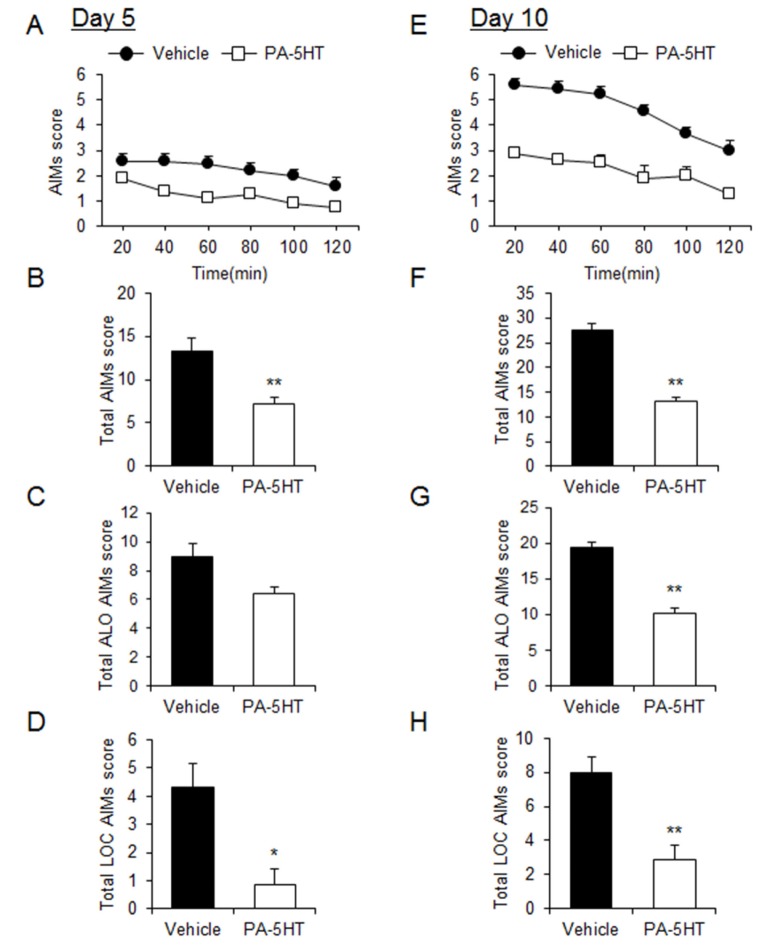
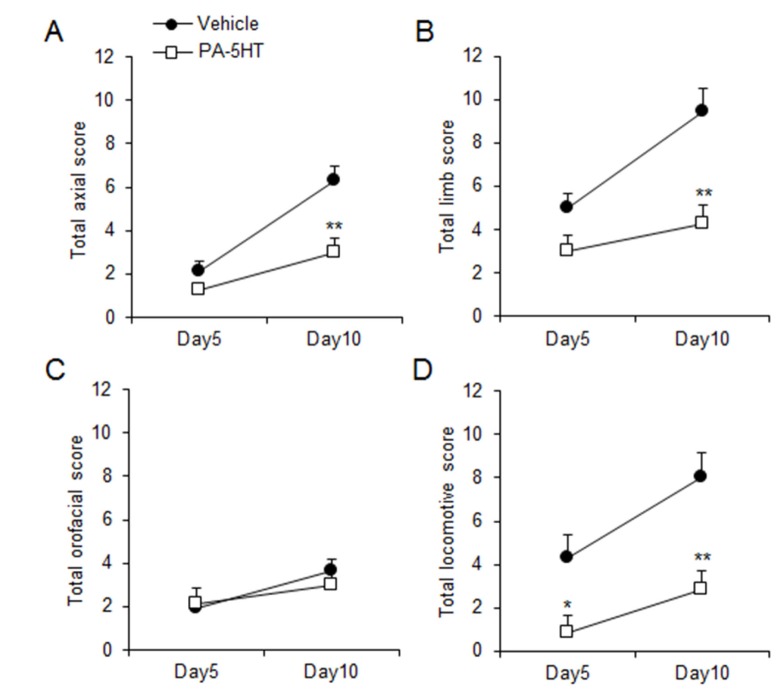
In order to investigate whether PA-5HT exerts an inhibitory effect on the LID of PD, the mice were administered L-DOPA (20 mg/kg/day) with vehicle or PA-5HT (0.3 mg/kg/day) for 10 days. On day 5, we performed the AIMs test in the vehicle-treated and PA-5HT-treated groups (Fig. 2A). Differences in the total AIM scores were observed between vehicle-treated and PA-5HT-treated groups (Fig. 2B). Differences in axial, limb, and orofacial AIM (ALO AIMs) scores were not significant at this time point (vehicle; 9±0.89 and PA-5HT 6.38±0.533, p=0.064, Fig. 2C), but locomotive AIMs (LOC AIMs) score was significantly decreased in the PA-5HT-treated group (vehicle; 4.33±0.84 and PA-5HT; 0.88±0.56, p<0.05, Fig. 2D). On day 10, differences in the total AIM scores were observed between vehicle-treated and PA-5HT-treated groups (Fig. 2E and F). ALO AIMs scores were significantly decreased in the PA-5HT-treated group (vehicle; 19.44±1.41 and PA-5HT; 10.25±0.88, p<0.01, Fig. 2G), and LOC AIMs score was also significantly decreased in the PA-5HT-treated group (vehicle; 8±0.96 and PA-5HT; 2.88±0.63, p<0.01, Fig. 2H). PA-5HT treatment significantly reduced axial, limb, and locomotive AIM scores on day 10 compared to the vehicle-treated group (Fig. 3A, B and D), but had no effect on orofacial AIM scores (Fig. 3C).
Fig. 2. Effects of PA-5HT on L-DOPA-induced dyskinesia in the PD animal model. (A and E) Time-course of the total AIMs scored every 20 min over a period of 120 min after the L-DOPA treatment on day 5 (A) and 10 (E). Two-way ANOVA: (A) effect of time, F(5, 75)=4.07, p=0.0025; effect of PA-5HT, F(1, 15)=8.99, p=0.0090; interaction, F(5, 75)=0.41, p=0.837. (E) Effect of time, F(5, 75)=9.76, p<0.0001; effect of PA-5HT, F(1, 15)=22.04, p=0.0003; interaction, F(5, 75)=1.05, p=0.3973. B and F, Total AIMs (sum of LOC and ALO AIMs) scored during 120 min after the L-DOPA treatment on day 5 (B) and 10 (F). C, D, G and H, ALO (axial, limb, and orofacial, C and G) and LOC (locomotive, D and H) AIMs scored during the 120 min period after L-DOPA treatment in both the groups on day 5 (C and D) and 10 (G and H) (n=8-9). Student's t test, *p<0.05 and **p<0.01. Data are presented as the mean±SEM. PA-5HT: Palmitoyl serotonin; L-DOPA: L-3,4-dihydroxyphenylalanine; PD: Parkinson's disease; AIMs: abnormal involuntary movements.
Fig. 3. Effect of PA-5HT on axial, limb, orofacial, and locomotive AIM scores induced by L-DOPA. Axial (A), limb (B), orofacial (C), and locomotive (D) AIMs scores were measured on day 5 and 10 after the L-DOPA treatment (n=8-9). AIMs scored during the 120 min period after L-DOPA treatment in both the groups on day 5. Student's t test,*p<0.05 and **p<0.01, Significant differences between the vehicle- and PA-5HT-treated groups. Data are presented as the mean±SEM. PA-5HT: Palmitoyl serotonin; AIMs: abnormal involuntary movements; L-DOPA: L-3,4-dihydroxyphenylalanine.
PA-5HT suppressed the hyper-phosphorylation of ERK1/2 in the 6-OHDA-lesioned striatum
Previous studies indicated that pERK1/2 is implicated in the degree of LID [37]. ERK is strongly activated by chronic treatment of L-DOPA in the 6-OHDA-lesioned striatum [38,39]. Plant extracts or compounds having therapeutic effects on LID reduced phosphorylation of ERK in the dopamine-denervated striatum [37,40]. We examined whether lower AIM scores in the PA-5HT-treated group are associated with the phosphorylation level of ERK1/2 protein. On day 11, the vehicle-treated and PA-5HT-treated mice were sacrificed after 30 min of L-DOPA treatment and western blot analysis was performed using the processed samples from the unlesioned and 6-OHDA-lesioned striatum. In the vehicle-treated group, phospho-ERK1/2 levels were significantly increased in the 6-OHDA-lesioned striatum compared with the unlesioned striatum (Fig. 4). In contrast, phosphorylation of ERK1/2 was not changed in the 6-OHDA-lesioned striatum of PA-5HT-treated mice (Fig. 4).
Fig. 4. Hyperphosphorylation of ERK1/2 by chronic L-DOPA is reduced in the 6-OHDA-lesioned striatum of PA-5HT-treated mice. Protein levels of phospho-extracellular signal-regulated protein kinase (pERK) 1/2 and total ERK1/2 were evaluated by western blots (A). pERK1/ERK1 (B) and pERK2/ERK2 (C) levels in the 6-OHDA lesioned versus unlesioned striata (n=8-9). One-way ANOVA followed by a Tukey-Kramer post hoc test. *p<0.05 and **p<0.01. Data are presented as the mean±SEM. L-DOPA: L-3,4-dihydroxyphenylalanine; 6-OHDA: 6-hydroxydopamine; PA-5HT: Palmitoyl serotonin.
PA-5HT suppressed the overexpression of FosB and ΔFosB in the 6-OHDA-lesioned striatum
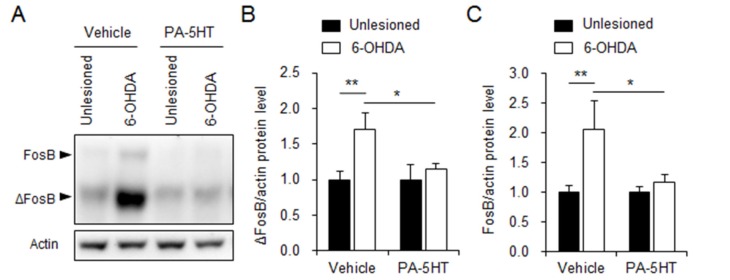
Previously, the increased FosB/ΔFosB expression induced by chronic treatment of L-DOPA in dopaminergic depletion striatum was reported [39,41]. Induction of their expression occurs in the striatal medium spiny neurons that express dopamine D1 receptors and is regulated by the activation of ERK1/2 [39,41,42]. On day 11, the vehicle-treated and PA-5HT-treated mice were sacrificed after 30 min of L-DOPA treatment and western blot analysis was performed using the processed samples from the unlesioned and 6-OHDA-lesioned striatum. The expression of FosB and ΔFosB was markedly increased in the 6-OHDA-lesioned striatum of the vehicle-treated mice, compared with the unlesioned striatum of the mice, but induction of FosB and ΔFosB expression in the lesioned striatum was not revealed in the PA-5HT-treated mice (Fig. 5).
Fig. 5. Overexpression of FosB/ΔFosB by chronic L-DOPA is reduced in the 6-OHDA-lesioned striatum of PA-5HT-treated mice. Protein levels of FosB and ΔFosB were evaluated by western blots (A). ΔFosB (B) and FosB (C) levels in the 6-OHDA lesioned versus unlesioned striata (n=8-9). One-way ANOVA followed by a Tukey-Kramer post hoc test.*p<0.05 and **p<0.01. Data are presented as the mean±SEM. L-DOPA: L-3,4-dihydroxyphenylalanine; 6-OHDA: 6-hydroxydopamine.
DISCUSSION
The present study demonstrated the protective effect of PA-5HT on LID in a 6-OHDA-induced hemi-Parkinsonian mouse model. The therapeutic effect of L-DOPA was not altered by PA-5HT treatment with L-DOPA in the PD animal model. Moreover, abnormal increase of ERK1/2 phosphorylation and FosB/ΔFosB expression induced by prolonged treatment of L-DOPA were significantly inhibited in the 6-OHDA lesioned striatum of PA-5HT-treated mice.
Previously, FAAH inhibitors have been shown to reduce hyperdopaminergia-related hyperactivity in mice [43,44]. The reduction of LID was seen when FAAH inhibition was combined with an antagonist of TRPV1 receptor [17]. Although the effect of PA-5HT on TRPV1 receptor has not been studied, PA-5-HT was found to be a potent FAAH inhibitor [13]. PA-5HT is one of the hybrid molecules patterned after arachidonoyl serotonin and is abundantly presents in the gastrointestinal tract of pigs and mice [13]. Arachidonoyl serotonin inhibits both FAAH and TRPV1 receptor [18,19]. Furthermore, FAAH and TRPV1 receptor are enriched in the basal ganglia, including the striatum, substantia nigra, and globus pallidus [45,46,47]. Under conditions in which FAAH activity is blocked, endocannabinoid anandamide is increased both intracellularly and extracellularly [48]. Increased anandamide level in the striatum may have benefits in the treatment of patients with PD [49]. However, amelioration of LID by FAAH inhibitors is consistent with some, but not all. Moreover, further studies are needed to understand biological roles of PA-5HT and investigate effects of FAAH by treatment with PA-5HT in vivo.
The long-term administration of L-DOPA to 6-OHDA-lesioned mice triggered activation of dopamine D1 receptor dependent signalling pathway [20,22,25]. The pharmacological inhibition of D1R-associated pathway, protein kinase A, ERK1/2, dopamine- and cAMP-regulated phosphoprotein (Mr 32 kDa), and adenylyl cyclase type 5 significantly attenuated dyskinesia in PD animal models [22,32,50]. FosB/ΔFosB expression occurred in neurons that express D1Rs in LID [35] and decreased in dyskinesia by the inhibition of D1R-dependent proteins [32,51]. Moreover, LID-associated phosphorylation of ERK1/2 is implicated in D1R-mediated pathway and attenuated in DARPP32-null mice [25]. PA-5HT markedly suppressed the glutamate-induced activation of ERK in the late phase [13]. In the present study, enhanced ERK1/2 phosphorylation and FosB/ΔFosB expression induced by chronic L-DOPA treatment were effectively suppressed by the co-treatment of PA-5HT in the hemi-Parkinsonian mouse model. However, the regulatory mechanism of PA-5HT on these signalling pathways in the striatum has not yet been elucidated. Further studies are needed to investigate the effects of PA-5HT on the striatal synaptic plasticity and dopaminergic system. In previous reports, systemic and local administration of anandamide affected the striatal synaptic plasticity [52], tyrosine hydroxylase activity [53], and dopamine release and cAMP synthesis [54] in the striatum. Enhancing the anandamide level by PA-5HT-mediated inhibition of FAAH in the striatum may affect striatal supersensitivity to L-DOPA and AIM caused by chronic treatment of L-DOPA. Moreover, TRPV1 receptor inhibitor reduced L-DOPA-induced FosB/ΔFosB expression in the dopaminedepleted striatum [55].
In conclusion, the co-treatment of PA-5HT with L-DOPA, the N-acyl serotonin with palmitoyl chain, can suppress the AIM induced by prolonged administration of L-DOPA in the 6-OHDA lesioned mouse model. Furthermore, the associated molecular changes, pERK and FosB/ΔFosB expression, mediated by D1R signalling in mice were reduced by the systemic administration of PA-5HT. These data suggest that the PA-5HT may have beneficial effect on dopamine replacement therapy in PD.
ACKNOWLEDGEMENTS
This work was supported by the National Research Foundation of Korea grant funded by the Korea government (MSIP; 2014R1A2A2A04005236 and 2015M3C7A1029113).
References
- 1.Fahn S. Medical treatment of Parkinson's disease. J Neurol. 1998;245:P15–P24. doi: 10.1007/pl00007742. [DOI] [PubMed] [Google Scholar]
- 2.Obeso JA, Rodríguez-Oroz MC, Rodríguez M, Lanciego JL, Artieda J, Gonzalo N, Olanow CW. Pathophysiology of the basal ganglia in Parkinson's disease. Trends Neurosci. 2000;23:S8–S19. doi: 10.1016/s1471-1931(00)00028-8. [DOI] [PubMed] [Google Scholar]
- 3.Obeso JA, Olanow CW, Nutt JG. Levodopa motor complications in Parkinson's disease. Trends Neurosci. 2000;23:S2–S7. doi: 10.1016/s1471-1931(00)00031-8. [DOI] [PubMed] [Google Scholar]
- 4.Chase TN, Oh JD. Striatal dopamine- and glutamate-mediated dysregulation in experimental parkinsonism. Trends Neurosci. 2000;23:S86–S91. doi: 10.1016/s1471-1931(00)00018-5. [DOI] [PubMed] [Google Scholar]
- 5.Rylander D, Recchia A, Mela F, Dekundy A, Danysz W, Cenci MA. Pharmacological modulation of glutamate transmission in a rat model of L-DOPA-induced dyskinesia: effects on motor behavior and striatal nuclear signaling. J Pharmacol Exp Ther. 2009;330:227–235. doi: 10.1124/jpet.108.150425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Carta M, Carlsson T, Kirik D, Björklund A. Dopamine released from 5-HT terminals is the cause of L-DOPA-induced dyskinesia in parkinsonian rats. Brain. 2007;130:1819–1833. doi: 10.1093/brain/awm082. [DOI] [PubMed] [Google Scholar]
- 7.Arai R, Karasawa N, Geffard M, Nagatsu T, Nagatsu I. Immunohistochemical evidence that central serotonin neurons produce dopamine from exogenous L-DOPA in the rat, with reference to the involvement of aromatic L-amino acid decarboxylase. Brain Res. 1994;667:295–299. doi: 10.1016/0006-8993(94)91511-3. [DOI] [PubMed] [Google Scholar]
- 8.Navailles S, De Deurwaerdère P. Imbalanced dopaminergic transmission mediated by serotonergic neurons in L-DOPA-induced dyskinesia. Parkinsons Dis. 2012;2012:323686. doi: 10.1155/2012/323686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lindgren HS, Andersson DR, Lagerkvist S, Nissbrandt H, Cenci MA. L-DOPA-induced dopamine efflux in the striatum and the substantia nigra in a rat model of Parkinson's disease: temporal and quantitative relationship to the expression of dyskinesia. J Neurochem. 2010;112:1465–1476. doi: 10.1111/j.1471-4159.2009.06556.x. [DOI] [PubMed] [Google Scholar]
- 10.Navailles S, Benazzouz A, Bioulac B, Gross C, De Deurwaerdère P. High-frequency stimulation of the subthalamic nucleus and L-3,4-dihydroxyphenylalanine inhibit in vivo serotonin release in the prefrontal cortex and hippocampus in a rat model of Parkinson's disease. J Neurosci. 2010;30:2356–2364. doi: 10.1523/JNEUROSCI.5031-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Burstein S. The elmiric acids: biologically active anandamide analogs. Neuropharmacology. 2008;55:1259–1264. doi: 10.1016/j.neuropharm.2007.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang SM, Bisogno T, Trevisani M, Al-Hayani A, De Petrocellis L, Fezza F, Tognetto M, Petros TJ, Krey JF, Chu CJ, Miller JD, Davies SN, Geppetti P, Walker JM, Di Marzo V. An endogenous capsaicin-like substance with high potency at recombinant and native vanilloid VR1 receptors. Proc Natl Acad Sci U S A. 2002;99:8400–8405. doi: 10.1073/pnas.122196999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Verhoeckx KC, Voortman T, Balvers MG, Hendriks HF, M Wortelboer H, Witkamp RF. Presence, formation and putative biological activities of N-acyl serotonins, a novel class of fatty-acid derived mediators, in the intestinal tract. Biochim Biophys Acta. 2011;1811:578–586. doi: 10.1016/j.bbalip.2011.07.008. [DOI] [PubMed] [Google Scholar]
- 14.Jin MC, Yoo JM, Sok DE, Kim MR. Neuroprotective effect of N-acyl 5-hydroxytryptamines on glutamate-induced cytotoxicity in HT-22 cells. Neurochem Res. 2014;39:2440–2451. doi: 10.1007/s11064-014-1448-2. [DOI] [PubMed] [Google Scholar]
- 15.Sieradzan KA, Fox SH, Hill M, Dick JP, Crossman AR, Brotchie JM. Cannabinoids reduce levodopa-induced dyskinesia in Parkinson's disease: a pilot study. Neurology. 2001;57:2108–2111. doi: 10.1212/wnl.57.11.2108. [DOI] [PubMed] [Google Scholar]
- 16.Johnston TH, Huot P, Fox SH, Wakefield JD, Sykes KA, Bartolini WP, Milne GT, Pearson JP, Brotchie JM. Fatty acid amide hydrolase (FAAH) inhibition reduces L-3,4-dihydroxyphenylalanine-induced hyperactivity in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned non-human primate model of Parkinson's disease. J Pharmacol Exp Ther. 2011;336:423–430. doi: 10.1124/jpet.110.169532. [DOI] [PubMed] [Google Scholar]
- 17.Morgese MG, Cassano T, Cuomo V, Giuffrida A. Anti-dyskinetic effects of cannabinoids in a rat model of Parkinson's disease: role of CB(1) and TRPV1 receptors. Exp Neurol. 2007;208:110–119. doi: 10.1016/j.expneurol.2007.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ortar G, Cascio MG, De Petrocellis L, Morera E, Rossi F, Schiano-Moriello A, Nalli M, de Novellis V, Woodward DF, Maione S, Di Marzo V. New N-arachidonoylserotonin analogues with potential "dual" mechanism of action against pain. J Med Chem. 2007;50:6554–6569. doi: 10.1021/jm070678q. [DOI] [PubMed] [Google Scholar]
- 19.Maione S, De Petrocellis L, de Novellis V, Moriello AS, Petrosino S, Palazzo E, Rossi FS, Woodward DF, Di Marzo V. Analgesic actions of N-arachidonoyl-serotonin, a fatty acid amide hydrolase inhibitor with antagonistic activity at vanilloid TRPV1 receptors. Br J Pharmacol. 2007;150:766–781. doi: 10.1038/sj.bjp.0707145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Westin JE, Vercammen L, Strome EM, Konradi C, Cenci MA. Spatiotemporal pattern of striatal ERK1/2 phosphorylation in a rat model of L-DOPA-induced dyskinesia and the role of dopamine D1 receptors. Biol Psychiatry. 2007;62:800–810. doi: 10.1016/j.biopsych.2006.11.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Darmopil S, Martín AB, De Diego IR, Ares S, Moratalla R. Genetic inactivation of dopamine D1 but not D2 receptors inhibits L-DOPA-induced dyskinesia and histone activation. Biol Psychiatry. 2009;66:603–613. doi: 10.1016/j.biopsych.2009.04.025. [DOI] [PubMed] [Google Scholar]
- 22.Picconi B, Centonze D, Håkansson K, Bernardi G, Greengard P, Fisone G, Cenci MA, Calabresi P. Loss of bidirectional striatal synaptic plasticity in L-DOPA-induced dyskinesia. Nat Neurosci. 2003;6:501–506. doi: 10.1038/nn1040. [DOI] [PubMed] [Google Scholar]
- 23.Aubert I, Guigoni C, Håkansson K, Li Q, Dovero S, Barthe N, Bioulac BH, Gross CE, Fisone G, Bloch B, Bezard E. Increased D1 dopamine receptor signaling in levodopa-induced dyskinesia. Ann Neurol. 2005;57:17–26. doi: 10.1002/ana.20296. [DOI] [PubMed] [Google Scholar]
- 24.Guigoni C, Doudnikoff E, Li Q, Bloch B, Bezard E. Altered D(1) dopamine receptor trafficking in parkinsonian and dyskinetic non-human primates. Neurobiol Dis. 2007;26:452–463. doi: 10.1016/j.nbd.2007.02.001. [DOI] [PubMed] [Google Scholar]
- 25.Santini E, Valjent E, Usiello A, Carta M, Borgkvist A, Girault JA, Hervé D, Greengard P, Fisone G. Critical involvement of cAMP/DARPP-32 and extracellular signal-regulated protein kinase signaling in L-DOPA-induced dyskinesia. J Neurosci. 2007;27:6995–7005. doi: 10.1523/JNEUROSCI.0852-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rangel-Barajas C, Silva I, Lopéz-Santiago LM, Aceves J, Erlij D, Florán B. L-DOPA-induced dyskinesia in hemiparkinsonian rats is associated with up-regulation of adenylyl cyclase type V/VI and increased GABA release in the substantia nigra reticulata. Neurobiol Dis. 2011;41:51–61. doi: 10.1016/j.nbd.2010.08.018. [DOI] [PubMed] [Google Scholar]
- 27.Alcacer C, Santini E, Valjent E, Gaven F, Girault JA, Hervé D. Gα(olf) mutation allows parsing the role of cAMP-dependent and extracellular signal-regulated kinase-dependent signaling in L-3,4-dihydroxyphenylalanine-induced dyskinesia. J Neurosci. 2012;32:5900–5910. doi: 10.1523/JNEUROSCI.0837-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hope BT, Nye HE, Kelz MB, Self DW, Iadarola MJ, Nakabeppu Y, Duman RS, Nestler EJ. Induction of a long-lasting AP-1 complex composed of altered Fos-like proteins in brain by chronic cocaine and other chronic treatments. Neuron. 1994;13:1235–1244. doi: 10.1016/0896-6273(94)90061-2. [DOI] [PubMed] [Google Scholar]
- 29.Chen J, Kelz MB, Hope BT, Nakabeppu Y, Nestler EJ. Chronic Fos-related antigens: stable variants of deltaFosB induced in brain by chronic treatments. J Neurosci. 1997;17:4933–4941. doi: 10.1523/JNEUROSCI.17-13-04933.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hiroi N, Marek GJ, Brown JR, Ye H, Saudou F, Vaidya VA, Duman RS, Greenberg ME, Nestler EJ. Essential role of the fosB gene in molecular, cellular, and behavioral actions of chronic electroconvulsive seizures. J Neurosci. 1998;18:6952–6962. doi: 10.1523/JNEUROSCI.18-17-06952.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cao X, Yasuda T, Uthayathas S, Watts RL, Mouradian MM, Mochizuki H, Papa SM. Striatal overexpression of DeltaFosB reproduces chronic levodopa-induced involuntary movements. J Neurosci. 2010;30:7335–7343. doi: 10.1523/JNEUROSCI.0252-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kim KS, Lee KW, Lee KW, Im JY, Yoo JY, Kim SW, Lee JK, Nestler EJ, Han PL. Adenylyl cyclase type 5 (AC5) is an essential mediator of morphine action. Proc Natl Acad Sci U S A. 2006;103:3908–3913. doi: 10.1073/pnas.0508812103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Park HY, Kang YM, Kang Y, Park TS, Ryu YK, Hwang JH, Kim YH, Chung BH, Nam KH, Kim MR, Lee CH, Han PL, Kim KS. Inhibition of adenylyl cyclase type 5 prevents L-DOPA-induced dyskinesia in an animal model of Parkinson's disease. J Neurosci. 2014;34:11744–11753. doi: 10.1523/JNEUROSCI.0864-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lundblad M, Andersson M, Winkler C, Kirik D, Wierup N, Cenci MA. Pharmacological validation of behavioural measures of akinesia and dyskinesia in a rat model of Parkinson's disease. Eur J Neurosci. 2002;15:120–132. doi: 10.1046/j.0953-816x.2001.01843.x. [DOI] [PubMed] [Google Scholar]
- 35.Kim KS, Kang YM, Kang Y, Park TS, Park HY, Kim YJ, Han BS, Kim CH, Lee CH, Ardayfio PA, Han PL, Jung BH, Kim KS. Pitx3 deficient mice as a genetic animal model of co-morbid depressive disorder and parkinsonism. Brain Res. 2014;1552:72–81. doi: 10.1016/j.brainres.2014.01.023. [DOI] [PubMed] [Google Scholar]
- 36.Park HY, Ryu YK, Kim YH, Park TS, Go J, Hwang JH, Choi DH, Rhee M, Lee CH, Kim KS. Gadd45β ameliorates L-DOPA-induced dyskinesia in a Parkinson's disease mouse model. Neurobiol Dis. 2016;89:169–179. doi: 10.1016/j.nbd.2016.02.013. [DOI] [PubMed] [Google Scholar]
- 37.Doo AR, Kim SN, Hahm DH, Yoo HH, Park JY, Lee H, Jeon S, Kim J, Park SU, Park HJ. Gastrodia elata Blume alleviates L-DOPA-induced dyskinesia by normalizing FosB and ERK activation in a 6-OHDA-lesioned Parkinson's disease mouse model. BMC Complement Altern Med. 2014;14:107. doi: 10.1186/1472-6882-14-107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Santini E, Alcacer C, Cacciatore S, Heiman M, Hervé D, Greengard P, Girault JA, Valjent E, Fisone G. L-DOPA activates ERK signaling and phosphorylates histone H3 in the striatonigral medium spiny neurons of hemiparkinsonian mice. J Neurochem. 2009;108:621–633. doi: 10.1111/j.1471-4159.2008.05831.x. [DOI] [PubMed] [Google Scholar]
- 39.Pavón N, Martín AB, Mendialdua A, Moratalla R. ERK phosphorylation and FosB expression are associated with L-DOPA-induced dyskinesia in hemiparkinsonian mice. Biol Psychiatry. 2006;59:64–74. doi: 10.1016/j.biopsych.2005.05.044. [DOI] [PubMed] [Google Scholar]
- 40.Teng L, Hong F, Zhang C, He J, Wang H. Compound formula rehmannia alleviates levodopa-induced dyskinesia in Parkinson's disease. Neural Regen Res. 2014;9:407–412. doi: 10.4103/1673-5374.128246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Andersson M, Hilbertson A, Cenci MA. Striatal fosB expression is causally linked with l-DOPA-induced abnormal involuntary movements and the associated upregulation of striatal prodynorphin mRNA in a rat model of Parkinson's disease. Neurobiol Dis. 1999;6:461–474. doi: 10.1006/nbdi.1999.0259. [DOI] [PubMed] [Google Scholar]
- 42.Fasano S, Bezard E, D'Antoni A, Francardo V, Indrigo M, Qin L, Doveró S, Cerovic M, Cenci MA, Brambilla R. Inhibition of Ras-guanine nucleotide-releasing factor 1 (Ras-GRF1) signaling in the striatum reverts motor symptoms associated with L-dopa-induced dyskinesia. Proc Natl Acad Sci U S A. 2010;107:21824–21829. doi: 10.1073/pnas.1012071107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tzavara ET, Li DL, Moutsimilli L, Bisogno T, Di Marzo V, Phebus LA, Nomikos GG, Giros B. Endocannabinoids activate transient receptor potential vanilloid 1 receptors to reduce hyperdopaminergia-related hyperactivity: therapeutic implications. Biol Psychiatry. 2006;59:508–515. doi: 10.1016/j.biopsych.2005.08.019. [DOI] [PubMed] [Google Scholar]
- 44.Johnston TH, Huot P, Fox SH, Wakefield JD, Sykes KA, Bartolini WP, Milne GT, Pearson JP, Brotchie JM. Fatty acid amide hydrolase (FAAH) inhibition reduces L-3,4-dihydroxyphenylalanine-induced hyperactivity in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned non-human primate model of Parkinson's disease. J Pharmacol Exp Ther. 2011;336:423–430. doi: 10.1124/jpet.110.169532. [DOI] [PubMed] [Google Scholar]
- 45.Mezey E, Tóth ZE, Cortright DN, Arzubi MK, Krause JE, Elde R, Guo A, Blumberg PM, Szallasi A. Distribution of mRNA for vanilloid receptor subtype 1 (VR1), and VR1-like immunoreactivity, in the central nervous system of the rat and human. Proc Natl Acad Sci U S A. 2000;97:3655–3660. doi: 10.1073/pnas.060496197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Egertová M, Cravatt BF, Elphick MR. Comparative analysis of fatty acid amide hydrolase and cb(1) cannabinoid receptor expression in the mouse brain: evidence of a widespread role for fatty acid amide hydrolase in regulation of endocannabinoid signaling. Neuroscience. 2003;119:481–496. doi: 10.1016/s0306-4522(03)00145-3. [DOI] [PubMed] [Google Scholar]
- 47.Tsou K, Nogueron MI, Muthian S, Sañudo-Pena MC, Hillard CJ, Deutsch DG, Walker JM. Fatty acid amide hydrolase is located preferentially in large neurons in the rat central nervous system as revealed by immunohistochemistry. Neurosci Lett. 1998;254:137–140. doi: 10.1016/s0304-3940(98)00700-9. [DOI] [PubMed] [Google Scholar]
- 48.Kathuria S, Gaetani S, Fegley D, Valiño F, Duranti A, Tontini A, Mor M, Tarzia G, La Rana G, Calignano A, Giustino A, Tattoli M, Palmery M, Cuomo V, Piomelli D. Modulation of anxiety through blockade of anandamide hydrolysis. Nat Med. 2003;9:76–81. doi: 10.1038/nm803. [DOI] [PubMed] [Google Scholar]
- 49.Fernandez-Espejo E, Caraballo I, Rodriguez de Fonseca F, Ferrer B, El Banoua F, Flores JA, Galan-Rodriguez B. Experimental parkinsonism alters anandamide precursor synthesis, and functional deficits are improved by AM404: a modulator of endocannabinoid function. Neuropsychopharmacology. 2004;29:1134–1142. doi: 10.1038/sj.npp.1300407. [DOI] [PubMed] [Google Scholar]
- 50.Lebel M, Chagniel L, Bureau G, Cyr M. Striatal inhibition of PKA prevents levodopa-induced behavioural and molecular changes in the hemiparkinsonian rat. Neurobiol Dis. 2010;38:59–67. doi: 10.1016/j.nbd.2009.12.027. [DOI] [PubMed] [Google Scholar]
- 51.Feyder M, Södersten E, Santini E, Vialou V, LaPlant Q, Watts EL, Spigolon G, Hansen K, Caboche J, Nestler EJ, Fisone G. A role for mitogen- and stress-activated kinase 1 in L-DOPA-induced dyskinesia and ΔFosB expression. Biol Psychiatry. 2016;79:362–371. doi: 10.1016/j.biopsych.2014.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ronesi J, Gerdeman GL, Lovinger DM. Disruption of endocannabinoid release and striatal long-term depression by postsynaptic blockade of endocannabinoid membrane transport. J Neurosci. 2004;24:1673–1679. doi: 10.1523/JNEUROSCI.5214-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Romero J, García L, Cebeira M, Zadrozny D, Fernández-Ruiz JJ, Ramos JA. The endogenous cannabinoid receptor ligand, anandamide, inhibits the motor behavior: role of nigrostriatal dopaminergic neurons. Life Sci. 1995;56:2033–2040. doi: 10.1016/0024-3205(95)00186-a. [DOI] [PubMed] [Google Scholar]
- 54.Cadogan AK, Alexander SP, Boyd EA, Kendall DA. Influence of cannabinoids on electrically evoked dopamine release and cyclic AMP generation in the rat striatum. J Neurochem. 1997;69:1131–1137. doi: 10.1046/j.1471-4159.1997.69031131.x. [DOI] [PubMed] [Google Scholar]
- 55.González-Aparicio R, Moratalla R. Oleoylethanolamide reduces L-DOPA-induced dyskinesia via TRPV1 receptor in a mouse model of Parkinson's disease. Neurobiol Dis. 2014;62:416–425. doi: 10.1016/j.nbd.2013.10.008. [DOI] [PubMed] [Google Scholar]